Abstract
We investigated tail-belting (TB), the newly-discovered freeze avoidance behavior among wild rodents. When temperatures dropped to -6 °C, wild mice (Apodemus agrarius and Apodemus flavicollis) were observed curling their tails inward and positioning it on the back. A literature search suggested TB had never been documented, presumably because rodents, especially in the laboratory, are seldomly assayed under cold stress. Due to the infrequent occurrence of the behavior, we used infrared and thermal cameras to confirm observations. We also collected tail-skin samples to investigate whether any physiological mechanisms might co-occur with TB. If such mechanisms were found, they could inform wider debate involving freeze protection among mammals, and could potentially lead to understanding mammal susceptibility or resilience to sudden temperature changes such as those associated with climate change. Lastly, we scored behaviors by bank voles (Myodes glareolus) which unexpectedly visited chambers. Across four winters, we observed TB in both Apodemus species during subzero conditions, but bank voles never performed the behavior. We also confirmed that TB occurs as an adaptive reflex which warms the tail. From tissue samples, we found that free amino acids, peptides, and glycoproteins were significantly higher during cold-stress. Thus, TB may have been accompanied by the expression of cold-protective proteins which ostensibly enable the peripheral body parts of mammals to survive temperatures well below 0 °C. These findings should inspire new dialogue regarding the role of lipids in tissues of peripheral organs in mammals. By extension, our findings may lead to the discovery of a putative cryoprotection mechanism among mammals.
Similar content being viewed by others
Introduction
Over the last six years, our team has been using naturalistic field assays to document rodent behavior in the field under a range of ambient conditions as an adjunct to laboratory studies1,2. Such a naturalistic approach allowed the testing of hypotheses that would be difficult to examine within a laboratory setting, and often resulted in findings with higher ecological validity. Moreover, we were able to observe and describe phenomena that are not observable in laboratory conditions3. Specifically, during the winter of 2021, we viewed a peculiar behavior that had not previously been documented. We first witnessed “tail-belting”; henceforth referred to as “TB”4 while scoring behaviors of the striped field mouse (Apodemus agrarius) and yellow-necked mouse (Apodemus flavicollis). The peculiar and obvious behavior, which only occurred in subzero temperatures, comprised the curling of the tail inward and positioning it on the dorsal, medial, or lateral area of the rump or back (Fig. 1B,C,E, & F). Upon examination, we learned that TB only occurred after the tail temperature fell below -6 °C. We further documented that animals exhibiting this behavior were either resting or feeding on highly-palatable food lures that had been used to attract animals into experimental, laboratory-style enclosures4.
(A) Image of a striped field mouse (A. agrarius); (B) Infrared video frame capturing tail-belting behavior in A. agrarius. (C) Thermal imaging video frame depicting tail-belting expression at –9 °C in A. agrarius. (D) Image of a yellow-necked mouse (A. flavicollis); (E) Infrared video frame showing tail-belting behavior in A. flavicollis. (F) Thermal imaging video frame displaying tail-belting expression at –7 °C in A. flavicollis.
TB has never been documented under laboratory conditions, even though Apodemus is related to the common laboratory mouse (Mus musculus), one of the most common laboratory animals in the world5,6. Our persistent lack of observing the behavior is likely because laboratory rodents are seldom subjected to cold stress. After all, animal welfare regulations necessitate maintaining temperatures within a range of 20-24ºC7. Additionally, the literature relating to cold stress and rodents has been limited to physiological mechanisms related to the protection of the body, with the focal point centered around precise thermal damage to structures (e.g., a selected area of the skin8), and not the examination of behaviors. Furthermore, those studies would not document the behavior of wild animals under natural conditions. Importantly, because TB behavior has not been previously described in any mammalian literature, its adaptive function has not been determined either.
TB was expressed solely under conditions that could cause cold stress. Therefore we assumed its principal function was to increase the temperature of the tail tissues from the outer surface. Given that rodent tail damage appears to be quite rare among the hundreds of rodents we have studied over the years—even during severe winters in the temperate climate zone—we can assume this behavior is part of a strategy to protect tails. This strategy appears effective because, even though the duration of the TB is usually quite short, it only occurred during extended feeding bouts longer than a few seconds4. Due to these experience and observations, we couldn’t help but wonder whether TB had been documented in other rodent species in cold climates, or if this behavior had simply gone undiscovered or ignored. Following an extensive search in the literature, we found no record of any similar behavior under laboratory or field conditions. We now question whether this behavior could be present among other similar long-tailed rodents, potentially even progenitors of the common laboratory mouse. But since there are so few assays that test mice under cold conditions, presence or absence of TB could remain unknown unless we provide compelling reasons to undertake such investigations. Our first step to justifying further tests, therefore, is to better understand the conditions that drive the expression of the behavior.
With so many questions facing a potential new discovery, we determined that our first step would be to undertake comprehensive thermal monitoring of ground and ambient temperature. Thermal imaging of the tail before the expression of TB, during the time when the tail is thrust over the back, and immediately after it has been retracted is essential. At the same time, because our initial findings were more serendipitous than experimental, we also wanted to improve the working protocol to more accurately document TB and the conditions it occurs within. Given the infrequent occurrences of this behavior, and the lack of reported frostbite data among wild rodent species, we also wanted to know if there were also active physiological mechanisms that protect peripheral body parts against low temperatures. We also wanted to know whether these mechanisms were different from those previously described in the literature, such as those involving blood flow regulation.
Importantly, during our previous research4, we documented an odd occurrence that may indicate other non-behavioral mechanisms are indeed protecting the tail, even before the rare TB behavior is expressed. Principally, we found that prior to lifting the tail, the surface temperature of A. agrarius tails are similar to the ambient temperature even when the temperature has dropped well below 0 °C. This perplexing finding has been guiding our exploratory search for possible mechanisms operating at the molecular level and tissue biochemistry, to explain how tissue survival is possible. For instance, we wondered how such regulation might operate because simple blood flow-based explanations appear inadequate given the persistent drop in tail surface temperature far below the freezing point of water (0 °C) as well as blood and other body fluids of mammals (0.5—to slightly above -2.5 °C, depending on the sources9,10. Likewise, the freezing point of non-perfused skin is below 0 °C (ranging from -0.53 to -0.65 °C11.
