Abstract
Heavy metal pollution causes severe abiotic stress in cereal crops around the world. This study investigated the effects of different concentrations (0, 100, 200, and 300 mg·kg–1) of nickel, lead, and copper stress on the growth and biochemical responses of Aegilops tauschii seedlings, to provide a reference for research on the mechanism of invasion and screening potential sources of wheat tolerance genes. The results showed that nickel, lead, and copper stress caused a significant decrease in the contents of chlorophyll a, chlorophyll b, and chlorophyll (a + b) in A. tauschii, thereby inhibiting photosynthesis to different degrees and hindering seedling growth, which was reflected in significant reductions in plant height and root length, with the most notable effect observed under stress by 300 mg·kg–1 lead. As the concentration of heavy metals increased, the activities of antioxidant enzymes (SOD, POD, and APX), non-enzymatic antioxidants (GSH and AsA), and the contents of osmotic regulatory substances (proline and soluble proteins) in A. tauschii significantly increased. Additionally, heavy metal stress increased H2O2 and TBARS levels. However, when the nickel, lead, and copper concentrations reached 300 mg·kg–1, no significant differences were found in H2O2 or TBARS levels compared to those in the CK group. To summarize, A. tauschii can mitigate the accumulation of ROS and membrane lipid peroxidation caused by heavy metal stress through self-regulation, thus exhibiting a certain degree of tolerance to stress caused by different concentrations of nickel, lead, and copper. Finally, the evaluation using the membership function method revealed that among the three heavy metals, A. tauschii exhibited the strongest adaptation to Cu, followed by Ni and Pb.
Similar content being viewed by others
Introduction
Food security is necessary for sustainable human development. Industrialization, mining, and modern agricultural techniques intensify the release of heavy metals. Toxic heavy metals can affect soil properties, reduce the effectiveness of essential nutrients for plants, and consequently affect plant morphology, structure, and biochemical responses1. Heavy metal pollution is a major abiotic stress faced by cereal crops worldwide2. Nickel and copper are micronutrients essential for plant growth and development3,4, but they can also be toxic to plants at high concentrations5,6. In contrast, lead, cadmium, and other nonessential elements can cause plant morphological, physiological, and biochemical dysfunctions at relatively low concentrations7. As a globally important strategic crop, wheat is sensitive to various biotic stressors, including heavy metals8. With the recent modification in agriculture, industrialization and urbanization, wheat production is severally declined due to excessive agricultural practices and various industrialized anthropogenic activities9. Once wheat interacts with heavy metals it triggers stress and affects plant growth, germination, plant biochemicals and eventually the loss of crop yield to biochemical responses and yield losses in wheat10. Previous research has shown that the content of heavy metals in wheat is mainly conditioned by their content in the soil11, as well as the influence of other factors, such as the pH of the soil, capacity of exchangeable cations, the cultivation system, soil moisture, temperature etc.12. Due to the lack of resistance genes in common wheat and with the development of gene editing breeding technology, wild relatives and local varieties of wheat have attracted significant attention as potential sources of ideal genes for tolerance to abiotic stress.
Aegilops tauschii, belonging to the family Poaceae, is native to Eastern Europe and Western Asia13. As a wild relative of common wheat, it possesses many valuable genes. Its D chromosome is completely homologous to that of wheat, allowing its superior genes to be transferred to wheat through gene exchange or free recombination14,15. As an invasive weed, A. tauschii has spread to several major wheat-producing provinces in China, including Henan, Shandong, and Hebei, where it affects 330,000 hectares of wheat fields16. This weed has increased from being a regional noxious weed to being a noxious weed, becoming one of the most harmful weeds in China’s winter wheat-growing regions17. Owing to inadequate prevention measures and limited control strategies, A. tauschii severely threatens the safety of wheat production in China18. However, no reports exist on the effects of heavy metal stress on A. tauschii. Therefore, this study investigated the effects of nickel, lead, and copper stress on the growth and biochemical responses of A. tauschii seedlings to provide a reference for research on the mechanism of invasion and identification of potential sources of wheat tolerance genes.
Results
The plant height and root length of A. tauschii decreased as the heavy metal concentration increased (Fig. 1). When the heavy metal concentration reached 300 mg·kg–1, plant height and root length decreased by 46% and 65% under Pb stress compare whit CK, which was greater than the 37% and 48% under Ni stress, and the 20% and 34% under Cu stress. All of these differences were statistically significant (P < 0.05).
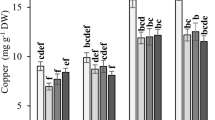
The contents of Chl a, Chl b, and Chl a + b in A. tauschii decreased as the heavy metal concentration increased (Fig. 2). Specifically, when the heavy metal concentration reached 300 mg·kg–1, the Chl a content under Pb stress decreased by 63% compared with CK, and the difference was significant (P < 0.05), while the Chl a content under Ni and Cu stress decreased by 12% and 16%, respectively, and the difference was not significant. The content of Chl b and the Chl a + b under Ni, Pb, and Cu stress decreased by 46%, 79%, 54% and 37%, 75%, 43%, respectively compared with CK, respectively, all of these difference were statistically significant (P < 0.05).
Content of Chlorophyll a, Chlorophyll b and total Chlorophyll in A. tauschii leaves induced by nickel, lead and copper stress. Mean (± SD) was calculated from three replicates for each treatment. Within each set of experimental, bars with different letters are significantly different at the 0.05 level.
