Abstract
Microplastics (MPs) are widespread in oceans worldwide, posing a significant threat to aquatic ecosystems. The abundance of these particles in water is related to population density and urban development, functioning as a sink for MPs. However, the pollution levels due to the accumulation of MPs in the estuarine in Amazonian mangrove areas remain unknown. Here, we show that population density and fishing activities influence the distribution of MP pollution in these waters. We found the highest abundance of MPs in the inner (1.03 items/m3) and outer (0.82 items/m3) portions of the estuary, corresponding to the areas with the highest population density and fishing activity, respectively. The main morphological characteristics of MPs are fibers (89.8%), blue color (55.2%), and size of 1000–2000 μm (31.7%). The risk analysis showed a low level of danger, suggesting that the potential impact on the ecosystem is still small. Our results demonstrate that the presence of human populations and their fishing activities significantly influence the accumulation of MPs in the estuarine waters on the Amazon coast. Based on our findings, more sophisticated analysis using MPs’ spatial distribution models can be associated with hydrodynamic processes, aiming to define pollution hotspots and support the mitigation of their emerging negative effects. In addition, monitoring and generating valuable information are the main targets for developing comprehensive strategies to preserve biodiversity and sustainability in the Amazon coastal zone.
Similar content being viewed by others
Introduction
Estuaries represent transitional environments in which the fresh waters of rivers meet the ocean’s salty waters. This unique ecosystem is home to a diversity of plant and animal species adapted to dynamic environmental conditions1,2. Globally, estuaries play a crucial role in coastal regions, supporting activities such as navigation, transportation, agriculture, fishing, recreation and tourism3. However, the substantial and variable rates of primary productivity in these environments are subject to pollution, with plastic standing out as a prominent pollutant4,5,6.
Plastics, due to their versatility, resistance, and affordability, play a significant role in various spheres of human life7,8,9. However, the mismanagement of plastics, from inadequate disposal methods to the absence of efficient solid waste management, has resulted in a significant worsening of plastic pollution10. The presence of plastics constitutes 80% of the global waste identified along coastlines, and this proportion tends to increase with the growth of plastic production11.
Plastic materials fragment into microplastics (MPs), tiny particles less than 5 mm in size12,13 due to natural factors such as abrasion and weathering, MPs are widespread in the oceans, posing a significant threat to aquatic ecosystems14,15. Their presence has been detected in surface waters16,17,18,19, sediments20,21,22,23,24, plants25, drinking water, food26,27,28, fish29, mussel30, oyster31, cattle, poultry, and pig32 and even in the human body (blood, feces, urine, human milk, lung tissue)33,34,35,36.
The scientific literature on MPs in coastal and estuarine ecosystems has grown in recent decades, reflecting the urgency of understanding the dynamics and impacts of these pollutants37. These ecosystems are particularly vulnerable to MP pollution due to their direct exposure to the local pollution source38. The abundance of MPs in inland waters is correlated with population density and urban development39. Since communities around estuaries are often densely populated, these environments can act as sinks for MP debris. It is suggested that these particles can be transferred along the food chain, resulting in significant damage to organisms and ecosystems40. Consequently, the ingestion of MPs poses a potential risk to both the health of aquatic organisms and to human health40,41. Furthermore, many researchers have used the Local Pollution Index (PLI) to assess the risk of MP pollution in their regions40,41,42.
Brazilian research on microplastic pollution in coastal and estuarine waters is still limited16,43,44,45. Here, we identify the presence and abundance of MPs in the estuarine waters of the tidal channels on the Ajuruteua Peninsula, considering the influence of the Taperaçu and Caeté River estuaries. Located in the northern region of the Brazilian Amazon, here we tested our hypothesis that tidal channels close to towns and cities with the highest density of human population and fishing activities are key points for the accumulation of MPs through analysis of the abundance, size, shape, color, and types of polymers. The objectives of this study involve i) the distribution of the abundance of MPs in the different sampling points that represent the tidal channels in the two estuaries and ii) the risk assessment of the microplastic pollution load in these estuaries.
Material and methods
Study site
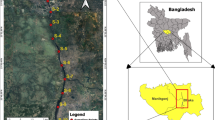
The Ajuruteua Peninsula, located in Bragança in the state of Pará, is 200 km from the mouth of the Amazon River in northern Brazil (Fig. 1). This peninsular region is significantly shaped by the hydrodynamic influence of two prominent estuaries. To the west is the Taperaçu estuary, whereas to the east, the region is influenced by the Caeté River estuary (Fig. 1)46. These two estuaries play crucial roles in coastal dynamics, influencing not only the topography but also the local ecology47,48.
It is interesting to note that these estuaries are interconnected through tidal channels, among which are the "Furo do Taici", “Furo Grande”, and the “Furo do Meio”. These tidal channels facilitate the hydraulic interconnection between the estuaries and play a vital role in transporting sediments and nutrients, directly affecting biodiversity and environmental conditions along the peninsula. This connection between estuaries and tidal channels creates a unique environment where the interactions between coastal and river waters play a fundamental role in the sustainability of the local ecosystem.
The region is characterized by a macrotidal regime49,50. Extending for approximately 30 km in length and 15 km in width, the area comprises 166 km2 of mangroves. The predominant vegetation is typically mangrove, represented by three species: Rhizophora mangle L., Avicennia germinans (L.) L., and Laguncularia racemosa (L.) C.F. Gaertn. and are subject to different flood frequencies, tidal ranges, and salinities51.
The region has an equatorial climate characterized by warm temperatures and high humidity. Annual rainfall varies between 2,300 and 2,800 mm, with up to 30% inter-annual variation50. This pattern is strongly influenced by the Intertropical Convergence Zone (ITCZ) shifts, resulting in a monsoon-like climate with significant rainfall and northeast trade winds from January to June50. On the other hand, between July and December, rainfall is substantially reduced while southeasterly trade winds prevail. This seasonal alternation contributes to the region’s unique climate dynamics, directly influencing vegetation, water resources, and other aspects of the local ecosystem47,49.