We hope that the line of inquiry we are proposing will encourage new approaches to shed light on the physiological mechanisms that rodents use to protect peripheral appendages from frostbite. In this way, the findings might help shape the discourse regarding factors previously not considered to play a significant role in safeguarding mammals against frostbite injuries. Perhaps they will guide research towards previously unconsidered directions that are crucial for addressing this issue, thereby outlining pathways for further study planning by our team and others.
Therefore, we conducted several pilot ‘exploratory’ tests aimed at determining the appropriate research directions to follow. We began with biochemical analyses to help identify causative elements. Proteins, including glycoproteins, and compounds encompassing their metabolites (amino acids and peptides) play a crucial role in protecting animal tissues from damage caused by the drop in temperatures below the freezing point. Thus, based on the literature12,13,14,15,16,17, we aimed to determine the potential involvement of quantitative changes in amino acids, lipids, and proteins, as well as modifications of the latter, commonly known as anti-freeze factors, among several non-mammalian animal species as there is ample data available on this topic concerning fish from polar regions, hibernating amphibians, and primarily insects in winter diapause12,13,14,15,16,17. Thus, behavioral studies with detailed temperature measurements may determine the direction of further research on this novel finding. We therefore, decided to perform a series of tests comparing selected parameters in the tail tissues of rodents captured during winter and contrasted with those caught during the summer.
Our goal was to support preliminary findings, particularly concerning the role of TB in tail warming. Previous observations we gathered, reveal that tail tissue temperatures might drop significantly below 0 °C, beyond levels observed in any mammal, even hibernators. In light of this, we consider a detailed examination of the tails’ surface temperature before TB expression a crucial goal. The finding that tail tissues reach subzero temperatures led us to hypothesize that a series of biochemical factors (likely contributing to lowering the freezing point of bodily fluids) at the tissue level, might protect the tail skin from frostbite.
Methods
Species identification
We deployed laboratory-style field chambers beside active colonies of two species, A. agrarius, which has a long, dorsal black stripe, and A. flavicollis, which has a yellow stripe around the neck, and no stripe on the back. A. flavicollis may rarely be confused with the similar looking wood mouse (A sylvaticus18). Therefore, to have the utmost level of certainty in, we performed genetic analysis on tail samples of 18 A. flavicollis. Tail samples were collected (from within 150 m of our chambers) between December 2021 and June 2023. Species level identification was determined based on cytochrome b gene (cytb) as a molecular marker (GeneBank accession numbers are AB032853 and AB033695 for cytb of A. flavicollis and A. sylvaticus respectively). PCR testing revealed that 100% of samples were indeed identified as A. flavicollis. Additionally, a sample of animals (N = 6) was captured and subjected to testing for the presence of toxoplasmosis (4 females and 2 males). However, all six individuals were negative for toxoplasmosis, ensuring that infection would not influence the behaviors.
Behavioral observation
After deploying the experimental chambers, we recorded the behaviors of free-living colonies from both species, the striped field mouse (A. agrarius; Fig. 1A) and the yellow-necked mouse (A. flavicollis; Fig. 1D). The controlled observations took place on a private, partly-wooded, suburban property in Warsaw, central Poland (52°20’N 21°03’E, altitude: 80 m). For a general and schematic representation of the protocol, see Fig. 2. The experimental observations took place between November 27, 2021 and February 14, 2022, and therefore covered the coldest period of the late-autumn and winter seasons in Poland. Temperatures during this period dropped down to -14 °C. Based on video observations (differences in body size, coat patterns, scars etc.) over 3 months, and the number of animals trapped directly after the cease of observation (N = 7), we estimate the number of mice visiting the chamber at or above the minimum of 10 individuals. Subsequent observations were also conducted during the winters of 2022/23 (with temperatures maximally dropping to -13 °C) and 2023/24 (with a minimum temperature of -15 °C).
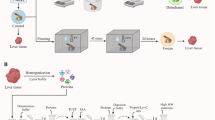
Schematic drawing of the experimental setup for the behavioural aspect of the study. Free-living colonies of A. agrarius and A. flavicollis had access to an experimental chamber placed in a forested area and equipped with a thermal imaging and CCTV cameras and thermometer. During the autumn-winter period, a highly palatable food (Nutella) was presented inside the chamber. Based on observations of individual differences in body shape and size, as well as post-experiment live trapping, we estimate the minimum number of animals visiting the experimental chamber and expressing TB to be 10 individuals. Occasionally, the experimental chamber was also visited by bank voles (Myodes glareolus), whose behavior we documented and refer to as a curiosity later in the text of the paper.
We used continuous video recording in a laboratory-style field chamber (Fig. 3) constructed from 12 mm waterproof plywood and painted with Luxens – odorless acrylic paint (Unicell international, Wasilków, Poland). The internal floor dimensions were 35 × 40 cm with a wall height of 70 cm. Chambers were deployed near cover beside bushes in the natural habitat of neighboring forest and meadows. Sewer pipe (70 mm diameter and 50 cm length; Certus, Cieszyn, Poland) was connected to the entrance hole. The lower surface of the chamber was covered with 1 cm of rinsed sand. Animals had 24/7, ad libitum access to a chocolate-nut cream (Nuss Milk Krem; i.e., Nutella™, Ferrero SpA, Alba, Italy) placed on a 70 mm Petri dish (PRO Scientific Inc., Oxford, CT, USA) (Fig. 3B). No light source was added, so the chamber remained dark throughout the entire study.
(A) The chamber used in the study with an outdoor temperature and humidity data logger (yellow), entrance pipe (grey), and two boxes with power supply, cables, and Android device recording thermal images via Wi-Fi connection with thermal camera; (B) The inside of the chamber with an entrance pipe (grey), internal data logger for temperature and humidity measurement (yellow), two infrared cameras recording from different angles, a Nutella as bait placed on a Petri dish, and a thermal camera, the screen of which was covered during the recording to keep the chamber totally dark.
