Abstract
3-Hydroxyanthranillic acid (3HAA) is one of the key metabolites from the tryptophan (TRP) metabolism pathway and is associated with aging, age-associated diseases, and healthy lifespan extension. This study aims to detect 3HAA in the sweat of healthy older adults using simple, high-performance liquid chromatography (HPLC) method. Chromatographic separation using 20 mmol/L sodium acetate, 3 mmol/L zinc acetate, and 7% (v/v) acetonitrile as mobile phase is possible to simultaneous detect 3HAA, KYN, and TRP with UV and fluorescence detection, respectively, under 6 min. This method demonstrated excellent linearity with coefficient of determination (r2) greater than 0.998 for all analytes. The linear range were 0.05–6 µg/mL for TRP, 0.1–6 µg/mL for KYN and 0.2–6 µg/mL for 3HAA. Percentage recoveries from spiked in human sweat ranged from 90 ± 7–101 ± 3 for TRP, 86 ± 1–92 ± 3 for KYN, and 96 ± 1–103 ± 4 for 3HAA. The precision (%RSD) of repeatability and reproducibility is less than 3% and 6%, respectively. This method was used in a cross-sectional study with 81 participants aged 50–79 years, selected randomly from a local primary healthcare hospital’s sampling frame. A detectable amount of 3HAA was observed in all sweat samples, marking the first report of 3HAA presence in human sweat. Additionally, the results revealed that the 3HAA sweat levels increased with age analyzed in three different age groups ranging from 50–59, 60–69, and 70–79. These findings enhance our understanding of sweat profiles and their correlation with aging, potentially further improving early diagnosis, disease monitoring, and development of customized treatment programs for older adults.
Similar content being viewed by others
Introduction
The older adult population is increasing worldwide, and chronic conditions associated with aging have also increased. Many adaptive metabolic systems experience physiological alterations that typically result in functional decline with aging. The Kynurenine pathway (KP) is an important metabolic pathway for the peripheral and central catabolism of TRP, an essential amino acid, to generate KYN and its several metabolites, including 3HAA (Fig. 1). Several studies reported a correlation between an imbalance of KP metabolism and various physiological and pathological conditions such as cancer, neuroinflammation, cognitive impairment, Alzheimer’s, cardiovascular disease, obesity, diabetes, etc1,2,3,4,5,6.
For example, assays of plasma and cerebrospinal fluid (CSF) of older adults with mild cognitive impairment (MCI) show a lower level of TRP, whereas the amount of KYN was higher compared to a comparison group of healthy individuals7,8. In addition, MCI progressors were found to have 4-fold higher levels of 3HAA in plasma than non-progressors, suggesting that this elevated KP activity may lead to neurotoxicity and cognitive decline9. By contrast, it was found that the level of 3HAA in the plasma of patients with osteoporosis was almost eight times lower than in the control group10, and the 3HAA levels were also decreased in cases of coronary heart disease, stroke, and clinical depression10. Recently, it was reported that introducing 3HAA could sensitize hepatocellular carcinoma (HCC) cells and xenografts to sorafenib, the first-line drug for HCC patients, by decreasing tumor cell stemness11. In addition, 3HAA is associated with the increased lifespan of Caenorhabditis elegans and mice by increasing the resistance to oxidative stress12. These findings make the 3HAA a promising biomarker in aging and age-associated diseases.
Therefore, TRP and its metabolites can be important metabolite biomarkers for early detection or follow-up of the progression of such diseases. Even though monitoring the biomarker levels in plasma or CSF has been widely used to assess the KYN pathway and its potential implications in health and disease, the standard methods used to take samples can be invasive and difficult procedures. Recently, non-invasive monitoring of biochemical markers in other biological fluids, especially in human sweat, has recently gained significant attention in the medical field13,14,15,16. It has been previously reported that TRP and KYN can be detected in human sweat using the HPLC technique16. However, to the best of our knowledge, studies of the detection of the presence of 3HAA in sweat have not been reported, even though this compound is one of the important metabolite biomarkers in the KYN pathway. Therefore, in this work, we propose a simple HPLC protocol for the determination of 3HAA in human sweat.
Results
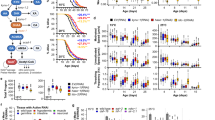
The overall scheme of this study is shown in Fig. 2. The objective is to validate the proposed method’s linearity, accuracy, precision, limit of detection (LOD), and limit of quantification (LOQ) using sweat samples.