As the concentration of heavy metals increased, the activities of SOD, POD, and APX in A. tauschii seedling roots continuously increased (Fig. 3). When the heavy metal concentration reached 300 mg·kg–1, the activities of SOD, POD, and APX increased significantly under Ni, Pb, and Cu stress compared with CK (P < 0.05). The CAT activity also increased with increasing heavy metal concentration. However, when the heavy metal concentration was 300 mg·kg–1, CAT activity was not significantly different from that of CK.
As the heavy metal concentration increased, the contents of GSH and AsA in the roots of A. tauschii plants increased (Fig. 4). Specifically, when the concentration of Ni and Pb was 300 mg·kg–1, the content of GSH and AsA were significantly different with CK (P < 0.05). However, the difference was not significant under Cu stress.
The H2O2 and TBARS contents in the roots of A. tauschii seedlings increased with increasing concentrations of heavy metals (Fig. 5). However, when the heavy metal concentration reached 300 mg·kg–1, the content of H2O2 and TBARS were not significantly different from CK.
The proline content in the roots of A. tauschii seedlings increased with increasing heavy metal concentration (Fig. 6). Specifically, when the heavy metal concentration reached 300 mg·kg–1, the proline content increased significantly (P < 0.05; by 88%, 91%, and 61%, respectively) under Ni, Pb, and Cu stress, compared to CK. The soluble protein content increased as the concentration of Ni and Cu, whereas it initially increased but then decreased under Pb stress (Fig. 6). When the heavy metal concentration reached 300 mg·kg–1, the soluble protein content under Ni and Cu stress increased by 22% and 2% respectively compared to CK, but the difference was not significant.
The membership function values of SOD, POD, CAT, and APX activities, as well as proline, soluble protein, H2O2, TBARS, GSH, and AsA contents of A. tauschii under Ni, Pb, and Cu stress were calculated using the fuzzy mathematics membership function method (Table 1). These values revealed that, A. tauschii had the strongest ability to adapt to Cu, followed by Ni and Pb.
Discussion
In the seedling growth and development phase, it is well known that metals cause indirect and indirect alterations in the growth of individuals by mainly affecting root development19. Tolerance to heavy metals is evaluated on the basis of the degree of root or stem growth restriction caused by toxic metals in the growth medium20. Studies have also shown that root growth is a more sensitive indicator of metal tolerance than chlorophyll determination21. The reduction in height of individual plant may be associated with reduced root growth, which in turn affected physiological and biochemical processes, such as water nutrient and absorption22. In this study, the growth of plant height and root length of A. tauschii seedlings were significantly inhibited by Ni, Pb, and Cu stress. These findings are also consistent with reports that heavy metal stress disrupts plant metabolic systems and rapidly inhibits cell division and elongation, resulting in a decrease in leaf area and plant stem growth, accompanied by a decrease in the number of flowers and fruits, as well as, a reduction in the growth of aboveground parts and roots7.
Chlorophyll is the main pigment in plant photosynthesis. Heavy metal stress can reduce the chlorophyll content, possibly through changes in the ultrastructure of chloroplasts under metal toxicity, the inhibition of enzymes involved in chlorophyll biosynthesis, and impaired absorption of essential elements, such as iron and magnesium5,23. Chlorophyll is an essential parameter used to measure photosynthetic injury caused by HM toxicity and other environmental stresses to plant. In this study, the chlorophyll content (Chl a, Chl b, and Chl a + b) decreased as the heavy metal concentration increassed, which was consistent the findings of studies on the wheat under Ni3, Pb24, and Cu stress25. Additionally, the adverse effects of heavy metal on photosynthetic pigments (chlorophyll, carotenoid) could inhibit plant growth and yield25,26. When the heavy metal concentration reached 300 mg·kg–1, Pb stress caused the greatest reduction in chlorophyll content (Chl a, Chl b, and Chl a + b), indicating the greatest inhibition of photosynthesis in seedlings by Pb stress, which was also consistent with the previous conclusions of plant height and root length.
Multiple studies have shown that when plants are stressed by heavy metals, the associated excessive production of reactive oxygen species (ROS) causes serious damage27. Plants have unique mechanism to maintain ROS at the physiological limit, counteracting their overexpression beyond a certain limit28. And this mechanism is called plant antioxidant defense system which involves enzymatic and non-enzymatic antioxidants29. The enzymatic components are superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), whereas the non-enzymatic antioxidants are ascorbate, glutathione (GSH), salicylic acid (SA)30. The activities of SOD, POD, CAT, and APX enzymes and the contents of the nonenzymatic antioxidants (GSH and AsA) in the roots of A. tauschii increased continuously with the increase of heavy metal concentration, which supports the findings of other studies that an increase in antioxidant enzyme activities and nonenzymatic antioxidant levels contributes to alleviating oxidative stress31. When the heavy metal concentration reached 300 mg·kg–1, only SOD activity significantly increased compared to CK among the four protective enzyme (P < 0.05). This indicated that A. tauschii primarily relies on increased SOD activity to cope with heavy metals stress, which might be related to the fact that SOD is generally considered to be the first line of defense against oxidative stress32. CAT converts H2O2 into H2O and O2, POD catalyzes the oxidation of various phenolic compounds using H2O2, and APX uses ascorbate as an electron donor to catalyze the reduction of H2O2. When the heavy metal concentration reached 300 mg·kg–1, the CAT activity under all three metal stress conditions was lower than that of CK, which suggested that A. tauschii may not rely primarily on CAT to detoxify H2O2 under heavy metal stress, similarly to the findings of studies on the wheat under Ni stress33.
