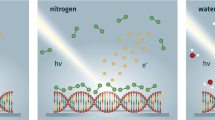
Abstract
A laser-plasma source emitting photons with energies in the water window spectral range has been used to reveal the radiation chemical yields of single-strand breaks in plasmid DNA as a function of ·OH radical scavenger concentration. Direct and indirect effects were investigated separately using DNA samples with various levels of hydration. We experimentally determined the value of the efficiency factor for strand cleavage in DNA caused by the reaction with ·OH radicals at 0.11, which was previously found in the theoretical studies. Additionally, the radiation chemical yield of ·OH radicals specific to the water window radiation emission of the source was determined by comparison with the gamma radiation-induced strand break yields. The ·OH radical yield determined using the plasmid DNA samples as a model was similar to the yield found using sensitive fluorescent dosimeters in previous experiments.
Similar content being viewed by others
Introduction
Ionizing radiation in the soft X-ray (SXR) spectral range has been used in radiation biology in many types of experiments. Those of particular interest were studies of the dependence of the radiation chemical yields of single- and double-strand breaks (SSB and DSB, respectively) in a simple deoxyribonucleic acid (DNA) molecule on the wide spectral range of photon energy, extending a few eV to several keV, to determine the values of the so-called “action spectrum” of strand breaks (see, e.g., original works1,2,3,4,5,6). The beneficial use of SXR radiation in radiobiology has been proposed due to its several typical characteristic properties. The interaction is mediated only by released photoelectrons and Auger electrons, the energy of which largely depends on the wavelength of the incident radiation. Low-energy secondary electrons travel only a few tens of nanometers, creating very short tracks7. This property is quantitatively similar to the track-ends of the high linear energy transfer (LET) radiation, where most of the energy lost by the primary charged particle comes from low-energy electrons8. Additionally, when the soft X-rays are delivered in short pulses an ultra-high dose rate irradiation conditions can be achieved in which the production of the ·OH radical is minimized or “saturated”. As previously shown, irradiation with ultra-high dose rates (UHDR) of pulsed radiation (> 40 Gy s−1) leads to biological effects referred to as the FLASH effect9, in which indirect effects caused by radical products are reduced and, in addition, mutual radical recombination appears to be favored, leading to higher yields of molecular products, as shown by experiments10,11,12,13 and theoretical studies14,15. The only exception is, if only pure water is considered16. The rapid dose delivery may result in differential response of normal and cancer cells, less oxygen depletion in normal tissues and could influence cellular repair mechanisms. Although many studies have been published on this topic so far, the physical and physico-chemical and biological phases of the FLASH effect are still under investigation. Accelerators of charged particles (e.g., LINAC, microtron, cyclotron) and conventional pulsed X-ray sources (e.g., synchrotron radiation, free-electron lasers and hot dense plasmas) deliver pulses irradiating samples at dose rates in the order of 109 Gy s−1. When tuned to a specific wavelength, SXR and X-rays simulate track-ends properties and enable radiation-driven processes to be studied as an isolated system.
The water window represents a region of the electromagnetic spectrum where soft X-rays penetrate water very easily. It is defined as the spectral range between the photon energies of the K-edge of carbon and oxygen, i.e., between the photon energies of 284 and 543 eV, respectively. It is of interest for radiobiology because the absorption of these photons in carbon- and oxygen-containing materials differs significantly. This property is used in the technique of soft X-ray microscopy, which allowed for high contrast enhancement of images of carbon structures in biological objects containing water as originally proposed by17. Considering this effect from the point of view of radiation biology, the difference in absorption causes selective energy distribution be-tween biomolecules and bulk water molecules, which may result in unexplored radiation-chemical phenomena.
Recent progress in the development of compact laser-plasma sources, emitting the photon energies in the water window, made it possible to scale the sources down to desktop dimensions18. This achievement has allowed radiobiological experiments to be conducted in a common laboratory room instead of using large-scale facilities such as synchrotrons or high pulsed power laser systems19,20. The only minor drawback may be the lower photon flux delivered by a single laser pulse, but this is compensated by a higher repetition rate, allowing for longer sample exposure.
Irradiating liquid samples with the water window soft X-rays is usually a challenging task because a vacuum is required for photon propagation. To overcome this limitation, we used a laser-plasma source capable of exposing the liquid sample placed in a small irradiation compartment filled with helium. The helium atmosphere is separated from the source placed inside a vacuum chamber by a 200-nm \(\hbox {Si}_{3}\hbox {N}_{4}\) membrane. The source design and the parameters of the produced water window radiation are presented in work of Ayele at al.21. In our previous studies (see22) we have shown how the absorbed energy generated by this desktop laser-plasma source is converted to the dose registered by chemical dosimeters and determined the radiation chemical yield of the most reactive species, the ·OH radical.
The presence of an ·OH radical scavenger in the irradiated DNA-containing system, such as Tris (tris(hydroxymethyl) aminomethane) and EDTA (ethylene diamine tetraacetic acid), may alter the reaction kinetics pathway and, consequently, the radiation chemical yield of DNA strand breaks. Considering a simple competition kinetics scheme, the ·OH radical in the irradiated system can react either with the DNA or with the scavenger molecules. Numerous earlier studies demonstrated the competition kinetics with plasmid DNA without considering the effect of EDTA in the system. The rate constant for the (Tris + ·OH) reaction is well established23, however, for EDTA, there are plenty of different published rate constants corresponding to different metal-EDTA complexes and different complex charge states at various pH levels24. In this study, we have estimated the rate constant of EDTA with the ·OH radical based on the competition kinetics with plasmid DNA under 60Co gamma irradiation.
The present study shows how the radiation chemical yields of single-strand breaks induced by soft X-rays in the water window radiation, through indirect and direct effects on a simple plasmid DNA molecule, depends on the concentration of the ·OH radical scavenger. The added ·OH radical scavengers Tris and EDTA revealed the predicted radiation chemical yield of strand breaks in a cell-like environment. A comparison with 60Co gamma radiation is made and the impact on non-homogeneous diffusion kinetics is discussed. Finally, based on the results of gamma radiation experiments and theoretical predictions, the time and scavenging capacity dependence of the ·OH radical yield induced by water window radiation is reported. We are going to utilize soft X-rays as a tool to study the formation and reactivity of the ·OH radical. In addition to radiobiology and radiation chemistry, ·OH radical plays a key role in the biochemistry of aging and oxidative stresses25, ·OH radical footprinting for mapping biopolymer structures26, and other biomedical problems and research activities27. Further development and improvement of methods, tools and techniques (e.g., refs.28,29 and present study) for a quantitative description of ·OH radical production, transport and reactions in complex aqueous systems is still necessary because many roles of ·OH radical in living matter remain unanswered or are questionable, see for example30.
