Abstract
Edible insects offer opportunities for food production, as they are an interesting source of many nutrients. In this study, the effect of pulsed electric field (PEF) and convective drying on the chemical composition with emphasizing the fat properties as well as physical, techno-functional, and thermal properties of yellow mealworm powders was investigated. The chemical composition of the yellow mealworm powders differed by PEF. When PEF was applied at 20 and 40 kJ/kg, the moisture, ash, and protein content were significantly lower, while the fat extraction yield significantly increased compared to the control sample. Furthermore, the fat extracted from these samples was characterized by a higher proportion of saturated and monounsaturated fatty acids as well as a higher thrombogenicity index, which is not beneficial from a nutritional point of view. After treatment with PEF at 5 kJ/kg, the powder was the lightest, redness and yellowness. Moreover, the highest hygroscopicity, water activity, and water and oil binding capacity for this powder were determined. The results revealed that yellow mealworm powders are a good source of macronutrients and exhibit beneficial techno-functional properties, nevertheless, the drawback is their high cohesiveness (1.27–1.44), which can be difficult to apply under industrial conditions.
Similar content being viewed by others
Introduction
Edible insects are gaining attention due to their high nutritional value and potential health benefits. They are rich in protein, unsaturated fatty acids, amino acids, vitamins, and minerals, making them a valuable source of food1,2,3,4. Studies have shown that edible insects have various pharmacological properties, including immune enhancement, antioxidant capacity, and regulation of blood lipid levels, which can contribute to human health5. In addition, insects are considered a sustainable and environmentally friendly food source that can solve food security problems4,6,7. While the safety aspects and regulations surrounding the consumption of edible insects are still under investigation8, their nutritional value, and potential health benefits make them a promising alternative food source.
Among four insect species (yellow mealworm, migratory locust, house cricket, and lesser mealworm) authorized as novel foods in the European Union9, Tenebrio molitor larvae (yellow mealworm) are considered one of the most promising species due to their nutritional value and mass production capacity1,10. Furthermore, it is one of the most farmed holometabolous insect species, such as in the food and feed industry11. Currently, the best-selling products on the European market are entire insects and powders obtained by grinding insects, mainly Acheta domesticus (house cricket) and Tenebrio molitor (yellow mealworm) 7,12. When used as a food ingredient, consumers accept insects more as powder or flour than consuming them whole10,13. Additionally, insect powders have a positive effect on the functional and sensory properties of foods and have a potential role in preventing diseases related to hyperglycemia and hyperlipidemia2. However, before ground insect powders and flours can be used in various food applications, information on their physicochemical quality (such as chemical composition and techno-functional properties) is essential.
To meet growing consumer interest, well-designed manufacturing processes are required to produce high-quality insect powders while maintaining food safety and quality. Drying is the most widely used technology to extend the shelf life of insects, whether consumed whole or as ingredients14. The drying of insects is of great importance throughout the insect processing chain and therefore must be carried out properly to reduce potential microbiological, chemical, and allergenic risks while maintaining nutritional properties and consumer acceptability10,15. Different drying techniques are reported for insect flours and powders, including freeze drying, sun drying, oven drying, and non-conventional drying methods16,17,18,19. Therefore, research is needed to improve processing on an industrial scale to process insects in the best possible way, both in nutritional value, physical and techno-functional properties, and from an economic point of view20,21.
Non-thermal technologies applied to edible insect-derived foods, such as pulsed electric field (PEF), high hydrostatic pressure (HHP), ultrasound (US), and cold atmospheric pressure and plasma (CAPP), could be a promising and powerful solution to positively influence protein profile, techno-functional properties, and fatty acid composition, thus creating new opportunities for the food industry15,22,23. The application of PEF as a pretreatment is useful for reducing the drying process’s energy consumption and improving the final product16,24. Previous research showed that use of PEF applied before convective and infrared-convective drying allows dried yellow mealworm larvae to be obtained with a high content of valuable compounds25. Furthermore, PEF treatment at 4.9 kJ/kg increased extraction yields of protein (> 18%) and fat (> 40%), while treatment at 24.53 kJ/kg increased the oil binding and emulsifying capacity and antioxidant activity of cricket flour26.
Therefore, this investigation aimed to assess the quality of yellow mealworm (Tenebrio molitor L.) powders processed by pulsed electric field and convective drying. For this purpose, properties such as chemical composition (moisture, protein, fat, and ash content), fat properties (fatty acid composition, fatty acid positional distribution), and other properties affect health aspects (atherogenicity index, thrombogenicity index, oxidative stability, acid value, peroxide value), thermal properties, as well as physical properties (water activity, hygroscopicity, color, particle size, bulk density, and flowability), and techno-functional properties (water holding capacity, oil holding capacity) were analyzed.
Materials and methods
Materials
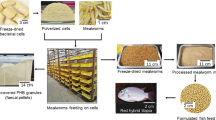
The alive yellow mealworm larvae (Tenebrio molitor L.) were purchased from a local Polish producer (Cirwins, Kamień Duży, Poland). The material was stored in a cold storage (4 ± 1 °C) for 24 h of fasting to discard their bowel content. The material came from the same batch, ensuring the same feeding (vegetables, cereal products) and environmental conditions, such as temperature and humidity. Before use, the residual feces and other impurities were removed by dipping the material in tap water for 30 s. After that, the material was gently dried with filter paper. The material was then immediately subjected to different processing conditions (Fig. 1). The yellow mealworm larvae were still alive after washing. The pulsed electric field (PEF) or hot air (90 °C) was applied to sacrificing insects24. Generally, sacrificing insects does not follow any special ethical considerations since it is considered that they do not suffer pain14. Moreover, insects as non-vertebrates are not included in Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes.
Pulsed electric field treatment
The utilization of pulsed electric field (PEF) treatment was performed using a PEF Pilot™ unit (Elea GmbH, Quakenbrück, Germany). The voltage generated by the system was 30 kV. The gap between the electrodes was 280 mm. Monopolar, exponential decay pulses were generated with a frequency of 2 Hz and a pulse width of 7 µs. The larvae in an amount of approx. 350 g were placed inside the treatment chamber, and tap water (20 ± 1 °C, conductivity of 220 µS/cm) was added until the larvae were completely immersed. The samples were treated with an electric field intensity of 1.07 kV/cm, and the specific energy consumption (delivered by applying the number of pulses) was 5, 20, and 40 kJ/kg27. The PEF treatment was performed in duplicate.
Convective drying
The material was dried using the prototype laboratory dryer (Warsaw, Poland) at a temperature of 90 °C and parallel to the material layer air flow of 2 m/s. The sieve load was 2.71 kg/m2. The drying process was performed until the material achieved a constant weight in two repetitions for each sample. After drying, the material was grounded using an analytical grinder IKA Tube Mill (IKA-Werke GmbH & Co., Staufen, Germany) for 20 s with a speed of 8,500 rpm, and then stored in air and light barrier PET/AL/PE bags (Pakmar, Warsaw, Poland).