Sampling methodology
The water samples were collected at nine sampling stations (S1—S9) along the Ajuruteua Peninsula, located in Bragança, in the state of Pará (Fig. 1). Three tidal channels in the study area were investigated: Furo do Taici (S1, S2, and S3), Furo do Meio (S4, S5, and S6), and Furo Grande (S7, S8, and S9). The collections were carried out in the dry season during the spring tide in 2022. Water samples were collected using horizontal trawls in the sub-surface layer of the water column (20 cm). For this purpose, a conical-cylindrical net with sisal rope (300 µm mesh opening and 30 cm mouth diameter) was equipped with a mechanical flow meter to calculate the volume of water sampled. Three trawls were carried out at each station, totaling 27 samples. The aluminum boat, equipped with an outboard motor, moved at 1.5 knots for approximately three minutes during each trawl. After each trawl, the net was washed with ultrapure water (UPW) to concentrate all the materials retained in the mesh, and the contents were transferred to a sterilized glass jar and stored for later analysis, covered with aluminum foil. All containers and tools were cleaned with UPW before each trawl.
Extraction of MPs
Aliquots of the water samples (50 mL) were treated with 10 mL of 30% hydrogen peroxide (H2O2) for 24 h at room temperature, in the dark, in glass beakers, to degrade the organic matter (Fig. S1). The supernatant was then filtered with a vacuum pump using a filter funnel with GF/F glass fiber paper (47 mm in diameter). After filtering, 100 mL of UPW was added to the funnel to remove all the plastic particles (Fig. S1). The filters were then placed in Petri dishes covered with aluminum foil and left to dry at room temperature for 24 h to allow the particles to be observed.
Identification of MPs
The dried samples were thoroughly examined under a Leica S8APO stereoscopic microscope (with magnification ranging from 1.0 × to 8.0x) equipped with LAS® software. This procedure is in line with the protocol outlined by Peng et al.52 and was used to identify, classify, and measure the material collected. The MPs were categorized into four groups based on shape (fiber, fragment, film, and sphere) and seven categories based on color (blue, transparent, black, white, yellow/green, shiny, and pink/red). The maximum size of each particle was measured (µm) using a stereoscopic microscope.
In addition, the polymers present in the samples were identified using Fourier transform infrared spectroscopy (FTIR) and Raman spectrometry. A Frontier spectrometer (PerkinElmer, USA) was used for the FTIR analysis, which scans the 8,300 to 350 cm-1 range. The spectrometer, equipped with a DynasCAn interferometer, sealed and desiccated optics coated with Ge KBr, provides a resolution of 0.4 cm-1 and a signal-to-noise ratio of 10,000:1 in 5 s of scanning, assisted by the extender accessory.
As for Raman spectroscopy, the spectra were recorded in the 100 to 4000 cm-1 range using LabRam HR Evolution equipment, an excitation laser with a wavelength of 785 nm, a power of 50 mW, and a diffraction grating with 600 lines/mm. The acquisition time was set at 10 s, with 10 accumulations. Precision in the location of the sample analysis was ensured by an optical microscope attached to the equipment, using a 50 × magnification objective lens. This set of techniques provides a comprehensive and detailed analysis of the physical and chemical characteristics of the microplastics present in the samples.
Risk analysis
To quantify the extent of MP contamination in the surface waters of tidal channels, an integrated Pollutant Load Index (PLI) was used, following the methodology proposed by Tomlinson et al.53. PLI values allow the classification of the risk associated with pollutant levels, which can be categorized as follows: PLI below 10 is considered as (I) low risk; PLI between 10 and 20 is (II) moderate risk; PLI between 20 and 30 is (III) high risk; and PLI above 30 is classified as (IV) very high risk (Table S1). This index was calculated using the following formula:
The expression "Csi" denotes the level of MP measured at each sampling site, while "C0" represents the lowest abundance of MP observed in the current field study, corresponding to the uncontaminated condition since there is no absolute zero reference value.
Quality control
All sample collection procedures in the laboratory were carried out in a strictly sterile environment, under a laminar flow hood, to avoid any potential contamination37,54. In addition, the laboratory benches were cleaned regularly, and all the equipment and utensils used, including beakers, funnels, and tools, were thoroughly washed and rinsed with UPW before and after each use. These materials were stored covered with aluminum foil37. As an additional precaution, all materials used in the analysis were made of glass or stainless steel, when possible, and the clothes used in the laboratory were made from cotton or natural fibers. These rigorous practices aimed to ensure the integrity and reliability of the results obtained, contributing to quality and precision.
Statistical analysis
The abundance of MPs in the water samples is expressed in MPs/m3 as the mean and standard deviation. Statistical analysis was conducted using non-parametric methods in Statistica 7 software. The normality of the raw data between MP abundance and the locations (Furo do Taici, Furo do Meio, and Furo Grande) was verified using the Shapiro–Wilk test, and the homoscedasticity was verified using Levene’s test. Analysis of variance (One-way ANOVA) was used to compare locations, and the post hoc test LSD (least significant difference) was used to determine which data groups were responsible for differences in the analyses. Two-way ANOVA with replication was used to compare locations (column factors) and stations (row factors). In a multivariate approach, the Penrose (P) and Mahalanobis (M) distances were also used to test the distances between the locations, considering the means, variances, covariances, and correlations between these variables. These statistical approaches were chosen to ensure a robust and reliable assessment of the differences between the sampling locations and stations, considering the specific characteristics of our data.