Materials and methods
Plasmid DNA (pBR322, 4361 base pairs) stored in 1 × TE buffer (10 mM Tris, 1 mM EDTA, pH 8.0) was purchased from New England Biolabs. The concentration and purity of the stock solution were checked using UV absorption at 260 nm and 280 nm, respectively. The absorbance ratio at 260 to 280 nm wavelength was 1.85, indicating low protein concentration (~ 10%). Before irradiation, >97% of the plasmids were in a supercoiled conformation (without any strand breaks). This was verified by running agarose electrophoresis prior the experiment. Tris solution was prepared with Tris Pufferan (Carl Roth GmbH, GE) in a concentration of 0.01 M; pH was adjusted to 8.1 by adding 35% HCl (Penta, CZ). EDTA (Chelaton II) was purchased from Penta (CZ) and prepared in a concentration of 0.5 M by adding 5 M NaOH until dissolved and pH 8.0 was reached. The EDTA solution was further diluted to a working concentration of 0.01 M. TE buffer (10 mM Tris, 1 mM EDTA, pH 8.0) was purchased from Sigma Aldrich (GE) and used as received. All reagents were of analytical grade.
Plasmids in buffered solution
The plasmid DNA working solution intended for measuring the indirect effect was prepared by diluting the stock solution (980 ng µL−1) with deionized water to a concentration of 10 ng µL−1. Samples for SXR irradiation were pipetted in a volume of 15 µL into a small polypropylene dish attached to the target holder. After irradiation, 10 µL was recovered for strand break analysis.
To estimate the fraction of plasmid DNA that was damaged during irradiation due to solvent evaporation, the same number of samples were placed in the irradiation compartment for the same period of time as the irradiated samples (reference set). Laser pulses were accumulated in the sample as integer multiples of 500 pulses, followed by 10-second pause, up to a maximum of 3000 pulses. Irradiated and non-irradiated samples were treated and evaluated in the same way. Both procedures were repeated three times for statistical purposes.
Plasmid samples for gamma irradiation were prepared using a constant volume of plasmid DNA stock solution and topping up to the required volume of Tris, EDTA, or TE buffer to 50 µL. Therefore, concentration of plasmid DNA always remained constant at 10 µg mL−1. The residual TE concentration from the added stock solution was taken into account when calculating the working concentrations of Tris and EDTA.
Dry plasmids
Samples were pipetted from a working solution containing 20 ng µL−1 of plasmid DNA in a volume of 5 µL onto a glass coverslip. Four sample compositions were prepared, each corresponding to a different DNA/TE buffer ratio. Plasmid DNA was maintained at the same concentration in each sample composition. Coverslips with sample droplets were transferred to a nitrogen-filled cabinet and were allowed to dry completely for 60 minutes.
Coverslips with dry samples were placed on a rotary stage in the irradiation chamber with the side containing the sample facing the source. The sample irradiation setup allowed a single sample to be exposed while others were shielded from the radiation by a 2-mm thick polypropylene sheet. After irradiating one sample, the disc was rotated to the position with an unirradiated sample. Irradiated samples were removed from the chamber, the coverslip was washed with 10 µL of water, and stored in polypropylene tubes in a cooled tube rack.
Both dry and liquid samples were analyzed for strand breaks using agarose gel electrophoresis. Details of the electrophoresis assay and quantification of strand breaks are described in our previous paper31.
Dosimetry of the gamma source
In this study, we used the therapeutical 60Co radionuclide source Chisostat S01 (Chirana, CZ). The dose delivered by gamma radiation was measured using a standard air-saturated Fricke dosimeter (1 mM \(\hbox {FeSO}_{4}\), 1 mM \(\hbox {NaCl}\) in 0.4 M \(\hbox {H}_{2}\hbox {SO}_{4}\), ~ 0.25 mM \(O_{2}\)) enclosed in a 1.5 mL polypropylene tube placed in a tube rack at the reference position from the radionuclide source collimator. Fricke solution was irradiated in the same tubes and the same configuration as the samples. The volume of a single tube containing the Fricke dosimeter was 1 mL. Absorbances of the irradiated solutions at 303 nm were measured using Shimadzu UV2600 spectrometer. From the known radiation chemical yield of \(\hbox {Fe}^{3+}\) ions for gamma radiation (1.62 µmol J−1), their extinction coefficient at 303 nm (2164 M−1 cm−1) and dosimeter density (1.024 g cm−3) the slope of the linear trend was converted to the dose rate of 1.36 × 10−2 Gy s−1.
Dosimetry of the SXR source
We used a stationary pulsed laser-plasma source to generate water window radiation21. The water window radiation dose produced by the laser-plasma delivered to the liquid samples was previously determined using a Fricke dosimeter22. Briefly, from the known energy measured by the Si photodiode absorbed in the irradiated do-simeter and the measured concentration of \(\hbox {Fe}^{3+}\) ions formed, we determined their water-window-specific radiation chemical yield. The energy deposited in the liquid samples containing plasmid DNA was then found by simple scaling the dose obtained by the Fricke dosimeter to 52 mGy per pulse.
The dose delivered to the liquid sample was determined previously using the Fricke system and corresponded to 52 mGy in the present irradiated configuration. However, this dose is based on the whole volume of the irradiated sample as the Fricke and the plasmid-containing solutions could not be analyzed in smaller volumes. The local interaction of the incident radiation can be characterized using two similar dose definitions. One uses a surface dose defined as a product of the incident energy fluence and the mass absorption coefficient as given by Eq. (1) yielding 196 Gy per pulse. The other may be defined as the amount of energy absorbed per layer confined by one attenuation length, i.e., surface dose multiple of 1-1/e (~ 63% of the surface dose: 124 Gy per pulse). In both cases, during 3-ns pulse, the liquid sample is exposed locally to a dose rate in the order of \({10^{10}}\; \text {Gy}\, \text{s}^{-1}\).