Validation
Linearity
Linearity and detection limit were determined by analyzing a series of mixed TRP and KYN standard solutions, and individual along with 3HAA. These solutions were used to generate calibration curves at concentration from 0.05 to 6 µg/mL of 3HAA, KYN, and TRP. The signal intensities of all analytes were proportional to the concentrations in these ranges by linear regression with coefficient of determination (R2) greater than 0.999. Limit of detection (LOD) and limit of quantification (LOQ) for 3HAA, KYN, and TRP were calculated from the calibration line at the lowest concentrations using the following equations: LOD = 3 × SD, LOQ = 10 × SD, (SD: standard deviation) as previously reported17. The results are summarized in Table 1.
Accuracy
The evaluation of accuracy was conducted through a three-point recovery test. Known quantities of TRP, KYN and 3HAA were prepared at final concentrations of 1.0, 2.5 and 5.0 µg/mL. These standard solutions were spiked into the sweat sample before deproteinization. The percentage recovery of spiked samples was calculated using the formula: % recovery = [(Cspiked sample – Csample) / Cspiked standard] × 100, where C is concentration of analyte). The mean of each concentration point was calculated based on the determinations of triplicate. The results are shown in Table 2, and indicate that the described method has an efficient recovery of 90 ± 7–101 ± 3 for TRP, 86 ± 1–92 ± 3 for KYN, and 96 ± 1–103 ± 4 for 3HAA. In addition, the results of the percentage recoveries for the three analytes in six human sweat samples were also presented in Supplementary Table S2. Efficient recovery was determined to be within the range of 80–110% according to the AOAC official method of analysis18. In addition, randomly picked six sweat samples were spiked with 3.0 µg/mL standard solution and calculated for percentage of recovery (Supplementary Table S2). The accuracy of method was carried out by calculation of percentage recoveries of spiked sample. In addition, selectivity, carry over effect, and matrix effects to 3HAA were also investigated and shown in supplementary Figure S1-S3.
Precision
Precision of the method was measured by the repeatability (intra-day, n = 7) and inter-day reproducibility of standard analytes used as substitutes for human sweat. Analysis repeatability was conducted by consecutive measurements of TRP, KYN, and 3HAA. The standards were prepared in two concentrations (0.05 and 1.0 µg/mL). Each concentration had seven replicates and was measured on a workday and alternate days. Calculation of precision was based on the mean of seven replicates in each day. Precision of the method is shown in Table 3. The developed method provides good repeatability (%RSD < 3) and good reproducibility (%RSD < 6).
Characteristics of the study subjects
The study group consisted of 81 Thai adults, including 54 females and 27 males, with age ranging from 50 to 79 years (Table 4).
Application of the HPLC method on detection of TRP, KYN, and 3HAA in human sweat
The proposed method was applied to determine TRP, KYN, and 3HAA in human sweat from 81 participants. It should be noted that enzyme activity from 14 cases was not reported due to undetectable levels of KYN or TRP. The data are presented as the content of TRP and KYN in fmol per µg of Na, while 3HAA in pmol per µg of Na. Activity of enzymes IDO&TDO was defined as the ratio of KYN/TRPx100. The results are summarized by gender and age group (Tables 5 and 6, and Fig. 3). Male participants showed no statistically significant differences from females in sweat levels of Na, TRP, KYN, 3-HAA, and the KYN/TRP ratio. However, the average sweat level of TRP and KYN in females was higher than in males. Conversely, the level of Na, 3-HAA, and the KYN/TRP ratio in the sweat of females were lower than those in males. There was a difference in levels of 3HAA, KYN, and TRP between age groups. The average TRP sweat level and KYN//TRP ratio in different age groups were not significantly different. The mean KYN sweat level in the older participants (70–79 years) was significantly higher than in the middle-aged adults (50–59 years). In addition, there was a significantly higher level of 3HAA in the age group 70–79 years than those age 50–59 years.