Results and discussion
Abundance of MPs
The average abundance of MPs in the water was 1.03 items/m3. MP distribution varied significantly between locations (Furo do Taici, Furo do Meio, and Furo Grande – One-way ANOVA, F = 3.72; P = 0.0381) (Fig. 2a). LSD’s post hoc comparison indicated Furo do Taici had higher MP abundance than the others (Fig. 2a). A comparative analysis by using Two-way ANOVA also confirmed significant difference between locations (F = 6.20; P = 0.0090) and, on the other hand, no significant difference between stations considering all locations (F = 1.72; P = 0.2044) were found (Fig. 2b–d). Considering a multivariate approach, results showed that the largest distances were found between Furo do Meio and Furo Grande (P = 10.91, M = 34.21) and the smallest between Furo do Taici and Furo do Meio (P = 4.29, M = 12.12).
Variations in MP concentrations in water ranged between 0.77 and 1.50 items/m3 in Furo do Meio and Furo do Taici, respectively. Concerning the Amazon River (8 to 39 MPs/m3; 16), concentrations in our study are substantially lower. A study undertaken on the Amazon River about the risk assessment of MPs revealed that urban cities near tidal channels can generate high concentrations of MPs throughout the main water body. These concentrations are high enough to cause ecotoxicological effects in aquatic organisms, potentially16. In Belém, in the state of Pará, urban rivers have MP concentrations of 167—3095 MPs/m3 16, which are higher than those of the Amazon River and the Ajuruteua Peninsula’s tidal channels in Bragança, Pará.
Concentrations of MP on the Amazon Continental Shelf range from 323 to 5733 MPs/m3 44. This difference demonstrates the influence of the methodology used on the results obtained in each area16,44. The specific characteristics of environments can significantly influence the results obtained. A notorious example of this variability is observed in the Yangtze River in China, where concentrations of MPs differed substantially between different studies. Whereas Xiong et al.55 reported an average abundance of 0.9 items/m3 (using a 333 µm net), Zhao et al.56 found an average of 4,137.3 ± 2,461.5 items/m3 (using a 12 V Teflon pump) for the same estuary. Thus, it is important to critically evaluate MP sampling methodologies when interpreting and comparing data, as differences in methods can lead to differing results. Furthermore, assessing the proximity of sampling points to urban areas is important due to the positive relationship between the abundance of MPs in surface waters and the surrounding population density, such as in the Yangtze Estuary57.
Characteristics and composition of MPs
Morphological characteristics of MPs
Using protocols and standardized methods related to color and shape12, the MPs in this research were classified into seven distinct color groups: blue, transparent, black, white, shiny, yellow/green, and pink/red; this last group takes into account initial similarities and also nuances associated with the effects of degradation58 (Fig. 3a).
Microplastic colors and forms in the samples: (a) Presence of MPs with their contribution by color (transparent, blue, black, white, shiny, pink/red, and others); (b) Presence of MPs with the contribution form (fiber, fragment, film, and others) for each tidal channel in the dry season during the spring tide in 2022, on the Ajuruteua Peninsula, Bragança, Pará, Brazil.
In tidal channels on the Ajuruteua Peninsula, blue (55.2%) and transparent (31.9%) emerged as the predominant colors among the particles found. Next, black (8.6%) was also notable, whereas pink/red (2.1%), white (0.9%), yellow/green (0.9%), and shiny (0.4%) were rarely identified. This same pattern was observed along the Furo do Taici, Furo do Meio, and Furo Grande (Fig. 3a). MPs with different colors may originate from white MPs, which may indicate the possibility that they were affected by light exposure and biological effects58. The blue and transparent MPs may be from fishing materials widely used in this region, such as nets, PET bottles, and fishing lines. Around 30% of the MP samples in the Amazon River were blue16. These particles often contain polymers such as polyethylene (PE), polypropylene (PP), high-density polyethylene (HDPE), low-density polyethylene (LDPE), and in some cases, polymer chloride ethylene (CPE)59. White and transparent MPs may have their origin associated with food packaging60. Distinct color distributions of MPs in watersheds result from anthropogenic sources and activities specific to the region, highlighting the need for targeted mitigation strategies. For example, in the middle and downstream sections of the Yangtze River, blue-colored MPs predominated, attributed to rice cultivation, in which blue plastic film is used to prepare seedlings61.
Morphologically, the MPs were classified into four shape categories: fragment, fiber, film, and other (composed of sphere, pellet, and/or granule). The shape is an essential characteristic when identifying MPs, as it provides us with information on the origin of this material, i.e., whether the particle is of primary or secondary origin62. Fibers tend to predominate in most estuaries16,63,64,65,66. Fibers were the dominant form of MPs, accounting for 89,8% of MPs on the Ajuruteua Peninsula, followed by fragments (6.3%), films (2.5%), and others (1.4%). Many studies have reported fiber as the predominant form of MPs in river ecosystems since fibers can enter water bodies through multiple sources. In Furo do Taici, the percentage of fiber was 91.5%; in Furo do Meio, it was 89.4%; and in Furo Grande, it was 88.6% (Fig. 3b; Fig. S2).
The abundance of fibrous material in tidal channel waters can be attributed, in part, to the contribution of fishing lines or nets67. However, more recent scientific studies emphasize that fibers originate primarily from domestic wastewater68. Specifically, washing clothes can be a significant MPs source, with substantial releases into aquatic environments. Washing clothes can release a considerable amount of MPs, estimated at between 124 and 308 mg/kg, which, through municipal sewage systems, can find their way into rivers69. The significant presence of microplastic fibers in the Amazon region can be attributed, in part, to the absence or limited capacity of wastewater treatment plants in the area16,44. Fibers, although generally retained during wastewater treatment in more developed urban regions, can occur highly in areas where treatment infrastructure is insufficient68.