The dose estimate in the dry sample DS in Gy relies on the energy-dependent mass absorption coefficient \((\mu (E)/\rho )_S\) given by the relation
where \(\Psi _0(E)\) is the spectral energy fluence of the incident water window radiation and f(E) is the energy-dependent absorption correction factor (see, e.g., Eq. A5 in6). The energy-dependent mass absorption coefficient was calculated using data from the Center for X-ray Optics database32 based on the atomic and mass composition of the sample (DNA:Tris:EDTA). The integration takes place between energies \(E_1\) to \(E_2\) (130 to 1200 eV, respectively), which cover the intensity of the spectrum. To obtain the dose in grays (J kg−1) all quantities must be expressed in SI units. A detailed description of the irradiation setup for dry samples can be found in33. Dose in the dry sample was estimated to be in a range of 24 to 44 Gy depending on the composition.
Results
Gamma irradiation
First, we have determined the rate constant of the (EDTA + ·OH) reaction. The change in the cooperative effect of scavengers was achieved in three stages: in the first one, the concentration of Tris was varied while the concentration of EDTA was kept constant; in the second, the EDTA concentration was varied and the Tris concentration was kept constant; in the third, the concentrations of Tris and EDTA were varied by changing the concentration of the TE buffer in the solution. All samples were evaluated in triplicate and the results were presented as average values.
Since both Tris and EDTA act as efficient ·OH radical scavengers, the total scavenging capacity was considered according to a general relation34
where i is the scavenging species reacting with ·OH radical. The rate constants for the scavenging reactions were taken from24 as \(k_{\text {Tris }+ ^{\cdot}\hbox {OH}} = {1.5 \times 10^{9}}\;\text {M}^{-1} \, \text{s}^{-1}\) and \(k_{\text {EDTA +} ^{\cdot}\hbox {OH}} = {3 \times 10^9}\;\text{M}^{-1} \, \text{s}^{-1}\). The rate constant for (EDTA + ·OH) reaction was chosen as a reaction rate for the EDTA-metal complex with ·OH at pH value which corresponds to a similar EDTA charge state as in the irradiated solution, i.e., pH 8.0.
We compared our data with those obtained earlier for various plasmid DNA substrates of different masses35,36. Figure 1 shows the radiation chemical yield of SSBs as a function of the scavenging capacity. We calculated the theoretical yields of SSBs according to Eq. (3) based on the published data of \([\text{DNA}]_0\) (DNA concentration in nucleotides) and SSB yield induced by the direct effect of the radiation, \(G_{\text{SSB}}^{\text{dir}}\), in references35,36. \(G_{^{\cdot}\hbox {OH}}(\sigma _{^{\cdot}\hbox {OH}})\) was taken for aerated water from ref.37 and \(\epsilon _{\text{SSB}}\) = 0.11 as suggested by LaVerne and Pimblott according to Eq. (4) in34 and the rate constant \(k = {5\times 10^9}\;\text {M}^{-1} \, \text{s}^{-1}\). Another approach to estimate the efficiency of the ·OH radical reaction for SSB formation, \(\epsilon _{\text{SSB}}\), is to fit theoretical yields to the measured data, leaving the efficiency as a free parameter. An uncertainty of 5% was obtained for the dataset used.
\(G_{\text{SSB}}\) values as measured with plasmids of three different molecular masses, which have three different concentrations of nucleotides irradiated with 60Co gamma radiation. Each data point in the dataset of our study represents an experiment for different Tris/EDTA ratios. Dashed lines are theoretical predictions for \(G_{\text{SSB}}\) based on the individual DNA concentrations (in nucleotides: \([\text{DNA}]_0\)), plasmid length (gyration radius, diffusion coefficient), diffusion rate constant defined by Eq. (5), from37, \(\epsilon _{\text{SSB}}\) = 0.11 as suggested in34 and \(G_{\text{SSB}}^{\text{dir}}\) estimate. The theoretical yields do not represent fits to the data.
The theoretical radiation chemical yield of SSB can be calculated as
where \([\text{DNA}]_0\) is the DNA concentration expressed in a number of nucleotides, \(G_{^{\cdot}\hbox {OH}}(\sigma _{^{\cdot}\hbox {OH}})\) is the scavenging-capacity-dependent radiation chemical yield of the ·OH radical, \(\epsilon _{\text{SSB}}\) is the conversion efficiency of the reacted radical to SSB, \(\sigma _{^{\cdot}\hbox {OH}}\) is the scavenging capacity, and \(k_{\text{exp}}\) is the experimental rate constant of the ·OH reaction with DNA derived using a spherical model of Mark et al.38. The experimental rate constant \(k_{\text{exp}}\) is calculated according to the relation
where k is the rate constant which is to be compared with slower non-diffusion-controlled reactions (see p. 199 in39). The diffusion rate constant, \(k_{\text{diff}}\), is derived from non-homogeneous steady-state kinetics as
where R is the DNA gyration radius calculated from its mass by the method developed by Latulippe and Zydney40, \(D_{^{\cdot}\hbox {OH}}\) and \(D_{\text{plasmid}}\) are the diffusion coefficients of the ·OH radical and the plasmid molecule, respectively, and l is plasmid length in base pairs. The diffusion coefficient of the plasmid molecule was calculated by the method developed by Prazeres41. The parameter L represents the mean diffusion distance of the ·OH radical before the reaction and is given by
The product of the Avogadro’s number, \(N_A\), in Eq. (5), and numerical factor divided by the number of nucleotides, which is twice the plasmid length in bp, converts the diffusion rate constant to units of \(\text {M}^{-1} \, \text{s}^{-1}\) per nucleotide. Note that the definition of the diffusion rate constant is implicit, meaning, that its value has to be found iteratively, since the value of L is dependent on the rate constant itself.
Irradiation in the water window
In the case of liquid samples, the evaluated plasmid form fractions were plotted as a function of the dose, shown in Fig. 2. The probability coefficients \(\beta _{\text{SSB}}\) and \(\beta _{\text{DSB}}\), which represent the average number of SSBs and DSBs per Gy, respectively, were found by fitting dose response curves to a set of equations describing the dose-dependent kinetics of all three plasmid forms developed in31. The values of the probability coefficients, which describe the action of radiation only, were found by subtracting the values of the background response from the values given by the irradiated response, e.g., \(\beta = \beta ^{\text{irr}} - \beta ^{\text{bck}}\). This correction had to be applied because the helium flow in the irradiation compartment caused the sample solvent to evaporate, increasing the DNA concentration.
Fraction of the supercoiled form decreasing as the function of the dose. Top abscissa indicates the time the samples spent in He (see details of irradiation by SXR in22.
