Abstract
Flexible conductive hydrogel strain sensors are gaining popularity due to their exceptional stretchability, sensitivity, and potential for wearable devices. However, their widespread use is hindered by significant issues, such as poor electrical conductivity and weak response time. To address these challenges, new hydrogels based on guar gum, borax, and glycerol have been fabricated via a green synthesis technique. These hydrogels were reinforced with functionalised multiwalled carbon nanotubes (f-MWCNTs) and silver nanoparticle decorated multiwalled carbon nanotubes (AgNP-MWCNTs). The resulting conductive hydrogels exhibited a self-healing capability of 83.2% and effective strain sensing with a gauge factor of 6.58. The incorporation of AgNP-MWCNTs significantly improved the electrical conductivity up to 3.05 ± 0.02 S m− 1, thanks to the tunnelling effect between f-MWCNTs and the synergic interaction of AgNP-MWCNTs. Moreover, the hydrogel sensors displayed excellent durability, enduring 3000 cycles of tensile loading and unloading at 50% strain. This innovative use of green design principles offers a straightforward, cost-effective, and environmentally friendly process for producing high-performance soft materials. These materials hold significant promise for various practical applications, including artificial skins, flexible electronics, and healthcare monitoring, highlighting the high relevance and impact of this research.
Similar content being viewed by others
Introduction
Flexible and wearable electronics have recently received plenty of interest due to their versatile applications across various engineering and industrial fields, such as wearable devices1,2, energy storage3,4, artificial skin5,6, touch panels7,8, and motion-detecting sensors9,10. Among these materials, strain sensors stand out for their ability to respond to external stimuli with electrical impulses, making them vital for developing electronic skin. Their growing usage also holds great potential in improving human-machine interfaces11, temperature detection12, health monitoring13, motion detection14, humidity monitoring15, and remote health management16,17. These devices can now be manufactured using diverse materials, for instance, carbon-based materials, conductive polymers, and metal nanowires, which are integrated with elastomeric substrates18,19,20.
Hydrogel-based strain sensors are promising materials for sensing applications, offering compatibility with bio-tissues while eliminating the requirement for elastomers in water-containing systems21. These soft, water-absorbing hydrogels can be made from natural materials, such as chitosan22, agarose23,24, sodium alginate25, cellulose26, and guar gum27. Guar gum, derived from guar seeds, offers outstanding characteristics, such as water-solubility, high molecular weight, and low cost. Its abundance of side chains enhances mechanical strength through enhanced entanglement and intramolecular crosslinks, making it a potential candidate for smart wearable hydrogels28. Wang et al.29 have developed a novel self-healing polysaccharide hydrogel using cationic guar gum and CuCl2. The CG-Cu hydrogels exhibited rapid gelation, excellent mechanical strength, biocompatibility, and antibacterial properties. In vivo tests confirmed complete degradation in 21 days, aiding skin regeneration and infection prevention. Moreover, Maroufi et al.30 have synthesised a chitosan (CS) and dialdehyde guar gum (DAGG) hydrogel via Schiff base reaction, achieving 12,000% swelling capacity with a 30:70 CS ratio. This hydrogel exhibited pH-independent swelling, porous structure, improved strength, and strong antioxidant and antimicrobial properties when loaded with 5% pomegranate peel extract.
Sodium tetraborate, or borax (Na2B4O7·10H2O), plays a vital role in the physical and chemical crosslinks of the hydrogel structures through their hydroxyl functional groups31. Tanpichai et al.32 developed eco-friendly hydrogels from water hyacinth cellulose using sodium hydroxide, urea, and borax to form crosslinks. The crosslinked hydrogels achieved a swelling ratio of ~ 900%, compared to 325% in uncrosslinked versions. Borax improved superabsorbency, increased gel fraction, delayed flame propagation, and provided antibacterial activity. These hydrogels showed potential for wound dressings, agriculture, and flame-retardant coatings. To improve hydrogel durability under severe environmental conditions, glycerol as a nontoxic anti-freezing agent can prevent water evaporation at high temperatures and ice formation at low temperatures33. Also, glycerol can form strong hydrogen bonds with water molecules, competing with the hydrogen bonds found in water34. Liu et al.35 developed high-performance PGSC ionic hydrogel sensors with dual crosslinked networks. Glycerol crosslinked PVA formed the framework, while ionic crosslinking between sodium alginate and Ca2+ served as sacrificial bonds for energy dissipation. The hydrogel demonstrated excellent mechanical properties, conductivity, self-recovery, and sensitivity, making it suitable for long-term monitoring of physiological signals in extreme conditions.