This lack of adequate infrastructure highlights the urgent need for investment and environmental management measures to address the presence of fibers and the broader problem of MP pollution in Amazon estuaries. This consideration underscores the importance of sustainable management policies and practices to preserve water quality in this region. It is important to reduce plastic pollution at its source to address the issue of MPs in river and tidal channel water70. This can involve initiatives such as improving waste management and recycling practices, eliminating single-use plastics, and promoting the use of environmentally friendly alternatives70.
Size of the MPs
Most MPs on the Ajuruteua Peninsula ranged in size from 1000 to 2000 µm (31.7%), as shown in Fig. 4. Notably, Furo Grande recorded the highest proportion, accounting for 38.0%, followed by Furo do Meio (30.3%) and Furo do Taici (27.0%). In addition to the size range mentioned, other relevant categories were 500 to 1000 µm and 2000 to 5000 µm, representing 26.2% and 19,8% of the distribution, respectively (Fig. 4). In the Changjiang River estuary, MPs with an average size of less than 1000 μm predominate in the region’s surface waters71. The size range of MPs plays a crucial role in assessing the potential impact of these contaminants on the biota of an ecosystem72. The prevalence of smaller particles intensifies the risks associated with the frequency of interactions, highlighting the importance of considering not only the presence but also the specific characteristics of particles when assessing the impact of MPs on ecosystem biota73. Studies in the Amazon region have shown that MPs smaller than 5 mm were the most commonly found and most likely to be ingested by fish. In addition, the order of length of the MPs accumulated in different fish gastrointestinal contents in the region was predominantly in the stomach (92.1%) compared to the intestines (7.9%)29. These differences in the distribution of MPs, considering their specific sizes, highlight the need for a more comprehensive assessment of the impact of these contaminants on local ecology.
Polymer composition of the MPs
The results obtained using the FTIR technique revealed the presence of the polymers CPE and HDPE in the samples analyzed (Fig. 5a). The Raman technique revealed the presence of LDPE (Fig. 5b), a polymer often used in the manufacture of plastic bags and films. This identification is consistent with the widespread use of these materials in the production of packaging, household utensils, and various everyday plastic products74.
CPE is highly versatile due to its resistance to high temperatures, chemicals, and weather and its flame-retardant properties, making it ideal for coatings, waterproof membranes, pipes, wires, cables, and rubber products75. However, CPE’s non-biodegradable nature poses environmental challenges, as it persists and breaks down into MPs over time, contributing to pollution76,77,78,79.
LDPE and HDPE are widely produced due to their distinct properties. LDPE is more flexible and used in films and packaging, while HDPE is more rigid and suitable for pipes and containers80,81. Both can release harmful additives, posing ecotoxicological risks79,82,83.
The presence of CPE, HDPE, and LDPE MPs in tidal channels raises concerns about their impacts on aquatic ecosystems due to their environmental persistence74. Combining FTIR and Raman techniques allows for better characterization of MPs, underscoring the need for complementary methods in future studies84. Understanding the composition and sources of MPs is essential for effective management and mitigation strategies to protect aquatic ecosystems.
Risk assessment of MPs
Pollutant Load Index values (PLIs) to assess ecological risk are presented for tidal channels along the Ajuruteua Peninsula (Fig. 6a). Ecological risk indices are currently being applied to assess the risk of MPs in various ecosystems, including mangroves85, coastal sediments86, atmosphere87 and soil88. Therefore, using the PLI, this study used the same model to assess the potential risk associated with the MP observed on the Ajuruteua Peninsula.
Pollution load index (PLI) of the MPs in the tidal channels in the dry season during the spring tide: (a) considering the sampling stations (A = stations S1, S4, and S7; B = stations S2, S5, and S8; and C = stations S3, S6, and S9), (b) for each sampling site (Furo do Taici, Furo do Meio, Furo Grande, and for the entire Peninsula) in 2022.
The results indicated that all the sampling stations had low levels of MP contamination, with PLI values ranging from 2.425 (Furo Grande) to 3.498 (Furo do Taici) (Fig. 6a,b). In general terms, the analysis of the PLIlocal revealed that the Ajuruteua Peninsula and the estuary areas had low pollutant loads, with an average PLI value of 2.772 (Fig. 6b), classified as "Hazard Level I" (< 10 on the hazard classification scale – Table S1). Despite a low pollutant load classification, MPs can significantly impact ecosystem health. This impact poses a danger to the biota of estuarine ecosystems, resulting in adverse effects such as obstruction of the digestive tract, reduced nutrient absorption, and release of toxic chemicals. Consequently, this can lead to a decrease in populations of key species and changes in the ecological dynamics of ecosystems in the medium and long term. Furthermore, MPs can act as vectors of other polluting substances, including heavy metals and persistent organic compounds, further increasing the risk to biota5,53,89,90. The presence of these contaminants can directly affect human health since many of the contaminated species are consumed by local communities that depend on these resources for their subsistence29,91.
Commercial activities, such as public markets, ice factories, fishing, and boat repair areas, among others, are the main sources of the continuous release of MPs to the environment92,93. Thus, monitoring these activities to minimize MP release is recommended to avoid an increase in PLI levels, which could have increasingly harmful impacts on the environment and the coastal population in the future.
Influence of estuaries on the spatial distribution of MPs
The spatial distribution of MPs in the Caeté River estuary and Taperaçu is shaped by interactions between natural and anthropogenic factors, such as hydrodynamic characteristics that affect transport and sedimentation94, tidal currents, salinity gradients, and estuarine morphology influence MP patterns95. Stations S1, S2, and S3 at Furo do Taici are located in the innermost portion of the peninsula and closest to the mainland. These stations are influenced by continental drainage, the Caeté River, and the negative impacts from the municipalities they pass, especially Bragança, due to the effluents discharged into the water bodies and local fishing activities. In contrast, stations S4, S5, and S6 at Furo do Meio are located in the middle portion of the peninsula. This region receives less anthropogenic influence and has less human activity than Furo do Taici. Stations S7, S8, and S9 at Furo Grande, located in the outermost portion of the peninsula, are farther from the mainland and closer to the beach. These stations experience greater influence from the salinity of the sea and human activities such as fishing and tourism. All locations are connected to the waters of the Caeté River to the east and the Taperaçu to the west of the peninsula. This connection reflects the hydrodynamics of sediment flow transported by the waters of the Caeté River and Taperaçu, whose characteristics and anthropogenic influences are distinct.
