Abstract
Polycyclic aromatic hydrocarbons (PAH) are semi-volatile, lipophilic, and harmful compounds that can persist for decades in a range of marine environments. There are several marine and soil microorganisms that possess enzymes involved in arene degradation. Here, we analyzed the structure (16S rRNA amplicons) and metabolic potential (inferred using phylogenetic placement) of the bacterial community in surface marine sediments from coastal waters off Concepción, Chile, and describe how microbial community patterns are shaped and altered by PAH contamination. Two depositional zones were identified, a “High PAH” area containing a mix of high and low molecular weight PAH of up to 10,350 ng∑PAH gdw−1 and with high organic matter content; and a “Low PAH” zone mostly characterized by low molecular weight PAH of up to 1810 ng∑PAH gdw−1 and lower levels of organic matter. We identified 53 hydrocarbonoclastic bacteria genera, with eight showing relatively high abundances at High PAH sites, although known PAH degrader clades were also present at Low PAH sites. With potential enzymes inferred in almost all samples, we suggest that breakdown of PAH is widespread in this area, likely resulting from the long history of local PAH emissions that may have promoted a stored microbial capacity for these degradation processes.
Similar content being viewed by others
Introduction
More than 350,000 compounds, or mixtures of compounds, have been registered for production worldwide1. Among these, several organic molecules with potentially negative environmental impacts have been detected in marine ecosystems, including polar regions, abyssal sediments, and the surface mixed layer of open oceans2. Chemical pollution has been recognized as one of the planetary boundaries that need to be delineated to provide limits in which humanity can safely operate3. Organic pollutants such as polycyclic aromatic hydrocarbons (PAH) can persist in the environment for several years4,5. These are semi-volatile, lipophilic compounds containing between two and seven fused aromatic rings. Pyrogenic PAH are formed during incomplete combustion of organic matter and are the most common present source of PAH. Petrogenic PAH are present in petroleum and its by-products and are associated with local point sources. Diagenetic PAH are formed from biogenic precursors and dominate sediments that were deposited before industrialization6,7. A variety of indicator ratios are used to discriminate these PAH derived from combustion of biomass and liquid fossil fuel8. PAH can be introduced into marine environments through oil spills, wastewater/sludge discharge, urban run-off, ship ballast cleaning, offshore oil exploration, natural seepage, and atmospheric deposition9,10. From over 100 known PAH compounds, 16 have shown deleterious effects on aquatic and terrestrial organisms, and on human health9, but these hydrocarbons are susceptible to degradation by microorganisms. Sediment quality guidelines focusing on organic pollutants for protection of benthic fauna have been previously described11 and can be compound-specific (e.g., phenanthrene) or class-specific (∑PAH). These guidelines provide threshold concentrations above which adverse effects might be expected12 and vary by almost an order of magnitude depending on the calculation approach. For example, the concentration guideline above which ∑PAH can cause detectable effects on benthic fauna ranges from 197 ng g−113 to 4,022 ng g−111.
Biodegradation of aromatic hydrocarbons has been studied for over a century since the first isolation of hydrocarbon-degrading bacteria14. Since then, ca. 500 bacterial, cyanobacterial, algal, and fungal strains have been identified and isolated from oil spills, and these include facultative and obligate hydrocarbon degraders15. In marine ecosystems, the group of hydrocarbonoclastic bacteria (HCB) include cosmopolitan marine genera such as Alcanivorax, Marinobacter, Thallassolituus, Cycloclasticus and Oleispira15. Degradation of PAH can occur under oxic conditions through activity of oxygenase enzymes (mono or dioxygenase) or by the addition of fumarate, as well as carboxylation, methylation, hydroxylation, or through intra-aerobic hydroxylation under anoxic conditions16. PAH are incorporated into the microbial metabolism through transformations to precursors of TCA-cycle intermediates, such as cathecol17. During aerobic catabolism, pre-conditioning for ring cleavage of PAH is initiated by dihydroxylation catalyzed by dioxygenases, whereas during anaerobic catabolism, methylation and addition of fumarate, carboxylation, and hydroxylation are the initial activation reactions18.
During the 2010 Deepwater Horizon oil spill in the Gulf of Mexico, an increase in the abundance of autochthonous aerobic hydrocarbon degraders was observed in the water column within the oil plume19. Evidence of an increase in genes associated with PAH degradation has been observed in incubation experiments amended with crude oil, using sediments collected 17 km from the site of the Deep-Water Horizon oil spill. Certain monooxygenases and dehydrogenases increased some 2.5 to 4.5-fold in comparison to controls, and genes associated with anaerobic metabolism of aromatic molecules, such as benzoyl-CoA reductase were also enriched20. Additionally, within three km of the spill, an increase was detected in sedimentary bacterial clades (particularly Deltaproteobacteria) and in genes associated with anaerobic degradation of aliphatic and aromatic hydrocarbons. These observations, together with previous evidence from the terrestrial environment21, suggest that the presence of pollutants could promote microbial communities fostering hydrocarbonoclastic taxa22. It has been suggested that microbial degradation of hydrocarbons prevents the presence of a permanent oily layer over the entire ocean surface23, but to date it is unknown which taxa are primarily responsible for the decay of contaminants, and how these microbes are distributed in chronically contaminated marine environments.
In the present study, we examined sediments from the coastal ocean off central Chile (Fig. S1). Previous studies in this area have indicated the presence of persistent organic pollutants and PAH both in the atmosphere24 and in sediments25,26, although the extension and/or dilution of the PAH signature from their potential sources along the coastal-ocean gradient remains unknown. Likely sources of PAH to the coast off central Chile include industrial activities (thermoelectric plants, oil refineries, fishing ports, shipyards, and production of fish meal, pulp and steel), maritime traffic, wildfires from pine plantations (particularly during austral summer), and urban activities (transportation and heating). Acute environmental perturbations are known to induce deterministic changes in microbial community composition27, although PAH concentrations in the study area did not generally exceed sediment quality guidelines25. However, chronic effects of PAH presence may promote development of alternative community assemblies28.
The principal goal of the present study is to determine the fingerprint and spatial distribution of PAH within surface sediments of a coastal environment perturbed by anthropogenic activities, and to determine how the presence of these compounds influences the structure and putative functions of the associated microbial community. We primarily categorized our sedimentary environment by PAH content and composition, and geochemical properties. We also evaluated potential selective pressure on microbial community composition, focusing on known taxa of hydrocarbonoclastic bacteria, particularly those showing a preference for environments enriched in PAH. This approach should advance our understanding of the interplay between the environmental microbiome and its biogeochemical background.