Baseline Mean Metabolite Level Comparison by Age Group. (a) TRP (Tryptophan), (b) KYN (Kynurenine), (c) 3-HAA (3-hydroxyanthranilic acid), (d) KYN/ TRP (Kynurenine/ Tryptophan ratio) or (IDO (Indoleamine-2,3-dioxygenase) and TDO (Tryptophan-2,3-dioxygenase) activity), (e) Na (Sodium). Color re-coded by Age: 50–59 (green), 60–69 (yellow), 70–79 (red). (Statistical significance denoted by * p < 0.05). Results from 14 cases that cannot be determined either TRP or KYN in sweat were excluded.
Discussion
TRP catabolism via the kynurenine pathway has become one of the major routes in which certain metabolites are associated with diseases such as cardiovascular disease19, inflammatory diseases, and cancer20,21. Several chromatographic techniques such as Liquid chromatography-tandem mass spectrometry (LC-MS/MS) have been developed for the detection of TRP and its metabolites to provide high sensitivity and high throughput screening of the metabolites22,23,24. However, LC-MS/MS requires expensive and sophisticated equipment. Moreover, the samples are usually assessed in plasma, serum, CSF, or brain, requiring invasive methods to collect them22,23. Therefore, the main objective of this study was to evaluate the use of HPLC for sweat analysis as a simpler, non-invasive, and more economical approach than LC-MS/MS, while still producing robust, and reliable data on metabolites. Several studies have reported the potential use of HPLC-UVD and FLD to demonstrate the presence of TRP and KYN in biological fluids, e.g., mouse brain, human plasma, human CSF25, human serum14, human sweat16. However, the detection of 3HAA in human sweat has not been reported. Therefore, the HPLC was applied to analyze three key metabolites in KP: TRP, KYN, and 3HAA. The results show that this method is simple, rapid, precise, and robust as the analysis can be completed within six minutes, with linear regression of R2 greater than 0.99 for all analytes. The spike samples provided high recovery: 82.0-87.2% for TRP, 86.0-91.7% for KYN, and 95.7-103.2% for 3HAA, good repeatability (%RSD < 3), and good reproducibility (%RSD < 6).
A challenge for the development of this method was the different amounts of sweat collected from each participant, especially in older adults who tend to have less sweat compared to younger adults due to sweat gland atrophy26. Therefore, the amount of sodium was used to normalize all metabolites in each sample to compare the level of metabolites.
The results show that TRP remained unchanged with age. In addition, TRP level in this study was about 3–5 times higher than previously reported16. The higher levels of TRP observed in this study suggest that TRP can be detected using UV detection, as demonstrated previously16. However, fluorescence detection offers greater sensitivity, which is particularly advantageous for detecting low concentrations of TRP in sweat samples. A comparison between TRP detection in UV and fluorescence detectors is provided in Supplementary Fig. 4. The higher amount of TRP in this study could be due to the different exercises that participants engaged in before sweat collection. The participants in Tomasz Saran et al., performed vigorous exercise; it has been reported that people who engage in acute vigorous exercise or exhaustive exercise have lower TRP in their plasma27,28. Moreover, several factors, e.g., age, race, nutritional/health status, etc., could affect the detectable amount of TRP.
The KYN level was in a similar range as previously reported by Tomasz Saran, et al.16. It is noteworthy that the current study found that the content of KYN and 3HAA increased with age. The level of KYN in sweat showed a similar trend as that previously reported in serum and urine29,30. One study found that the increased level of KYN with age was associated with muscle atrophy and oxidative stress in in vitro and mouse model31. In addition, a higher level of KYN was observed in the serum of older adults with frailty syndrome30. Therefore, detectable KYN in sweat with increasing age could be an alternative measurement of serum KYN.
In this study, 3HAA, one of the key metabolites in KP, presents the first evidence in human sweat. This novel finding opens new possibilities for non-invasive monitoring of TRP metabolism. The presence of 3-HAA in sweat is particularly intriguing given its reported roles in nervous and immune system function10,32. Recent studies have highlighted 3HAA as a potential therapeutic target for age-associated diseases10, with correlations found between 3HAA levels and disease conditions such as Huntington’s disease33, brain injury34, stroke35, and depression36. In addition, introducing 3HAA could help to increase the lifespan in C. elegans and mouse models, suggesting its potential importance in aging processes12.
