Abstract
Hematological changes is one of the most common complications occurred in cancer patients. Therefore, the current study aimed to assess the hematological toxicity of cervical cancer patients before and after the initiation of treatment. The retrospective cohort study was conducted from 2015 to 2022 at the University of Gondar Comprehensive Specialized Hospital. The hematological profile and sociodemographic and clinical data of the cervical cancer patients were collected using data extraction sheets. The Epidata version 3.1 and SPSS version 25 were used for data entry and analysis, respectively. Descriptive statistics were employed to summarize the data. To compare the median differences in hematological parameters before and after treatment, the Wilcoxon rank test was used. In addition, to assess the presence of an independent association between hematological abnormalities and the independent variables, logistic regression models were used. A p value less than 0.05 was considered to indicate statistical significance for all tests. In current study, the median (Interquartile range) of hemoglobin levels, white blood cell counts, and platelet counts, before treatment were 13.2 (12.1, 15) g/dl, 7.5 (5.8, 11.8) *109/L, and 330 (252, 383) *109/L, respectively. However, after treatment the median (Interquartile range) value of hemoglobin levels, white blood cell counts, and platelet counts were significantly lowered. On the other hand, red cell distribution width was significantly greater after treatment. At baseline, the magnitude of leucocytosis, anemia, and thrombocytosis were 27.9%, 24.6%, and 18.7%, respectively. After the treatment, anemia increased to 44.3%, but leucocytosis and thrombocytosis were replaced by leucopenia 18.3% and thrombocytopenia 17.8%, respectively. Hematological abnormalities such as anemia, leucopenia and thrombocytopenia were high after chemo-radiotherapy, and surgery. As the stage of cancer advances, the risk of developing anemia, leucocytosis, and thrombocytosis increased in 7.6, 6.9 and 9 times, respectively. Furthermore, being HIV patients and rural resident increased the risk of developing anemia about twofold. In conclusion hematological abnormalities were observed before and after cervical cancer treatment, with significance increment after chemo-radiotherapy and surgery. As the stage of cancer advances, the risk of developing hematological abnormalities increases. Therefore, routine monitoring of hematological changes before and after treatment and screening for major risk factors are important for improved patients’ management.
Similar content being viewed by others
Introduction
Cervical cancer is a prevalent gynecological cancer that originates from the abnormal multiplication and spread of cells around the female reproductive organ1. The complications of cervical cancer ranged from asymptomatic to pelvic pain, unexplained weight loss, unexpected vaginal discharge, bleeding and pain during sexual intercourse, organ failure, biochemical abnormalities, hematological abnormalities, and death1,2,3,4. These complications may be directly related to disease pathogenesis and/or treatment-associated adverse effects5. Hematological changes are among the most common complications that occur in cervical cancer patients. This is due to either direct consequences of disease or adverse effects of treatment2,6,7,8.
The cervical cancer pathogenesis directly affects almost all hematological parameters9,10,11. This is because of the malignant cell infiltration of different organs involved in hematopoiesis12,13,14. In addition, tumor-associated bleeding, tumor-associated malnutrition, tumor-associated activation of the coagulation system, and impaired iron and folate metabolism due to tumor competition for nutrients are also possible malignant-related causes for changes7,13,14,15. Furthermore, tumor-associated production and activation of different cytokines and growth factors also involved to the changes3,14,16,17.
Heavy bleeding during cryotherapy and surgical removal of tumors can lead blood cells to be suppressed3,18,19. In addition, chemoradiotherapy used for treatment can irradiate bone marrow cavities such as the lumbar sacrum, pubis, ischium, proximal femur, and ilium8,20. This can induce the suppression of almost all blood cells since those cavities are involved in approximately 50% of hematopoiesis3,9,21. Similarly, the myelotoxic effect of chemoradiotherapy on different organs, such as the liver, kidney, and gastrointestinal tract, induces the suppression of different cellular components and reduces hematopoiesis8,22. Furthermore, chemoradiotherapy can directly cause cell death, stimulate different inhibitory cytokines, and prevent cellular growth8,14,23. This can also decrease the hemoglobin concentration, platelet count, and leukocyte count after treatment24,25,26.
The chemotherapy reduces the production of RBC through damaging DNA of immature rapidly dividing hematopoietic stem cells which involved for production of RBC cells. Additionally, chemotherapy targeted for programmed cell death of cancerous cells but it can also lead normal erythroid progenitor cells to undergo programmed cell death27,28.