Results
PAH distribution in surface sediments
Contents of PAH from 31 sediment samples ranged between 81 and 10,300 ng PAH gdw−1, with the highest values recorded on the eastward side near the coast of the Gulf of Arauco (Fig. 1D, Table S1), and on the eastward side of the Bay of Concepción. Indicators of biological production (organic carbon, nitrogen, and protein-amino acids) were also higher within both the Bay of Concepción and the Gulf of Arauco (Fig. 1A, B, C), with spatial distribution resembling that of PAH. Low molecular weight PAH (three rings, < 200 g mol-1) accounted for 82 ± 23% of ∑PAH for all sampled stations (Table S1).
(A) Study area depicting sampling stations (triangles = Low PAH sites, circles = High PAH sites) and C-org (%), (B) Total nitrogen (%), (C) Proteins as total hydrolysable amino acids (µg THAA gdw−1) and D) Polycyclic aromatic hydrocarbons content (ΣPAH in Log10 ng gdw−1). Color bars represent contents for each parameter.
An analysis of principal components (PCA) was used to classify the 31 sampling sites based on relative contents of 15 PAH compounds. Those of low molecular weight were: naphthalene (Nap), acenaphthylene + acenaphthene (Acy + Ace), fluorene (Flo), phenanthrene (Phe), anthracene (Ant). High molecular weight compounds were: fluoranthene (Flu), pyrene (Pyr), benzo(a)anthracene (BaA), chrysene (Chr), benzo(b)fluoranthene (BbF), benzo(k)fluoranthene (BkF), benzo(a)pyrene (BaP), indene(1,2,3,c,d)pyrene (IP), dibenz(a,h)anthracene (DB(ah)A), and benzo(g,h,i)perylene (B(g,h,I)P). Sampling sites were separated into two clusters along the principal component 1, which explained 84% of variability (Fig. 2A). PCA resulted in a segregation of sites consistent with distributions of sedimentary organic carbon (%C-org), nitrogen (%N), δ13C (‰) and δ15N (‰), proteins from total hydrolysable amino acids (THAA), depth (m), ∑PAH, and relative content of low and high molecular weight PAH (Fig. 2B).
(A) Principal Component Analysis (PCA) of relative content of each PAH by sampling site, separated along the PC1 axes. Ellipses are generated at the 95% confidence limit leaving nine sites with high contents of PAH (yellow circles) and 22 sites with predominantly low PAH content (blue triangles). (B) Environmental fitting (Envifit) built onto the PCA of parameters %C, %N, water depth, protein as THAA, δ13C, δ15N, and relative content of low and high MW-PAH.
Nine sampling sites, hereafter named “High PAH” sites, were characterized by elevated contents of carbon and protein amino acids, with ΣPAH > 1,300 ng gdw-1 of which 50 ± 12% were high MW PAH (Fig. S2). Twenty-two sampling sites were labelled “Low PAH” sites and had lower contents of carbon and protein amino acids, with ΣPAH < 1,300 ng gdw−1 (except for site M70) consisting of almost 100% low molecular weight PAH (96 ± 7%, Fig. S2). Overall, only one sample from High PAH sites reached the Effect Range Low threshold of ∑PAH 4022 ng gdw−1 of the sediment quality guideline11. Diagnostic ratios of PAH (Fig. S3) show the principal source to be combustion of organic matter (grass, wood, coal) since ANT/178 ratio was > 0.1, BAA/228 ratio was > 0.2, IP/276 ratio > 0.5 and FLU/202 ratio > 0.5.
Microbial communities of surface sediments
A total of 71,830 different amplicon sequence variants (ASVs) were obtained from 6,087,637 16S rRNA reads. A non-metric multidimensional scaling (NMDS) analysis based on similarities of microbial community composition at the ASV level showed that all samples with high PAH content were grouped together (ANOSIM R = 0.4, p < 0.0001, Fig. 3). The low PAH samples were more dispersed on the ordination, indicating more variable community composition. There were no significant differences (Kruskal Wallis p > 0.05) in either community diversity (Shannon index) or richness (Chaos1 index) between high and low PAH sediments (Fig. S4).
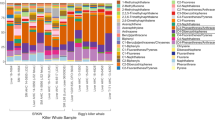
Low PAH sites contained 64% of unique ASVs, with only 21% at High PAH sites, and the remaining 15% being shared between categories (Fig. S5). Based on the taxonomic annotation, we identified 592 genera from the entire data set, 53 of which have been previously reported to degrade PAH (Fig. 4). Among these hydrocarbonoclastic genera, 13 were significantly different in relative abundance between High and Low PAH sites (eight for High PAH and five for Low PAH, Fig. S6). In High PAH sites, genera belonging to the phylum Firmicutes were: Bacillus (0.2% ± 0.1%), Desulfosporosinus (0.02% ± 0.01%) and Ruminococcus (0.001% ± 0.001%). Those belonging to the phylum Actinobacteriota were: Mycobacterium (0.02% ± 0.01%), Nocardioides (0.01% ± 0.02%), Atopobium (0.004% ± 0.01%) and Collinsella (0.002% ± 0.003%), and Gammaproteobacteria Microbulbifer (0.003% ± 0.01%). In contrast, the genera significantly more abundant in Low PAH sites included the Gammaproteobacteria Woeseia (3.5% ± 2.3%), Vibrio (0.05% ± 0.2%) and Colwellia (0.04% ± 0.07%), Spirochaeta (0.3% ± 0.2%) from the phylum Spirochaetota, and (Delta)Proteobacteria Desulfobacca (0.03% ± 0.05%).
Significant correlations (t-test, p < 0.05) between relative abundances of each of 592 genera and Principal Component 1 (Fig. 2A) resulted in 81 genera showing positive correlations (Fig. S7), with seven of them already described as HCB (Bacillus, Collinsella, Desulfosporosinus, Mycobacterium, Paenibacillus, Streptomyces and Syntrophomonas). However, the relative abundances of those 81 genera were less than 1% of total, and these were present at both High PAH and Low PAH sites. Negative correlations of genera and PC1 were significant for 58 genera, of which Woeseia and Spirochaeta had been previously described as HCB (Fig. S8).