Our findings may have significant implications for the study of neurodegenerative diseases, particularly early-stage Alzheimer’s disease (AD). Previous research has shown that the 3-HAA/ anthranilic acid (AA) ratio strongly influences the odds of an individual progressing from cognitive normal or mild cognitive impairment (MCI) to MCI or AD, respectively9. Higher levels of 3-HAA and the 3-HAA/AA ratio in plasma have been associated with disease progression9. In contrast, previous single-point studies of the KP, conducted after disease diagnosis, found a significant decrease (30%) in 3-HAA in the plasma of AD patients37 while it was increased in CSF38. Interestingly, the previous study also found that 3-HAA was decreased in symptomatic AD study participants relative to progressors9. Moreover, 3-HAA has immunomodulatory effects, including inducing the death of activated T-cells while sparing non-activated ones and reducing pro-inflammatory cytokines39. These actions suggest that 3-HAA might regulate immune responses, especially relevant in aging and age-related diseases. Since the immune system helps remove amyloid plaques associated with dementia, high levels of 3-HAA could hinder this process. This suggests that 3-HAA might be a potential indicator of a weakened immune response in people who have not yet shown dementia symptoms. Interestingly, 3-HAA has been associated with lower odds of cognitive decline in prediabetes and type 2 diabetes, further emphasizing its complex relationship with various health conditions. The ability of 3-HAA to coordinate aspects of the immune response and modulate T-cell activity in the immune system adds another layer of complexity to its potential roles in health and disease.
It is important to address the limitations of our study and areas for future research. While we have successfully detected 3-HAA in sweat, we need to consider how these levels compare to those in blood and other bodily fluids. Previous studies have shown that TRP levels in plasma are typically 1800–3000 times higher than 3-HAA levels40,41. Despite variations in study design and some conflicting results, research on the kynurenine pathway (KP) in Alzheimer’s disease has reached a consensus on KP activation, evidenced by elevated kynurenine to tryptophan ratios. Most studies also report increased levels of neurotoxic metabolites quinolinic acid and 3-hydroxykynurenine in AD patients, indicating a consistent pattern of KP dysregulation in the disease1,9. Nevertheless, the mechanisms underlying variations in 3-HAA levels, such as changes in 3-HAO function, deserve further study. Understanding these mechanisms could help explain contradictory findings regarding 3-HAA levels in different disease states and potentially reveal new therapeutic targets.
Additionally, we observed that 3-HAA levels increase significantly with age in our study population. This finding warrants further investigation, as it may provide insights into age-related changes in tryptophan metabolism and their potential health implications.
Conclusion
The HPLC method we developed for this analysis proved to be simple, rapid, precise, and robust, with high recovery rates and good repeatability and reproducibility for 3-HAA, as well as for tryptophan (TRP) and kynurenine (KYN). Our pilot study demonstrating the first-time presence of 3-HAA in human sweat represents an important step forward in the non-invasive monitoring of tryptophan metabolism. While the implications of this finding are potentially far-reaching, particularly in the context of aging and neurodegenerative diseases, further research is needed to fully understand the significance of sweat 3-HAA levels and their relationship to health and disease processes. Future studies should focus on validating these findings in larger and more diverse populations, exploring the relationship between sweat and plasma 3-HAA levels, and investigating how sweat 3-HAA levels change in various disease states. This line of research could ultimately lead to the development of new diagnostic tools and therapeutic strategies for age-related diseases.
Methods
Materials and reagents
TRP, KYN, 3HAA, zinc acetate, and perchloric acid were purchased from Sigma-Aldrich, USA. Sodium acetate trihydrate was purchased from Merck, USA. Sodium chloride was purchased from VWR Chemicals, Belgium. Acetonitrile (HPLC grade) was acquired from Fisher Scientific, Korea.
The stock standard solutions of all compounds were prepared at 1 mg/mL. 3HAA was dissolved in 0.1 M of NaOH. KYN and TRP were dissolved in deionized (DI) water. The standard solutions were diluted to the concentrations of 200 µg/mL of 3HAA, 20 µg/mL of KYN, and 20 µg/mL of TRP with DI water before preparing the mixed standard solutions. Working standard solutions were mixed and freshly prepared before use by diluting the standard solutions with 6% (v/v) perchloric acid. The concentrations of the calibration curves were 0.05–6 µg/mL for TRP, 0.1–6 µg/mL for KYN, and 0.2–6 µg/mL for 3HAA.