Chemical composition
The protein content was assessed by the Kjeldahl method using a KjelFlex K-360 nitrogen analyzer (Büchi, Flawil, Switzerland) coupled with a TitroLine 5000 automatic titrator (SIAnalytics, Weilheim, Germany). Nitrogen concentration was converted to protein content by multiplying by a conversion factor of 4.7628. The fat extraction yield was assessed by the Soxhlet method using a Behrotest ET2 Control Unit (Behr Labor-Technik GmbH, Düsseldorf, Germany). The petroleum ether was used as a solvent, and an extraction process lasted 6 h. The ash and moisture content were determined by the gravimetric method29. The analysis was conducted in triplicate for each sample.
Fatty acid composition
The oil was extracted according to the Folch method described by Kozłowska et al.30. The fatty acid composition was determined as fatty acid methyl esters (FAMEs), according to the ISO method31. The FAMEs thus prepared were injected onto the BPX-70 capillary column (length 60 m, internal diameter 0.25 mm, film thickness 0.25 μm) of the YL6100 GC gas chromatograph (Young Lin Bldg., Anyang, Hogye-dong, Korea) coupled with a flame ionization detector (FID). The separation conditions for the FAMEs were as follows: 70 °C for 5 min, 15 °C/min to 160 °C, 1.1 °C/min to 200 °C, 200 °C for 12 min, 30 °C/min to 225 °C and 225 °C for 1 min. The injector and detector temperature were set at 225 °C and 250 °C, respectively32. The detected FAMEs were identified by comparing their retention times with the standard FAMEs mixture (Supelco 37 Component FAME Mix, Sigma-Aldrich GmbH, Schnelldorf, Germany). Calculations of each fatty acid area under the obtained chromatogram were applied to obtain the fatty acid composition. The analysis was conducted in duplicate for each sample.
Health indices
The atherogenicity index (AI) is related to the relationship between the sum of saturated fatty acids (SFA) and the sum of unsaturated fatty acids (UFA) present in lipid fraction of food33. The AI was calculated with the following formula:
The thrombogenicity index (TI) is related to the relationship between pro-thrombogenic fatty acids (SFA) and anti-thrombogenic fatty acids (MUFA, n-3 PUFA, and n-6 PUFA) present in lipid fraction of food33. The TI was calculated with the following formula:
The hypocholesterolemic/hypercholesterolemic (HH) ratio is related to the relationship between hypocholesterolemic fatty acids (the sum of oleic acid (C18:1 n-9) and polyunsaturated fatty acids PUFA) and hypercholesterolemic fatty acids (saturated fatty acids from C12:0 to C16:0) present in the lipid fraction of food33. The HH was calculated with the following formula:
Fatty acid positional distribution
The positional distribution in triacylglycerols (TAG) was assessed by the partial hydrolysis method with a regiospecific pancreatic lipase, which hydrolyzes the ester bonds in sn-1,3 positions of TAG32,34. The products of hydrolysis were isolated by preparative thin-layer chromatography (TLC) technique using silica gel plates and a chromatographic chamber. The sn-2 monoacylglycerols were scratched off from plates with silica gel and extracted with diethyl ether. The composition of fatty acids in the sn-2 position of monoacylglycerols was performed by the GC method described above.
Oxidative stability
The oxidative stability was assessed by the Pressure Differential Scanning Calorimetry (PDSC) method using a DSC Q20 TA Instruments (Newcastle, WA, USA). An aluminum pan with fat in an amount of approx. 3 mg and an empty pan (as a reference) were placed in a high-pressure cell35. The oxidative stability was performed at 140 °C and under an oxygen atmosphere with an initial pressure of 1400 kPa. Measurement of each sample was made in duplicate.
Acid value
The acid value (AV) was measured by the titration method. A solution of 0.1 mol/L ethanolic potassium hydroxide (KOH) and a TitraLab AT100 titrator (HACH LANGE, Wrocław, Poland) were used. The results were expressed as mg KOH per 1 g of fat35. Measurement of each sample was made in triplicate.
Peroxide value
The peroxide value (PV) was measured by the titration method. A solution of 0.001 mol/L sodium thiosulfate (Na2S2O3) and a TitraLab AT100 titrator (HACH LANGE, Wrocław, Poland) wasused. The results were expressed as meq O2 (milliequivalents of oxygen) per 1 kg of fat35. Measurement of each sample was made in triplicate.
Water activity
The water activity was measured at a temperature of 25 ± 1 °C, using a HygroLab C1 hygrometer (Rotronic, Bassersdorf, Switzerland). The measurement of each powder was made in four repetitions.
Hygroscopicity
The powder in an amount of approx. 0.3 g was weighed on an analytical scale into an aluminum vessel and then placed in the desiccator with distilled water, giving a water activity of 1.0. The mass of the samples was remeasured after 1, 2, 3, 6, 9, 12, 24, 48, and 72 h29. Measurement of each powder was made in three repetitions. The hygroscopicity was determined as the mass of the powder after each adsorption time (g) in relation to the initial mass of the powder (g). The hygroscopicity (H) was calculated as follows:
where mτ – is the mass of the sample after adsorption time (g); and m0 – is the initial mass of the powder (g).
Particle size
The particle size of yellow mealworm powders was determined using a CILAS 1190 laser particle size analyzer (CILAS, Orleans, France). Measurements were carried out in a recirculation chamber with isopropanol as the dispersing agent. The mean particle size was determined as the volume mean diameter d4.3.
Bulk density and flowability
The loose bulk density ρL was calculated as a relationship between the volume of powder and its given mass, while the tapped bulk density ρT100 was measured after tapping 100 times using STAV 2003 automatic volumeter (Engelsmann AG, Ludwigshafen, Germany). The flowability of powder was determined as the tapped bulk density in relation to the loose bulk density and was expressed as the Hausner ratio (HR100). The measurement of each powder was made in at least three repetitions.
Color
The color parameters were measured using a Konica Minolta CR-5 chromameter (Konica Minolta, Japan) working in the reflectance mode in a CIE L*a*b* system (light source: D65, standard observer: 2°, diameter: 30 mm). The measurement of each powder was made in ten repetitions. Based on L*a*b* color parameters, the total color difference (∆E) was calculated:
where L* is the lightness (0 – black, 100 – white), a* is the chromaticity coordinate (red – positive values, green – negative values), b* is the chromaticity coordinate (yellow – positive values, blue – negative values), ΔL*, Δa*, Δb* are the differences between the values of L*, a* and b* measured for untreated and PEF-treated yellow mealworm powders.