Potential abundance of enzymes for degradation of PAH and organic matter
Using phylogenetic placement based on amplicons sequences, we identified 19 enzymes related to PAH degradation, including dehydrogenases, oxidoreductases, dioxygenases, methyltransferases, carboxy, aldehyde, and oxo-acid lyases, and synthases, with potential relative abundances ranging from undetectable to ca. 6% (Fig. 5). Six enzymes were significantly enhanced (Mann–Whitney p-value < 0.05) in High PAH sediments, four enzymes were significantly higher in Low PAH sediments, whereas nine others – although present – were not statistically different between sampling site categories (Fig. 5B). Two enzymes associated with protein and carbohydrate degradation, leucyl aminopeptidase and β-glucosidase, were found to be significantly higher by ca. 4% in Low PAH sediments (Fig. 5B). A correspondence analysis showed that predicted HCB enzymes are distributed homogeneously in all sampling sites and revealed a low contribution of PAH-degrading enzymes to variability among samples (Fig. S9).
(A) Predicted enzymes associated with degradation of PAH, carbohydrates (β-glucosidase) and proteins (Leucyl aminopeptidase). Color bar scale represents relative abundance of enzymes by sampling site; (B) Relative abundance of enzymes between High and Low PAH sites is shown as an anomaly between High PAH sediments (positive values) and Low PAH sediments (negative values). Enzyme anomalies in dark red are significantly different between High and Low PAH sites (Mann–Whitney p < 0.05).
Discussion
Contents of PAH in the present study area were within the range observed in other coastal areas with high industrial activity, such as Santander Bay in northern Spain29, Masan Bay in South Korea30, the Yangtze and Yellow River estuaries in China31, and the Baltic Sea, North Sea, and Chinese Bohai and Yellow Seas32. Previous work in the vicinity of the present study area has reported up to 200 ng ∑PAH gdw−1 in San Vicente Bay26 (Fig. S1), where major sources were attributed to both combustion products and petrogenic sources. In a brackish wetland (Lenga Estuary) connected to San Vicente Bay, PAH concentrations were between 300 to 6,000 ng PAH gdw−1, where PAH levels were positively correlated with sedimentary organic matter content25. In Concepción Bay (Fig. S1), levels up to 160 ng anthracene gdw−1, 280 ng phenanthrene gdw−1, 700 ng fluoranthene gdw−1, and 570 ng pyrene gdw-1 have been previously reported33. Overall, concentrations are typical of those found in marine sediments impacted by industrial activity and forest fires, although PAH levels were categorized below the threshold of Effect Range Low of the Sediment Quality Guideline11.
Composition of atmospheric hydrocarbons along the coastline within the study area has shown that high MW compounds account for 40 to 60% of ∑PAH24, similar to distributions observed at our High PAH sampling sites (Fig. S2). Although freshwater input represents a potentially significant input into the study area from the Itata, Andalién and Biobío rivers, no information is currently available on local riverine PAH concentrations. We suggest that at High PAH sampling sites, contaminants rapidly accumulate approximately reflecting the original composition due to favored adsorption onto biogenic particles. This is supported by observations of elevated organic carbon contents in sediments, and by a positive and significant correlation of sedimentary PAH with organic carbon in High PAH sites (r = 0.7, p < 0.001), a correlation that has also been previously observed with suspended organic carbon25. In contrast to the High PAH sites, low MW compounds predominate (ca. 90%) in Low PAH sites, with contents averaging 690 ng ∑PAH gdw−1 (Table S1). At these sites, PAH do not appear to rapidly deposit, likely due to hydrodynamics and lower concentration of biogenic particles in the water column (inferred from the lower organic carbon contents observed in sediments, Fig. 2 and Table S1). Atmospheric deposition represents the principal source of pyrogenic PAH into surface waters because of long-range transport from sources34. Low MW PAH may be transported further in the atmosphere and dispersed more widely – both in the atmosphere and water column–due to their higher volatility and solubility7. Essentially, we suggest that Low PAH sampling sites represent background contamination by PAH.
We observed that the most abundant bacterial groups in all sampling sites were the common sulfate reducers typically encountered in coastal marine sediments35, including members of Gammaproteobacteria, Deltaproteobacteria and Bacteroidota that have previously been reported in this area36. From the 53 bacterial genera previously classified as hydrocarbonoclastics, significant differences between sedimentary categories were observed for specific keystone genera, and particularly those from the Firmicutes, Actinobacteria and Gammaproteobacteria taxa (Fig. 4). Members of these taxonomic groups, along with Planctomycetes, Chloroflexi and Bacteroidetes, have been described as major components of bacterial communities in PAH biodegradation experiments conducted with anoxic soils37 and with mangrove sediments38.
However, Gammaproteobacteria (Woeseia, Vibrio and Colwellia), Spirochaetota (Spirochaeta) and Deltaproteobacteria (Desulfobacca) were more abundant in Low PAH than in High PAH sediments (Fig. S6). Although our Low PAH sites had lower contents of PAH, known clades of PAH degraders were present, with no major evidence of differences in diversity and abundance of the prokaryote community detected between PAH categories (Fig. S6). One reason could be that the ambient abundance of PAH degraders might be controlled by low substrate availability (fraction of PAH-carbon to plankton-carbon averages 0.01% in our study site). A supporting interpretation has been provided by Jeanbille et al. (2016)39 who showed that PAH presence did not promote prokaryote diversity in coastal sediments undergoing chronic contamination. Previously, Gillan et al.40 has also evidenced that microbial communities could adapt to long-term heavy metal pollution, ultimately showing similar diversity to uncontaminated sites. This interpretation is consistent with observations at our study site, which can be considered to be under chronic contamination; documented PAH emissions started ca. 70 years ago, a period representing in excess of 2000 microbial generations41.
The study of the influence of organic pollutants on the diversity of microorganisms with known degradation capabilities is a growing field42. Out of 81 genera positively correlated with PC1, only seven out of the 53 detected HCB genera showed this correlation (Fig. S7). This low representation is due to scarcity of data for certain genera showing low abundance and scattered distributions, thus precluding effective statistical analyses. Other genera such as Nocardioides and Microbulbifer did not show significant associations with PC1 because of their aerobic nature, whereas the majority of sediments were severely hypoxic or anoxic. Twenty-eight genera of the Firmicutes were positively correlated with PC1, indicating their association with environmental conditions represented by this variable. Interestingly, the Firmicutes phylum has also been shown to be an important component of bacterial communities in PAH anaerobic degradation experiments in soils37. Seven genera from the Bacteroidota also showed positive correlations with PC1; this is a phylum shown to be abundant in sediments within the study area36 and whose abundance have been shown to increase in sediments experimentally amended with PAH42. Members of the Cyanobacteria, Actinobacteria and Spirochaetota phyla have been previously shown to be capable of PAH degradation43,44,45, whereas the genus Nostoc PCC-7524 (Cyanobacteria), genera Allocatelliglobosispora, Amycolatpsis, Blastococcus, Glutamicibacter and IMCC26207 (Actinobacteria), and genera GWE2-31-10, and RBG-16-49-21 (Spirochaetota) are not known PAH degraders.