According to the available evidences, the presence of hematological abnormalities in cervical cancer patients worsen the outcomes and leads to treatment failure2,9,29. As a result, assessing the magnitude of hematological abnormalities is critical for reducing mortality and morbidity. Additionally, it ensuring poor treatment prognosis in cervical cancer patients; hence, the degree of magnitude of hematological abnormalities varies based on different situations30. Nevertheless, there are insufficient data on the impact of this disease and its treatment on hematological parameters in the study setting. Thus, this study aimed to determine hematological abnormalities before and after cervical cancer treatment.
Methods and materials
Study design, period and setting
The retrospective cohort study was conducted from January 1, 2015 to December 30, 2022 at University of Gondar, Comprehensive Specialized Hospital Cancer Treatment Center, Northwest Ethiopia.
Populations
All cervical cancer patients who visited a treatment center and confirmed as having cervical cancer were considered as the source population. Moreover, in the present study, cervical cancer patients who had visited the treatment center during the study period and had complete hematological data before and after treatment were considered as the study population.
Eligibility criteria
Patients who were newly diagnosed with cervical cancer and had follow-up data in medical records were included. Patients whose hematological profile data before and after treatment incomplete were excluded from the study. In addition, unreadable patient data from medical records and patients who started treatment elsewhere and who were followed up in the study setting were excluded from this study.
Sample size determination and sampling technique
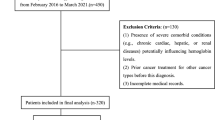
As a result of the retrospective nature of the study, all cervical cancer patients (219) who attended the University of Gondar, Comprehensive Specialized Hospital Cancer Treatment Center from 2015 to 2022 and fulfilled the eligibility criteria were included in the present study. A convenient sampling technique were used to include study participants.
Data collection procedure and quality control
Data extraction checklists, which included sociodemographic variables, clinical characteristics, and hematological parameters, were prepared to extract information from the medical records of cervical cancer patients. The data extraction sheets were pretested to ensure its validity and accuracy. Before data collection, all medical charts (360) of cervical cancer patients which recorded within the study period were evaluated to determine the completeness of all necessary information. Nevertheless, only 219 medical records fulfilled the inclusion criteria, and eligible for data collection. From eligible medical charts, data such as sociodemographic, clinical, and hematological parameters were extracted by two data collectors through observation. To ensure data quality, training was given to all the data collectors before starting the data collection process. In addition, to assure the quality of the data such clarity, accuracy, completeness, and consistency, the supervisors and principal investigators inspected the data collection process on the spot.
Data analysis and interpretations
First, the completeness of the collected data was checked manually. The data were subsequently coded and entered into Epidata version 3.1 and analyzed with SPSS version 25 software. Descriptive statistics such as frequencies, percentages, medians, and interquartile ranges (IQRs) were utilized to summarize the data. The data were presented in text, tables, and charts. The normality of the data was assessed by the Shapiro‒Wilk test. The Wilcoxon rank test was used to compare the median differences in hematological parameters before and after initiation of cervical cancer treatment. In addition, to assess the presence of an independent association between hematological abnormalities and independent variables, both bivariate and multivariate logistic regression models with 95% confidence intervals (CIs) were used. All logistic regression model assumptions were checked and fulfilled. A p value less than 0.05 was considered to indicate statistical significance for all tests.
Operational definitions
Based on the World Health Organization (WHO), anemia is defined as a hemoglobin level less than 12.0 g/dL in women after adjusting for altitude. The severity of anemia was classified as mild (11–11.9 g/dl), moderate (8–10.9 g/dl), and severe (< 8 g/dl) by considering hemoglobin level based on WHO definition31. However, other hematological abnormalities were defined based on the local reference range. In addition, cytopenia was defined as a decrease in total blood cell count and indicated by anemia, leucopenia, or thrombocytopenia based on the local reference range32.
Ethical consideration
An ethical clearance letter was obtained from the ethical review committee of the School of Biomedical and Laboratory Science, College of Medicine and Health Science, University of Gondar (Ref. No./SBLS/2897/2015) after the study protocol was reviewed. Furthermore, a permission letter was obtained from the hospital medical and cancer treatment center directors. Since the study was retrospective, informed consent was waived from ethical review committee. To keep confidentiality of the collected data the personal identifiers were removed and unauthorized persons limited in their access to the data.
Results
Sociodemographic and clinical characteristics of the study participants
Out of the 360 records retrieved from patient charts, only 219 had complete data and included in this study. The majority of patients admitted to the treatment center were urban residents 170(77.6%) and had cervical cancer 137(71.3%) at the early stage (stages one and two). The median (IQR) age of the study participants was 50 (49.2–52) years. Fifty-six (25.6%) patients admitted in treatment center was with complication of metastasis to the local site. Concerning coinfections, 25 (11.4%) and 16 (7.3%) patients had human immunodeficiency virus (HIV) and intestinal parasite infections, respectively. Majority of patients included this study were who took chemo-radiotherapy and cryotherapy with frequency of 82(37.5%) and 71 (32.4%), respectively. Among patients who took radiotherapy regimen, majority 37 (39.4%) of patients were took 20 Gy in to 10 Gy fractions (Table 1).