Water holding capacity (WHC)
The powder in an amount of approx. 0.5 g was weighed into a pre-weighed 15 mL centrifuge tubes and mixed with 2.5 mL of water at least in three repetitions. The mixtures were vortexed for 60 s and then centrifuged at 3000× g for 20 min. After the supernatant was removed, the tube was weighed. The WHC was calculated as follows:
where m is the mass of powder after water binding (g), m0 is the initial mass of powder (g).
Oil holding capacity (OHC)
The powder in an amount of approximately 0.5 g was weighed into a pre-weighed 15 mL centrifuge tubes and mixed with 2.5 mL of commercial rapeseed oil at least in three repetitions. The mixtures were vortexed for 120 s, and then centrifuged at 3000× g for 20 min. After the supernatant was removed, the tube was weighed. The OHC was calculated as follows:
where m is the mass of powder after oil holding (g), m0 is the initial mass of powder (g).
Thermal properties
The powder in an amount of approx. 6 mg was weighted in 70 µL alumina crucibles and heated from 30 to 600 °C at a rate of 5 °C/min in a nitrogen atmosphere with a flow at 50 mL/min using a TGA/DSC 3 + thermogravimeter (Mettler Toledo, Greifensee, Switzerland)36. Thermograms TGA/DTG were evaluated using STAR 16.10 software (Mettler Evaluation). The measurement of each powder was made in two repetitions.
Statistical analysis
The one-way analysis of variance ANOVA and the post hoc Tukey’s HSD test were applied to assess significant differences between investigated samples using STATISTICA 13.1 (TIBCO Software, Palo Alto, CA, USA). The significance level was set at α = 0.05.
Results and discussion
Chemical composition
The nutritional properties of insects are important in terms of their utilization as an alternative source of food. The most important macronutrient present in insects is protein. Compared to the untreated sample, significantly lower protein content was observed for materials preceded with PEF at 20 and 40 kJ/kg before drying (Table 1). This outcome may be related to the electroporation phenomenon, which resulted in a cleavage of the peptide bonds, some changes in the protein structure, and a reduction in protein extractability due to unfolding of the protein structures and their denaturation23,37. In turn, a lower dose of PEF (5 kJ/kg) may lead to the destruction of the material structure, better solvent availability, and higher protein extractability. A similar effect has been reported for house cricket (Acheta domesticus) flour treated with PEF at 4.9 kJ/kg before the extraction process26. On the other hand, the possible release of chitin nitrogen from the insect exoskeleton and nitrogen-containing compounds from the cellular structure phospholipids may be the reason for higher protein content in the case of PEF_5kJ/kg powder. However, this needs further detailed research.
The fat extraction yield increased for samples subjected to PEF at 20 and 40 kJ/kg before drying and was significantly higher compared to untreated sample. These results are similar to those reported for olives38,39, sunflower seeds40,41, sesame seeds42, pecan nuts43, or insect biomass24,44. Changes in the material structure could provide better solvent penetration and therefore higher fat extraction yield. The extraction yield is also affected by the extraction time, temperature, particle size, and lipid source: solvent ratio45. In turn, for the sample marked as PEF_5kJ/kg, the lowest fat extraction yield was determined. This observation may be related to the interactions with other compounds such as proteins or carbohydrates. The increase in the hydrophobicity of the surface of protein molecules as a result of denaturation could result in a greater ability to bind to fat molecules13,17 and thus a lower extractability from powder.
Extracted fat properties
Fatty acid composition and health indices
Fats are a mixture of substances dominated by triacylglycerols (TAG). The key components of triacylglycerols are fatty acids. Their composition and structural distribution in TAG are related to the properties of fat35. Different food products are composed of different types of fatty acids, which can be influenced by processing, storage, and cooking techniques46. Fat extracted from yellow mealworm powders was dominated by unsaturated fatty acids (Table 2). The percentage of fatty acids from particular groups of saturated fatty acids (SFA), monounsaturated fatty acids (MUFA), and polyunsaturated fatty acids (PUFA) was in proportions of 20.22–21.81%, 41.17–42.39%, and 34.48–47.21%, respectively. Among the tested samples, the fat extracted from the powder marked PEF_5kJ/kg was characterized by the lowest percentage of SFA and the highest percentage of PUFA. The main fatty acids determined in insect fats were palmitic acid (16:0), linoleic acid (18:2 n-6c), and oleic acid (18:1 n-9c). Furthermore, a medium-chain fatty acid was found, such as lauric acid (C12:0). After PEF treatment at 20 and 40 kJ/kg, the percentage of linoleic acid decreased, while the percentage of lauric acid, palmitic acid, oleic acid, and α-linolenic acid increased.
The results obtained in the current study are consistent with those presented by other researchers in the literature. For instance, similar proportions of lauric acid (C12:0)47, stearic acid (C18:0)21,48, and α-linolenic acid (C18:3 n-3)21,47,48 in yellow mealworm fat were found. In turn, a slightly lower percentage of myristic acid (C14:0) was provided at a level of 2.01–2.26% and oleic acid (C18:1 n-9c) at a level of 32.38–36.75%, and a slightly higher percentage of linoleic acid (C18:2 n-6c) at the level of 36.44–40.28% was provided21,47,48. Generally, a high content of unsaturated fatty acids, especially n-3 PUFA, is desirable in the human diet. Their role is very well known, as they have a positive effect in preventing the development of diseases related primarily to the cardiovascular system46,49,50. Nevertheless, different fatty acids can play an important role. For instance, lauric acid (C12:0) is a medium-chain fatty acid that is more easily used for energy than long-chain saturated or unsaturated fatty acids. Furthermore, lauric acid can affect intestinal bacteria due to its potential antimicrobial properties51. In turn, pentadecanoic acid (C15:0) is an essential long-chain saturated fatty acid that has broad activity to prevent the development of cardiovascular, immune, metabolic, and liver diseases52.
When talking about fat quality, it is also important to mention its nutritional value, which informs us about its impact on human health. For this purpose, based on the fatty acid composition, three health indices are usually calculated such as the atherogenicity index (AI), the thrombogenicity index (TI), and the hypocholesterolemic: hypercholesterolemic ratio (HH). Fats with favorable nutritional value are those characterized by low values of the AI and TI indices and high values of the HH. The AI and TI values obtained in the current study were relatively low (Table 2) and met the recommended fat requirements in the human diet (AI below 1.0 and TI below 0.5). A similar AI value (0.40) was observed for avocado fruit oil, while a similar TI value (0.45) was found for argan and borage oils53. The HH ratio obtained for insect fats was between 4.11 and 4.69 (Table 2). The values obtained were noticeably lower than those obtained, for example, camelina oil (11.7–14.7)54, hemp oil (14.88)53, coriander seed oil (28.34)55 or strawberry seed oil (18.81)32. Considering the results obtained in our study, it can be said that of the fats tested, those extracted from the samples PEF_0kJ/kg and PEF_5kJ/kg showed the most favorable nutritional properties. However, low values of the HH ratio suggest that these fats could potentially cause cardiovascular disease if consumed in greater amounts.