For continuous surveillance, we used two separate recording systems – an Easycam EC-116-SCH connected to an Easycam EC-7804 T recorder (Eltrox Sp. z o.o., Częstochowa, Poland) and a GISE 4W1 GS-2CMD4-VF-V2 camera (Eltrox Sp. z o.o.) connected to a KENIK KG-1154L recorder (Eltrox Sp. z o.o.). This setup enabled 24/7 motion detection recording for the duration of the study. When the ambient temperature dropped below 0 °C, we deployed a thermal camera (Seek Thermal Shot SW-AAA camera; Santa Barbara, CA, USA), placed 30 cm above the Petri dish for continuous recording (Fig. 3B). The thermal image was transferred via Wi-Fi to an Android device (Samsung Galaxy A8 2018, Samsung Electronics Co., Suwon, South Korea), and video was saved via 24/7 screen recording with a Super Screen Recorder (Dream Heli Technology Co., Ltd., Beijing, China). To prevent the Android device from shutting down due to low temperatures, it was sometimes kept warm inside a tool box with the assistance of a 50W infrared Terrarium Heat Lamp. The temperature and humidity measurements were taken at 10–15 above the ground level inside while outside the chamber we deployed two CEM DT-172 data loggers to collect external temperature and humidity (Shenzhen Everbest Machinery Industry CO. LTD, Shenzhen, China). Both data loggers were validated by examining the convergence of measurements between them and an additional device, the Thermohygrometer BC06 (Trotec, Heinsberg, Germany). The maximum observed differences were 0.1 °C. The accuracy of the thermal imaging camera was validated using two temperature probes TP-04 (Tenmars Electronics Co. LTD., Taipei, Taiwan) and STP-03/K (Termoprodukt, Bielawa, Poland) and a laboratory thermometer DT-31 (Termoprodukt). This validation was carried out at both positive and negative temperatures by measuring the temperature of the sand. The results demonstrated complete consistency between the two methods.
Tissue collection for biochemical measurements
Rodents were live-trapped using Trip traps (Trixie, Heimtierbedarf GmbH & Co. KG, Tarp, Germany) and Vaco Professional live traps (Vaco, Wrocław, Poland). Six individuals (female and male) of A. agrarius were captured twice in the winter season (when the observations were carried out) and twice in the summer season. Four individuals from each season (2021/2022 and 2022/2023) were used in the studies (a total of eight individuals—four females and four males—for each analysis). Because no differences were observed between males and females for any of the parameters examined, the results are presented collectively for both sexes in the figures. The same procedure was followed for C57BL/6 J laboratory mice (M. musculus) obtained from the Animal Facility of the Faculty of Biology, University of Warsaw. The results from these mice were considered as the reference parameters in the winter and summer groups for A. agrarius. Both male and female C57BL/6 J mice, aged 24–26 weeks, were used; for these analyses, we used tissues from mice (female:male, 50:50 ratio) euthanised during the winter and summer seasons (eight individuals in total). Animals were maintained at a temperature of 22 °C, a 12 h light: 12 h dark photoperiod, and given ad libitum access to food and water throughout their development. For an overview of the number of animals used for biochemical analysis, see Fig. 4. Both wild and laboratory mice were euthanized with an overdose of the inhalational anesthetic Forane (isoflurane; AbbVie Inc., North Chicago, IL, USA). Three 2–2.5 cm fragments of tails from the central part were extracted with a razor blade and immediately frozen in liquid nitrogen, where the material was kept until analyses.
Tail tissue collection and biochemical analysis. Samples were taken from free-ranging A. agrarius and laboratory M. musculus (C57BL/6J) during cold and warm seasons. Biochemical analysis included measurements of free amino acids, peptides, glycoprotein moieties, total lipids, phospholipids and glycerol.
Just prior to the extraction of the investigated factors, tissues were prepared on dry ice. Separated skin fragments (including subcutaneous tissue) were then used for the analysis of free amino acids, peptides, glycosylated proteins (sugar moieties linked to proteins), total lipids, phospholipids and glycerol content using dedicated commercial kits or biochemical methods described below. The total protein content in tissue samples or the wet weight of tissue fragments served as a reference during the validation of the quantities of the studied factors. For protein extraction, we used the truXTRAC® Protein Extraction Buffer Super B (Covaris, Woburn, MA, USA), while for lipids and their metabolites, we employed the buffers recommended by the manufacturers of the respective kits or following the described procedure. The protein content in the samples was determined using the Cayman Protein Determination Kit (Cayman Chemical, Ann Arbor, MI, US) which utilizes the Bradford method, involving the measurement of sample absorbance at 595 nm on transparent 96-well plates, Corning™ NBS™ Microplates (Corning Inc., Corning, NY, USA), in spectrophotometer/spectrofluorometer –VICTOR3 V™ multilabel counter (Perkin Elmer–Wellesley, MA, USA). To determine the protein content in the samples, which was required for specific tests, we employed standard Bovine Serum Albumin (Capricorn Scientific GmbH, Ebsdorfergrund, Germany) solutions. For normalizing values with respect to tissue mass, we employed an analytical laboratory scale—Gram FV (Waagenet AF GmbH, Berlin, Germany). We consistently used either 25 or 50 mg of tissue for the analyses, depending on the procedural requirements.