Hematological changes in cervical cancer patients before and after treatment
The median (interquartile range (IQR)) of Hb before and after treatment were 13.2 (12.1, 15) g/dl and 12.2 (10.7, 14.2) g/dl, respectively. Meanwhile, the median (IQR) of white blood cell (WBC) before and after treatment were 7.5 (5.8, 11.8) *109/L and 4.8 (3.8, 5.7) *109/L, respectively. Before and after treatment, the median (IQR) of neutrophil (Nue) were 4.5 (2.8, 8.0, 11.8) *109/L and 2.4 (2.1, 3.1) *109/L, whereas the median (IQR) of lymphocyte (Lym) before and after treatment were 1.9 (1.6, 2.5) *109/L and 1.7 (1.6, 1.8) *109/L, respectively. Regarding the platelet (PLT) count, the median (IQR) values detected before and after treatment were 330 (252, 383) *109/L and 298 (170, 365) *109/L, respectively. In addition, the median hematological parameters, such as Hb levels, WBC, Nue, Lym, PLTs, and platelet mean volume (PMV), were significantly lower after treatment than before treatment. In contrast, the red cell distribution width (RDW) was significantly greater after treatment than before treatment (Table 2).
Hematological abnormalities of cervical cancer patients before and after treatment
In this study, before treatment, the magnitude of leucocytosis, anemia, and thrombocytosis were 27.9% (95% CI: 21.9, 34.7), 24.6% (95% CI: 18.7, 30.6), and 18.7% (95% CI: 14.2, 23.7), respectively. The magnitude of anemia increased to 44.3% (95% CI: 37.9, 50.7) after treatment. The severity of anemia before 21 (38.9%) and after 45 (46.4%) treatment was moderate (Fig. 1). On the other hand, leucocytosis and thrombocytosis were changed to leucopenia (18.3%; 95% CI: 13.2, 23.7) and thrombocytopenia (17.8%; 95% CI: 12.8, 23.3), respectively, after treatment. With regard to WBC abnormalities, 35.2% (95% CI: 28.3, 41.6) of the patients were diagnosed with neutrophilia at baseline, and 16% (95% CI: 11.4, 21.0) of the patients were diagnosed with lymphopenia after treatment (Table 3). In addition, the current study revealed 7.3% (95% CI: 4.1, 11) cytopenias among cervical cancer patients after treatment (Fig. 2).
Distribution of hematological abnormalities in cervical cancer patients based on treatment group
The magnitude of anemia in patients who took chemoradiotherapy and surgery were 59 (60.8%) and 21 (21.6%), respectively. Eighty-eight (93.6%) of patients taking the RT regimen had anemia. The magnitude of anemia in patients taking 40–50 Gy in 2-Gy fractions + brachytherapy, 40–50 Gy in 2-Gy fractions + 20–26 Gy in 2-Gy fraction boost, and 20 Gy in 10-Gy fractions RT regimen were 14 (15.9%), 33 (37.5%), and 41 (46.6%), respectively. The majority of patients with leucopenia took chemoradiotherapy 23 (57.5%) and surgery 7 (17.5%) as treatment. Regarding differential WBC abnormalities, neutropenia and lymphopenia were the most prevalent abnormalities observed following chemoradiotherapy, with a frequency of 6 (54.5%) and 42.6%, respectively. Furthermore, the magnitude of thrombocytopenia after surgery and chemoradiotherapy were 19 (48.7%) and 11 (28.2%), respectively (Fig. 3).
Associations between hematological abnormalities and the sociodemographic and clinical characteristics of cervical cancer patients before treatment
Multivariable logistic regression revealed that patients admitted to the treatment center during the late stage of cervical cancer were at a greater risk of developing anemia (adjusted odds ratio (AOR) = 7.6, 95% CI (3.7, 15.5), leucocytosis (AOR = 6.9, 95% CI (3.6, 13.6)), and thrombocytosis (AOR = 9, 95% CI4,20) than admitted during the early stage. In addition, cervical cancer patients with HIV coinfection were about two times (AOR = 2.3, 95% CI (1.1, 6.2)) more likely develop anemia than were non-HIV-coinfected patients. Moreover, being a rural resident increased the risk of developing anemia (AOR = 2.5, 95% CI (1.14, 5.5)) compared with urban resident before treatment (Table 4).