As a nonenzymatic water-soluble antioxidant, AsA can directly interact with ROS in cells to reduce their accumulation34. GSH acts as a heavy metal chelator and is an important low-molecular-weight antioxidant involved in the GSH-AsA cycle that plays a crucial role in regulating H2O2 levels in plant cells35. Maintaining sufficient ascorbic acid and glutathione is vital under adverse conditions36. In this study, the contents of GSH and AsA increased with increasing heavy metal concentration. However, when the heavy metal concentration reached 300 mg·kg–1, there were no significant difference in GSH and AsA contents with CK under Ni, Cu and Pb stress. which might be related to the high stress tolerance adaptation of A. tauschii, a lower nonenzymatic antioxidant level sufficiently resisted the stress caused by the different concentrations of heavy metals used in this study.
TBARS is generally used to indicate the degree of plant cell membrane damage as one final decomposition product of lipid peroxidation, and reflected the ROS level under heavy metal stress37. Studies have shown that abiotic stressors, including heavy metals, increase H2O2 and TBARS levels in plant tissues38,39. In this study, the contents of H2O2 and TBARS increased with increasing concentrations of heavy metals, which was consistent with the findings of studies on wheat under Ni33, Pb40, and Cu stress41. However, when the heavy metal concentration reached 300 mg·kg–1, the contents of H2O2 and TBARS compared to CK did not reach a significant level, this indicates that A. tauschii can regulate and suppress the production of ROS and lipid peroxidation caused by heavy metal stress. Additionally, the contents of H2O2 and TBARS were significantly lower than that of CK under low concentrations of Ni, Pb, and Cu, it may be related to the enhanced antioxidant activity of A. tauschii.
Soluble proteins and proline are important osmotic regulators in plants42. Proline also scavenges ROS, stabilizes protein, chelate metal, inhibits lipid peroxidation, and acts as a redox signaling molecule. Proline plays a major role in protecting plants against heavy metal stress29. Several studies have shown that proline content is positively correlated with plant adaptation to heavy metal stress31,43. In this study, when the heavy metal concentration reached 300 mg·kg–1, the proline content of A. tauschii increased significantly compared to CK, indicating that A. tauschii relies on the accumulation of proline content to cope with Ni, Pb, and Cu stress, and this also highlights the important role of proline in alleviating the toxicity of heavy metals44. Soluble proteins act as important osmotic regulators in plant cells, and their content is considered to be a good indicator of oxidative stress under heavy metal stress45. In this study, the soluble protein content of A. tauschii continuously increased under Ni, Cu, and low-concentration Pb stress, which was attributed to moderate heavy metal stress promoting protein synthesis in the root of A. tauschii, which aligns with existing research findings3. However, as the Pb concentration increased, stress led to damage to the cell structure, thus disrupting osmotic regulation balance and inhibiting protein biosynthesis, resulting in a decrease in the soluble protein content. And this finding was similar to those reported in studies on the Phragmites australis under Cu, Pb, and Cd stress46.
The adaptation of plants to stress involves the coordinated regulation of multiple physiological indicators. To avoid the bias of using a single indicator for assessing plant tolerance, the fuzzy membership function method is commonly used to evaluate the adaptability of plants to heavy metal stress47,48,49. In this study, ten highly correlated indices, including SOD, H2O2, and GSH, were calculated using the membership function method. The results showed that the adaptability of A. tauschii to the three heavy metals followed the sequence Cu > Ni > Pb.
Conclusion
To summarize, Ni, Pb, and Cu stress led to a decrease in the contents of chlorophyll, thus inhibiting photosynthesis to different degrees and resulting in stunted growth, as determined by significantly reduced root length and plant height of A. tauschii. In addition, the increase of protective enzymes, nonenzymatic antioxidants and osmotic regulatory substances indicated that A. tauschii can reduce the production of H2O2 and TBARS through self-regulation, so as to adapt to a certain degree of heavy metal stress. Finally, a comprehensive evaluation using the membership function method indicated that the adaptability of A. tauschii to the three heavy metals followed the sequence Cu > Ni > Pb.
Materials and methods
Materials
Seeds of A. tauschii were collected from a breeding experimental field (35°18’N, 113°52’E) at the Xinjiang Academy of Agricultural Sciences, Henan Province. The thousand-grain mass of A. tauschii seeds was 11.42 g, and the average longitudinal and transverse diameters of the seeds were 5.87 and 2.46 mm, respectively. The heavy metal stress experiments were conducted in the garden plant laboratory of the College of Horticulture and Plant Protection, Henan University of Science and Technology.