Other genera that were associated with PC1 (Fig. S7) are bacteria previously identified in coastal sediments off the Chilean coast: Subgroup 23, Lutimonas, TPD-58, Desulfobulbus, Desulfosarcina and LCP-8036. These genera are diverse, and Lutimonas–an obligate aerobe–was one of the most abundant genera in our samples, whereas Desulfobulbus, Desulfosarcina and LCP-80 are sulfate reducing bacteria. Significant positive correlations for certain taxa are indicative of enhancement in relative content of PAH to the environment as described by PC1: high contents of carbon, protein and PAH (Fig. 2). These potentially new HCB taxa show low relative abundances (< 1% of bacteria), as has been previously shown in environments undergoing chronic contamination. For instance, the presence of HCB in abundances < 1% increased to 5–10% following the Deepwater Horizon oil spill19,46. Further analyses will be required to confirm whether those positive PC1-correlated genera have PAH-degrading enzymes that are in fact functional under these conditions.
Fifty-eight genera were negatively associated with PC1 (Pearson correlation index < −0.4, Fig. S8), of which two were known HCB (Woeseia and Spirochaeta) and four were among the most abundant in our samples (Subgroup 10, Rhodopirellula, Woeseia, Blastopirellula). Another negatively correlated genus is Clade OM60(NOR5), which has also been previously negatively associated with PAH content in surface sediments of polar and subpolar regions47. We interpreted these observations as an indication that certain environments are less than favorable to certain taxa. Thus, 31 genera show aerobic metabolism, whereas others such as Woeseia and Spirochaeta are facultative anaerobic and likely to be affected by limited oxygen availability in these sediments.
Overall, the capability for degradation of PAH appears to be widespread in the present study area, with potential enzymes detected in almost all samples (Fig. 5) and likely resulting from the long local history of PAH emissions. This area is surrounded by industrial developments that began around 70 years ago and continues today, and this history possibly contributes to a stored microbial potential for PAH degradation. Slight differences in relative abundance of predicted enzymes between sediment categories were noted (Fig. 5); oxidoreductases and oxygenases were slightly less abundant than dehydrogenases, methyltransferases and lyases in all samples, suggesting that anaerobic metabolism prevails in the study area. This is consistent with the oxygenation and productivity regime of the local water column that leads to anoxic and suboxic surface sediments48.
In order to assess the reliability of our approach – which infers the potential occurrence of predicted enzymes in the microbial community – we searched for the presence of leucyl aminopeptidase (EC 3.4.11.1) and β-glucosidase (EC 3.2.1.21) in our samples (Fig. 5). These are universal extracellular enzymes that catalyze hydrolysis of proteins and carbohydrates, considered to be the initial step in degradation of planktonic organic matter, principally macromolecules49. Both enzymes were consistently present in all samples and were significantly more abundant in Low PAH sampling sites by 4% (Fig. 5). Although this distribution cannot be explained by the PAH concentrations to which our study area is exposed, a deleterious effect of PAH on enzymatic activity has been reported during incubation of freshwater sediment exposed to extremely high PAH concentrations of 300,000 ng PAH gdw−150.
One caveat of the present approach is that potential function (i.e., enzymes) is based on alignment of reads against reference genomes of bacteria. The approach provides no evidence of enhancement of enzymatic activity (for instance the half saturation constant, Km) associated with the increased potential abundance of anaerobic enzymes – relative to those for aerobic degradation of PAH – that is coincident with the oxygenation regime of surface sediments in the study area48,51. However, those patterns are likely a weighted average of the influence of all biogeochemical and geophysical conditions on the bacterial community of surface sediments.
This study evidences a widespread potential for PAH degradation in surface marine sediments distributed across the whole study area regardless of PAH content and composition. The hydrocarbonoclastic bacteria detected were both anaerobic and aerobic, and we expect changes in their abundance to occur throughout the year based on seasonal dynamics of oxygenation within these coastal sediments48. These environmental constraints, along with interactions of HCB within consortia of sedimentary microorganisms52,53, are two key aspects that could provide critical information for elucidating potential strategies for bioremediation of contaminated coastal sediments. Caution should be applied however, as the stored degradation capacity may only result from long term chronic pollution and may not represent a straightforward mechanism for biodegradation of anthropogenic chemicals. In fact, future studies in the area should also include heavy metals and other persistent organic pollutants, as well as comparison of these results with pristine areas.
Conclusions
We found that the study area exhibits levels of PAH contamination comparable to other industrialized coastal regions globally. Additionally, our results highlight the differential accumulation of PAH compounds based on their molecular weight, with high MW-PAH predominating in sites with elevated PAH contents, whereas low PAH sediments appear to represent background contamination by PAH. A widespread potential for PAH degradation was detected in the study area. Despite lower PAH contents, sites with background contamination still harboured significant populations of known PAH degraders, thus providing a study site to test whether there is microbial adaptation to chronic pollution. Certain taxa showed significant associations with concentrations of PAH, indicating their potential role as markers of hydrocarbon pollution in coastal sediments. Detection of predicted enzymes associated with PAH degradation – in almost all our samples – underscores the long-term impact of historical PAH emissions on microbial metabolic pathways in this region. Overall, anaerobic metabolism of PAH appears to be favored because predicted levels of oxidoreductases and oxygenases were slightly lower than dehydrogenases, methyltransferases and lyases. This concurs with the known oxygenation regime of the water column and high local carbon sedimentation rates that lead to anoxic sediments.
Online methods
Study area and sampling strategy
The study area encompasses a geographical area of approximately 770 km2 located in the seasonal upwelling system off Concepción, Central Chile (36°31.759’S, 72°56.967’W, Fig. S1). Here, the continental shelf is intersected by two submarine canyons of the Biobío and Itata rivers (Fig. S1) with the area subject to seasonal upwelling that promotes high primary production rates (ca. 10 g C m−2 d−1)54. Sampling was conducted during austral spring (September 2021) on board the research vessel Kay Kay II (Department of Oceanography, University of Concepción) from which 31 surface sediment samples were collected at depths ranging from 10 to 90 m (Fig. S1). Sediments were collected with a Van Veen grab, with a sample of the top centimeter removed on board and then stored in the dark at 4 °C in acid-cleaned 50 mL glass centrifuge tubes. Once in the laboratory, tubes were frozen at −80 °C prior to analyses.