Chromatographic conditions
The method for analysis of TRP, KYN and 3HAA by using HPLC was modified from Tomasz Saran, et al.16. The HPLC equipment was an Agilent Waters 2695 Separations Module composed of autosampler, UV-VIS detector (Water 2487 Dual λ Absorbance, USA) and fluorescence detector (Jasco FP-2020 Plus Intelligent Fluorescence Detector, Japan). An Agilent column Zorbax Eclipse Pluse C18 (4.6 × 150 mm, i.d.; 5-micron particle size) was set at 30 ºC. The mobile phase consisted of 20 mmol/L sodium acetate, 3 mmol/L zinc acetate and 7% (v/v) acetonitrile without pH adjustment (isocratic elution). The sample was filtered through a 0.45 μm membrane filter (Filtrex, Cellulose Acetate membrane, Singapore) before injection. The flow-rate was set to 1 mL/min and the injection volume was 20 µL. The wavelength of the variable wavelength (VWD) was set at 250 nm for 3HAA and KYN, while fluorescence detector (FLD) condition was used for analysis of TRP at excitation wavelength at 280 nm and emission wavelength at 354 nm. The samples were kept at 4 ºC during the analysis. The total analysis time for sweat samples was approximately six minutes, and concentrations were determined based on peak areas. Empower (version 3) software was utilized to manage the HPLC system and process chromatographic data.
Determination of sodium content in human sweat
Sodium content in sweat was measured by using Microwave Plasma-Atomic Emission Spectrometer (MP-AES) (Model: 4100, Agilent Inc. Mulgrave, VIC, Australia) with parameters of the measurement system: plasma gas-nitrogen, replicates 3 times, pump speed 15 rpm, uptake time 15 s and stabilization time 15 s. The plasma sodium was absorbed at 588.995 nm for analyzation. The sodium standard was prepared from sodium chloride.
Data of 3HAA, KYN, and TRP are presented as the content of determined substance of TRP and KYN in fmol per µg of Na, while 3HAA in pmol per µg of Na. Activity of enzymes IDO&TDO was defined as the ratio of KYN/TRP x 100. Data of participants are presented as a mean ± standard deviation (SD).
Study subjects and enrollment protocol
This study was approved by the Institutional Review Board and the Clinical Research Ethics Committee, Mahidol University (COA. No. 2023/02–027) and complied with the Declaration of Helsinki and good clinical practice guidelines. A total of 81 healthy Thai adults (27 male, 54 female) were recruited from two villages in Nakhon Pathom Province (about one hour drive from Bangkok). This was a cross-sectional study, and participants were selected randomly (with replacement) using the sampling frame of the local primary healthcare hospital. All participants received an explanation of the objective of the study and data collection protocol before being asked to sign the voluntary consent form. Informed consents were obtained from all participants. Participants who could not communicate clearly in Thai or had a disqualifying disability e.g. dementia, Alzheimer’s disease, and acute or chronic conditions that could limit the ability to participate in the study were excluded from the study.
Samples collection
The absorbent patch method modified from a previous report by Lindsay B. Baker, et al.. was used to collect regional sweat for this study42. The sample collection was performed in a private room at a local hospital with two practical nurses. Both sides of the medial border of the scapular spine of participants were cleaned with alcohol pad, prior to placing the sweat patch on both sides (35 cm2 absorbent pad, 3 M Tegaderm + Pad, USA). Each participant performed moderate exercise to induce perspiration for 30 min before the patches were removed, and the sites were cleaned with an alcohol pad. Within one minute of removal, the collection pad was removed from the patch, placed in a collection tube labelled with participant unique identifier code, date, placement, and removal time, and deposited in an iced container before storage at -20 °C prior to extraction.
Sample preparation
The obtained pads were defrosted on ice, then 0.5 mL of de-ionized water was added. The solution was separated from pads using centrifugation at 18,514 g, 4 °C for 15 min. The solutions were deproteinized with 6% (v/v) perchloric acid in a 1:1 ratio and centrifugation at 13,266 g, 4 °C for 30 min to obtain the supernatant for further analysis of sodium, TRP, KYN and 3HAA with the method described above.