Direct effect
We note that by the term “direct effect” in the context of the present study we mean a process that has been previously termed “direct-type effect”42. Ionizations can take place both on the DNA molecule and in its solvation shell. Holes created in the inner solvation shell (2.5 - 8 water molecules per nucleotide) transfer directly to the DNA backbone, where they cannot be distinguished from those created directly in the molecule43.
Dry samples served as a probe for the estimation of the radiation chemical yield of strand breaks caused by the direct-type effect. The concentration of the buffer salts was varied while the mass of the plasmid DNA was kept constant. Tha radiation chemical yields of strand breaks in \(\text {mol} \, \text{J}^{-1}\) were calculated from the value of \(D_{37}\), i.e., the dose responsible for the induction of a single SSB or DSB in one plasmid on average. Details of the dry plasmid analysis are described in the Additional information.
After measuring the \(D_{{37}}\) of all four sets of samples containing a different fraction of DNA and TE it was possible to find the value of \(G_{\text{SSB}}^{\text {dir*}}\) which corresponds to the sample containing pure DNA by extrapolation of the values as a function of the mass fraction of DNA in the sample, \(w_{\text{DNA}}\). We have chosen to fit the data with a simple power function in a form: \(a(w_{\text{DNA}})^b\). The value of the radiation chemical yield of the pure DNA is then represented by parameter a. The measured \(G_{\text{SSB}}^{\text {dir*}}\) value and the fit are shown in Fig. 3. The resulting value of the SSB yield induced by the direct effect is then obtained by taking a product of parameter a, i.e., \(G_{\text{SSB}}^{\text {dir*}}(w_{\text{DNA}} = 1)\), with the mass fraction of the DNA in the liquid sample \(w_{\text{DNA}} \approx 10^{-5}\), which yields \(G_{\text{SSB}}^{\text{dir}} = {2.1 \times 10^{-13}}\; \text {mol} \, \text{J}^{-1}\).
Indirect effect
The radiation chemical yields of SSBs obtained by irradiating liquid samples with water window radiation were plotted together with the yields obtained by irradiating the same plasmid samples (pBR322) at the same concentration (10 \(\text {ng}\) µL−1) with an Al Kα radiation source (emission at 1.5 keV photon energy) taken from the work of Souici et al.44. The compared data are shown in Fig. 4. The numerical values from water window irradiation are summarized in Table 1. The function for fitting the water window data was again of the form \(a\sigma ^b\) with fitting parameters \(a = {1.77 \times 10^{-9}}\; \text {mol} \, \text{J}^{-1}\) and b = -0.3.
\(G_{\text{SSB}}\) as a function of the scavenging capacity for radiation of different qualities. Data for the gamma radiation are depicted together with the theoretical predictions by Mark’s spherical model. Data for the water window and Al Kα were fitted using a function in a form \(a\sigma ^b\). All data were obtained using the same pBR322 plasmid and concentration of 10 \(\text {ng}\) µL−1. The scavenging capacity corresponding to the cell mimetic condition (\(\sigma = {3 \times 10^8}\;\text {s}^{-1}\)) is marked with a dotted arrow.
With the plasmid DNA single-strand breaks yields determined for both water window radiation and gamma radiation, it was possible to determine the radiation chemical yield of the ·OH radical for water window and Al Kα radiation, \(G_{^{\cdot}\hbox {OH}}\), as a function of the scavenging capacity by scaling the radiation chemical yield of ·OH radical determined for gamma radiation, \(G_{^{\cdot}\hbox {OH}}^{ {\gamma }}\), defined in37 as:
where \(G_{\text{SSB}}\) is the SSB yield induced by the water window or the Al Kα radiation and \(G_{\text{SSB}}^{ {\gamma }}\) is the SSB yield induced by the gamma radiation given by the Mark’s model. All G values in Eq. (7) are functions of \(\sigma _{^{\cdot}\hbox {OH}}\). The practical range of scavenging capacity within which we performed the calculation was taken between \(10^3\) and \(10^9\) \(\text {s}^{-1}\). The data obtained by the above-mentioned method were fitted using an empirical relationship for general ·OH dependence on scavenging capacity introduced in45 with the assumption that the value of the initial yield \(G^{{0}}\), i.e., the yield at the time before spore diffusion (\(t < 10^{-12}\) s), is 5.5 molecules per 100 eV (\({5.7 \times 10^{-7}}\; \text {mol} \, \text{J}^{-1}\)) as was revealed by simulations using a wide range of electron energies46. The time dependence of the ·OH radical decay was reconstructed using fitted parameters in the inverse Laplace transform (Eq. IV in45). The scavenging and the temporal dependence of the radiation chemical yield of the ·OH radical determined using this approach are shown in Figs. 5 and 6. For comparison, we have added \(G_{^{\cdot}\hbox {OH}}\) in aerated water induced by gamma radiation determined previously in37 and dependencies corresponding to ·OH yield induced by high-LET radiation of 10 and 100 \(\text {eV} \, \text{nm}^{-1}\). Data for particles were obtained by means of diffusion kinetics simulations (for aerated water) described in47.
\(G_{^{\cdot}\hbox {OH}}\) for gamma, water window and Al Kα radiation determined using Eq. (7) in the scavenging capacity range of \({10^3 - 10^9}\;\text {s}^{-1}\). The dotted projection is the yield based on the fitted parameters and fixed value of \(G^{{0}}\) = 5.5 molecules per 100 eV. \(G_{^{\cdot}\hbox {OH}}^{ {\gamma }}\) was taken from reference37. The previously determined \(G_{^{\cdot}\hbox {OH}}\) value22 using terephthalic acid (TA) as ·OH scavenger is marked by the red cross. ·OH yields for 10 and 100 \(\text {eV} \, \text{nm}^{-1}\) radiation represented by dotted and dashed gray lines, respectively, added for comparison.
Time dependence of the ·OH radical decay reconstructed from the scavenging capacity dependence using inverse Laplace transform. For comparison, we have added dependencies corresponding to high-LET radiation with 10 and 100 \(\text {eV} \, \text{nm}^{-1}\) (dotted and dashed gray lines, respectively).
Discussion
Strand breaking efficiency
There are extensive data available on the strand cleavage yields for 60Co and \(^{137}\hbox {Cs}\) gamma radiation, however, very few experiments devoted to irradiation of aqueous samples using SXR radiation (Al Kα emission at 1.5 keV) were performed44,48,49. In the work of other research groups, photon energies within the water window spectral range were used for cell inactivation50,51,52,53, but only a single study was very close to the water window energies which used plasmid DNA as a probe to determine ·OH radical yield54.