Urban centers near the estuary increase MP inputs through runoff, industrial discharges, and poor waste management43,90. In addition, the influence of freshwater results in a more substantial load of MP from continental sources43,90, evidenced at the sampling points closest to the Caeté River estuary in this study (Fig. S3). Similar patterns are observed in the Goiânia River estuary in Brazil, with higher MP values during the rainy season (1,400 items/m3) compared to the dry season (133 items/m3) due to increased freshwater flow90. Studies in Malaysia show a threefold MP increase after floods, linked to heavy rainfall and river discharge96.
The main pressure factors were identified on the Ajuruteua Peninsula, where the distribution of MPs is noticeably greater at the sampling stations that the Caeté River most influences: the innermost portions (Furo do Taici) and at the stations along the Caeté estuary (S1, S4, and S7). Station S6, directly influenced by Taperaçu, had a higher abundance than the others (S3 and S9). This relationship concerns the fishing activities near the sampling station, such as fishing weirs. Local hydrodynamics and human activities are crucial in MP distribution, particularly in river areas with frequent human interaction. These findings enrich and support the better understanding of each study area since each one has its dynamics, making this multiparametric approach much more realistic and essential for developing effective management strategies to reduce MP pollution in estuarine environments97.
Conclusion
This is the first investigation into the presence and abundance of MPs in the surface waters of the tidal channels on the Ajuruteua Peninsula, considering the influence of the Caeté River estuary and Taperaçu as potential sources of MPs in the mangrove region, providing a more complete picture of the pollutant load and associated hazard risk. An increase in MP levels was observed towards the mouth of the river, a phenomenon influenced by the different characteristics of the collection stations and the surrounding human activities. When applying the Pollution Level Index (PLI) to assess ecological risk, we found that the region falls into the "Hazard Level I" classification, indicating that MP contamination is in the lower range of pollution. It is worth noting that runoff from urban areas significantly impacts the accumulation and transport of MP from the interior to the estuarine region of the peninsula. Given this scenario, the proper management of plastic waste emerges as a crucial measure to mitigate MP emissions into the environment, helping to keep the region within the lower pollution range. These findings highlight the urgency of more extensive research into MP pollution in the Brazilian Amazon’s fresh waters and estuarine ecosystems. A thorough understanding of this scenario is vital for implementing effective conservation and environmental management strategies in this coastal region, ensuring biodiversity preservation and sustainability of water resources.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
da Silva Montes, C. et al. Investigating spatial-temporal contamination for two environments of the Amazon estuary: A multivariate approach. Mar Environ Res 185, 105883 (2023).
Li, B. et al. Dynamic characteristics of microplastics under tidal influence and potential indirect monitoring methods. Science of the Total Environment 869, 161869 (2023).
Bhavan, S. G. et al. Seen from the Stakeholder’s revelation—Valuation of ecosystem services in a small tropical Indian estuary. Reg Stud Mar Sci 62, 102905 (2023).
Chen, L. et al. Spatiotemporal dynamics of coastal wetlands and reclamation in the Yangtze Estuary During Past 50 Years (1960s–2015). Chin Geogr Sci 28, 386–399 (2018).
Nishitha, D. S. et al. Study of trace metal contamination and ecological risk assessment in the sediments of a tropical river estuary, Southwestern India. Environ Monit Assess 194, 1–15 (2022).
Wang, Z. B. et al. Human impacts on morphodynamic thresholds in estuarine systems. Cont Shelf Res 111, 174–183 (2015).
Hale, R. C., Seeley, M. E., La Guardia, M. J., Mai, L. & Zeng, E. Y. A Global perspective on microplastics. J Geophys Res Oceans 125 (2020).
Hale, R. C., Seeley, M. E., King, A. E. & Yu, L. H. Analytical Chemistry of Plastic Debris: Sampling, Methods, and Instrumentation. in 17–67 (Springer, Cham, 2022). https://doi.org/10.1007/978-3-030-78627-4_2.
Shen, M. et al. Are biodegradable plastics a promising solution to solve the global plastic pollution?. Environmental Pollution 263, 114469 (2020).
Kibria, M. G., Masuk, N. I., Safayet, R., Nguyen, H. Q. & Mourshed, M. Plastic waste: Challenges and opportunities to mitigate pollution and effective management. Int J Environ Res 17, 1–37 (2023).
Derraik, J. G. B. The pollution of the marine environment by plastic debris: A review. Mar Pollut Bull 44, 842–852 (2002).
Prata, J. C., da Costa, J. P., Duarte, A. C. & Rocha-Santos, T. Methods for sampling and detection of microplastics in water and sediment: A critical review. TrAC Trends in Analytical Chemistry 110, 150–159 (2019).
Thompson, R. C. Microplastics in the marine environment: Sources, consequences and solutions. Marine Anthropogenic Litter (2015) https://doi.org/10.1007/978-3-319-16510-3_7/FIGURES/6.
Chau, H. S. et al. Microplastic occurrence and ecological risk assessment in the eight outlets of the Pearl River Estuary, a new insight into the riverine microplastic input to the northern South China Sea. Mar Pollut Bull 189, 114719 (2023).
Jambeck, J. R. et al. Plastic waste inputs from land into the ocean. Science 1979(347), 768–771 (2015).
Rico, A. et al. Large-scale monitoring and risk assessment of microplastics in the Amazon River. Water Res 232, 119707 (2023).