Peptides and free amino acids content
The measurements were performed on samples standardized for equal amounts of total proteins, as determined using the Cayman Protein Determination Kit. To quantify peptides, we employed the Pierce™ Quantitative Peptide Assays & Standards kit (Thermo Fisher Scientific Inc., Waltham, MA, USA), and for determining the concentration of free L-amino acids, we employed the Abnova™ L-Amino Acid Quantitation Kit (Abnova, Taipei, Taiwan). The first method is based on the reduction of copper ions (from Cu+2 to Cu+1) through the amide backbone of peptides in the Biuret reaction (under alkaline conditions)19. Afterward, chelator coupling with the reduced copper occurs to generate a bright red complex, the level of which is measured at 480 nm. The second method employs an enzyme-catalyzed oxidation of L-amino acids to convert a dye into a colored state. The absorbance at 570 nm is in direct proportion to the L-amino acid content in the samples under examination. Both, peptides and amino acids analyzes were performed in 96-well Corning™ NBS™ Microplates in VICTOR3 V™ reader.
Content of glycopeptides/glycoproteins
Pierce™ Glycoprotein Carbohydrate Estimation Kit (GCE) (Thermo Fisher Scientific Inc.) was used to measure level of protein glycosylation likely to be related with cells protection against frostbite in the tail tissues. The measurements were conducted on samples standardized based on the total protein content. GCE is designed to effectively estimate the amount of oxidizable glycosylation (percent carbohydrate by weight) of purified protein samples. Sugar groups in the glycoprotein sample are first oxidized with sodium meta-periodate to produce detectable aldehyde groups that, upon reacting with a detection reagent, yield a purple-colored product, the concentration of which can be determined colorimetrically at 550 nm. All of these measurements we conducted on multi-well Pierce™ 96-Well Polystyrene Plates, shaped for universal use -with corner notch (Thermo Fisher Scientific Inc.), using the Victor3 plate reader.
Total lipids, phospholipids and glycerol content
For extraction of total lipids, fragments of tail skin weighing 50 mg were homogenized at room temperature in chloroform–methanol (1:2, v/v) as described earlier for the extraction of lipids from rodent skin (Wilkinson 1966). Following centrifugation of the suspension, lipids were recovered from the solution by evaporation to dryness in vacuo using a rotary drier at 50 °C in SpeedVac Concentrator AES1010 (SAVANT Instruments Inc., New Castle, DE, USA), and purified by extraction with hexane-dichloromethane (1:1, v/v); all organic solvents were of analytical grade and purity (POCH S.A., Gliwice, Poland). The total lipid content of the samples was then determined by measuring their mass.
The phospholipids in tail skin samples weighing 25 mg were measured using Cell Biolabs’ total Phospholipid Assay Kit (Cell Biolabs, Inc., San Diego, CA, USA) in accordance with the manufacturer’s procedure based on a series of enzyme driven reactions. In this method a hydrolyzing enzyme reacts with phospholipids, breaking the phosphodiester bond within their structure. Next, an oxidoreductase reacts to generate a peroxide, which is subsequently detected with a highly specific fluorescence probe. The content of the reaction product is measured at 530/585 nm wavelength.
In order to measure the glycerol in the samples of 50 mg of tails skin, we used the Free Glycerol Assay Kit (abcam, Cambridge, UK). In this procedure, glycerol is enzymatically oxidized to generate a product which reacts with the probe to generate color product whose quantity was quantified at a wavelength of 570 nm. Phospholipids and glycerol analyses mentioned above were performed in 96-well Corning™ NBS™ Microplates in VICTOR3 V™ reader.
Statistics
Data from biochemical studies were analyzed using Mann–Whitney test (IBM Corp. IBM SPSS Statistics for Windows version 29.0.2.0 Armonk, NY, USA).
Results
Behavioral
Observed animals spent considerable amounts of time, up to 15 min, with their tails immobile on frozen ground at temperatures as low as around -6 or -7 °C (Fig. 5). Seventy-four new instances of TB were recorded during 22 visits inside field chambers; 37 instances occurred by striped field mice (A. agrarius, see example recording in Supplementary file 1) and 34 by yellow-necked mice (A. flavicollis, see example recording Supplementary file 2). The highest temperature at which A. agrarius exhibited tail-belting (TB) was -2.8 °C inside the chamber (compared to -3.7 °C outside). A. flavicollis displayed TB at -3.7 °C (while the outside temperature was -4.7 °C; see Supplementary file 3).
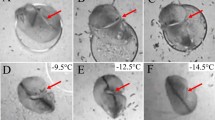
Video stills from thermal imaging depict striped field mice (A. agrarius) (A-C) and yellow-necked mice (A. flavicollis) exhibiting tail temperatures significantly below the freezing point of water. Tail temperatures were comparable to ground temperatures (for NETD = 70mK), i.e., (A) -3°C; (B) -4°C; (C) -6°C for A. agrarius, and (D) -5°C; (E) -7°C; (F) -8°C for A. flavicollis. Precise measurement was sometimes hindered due to mouse movement. Red arrows indicate the location of the tails. NETD = noise equivalent temperature difference.
There were several several (e.g., fewer than 10) unexpected visits from bank voles (Myodes glareolus) during subzero temperatures (Fig. 6A,B; see example recording in Supplementary file 4). After scoring these visits, we determined they did not express TB, despite the temperature dropping all the way to -9 °C. Similar to A. agrarius and A. flavicollis, the surface temperature of bank voles’ tails was comparable to the ambient temperature, i.e., -5 °C and -7 °C (Fig. 6C,D).
Despite the relatively low number of recorded visits to the experimental chamber, we observed instances of bank voles(M. glareolus) exploring the chamber in sub-zero temperatures.No tail belting was observed; however, similar to A. flavicollisand A. agrarius, the tail temperatures of bank voles alsodropped well below zero degrees Celsius during our observations. (A) Photograph of a bank vole; (B)Infrared camera still showing a bank vole feeding in the experimental box; (C, D) Thermal images depicting thetemperatures of bank voles' tails, reaching as low as -5 °C and -7 °C respectively (noise equivalent temperaturedifference = 70mK).
Thermal camera recordings further confirmed our hypothesis that the function of tail-belting is to heat the tail to a temperature above the frostbite risk threshold in both studied species (Fig. 7). Exemplary recordings of tail-belting expression in both infrared and mid-infrared (i.e., thermal) for both species can be found in Supplementary file 1 and Supplementary file 2.