In all samples irradiated in the water window, except for the one with the lowest scavenging capacity, only single-strand breaks were detected. The ratio of SSB to DSB at the lowest scavenging capacity (\({1.8\times 10^5}\;\text {s}^{-1}\)) was 73 which is slightly higher than found for gamma radiation (data not shown) at the same \(\sigma _{^{\cdot}\hbox {OH}}\) where (SSB/DSB = ~ 40). For gamma radiation, this ratio increases as a function of the scavenging capacity from ~ 40 at \({1.8\times 10^5}\;\text {s}^{-1}\) to ~ 200 at \({3.0\times 10^7}\;\text {s}^{-1}\). In other words, as the number of SSBs decreases while the scavenging capacity increases the DSBs decreases even further. This indicates that double-strand breaks are induced solely by a combination of single-strand breaks or a combination of single-strand break and an oxidized base on opposite strands as suggested by Milligan et al.55. The most plausible explanation may be the increasing contribution of the direct effect, which has been estimated to be ~ \(10^{-13}\; \text {mol} \, \text{J}^{-1}\) for SSBs as the scavenging capacity is increased and the indirect effect is suppressed due to the short diffusion distance of ·OH radical or by their low production due to effects attributed to UHDR conditions as discussed in the text below.
As the photon energy of the incident radiation decreases, the exponent of the power function describing the dependence of \(G_\text{SSB}\) becomes less negative, as shown in Fig. 5. This can be explained by a change in the radiation quality, i.e., by an increase in LET of the secondary and Auger electrons as they develop shorter tracks with nanometer-sized distances. The similarity between high LET and SXR radiation is evident from Fig. 6, where the radiation chemical yield of ·OH radicals induced by the Al Kα shows a very similar pattern at LET of 100 \(\text {eV} \, \text{nm}^{-1}\) in both the scavenging and time domain. Similar observation has been made by Monte Carlo simulations56.
Although radiation in the water window under cellular scavenging conditions (\({3\times 10^8}\;\text {s}^{-1}\)) has the lowest predicted SSBs yield of the sources shown, its absorbed dose, or rather the absorbed energy density, is comparable to the absorbed energy density from Al Kα radiation. The contribution of the indirect effect, measured as the ratio between \(G_{\text{SSB}}\) and \(G^{\text{dir}}_{\text{SSB}}\) in the cellular scavenging capacity range, is about ten times higher than the determined direct effect. These results suggest processes underlying the FLASH effect, i.e., suppression of the indirect radical effects and an increase in the contribution of the direct effect to the formation of radiation damage, here DNA strand breaks. This property makes the water window radiation source a promising therapeutical tool for treating the surface of cells or tissues, where surface-localized energy deposition is of interest.
·OH radical yield
It clearly follows from the experimental conditions, i.e., pulsed irradiation, that the reaction rate constant in the theoretical description of the water window radiation action cannot be based on the assumption of a steady state, as in the case of using a continuous source. High absorption coefficients together with a discontinuity due to the presence of the oxygen K-edge in the water window spectral range are responsible for the strong attenuation of the beam in the water environment, which results in a non-homogeneous spatial distribution of primary events in the sample. Moreover, the nanosecond pulse duration also contributes to temporal non-homogeneity, meaning, that energy-deposition events cannot be treated as time-independent.
Without considering the dose rate effect, the lifetime of the ·OH radical in the irradiated system strongly depends on the scavenger concentration. The working solution containing DNA at a concentration of 10 \(\text {ng}\) µ\(\text {L}^{-1}\) combined with \({10^{-4}}\;\text{M}\) Tris and \({10^{-5}}\;\text{M}\) EDTA restricts the ·OH radical lifetime to 10 µs (\(k_{^{\cdot}\hbox {OH}+\text {Tris}} = {1.5\times 10^{9}}\;\text {M}^{-1}\, \text{s}^{-1}\) and \(k_{^{\cdot}\hbox {OH}+\text {EDTA}} = {3.0\times 10^{9}}\;\text {M}^{-1}\, \text{s}^{-1}\)). The intraspur radical-radical reactions occur in times \({< 10^{-8}}\;\text{s}\), thus the DNA damage is expected to be produced by diffusion-controlled reactions only. After a 3-ns pulse, all the interspur reactions are completed and the ·OH radicals are assumed to be homogeneously distributed in the solution. See, for example, computer simulations46. Other theoretical studies15,57, focusing on simulations of track overlaps (high dose rate) predict increased \(\hbox {H}_{2}\hbox {O}_{2}\) yield with increasing dose rate (inter-track distance or particle fluence) versus conventional ones. However, this is not consistent with experiments performed under FLASH conditions, which report reduced radiation chemical yields of \(\hbox {H}_{2}\hbox {O}_{2}\) when compared to conventional dose rates12,13. It is therefore likely that (·OH + ·OH) reaction is not the leading channel that produces the hydrogen peroxide under FLASH and that other reaction need to be considered to explain the low radiation chemical yield of ·OH radical when using a combination of high LET and UHDR as in the case of the water window radiation. As pointed out in46 a significant fraction of the \(\hbox {H}_{2}\hbox {O}_{2}\) can be eliminated by the reaction with aqueous electron (\(\hbox {e}^{-}_{\hbox {aq}}\,+\, \hbox {H}_{2}\hbox {O}_{2}\)) which produces additional ·OH radical. The reaction of ·OH with aqueous electron further decreases its yield due to the slightly faster reaction rate compared to the mutual ·OH recombination so that in the case of SXR interaction, it could lead to a further decrease in ·OH yield due to the high number of low-energy electron produced. From the simulated spectrum of secondary electrons liberated by the water window radiation source in water (see appendix in22) can be estimated that about 56% of electrons are in the energy range of 200 - 450 eV, 40% is between 15-100 eV and the rest represents electrons with energy < 15 eV. As the water window radiation is absorbed in water within a depth of few micrometers the tracks of the secondary electrons are localized in relatively small volume increasing the probability of their spatial overlap. An increased overlap also develops between clusters of reactants as a result of the decrease in the separation of the energy-loss events induced by slowing-down electrons. The initial spur radius of \(\hbox {e}^{-}_{\hbox {aq}}\) reactants is about three times smaller than that of ·OH58, meaning that as the energy of the electron decreases the amount of reactions between ·OH radicals increases. This effect is further multiplied by the relatively short period of time (3 ns) during which the energy is deposited causing temporal track overlaps which promotes further (·OH + ·OH) reactions.