Experimental design
First, bud induction treatment was conducted on seeds of A. tauschii using the Petri dish filter paper method50. Plump seeds were selected from spikelets, disinfected in a 5% sodium hypochlorite solution for 10 min, and rinsed thrice with distilled water; then, excess surface moisture was removed using filter paper. The seeds were placed in Petri dishes lined with two layers of filter paper (12 cm in diameter), with 25 seeds per dish. Germination was induced in a growth chamber under a photoperiod of 12-h/12-h light/dark cycles, with temperatures set at 25 °C during the day and 15 °C at night. Germination was considered successful when the length of the primary root reached half the length of the seed. Next, the germinated seeds were sown in plastic cups containing 200 g of coarse sand, which had been sterilized by washing, and then treated in an oven at 120 °C for 120 min. Three plants were planted in each cup, and the cups were supplemented with half-strength Hoagland nutrient solution. After three days of cultivation in the growth chamber under the same conditions as the bud induction treatment, the seedlings were treated with 0 (CK), 100, 200, or 300 mg·kg–1 nickel chloride (NiCl2), lead acetate trihydrate (Pb (CH3COO)2-3H2O), or copper sulfate pentahydrate (CuSO4-5H2O). Five cups were used for each treatment, for a total of 60 cups. Each group was subsequently supplemented with half-strength Hoagland nutrient solution to maintain the substrate moisture at 75–80%.
Measurement indicators and methods
After treatment with heavy metals for 25 days, the plants were harvested. For each treatment, 10 randomly selected seedlings were used, five for morphological measurements and five for root separation. The samples were subsequently frozen in liquid nitrogen and stored at − 80 °C for physiological and biochemical analysis.
Morphological indicators
Plant height and root length were measured using a ruler. Plant height was measured from the base of the stem to the highest point of the plant. The root length was measured as the length of the longest root of each seedling51.
Physiological indices
Chlorophyll content
Following the method adopted by Li52, 0.5 g of leaves were ground in 95% ethanol and then centrifuged at 10,000 r·min–1 for 10 min. The supernatant was collected after washing, and the supernatants were pooled twice; the final volume was adjusted to 25 mL. After the final volume was adjusted, the solution was measured for light absorption at 649 nm and 665 nm, which was used to calculate the contents of chlorophyll a, chlorophyll b, and chlorophyll (a + b).
Proline content
The proline content was determined using the acid ninhydrin colorimetric method52. Then, 0.1 g of the sample was ground with 3% sulfosalicylic acid, followed by extraction in a boiling water bath for 10 min. After cooling, the supernatant was collected as the extract. Next, 2 mL of the extract was removed, and 2 mL of glacial acetic acid and 3 mL of 2.5% acid ninhydrin were added. The mixture was heated in a boiling water bath for 30 min for color development. The mixture was cooled, and 5 mL of toluene was added for extraction. The upper layer of the solution was collected, and its absorbance was measured at 520 nm.
Antioxidant enzyme activity
The enzyme extraction buffer used was 50 mM phosphate buffer (pH 7.8) containing 20% glycerol, 1 mmol·L–1 EDTA, 1 mmol·L–1 ascorbic acid (AsA), 1 mmol·L–1 reduced glutathione, 5 mmol·L–1 MgCl2, and 1 mmol·L–1 dithiothreitol. In total, 0.5 g of fresh sample was weighed, and 5 mL of extraction buffer was added. The sample was ground in an ice bath and centrifuged at 10,000 r·min–1 for 20 min at 4 °C. Finally, the supernatant was collected for analysis of enzyme activity.
Superoxide dismutase activity was determined using the nitroblue tetrazolium photoreduction method, peroxidase activity was determined using the guaiacol method, and catalase activity was determined using the hydrogen peroxide reduction method. The soluble protein content was determined using the Coomassie Brilliant Blue G-250 method, and ascorbate peroxidase activity was measured using the UV spectrophotometric method.
Hydrogen peroxide (H2O2) and thiobarbiturate (TBARS) content
The H2O2 content was measured using the method described by Li52. Briefly, 0.2 g of sample was mixed with 2 mL of precooled acetone. After thorough grinding, the mixture was centrifuged at 10,000 r·min–1 for 10 min. Next, 1 mL of the supernatant was removed, and titanium sulfate and concentrated ammonia were added. After the mixture was centrifuged at 5,000 r·min–1 for 5 min, the precipitate was dissolved in 3 mL·L–1 sulfuric acid, and the absorbance was measured at 410 nm.
The thiobarbituric acid method was used to determine the content of thiobarbituric acid40. Briefly, 0.1 g of sample was finely chopped and then ground into a homogenate. The homogenate was centrifuged at 4,000 r·min–1 for 10 min, and the supernatant was collected and mixed with TBARS. The mixture was boiled in a water bath for 12 min, followed by centrifugation at 5,000 r·min–1 for 15 min. The absorbance of the supernatant was measured at 450, 532, and 600 nm to calculate the TBARS content.
Nonenzymatic antioxidant content
Of precooled 6% perchloric acid buffer (containing 1 mmol·L–1 EDTA, pH 2.8), 3 mL and a small amount of quartz sand were added to each 0.5 g sample. The mixture was ground into a homogenate in a precooled mortar and centrifuged at 10,000 r·min–1 for 20 min at 4 °C. The antioxidant content was measured using the method described by Anderson et al.53. The supernatant was used to determine the levels of reduced glutathione and ascorbic acid.
Data analysis
The fuzzy membership function was used to comprehensively evaluate the adaptability of A. tauschii to different types of heavy metals54, using the following equation:
Here, Zij represents the determined value of index j for plant i, Xmin represents the minimum value of the measured index, and Xmax represents the maximum value. SOD, POD, CAT, proline, protein, APX, GSH, and AsA were positively correlated with resistance to heavy metal stress, with their membership function values denoted as Zij. In contrast, H2O2 and TBARS were negatively correlated with resistance to heavy metal stress, with their membership function values expressed as 1-Zij. The mean value of all the index membership function values was calculated. A higher mean value indicated greater adaptability to heavy metal stress, whereas a lower average value indicated weaker adaptability.