Rose, D. & Webber, M. Characterization of microplastics in the surface waters of Kingston Harbour. Science of the Total Environment 664, 753–760 (2019).
Wang, G. et al. Seasonal variation and risk assessment of microplastics in surface water of the Manas River Basin, China. Ecotoxicol Environ Saf 208, 111477 (2021).
Xu, P. et al. Microplastic risk assessment in surface waters: A case study in the Changjiang Estuary, China. Mar Pollut Bull 133, 647–654 (2018).
Firdaus, M., Trihadiningrum, Y. & Lestari, P. Microplastic pollution in the sediment of Jagir Estuary, Surabaya City, Indonesia. Mar Pollut Bull 150, 110790 (2020).
Leads, R. R. et al. Spatial and temporal variability of microplastic abundance in estuarine intertidal sediments: Implications for sampling frequency. Science of the Total Environment 859, 160308 (2023).
Radhakrishnan, K. et al. Characterization and distribution of microplastics in estuarine surface sediments, Kayamkulam estuary, southwest coast of India. Mar Pollut Bull 168, 112389 (2021).
Sajad, S., Allam, B. K., Mushtaq, Z. & Banerjee, S. Microplastic detection and analysis from water and sediment: A review. Macromol Symp 407, 2100367 (2023).
Paes, E. da S. et al. Widespread microplastic pollution in mangrove soils of Todos os Santos Bay, Northern Brazil. Environ Res 210, 112952 (2022).
Duan, J. et al. How mangrove plants affect microplastic distribution in sediments of coastal wetlands: Case study in Shenzhen Bay, South China. Science of the Total Environment 767, 144695 (2021).
Karami, A. et al. The presence of microplastics in commercial salts from different countries. Scientific Reports 7, 1–11 (2017).
Kosuth, M., Mason, S. A. & Wattenberg, E. V. Anthropogenic contamination of tap water, beer, and sea salt. PLoS ONE 13, e0194970 (2018).
Nakat, Z., Dgheim, N., Ballout, J. & Bou-Mitri, C. Occurrence and exposure to microplastics in salt for human consumption, present on the Lebanese market. Food Control 145 (2023).
Pegado, T. de S. e. S. et al. First evidence of microplastic ingestion by fishes from the Amazon River estuary. Mar Pollut Bull 133, 814–821 (2018).
Rodrigues, J. M. S. et al. First evidence of microplastics in commercial mussels from Amazonian estuaries. Reg Stud Mar Sci 70, 103379 (2024).
Pantoja, J. C. D. et al. First register of microplastic contamination in oysters (Crassostrea gasar) farmed in Amazonian estuaries. Mar Pollut Bull 201, 116182 (2024).
Prata, J. C. & Dias-Pereira, P. Microplastics in terrestrial domestic animals and human health: Implications for food security and food safety and their role as sentinels. Animals 13, 661 (2023).
Amato-Lourenço, L. F. et al. Presence of airborne microplastics in human lung tissue. J Hazard Mater 416, 126124 (2021).
Pérez-Guevara, F., Roy, P. D., Kutralam-Muniasamy, G. & Shruti, V. C. A central role for fecal matter in the transport of microplastics: An updated analysis of new findings and persisting questions. Journal of Hazardous Materials Advances 4, 100021 (2021).
Ragusa, A. et al. Raman microspectroscopy detection and characterisation of microplastics in human breastmilk. Polymers (Basel) 14, (2022).
Yang, Y. et al. Detection of various microplastics in patients undergoing cardiac surgery. Environ Sci Technol 57, 10911–10918 (2023).
Mendes, D. S., Beasley, C. R., Silva, D. N. N. & Fernandes, M. E. B. Microplastic in mangroves: A worldwide review of contamination in biotic and abiotic matrices. Mar Pollut Bull 195, 115552 (2023).
Rajan, K., Khudsar, F. A. & Kumar, R. Urbanization and population resources affect microplastic concentration in surface water of the River Ganga. Journal of Hazardous Materials Advances 11, 100342 (2023).
da Costa, I. D., Costa, L. L. & Zalmon, I. R. Microplastics in water from the confluence of tropical rivers: Overall review and a case study in Paraiba do Sul River basin. Chemosphere 338, 139493 (2023).
Ye, Y. et al. Pollution characteristics and ecological risk of microplastic in sediments of Liaodong Bay from the northern Bohai Sea in China. Mar Pollut Bull 187, 114505 (2023).
Pan, Z. et al. Microplastic pollution and ecological risk assessment in an estuarine environment: The Dongshan Bay of China. Chemosphere 262, 127876 (2021).
Mendes, D. S. et al. Comprehensive risk assessment of microplastics in tidal channel sediments in amazonian mangroves (northern Brazil). J Environ Manage 366, 121826 (2024).
Lima, A. R. A., Barletta, M. & Costa, M. F. Seasonal distribution and interactions between plankton and microplastics in a tropical estuary. Estuar Coast Shelf Sci 165, 213–225 (2015).
Queiroz, A. F. dos S. et al. First assessment of microplastic and artificial microfiber contamination in surface waters of the Amazon Continental Shelf. Science of the Total Environment 839, 156259 (2022).
Trindade, L. dos S. et al. Microplastics in surface waters of tropical estuaries around a densely populated Brazilian bay. Environmental Pollution 323, 121224 (2023).
Gomes, V. J. C., Freitas, P. T. A. & Asp, N. E. Dynamics and seasonality of the middle sector of a macrotidal estuary. Journal of Coastal Research https://doi.org/10.2112/SI65-193.1 (2013).
Moraes, B. C. et al. Variação espacial e temporal da precipitação no Estado do Pará. Acta Amazon 35, 207–214 (2005).