While collecting tissue samples, we measured the tail length and body size of each individual. The mean tail length for A. agrarius and A. flavicollis living was 71.8 mm and 97.6 mm, respectively, while the mean tail length of M. glareolus was 40.5 mm (N = 12 for each species). Detailed data can be found in Supplementary file 5.
Behaviors were documented during winter 2021/2022, with 3 additional instances from the two more recent winters. The latter three were incidental and not part of the experiment, but they were noted as additional evidence. For each instance, we recorded animals lifting and/or curling the tail inward and positioning it on the dorsal, medial, or lateral area of the rump or back. Despite not performing systematic observations, it appears the speed and the degree of tightness (the force applied to press the tail against the body) varied considerably and was likely inversely correlated with temperature, with greater speed and pressure exerted on the tail against the body at lower temperatures. There were no instances of this behavior observed among bank voles, despite their occasional visits to the experimental chamber in temperatures well below zero (Fig. 6).
Biochemical analyses
We observed significant increases in A. agrarius tissues collected during winter as compared to summer. All increases were accompanied by large effect sizes in several biochemical measures. These include a 117% rise in free amino acids (U = 70.5, Z = 2.15, p = 0.032, Cohen’s r = 0.54; Fig. 8A), 71% higher free peptides (U = 73, Z = 2.05, p = 0.040, Cohen’s r = 0.51; Fig. 8B), a 135% increase in glycoproteins (U = 61, Z = 2.51, p = 0.012, Cohen’s r = 0.63; Fig. 8C), 31% more fat (U = 72, Z = -2.09, p = 0.037, Cohen’s r = 0.52r; Fig. 8D), a 50% rise in phospholipids (U = 64, Z = 2.39, p = 0.017, Cohen’s r = 0.60; Fig. 8E), and 162% higher glycerol levels (U = 57, Z = 2.66, p = 0.008, Cohen’s r = 0.67; Fig. 8F).
The concentration of biochemical factors considered to possibly play a role in frost protection for in A. agrarius over two seasons, summer and winter, normalized to their average concentration in tail tissues of laboratory mice (M. musculus): (A) free amino acids – yellow dots; (B) peptides – orange dots; (C) glycoprotein carbohydrate content – red dots; (D) total lipids – brown dots including (E) phospholipids – beige dots and (F) glycerol – blue dots; each dot represents the mean value from five measurements of the examined parameter in one individual. In all biochemical studies, the mean value of a given parameter for M. musculus was considered 100%, and the average values for individuals of A. agrarius specimens were expressed in relation to this standard. The horizontal longest lines represent the mean, upward and downward whiskers (bounded by horizontal lines) indicate + /- SEM. Data were analyzed using the Mann–Whitney U test; significance levels: *p < 0.05, p < 0.025.
Discussion
Our findings suggest a gradual increase in tail temperature (Fig. 7) and reinforces our hypothesis that TB’s function is frostbite prevention. Additionally, the 74 new occurrences of TB support the 115 instances previously observed by Stryjek et al. (2021a). Given the relatively short lifespan of both species, rarely exceeding 1 year20, we almost certainly observed different groups of animals each winter. We also found that tail temperature can drop well below the freezing point of water and remain at such temperatures without frostbite (Fig. 5). It is worth noting that although thermal imaging illustrates surface temperature, it is probable that the internal tissue temperatures of the tail also dropped, however, the question remains as to how close it got to 0 °C, or if it even fell below that level (since the freezing point of blood is below 0 °C). It is conceivable that circulation could still be sustained in these conditions, at least theoretically, since the complete cessation of circulation in the organs of active mammals has not been described until now; even in hibernating Arctic ground squirrels, whose body temperature drops below 0 °C, blood circulation is maintained, although driven by only 3–4 heartbeats per minute (for comparison, during periods of activity—in summer, it reaches 200–300 beats per minute;21. It should be noted that, to date, the state of blood flow through any peripheral organs and the circulation within any organs of these hibernating squirrels have not been analyzed. We also observed that in many instances, animals held their tails in subzero temperatures against surfaces for extended periods up to 10 min, as indicated by several observations recorded at -4 °C and -6 °C.
It is important to consistently consider the exceptionally unfavorable surface area-to-volume ratio (in terms of heat loss and body cooling) in small rodents. Moreover, given the highly efficient circulation in active small mammals, peripheral structures like a long tail are especially disadvantageous for maintaining a non-frozen state if the core body temperature alone is expected to keep the tail tissues from freezing. This leads us to hypothesize that the temperature of the deeper layers of the tail skin, along with other protective mechanisms against frostbite, play a role – beyond those related solely to heat dissipation through the blood. These observations steer the discussion toward the likely presence of physiological adaptations in tissues to subzero temperatures which may be based on biochemical and molecular mechanisms that we are familiar with, but did not know for certain that they are employed in the skin of mammals exposed to dangerously low temperatures.
We found that TB may occur at higher temperatures than described in the previous study4. The data from our weather stations suggest that each species might have different thresholds for the behavior. The temperature at which TB was expressed was also variable due to possible correlations with time spent outside the nest, exhaustion, and hunger levels. Equally interesting are the data obtained from the unexpected visits from bank voles. Like the two Apodemus species, bank voles were observed to keep their tail temperatures consistent with ambient temperatures. However, they did not reveal any instances of tail-belting (TB). The lack of TB may be due to their dynamic movement patterns22, shorter tails, or possibly the limited number of observations we conducted. Therefore, we cannot definitely say that bank voles do not express TB.
The TB behavior appears to require no learning and is executed reflexively, thus seemingly meeting the criteria of an unconditional reflex, with the speed and force of tail-belting increasing with decreasing temperatures (RS, unpublished observations). Further investigation could seek to determine the corresponding neural mechanism underlying this reflexive response, along with the physiology of cold receptors in the peripheral organs of Apodemus mice.