The calculated \(G_{^{\cdot}\hbox {OH}}^{\text{WW}}\) is based on the assumption that both \(k_{\text{exp}}\) and \(\epsilon _{\text{SSB}}\) are independent of the incident radiation quality. Previously determined \(G_{^{\cdot}\hbox {OH}} = {2.3\times 10^{-8}}\;\text {mol}\, \text{J}^{-1}\) at a scavenging capacity of \({4.4\times 10^{5}}\;\text {s}^{-1}\) (scavenger concentration of \({10^{-4}}\;\text{M}\) with second order rate constant \({4.4\times 10^{9}}\;\text {M}^{-1}\, \text{s}^{-1}\)) using terephthalic acid as ·OH radical scavenger22 can be compared to \(G_{^{\cdot}\hbox {OH}} = {1.0\times 10^{-8}}\; \text {mol}\, \text{J}^{-1}\) in the present study using plasmid DNA as a target molecule for ·OH radical attack. The difference might be due to small variations in the rate constant as well as the efficiency factor or the second order rate constant for the terephthalic acid scavenging reaction, which have a slightly higher value than those (\({3.9\times 10^{10}}\;\text {M}^{-1}\, \text{s}^{-1}\) in particular) referenced in59.
Conclusion
We have demonstrated the capabilities of a laser-plasma source emitting photons with energies in the water window spectral range to study DNA damage mediated via both direct and indirect effects. Strand breaks induced by water window radiation showed lower yields when compared to Al Kα and 60Co gamma radiation. Comparing the data achieved using water window and Al Kα radiation with gamma radiation, we have estimated the dependence of the radiation chemical yield of the ·OH radical on the scavenging capacity of the irradiated system and, using inverse Laplace transform, we were able to reconstruct its time dependence. This technique showed that the action of water window radiation in biological system is similar to the action of particle radiation with moderately high LET (> 100 eV nm−1). Moreover, the localized dose distribution in order of a few micrometers in depth and nanosecond pulse duration makes this source ideal for studying the ultra-high-dose-rates effects and to study mechanisms behind FLASH conditions, that still remain unresolved.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Hieda, K. DNA damage induced by vacuum and soft X-ray photons from synchrotron radiation. Int. J. Radiat. Biol. 66, 561–567. https://doi.org/10.1080/09553009414551631 (1994).
Hieda, K., Suzuki, K., Hirono, T., Suzuki, M. & Furusawa, Y. Single- and double-strand breaks in pBR322 DNA by vacuum-UV from 8.3 to 20.7eV. J. Radiat. Res. 35, 104–111. https://doi.org/10.1269/jrr.35.104 (1994).
Hieda, K., Hirono, T. & Azami, A. Single- and double-strand breaks in pBR322 plasmid DNA by monochromatic X-rays on and off the K-absorption peak of phosphorus. Int. J. Radiat. Biol. 70, 437–445. https://doi.org/10.1080/095530096144914 (1996).
Yokoya, A., Watanabe, R. & Hara, T. Single- and double-strand breaks in solid pBR322 DNA induced by ultrasoft X-rays at photon energies of 388, 435 and 573 eV. J. Radiat. Res. 40, 145–158. https://doi.org/10.1269/jrr.40.145 (1999).
Prise, M. et al. Critical energies for SSB and DSB induction in plasmid DNA by low-energy photons: Action spectra for strand-break induction in plasmid DNA irradiated in vacuum. Int. J. Radiat. Biol. 76, 881–890. https://doi.org/10.1080/09553000050050891 (2000).
Yokoya, A., Cunniffe, S. M. T., Watanabe, R., Kobayashi, K. & O’Neill, P. Induction of DNA strand breaks, base lesions and clustered damage sites in hydrated plasmid DNA films by ultrasoft X-rays around the phosphorus K-edge. Radiat. Res. 172, 296–305. https://doi.org/10.1667/RR1609.1 (2009).
Meesungnoen, J., Jay-Gerin, J., Filali-Mouhim, A. & Mankhetkorn, S. Low-energy electron penetration range in liquid Water. Radiat. Res. 158, 657–660. https://doi.org/10.1667/0033-7587(2002)158[0657:LEEPRI]2.0.CO;2 (2002).
Pimblott, S. M. & LaVerne, J. A. Production of low-energy electrons by ionizing radiation. Radiat. Phys. Chem. 76, 1244–1247. https://doi.org/10.1016/j.radphyschem.2007.02.012 (2007).
Favaudon, V. et al. Ultrahigh dose-rate FLASH irradiation increases the differential response between normal and tumor tissue in mice. Sci. Transl. Med. 6, https://doi.org/10.1126/scitranslmed.3008973 (2014).
Kusumoto, T., Kitamura, H., Hojo, S., Konishi, T. & Kodaira, S. Significant changes in yields of 7-hydroxy-coumarin-3-carboxylic acid produced under FLASH radiotherapy conditions. RSC Adv. 10, 38709–38714. https://doi.org/10.1039/D0RA07999E (2020).
Kusumoto, T. et al. Radiation Chemical yields of 7-hydroxy-coumarin-3-carboxylic acid for proton- and carbon-ion beams at ultra-high dose rates: Potential roles in FLASH effects. Radiat. Res. 198, https://doi.org/10.1667/RADE-21-00.230.1 (2022).
Blain, G. et al. Proton irradiations at ultra-high dose rate vs. conventional dose rate: Strong impact on hydrogen peroxide yield. Radiat. Res. 198, https://doi.org/10.1667/RADE-22-00021.1 (2022).
Montay-Gruel, P. et al. Long-term neurocognitive benefits of FLASH radiotherapy driven by reduced reactive oxygen species. Proc. Natl. Acad. Sci. 116, 10943–10951. https://doi.org/10.1073/pnas.1901777116 (2019).
Labarbe, R., Hotoiu, L., Barbier, J. & Favaudon, V. A physicochemical model of reaction kinetics supports peroxyl radical recombination as the main determinant of the FLASH effect. Radiother. Oncol. 153, 303–310. https://doi.org/10.1016/j.radonc.2020.06.001 (2020).