Characterization of sediment organic matter
Organic carbon, nitrogen, and C and N stable isotope composition were analyzed in the Laboratory of Biogeochemistry and Applied Stable Isotopes (LABASI) of the Pontificia Universidad Católica de Chile using a Thermo Delta Advantage Isotope Ratio Mass Spectrometer coupled with Elemental Flash Analyzer (EA2000). Carbon stable isotopic ratios are reported in the δ notation with respect to Pee Dee Belemnite (δ13C = [(Rsample/Rstandard)-1], where R is 13C/12C). Nitrogen stable isotopic ratios are reported in the δ notation with respect to air (δ15N = [(Rsample/Rstandard)−1], where R is 15N/14N). Typical precision of these analyses was 0.2%.
Sedimentary proteins were determined as Total Hydrolysable Amino Acids (THAA) following Pantoja and Lee55 from ca. 200 mg subsamples of freeze-dried sediment. Briefly, after hydrolysis (6 M hydrochloric acid at 150 °C for 90 min under N2 gas), the supernatant was removed and neutralized with 6 M potassium hydroxide. Identification and quantification of amino acids was conducted using high-performance liquid chromatography (Shimadzu Nexera) following precolumn derivatization with o-phthaldialdehyde and 2-mercaptoethanol and detection by fluorescence55. Fourteen amino acids were identified and quantified: Aspartic acid, glutamic acid, serine, histidine, glycine, threonine, arginine, alanine, tyrosine, methionine, valine, phenylalanine, isoleucine, and leucine.
Determination of Polycyclic aromatic hydrocarbons (PAH)
Two-gram samples of freeze-dried and homogenized sediment were vortexed with acetone for 10 min and allowed to extract in an ultrasonic bath (40 kHz, 550 W) for 30 min 56. Samples were then centrifuged (2500 rpm, 5 min) and 1 mL of supernatant filtered (PTFE 13 mm diameter filter, 0.22 µm pore size) and injected into the HPLC (Shimadzu Nexera X2) with fluorescence detection set at three excitation and emission wavelengths (Table S2). A silica 18-C reverse phase column (Shim-pack XR-ODS III, 2.0 mm × 200 mm) was maintained at 35 °C with acetonitrile (HPLC grade) and ultrapure Milli-Q water used as mobile phase in a gradient program.
Calibration curves were constructed using a series of standard solutions (Sigma Aldrich) dissolved in acetone at concentrations ranging from 1.25 µg L−1 to 20 µg L-1. Blank samples of acetone and standard solutions were injected at the beginning and end of each batch.
To infer the origin of PAH compounds, we calculated the ANT/178, BAA/228, IP/276 and the FLU/202 ratios8. These indices relate the content of two thermodynamically similar PAH compounds based on respective molecular weights (Ant and Phe; BAA and Chr; IP and BghiP; Flu and Pyr, respectively) to determine whether sources could be of pyrolytic or petrogenic origin. Ranges of values for each index are shown in Table S3.
16S rDNA amplicon sequencing
To characterize the microbial community and composition within sampled sediments, DNA extraction was performed using a Soil DNA isolation kit (QIAGEN PowerSoil DNA Kit). DNA concentration was measured using a Nanodrop spectrophotometer and expressed as µg nL-1. Amplicon sequencing of 16S rDNA subunits v4 and v5 (primers: 515FB, 936R) were performed in Illumina MiSeq (2 × 300 PE) at the Integrated Microbiome Resource (IMR) facility using primers: 515FB and 926R57,58. Raw sequences were demultiplexed at the sequencing facility. Quality checks, trimming, chimaeras removal, read merging and taxonomy assignment (using SILVA nr99 v138.1 database) were performed using the DADA2 package in R59. Redundant fasta files were then generated using “deunique_dada2.py” phyton script (https://github.com/bowmanlab/seq_data_scripts) and used as input for the PAthway PRediction by phylogenetIC plAcement (PAPRICA) workflow60 to predict potential enzymes present in the samples. Briefly, this method aligns 16S rRNA gene reads against phylogenetic trees from all complete bacterial and archaeal genomes present in GenBank60,61 since potential enzymes in reference genomes are known. Relative abundances of amplicon sequence variants (ASV) and potential enzymes were calculated after Hellinger transformation using the Vegan package in R. Sequences obtained in the present study were deposited in the European Molecular Biology Laboratory under project accession number PRJEB57258.
The detection of hydrocarbonoclastic genera followed the method of Martinez-Varela et al.42. Briefly, a list was compiled of hydrocarbonoclastic bacteria from the available literature42,62,63 including: (i) genera identified in environments polluted by hydrocarbons from oil spills, (ii) genera observed to have growth stimulated by hydrocarbon exposure and (iii) genera observed to display hydrocarbon catabolic activity. ASVs were filtered at the genus level and genera selected that match with the hydrocarbonoclastic list. A non-parametric U-Mann Whitney test was performed to compare and differentiate hydrocarbonoclastic genera between sedimentary categories.
Relative abundance of enzymes potentially associated with PAH degradation derived from the PAPRICA pipeline was calculated as a relative anomaly between High and Low PAH sites as follows:
where mrel_ec_i_High represents the mean relative abundance of i-enzyme from High PAH sites, while mrel_ec_i_Low is the mean of the relative abundance of i-enzyme at the Low PAH sites. Thus, a positive anomaly represents greater abundance in High PAH sites, while a negative anomaly indicates that the enzyme is more prevalent in Low PAH sites. These anomalies were compared statistically between sedimentary categories using a Non-parametric U-Mann Whitney test.
Statistical analyses
Statistical analyses were performed using Vegan64 and custom R scripts (R package version 4.2.1). A Principal Component Analysis (PCA) of the relative content of each PAH at each sampling site resulted in grouping of two categories of samples. The sedimentary organic matter content (C-org, %N), protein content measured as THAA, C to N molar ratio (C/N), δ13C, δ15N, and ΣPAH content were superimposed on the PCA described before. PCA was conducted using the pcrcomp tool in R, after standardization using decostand from the Vegan package for R.
In order to identify whether microbial assemblages differed significantly between the High and Low PAH samples, a Non-metric MultiDimensional Scaling (NMDS) analysis was conducted by grouping the samples into these two categories and calculating Bray–Curtis distances, using the Vegan package. Briefly, this NMDS analysis attempts to represent the pairwise dissimilarity between samples in a low-dimensional space65. Environmental adjustment to the NMDS was performed using the envfit function in the Vegan package. Significance of differences were evaluated through Analysis of Similarities (ANOSIM). To explore relationships between potential predicted enzymes and PAH content, a Correspondence Analysis was performed for all predicted enzymes using the ca function in R66.