Biochemical
When discussing the potential involvement of factors that protect tissues from freezing below 0 °C, it is essential to emphasize the lack of data on molecular cryoprotective factors in mammals. Even when this topic is considered, it focuses on hibernating mammals (which are in a completely different physiological state than the mice we studied), and such factors have not been described in these species. The vertebrates that have evolved such factors and are intensively studied in this regard are limited to fish, amphibians and a limited number of reptiles23. Additionally, in the search for analogies, their cryoprotective factors are compared to those found in insects, the group of animals that has received the most attention in this field. Therefore, we believe our findings represent are novel and noteworthy, and may establish a new direction in research on mammals concerning the protection of tissues from frostbite-induced damage.
The dramatic increases in proteins, their metabolites, and lipids (Fig. 8) during the winter period validate our hypothesis that these factors are working in conjunction with TB in order to keep peripheral tissue from freezing. Importantly, these findings also hint at the possible presence of a cryo-protein factor, previously unknown among mammals. For instance, after conducting a literature review on antifreeze peptides and proteins, we found many of them are glycosylated factors characterized by high antifreeze activity due to sugar moieties16. This prompted us to measure the level of glycoproteins in the tail skin tissues of rodents in the winter and summer seasons. Results were consistent with our hypothesis that the quantity of glycoproteins and probably glycopeptides in the examined samples from winter is significantly higher than in samples from summer. It is noteworthy that their quantity in the bodies of insects and fish also changes seasonally12,13,14,15.
Physiologically, lipids and their metabolites can also serve protective functions for tissues under conditions of temperature dropping below 0 °C. This is due to their role as substrates for aerobic cellular respiration in such conditions, and their chemical properties can increase the flow of oxygen through cells, as demonstrated in Antarctic fish17. The higher level of total lipids observed in the tail skin tissues in winter compared to summer is most likely a result of changes in the volume of the adipose tissue located beneath the dermis. It seems that the next step in these studies should be to determine the level of lipolytic activity in the tail tissues during the winter season and compare this data with knowledge about such activity in the summer, which is also supported by data obtained from the study of Antarctic fish24. There is very little data on the relationship between lipids in mammals in response to temperature drops, particularly at the local tissue level, and the available data mostly pertains to hibernation or hypothermia. An example of this is the changes in the levels of cholesterol esters, phospholipids, and ketone bodies in the little ground squirrel (souslik) Citellus pygmaeus (Spermophilus pygmaeus)25.
In addition to lipids, other compounds in mammals also undergo changes in response to decreases in body temperature or the temperature of its peripheries, but these data concern hibernating individuals. Thus, remaining in a state entirely different from the non-hibernating Apodemus species that we are studying. Moreover, these data focus on answering questions about changes in tissue metabolism resulting from temperature drops, primarily concerning the secondary metabolites produced, which can be dangerous in this state (such as reactive oxygen species), as well as factors that protect against toxic substances and slow down metabolism (such as H2S) and many proteins with protective roles—anti-apoptotic and anti-inflammatory26 and the works cited therein). However, none of these phenomena play a cryoprotective role, making it difficult to apply this knowledge in the context of our discussion. Nevertheless, there is some hope in the re-presentation of a 1989 report, which involved changes in body temperature in the Arctic ground squirrel Spermophilus parryii27. This was verified again in 2023 with the addition of several new insights28 indicating a possible drop in body temperature below 0 °C during hibernation in these mammals. Although this finding is not directly related to ours, as we do not characterize factors associated with hibernation physiology, these studies strongly suggest the need to search for factors responsible for protecting tissues from freezing in mammals, as they have not yet been identified in this group.
It may also be important to consider the potential functional differences between subcutaneous white adipose and dermal white adipose tissues, which remain relatively poorly understood physiologically29,30,31,32. While their role in cold protection is at least debated in rodents when considering their role in the trunk, almost nothing is known about their role in peripheral organs under challenging conditions resulting from a significant drop in temperature.
Metabolites of lipids, acting as cryoprotectants, are utilized by amphibians. Studies have shown that the level of glycerol in the tissues of hibernating Salamandrella keyserlingii, Rana arvalis, Hyla versicolor, Pseudacris crucifer, and Dryophytes chrysoscelis at temperatures below 0 °C is very high. Additionally, Rana sylvatica, with a high level of this alcohol in its system, can hibernate at temperatures below -18 °C23,33,34,35. The increase in glycerol levels in tissues has also been described in diapausing arthropods, providing evidence of its cryoprotective role23,36. Whether glycerol locally, i.e., in the tissues of peripheral organs in mammals, plays a protective role against low temperatures is not clear. However, the local production and release of glycerol into the extracellular space by adipose tissue cells located in various organs have been documented in the literature37,38,39,40,41,42,43. Interestingly, adipose tissue releasing glycerol has a significantly reduced ability to recycle it, and therefore, it may linger locally in the intercellular space for some time44. Therefore, measuring the amount of glycerol in tail skin tissues seemed justified to us, and as it turned out, the obtained result is intriguing and encourages further research in this area. This is because the glycerol levels in the tail tissues of winter mice were significantly higher (162% increase) as compared to mice captured during the summer.
Our findings from the initial biochemical analyses of A. agrarius skin tissue justifies further exploration of the mechanisms by which mammalian peripheral structures defend against frostbite—when their local temperature drops below the freezing point of water, and the issue pertains to molecular-level regulation. This process appears to be essential and most likely accompanies systemic solutions (blood flow in the deeper layers of organs) and behavioral adaptations, such as TB. Although there are numerous reports of physiological protections against frostbite and heat loss (related to blood circulation and energy production)—information so well known that it can be found in any general and comparative physiology textbook—the issue of the molecular basis of the processes we are discussing here, specifically and exclusively concerning active (non-hibernating) mammals, is represented quite differently. To our knowledge, there is essentially no data on this topic.
Nonetheless, it is very interesting to also consider the situation studied in the Arctic ground squirrels, where the phenomenon of supercooling of tissues and body fluids has been postulated21. Such supercooling, being a particularly dangerous physical phenomenon for the integrity of tissues and cells, seems to require the existence of factors that would protect against the formation of ice crystals or safeguard against those that are already forming. We believe that the continuation of the research we propose here is an important and intriguing direction in this context.