Discussion
Globally, cervical cancer is one of the most common causes of death for women33. Cervical cancer patients suffer not only from disease but also from adverse effects from treatment. Hematological abnormalities are complications resulting from both disease and adverse effects of treatment and can lead to life-threatening conditions. As a result, assessing the magnitude of hematological abnormalities is essential for improving patient outcomes. The current study also aimed to assess the magnitude of hematological abnormalities before and after treatment among cervical cancer patients34.
The present study revealed that the Hb concentration of cervical cancer patients was significantly lower after treatment than before treatment. These findings are comparable to those of studies conducted in Korea28,35, Poland36, Canada37, Belgium28, and Germany38,39. The possible reason for the decrease in Hb after treatment could be related to the low RBC count due to the side effects of treatment. In addition, iron loss due to bleeding during surgery may cause a decrease in hemoglobin after treatment40,41. However, a study conducted in Turkey reported that there was no significant difference in Hb level after treatment23. The variability of the findings may be due to differences in sample size and the inclusion of more frequently blood transfused patients in the Turkish study. On the other hand, the finding of this study showed that RDW was significantly greater after treatment than before treatment. This finding is supported by studies conducted in Canada37 and Belgium28. It could be a side effect of cancer therapy, as cell hemolysis due to side effects of therapy results in a low RBC count and increased RDW after treatment42.
With regard to WBC, the current study showed that the total WBC count, Nue count, and Lym count significantly decreased after treatment. These findings concur with studies conducted in Korea35,43, Belgium28, Turkey23, and Germany39. Similarly, in the present study, the PLT and PMV were significantly lower after treatment. Previous studies conducted in India44, China45, Korea35, and Germany39 reported similar finding. The possible reasons for the low WBC and PLT could be the adverse effects of the treatments. Bleeding during cryotherapy, surgery, and tissue damage caused by drugs may lower the total blood count after treatment. The findings of the MPV study conducted in China support this finding45. The presence of large PLTs due to the inflammatory response of irritated tissue to treatment may lead to a decrease in the MPV after treatment45.
The baseline magnitude of anemia detected in this study was 24.6% (95% CI: 18.7, 30.6). This finding is in agreement with studies conducted in Kenya20, Belgium28, and Canada22,37. This could be attributed to impaired hematopoiesis, tumor-associated loss of blood by bleeding, tumor-associated malnutrition, and impaired iron and folate metabolism due to tumor competition for nutrients13,15. However, the figure is lower than those of studies carried out in Ethiopia (51%)13, Korea (36.5%)11, Poland (33%)36, and Canada (40%)46. The discrepancy between the findings could be attributed to differences in study time, sample size, health care delivery policy, and clinical characteristics.
The magnitude of anemia increased to 44.3% (95% CI: 37.9, 50.7) after treatment. this finding is also in lined with studies conducted in Korea (36.4%)11, Poland (43%)36, and Belgium (38%)28. The possible reason for the increase in the magnitude of anemia after treatment is the impaired production of red blood cells (RBCs) due to the toxicity of the hematopoietic organs. In contrast, studies conducted in China (50% and 79.7%)3,47 and Thailand (57.3%)48 reported a much greater magnitude of anemia after treatment. A possible explanation for the discrepancy could be related to the variability in sample size, treatment modalities, and stages of cancer; hence, those studies included a very small sample size and exclusively included late-stage patients who were treated with chemo-radiotherapy.
On the other hand, magnitude of anemia in patients who took chemoradiotherapy and surgery were 59 (60.8%) and 21 (21.6%), respectively. The studies conducted in Australia49 and Mexico46 supports the finding. The possible reason for high magnitude of anemia after chemoradiotherapy and surgery might be suppression of RBC production by toxic effect of chemo-radiotherapy and loss of blood cell during surgery, respectively50. The production of RBC suppressed following chemotherapy by various mechanisms. One of the mechanisms is suppression of bone marrow by damaging the DNA of erythroid precursor cells due to chemoradiotherapy. Furthermore, the production of RBC suppressed following chemotherapy as a result of apoptosis of RBC cells, induction of interleukin 6 and tumor necrosis factor alpha secretion which reduce the activity of erythropoietin, induction of iron sequestration, induction of oxidative stress and hemolysis of RBCs, nutritional deficiencies and kidney disease51,52,53.
The magnitude anemia in patients who took 20 Gy in 10-Gy fractions and 40–50 Gy in 2-Gy fractions + 20–26 Gy in 2-Gy fraction RT regimen were high. This could be related to high dose of RT, since it well known high RT dose leads to more tissue damage46.