Fatty acid positional distribution
The nutritional properties of fat are influenced not only by the composition of fatty acids but also by the distribution of the fatty acids in the different TAG positions56,57. The study conducted on yellow mealworm fats showed that the sn-1,3 TAG positions were occupied by unsaturated fatty acids, e.g., oleic and linoleic acids, with a distribution of linoleic acid close to statistical (Table 3). The acid whose proportion in the sn-1,3 TAG position was also significant was palmitic acid. Its percentage in the external positions of the TAG, in contrast to that of unsaturated acids, increased statistically after PEF treatment. Oleic acid and linoleic acid also occupied the inner (sn-2) TAG position of these fats. Moreover, their percentage in the sn-2 position was higher than in the sn-1,3 TAG positions. Based on the distribution of fatty acids, it can be concluded that unsaturated fatty acids, such as oleic and linoleic acid prefer the central position of the TAG, while saturated fatty acids, such as palmitic, stearic, and myristic acids are located mainly in the external positions of the TAG. This structure of TAG is not the most favorable in terms of their digestibility because the saturated fatty acids released by hydrolysis from the sn-1,3 TAG positions form insoluble calcium salts, which are removed from the body56,58. It was observed that the application of PEF influenced the distribution of the fatty acids in TAG positions. This may be related to the stimulation of endogenous lipases, which promoted the process of transesterification and moving these fatty acids within and between TAG molecules59.
Acid value, peroxide value and oxidative stability
The quality and freshness of the fat is very important. Various methods are used to assess it, among them titration methods such as acid value (AV) or peroxide value (PV). The acid value indicates the degree of hydrolysis and the amount of free fatty acids formed. The peroxide number indicates the degree of oxidation and the amount of primary oxidation products present54,60. Higher acid values were determined in the fat extracted from insects treated with PEF (Table 4). This increase can be due to the promotion of endogenous lipase activity59. This resulted in more intensive hydrolysis of the ester bonds of TAG molecules and the formation of more free fatty acids35. The highest AV was determined in the fat from the sample marked as PEF_5kJ/kg, which could be due to the significantly higher water content, which promotes fat hydrolysis54. The peroxide values were dependent on PEF energy input (Table 4). Lower PEF energy input (5 and 20 kJ/kg) resulted in lower PVs compared to the control material. On the contrary, the PEF application at the highest energy input contributed to the highest PV for the extracted fat, which could be linked to interactions between free radicals and the antioxidant compounds present61. Considering the results, no relationship was observed between the fat values tested, confirming the complexity of the oxidation process and the need to control it with different assays.
To date, there are no guidelines on the quality of fat extracted from insects. EFSA’s Scientific Opinion for blanched and thermal dried yellow mealworm larvae indicates acid value and peroxide value in the range of 0.5–4.8 mg KOH/g and 1.0–16.3 meq O2/kg, respectively62. The general quality requirements for fats and oils can be found in the Codex Alimentarius. Based on this, the tested fats exceeded the acceptable acid value (4 mg KOH/g), while meeting the requirements for the peroxide value (15 meq O2/kg).
The oxidation stability of fats is one of the key characteristics that indicate the degree of susceptibility of fats to oxidative processes and contribute to their shelf life. Oxidative stability is mainly related to the composition of fatty acids and their distribution in the TAG structure53,59. The highest oxidative stability (10.13 min) was obtained for the fat extracted from the sample treated with PEF at 5 kJ/kg (Table 5). For the other samples, oxidative stability was generally similar. The results are interesting because, in general, fats with a higher content of unsaturated fatty acids and acid value are characterized by lower oxidative stability. Fat extracted from sample PEF_5kJ/kg consisted of the largest proportion of fatty acids from the PUFA group and was presented with the highest acid value, which should result in the lowest oxidative stability. A higher acid value (higher content of free fatty acids) results in lower oxidative stability, which was demonstrated, e.g., for quinoa seed oil63. Another explanation could be the oleic acid (C18:1 n-9) content, which contributes to the greater oxidative stability of fats32. However, the fat in question had the lowest percentage of it, which also does not support this theory.
Water activity, particle size, bulk density and flowability
The water activity gives information about the present and available water in a food product to facilitate the appearance of chemical and enzymatic reactions or the growth of microorganisms6. The insect powders exhibited water activity below 0.214 (Table 5). The results obtained were presented with values lower than 0.6, which indicates that insect powders are not susceptible to the growth of microorganisms6. The highest water activity was determined in powder marked as PEF_5kJ/kg. Greater interactions with the water molecules could be explained due to the breaking of the hydrogen and hydrophobic bonds and a decrease in protein molecular weight related to the partial unfolding of protein molecules23,64. The water activity of samples was similar to the values obtained by other research for Tenebrio molitor larvae powders (0.08–0.17)10.
Table 5 Shows yellow mealworm powders’ particle size, bulk density, and flowability. The PEF-treated samples had a smaller particle size than the control sample. The PEF treatment loosens the material’s structure during drying, resulting in higher grinding efficiency and smaller particle size. Ando et al.19 determined the particle size of the powder obtained by two different drying methods (hot air drying and freeze drying) in two species of crickets and silkworm larvae. The results showed significant shrinkage and hardening of the material during hot air drying, which could result in lower grinding efficiency and larger particle size compared to samples obtained by freeze drying. The particle diameter of the yellow mealworm powders in this study was smaller compared to the powders of crickets (261.4–266.4 μm) and silkworm larvae (209.3 μm)19 and similar to the defatted yellow mealworm powders (86.95–161.84 μm)65 obtained by hot air drying. A lower particle diameter is advantageous from the point of view of product acceptability and affects perception in the mouth. Particles smaller than 100 μm give a creamy impression and a smooth texture that is more pleasant/acceptable66. On the other hand, fine powders (< 100 μm) become very cohesive, and small particles no longer flow individually, but as aggregates67.
Bulk density is the mass of a solid that occupies a unit volume of a bed, including the volume of all interparticle voids. Bulk density is determined in the bulk materials trade when evaluating packaging requirements, material handling, and processing operations. The bulk density of the PEF-treated samples was lower than that of the control sample (Table 5). The lowest bulk density values were recorded for the powder PEF_5kJ/kg, which may be influenced by interparticle forces (such as van der Waals, capillary, electrostatic, mechanical interlocking, and other forces). These forces keep the particles close together and can cause the particles to agglomerate, thus changing the properties of the powder (including reducing bulk density). Compared to black soldier fly larvae flours6,68, the bulk density of the yellow mealworm powders obtained was lower. On the other hand, house cricket powders16 had the lowest bulk density. Differences in bulk density between powders may result from differences in insect species and production methods. For example, the grinding techniques used differentiate the powder in terms of particle size, which in turn affects its bulk density. For defatted yellow mealworm powders, the jet mill machine produced smaller particle sizes, and the cutter mill produced larger particles than other milling machines65. Additionally, food materials with low moisture content have high grinding efficiency because these materials are more brittle. Furthermore, an increase in moisture content causes an increase in plasticity, which determines ductile behavior during grinding, resulting in increased grinding difficulty69.