Asp, N. E., de Freitas, P. T. A., Gomes, V. J. C. & Gomes, J. D. Hydrodynamic overview and seasonal variation of estuaries at the eastern sector of the Amazonian coast. Journal of Coastal Research https://doi.org/10.2112/SI65-185.1 (2013).
Souza-Filho, P. W. M., Lessa, G. C., Cohen, M. C. L., Costa, F. R. & Lara, R. J. The subsiding macrotidal barrier estuarine system of the Eastern Amazon Coast, Northern Brazil. Lecture Notes in Earth Sciences 107, 347–375 (2009).
Pereira, L. C. C., de Oliveira, S. M. O., da Costa, R. M., da Costa, K. G. & Vila-Concejo, A. What happens on an equatorial beach on the Amazon coast when La Niña occurs during the rainy season?. Estuar Coast Shelf Sci 135, 116–127 (2013).
Virgulino, P. C. C., Carneiro, D. N., Nascimento, W. R., Cougo, M. F. & Fernandes, M. E. B. Biomass and carbon estimation for scrub mangrove forests and examination of their allometric associated uncertainties. PLoS ONE 15, e0230008 (2020).
Peng, G. et al. Microplastics in sediments of the Changjiang Estuary, China. Environmental Pollution 225, 283–290 (2017).
Tomlinson, D. L., Wilson, J. G., Harris, C. R. & Jeffrey, D. W. Problems in the assessment of heavy-metal levels in estuaries and the formation of a pollution index. Helgoländer Meeresuntersuchungen 33, 566–575 (1980).
Gago, J., Filgueiras, A., Pedrotti, M. L., Caetano, M. & Frias, J. Standardised protocol for monitoring microplastics in seawater. Deliverable 4.1 (2019) https://doi.org/10.25607/OBP-605.
Xiong, X., Wu, C., Elser, J. J., Mei, Z. & Hao, Y. Occurrence and fate of microplastic debris in middle and lower reaches of the Yangtze River – From inland to the sea. Science of the Total Environment 659, 66–73 (2019).
Zhao, S., Zhu, L., Wang, T. & Li, D. Suspended microplastics in the surface water of the Yangtze Estuary System, China: First observations on occurrence, distribution. Mar Pollut Bull 86, 562–568 (2014).
Zhao, S. et al. Analysis of suspended microplastics in the Changjiang Estuary: Implications for riverine plastic load to the ocean. Water Res 161, 560–569 (2019).
Hidalgo-Ruz, V., Gutow, L., Thompson, R. C. & Thiel, M. Microplastics in the marine environment: a review of the methods used for identification and quantification. Environ Sci Technol 46, 3060–3075 (2012).
Andrady, A. L. Microplastics in the marine environment. Mar Pollut Bull 62, 1596–1605 (2011).
Tsering, T., Sillanpää, M., Viitala, M. & Reinikainen, S. P. Variation of microplastics in the shore sediment of high-altitude lakes of the Indian Himalaya using different pretreatment methods. Science of the Total Environment 849, 157870 (2022).
Tian, L., Jinjin, C., Ji, R., Ma, Y. & Yu, X. Microplastics in agricultural soils: sources, effects, and their fate. Curr Opin Environ Sci Health 25, 100311 (2022).
Park, J., Hong, S., Shim, W. J., Khim, J. S. & Park, J. Distribution, compositional characteristics, and historical pollution records of microplastics in tidal flats of South Korea. Mar Pollut Bull 189, 114741 (2023).
Govender, J. et al. Towards Characterising Microplastic Abundance, Typology and Retention in Mangrove-Dominated Estuaries. Water 2020, Vol. 12, Page 2802 12, 2802 (2020).
Jiao, M. et al. Mangrove forest: An important coastal ecosystem to intercept river microplastics. Environ Res 210, 112939 (2022).
Kannankai, M. P. et al. Urban mangrove ecosystems are under severe threat from microplastic pollution: a case study from Mangalavanam, Kerala, India. Environmental Science and Pollution Research 1–13 (2022). https://doi.org/10.1007/S11356-022-21530-1/FIGURES/8.
Sutton, R. et al. Microplastic contamination in the San Francisco Bay, California, USA. Mar Pollut Bull 109, 230–235 (2016).
Browne, M. A. et al. Accumulation of microplastic on shorelines woldwide: Sources and sinks. Environ Sci Technol 45, 9175–9179 (2011).
Schell, T. et al. Effects of polyester fibers and car tire particles on freshwater invertebrates. Environ Toxicol Chem 41, 1555 (2022).
De Falco, F., Di Pace, E., Cocca, M. & Avella, M. The contribution of washing processes of synthetic clothes to microplastic pollution. Scientific Reports 9, 1–11 (2019).
Montagner, C., Dias, M., Paiva, E. & Vidal, C. Microplásticos: Ocorrência ambiental e desafios analíticos. Quim Nova https://doi.org/10.21577/0100-4042.20170791 (2021).
Li, Y. et al. The fate of microplastic pollution in the Changjiang River estuary: A review. J Clean Prod 425, 138970 (2023).
Mohamed Nor, N. H. & Obbard, J. P. Microplastics in Singapore’s coastal mangrove ecosystems. Mar Pollut Bull 79, 278–283 (2014).
Li, T. et al. Environmental fate of microplastics in an urban river: Spatial distribution and seasonal variation. Environmental Pollution 322, 121227 (2023).
Ivleva, N. P. Chemical analysis of microplastics and nanoplastics: Challenges, advanced methods, and perspectives. Chem Rev 121, 11886–11936 (2021).
Jagadeesh, P. et al. Sustainable recycling technologies for thermoplastic polymers and their composites: A review of the state of the art. Polym Compos 43, 5831–5862 (2022).
Ahmed, K. Eco-thermoplastic elastomer blends developed by compatibilizing chlorinated polyethylene into industrial-waste-filled polypropylene/acrylonitrile butadiene rubber system. Arab J Sci Eng 40, 2929–2936 (2015).