Hydrogels face significant challenges, such as lack of inherent conductivity, poor mechanical properties, and sensitivity, which are crucial factors for tracking human movement and detecting minute motions or physiologic signals. To address these issues, nanocomposite conductive hydrogels have been developed. By integrating nanomaterials, such as functionalised multi-walled carbon nanotubes (f-MWCNTs), into the hydrogel matrix, both Young’s modulus and tensile strength are improved36,37. These hydrogels also enhance sensitivity, as stretching induces resistance and the tunnelling effect38,39. However, to fully benefit from nanofillers, these materials must be dispersed uniformly in high concentrations, overcoming percolation barriers to form conductive channels40,41.
Silver nanoparticles (AgNPs) are valuable conductive materials that can improve the sensing characteristics of hydrogels. These fillers possess exceptional optical, electrical, and magnetic properties and a high surface-to-volume ratio that contributes to quantum effects42. Besides, AgNPs have been fabricated using the green synthesis method using bio-reductive materials or non-toxic plant extracts. However, AgNPs have suffered morphological flaws, such as agglomeration, resulting in low efficiency43. Also, due to their high surface energy, these materials are reactive and sensitive to oxidation, leading to aggregation into larger particles and loss of their desired characteristics. While incorporating AgNPs into polymer nanocomposite systems can enhance some properties, it may also result in metal leaching and further aggregation44. To overcome these difficulties, a nanoconfinement method has been proposed, which restricts AgNPs around MWCNTs. This approach lessens metal aggregation and leaching within the hydrogel matrix, improving overall performance45.
Self-healing refers to the intelligent ability of materials to autonomously repair themselves after physical damage, either without human intervention or in response to external stimuli46. The self-healing materials possess excellent mechanical and electrical healing capabilities, high toughness, good stretchability, and environmental stability, making them suitable for various engineering applications47. Notably, self-healable polymers present intriguing frameworks for repairing their structures, utilising approaches such as capsule-based healing, vascular healing, and intrinsic healing, depending on the microscopic structures and specific purposes48. Recently, Rammal et al.49 have developed hydrogels with self-healing characteristics, enhancing the boundaries of functionalisation. These materials are exploited particularly in tissue engineering and medical applications due to their biocompatibility and sensing devices.
To advance a field of smart sensing applications, a new nanocomposite conductive hydrogel with self-healing characteristic has been developed using guar gum, borax, and glycerol. The hydrogels were separately incorporated by AgNPs-MWCNTs and f-MWCNTs with different weight contents. The AgNPs decorated on the surface of MWCNTs (AgNPs-MWCNTs) through a green synthesis technology using Withania coagulans seed extract. Comprehensive characterisations of physical, mechanical, electrical, self-healing properties and sensitivity of the composite hydrogels were conducted. These hydrogel materials are poised to serve as smart, eco-friendly devices for human motion detection and health monitoring.