Despite the need for more data, it is important to note that biochemical analyses have so far only been conducted on samples obtained from A. agrarius. Although we expect similar results in A. flavicollis (and it is likely the same applies to M. glareolus as well), these assumptions require empirical confirmation. Further comprehensive studies conducted also in controlled laboratory environments are recommended to assess the factors triggering tail-belting (TB), as well as to precisely determine the temperature of tail tissues. This could be crucial due to uncertainty regarding whether the measured temperature reflects the core temperature of the tail or its surface temperature. We expect to further answer to the question of whether antifreeze proteins and other active factors set up the thermal hysteresis of tissues in those most prone to frostbite extremities such as limbs, ears, noses and tails. This phenomenon has not yet been characterized in mammals, and would certainly be an interesting find. The expression of the tail-belting behavior, supported by cryoprotective physiological adaptations at the tissue level, further elucidates the evolutionary success of rodent taxa and their remarkable adaptability to diverse environments.
Ethics statement
This observational study involved minimally invasive methods, focusing on monitoring free-ranging animals that could choose to enter or avoid experimental chambers equipped with food and video cameras. As a result, it did not require approval from the local ethics committee for animal experimentation. This molecular research was conducted at the University of Warsaw’s Faculty of Biology, which is registered as institution number 0031 by the Polish Ministry of Science for authorized rodent studies. Tissue collection was conducted by certified staff. According to the Act on the Protection of Animals Used for Scientific or Educational Purposes, enacted on January 15, 2015 (Article 2, point 6), euthanizing mice for organ or tissue collection does not require ethics committee approval.
The study took place on private land with the owners’ permission, adhering to the Polish Animal Protection Act (August 21, 1997). All procedures followed the ARRIVE guidelines (Kilkenny et al. 2010) to ensure ethical and methodological standards were met.
Data availability
The raw data are available as supplemental tables.
References
Stryjek, R., Parsons, M. H., Fendt, M., Święcicki, J. & Bębas, P. Let’s get wild: A review of free-ranging rat assays as context-enriched supplements to traditional laboratory models. J. Neurosci. Methods 362, 109303 (2021).
Parsons, M. H. et al. Making a case for the free exploratory paradigm: animal welfare-friendly assays that enhance heterozygosity and ecological validity. Front. Behav. Neurosci. 17, 1228478 (2023).
d’Isa, R., Parsons, M. H., Chrzanowski, M., Bebas, P. & Stryjek, R. Catch me if you can: Free-living mice show a highly flexible dodging behaviour suggestive of intentional tactical deception. Royal Soc. Open Sci. 11, 231692 (2024).
Stryjek, R., Parsons, M. H. & Bebas, P. A newly discovered behavior (‘tail-belting’) among wild rodents in sub zero conditions. Sci. Rep. 11, 1–7 (2021).
Rubtsov, N. et al. Comparative analysis of DNA homology in pericentric regions of chromosomes of wood mice from genera Apodemus and Sylvaemus. Russ. J. Genet. 51, 1233–1242 (2015).
Knitlová, M. & Horáček, I. Late pleistocene-holocene paleobiogeography of the genus Apodemus in central Europe. PloS One 12, e0173668 (2017).
Curzer, H. J., Perry, G., Wallace, M. C. & Perry, D. The three Rs of animal research: what they mean for the institutional animal care and use committee and why. Sci. Eng. Ethics 22, 549–565 (2016).
Auerbach, L. J. et al. A novel mouse model for frostbite injury. Wilderness Environ. Med. 24, 94–104 (2013).
Collins, D. A. & Scott, F. The freezing points of serum and corpuscles. J. Biol. Chem. 97, 189–213 (1932).
Bjerg, M., Rasmussen, M. D. & Nielsen, M. Changes in freezing point of blood and milk during dehydration and rehydration in lactating cows. J. Dairy Sci. 88, 3174–3185 (2005).
Keatinge, W. & Cannon, P. Freezing-point of human skin. The Lancet 275, 11–14 (1960).
Wilkens, C. & Ramløv, H. Seasonal variations in antifreeze protein activity and haemolymph osmolality in larvae of the beetle Rhagium mordax (Coleoptera: Cerambycidae). CryoLetters 29, 293–300 (2008).
Qiu, L., Mao, X., Hou, F. & Ma, J. A novel function–thermal protective properties of an antifreeze protein from the summer desert beetle Microdera punctipennis. Cryobiology 66, 60–68 (2013).
Xiang, H., Yang, X., Ke, L. & Hu, Y. The properties, biotechnologies, and applications of antifreeze proteins. Int. J. Biol. Macromol. 153, 661–675 (2020).
Walker, V. K. et al. in Insect timing: circadian rhythmicity to seasonality 199–211 (Elsevier, 2001).
Urbańczyk, M., Góra, J., Latajka, R. & Sewald, N. Antifreeze glycopeptides: From structure and activity studies to current approaches in chemical synthesis. Amino Acids 49, 209–222 (2017).
Sidell, B. in Biology of Antarctic fish 220–231 (Springer, 1991).
Bartolommei, P. et al. Field identification of Apodemus flavicollis and Apodemus sylvaticus: A quantitative comparison of different biometric measurements. Mammalia 80, 541–547 (2016).
Shen, C.-H. Quantification and analysis of proteins. Diagnostic Mol. Biol., 187–214 (2019).
Haitlinger, R. Morphological variability in Apodemus agrarius (Pallas 1771); Zmienność morfologiczna Apodemus agrarius (Pallas 1771). Acta Theriologica 6, 239–255 (1962).
Boyer, B. B. & Barnes, B. M. Molecular and metabolic aspects of mammalian hibernation: Expression of the hibernation phenotype results from the coordinated regulation of multiple physiological and molecular events during preparation for and entry into torpor. Bioscience 49, 713–724 (1999).