Alanazi, A., Meesungnoen, J. & Jay-Gerin, J.-P. A computer modeling study of water radiolysis at high dose rates. Relevance to FLASH radiotherapy. Radiat. Res. 195, https://doi.org/10.1667/RADE-20-00168.1 (2020).
Kusumoto, T. et al. Dose rate effects on hydrated electrons, hydrogen peroxide, and a OH radical molecular probe under clinical energy protons. Radiat. Res. 201, https://doi.org/10.1667/RADE-23-00244.1 (2024).
Wolter, H. Spiegelsysteme streifenden Einfalls als abbildende Optiken für Röntgenstrahlen. Annalen der Physik 445, 94–114. https://doi.org/10.1002/andp.19524450108 (1952).
Wachulak, P. et al. “Water window’’ compact, table-top laser plasma soft X-ray sources based on a gas puff target. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 268, 1692–1700. https://doi.org/10.1016/j.nimb.2010.02.002 (2010).
Davídková, M. et al. A high-power laser-driven source of sub-nanosecond soft X-ray pulses for single-shot radiobiology experiments. Radiat. Res. 168, 382–387. https://doi.org/10.1667/RR0676.1 (2007).
Zielbauer, B. et al. Strand breaks in DNA samples induced with LASERIX. In X-Ray Lasers 2008, Proceedings (eds. Lewis, C. L. S. & Riley, D.), vol. 130, 409–411, https://doi.org/10.1007/978-1-4020-9924-3_47 (Springer, 2009).
Ayele, M. et al. Development and characterization of a laser-plasma soft X-ray source for contact microscopy. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 411, 35–43. https://doi.org/10.1016/j.nimb.2017.03.082 (2017).
Vyšín, L. et al. Chemical dosimetry in the “Water Window’’: Ferric ions and hydroxyl radicals produced by intense soft X-rays. Radiat. Res. 193, 372. https://doi.org/10.1667/RR15520.1 (2020).
Hicks, M. & Gebicki, J. M. Rate constants for reaction of hydroxyl radicals with Tris, Tricine and Hepes buffers. FEBS Lett. 199, 92–94. https://doi.org/10.1016/0014-5793(86)81230-3 (1986).
Buxton, G. V., Greenstock, C. L., Helman, W. P. & Ross, A. B. Critical review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (·OH/·O - in aqueous solution). J. Phys. Chem. Ref. Data 17, 513–886. https://doi.org/10.1063/1.555805 (1988).
Jomova, K. et al. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: Chronic diseases and aging. Arch. Toxicol. 97, 2499–2574. https://doi.org/10.1007/s00204-023-03562-9 (2023).
Tullius, T. D. & Greenbaum, J. A. Mapping nucleic acid structure by hydroxyl radical cleavage. Curr. Opin. Chem. Biol. 9, 127–134. https://doi.org/10.1016/j.cbpa.2005.02.009 (2005).
Halliwell, B. & Gutteridge, J. M. C. Free Radicals in Biology and Medicine (Oxford University Press, 2015).
Cheng, F.-C., Jen, J.-F. & Tsai, T.-H. Hydroxyl radical in living systems and its separation methods. J. Chromatogr. B 781, 481–496. https://doi.org/10.1016/S1570-0232(02)00620-7 (2002).
Zhao, Z. Hydroxyl radical generations form the physiologically relevant Fenton-like reactions. Free Radic. Biol. Med. 208, 510–515. https://doi.org/10.1016/j.freeradbiomed.2023.09.013 (2023).
Fleming, A. M. & Burrows, C. J. On the irrelevancy of hydroxyl radical to DNA damage from oxidative stress and implications for epigenetics. Chemical Society Reviews 49, 6524–6528. https://doi.org/10.1039/D0CS00579G (2020).
Vyšín, L. et al. Dose-rate effects in breaking DNA strands by short pulses of extreme ultraviolet radiation. Radiat. Res. 189, 466–476. https://doi.org/10.1667/RR14825.1 (2018).
Henke, B., Gullikson, E. & Davis, J. X-Ray interactions: Photoabsorption, scattering, transmission, and reflection at E = 50–30,000 eV, Z = 1–92. Atomic Data Nucl. Data Tables 54, 181–342. https://doi.org/10.1006/adnd.1993.1013 (1993).
Adjei, D. et al. DNA strand breaks induced by soft X-ray pulses from a compact laser plasma source. Radiat. Phys. Chem. 120, 17–25. https://doi.org/10.1016/j.radphyschem.2015.11.021 (2016).
LaVerne, J. A. & Pimblott, S. M. Yields of hydroxyl radical and hydrated electron scavenging reactions in aqueous solutions of biological interest. Radiat. Res. 135, 16. https://doi.org/10.2307/3578391 (1993).
Milligan, J. R., Aguilera, J. A. & Ward, J. F. Variation of single-strand break yield with scavenger concentration for plasmid DNA irradiated in aqueous solution. Radiat. Res. 133, 151. https://doi.org/10.2307/3578350 (1993).
Klimczak, U., Ludwig, D., Mark, F., Rettberg, P. & Schulte-Frohlinde, D. Irradiation of plasmid and phage DNA in water-alcohol mixtures: Strand breaks and lethal damage as a function of scavenger concentration. Int. J. Radiat. Biol. 64, 497–510. https://doi.org/10.1080/09553009314551711 (1993).
LaVerne, J. A. & Pimblott, S. M. Diffusion-kinetic modelling of the cooperative effect of scavengers on the scavenged yield of the hydroxyl radical. J. Chem. Soc. Faraday Trans. 89, 3527. https://doi.org/10.1039/ft9938903527 (1993).
Mark, F., Becker, U., Herak, J. N. & Schulte-Frohlinde, D. Radiolysis of DNA in aqueous solution in the presence of a scavenger: A kinetic model based on a nonhomogeneous reaction of OH radicals with DNA molecules of spherical or cylindrical shape. Radiat. Environ. Biophys. 28, 81–99. https://doi.org/10.1007/BF01210293 (1989).
Espenson, J. H. Chemical Kinetics and Reaction Mechanisms 2nd edn. (McGraw-Hill, Inc., 1995).
Latulippe, D. R. & Zydney, A. L. Radius of gyration of plasmid DNA isoforms from static light scattering. Biotechnol. Bioeng. 107, 134–142. https://doi.org/10.1002/bit.22787 (2010).
Prazeres, D. M. F. Prediction of diffusion coefficients of plasmids. Biotechnol. Bioeng. 99, 1040–1044. https://doi.org/10.1002/bit.21626 (2008).