Materials, preparations, and characterisations
Materials
Guar gum (viscosity of 3500–4000 CPs) and borax (Na2B4O7·10H2O, > 99.5% purity, Mw of 381.37 g/mol) were obtained from Chemipan Corporation Co., Ltd (Bangkok, Thailand). Glycerol (1,2,3-propanetriol, > 99% purity, Mw of 92.09 g/mol) was purchased from Thermo Fisher Scientific (Thailand). Withania coagulans seeds were obtained from Organic Herbs (Thailand). Multi-walled carbon nanotubes (MWCNTs) (99% purity, 12.9 nm diameter) were obtained from Nanogen, Thailand. Silver nitrate (AgNO3, > 99.9% purity) was purchased from Fisher Chemicals, Thailand. Nitric acid (HNO3) 70% and sulfuric acid (H2SO4) 98% were purchased from RCI Labscan Co., Ltd., Thailand. All chemicals are analytical grade. Distilled water was used throughout the experiment.
Materials preparations
Acidic functionalisation of MWCNTs
300 mg pristine MWCNTs (p-MWCNTs) were mixed with 20 mL of 15.8 M HNO3 and 60 mL of 18.4M H2SO4, according to the previous reported50. The mixture was sonicated for 3 h in an ultrasonic bath at a temperature range of 40–50 °C. After sonication, the mixture was washed with distilled water until the pH reached neutral. Finally, the functionalised MWCNTs (f-MWCNTs) were vacuum-dried in an oven at 70 °C overnight.
Withania coagulans extraction
Dry Withania coagulans were milled into a 100-mesh size powder. Then, 5 g of dry fruit powder was mixed with 100 mL of distilled water and boiled at a temperature of 100 °C for 30 min. After boiling, the mixture was cooled down to room temperature and filtered by a vacuum filter using a Whatman filter paper to obtain the fruit extract solution. The solution was then stored at 4 °C for further use.
Green synthesis of silver nanoparticles (AgNPs)
Silver nanoparticles (AgNPs) were prepared by adding 2 mL Withania coagulans extract solution to 1 mL of 0.1 M AgNO3 solution. The mixture was constantly stirred on a hot plate at 80 °C for 24 h under UV light. The formation of AgNPs was observed by a colour change from light yellow to dark brown.
Green synthesis of silver nanoparticles decorated multi-wall carbon nanotubes (AgNP-MWCNTs) nanocomposites
To synthesise AgNP-MWCNTs, 20 mg f-MWCNTs were added to 50 mL of distilled water and sonicated in an ultrasonic bath until homogenously dispersed. Then, 20 mL of f-MWCNTs mixture and 10 mL of 0.1 M AgNO3 were mixed and stirred for 1 h at a room temperature. Next, 20 mL of Withania coagulans extract solution, serving as a reducing and stabilising agent, was gradually added by a burette. The mixture was constantly stirred for 24 h under UV light at 80 °C. The formation of AgNPs on MWCNTs surface was confirmed by a distinct colour change from black to brown-reddish.
Preparation of guar gum/glycerol/borax hydrogel
First, a mixture was prepared by dissolving 6 mL of glycerol in 30 mL of distilled water. Next, 900 mg of guar gum powder was added to the mixture while stirring. Second, a 20wt% borax solution was prepared by dissolving borax salt in distilled water and gradually adding it to the guar gum solution until the hydrogel was fully formed. The resulting hydrogel was placed into a mould and dried in the oven at 50 °C for 48 h.
Preparation of the hydrogel filled with f-MWCNTs and the hydrogel filled with AgNP-MWCNTs
To prepare the hydrogel composites with f-MWCNTs, 0.3wt%, 0.6wt%, 0.9wt%, and 1.2wt% of f-MWCNTs were separately added in 30 mL of distilled water and sonicated for 1 h to achieve homogenous dispersion. Then, 6 mL of glycerol was added to the solution, which was kept sonicated until completely dissolved. Next, 900 mg of guar gum powder was added to the mixture. A 20wt% borax solution, prepared by dissolving borax salt in distilled water, was gradually added to the solution until the hydrogel was fully formed. The resulting hydrogel was then placed into a mould and dried in the oven at 50 °C for 48 h.
The synthesis process was repeated to prepare the samples of hydrogel filled with AgNP-MWCNTs, using identical weight contents to those of the f-MWCNTs.