Correlation between identified genera and PC1
Taxonomic assignment at the level of genera from ASV were correlated with the scores of PC1 (Fig. 2A), which represents segregation of High and Low-PAH sampling sites. Significance of correlations was evaluated using a t-test from the cor.test function in R. Only significant correlations (p value < 0.05) between genera and PC1 were considered.
Data availability
Sequence data that support the findings of this study have been deposited in the European Molecular Biology Laboratory under project accession number PRJEB57258. Geochemical data is provided within the supplementary information files.
References
Wang, Z., Walker, G. W., Muir, D. C. G. & Nagatani-Yoshida, K. Toward a global understanding of chemical pollution: a first comprehensive analysis of national and regional chemical inventories. Environ. Sci. Technol. 54, 2575–2584. https://doi.org/10.1021/acs.est.9b06379 (2020).
Farrington, J. W. & Takada, H. Examples of the status, trend, and cycling of organic chemicals of environmental concern in the ocean. Oceanography 27, 196–213. https://doi.org/10.5670/oceanog.2014.23 (2014).
Richardson, K. et al. Earth beyond six of nine planetary boundaries. Sci. Adv. 9, eadh2458. https://doi.org/10.1126/sciadv.adh2458 (2024).
Zhang, Y., Wu, R. S. S., Hong, H.-S., Poon, K.-F. & Lam, M. H. W. Field study on desorption rates of polynuclear aromatic hydrocarbons from contaminated marine sediment. Environ. Toxicol. Chem. 19, 2431–2435. https://doi.org/10.1002/etc.5620191006 (2000).
Vila-Costa, M., Cerro-Gálvez, E., Martínez-Varela, A., Casas, G. & Dachs, J. Anthropogenic dissolved organic carbon and marine microbiomes. ISME J. 14, 2646–2648. https://doi.org/10.1038/s41396-020-0712-5 (2020).
Meyers, P. A. & Ishiwatari, R. Lacustrine organic geochemistry—An overview of indicators of organic matter sources and diagenesis in lake sediments. Org. Geochem. 20, 867–900. https://doi.org/10.1016/0146-6380(93)90100-P (1993).
Burgess, R. M., Ahrens, M. J. & Hickey, C. W. Geochemistry of PAHs in aquatic environments: source persistence and distribution. In PAHs: An Ecotoxicological Perspective (ed. Peter, E. T. D.) (Wiley, 2003).
Yunker, M. B. et al. PAHs in the Fraser River basin: a critical appraisal of PAH ratios as indicators of PAH source and composition. Org. Geochem. 33, 489–515. https://doi.org/10.1016/S0146-6380(02)00002-5 (2002).
U.S. Environmental Protection Agency. Quality criteria for water 1986 (“Gold Book”). USEPA Rep 440/ 5-86-001, Office of Water Regulations and Standards, USEPA.
Louvado, A. et al. Polycyclic aromatic hydrocarbons in deep sea sediments: Microbe–pollutant interactions in a remote environment. Sci. Total Environ. 526, 312–328. https://doi.org/10.1016/j.scitotenv.2015.04.048 (2015).
Long, E. R., Macdonald, D. D., Smith, S. L. & Calder, F. D. Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environ. Manag. 19, 81–97. https://doi.org/10.1007/BF02472006 (1995).
McGrath, J. A., Joshua, N., Bess, A. S. & Parkerton, T. F. Review of polycyclic aromatic hydrocarbons (PAHs) sediment quality guidelines for the protection of benthic life. Integr. Environ. Assess. Manag. 15, 505–518. https://doi.org/10.1002/ieam.4142 (2019).
Menchaca, I. et al. Determination of polychlorinated biphenyl and polycyclic aromatic hydrocarbon marine regional sediment quality guidelines within the European water framework directive. Chem. Ecol. 30, 693–700. https://doi.org/10.1080/02757540.2014.917175 (2014).
Söhngen, N. L. Benzin, petroleum, paraffinöl und paraffin als kohlenstoff-und energiequelle für mikroben. Zentr Bacteriol. Parasitenk Abt II (37), 595–609 (1913).
Yakimov, M. M., Timmis, K. N. & Golyshin, P. N. Obligate oil-degrading marine bacteria. COBIOT 18, 257–266. https://doi.org/10.1016/j.copbio.2007.04.006 (2007).
Kimes, N. E., Callaghan, A. V., Suflita, J. M. & Morris, P. J. Microbial transformation of the deepwater horizon oil spill—past, present, and future perspectives. Front. Microbiol. 5, 603. https://doi.org/10.3389/fmicb.2014.00603 (2014).
Mallick, S., Chakraborty, J. & Dutta, T. K. Role of oxygenases in guiding diverse metabolic pathways in the bacterial degradation of low-molecular-weight polycyclic aromatic hydrocarbons: A review. Crit. Rev. Microbiol. 37, 64–90. https://doi.org/10.3109/1040841X.2010.512268 (2011).
Foght, J. Anaerobic biodegradation of aromatic hydrocarbons: pathways and prospects. J. Mol. Microbiol. Biotech. 15, 93–120. https://doi.org/10.1159/000121324 (2008).
Hazen, T. C. et al. Deep-Sea oil plume enriches indigenous oil-degrading bacteria. Science 30, 204–208. https://doi.org/10.1126/science.1195979 (2010).
Bacosa, H. P. et al. Hydrocarbon degradation and response of seafloor sediment bacterial community in the northern Gulf of Mexico to light Louisiana sweet crude oil. ISME J. 12, 2532–2543. https://doi.org/10.1038/s41396-018-0190-1 (2018).
Johnsen, A. R., Wick, L. Y. & Harms, H. Principles of microbial PAH-degradation in soil. Environ. Pollut. 133, 71–84. https://doi.org/10.1016/j.envpol.2004.04.015 (2005).
Das, N. & Chandran, P. Microbial degradation of petroleum hydrocarbon contaminants: An overview. Biotechnol. Res. Int. 2011, 941810. https://doi.org/10.4061/2011/941810 (2011).
Head, I. M., Jones, D. M. & Röling, W. F. M. Marine microorganisms make a meal of oil. Nat. Rev. Microbiol. 4, 173–182. https://doi.org/10.1038/nrmicro1348 (2006).
Pozo, K. et al. Survey of persistent organic pollutants (POPs) and polycyclic aromatic hydrocarbons (PAHs) in the atmosphere of rural, urban and industrial areas of Concepción, Chile, using passive air samplers. Atmos. Pollut. Res. 3, 426–434. https://doi.org/10.5094/APR.2012.049 (2012).