All the statistical analyses were conducted using SPSS 18.0. One-way analysis of variance was performed to compare the differences in the same indices after heavy metal stress treatment. The level of significance was tested using the least significant difference test at p = 0.05. The data were summarized, and figures were plotted using Microsoft Excel. The data are expressed as the means (± SE).
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
1. Nazir, A., Rafique, F., Ahmed, K., Khan, S. A., Khan, N., Akbar, M., Zafar, M. Evaluation of heavy metals effects on morpho-anatomical alterations of wheat (Triticum aestivum L.) seedlings. Microsc. Res. Techniq. 84(11): 2517–2529. doi:10.1002/jemt.23801 (2021).
2. Din, A. M. U., Mao, H. T., Khan, A., Raza, M. A., Ahmed, M., Yuan, M., Zhang, Z. W., Yuan, S., Zhang, H. Y., Liu, Z. H., Su, Y. Q., Chen, Y. E. Photosystems and antioxidative system of rye, wheat and triticale under Pb stress. Ecotox. Environ. Safe. 249: 114356. doi:10.1016/j.ecoenv.2022.114356 (2023).
3. Atta, N., Shahbaz, M., Farhat, F., Maqsood, M. F., Zulfiqar, U., Naz, N., Ahmed, M. M., Hassan, N. U., Mujahid, N., Mustafa, A. E. M. A., Elshikh, M. S., Chaudhary, T. Proline-mediated redox regulation in wheat for mitigating nickel-induced stress and soil decontamination. Sci. Rep. 14(1): 456. doi:10.1038/s41598-023-50576-5 (2024).
4. Saleem, M. H., Kamran, M., Zhou, Y. Y., Parveen, A., Rehman, M., Ahmar, S., Malik, Z., Mustafa, A., Anjum, R. M. A., Wang, B., Liu, L. J. Appraising growth, oxidative stress and copper phytoextraction potential of flax (Linum usitatissimum L.) grown in soil differentially spiked with copper. J. Environ. Manage. Journal of environmental management, 257: 109994. doi:10.1016/j.jenvman.2019.109994 (2020).
5. Mishra, S. R., Chandra, R., Prusty, B. A. K. Chelate-assisted phytoaccumulation: growth of Helianthus annuus L., Vigna radiata (L.) R. Wilczek and Pennisetum glaucum (L.) R. Br. in soil spiked with varied concentrations of copper. Environ. Sci. Pollut. R. 27: 5074–5084. doi:10.1007/s11356-019-07257-6 (2020).
6. Farhat, F., Fry, S. C. Copper, cadmium and nickel pollution inhibit growth and promote ascorbate catabolism in cell cultures of Arabidopsis thaliana and Zea mays. Plant. Biosyst. 157(3): 699–710. doi:10.1080/11263504.2023.2188273 (2023).
7. Saleh, S. R., Kandeel, M. M., Ghareeb, D., Ghoneim, T. M., Talha, N. I., Alaoui-Sosse, B., Aleya, L., Abdel-Daim, M. M. Wheat biological responses to stress caused by cadmium, nickel and lead. Sci. Total. Environ. 706: 136013. doi:10.1016/j.scitotenv.2019.136013 (2020).
8. Alzahrani, Y., Rady, M. M. Compared to antioxidants and polyamines, the role of maize grain-derived organic biostimulants in improving cadmium tolerance in wheat plants. Ecotox. Environ. Safe. 182: 109378. doi:10.1016/j.ecoenv.2019.109378 (2019).
9. Akhtar, S., Khan, Z. I., Ahmad, K., Nadeem, M., Ejaz, A., Hussain, M. I., Ashraf, M. A. Assessment of lead toxicity in diverse irrigation regimes and potential health implications of agriculturally grown crops in Pakistan. Agr. Water. Manage. 271: 107743. doi: 10.1016/j.agwat.2022.107743. (2022).
10. Shahzad, A., Aslam, U., Ferdous, S., Qin, M. Z., Siddique, A., Billah, M., Naeem, M., Mahmood, Z., Kayani, S. Combined effect of endophytic Bacillus mycoides and rock phosphate on the amelioration of heavy metal stress in wheat plants. BMC Plant. Biol. 24(1): 125. doi: 10.1186/s12870-024-04812-3. (2024).
11. Yang, L., Ren, Q., Ge, S. J., Jiao, Z. Q., Zhan, W. H., Hou, R. X., Ruan, X. L., Pan, Y. F., Wang, Y. Y. Metal(loid)s Spatial Distribution, Accumulation, and Potential Health Risk Assessment in Soil-Wheat Systems near a Pb/Zn Smelter in Henan Province, Central China. Int. J. Environ. Res. Pub. He. 19, 2527. doi:10.3390/ijerph19052527 (2022).
12. Mickovski Stefanović, V., Roljević Nikolić, S., Matković Stojšin, M., Majstorović, H., Petreš, M., Cvikić, D., Racić, G. Soil-to-wheat transfer of heavy metals depending on the distance from the industrial zone. Agronomy. 13(4): 1016. doi: 10.3390/agronomy13041016. (2023).