In this work, the guar gum hydrogel refers for guar gum/ borax/ glycerol hydrogel nanocomposites, the hydrogel filled with f-MWCNTs is referred to as f-MWCNT-filled guar gum/ borax/ glycerol hydrogel, and the hydrogel filled with AgNP-MWCNTs is referred to as AgNP-MWCNT-filled guar gum/ borax/ glycerol hydrogel. The schematic of the materials preparation and characterisation is shown in Fig. 1.
Materials characterisations
The formation of AgNPs was confirmed by UV-visible spectroscopy analysis (UV-visible spectrophotometer Agilent 8453, United States). The samples were filled in a 3.5 mL quartz cuvette. The absorption was measured at a wavelength range of 200–800 nm. Distilled water was employed as a blank.
X-ray diffraction (XRD) patterns of the samples were measured using a (Bruker AXS D8 advance, Germany) operating at 40 kV and 40 mA with Cu Kα radiation (λ = 1.5406 Å). The diffraction pattern was captured over a 2θ range of 5° to 80°.
Field emission scanning electron microscopy (JEOL JSM-7610 F, Oxford X-Max 20, Japan) combined with energy-dispersive spectroscopy (EDS) mapping were used to assess the appearance and interface of the produced samples after air drying. The samples were coated with gold, and the magnification was set to ×100000 at an accelerating voltage of 5 kV.
FTIR spectra of p-MWCNTs, f-MWCNTs, AgNP-MWCNTs, guar gum hydrogel, hydrogel filled with f-MWCNTs, and hydrogel filled with AgNP-MWCNTs were measured using a Nicolet 380 FTIR spectrometer (ThermoElectron Instruments Co., Ltd., USA) in the frequency range of 4000 –500 cm− 1. For the hydrogel samples, the samples were freeze-dried and mixed with KBr to form an ultrafine powder before characterisation. The FTIR spectra of p-MWCNTs, f-MWCNTs, AgNP-MWCNTs were included in Figure S1 in the Supplementary Information.
The mechanical properties of the original and self-healed guar gum hydrogel, the hydrogel filled with f-MWCNTs and the hydrogel filled with AgNP-MWCNTs were evaluated using a universal testing machine (Instron, Model 5567) equipped with a 5-kN load cell, following the ASTM D 638 standard. The distance between the grips was initially set at 50 mm, and a crosshead speed of 50 mm/min was used. The tests were repeated with 3 samples.The swelling rate (SR) of the hydrogels was measured by a classic gravimetricmethod. First, the hydrogel was placed in an oven at 40 °C to remove the water. Then, the dried hydrogels were weighed (W0) and soaked in DI water at room temperature until the swelling equilibrium was reached. During this process, the swollen hydrogels were regularly taken out and weighed (Wt). The swelling rate was calculated according to Eq. 1:
The water absorption is a parameter that indicates the long-term stability of hydrogels. The as-prepared hydrogel was weighed and kept at room temperature until the water-retaining capacity reached equilibrium. The hydrogels were regularly taken out and weighed. The water-retention capacity can be calculated using Eq. 2:
Where Wt and Wo are the instantaneous weight and the original weight of the samples, respectively.
Conductive hydrogel samples with the dimensions of 50 × 12 × 1 mm3 (L × W × T) were used to record electrical signals. The signals were captured utilising a digital source device (Palmsens4, The Netherlands). The rate of change in relative resistance gauge factor (GF) was calculated using Eqs. 3 and 4:
Where Ro was the original resistance without strain and R was the real-time resistance under applied strain ε.
The electrical properties of guar gum hydrogel, hydrogel filled with f-MWCNTs, and hydrogel filled with AgNPs-MWCNTs were measured using a digital source device. The electrical conductivity is measured using Eq. 5:
Where L is the length of the hydrogel sample (cm), A is the area of the hydrogel sample (cm2), and R is the resistance of the hydrogel samples.
The self-healing performance of the guar gum hydrogel, the hydrogel filled with f-MWCNTs, and the hydrogel filled with AgNP-MWCNTs was assessed through visual observation, optical microscopic, LED indicator test, and weight holding test at room temperature. The samples were cut into two pieces and then reconnected without the addition of any chemical agents or heat.