In this study, the magnitude of leucocytosis was 27.9% (95% CI: 21.9, 34.7), with a predominance of neutrophilia 35.2% (95% CI: 28.3, 41.6) at baseline. These findings are in agreement with studies conducted in Canada22 and France16. As pervious evidence shows, the occurrence of leucocytosis is expected because of the upregulation of leucocyte growth factors, such as interleukins, colony-stimulating factors of granulocytes-macrophages, and tumor necrosis factor-alpha, due to tumors17. On the other hand, this study revealed 18.3% (95% CI: 13.2, 23.7) leucopenia after treatment. Out of leucopenia patient’s, majority were who took chemoradiotherapy 23 (57.5%) as a treatment. This figure is in consistent with studies recently conducted in Kenya20 and Canada22. A possible explanation for the occurrence of leucopenia after treatment is related to the side effects of treatment, specifically chemoradiotherapy. However, this number is much lower than that reported in studies conducted in China3 and Thailand48. A possible explanation for this variability might be differences in sample size, treatment modalities, operational definitions of leucocytosis, and cancer stages.
In the present study, 18.7% (95% CI: 14.2, 23.7) of patients had thrombocytosis at baseline, but this figure changed to 17.8% (95% CI: 12.8, 23.3) after treatment with high magnitude in patients who took surgery 19 (48.7%) and chemoradiotherapy 11 (28.2%). These findings are supported by studies conducted in Korea43, China3, and Canada22. This may be because of the induction of various tumor-secreted growth factors and proinflammatory cytokines, which promote an increase in blood thrombopoietin levels, causing thrombocytosis54. The occurrence of thrombocytopenia after chemoradiotherapy treatment is explained by a decrease in the production of blood cells due to the negative effect of treatment on stem cells in the bone marrow, whereas occurrence of thrombocytopenia following surgery might explained high platelet loss and consumption during surgery50,55.
The findings of this study showed that as the cervical cancer stage advanced, the odds of developing anemia, leucocytosis, and thrombocytosis increased by 7.6 times, 6.9 times, and 9 times, respectively. These findings are in consistent with those of studies conducted in Canada22,37, Romania56, Turkey57, Belgium28, China21, Korea11,35, India9, Kenya20 and Ethiopia13. The increased likelihood of anemia in the late stage may be due to heavy bleeding, multiple medications, or the presence of high malignant cell competition for nutrients in the late stage of cancer compared with the early stage. On the other hand, the production of additional growth factors and cytokines in the advanced stage might increase the risk of leucocytosis and thrombocytosis. Similarly, cancer-HIV coinfection and being a rural resident increase the risk of developing anemia approximately twofold. These findings are supported by previous studies conducted in Nigeria and Ethiopia20,58 in the case of cervical cancer-HIV coinfection; however, in the case of rural residents, we did not find published supporting findings. The association between cancer and HIV coinfection could be due to the suppression of blood cell production caused by the side effects of HIV drugs. On the other hand, the differences in socioeconomic status, dietary characteristics, and admission stage between rural and urban residents might cause variability.
Limitations of the study
The retrospective nature of the study might cause errors; there might be errors in recording since the data were not collected for the specific aim of the study. In addition, the loss of follow-up and the transfer of patients to another center led to a reduced sample size. This may have affected the representativeness of our study. Another limitation of this study was being single center study, this may also minimize representativeness of findings.
Conclusion
In conclusion, after treatment hematological parameters such as Hgb concentration, WBC count, Nue, Lym, PLT, and MPV were significant decreased after treatment. In addition, the magnitude of anemia is relatively greater after treatment than before treatment. Moreover, this study revealed moderate leucocytosis and thrombocytosis, but these conditions were replaced by leucopenia and thrombocytopenia after treatment. Furthermore, the study revealed that patients with advanced-stage cancer were at high risk for developing anemia, leucocytosis, and thrombocytosis. The magnitude of anemia, leucopenia and thrombocytopenia were high following chemo-radiotherapy and surgery. Therefore, strict follow-up and early detection of hematological abnormalities before and after treatment giving more emphasis to patients who took chemo-radiotherapy and surgery are necessary for better patient management and improved quality of life. Additionally, physicians should consider hematological complications during cervical cancer treatment. It is also important to conduct more conclusive, prospective and multicenter studies that can confirm the findings of this study.
Data availability
All the data supporting these findings are contained within the manuscript.
Abbreviations
- Hb:
-
hemoglobin
- HIV:
-
human immunodeficiency virus
- IQR:
-
interquartile range
- Lym:
-
lymphocyte
- MPV:
-
mean platelet volume
- Neu:
-
neutrophil
- PLT:
-
platelet
- RBC:
-
red blood cell
- RDW:
-
red cell distribution width
- WBC:
-
white blood cell and WHO: World Health Organization
References
Regauer, S. & Reich, O. The origin of human papillomavirus (HPV)—induced cervical squamous cancer. Curr. Opin. Virol. 51, 111–118 (2021).