Pozo, K. et al. Levels and spatial distribution of polycyclic aromatic hydrocarbons (PAHs) in sediments from Lenga Estuary, central Chile. Mar. Pollut. Bull. 62, 1572–1576. https://doi.org/10.1016/j.marpolbul.2011.04.037 (2011).
Pozo, K. et al. Multicompartmental analysis of POPs and PAHs in Concepciόn Bay, central Chile: Part I - Levels and patterns after the 2010 tsunami. Mar. Pollut. Bull. 174, 113144. https://doi.org/10.1016/j.marpolbul.2021.113144 (2022).
Dini-Andreote, F., Stegen, J. C., van Elsas, J. D. & Salles, J. F. Disentangling mechanisms that mediate the balance between stochastic and deterministic processes in microbial succession. PNAS 112, E1326–E1332. https://doi.org/10.1073/pnas.1414261112 (2015).
Potts, L. D. et al. Chronic environmental perturbation influences microbial community assembly patterns. Environ. Sci. Technol. 56, 2300–2311. https://doi.org/10.1021/acs.est.1c05106 (2022).
Viguri, J., Verde, J. & Irabien, A. Environmental assessment of polycyclic aromatic hydrocarbons (PAHs) in surface sediments of the Santander Bay Northern Spain. Chemosphere 48, 157–165. https://doi.org/10.1016/S0045-6535(02)00105-4 (2002).
Yim, U. H., Hong, S. H., Shim, W. J., Oh, J. R. & Chang, M. Spatio-temporal distribution and characteristics of PAHs in sediments from Masan Bay Korea. Mar. Pollut. Bull. 50, 319–326. https://doi.org/10.1016/j.marpolbul.2004.11.003 (2005).
Hui, Y., Zheng, M., Liu, Z. & Gao, L. Distribution of polycyclic aromatic hydrocarbons in sediments from Yellow River Estuary and Yangtze River Estuary China. J. Environ. Sci. 21, 1625–1631. https://doi.org/10.1016/S1001-0742(08)62465-1 (2009).
Wang, P. et al. Overall comparison and source identification of PAHs in the sediments of European Baltic and North Seas, Chinese Bohai and Yellow Seas. Sci. Total Environ. 737, 139535. https://doi.org/10.1016/j.scitotenv.2020.139535 (2020).
Rudolph, A., Franco, C., Becerra, J., Barros, A. & Ahumada, R. Assessment of organic matter and polynuclear aromatic hydrocarbons in surficial benthic sediments of Concepcion Bay, Chile|Evaluacion de materia organica e hidrocarburos aromaticos policlclicos en sedimentos superficiales Bahia Concepcion-Chile. Bol. Soc. Chil. Quim. 47, 403–410. https://doi.org/10.4067/S0366-16442002000400012 (2002).
Duran, R. & Cravo-Laureau, C. Role of environmental factors and microorganisms in determining the fate of polycyclic aromatic hydrocarbons in the marine environment. FEMS Microbiol. Rev. 40, 814–830. https://doi.org/10.1093/femsre/fuw031 (2016).
Zinger, L. et al. Global patterns of bacterial beta-diversity in seafloor and seawater ecosystems. PLoS One 6, e24570. https://doi.org/10.1371/journal.pone.0024570 (2011).
Fonseca, A., Espinoza, C., Nielsen, L. P., Marshall, I. P. G. & Gallardo, V. A. Bacterial community of sediments under the Eastern Boundary current system shows high microdiversity and a latitudinal spatial pattern. Front. Microbiol. 13, 1016418. https://doi.org/10.3389/fmicb.2022.1016418 (2022).
Gou, Y. et al. Enhanced degradation of polycyclic aromatic hydrocarbons in aged subsurface soil using integrated persulfate oxidation and anoxic biodegradation. Chem. Eng. J. 394, 125040. https://doi.org/10.1016/j.cej.2020.125040 (2020).
Zhang, X. Z., Xie, J. J. & Sun, F. L. Effects of three polycyclic aromatic hydrocarbons on sediment bacterial community. Curr. Microbiol. 68, 756–762. https://doi.org/10.1007/s00284-014-0535-6 (2014).
Jeanbille, M. et al. Chronic polyaromatic hydrocarbon (PAH) contamination is a marginal driver for community diversity and prokaryotic predicted functioning in coastal sediments. Front. Microbiol. https://doi.org/10.3389/fmicb.2016.01303 (2016).
Gillan, C. D., Danis, B., Pernet, P., Joly, G. & Dubois, P. Structure of sediment-associated microbial communities along a heavy-metal contamination gradient in the marine environment. Appl. Environ. Microbiol. 71, 679–690. https://doi.org/10.1128/AEM.71.2.679-690.2005 (2005).
Kevorkian, R., Bird, J. T., Shumaker, A. & Lloyd, K. G. Estimating population turnover rates by relative quantification methods reveals microbial dynamics in marine sediment. Appl. Environ. Microbiol. 84, e01443-e1517. https://doi.org/10.1128/AEM.01443-17 (2017).
Martinez-Varela, A. et al. Metatranscriptomic responses and microbial degradation of background polycyclic aromatic hydrocarbons in the coastal Mediterranean and Antarctica. Environ. Sci. Pollut. Res. 30, 119988–119999. https://doi.org/10.1007/s11356-023-30650-1 (2023).
Sundaram, S. & Soumya, K. K. Study of physiological and biochemical alterations in cyanobacterium under organic stress. Am. J. Plant Physiol. 6, 1–16. https://doi.org/10.3923/ajpp.2011.1.16 (2010).
Isaac, P., Martínez, F. L., Bourguignon, N., Sánchez, L. A. & Ferrero, M. A. Improved PAHs removal performance by a defined bacterial consortium of indigenous Pseudomonas and actinobacteria from Patagonia Argentina. Int. Biodeter. Biodegr. 101, 23–31. https://doi.org/10.1016/j.ibiod.2015.03.014 (2015).
Kümmel, S. et al. Anaerobic naphthalene degradation by sulfate-reducing Desulfobacteraceae from various anoxic aquifers. FEMS Microbiol. Ecol. 91, fiv006. https://doi.org/10.1093/femsec/fiv006 (2015).
Redmond, M. C. & Valentine, D. L. Natural gas and temperature structured a microbial community response to the Deepwater Horizon oil spill. PNAS 109, 20292–20297. https://doi.org/10.1073/pnas.1108756108 (2012).