13. Zhao, X. P., Bai, S. L., Li, L. C., Han, X., Li, J. H., Zhu, Y. M., Fang, Y., Zhang, D., Li, S. P. Comparative transcriptome analysis of two Aegilops tauschii with contrasting drought tolerance by RNA-Seq. Int. J. Mol. Sci. 21(10): 3595. doi:10.3390/ijms21103595 (2020).
14. Abbas, A., Hameed, R., Shahani, A. A. A., Ali, W., Huang, P., Du, Y. Z., Du, D. L. Analysis of physio-biochemical responses and expressional profiling of DREB transcription factors for drought tolerance in Aegilops tauschii Coss. Genet. Resour. Crop. Ev. 71(2): 811–822. doi: 10.21203/rs.3.rs-2775394/v1 (2024).
15. Ma, J., Wang, R., Zhao, H., Li, L., Zeng, F., Wang, Y. S., Chen, M. J., Chang, J. L., He, G. Y., Yang, G. X., Lin, Y. Genome-wide characterization of the VQ genes in Triticeae and their functionalization driven by polyploidization and gene duplication events in wheat. Int. J. Biol. Macromol. 243: 125264. doi:10.1016/j.ijbiomac.2023.125264 (2023).
16. Abbas, A., Yu, H. Y., Cui, H. L., Yu, H. L., Li, X. J. Assessment of the genetic diversity in Aegilops tauschii (coss.) By using ssr markers and morphysiological traits. Appl. Ecol. Env. Res. 18: 7011–7020. doi: 10.15666/aeer/1805_70117020 (2020).
17. Qiang, S., Zhang, H. Invasion and management of alien plants in agroecosystems in China. J. Nanjing Agric. Univ. 45(5):957–980. doi:10.7685/jnau.202206010 (2022).
18. Abbas, A., Hameed, R., Saeed, M., Shahani, A. A. A., Huang, P., Du, D. L., Zulfiqar, U., Alamri, S., Alfagham, A. T. Investigating the dynamic responses of Aegilops tauschii Coss. to salinity, drought, and nitrogen stress: a comprehensive study of competitive growth and biochemical and molecular pathways. Front. Plant. Sci. 14: 1238704. doi: 10.3389/fpls.2023.1238704 (2023).
19. Shabbir, Z., Sardar, A., Shabbir, A., Abbas, G., Shamshad, S., Khalid, S., Natasha Murtaza, G., Dumat, C., Shahid, M. Copper uptake, essentiality, toxicity, detoxification and risk assessment in soil-plant environment. Chemosphere. 259, 127436. doi:10.1016/j.chemosphere.2020.127436. (2020).
20. Gopal Rathor, G. R., Neelam Chopra, N. C., Tapan Adhikari, T. A. Effect of variation in nickel concentration on growth of maize plant: a comparative over view for pot and Hoagland culture. J. Chem. Res. R. 4(10):30–34 (2014).
21. Morgan, A. J., Evans, M., Winters, C., Gane, M., Davies, M. S. Assaying the effects of chemical ameliorants with earthworms and plants exposed to a heavily polluted metalliferous soil. Eur. J. Soil. Biol. 38(3–4): 323–327. doi:10.1016/S1164-5563(02)01168-8 (2002).
22. Mir, A. R., Pichtel, J., Hayat, S. Copper: uptake, toxicity and tolerance in plants and management of Cu-contaminated soil. BioMetals. 34, 737–759. doi:10.1007/s10534-021-00306-z. (2021).
23. Saleem, M. H., Fahad, S., Adnan, M., Ali, M., Rana, M. S., Kamran, M., Ali, Q., Hashem, I. A., Bhantana, P., Ali, M., Hussain, R. M. Foliar application of gibberellic acid endorsed phytoextraction of copper and alleviates oxidative stress in jute (Corchorus capsularis L.) plant grown in highly copper-contaminated soil of China. Environ. Sci. Pollut. R. 27: 37121–37133. doi:10.1007/s11356-020-09764-3 (2020).
24. Navabpour, S., Yamchi, A., Bagherikia, S., Kafi, H. Lead-induced oxidative stress and role of antioxidant defense in wheat (Triticum aestivum L.). Physiol. Mol. Biol. Pla. 26: 793–802. doi:10.1007/s12298-020-00777-3 (2020).
25. Riaz, M., Zhao, S., Kamran, M., Rehman, N. U., Mora-Poblete, F., Maldonado, C., Saleem, M. H., Parveen, A., Al-Ghamdi, A. A., Al-Hemaid, F. M., Ali, S., Elshikh, M. S. Effect of nano-silicon on the regulation of ascorbate-glutathione contents, antioxidant defense system and growth of copper stressed wheat (Triticum aestivum l.) seedlings. Front. Plant. Sci. 13: 986991. doi:10.3389/fpls.2022.986991 (2022).
26. Marzilli, M., Di, Santo. P., Palumbo, G., Maiuro, L., Paura, B., Tognetti, R., Cocozza, C. Cd and Cu accumulation, translocation and tolerance in Populus alba clone (Villafranca) in autotrophic in vitro screening. Environ. Sci. and Pollut. R. 25: 10058–10068. doi:10.1007/s11356-018-1299-5 (2018).