Results and discussion
UV-visible spectroscopy
UV–visible absorbance spectra of p-MWCNTs and f-MWCNTs were measured between wavelengths of 200–800 nm, as shown in Fig. 2a. No absorbance peak was observed for p-MWCNTs, likely due to their large particle size, aggregation, and bundling. Even after sonication, the van der Waals forces within the bundles could not be overcome due to the lack of functional groups. In contrast, f-MWCNTs exhibited an absorbance peak at 260 nm, attributed to the n-π transition of nonbonding pairs of electrons in phenolic groups (O-H) and carboxylic groups (-COOH), confirming successful functionalisation51.
The role of Withania coagulans extract as a reducing and capping agent in the green synthesis of AgNPs was also investigated. As shown in Fig. 2b, the maximum absorbance at 470 nm confirmed the formation of AgNPs after exposure to Withania coagulans extract, indicated by the colour change of the AgNO3 solution from pale yellow to colloidal brown. A successful synthesis of AgNP-MWCNT nanocomposites was confirmed by the absorbance peak at 435 nm, attributed to the surface plasmon resonance (SPR) effect of AgNPs. The slight shift to a lower wavelength suggested the formation of smaller AgNPs on the MWCNT surface52.
X-ray diffraction (XRD)
The XRD patterns of p-MWCNTs, f-MWCNTs and green-synthesised AgNP-MWCNTs are shown in Fig. 3. The p-MWCNTs displayed two distinct peaks at 2θ = 25.75° and 42.98°, corresponding to the (002) and (100) planes, respectively. After acid treatment, the intensity of the (002) peak of f-MWCNTs increased, indicating the successful attachment of carboxylic acid groups to the surface. In addition, f-MWCNTs exhibited two diffraction peaks at 2θ = 25.70° and 42.96°, suggesting a high degree of graphitisation.
In the case of the AgNP-MWCNTs nanocomposites, four prominent diffraction peaks were observed at 2θ = 38.20o, 44.37o, 64.57o and 77.51°, corresponding to (111), (200), (220), and (311) planes of the face-cantered cubic (fcc) crystal structure of metallic Ag. The Bragg diffraction peak of AgNP-MWCNTs at 2θ = 25.72° was retained, while a tiny peak at 2θ = 42.96° diminished, likely due to a significant magnification of AgNPs. These XRD results indicated the crystalline structure of the nanocomposites, consisting of both Ag and MWCNTs. The average crystallite size of the AgNP-MWCNTs nanocomposites, calculated using the Debye-Scherer Equation, as shown in Eq. 6, was determined to be 33.56 nm.
Where K is the Bragg’s constant, which is between 0.94 and 1, β is the whole width at half maximum of several diffraction peaks, θ stands for Bragg’s diffraction angle, and λ is the wavelength of target Cu-Kα (1.5406 Å).
FESEM and EDX analyses
FESEM was utilised to investigate the potential fragmentation of p-MWCNT after acid treatment and to detect morphological changes in MWCNT samples. In Fig. 4a, the p-MWCNT sample exhibited the typical flakes and clusters of entangled tubes due to the robust Van der Waals forces between each tube. In contrast, the acid-functionalised f-MWCNT sample presented a rougher surface, attributed to defect sites caused by the adhered functional groups, as shown in Fig. 4b. Figure 4c,d depict AgNP-MWCNTs, clearly exhibiting a spherical shape of AgNPs consistently distributed on the surface. This distribution resulted from the electrostatic or weak van der Waals forces that securely hold AgNPs to the f-MWCNTs, though some aggregations were observed. Despite functionalisation and the introduction of AgNPs, no significant surface or shape distortion of MWCNTs was observed.