Debije, M. G., Strickler, M. D. & Bernhard, W. A. On the efficiency of hole and electron transfer from the hydration layer to DNA: An EPR study of crystalline DNA X-irradiated at 4 K. Radiat. Res. 154, 163–70. https://doi.org/10.1667/0033-7587(2000)154[0163:oteoha]2.0.co;2 (2000).
Becker, D., Adhikary, A. & Sevilla, M. D. The role of charge and spin migration in DNA radiation damage. In Charge Migration in DNA (ed. Chakraborty, T.), chap. 7, 1st edn, 139–175. https://doi.org/10.1007/978-3-540-72494-0_7 (Springer, 2007).
Souici, M. et al. DNA strand break dependence on Tris and arginine scavenger concentrations under ultra-soft X-ray irradiation: The contribution of secondary arginine radicals. Radiat. Environ. Biophys. 55, 215–228. https://doi.org/10.1007/s00411-016-0642-9 (2016).
LaVerne, J. A. OH radicals and oxidizing products in the gamma radiolysis of water. Radiat. Res. 153, 196–200. https://doi.org/10.1667/0033-7587(2000)153[0196:oraopi]2.0.co;2 (2000).
Pimblott, S. M. & LaVerne, J. A. Effect of electron energy on the radiation chemistry of liquid water. Radiat. Res. 150, 159. https://doi.org/10.2307/3579851 (1998).
Pimblott, S. M. & LaVerne, J. A. Diffusion-kinetic theories for LET effects on the radiolysis of Water. J. Phys. Chem. 98, 6136–6143. https://doi.org/10.1021/j100075a016 (1994).
Fulford, J., Nikjoo, H., Goodhead, D. T. & O’Neill, P. Yields of SSB and DSB induced in DNA by Al K ultrasoft X-rays and α-particles: Comparison of experimental and simulated yields. Int. J. Radiat. Biol. 77, 1053–1066. https://doi.org/10.1080/09553000110069308 (2001).
Fromm, M. & Boulanouar, O. Low energy electrons and ultra-soft X-rays irradiation of plasmid DNA. Tech. Innov. Radiat. Phys. Chem. 128, 44–53. https://doi.org/10.1016/j.radphyschem.2016.05.025 (2016).
Hervé du Penhoat, M.-A. et al. Lethal effect of carbon K-shell photoionizations in Chinese hamster V79 cell nuclei: Experimental method and theoretical analysis. Radiat. Res. 151, 649. https://doi.org/10.2307/3580203 (1999).
Fayard, B. et al. Cell inactivation and double-strand breaks: The role of core ionizations, as probed by ultrasoft X-rays. Radiat. Res. 157, 128–40. https://doi.org/10.1667/0033-7587(2002)157[0128:ciadsb]2.0.co;2 (2002).
Gobert, F. N. et al. Chromosome aberrations and cell inactivation induced in mammalian cells by ultrasoft X-rays: Correlation with the core ionizations in DNA. Int. J. Radiat. Biol. 80, 135–45. https://doi.org/10.1080/09553000310001654710 (2004).
Hervé du Penhoat, M.-A. et al. Double-strand break induction and repair in V79-4 hamster cells: The role of core ionisations, as probed by ultrasoft X-rays. Int. J. Radiat. Biol. 86, 205–219. https://doi.org/10.3109/09553000903419296 (2010).
Fulford, J., Bonner, P., Goodhead, D. T., Hill, M. A. & O’Neill,. Experimental determination of the dependence of OH radical yield on photon energy: A comparison with theoretical simulations. J. Phys. Chem. A 103, 11345–11349. https://doi.org/10.1021/jp993087n (1999).
Milligan, J., Aguilera, J. A., Paglinawan, R. A., Ward, J. F. & Limoli, C. L. DNA strand break yields after post-high LET irradiation incubation with endonuclease-III and evidence for hydroxyl radical clustering. Int. J. Radiat. Biol. 77, 155–164. https://doi.org/10.1080/09553000010013445 (2001).
Goodhead, D. T. & Nikjoo, H. Track structure analysis of ultrasoft X-rays compared to high- and low-LET radiations. Int. J. Radiat. Biol. 55, 513–529. https://doi.org/10.1080/09553008914550571 (1989).
Kreipl, M. S., Friedland, W. & Paretzke, H. G. Interaction of ion tracks in spatial and temporal proximity. Radiat. Environ. Biophys. 48, 349–359. https://doi.org/10.1007/s00411-009-0234-z (2009).
LaVerne, J. A. & Pimblott, S. M. Scavenger and time dependences of radicals and molecular products in the electron radiolysis of water: Examination of experiments and models. J. Phys. Chem. 95, 3196–3206. https://doi.org/10.1021/j100161a044 (1991).
Gonzalez, D. H., Kuang, X. M., Scott, J. A., Rocha, G. O. & Paulson, S. E. Terephthalate probe for hydroxyl radicals: Yield of 2-hydroxyterephthalic acid and transition metal interference. Anal. Lett. 51, 2488–2497. https://doi.org/10.1080/00032719.2018.1431246 (2018).
Acknowledgements
Authors would like to acknowledge the financial support provided by the PPLZ project of the Czech Academy of Sciences (reg. no.: L100102051). This work was also partially supported by H2020 Laserlab Europe V (PRISES, Contract No. 871124) and NAWA project (Contract no. PPI/PRO/2019/1/00001/U/00001). Marie Davídková acknowledge the financial support by long-term conceptual development project of the Nuclear Physics Institute of the Czech Academy of Sciences, RVO: 61389005.
Author information
Authors and Affiliations
Contributions
L.V.: Conceptualization and supervision of the experiment, processing and interpretation of the data. P.W., H.F. and A.B.: Access to, operation and support of the water window radiation source. Laboratory access and support for sample analysis on site. V.H.: Preparation and analysis of the samples irradiated with the water window source. M.D.: Access to the calibrated gamma radiation source, analysis of the gamma-irradiated samples. L.J.: Financial support. All authors contributed in writing and revision of the manuscript.
Corresponding author
Ethics declarations
Competing interests
Te authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Vyšín, L., Wachulak, P., Hájková, V. et al. Breaking the DNA by soft X-rays in the water window reveals the scavenging and temporal behaviour of ·OH radicals. Sci Rep 14, 28515 (2024). https://doi.org/10.1038/s41598-024-79328-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-79328-9