27. Mzoughi, Z., Souid, G., Timoumi, R., Le Cerf, D., Majdoub, H. Partial characterization of the edible Spinacia oleracea polysaccharides: cytoprotective and antioxidant potentials against Cd induced toxicity in HCT116 and HEK293 cells. Int J Biol Macromol. 136:332–340. doi:10.1016/j.ijbiomac.2019.06.089. (2019).
28. Singh, S., Parihar, P., Singh, R., Singh, V. P., Prasad, S. M. Heavy metal tolerance in plants: role of transcriptomics, proteomics, metabolomics, and omics. Front. Plant. Sci. 6, 1143. doi: 10.3389/fpls.2015.01143. (2016).
29. Yeboah, A., Lu, J., Yang, T., Shi, Y. Z., Amoanimaa-Dede, H., Boateng, K. G. A., Yin, X. G. Assessment of castor plant (Ricinus communis L.) tolerance to heavy metal stress-a review. Phyton-int. J. Exp. Bot. 89(3): 453. doi: 10.32604/phyton.2020.09267. (2020).
30. Yan, L. J., Allen, D. C. Cadmium-induced kidney injury: Oxidative damage as a unifying mechanism. Biomolecules. 11(11): 1575. doi:10.3390/biom11111575 (2021).
31. Hasanuzzaman, M., Nahar, K., Rahman, A., Mahmud, J. A., Alharby, H. F., Fujita, M. Exogenous glutathione attenuates lead-induced oxidative stress in wheat by improving antioxidant defense and physiological mechanisms. J. Plant. Interact. 13(1): 203–212. doi:10.1080/17429145.2018.1458913 (2018).
32. Kamran, M., Danish, M., Saleem, M. H., Malik, Z., Parveen, A., Abbasi, G. H., Jamil, M., Ali, S., Afzal, S., Riaz, M., Rizwan, M., Ali, M., Zhou, Y. Y. Application of abscisic acid and 6-benzylaminopurine modulated morpho-physiological and antioxidative defense responses of tomato (Solanum lycopersicum L.) by minimizing cobalt uptake. Chemosphere. 263: 128169. doi:10.1016/j.chemosphere.2020.128169 (2021).
33. Gajewska, E., Skłodowska, M. Effect of nickel on ROS content and antioxidative enzyme activities in wheat leaves. Biometals. 20(1): 27–36. doi:10.1007/s10534-006-9011-5 (2007).
34. Al Mahmud, J., Hasanuzzaman, M., Nahar, K., Bhuyan, M. H. M. B., Fujita, M. Insights into citric acid-induced cadmium tolerance and phytoremediation in Brassica juncea L.: Coordinated functions of metal chelation, antioxidant defense and glyoxalase systems. Ecotox. Eenviron. Safe. 147: 990–1001. doi:10.1016/j.ecoenv.2017.09.045 (2018).
35. Gajewska, E., SkŁodowska, M. Differential effect of equal copper, cadmium and nickel concentration on biochemical reactions in wheat seedlings. Ecotox. Eenviron. Safe. 73(5): 996–1003. doi:10.1016/j.ecoenv.2010.02.013 (2010).
36. Nahar, K., Hasanuzzaman, M., Alam, M. M., Rahman, A., Suzuki, T., Fujita, M. Polyamine and nitric oxide crosstalk: antagonistic effects on cadmium toxicity in mung bean plants through upregulating the metal detoxification, antioxidant defense and methylglyoxal detoxification systems. Ecotox. Eenviron. Safe. 126: 245–255. doi:10.1016/j.ecoenv.2015.12.026 (2016).
37. Tauqeer, H. M., Ali, S., Rizwan, M., Ali, Q., Saeed, R., Iftikhar, U., Ahmad, R., Abbasi, G. H. Phytoremediation of heavy metals by Alternanthera bettzickiana: growth and physiological response. Ecotox. Environ. Safe. 126: 138–146. doi: 10.1016/j.ecoenv.2015.12.031. (2016).
38. Chen, Y. E., Mao, H. T., Wu, N., Khan, A., Din, A. M. U., Ding, C. B., Zhang, Z. W., Yuan, S., Yuan, M. Different tolerance of photosynthetic apparatus to Cd stress in two rice cultivars with the same leaf Cd accumulation. Acta physiologiae plantarum, 41: 1–13. doi:10.1007/s11738-019-2981-z (2019).
39. Rehman, M. Z., Zafar, M., Waris, A. A., Rizwan, M., Ali, S., Sabir, M., Usman, M., Ayub, M. A., Ahmad, Z. Residual effects of frequently available organic amendments on cadmium bioavailability and accumulation in wheat. Chemosphere. 244: 125548. doi:10.1016/j.chemosphere.2019.125548 (2020).
40. Liu, X., Zhang, S., Shan, X., Zhu, Y. G. Toxicity of arsenate and arsenite on germination, seedling growth and amylolytic activity of wheat. Chemosphere. 61(2): 293–301. doi:10.1016/j.chemosphere.2005.01.088 (2005).
41. Groppa, M. D., Tomaro, M. L., Benavides, M. P. Polyamines and heavy metal stress: the antioxidant behavior of spermine in cadmium-and copper-treated wheat leaves. Biometals. 20: 185–195. doi:10.1007/s10534-006-9026-y (2007).