The Hausner ratio is defined as the ratio of the tapped bulk density of a powder to its loose bulk density. This ratio can be used to determine the flowability of the powder70. The use of PEF treatment affects the flowability of yellow mealworm powders (Table 5). The powders PEF_20kJ/kg and PEF_40kJ/kg showed poor flowability (cohesive) compared to the powder without PEF treatment and PEF_5kJ/kg (passable flowability). Fine particles and the presence of fat on the surface of the powder reduces its flowability71, which seems to confirm this statement due to the increased fat extraction yield in the case of samples subjected to PEF at 20 and 40 kJ/kg. Son et al.65 showed that yellow mealworm powders had better flowability and other physical properties (such as emulsifying and foaming activities), when the fat was removed. The flowability of the powder is also affected by the moisture content. Moisture content is an important aspect in food powders, as it is associated with increased cohesion, which is partly due to liquid bridges between particles69.
Hygroscopicity
The hygroscopic properties of dried materials are related to the ability to adsorb water vapor in a humid environment and are very important in terms of stability during the storage of these materials72. The hygroscopic kinetics of the powders obtained are presented in Fig. 2. Based on the hygroscopic kinetics obtained, the highest ability for water vapor adsorption was observed for all tested samples within the first 24 h of measurement. The powder PEF_5kJ/kg was characterized by the highest hygroscopicity, which may be related to the lowest fat content and the greater ability to adsorb water vapor73. It could be due to a lower surface solubility of protein molecules after denaturation and thus higher water adsorption ability16,17. The powders PEF_20kJ/kg and PEF_40kJ/kg exhibited a higher hygroscopicity compared to powder without PEF treatment, however, these changes were not statistically significant. The PEF treatment with higher energy may have resulted in the unfolding of the secondary and tertiary structures of protein, its aggregation, and increased ability to adsorb water22,23. Our assumptions require further research, such as Fourier transform infrared spectroscopy (FTIR) of secondary and tertiary structures of protein as well as determination of its functional properties, including solubility and hydrophobicity.
Color
Color is one of the main crucial parameters that determine the consumer’s choice of products17,74. The macroscopic photographs of yellow mealworm powders are shown in Fig. 3, while the color parameters are in Table 6. The application of PEF did not show a clear trend in the changes in the color parameters. There was a significant increase in the value of the color parameters for the sample treated with PEF at 5 kJ/kg, while for the sample marked as PEF_20kJ/kg, these values were significantly lower compared to the untreated sample. Reflecting these differences, the ΔE was calculated to show the color differences compared to the sample without PEF treatment. The values of this parameter decreased with increasing PEF energy. The higher ΔE was obtained for the sample PEF_5kJ/kg. For samples PEF_5kJ/kg and PEF_20kJ/kg, the values of ΔE were greater than 2, which means that the changes in color can be noticed by an inexperienced observer3. The obtained results might be related to the water content as well as water activity, which could promote non-enzymatic browning processes and lipid oxidation75,76, enzymatically formation of dark pigments75 and phospholipid deprivation48. Furthermore, the high water content in the sample PEF_5kJ/kg may promote fat hydrolysis and the formation of free fatty acids, which resulted in a higher velocity of the fat oxidation process. In turn, the sample treated with the highest PEF energy (PEF_40kJ/kg) probably resulted in the inactivation of phenol oxidase, which is responsible for the browning and darkening77.
Techno-functional properties
The techno-functional properties such as water and oil holding capacity are regarded as pivotal characteristics in food applications, particularly in the context of texture and structure formation13. Table 5 illustrates the water holding capacity (WHC) and oil holding capacity (OHC) of yellow mealworm powders. Treatment with PEF increased WHC and OHC, but only in the case of the sample treated with an energy input of 5 kJ/kg (2.23 g/g). Further increases in energy input from 20 to 40 kJ/kg did not cause any changes in the values compared to the control sample. In turn, the research conducted by Psarianos et al.26 showed no effect of PEF treatment on the WHC and OHC of cricket flour. PEF-induced cell permeability enhances transport phenomena between the intracellular and extracellular environment. Therefore, once cell membranes are disrupted, water can only enter the cell more easily but cannot be held by the sample26. However, it seems that in the case of the PEF_5kJ/kg sample, a lower particle size (Table 5) resulted in higher WHC and OBC compared to the other samples. These findings are confirmed by other research, which showed that WHC values for very fine grinding Protaetia brevitarsis larvae powder were much higher than with traditional grinding samples78. The WHC and OHC values of the yellow mealworm powders were similar to those obtained by other researchers79. However, in other studies, the WHC value was lower (1.29 g/g) and the OHC value was higher (1.71 g/g) for Tenebrio molitor flour18, which is likely due to different production methods.
Thermal properties
Thermogravimetry (TGA) is a straightforward analytical technique for assessing the thermal stability of materials, including powders. It offers insights into the alterations in sample behavior that may arise from industrial processing44,80. The TGA and DTG curves in Fig. 4 shows the thermal stability of the obtained powders in the temperature range of 30–600 °C. The first weight loss, amounting to 2%, is observed from 30 to 120 °C and is related to the loss of free water and loosely bound water81. In the second stage, up to 240 °C, organic matter decomposes, mainly the intermolecular hydrogen bonds in proteins are broken44,81. In this event, the highest instability was observed in the sample PEF_5kJ/kg with the highest protein content, for which a 13% mass decrease and a maximum of the endothermic peak were observed at 210 °C. However, in the range of 240–520 °C, this sample showed the lowest mass loss of 60.5% as compared to the control sample (68.0%), which may indicate greater thermal stability of the material in a given range. The endothermic peak had two maxima: 316 and 356 °C related to polymer decomposition36.
Conclusions
The research performed determines how PEF and convective drying affect the nutritional value including fat properties, as well as physical, technological, and thermal properties of yellow mealworm powders. The application of PEF affected the studied properties: lower PEF energy (5 kJ/kg) significantly increased protein content and decreased fat extraction yield, while higher energy (20 and 40 kJ/kg) significantly decreased protein content and increased fat content compared to untreated material. As a result of PEF treatment, the proportion of saturated fatty acids (SFA) and monounsaturated fatty acids (MUFA) increased (PEF_5kJ/kg) or decreased (PEF_20kJ/kg and PEF_40kJ/kg). The increase of SFA and MUFA proportion is related to the oxidation of polyunsaturated fatty acids (PUFA) due to the formation of free radicals. An increase in hygroscopic properties and changes in the color of powders were also observed. The differences in color compared to powder without PEF treatment decreased with the increasing PEF energy. Furthermore, the application of PEF resulted in a significant decrease in loose bulk density and an increase in cohesiveness (Hausner ratio). These results suggest that for the industrial production of yellow mealworm powders, some flow-enhancing action will be required. The water and oil holding capacity initially increased (PEF_5kJ/kg) and then decreased to values comparable to powder without PEF treatment.