Ali, N. et al. Insight into microplastics in the aquatic ecosystem: Properties, sources, threats and mitigation strategies. Science of the Total Environment 913, 169489 (2024).
Zhang, L. et al. Tidal variation shaped microplastic enrichment patterns in mangrove blue carbon ecosystem of northern Beibu Gulf, China. Front Mar Sci 9, 927884 (2022).
Chen, H. et al. Physicochemical properties of environmental media can affect the adsorption of arsenic (As) by microplastics. Environmental Pollution 338, 122592 (2023).
da Silva, D. J. & Wiebeck, H. Predicting LDPE/HDPE blend composition by CARS-PLS regression and confocal Raman spectroscopy. Polímeros 29, e2019010 (2019).
Al-Salem, S. M., Lettieri, P. & Baeyens, J. The valorization of plastic solid waste (PSW) by primary to quaternary routes: From re-use to energy and chemicals. Prog Energy Combust Sci 36, 103–129 (2010).
Chen, Y. et al. Identification and quantification of microplastics using Fourier-transform infrared spectroscopy: Current status and future prospects. Curr Opin Environ Sci Health 18, 14–19 (2020).
Senathirajah, K., Kandaiah, R., Panneerselvan, L., Sathish, C. I. & Palanisami, T. Fate and transformation of microplastics due to electrocoagulation treatment: Impacts of polymer type and shape. Environmental Pollution 334, 122159 (2023).
Käppler, A. et al. Analysis of environmental microplastics by vibrational microspectroscopy: FTIR, Raman or both?. Anal Bioanal Chem 408, 8377–8391 (2016).
Li, R., Yu, L., Chai, M., Wu, H. & Zhu, X. The distribution, characteristics and ecological risks of microplastics in the mangroves of Southern China. Science of the Total Environment 708, 135025 (2020).
Ranjani, M. et al. Assessment of potential ecological risk of microplastics in the coastal sediments of India: A meta-analysis. Mar Pollut Bull 163, 111969 (2021).
Liu, K., Wang, X., Wei, N., Song, Z. & Li, D. Accurate quantification and transport estimation of suspended atmospheric microplastics in megacities: Implications for human health. Environ Int 132, 105127 (2019).
Jacques, O. & Prosser, R. S. A probabilistic risk assessment of microplastics in soil ecosystems. Science of the Total Environment 757, 143987 (2021).
Brennecke, D., Duarte, B., Paiva, F., Caçador, I. & Canning-Clode, J. Microplastics as vector for heavy metal contamination from the marine environment. Estuar Coast Shelf Sci 178, 189–195 (2016).
Barletta, M., Lima, A. R. A. & Costa, M. F. Distribution, sources and consequences of nutrients, persistent organic pollutants, metals and microplastics in South American estuaries. Science of the Total Environment 651, 1199–1218 (2019).
Pegado, T. et al. Ingestion of microplastics by Hypanus guttatus stingrays in the Western Atlantic Ocean (Brazilian Amazon Coast). Mar Pollut Bull 162, 111799 (2021).
Deng, H. et al. Microplastics pollution in mangrove ecosystems: A critical review of current knowledge and future directions. Science of the Total Environment 753, 142041 (2021).
Deng, J. et al. Microplastics and accumulated heavy metals in restored mangrove wetland surface sediments at Jinjiang Estuary (Fujian, China). Mar Pollut Bull 159, 111482 (2020).
Chen, L. et al. Characteristics and spatiotemporal distribution of microplastics in sediments from a typical mariculture pond area in Qingduizi Bay, North Yellow Sea, China. Mar Pollut Bull 176, 113436 (2022).
Gomes, V. J. C. et al. Suspended-sediment distribution patterns in tide-dominated estuaries on the eastern amazon coast: Geomorphic controls of turbidity-maxima formation. Water (Switzerland) 13, 1568 (2021).
Chen, H. L., Gibbins, C. N., Selvam, S. B. & Ting, K. N. Spatio-temporal variation of microplastic along a rural to urban transition in a tropical river. Environmental Pollution 289, 117895 (2021).
Liu, Z., Huang, Q., Wang, H. & Zhang, S. An enhanced risk assessment framework for microplastics occurring in the Westerscheldt estuary. Science of the Total Environment 817, 153006 (2022).
Acknowledgements
The authors would like to thank the Coordination for the Improvement of Higher Education Personnel (CAPES—process no. 88882.444924/2019-01) for awarding the doctoral scholarship and the Rufford Foundation for the grant (30854-1) that supported the fieldwork carried out during the doctoral thesis. Special thanks are extended to the Porous Media Science and Engineering Research Group (GCEMP) of the Federal University of Pará (UFPA), Salinópolis Campus, for providing the necessary facilities to quantify the samples’ particles. In addition, the authors would like to thank the support of the Analytical Center (Central Analítica) of the Chemistry Institute of the Federal University of Rio Grande do Norte (UFRN), where the FTIR and Raman spectroscopy analyses were carried out, and the project Mangues da Amazônia (Programa Petrobras Socioambiental) for supporting the research.
Author information
Authors and Affiliations
Contributions
D.S.M., M.E.B.F. conceptualized the study and planned for the study sites. D.S.M. collected the samples from the sites. D.S.M., D.N.N.S. processed the samples. D.S.M., D.N.N.S., M.G.S. performed the analyses. D.S.M., M.E.B.F., C.R.B. wrote the manuscript. D.S.M., M.E.B.F., C.R.B. contributed to the writing, editing, and revising of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mendes, D.S., Silva, D.N.N., Silva, M.G. et al. Microplastic distribution and risk assessment in estuarine systems influenced by traditional villages and artisanal fishery activities. Sci Rep 14, 29044 (2024). https://doi.org/10.1038/s41598-024-80468-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-80468-1