Araújo, D. F. B., Cavalcanti, I. D., Larrazabal-Hadj-Idris, B. R. & Peres, A. L. Hematological and biochemical toxicity analysis of chemotherapy in women diagnosed with cervical cancer. Jornal Brasileiro de Patologia e Med. Laboratorial. 56, e1772020 (2020).
Hui, B. et al. Association between bone marrow dosimetric parameters and acute hematologic toxicity in cervical cancer patients undergoing concurrent chemoradiotherapy: comparison of three-dimensional conformal radiotherapy and intensity-modulated radiation therapy. Int. J. Gynecol. Cancer. 24(9), 1648–1652 (2014).
Kumar, T. et al. Correlation between pelvic bone marrow radiation dose and acute hematological toxicity in cervical cancer patients treated with concurrent chemoradiation. Cancer Manag. Res. 11, 6285–6297 (2019).
Kantelhardt, E. J. et al. Cervical cancer in Ethiopia: survival of 1,059 patients who received oncologic therapy. Oncologist. 19 (7), 727–734 (2014).
Wondimneh, B. et al. Comparison of hematological and biochemical profile changes in pre-and post-chemotherapy treatment of cancer patients attended at Ayder comprehensive specialized hospital, Mekelle, Northern Ethiopia 2019: a retrospective cohort study. Cancer Manage. Res. 625–632. (2021).
Abushofa, F. A., Azab, A. E. & Al Ghawi, H. M. Haematological parameter alterations among cervical cancer patients and the relationship between blood groups and cervical cancer disease. Himal. J. Appl. Med. Sci. Res. 3(2). (2022).
Eifel, P. J. (ed) Editor Chemoradiotherapy in the Treatment of Cervical cancer. Seminars in Radiation Oncology (Elsevier, 2006).
Baruah, C., Paul, T., Sarma, B., Deka, K. J. & Patowory, P. P. Hematological toxicity in Carcinoma Cervix patients undergoing concurrent Chemo Radiation Therapy or Radiation Therapy alone in a Tertiary Hospital of North East India: a prospective study. Asian Pac. J. Cancer Care. 7 (4), 651–655 (2022).
Kumar, A. et al. Correlation of hematological parameters with clinical outcomes in cervical Cancer patients treated with Radical Radio (chemo) therapy: a retrospective study. Int. J. Radiation Oncology* Biology* Phys. (2023).
Lim, S., Lee, C-M., Park, J-M., Jung, S-Y. & Lee, K-B. An association between preoperative anemia and poor prognostic factors and decreased survival in early stage cervical cancer patients. Obstet. Gynecol. Sci. 57 (6), 471–477 (2014).
Mabuchi, S. et al. Elevated white blood cell count at the time of recurrence diagnosis is an indicator of short survival in patients with recurrent cervical cancer. Int. J. Gynecologic Cancer ;22(9). (2012).
Wassie, M., Aemro, A. & Fentie, B. Prevalence and associated factors of baseline anemia among cervical cancer patients in Tikur Anbesa Specialized Hospital, Ethiopia. BMC Women’s Health. 21 (1), 1–8 (2021).
Yanazume, S. et al. Tumor bleeding requiring intervention and the correlation with anemia in uterine cervical cancer for definitive radiotherapy. Jpn J. Clin. Oncol. 48 (10), 892–899 (2018).
Candelaria, M., Cetina, L. & Dueñas-González, A. Anemia in cervical cancer patients. Med. Oncol. 22 (2), 161–168 (2005).
Escande, A. et al. Neutrophilia in locally advanced cervical cancer: a novel biomarker for image-guided adaptive brachytherapy? Oncotarget. 7 (46), 74886 (2016).
Madeddu, C. et al. Pathogenic and prognostic roles of paraneoplastic leukocytosis in Cervical Cancer: can genomic-based targeted therapies have a role? A literature review and an emblematic case report. Diagnostics. 12 (8), 1910 (2022).
Porras, G. O. R., Nogueda, J. C. & Chacón, A. P. Chemotherapy and molecular therapy in cervical cancer. Rep. Practical Oncol. Radiotherapy. 23 (6), 533–539 (2018).
Ma, R. et al. Radiotherapy-induced haematological and intestinal toxicity in cervical cancer. (2021), unpublished.
Degu, A., Njogu, P., Weru, I. & Karimi, P. Assessment of drug therapy problems among patients with cervical cancer at Kenyatta National Hospital, Kenya. Gynecologic Oncol. Res. Pract. 4 (1), 1–15 (2017).