The results revealed that all yellow mealworm powders are a good source of nutritional compounds. Although the use of lower PEF energy (5 kJ/kg) improved the techno-functional properties of the powder, nevertheless, the problem is their high cohesiveness and ability to water vapor adsorption, which is not beneficial and can be problematic in terms of industrial application. Therefore, the application of PEF before convective drying is not advisable in this case. However, the results presented were obtained on a laboratory scale, so further research should focus on drying insects on an industrial scale, and then compare if the effect obtained is similar.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ordoñez-Araque, R., Quishpillo-Miranda, N. & Ramos-Guerrero, L. Edible insects for humans and animals: nutritional composition and an option for Mitigating Environmental damage. Insects. 13, 944 (2022).
Zhou, Y. et al. Nutritional composition, Health benefits, and application value of Edible insects: a review. Foods. 11, 3961 (2022).
Kowalczewski, P. Ł. et al. Nutritional Value and Biological Activity of Gluten-Free Bread enriched with cricket powder. Molecules. 26, 1184 (2021).
van Huis, A. et al. Edible insects. Future prospects for food and feed security. FAO Forestry Paper. 171, 1–186 (2013).
Tanga, C. M. & Ekesi, S. Dietary and therapeutic benefits of Edible insects: A Global Perspective. Annu. Rev. Entomol. 69, 303–331 (2024).
Vanqa, N., Mshayisa, V. V., Basitere, M. & Proximate Physicochemical, Techno-Functional and Antioxidant Properties of Three Edible Insect (Gonimbrasia belina, Hermetia illucens and Macrotermes subhylanus) Flours. Foods 11, 976 (2022).
Szulc, K. & Edible Insects A study of the availability of insect-based food in Poland. Sustainability. 15, 14964 (2023).
Gnana, M. et al. Edible insects as emerging food products–processing and product development perspective. J. Food Sci. Technol. 60, 2105–2120 (2023).
Kowalski, S. et al. Chemical Composition, Antioxidant Properties and Sensory Aspects of Sponge Cakes Supplemented with Edible Insect Flours. Antioxidants 12, 1912 (2023).
Yan, X. et al. Quality of Tenebrio molitor powders: effects of four processes on Microbiological Quality and Physicochemical factors. Foods. 12, 572 (2023).
Barrett, M., Godfrey, R. K., Schnell, A. & Fischer, B. Farmed yellow mealworm (Tenebrio molitor; Coleoptera: Tenebrionidae) welfare: species-specific recommendations for a global industry. J. Insects Food Feed. 1–46. https://doi.org/10.1163/23524588-20230104 (2023).
Pippinato, L., Gasco, L., Di Vita, G. & Mancuso, T. Current scenario in the European edible-insect industry: a preliminary study. J. Insects Food Feed. 6, 371–381 (2020).
Zielińska, E. Evaluating the functional characteristics of certain insect flours (Non-Defatted/Defatted flour) and their protein preparations. Molecules. 27, 6339 (2022).
Hernández-Álvarez, A. J., Mondor, M., Piña-Domínguez, I. A. & Sánchez-Velázquez, O. A. Melgar Lalanne, G. Drying technologies for edible insects and their derived ingredients. Drying Technol. 39, 1991–2009 (2021).
Ojha, S., Bußler, S., Psarianos, M., Rossi, G. & Schlüter, O. K. Edible insect processing pathways and implementation of emerging technologies. J. Insects Food Feed. 7, 877–900 (2021).
Psarianos, M., Iranshahi, K., Rossi, S., Gottardi, D. & Schlüter, O. Quality evaluation of house cricket flour processed by electrohydrodynamic drying and pulsed electric fields treatment. Food Chem. 138276 https://doi.org/10.1016/j.foodchem.2023.138276 (2023).
Lucas-González, R., Fernández-López, J. & Pérez-Álvarez, J. A. Manuel Viuda-Martos, ·. Effect of drying processes in the chemical, physico-chemical, techno-functional and antioxidant properties of flours obtained from house cricket (Acheta domesticus). Eur. Food Res. Technol. 245, 1451–1458 (2019).
Zielińska, E., Karaś, M. & Baraniak, B. Comparison of functional properties of edible insects and protein preparations thereof. LWT. 91, 168–174 (2018).
Ando, Y., Watanabe, T., Orikasa, T. & Tomita, S. Structural Differences between Hot Air-Dried and freeze-dried two-spotted cricket (Gryllus Bimaculatus), House Cricket (Acheta domesticus), and Silkworm (Bombyx mori) larvae and their effect on Powder properties after grinding. Food Bioproc Tech. 1, 1–12 (2023).
Villaseñor, V. M., Enriquez-Vara, J. N., Urías-Silva, J. E. & Mojica, L. Edible insects: Techno-functional Properties Food and feed applications and biological potential. Food Reviews Int. 38, 866–892 (2022).
Kröncke, N. et al. Effect of different drying methods on nutrient quality of the yellow mealworm (Tenebrio molitor L). Insects. 10, 84 (2019).
Mannozzi, C. et al. Nonthermal technologies affecting techno-functional properties of edible insect-derived proteins, lipids, and chitin: a literature review. Innovative Food Sci. Emerg. Technol. 88, 103453 (2023).
Rostamabadi, H. et al. Impact of emerging non-thermal processing treatments on major food macromolecules: Starch, protein, and lipid. Trends Food Sci. Technol. 141, 104208 (2023).
Alles, M. C. et al. Bio-refinery of insects with pulsed electric field pre-treatment. Innovative Food Sci. Emerg. Technol. 64, 102403 (2020).
Bogusz, R. et al. Nutritional Value and Microbiological aspects of dried yellow mealworm (Tenebrio molitor L.) Larvae pretreated with a Pulsed Electric Field. Appl. Sci. 14, 968 (2024).
Psarianos, M. et al. Effect of pulsed electric fields on cricket (Acheta domesticus) flour: extraction yield (protein, fat and chitin) and techno-functional properties. Innovative Food Sci. Emerg. Technol. 76, 102908 (2022).
Lammerskitten, A. et al. Pulsed electric field pre-treatment improves microstructure and crunchiness of freeze-dried plant materials: case of strawberry. LWT. 134, 110266 (2020).