Statistical analysis
GraphPad Prism software version 8.0.1 was used to calculate correlation coefficients by linear regression analysis and R square of linear regression curves. Data were analyzed as mean ± standard deviation (SD). The data were examined for normal distribution with the Kolmogorov-Smirnov test. One-way ANOVA was performed to compare the mean levels of each tryptophan metabolites between three groups of study. Whereas nonparametric data was used the jonckheere – terptra test for comparison between three groups. LSD test was used for post hoc analysis for finding the differences between all pairs of groups.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable.
Abbreviations
- AD:
-
Alzheimer’s disease
- 3HAA:
-
3-Hydroxyanthranillic acid
- CSF:
-
cerebrospinal fluid
- HCC:
-
hepatocellular carcinoma
- HPLC:
-
high-performance liquid chromatography
- KP:
-
Kynurenine pathway
- KYN:
-
kynureine
- LOD:
-
limit of detection
- LOQ:
-
limit of quantification
- MCI:
-
mild cognitive impairment
- RSD:
-
relative standard deviation
- TRP:
-
tryptophan
- UV:
-
ultraviolet
References
Mittal, S. (ed Sandeep Mittal) (Springer Internatinal Publishing, 2015).
Comim, C. M. et al. Inhibition of indoleamine 2,3-dioxygenase 1/2 prevented cognitive impairment and energetic metabolism changes in the hippocampus of adult rats subjected to polymicrobial sepsis. J. Neuroimmunol. 305, 167–171. https://doi.org/10.1016/j.jneuroim.2017.02.001 (2017).
Heisler, J. M. & O’Connor, J. C. Indoleamine 2,3-dioxygenase-dependent neurotoxic kynurenine metabolism mediates inflammation-induced deficit in recognition memory. Brain. Behav. Immun. 50, 115–124. https://doi.org/10.1016/j.bbi.2015.06.022 (2015).
Sorgdrager, F. J. H., Naudé, P. J. W., Kema, I. P., Nollen, E. A. & Deyn, P. P. Tryptophan Metabolism in Inflammaging: from biomarker to therapeutic target. Front. Immunol. 10, 2565. https://doi.org/10.3389/fimmu.2019.02565 (2019).
Yu, D. et al. The IDO inhibitor coptisine ameliorates cognitive impairment in a mouse model of Alzheimer’s disease. J. Alzheimer’s Disease: JAD. 43, 291–302. https://doi.org/10.3233/jad-140414 (2015).
Zwilling, D. et al. Kynurenine 3-monooxygenase inhibition in blood ameliorates neurodegeneration. Cell. 145, 863–874. https://doi.org/10.1016/j.cell.2011.05.020 (2011).
Solvang, S. H. et al. The kynurenine pathway and cognitive performance in community-dwelling older adults. The Hordaland Health Study. Brain. Behav. Immun. 75, 155–162. https://doi.org/10.1016/j.bbi.2018.10.003 (2019).
Whiley, L. et al. Metabolic phenotyping reveals a reduction in the bioavailability of serotonin and kynurenine pathway metabolites in both the urine and serum of individuals living with Alzheimer’s disease. Alzheimers Res. Ther. 13, 20. https://doi.org/10.1186/s13195-020-00741-z (2021).
Cespedes, M. et al. Systemic perturbations of the kynurenine pathway precede progression to dementia independently of amyloid-β. Neurobiol. Dis. 171, 105783. https://doi.org/10.1016/j.nbd.2022.105783 (2022). https://doi.org:.
Darlington, L. G. et al. On the Biological importance of the 3-hydroxyanthranilic acid: anthranilic acid ratio. Int. J. Tryptophan Res. 3, 51–59. https://doi.org/10.4137/ijtr.s4282 (2010).
Gan, G. et al. 3-hydroxyanthranic acid increases the sensitivity of hepatocellular carcinoma to sorafenib by decreasing tumor cell stemness. Cell. Death Discovery. 7, 173. https://doi.org/10.1038/s41420-021-00561-6 (2021).
Dang, H. et al. On the benefits of the tryptophan metabolite 3-hydroxyanthranilic acid in Caenorhabditis elegans and mouse aging. Nat. Commun. 14, 8338. https://doi.org/10.1038/s41467-023-43527-1 (2023).
Jankovskaja, S. et al. Non-invasive, topical sampling of potential, low-molecular weight, skin Cancer biomarkers: a study on healthy volunteers. Anal. Chem. 94, 5856–5865. https://doi.org/10.1021/acs.analchem.1c05470 (2022).