Karantanis, N.-E., Rychlik, L., Herrel, A. & Youlatos, D. Arboreal gaits in three sympatric rodents Apodemus agrarius, Apodemus flavicollis (Rodentia, Muridae) and Myodes glareolus (Rodentia, Cricetidae). Mamm. Biol. 83, 51–63 (2017).
Storey, K. B. & Storey, J. M. Molecular physiology of freeze tolerance in vertebrates. Physiol. Rev. 97, 623–665 (2017).
Sidell, B. D. & Hazel, J. R. Triacylglycerol lipase activities in tissues of Antarctic fishes. Polar Biol. 25, 517–522 (2002).
Otis, J. P., Sahoo, D., Drover, V. A., Yen, C.-L.E. & Carey, H. V. Cholesterol and lipoprotein dynamics in a hibernating mammal. PLoS One 6, e29111 (2011).
Giroud, S. et al. The torpid state: Recent advances in metabolic adaptations and protective mechanisms. Front. Physiol. 11, 623665 (2021).
Barnes, B. M. Freeze avoidance in a mammal: Body temperatures below 0 C in an arctic hibernator. Science 244, 1593–1595 (1989).
Lee, T. N., Kohl, F., Buck, C. L. & Barnes, B. M. Hibernation strategies and patterns in sympatric arctic species, the Alaska marmot and the arctic ground squirrel. J. Mammal. 97, 135–144 (2016).
Alexander, C. M. et al. Dermal white adipose tissue: A new component of the thermogenic response. J. Lipid Res. 56, 2061–2069 (2015).
Zhang, S. et al. Molecular cloning, sequence analysis and homology modeling of the first caudata amphibian antifreeze-like protein in axolotl (Ambystoma mexicanum). Zool. Sci. 30, 658–662 (2013).
Segalla, L., Chirumbolo, S. & Sbarbati, A. Dermal white adipose tissue: Much more than a metabolic, lipid-storage organ?. Tissue Cell 71, 101583 (2021).
Li, Y., Long, J., Zhang, Z. & Yin, W. Insights into the unique roles of dermal white adipose tissue (dWAT) in wound healing. Front. Physiol. 15, 1346612 (2024).
Costanzo, J. P., do Amaral, M. C. F., Rosendale, A. J. & Lee, R. E. Jr. Hibernation physiology, freezing adaptation and extreme freeze tolerance in a northern population of the wood frog. J. Exp. Biol. 216, 3461–3473 (2013).
do Amaral, M. C. F., Frisbie, J., Goldstein, D. L. & Krane, C. M. The cryoprotectant system of Cope’s gray treefrog, Dryophytes chrysoscelis: Responses to cold acclimation, freezing, and thawing. J. Comp. Physiol. B 188, 611–621 (2018).
Berman, D., Meshcheryakova, E. & Bulakhova, N. in Doklady Biological Sciences. 137–141 (Springer).
Storey, K. B. & Storey, J. M. Natural freezing survival in animals. Annual Rev. Ecol. Systemat. 27, 365–386 (1996).
Rotondo, F. et al. Glycerol is synthesized and secreted by adipocytes to dispose of excess glucose, via glycerogenesis and increased acyl-glycerol turnover. Sci. Rep. 7, 8983 (2017).
van der Merwe, M.-T.S. et al. Lactate and glycerol release from subcutaneous adipose tissue in black and white lean men. J. Clin. Endocrinol. Metab. 84, 2888–2895 (1999).
Hibuse, T., Maeda, N., Nagasawa, A. & Funahashi, T. Aquaporins and glycerol metabolism. Biochimica et Biophysica Acta (BBA)-Biomembranes 1758, 1004–1011 (2006).
Langin, D. Control of fatty acid and glycerol release in adipose tissue lipolysis. Comptes. Rendus. Biolog. 329, 598–607 (2006).
Maggs, D. G. et al. Interstitial fluid concentrations of glycerol, glucose, and amino acids in human quadricep muscle and adipose tissue. Evidence for significant lipolysis in skeletal muscle. J. Clin. Investig. 96, 370–377 (1995).
Gambert, S., Héliès-Toussaint, C. & Grynberg, A. Extracellular glycerol regulates the cardiac energy balance in a working rat heart model. Am. J. Physiol.-Heart Circulatory Physiol. 292, H1600–H1606 (2007).
Chiadak, J. D. et al. Lipopolysaccharide modifies glycerol permeability and metabolism in 3T3-L1 adipocytes. Int. J. Mol. Sci. 18, 2566 (2017).
Chakrabarty, K., Tauber, J. W., Sigel, B., Bombeck, C. T. & Jeffay, H. Glycerokinase activity in human adipose tissue as related to obesity. Int. J. Obes. 8, 609–622 (1984).
Acknowledgements
This research was self-funded by the authors, with in-kind assistance from the Centre for Urban ecological studies (CUES). Equipment funds were awarded to RS (Polish National Science Centre (NCN)—Grant Number: UMO-2013/09/B/HS6/03435) and PB (IDUB University of Warsaw—Grant Number: BOB-IDUB-622-322/2022).
Author information
Authors and Affiliations
Contributions
R.S., M.H.P, and P.B. Conceptualization; R.S. Data curation; R.S., M.H.P., and P.B. Formal analysis; R.S., M.H.P., and P.B.; Funding acquisition; R.S., P.B Investigation; R.S., M.H.P., and P.B. Methodology; R.S. Project administration; R.S. and P.B. Resources; R.S. Visualization; R.S., M.H.P., and P.B. Writing-original draft; R.S., M.H.P., and P.B. Writing-review & editing; R.S., M.H.P., and P.B. approved final version of manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Material 1
Supplementary Material 2
Supplementary Material 4
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Stryjek, R., Parsons, M.H. & Bebas, P. Insights into tail-belting by wild mice encourages fresh perspectives on physiological mechanisms that safeguard mammal tissues from freezing. Sci Rep 14, 28933 (2024). https://doi.org/10.1038/s41598-024-79594-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-79594-7