Janssen, R. H., Vincken, J. P., van den Broek, L. A. M., Fogliano, V. & Lakemond, C. M. M. Nitrogen-to-protein Conversion factors for three Edible insects: Tenebrio molitor, Alphitobius diaperinus, and Hermetia illucens. J. Agric. Food Chem. 65, 2275–2278 (2017).
Bogusz, R. et al. The Pulsed Electric Field Treatment Effect on Drying Kinetics and Chosen Quality aspects of Freeze-dried black soldier fly (Hermetia illucens) and yellow mealworm (Tenebrio molitor) Larvae. Appl. Sci. 13, 10251 (2023).
Kozłowska, M., Gruczyńska, E., Ścibisz, I. & Rudzińska, M. Fatty acids and sterols composition, and antioxidant activity of oils extracted from plant seeds. Food Chem. 213, 450–456 (2016).
ISO 12966-2:2017. Animal and Vegetable Fats and Oils—Gas Chromatography of Fatty Acid Methyl Esters—Part 2: Preparation of Methyl Esters of Fatty Acids.
Górska, A. et al. Berry Seeds—A By-Product of the Fruit Industry as a source of oils with Beneficial Nutritional Characteristics. Appl. Sci. 13, 5114 (2023).
Cichocki, W. et al. Chemical characteristics and thermal oxidative Stability of Novel Cold-pressed oil blends: GC, LF NMR, and DSC studies. Foods. 12, 2660 (2023).
Bryś, J. et al. Use of GC and PDSC methods to characterize human milk fat substitutes obtained from lard and milk thistle oil mixtures. J. Therm. Anal. Calorim. 130, 319–327 (2017).
Brzezińska, R. et al. Quality Evaluation of Plant Oil blends interesterified by using immobilized Rhizomucor miehei lipase. Appl. Sci. 12, 11148 (2022).
Zozo, B., Wicht, M. M., Mshayisa, V. V. & van Wyk, J. The Nutritional Quality and Structural Analysis of Black Soldier fly Larvae Flour before and after Defatting. Insects. 13, 168 (2022).
Vanga, S. K., Wang, J., Jayaram, S. & Raghavan, V. Effects of Pulsed Electric fields and Ultrasound Processing on proteins and enzymes: a review. Processes. 9, 722 (2021).
Tamborrino, A. et al. Pulsed Electric Fields for the Treatment of Olive Pastes in the oil extraction process. Appl. Sci. 10, 114 (2019).
Andreou, V. et al. Shelf-life evaluation of virgin olive oil extracted from olives subjected to nonthermal pretreatments for yield increase. Innovative Food Sci. Emerg. Technol. 40, 52–57 (2017).
Shorstkii, I., Khudyakov, D. & Mirshekarloo, M. S. Pulsed electric field assisted sunflower oil pilot production: impact on oil yield, extraction kinetics and chemical parameters. Innovative Food Sci. Emerg. Technol. 60, 102309 (2020).
Shorstkii, I., Mirshekarloo, M. S. & Koshevoi, E. Application of Pulsed Electric Field for Oil Extraction from sunflower seeds: Electrical Parameter effects on Oil Yield. J. Food Process. Eng. 40, e12281 (2017).
Sarkis, J. R., Boussetta, N., Tessaro, I. C., Marczak, L. D. F. & Vorobiev, E. Application of pulsed electric fields and high voltage electrical discharges for oil extraction from sesame seeds. J. Food Eng. 153, 20–27 (2015).
Rábago-Panduro, L. M., Morales-de la Peña, M., Martín-Belloso, O. & Welti-Chanes, J. Application of Pulsed Electric fields PEF on pecan nuts Carya illinoinensis Wangenh. K. Koch: oil extraction yield and compositional characteristics of the oil and its By-product. Food Eng. Rev. 13, 676–685 (2021).
Bogusz, R. et al. Effect of Pulsed Electric Field Technology on the composition and bioactive compounds of black soldier fly Larvae dried with convective and infrared–convective methods. Molecules. 28, 8121 (2023).
Brogan, E. N., Park, Y. L., Shen, C., Matak, K. E. & Jaczynski, J. Characterization of lipids in insect powders. LWT. 184, 115040 (2023).
Calder, P. C. Functional roles of fatty acids and their effects on Human Health. J. Parenter. Enter. Nutr. 39, 18S–32S (2015).
Mihaly Cozmuta, A. et al. Cricket and yellow mealworm powders promote higher bioaccessible fractions of mineral elements in functional bread. J. Funct. Foods. 99, 105310 (2022).
Selaledi, L. & Mabelebele, M. The influence of drying methods on the Chemical composition and body color of yellow mealworm (Tenebrio molitor L). Insects. 12, 333 (2021).
Radzikowska, U. et al. The influence of dietary fatty acids on Immune responses. Nutrients. 11, 2990 (2019).
Sardesai, V. M. Nutritional role of fatty acids. in Integrative and Functional Medical Nutrition Therapy 135–149 (Springer International Publishing, Cham, doi:https://doi.org/10.1007/978-3-030-30730-1_10. (2020).
Kim, Y. B. et al. Black soldier fly larvae oil as an alternative fat source in broiler nutrition. Poult. Sci. 99, 3133–3143 (2020).
Venn-Watson, S., Schork, N. J. & Pentadecanoic Acid :0), an essential fatty acid, Shares clinically relevant cell-based activities with leading longevity-enhancing compounds. Nutrients 15. (C15, 4607 (2023).
Ying, Q., Wojciechowska, P., Siger, A., Kaczmarek, A. & Rudzińska, M. Phytochemical content, oxidative Stability, and Nutritional properties of unconventional cold-pressed edible oils. J. Food Nutr. Res. 6, 476–485 (2018).
Ratusz, K., Symoniuk, E., Wroniak, M., Rudzińska, M. B. & Compounds Nutritional quality and oxidative Stability of Cold-pressed Camelina (Camelina sativa L.) oils. Appl. Sci. 8, 2606 (2018).
Symoniuk, E., Ksibi, N., Wroniak, M., Lefek, M. & Ratusz, K. Oxidative Stability Analysis of Selected Oils from unconventional raw materials using Rancimat Apparatus. Appl. Sci. 12, 10355 (2022).
Karrar, E. et al. Fatty acid composition and stereospecificity and sterol composition of milk fat from different species. Int. Dairy. J. 128, 105313 (2022).
Dima, C., Assadpour, E., Dima, S. & Jafari, S. M. Bioavailability of nutraceuticals: role of the food matrix, processing conditions, the gastrointestinal tract, and nanodelivery systems. Compr. Rev. Food Sci. Food Saf. 19, 954–994 (2020).
Cichońska, P., Bryś, J. & Ziarno, M. Use of natural biotechnological processes to modify the nutritional properties of bean-based and lentil-based beverages. Sci. Rep. 13, 16976 (2023).