Pi, L. G., Tang, A. G., Mo, X. M., Luo, X. B. & Ao, X. More rapid and sensitive method for simultaneous determination of tryptophan and kynurenic acid by HPLC. Clin. Biochem. 42, 420–425. https://doi.org/10.1016/j.clinbiochem.2008.11.011 (2009).
Sadok, I. & Staniszewska, M. Electrochemical determination of Kynurenine Pathway metabolites—challenges and perspectives. Sensors 21, 7152. https://doi.org/10.3390/s21217152 (2021).
Saran, T. et al. Effect of 4-week physical exercises on tryptophan, kynurenine and kynurenic acid content in human sweat. Sci. Rep. 11, 11092. https://doi.org/10.1038/s41598-021-90616-6 (2021).
Çevikkalp, S. A., Löker, G. B., Yaman, M. & Amoutzopoulos, B. A simplified HPLC method for determination of tryptophan in some cereals and legumes. Food Chem. 193, 26–29. https://doi.org/10.1016/j.foodchem.2015.02.108 (2016).
Latimer, G. W. Jr. & Latimer, G. W. Jr. (eds) in Official Methods of Analysis of AOAC INTERNATIONAL (Oxford University Press, 2023).
Gáspár, R. et al. Kynurenine Pathway metabolites as potential clinical biomarkers in coronary artery disease. Front. Immunol. 12, 768560. https://doi.org/10.3389/fimmu.2021.768560 (2021).
Yu, F. et al. Association between metabolites in tryptophan-kynurenine pathway and inflammatory bowel disease: a two-sample mendelian randomization. Sci. Rep. 14, 201. https://doi.org/10.1038/s41598-023-50990-9 (2024).
Shadboorestan, A., Koual, M., Dairou, J. & Coumoul, X. The role of the Kynurenine/AhR pathway in diseases related to metabolism and Cancer. Int. J. Tryptophan Res. 16, 11786469231185102. https://doi.org/10.1177/11786469231185102 (2023).
Wang, L. S. et al. LC-MS/MS-based quantification of tryptophan metabolites and neurotransmitters in the serum and brain of mice. J. Chromatogr. B Analyt Technol. Biomed. Life Sci. 1112, 24–32. https://doi.org/10.1016/j.jchromb.2019.02.021 (2019).
Chen, Y. et al. Ultra-performance liquid chromatography-tandem mass spectrometry quantitative profiling of tryptophan metabolites in human plasma and its application to clinical study. J. Chromatogr. B Analyt Technol. Biomed. Life Sci. 1128, 121745. https://doi.org/10.1016/j.jchromb.2019.121745 (2019).
Sadok, I., Gamian, A. & Staniszewska, M. M. Chromatographic analysis of tryptophan metabolites. J. Sep. Sci. 40, 3020–3045. https://doi.org/10.1002/jssc.201700184 (2017).
Cseh, E. K. et al. HPLC method for the assessment of tryptophan metabolism utilizing separate internal standard for each detector. Anal. Biochem. 574, 7–14. https://doi.org/10.1016/j.ab.2019.03.005 (2019). https://doi.org:.
Balmain, B. N., Sabapathy, S., Louis, M. & Morris, N. R. Aging and Thermoregulatory Control: The Clinical Implications of Exercising under Heat Stress in Older Individuals. Biomed Res Int 8306154 (2018). https://doi.org/10.1155/2018/8306154
Mudry, J. M. et al. Direct effects of exercise on kynurenine metabolism in people with normal glucose tolerance or type 2 diabetes. Diabetes Metab. Res. Rev. 32, 754–761. https://doi.org/10.1002/dmrr.2798 (2016).
Strasser, B. et al. Effects of Exhaustive Aerobic Exercise on Tryptophan-Kynurenine metabolism in trained athletes. PLOS ONE. 11, e0153617. https://doi.org/10.1371/journal.pone.0153617 (2016).
Chojnacki, C. et al. Tryptophan intake and metabolism in older adults with Mood disorders. Nutrients. 12 https://doi.org/10.3390/nu12103183 (2020).
Teixeira-Gomes, A. et al. Exploring early detection of Frailty Syndrome in older adults: evaluation of Oxi-Immune markers, clinical parameters and modifiable risk factors. Antioxid. (Basel). 10. https://doi.org/10.3390/antiox10121975 (2021).