Huang, J., Gu, F., Ji, T., Zhao, J. & Li, G. Pelvic bone marrow sparing intensity modulated radiotherapy reduces the incidence of the hematologic toxicity of patients with cervical cancer receiving concurrent chemoradiotherapy: a single-center prospective randomized controlled trial. Radiat. Oncol. 15, 1–9 (2020).
Koulis, T. A. et al. Anemia, leukocytosis and thrombocytosis as prognostic factors in patients with cervical cancer treated with radical chemoradiotherapy: a retrospective cohort study. Clin. Translational Radiation Oncol. 4, 51–56 (2017).
Yildirim, B. A., Guler, O. C., Kose, F. & Onal, C. The prognostic value of haematologic parameter changes during treatment in cervical cancer patients treated with definitive chemoradiotherapy. J. Obstet. Gynaecol. 39 (5), 695–701 (2019).
Cheng, J. et al. The association of pretreatment thrombocytosis with prognosis and clinicopathological significance in cervical cancer: a systematic review and meta-analysis. Oncotarget. 8 (15), 24327 (2017).
Kozasa, K. et al. Comparison of clinical utilities of the platelet count and platelet-lymphocyte ratio for predicting survival in patients with cervical cancer: a single institutional study and literature review. Oncotarget. 8 (33), 55394 (2017).
Sharma, D. & Singh, G. Thrombocytosis in gynecological cancers. J. Cancer Res. Ther. 13 (2), 193 (2017).
Sai Lalitha, B., Malini, M. & Botlagunta, M. (eds) Effect of chemoradiation on haematological parameters in cervical cancer. Proceedings of the 2nd International Conference on Computational and Bio Engineering: CBE 2020, (Springer, 2021).
Gennigens, C. et al. Correlation between hematological parameters and outcome in patients with locally advanced cervical cancer treated by concomitant chemoradiotherapy. Cancer Med. 9 (22), 8432–8443 (2020).
Boice, J. D. & Hutchison, G. B. Leukemia in women following radiotherapy for cervical cancer: ten-year follow-up of an international study. J. Natl Cancer Inst. 65 (1), 115–129 (1980).
Waggoner, S. E. Cervical cancer. Lancet. 361 (9376), 2217–2225 (2003).
Ahmed, S. S. & Mohammed, A. A. Effects of thyroid dysfunction on hematological parameters: Case controlled study. Annals Med. Surg. 57 (10), 52–55 (2020).
Enawgaw, B. et al. Haematological and immunological reference intervals for adult population in the state of Amhara, Ethiopia. Tropical Med. Int. Health. 23 (7), 765–773 (2018).
Hull, R. et al. Cervical cancer in low and middle–income countries. Oncol. Lett. 20 (3), 2058–2074 (2020).
Rose, B. S. et al. Normal tissue complication probability modeling of acute hematologic toxicity in cervical cancer patients treated with chemoradiotherapy. Int. J. Radiation Oncology* Biology* Phys. 79 (3), 800–807 (2011).
Lee, H. J. et al. Prognostic value of hematological parameters in locally advanced cervical cancer patients treated with concurrent chemoradiotherapy. Anticancer Res. 40 (1), 451–458 (2020).
Serkies, K., Badzio, A. & Jassem, J. Clinical relevance of hemoglobin level in cervical cancer patients administered definitive radiotherapy. Acta Oncol. 45 (6), 695–701 (2006).
Barkati, M. et al. Hemoglobin level in cervical cancer: a surrogate for an infiltrative phenotype. Int. J. Gynecologic Cancer 23(4). (2013).
Bishop, A. J., Allen, P. K., Klopp, A. H., Meyer, L. A. & Eifel, P. J. Relationship between low hemoglobin levels and outcomes after treatment with radiation or chemoradiation in patients with cervical cancer: has the impact of anemia been overstated? Int. J. Radiation Oncology* Biology* Phys. 91 (1), 196–205 (2015).
Meixner, E. et al. Age-dependent hematologic toxicity profiles and prognostic serologic markers in postoperative Radiochemotherapy treatment for uterine cervical Cancer. Technol. Cancer Res. Treat. 21, 15330338221118188 (2022).
Ramatillah, D. L., Gillani, S. W. & Suardi, M. Evaluation of patient treatment response in oncology ward of teaching hospital. Int. J. Pharm. Life Sci(IJPLS). 3 (11), 2082–2089 (2012).
Mell, L. K. et al. Dosimetric predictors of acute hematologic toxicity in cervical cancer patients treated with concurrent cisplatin and intensity-modulated pelvic radiotherapy. Int. J. Radiation Oncology* Biology* Phys. 66 (5), 1356–1365 (2006).