42. Bai, Y., Xiao, S., Zhang, Z., Zhang, Y., Sun, H., Zhang, K., Wang, X., Bai, Z., Li, C., Liu, L. Melatonin improves the germination rate of cotton seeds under drought stress by opening pores in the seed coat. PeerJ. 8: e9450. doi: 10.7717/peerj.9450 (2020)
43. Yang, Y. L., Zhang, Y. Y., Wei, X. L., You, J., Wang, W. R., Lu, J., Shi, R. X. Comparative antioxidative responses and proline metabolism in two wheat cultivars under short term lead stress. Ecotox. Environ. Safe. 74(4): 733–740. doi:10.1016/j.ecoenv.2010.10.035 (2011).
44. Guimarães-Soares, L., Pascoal, C., Cássio, F. Effects of heavy metals on the production of thiol compounds by the aquatic fungi Fontanospora fusiramosa and Flagellospora curta. Ecotox. Environ. Safe. 66(1): 36–43. doi:10.1016/j.ecoenv.2005.10.005 (2007).
45. Ismaiel, M. M. S., Said, A. A. Tolerance of Pseudochlorella pringsheimii to Cd and Pb stress: role of antioxidants and biochemical contents in metal detoxification. Ecotox. Environ. Safe. 164: 704–712. doi:10.1016/j.ecoenv.2018.08.088 (2018).
46. Alfadul, S. M. S., Al-Fredan, M. A. A. Effects of Cd, Cu, Pb, and Zn combinations on Phragmites australis metabolism, metal accumulation and distribution. Arab. J. Sci. Eng. 38: 11–19. doi:10.1007/s13369-012-0393-0 (2013).
47. Ye, W., Guo, G., Wu, F., Fan, T., Lu, H. Chen, H. Y., Li, X. D., Ma, Y. H. Absorption, translocation, and detoxification of Cd in two different castor bean (Ricinus communis L.) cultivars. Environmental Science and Pollution Research, 25, 28899–28906. doi: 07/s11356-018-2915-0. (2018).
48. Cao, Y., Zhang, Y., Ma, C. X., Li, H. M., Zhang, J. F., Chen, G. C. Growth, physiological responses, and copper accumulation in seven willow species exposed to Cu—a hydroponic experiment. Environ. Sci. Pollut. R. 25: 19875–19886. doi:10.1007/s11356-018-2106-z (2018).
49. Jun, Z., Wenke, W., Yani, G., Zhoufeng, W., Shumiao, C. Effect of Cd2+ stress on seed germination characteristics of ryegrass, Indian mustard and grain amaranth. Environ. Eng. Manag. J. 18(9): 1875–1884 (2019).
50. Wang, A. B., Baskin, C. C., Baskin, J. M., Ding, J. Q. Seed position in spikelet as a contributing factor to the success of the winter annual invasive grass Aegilops tauschii. Front. Plant. Sci. 13: 916451. doi:10.3389/fpls.2022.916451 (2022).
51. Dai, L., Xue, X., Li, X. L., Xiang, W. J., Fu, D. D., Zu, J. Q., Ou, X. Q. Effects of simulated water environment polluted by ionic liquid on growth and physiological characteristics of wheat seedlings. Ecol. Sci. 41(3): 166–171. doi:10.14108/j.cnki.1008-8873.2022.03.019 (2022).
52. Li, H. S. Principle and Technology of Plant Physiological Biochemical Experiment. Higher Education Press, Beijing (2000).
53. Anderson, J. V., Chevone, B. I., Hess, J. L. Seasonal variation in the antioxidant system of eastern white pine needles: evidence for thermal dependence. Plant. physiol. 98(2): 501–508. (1992).
54. Khan, J., Yang, Y., Fu, Q., Shao, W. Q., Wang, J. K., Shen, L., Huai, Y., Malangisha, G. K., Ali, A., Mahmoud, A., Lin, Y., Ren, Y. Y., Yang, J. H., Hu, Z. Y., Zhang, M. F. Screening of watermelon varieties for lead tolerance at the seedling stage. HortScience. 55(6): 858–869. doi:10.21273/HORTSCI14855-20 (2020).
Funding
This work was supported by the Natural Science Foundation of China (32271848) and Public Welfare Industry Special Research Projects of Luoyang (2202022A).
Author information
Authors and Affiliations
Contributions
N W: Data curation; methodology; formal analysis; writing; H C: Methodology; writing-review and editing; data curation; N W: writing-review and editing; investigation; Y T: Formal analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, N., Chen, H. & Tian, Y. Effects of nickel, lead, and copper stress on the growth and biochemical responses of Aegilops tauschii seedlings. Sci Rep 14, 24832 (2024). https://doi.org/10.1038/s41598-024-77143-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-77143-w
Keywords
This article is cited by
-
Protective role of Salicylic acid and sodium nitroprusside foliar application against copper stress in okra (Abelmoschus esculentus)
Scientific Reports (2025)
-
Investigating the role of bulk and nano nickel in amelioration of morphophysiology and photosynthetic activity of Triticum aestivum L.
Scientific Reports (2025)
-
Immobilizing heavy metals in contaminated soil by hydrochar modified with carboxymethyl cellulose-stabilized zinc nanoparticles
Cellulose (2025)
-
Emerging remediation approaches for mining contaminated soils by heavy metals: recent updates and future perspective
Environmental Geochemistry and Health (2025)
-
Chitosan-based Zeolitic Imidazolate Framework-8 for water remediation: kinetic and isotherm insights into the removal of organic and inorganic pollutants
Environmental Science and Pollution Research (2025)