Hurtado-Ribeira, R. et al. The Interaction of Slaughtering, Drying, and defatting methods differently affects oxidative quality of the Fat from Black Soldier fly (Hermetia illucens) Larvae. Insects. 14, 368 (2023).
Mahboubifar, M., Yousefinejad, S., Alizadeh, M. & Hemmateenejad, B. Prediction of the acid value, peroxide value and the percentage of some fatty acids in edible oils during long heating time by chemometrics analysis of FTIR-ATR spectra. J. Iran. Chem. Soc. 13, 2291–2299 (2016).
Symoniuk, E., Marczak, Z., Brzezińska, R., Janowicz, M. & Ksibi, N. Effect of the Freeze-Dried Mullein Flower Extract (Verbascum nigrum L.) Addition on Oxidative Stability and Antioxidant Activity of Selected Cold-Pressed Oils. Foods 12, 2391 (2023).
Turck, D. et al. Safety of dried yellow mealworm (Tenebrio molitor larva) as a novel food pursuant to regulation (EU) 2015/2283. EFSA J. 19, 1–29 (2021).
Wirkowska-Wojdyła, M., Ostrowska-Ligęza, E., Górska, A. & Bryś, J. Application of Chromatographic and Thermal methods to study fatty acids composition and positional distribution, Oxidation Kinetic parameters and Melting Profile as important factors characterizing Amaranth and Quinoa oils. Appl. Sci. 12, 2166 (2022).
Beniwal, A. S., Singh, J., Kaur, L., Hardacre, A. & Singh, H. Meat analogs: protein restructuring during thermomechanical processing. Compr. Rev. Food Sci. Food Saf. 20, 1221–1249 (2021).
Son, Y. J., Lee, J. C., Hwang, I. K., Nho, C. W. & Kim, S. H. Physicochemical properties of mealworm (Tenebrio molitor) powders manufactured by different industrial processes. LWT. 116, 108514 (2019).
Stokes, J. R., Boehm, M. W. & Baier, S. K. Oral processing, texture and mouthfeel: from rheology to tribology and beyond. Curr. Opin. Colloid Interface Sci. 18, 349–359 (2013).
Shah, D. S. et al. A concise summary of powder processing methodologies for flow enhancement. Heliyon. 9, e16498 (2023).
Mshayisa, V. V., Van Wyk, J., Zozo, B. & Nutritional Techno-Functional and Structural Properties of Black Soldier Fly (Hermetia illucens) Larvae flours and protein concentrates. Foods. 11, 724 (2022).
Jung, H., Lee, Y. & Yoon, W. Effect of moisture content on the grinding process and Powder properties in Food: a review. Processes. 6, 69 (2018).
Fitzpatrick, J. Powder Properties in food Production Systems. In Handbook of Food Powders 285–308 (Elsevier, 2013). https://doi.org/10.1533/9780857098672.2.285
Szulc, K., Nazarko, J., Ostrowska-Ligęza, E. & Lenart, A. Effect of fat replacement on flow and thermal properties of dairy powders. LWT - Food Sci. Technol. 68, 653–658 (2016).
Cichowska-Bogusz, J., Figiel, A., Carbonell-Barrachina, A. A., Pasławska, M. & Witrowa-Rajchert, D. Physicochemical properties of dried Apple slices: impact of Osmo-Dehydration, Sonication, and drying methods. Molecules. 25, 1078 (2020).
Bolat, B., Ugur, A. E., Oztop, M. H. & Alpas, H. Effects of High Hydrostatic pressure assisted degreasing on the technological properties of insect powders obtained from Acheta domesticus & Tenebrio molitor. J. Food Eng. 292, 110359 (2021).
Zalewska, M., Marcinkowska-Lesiak, M. & Onopiuk, A. Application of different drying methods and their influence on the physicochemical properties of tomatoes. Eur. Food Res. Technol. 248, 2727–2735 (2022).
Tobolková, B., Takáč, P., Mangová, B. & Kozánek, M. A comparative study of colour characteristics of thermally/non-thermally treated mealworm larvae (Tenebrio molitor) by means of UV/Vis spectroscopy and multivariate analysis. J. Food Meas. Charact. 15, 3791–3799 (2021).
Lenaerts, S., Van Der Borght, M., Callens, A. & Van Campenhout, L. Suitability of microwave drying for mealworms (Tenebrio molitor) as alternative to freeze drying: impact on nutritional quality and colour. Food Chem. 254, 129–136 (2018).
Janssen, R. H. et al. Involvement of phenoloxidase in browning during grinding of Tenebrio molitor larvae. PLoS One. 12, e0189685 (2017).
Zhang, Z. Q. et al. Effects of traditional grinding and superfine grinding technologies on the properties and volatile components of Protaetia brevitarsis larvae powder. LWT. 173, 114307 (2023).
Borremans, A. et al. Effect of blanching plus fermentation on selected functional properties of Mealworm (Tenebrio molitor) powders. Foods. 9, 917 (2020).
Ostrowska-Ligęza, E., Dolatowska-Żebrowska, K., Wirkowska-Wojdyła, M., Bryś, J. & Górska, A. Comparison of thermal characteristics and fatty acids composition in Raw and Roasted Cocoa beans from Peru (Criollo) and Ecuador (Forastero). Appl. Sci. 11, 2698 (2021).
Huang, C. et al. Impact of drying method on the nutritional value of the edible insect protein from black soldier fly (Hermetia illucens L.) larvae: amino acid composition, nutritional value evaluation, in vitro digestibility, and thermal properties. Eur. Food Res. Technol. 245, 11–21 (2019).
Acknowledgements
The research for this publication was carried out with the use of research equipment purchased as part of the “Food and Nutrition Centre–modernisation of the WULS campus to create a Food and Nutrition Research and Development Centre (CŻiŻ)” co-financed by the European Union from the European Regional Development Fund under the Regional Operational Programme of the Mazowieckie Voivodeship for 2014–2020 (project no. RPMA.01.01.00-14-8276/17).The authors would like to thank Maja Kaźmierczyk and Joanna Powichrowska for their help.
Author information
Authors and Affiliations
Contributions
R.B., M.N. and K.S. conceptualized the study. R.B., K.S., M.N. and J.B. prepared methodology. R.B. and M.N. visualized the study. R.B., J.B., K.R. conduct investigation. R.B., K.S., K.R. and J.B. conduct formal analysis. R.B., K.S., J.B., and K.R. wrote the original draft. M.N. and K.S. supervised the project. M.N. and K.S. reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bogusz, R., Nowacka, M., Bryś, J. et al. Quality assessment of yellow mealworm (Tenebrio molitor L.) powders processed by pulsed electric field and convective drying. Sci Rep 14, 27792 (2024). https://doi.org/10.1038/s41598-024-79412-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-79412-0