Li, J., Yang, X., Ma, J., Gong, F. & Chen, Q. Relationship of red blood cell distribution width with cancer mortality in hospital. BioMed Res. Int. 2018. (2018).
Noh, J. J. et al. The prognostic model of pre-treatment complete blood count (CBC) for recurrence in early cervical cancer. J. Clin. Med. 9 (9), 2960 (2020).
Shrivastava, R., Sharma, N., Rathore, S. & Rathore, R. Biochemical and hematological changes during the course of radiotherapy in cervix cancer patients in Gwalior region of India. Pharmacol. Online. 3, 211–214 (2009).
Wang, J. et al. Prognostic values of platelet-associated indicators in resectable cervical cancer. Dose-Response. 17 (3), 1559325819874199 (2019).
Candelaria, M., Cetina, L. & Dueñas-González, A. Anemia in cervical cancer patients: implications for iron supplementation therapy. Med. Oncol. 22, 161–168 (2005).
Shi, F. et al. Impact of hematologic toxicities during concurrent chemoradiation for cervical cancer. Obstet. Gynecol. Sci. 65 (2), 176–187 (2022).
Yuenyao, P. et al. Hematologic toxicities of cisplatin concurrent chemoradiation in cervical cancer at Ubonrajchathani Cancer Center. Srinagarind Med. J. 22 (2), 127–132 (2007).
Obermair, A. et al. Anemia before and during concurrent chemoradiotherapy in patients with cervical carcinoma: effect on progression-free survival. Int. J. Gynecologic Cancer 13(5). (2003).
Shahid, S. Review of hematological indices of cancer patients receiving combined chemotherapy & radiotherapy or receiving radiotherapy alone. Crit. Rev. Oncol. Hematol. 105, 145–155 (2016).
Silberstein, L. E., Anastasi, J. & Hematology Basic Principles and Practice E-Book: Basic Principles and Practice (Elsevier Health Sciences, 2017).
Tonia, T. et al. Erythropoietin or darbepoetin for patients with cancer. Cochrane Database Syst. Reviews 2012(12).
Adelstein, D. J. & Rodriguez, C. P. (eds) Current and Emerging Standards of Concomitant Chemoradiotherapy. Seminars in Oncology (Elsevier, 2008).
Cao, W. et al. Prognostic role of pretreatment thrombocytosis on survival in patients with cervical cancer: a systematic review and meta-analysis. World J. Surg. Oncol. 17, 1–10 (2019).
Kiptoo, V. K. & Prevalence Risk Factors and Interventional Strategies for Management of Thrombocytopenia among Adult Patients Treated for Cancer at (University of Nairobi, 2023).
Gavrilescu, M. M. et al. Clinical value of hematological biomarkers in uterine cervical cancer. Chirurgia. 111, 493–499 (2016).
Onal, C., Guler, O. C. & Yildirim, B. A. Prognostic use of pretreatment hematologic parameters in patients receiving definitive chemoradiotherapy for cervical cancer. Int. J. Gynecologic Cancer 26(6). (2016).
Tadesse, S. Preventive mechanisms and treatment of cervical cancer in Ethiopia. Gynecol. Obstet. (Sunnyvale). 5 (101), 2161–0932 (2015).
Acknowledgements
The authors would like to acknowledge the staff of the University of Gondar Specialized Hospital working in the Cancer Treatment Center.
Funding
No funding.
Author information
Authors and Affiliations
Contributions
“D.M.B. and M.A. and B.B.T. and B.B. and E.C. contributed to the design and implementation of the study, collected the data, performed the statistical analysis, performed the data interpretation, and drafted the manuscript; additionally, A.M. and N.C. T.E. and M.S. and M.T. contributed to the data interpretation and drafting. All the authors reviewed the main manuscript”.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent to participate and ethical approval
We confirm that all the procedures were performed in accordance with the relevant guidelines and regulations. Ethical approval was obtained from the Ethical Review Committee of the School of Biomedical and Laboratory Sciences, College of Medicine and Health Science, the University of Gondar (SBLS/194/2015). The objective and purpose of the study were explained to the medical director and cancer treatment director, and permission was obtained to collect the data. Since the study was retrospective, informed consent was waived by ethical review committee of the School of Biomedical and Laboratory Sciences, College of Medicine and Health Sciences University of Gondar. No personal identifiers were used for confidentiality. In addition, to keep patient information confidential, a password was used to limit access to the data.
Consent for publication
Not applicable. This manuscript does not contain any individual data.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Berta, D.M., Teketelew, B.B., Chane, E. et al. Hematological changes in women with cervical cancer before and after cancer treatment: retrospective cohort study. Sci Rep 14, 27630 (2024). https://doi.org/10.1038/s41598-024-75937-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-75937-6