Espínola, F. et al. Metagenomic analysis of subtidal sediments from polar and subpolar coastal environments highlights the relevance of anaerobic hydrocarbon degradation processes. Microb. Ecol. 75, 123–139. https://doi.org/10.1007/s00248-017-1028-5 (2018).
Srain, B. M. & Pantoja-Gutiérrez, S. Microbial production of toluene in oxygen minimum zone waters in the Humboldt current system off Chile. Sci. Rep. 12, 10669. https://doi.org/10.1038/s41598-022-14103-2 (2022).
Arnosti, C. et al. Extracellular enzymes in terrestrial, freshwater, and marine environments: perspectives on system variability and common research needs. Biogeochemistry 117, 5–21. https://doi.org/10.1007/s10533-013-9906-5 (2014).
Verrhiest, G. J., Clément, B., Volat, B., Montuelle, B. & Perrodin, Y. Interactions between a polycyclic aromatic hydrocarbon mixture and the microbial communities in a natural freshwater sediment. Chemosphere 46, 187–196. https://doi.org/10.1016/S0045-6535(01)00064-9 (2002).
Sobarzo, M., Bravo, L., Donoso, D., Garcés-Vargas, J. & Schneider, W. Coastal upwelling and seasonal cycles that influence the water column over the continental shelf off central Chile. Prog. Oceanogr. 75, 363–382. https://doi.org/10.1016/j.pocean.2007.08.022 (2007).
Gieg, L. M., Fowler, S. J. & Berdugo-Clavijo, C. Syntrophic biodegradation of hydrocarbon contaminants. COBIOT 27, 21–29. https://doi.org/10.1016/j.copbio.2013.09.002 (2014).
Zhang, C. et al. Marine sediments harbor diverse archaea and bacteria with the potential for anaerobic hydrocarbon degradation via fumarate addition. FEMS Microbiol. Ecol. 97, fiab045. https://doi.org/10.1093/femsec/fiab045 (2021).
Daneri, G. et al. Primary production and community respiration in the Humboldt current system off Chile and associated oceanic areas. Mar. Ecol. Prog. Ser. 197, 41–49 (2000).
Pantoja, S. & Lee, C. Amino acid remineralization and organic matter lability in Chilean coastal sediments. Org. Geochem. 34, 1047–1056. https://doi.org/10.1016/S0146-6380(03)00085-8 (2003).
Sarrazin, L. et al. Determination of polycyclic aromatic hydrocarbons (PAHs) in marine, brackish, and river sediments by HPLC, following ultrasonic extraction. J. Liq. Chromatogr. R T 29, 69–85. https://doi.org/10.1080/10826070500362987 (2006).
William, W. et al. Improved bacterial 16S rRNA gene (V4 and V4–5) and fungal internal transcribed spacer marker gene primers for microbial community surveys. mSystems https://doi.org/10.1128/msystems.00009-15 (2015).
Parada, A. E., Needham, D. M. & Fuhrman, J. A. Every base matters: assessing small subunit rRNA primers for marine microbiomes with mock communities, time series and global field samples. Environ. Microbiol. 18, 1403–1414. https://doi.org/10.1111/1462-2920.13023 (2016).
Callahan, B. J. et al. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 13, 581–583. https://doi.org/10.1038/nmeth.3869 (2016).
Bowman, J. S. & Ducklow, H. W. Microbial communities can be described by metabolic structure: a general framework and application to a seasonally variable, depth-stratified microbial community from the coastal West Antarctic Peninsula. PLoS One 10, e0135868. https://doi.org/10.1371/journal.pone.0135868 (2015).
Webb, S. J., Rabsatt, T., Erazo, N. & Bowman, J. S. Impacts of Zostera eelgrasses on microbial community structure in San Diego coastal waters. Elem. Sci. Anth. 7, 11. https://doi.org/10.1525/elementa.350 (2019).
Lozada, M., Marcos, M. S., Commendatore, M. G., Gil, M. N. & Dionisi, H. M. The bacterial community structure of hydrocarbon-polluted marine environments as the basis for the definition of an ecological index of hydrocarbon exposure. Microb. Environ. 29, 269–276. https://doi.org/10.1264/jsme2.me14028 (2014).
Karthikeyan, S. et al. Genome repository of oil systems: An interactive and searchable database that expands the catalogued diversity of crude oil-associated microbes. Environ. Microbiol. 22, 2094–2106. https://doi.org/10.1111/1462-2920.14966 (2020).
Oksanen, J. et al. vegan: Community ecology package. https://CRAN.R-project.org/package=vegan (2022).
Buttigieg, P. L. & Ramette, A. A guide to statistical analysis in microbial ecology: a community-focused, living review of multivariate data analyses. FEMS Microbiol. Ecol. 90, 543–550. https://doi.org/10.1111/1574-6941.12437 (2014).
Nenadic, O. & Greenacre, M. Correspondence analysis in R with two- and three-dimensional graphics: The ca package. J. Stat. Softw. 20, 1–13. https://doi.org/10.18637/jss.v020.i03 (2007).
Acknowledgements
We would like to thank Lilian Núñez, Víctor Acuña, Paulina Lincoñir and Marco Hidalgo for their help both in the field and in the laboratory. Special thanks to Dr. Corentin Hochart for his guidance on bioinformatic analyses and to Dr. David Crawford and two anonymous reviewers for improving the quality of this article.
Funding
The study was supported by Fondecyt grant 1200252. Special thanks to grant REDES190106 and CNRS International Associated Laboratory LIA MAST for setting up a bioinformatic internship at the Observatoire Océanologique de Banyuls-sur-Mer for CCI and GLJ. CCI acknowledges Doctoral Scholarship ANID 2020 (21200707). This research was partially supported by the supercomputing infrastructure of the NLHPC (ECM-02) and the Center for Oceanographic Research COPAS Coastal ANID FB210021.
Author information
Authors and Affiliations
Contributions
The scientific framework of this work was developed by SPG, MHG, MA, GLJ. The present study was initiated and planned by SPG and CCI. Field campaigns and laboratory analyses were conducted by CCI and AP, supervised by KHA, MA, MS, SPG. Bioinformatics analyses were conducted by CCI, supervised by GLJ, MHG, PEG. All authors contributed to data analysis and writing of the paper based on an initial version written by CCI, GLJ and SPG.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Castillo-Ilabaca, C., Gutiérrez, M.H., Aranda, M. et al. PAH contamination in coastal surface sediments and associated bacterial communities. Sci Rep 14, 29053 (2024). https://doi.org/10.1038/s41598-024-78905-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-78905-2