Kaiser, H. et al. Kynurenine, a Tryptophan Metabolite that increases with Age, induces muscle atrophy and lipid peroxidation. Oxid. Med. Cell. Longev. 2019 (9894238). https://doi.org/10.1155/2019/9894238 (2019).
S Espejo, L. et al. The emerging role of 3-Hydroxyanthranilic acid on C. Elegans Aging Immune function. bioRxiv. https://doi.org/10.1101/2024.01.07.574394 (2024).
Stoy, N. et al. Tryptophan metabolism and oxidative stress in patients with Huntington’s disease. J. Neurochem. 93, 611–623. https://doi.org/10.1111/j.1471-4159.2005.03070.x (2005).
Mackay, G. M. et al. Tryptophan metabolism and oxidative stress in patients with chronic brain injury. Eur. J. Neurol. 13, 30–42. https://doi.org/10.1111/j.1468-1331.2006.01220.x (2006).
Darlington, L. G. et al. Altered kynurenine metabolism correlates with infarct volume in stroke. Eur. J. Neurosci. 26, 2211–2221. https://doi.org/10.1111/j.1460-9568.2007.05838.x (2007).
Mackay, G. M. et al. Kynurenine metabolites and inflammation markers in depressed patients treated with fluoxetine or counselling. Clin. Exp. Pharmacol. Physiol. 36, 425–435. https://doi.org/10.1111/j.1440-1681.2008.05077.x (2009).
Giil, L. M. et al. Kynurenine Pathway metabolites in Alzheimer’s Disease. J. Alzheimer’s Disease: JAD. 60, 495–504. https://doi.org/10.3233/jad-170485 (2017).
Jacobs, K. R. et al. Correlation between plasma and CSF concentrations of kynurenine pathway metabolites in Alzheimer’s disease and relationship to amyloid-β and tau. Neurobiol. Aging. 80, 11–20. https://doi.org/10.1016/j.neurobiolaging.2019.03.015 (2019).
Lee, W. S. et al. The tryptophan metabolite 3-hydroxyanthranilic acid suppresses T cell responses by inhibiting dendritic cell activation. Int. Immunopharmacol. 17, 721–726. https://doi.org/10.1016/j.intimp.2013.08.018 (2013).
Brouns, R. et al. The role of tryptophan catabolism along the kynurenine pathway in acute ischemic stroke. Neurochem Res. 35, 1315–1322. https://doi.org/10.1007/s11064-010-0187-2 (2010).
Myint, A. M. et al. Tryptophan breakdown pathway in bipolar mania. J. Affect. Disord. 102, 65–72. https://doi.org/10.1016/j.jad.2006.12.008 (2007).
Baker, L. B. et al. Body map of regional vs. whole body sweating rate and sweat electrolyte concentrations in men and women during moderate exercise-heat stress. J. Appl. Physiol. (1985). 124, 1304–1318. https://doi.org/10.1152/japplphysiol.00867.2017 (2018).
Acknowledgements
This research project was supported by Mahidol University (Fundamental Fund: fiscal year 2023 by National Science Research and Innovation Fund (NSRF)) (Grant no. FF-139/2566). Partial support was also provided by the Center for Scientific Instrumentation and Platform Services grant, Faculty of Science, Mahidol University.
Author information
Authors and Affiliations
Contributions
K.P.K. designed and supervised overall research, data analysis, and criticism and prepared the manuscript (original draft, reviewed and edited), supported laboratory equipment and specimens. P.M. designed and performed the experiment, analyzed data and prepared the original draf, reviewed and edited. Y.T. designed and collected sweat samples prepared an original draft, reviewed and edited the manuscript. R.P. and S.M. designed and collected sweat samples. T.M. designed, analyzed, and critiqued HPLC and prepared the manuscript (original draft, reviewed, and edited). P.S. helped in design research, and manuscript preparation. P.K. designed and supervised overall research, data analysis, and criticism and prepared the manuscript (original draft, reviewed and edited), supported equipment, specimens and funding acquisition. All authors have approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Katewongsa, K.P., Manohong, P., Trangan, Y. et al. Determination of 3-hydroxyanthranilic acid in the sweat of healthy older adults. Sci Rep 14, 25396 (2024). https://doi.org/10.1038/s41598-024-76956-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-76956-z