The chemical purity and elemental composition of green-produced AgNP-MWCNTs were further investigated using EDX spectroscopy. The EDX images of AgNP-MWCNTs and various elemental compositions are shown in Fig. 4e–h. Table S1 summarises the mass and elemental percentages of Ag, C, and O and compares them to results reported by Anshori et al.53. The results showed that the percentages in this work were similar to the reported results, confirming the successful synthesis of AgNP-MWCNT material. Figure 4i shows a map summary of all components, demonstrating the abundance of AgNPs scattered throughout the outer wall of the MWCNTs, as indicated by the high intensities of C and Ag relative to O.
FTIR analysis
The FTIR spectra of the guar gum hydrogel, the hydrogel filled with f-MWCNTs and the hydrogel filled with AgNP-MWCNTs are illustrated in Fig. 5. Across the various hydrogel samples, no appreciable attenuation or disappearance of spectral peaks were observed, indicating that the chemical compositions and bonding strengths of different formulations are similar. This consistency further contributed to the stability and durability of hydrogels. In both the guar gum hydrogel and hydrogel filled with AgNP-MWCNTs, the peak at 3261 cm− 1 belonged to the O-H stretching vibration of the guar gum polymer. Meanwhile, in the hydrogel filled with f-MWCNTs, this peak shifted slightly to 3258 cm− 1. The 2928 cm− 1 and 2873 cm− 1 peaks were attributed to the C-H stretching of sp3 hybridised carbon within guar gum. The 1648 cm− 1 and 1409 cm− 1 peaks indicated O-H bending and C-H bending in the hydrogel structure54, respectively. In addition, the peaks at 1031 cm− 1, 953 cm− 1, and 854 cm− 1 corresponded to the asymmetric stretching vibration of B-O-C, B-O, and C-O-C connections within the network. The crystallinity of a polymer could be further determined by analysing the spectral region between 700 cm− 1 and 500 cm− 155.
Mechanical properties
The mechanical properties of the guar gum hydrogel with different amounts of borax and the hydrogels filled with different amounts of f-MWCNTs and AgNP-MWCNTs were evaluated. For the guar gum hydrogels, the percentages of borax were varied with 10wt%, 20wt%, and 30wt%. As shown in Fig. 6a, the tensile strength and elongation at the break increased with borax content up to 20wt% due to enhanced crosslinking between guar gum and borax, which improved the structural toughness. However, at 30wt% borax, excessive crosslinking resulted in a reduction in these properties due to increased network stiffness.
The hydrogel nanocomposites with different f-MWCNT and AgNP-MWCNT contents of 0.3wt%, 0.6wt%, 0.9wt%, and 1.2wt% were also investigated. Figure 6b illustrates that increasing f-MWCNT contents proportionally enhanced tensile strength and elongation at the break, as rigid particles reinforced the hydrogel matrix. The optimum tensile strength of 0.21 ± 0.01 MPa and elongation at a break of 208.6 ± 10.4% were observed at 0.9 wt% of f-MWCNTs. However, at 1.2 wt%, these properties decreased due to reduced water content and particle aggregation. Similarly, Fig. 6c shows that an integration of AgNP-MWCNTs increased the mechanical strength of the hydrogel due to the large surface area facilitating enhanced connection pathways56. The nanocomposite hydrogel exhibited the maximum tensile strength of 0.17 ± 0.01 MPa and elongation at a break of 228.5 ± 11.4% at 0.9wt% of AgNPs-MWCNTs.
Swelling and water retention
The swelling rates of the guar gum hydrogel, the hydrogel filled with f-MWCNTs, and the hydrogel filled with AgNP-MWCNTs over 10 h were investigated, as displayed in Fig. 7a. All samples exhibited a rapid increase in swelling during the first hour and reached equilibrium after approximately 6 h. The final mass of the guar gum hydrogel was 3.6 times its initial mass, while the hydrogels filled with f-MWCNTs and AgNP-MWCNTs swelled to 2.3 times and 2.9 times their initial mass, respectively. The absence of conductive fillers in the guar gum hydrogel allowed for greater water penetration, leading to its higher swelling rate. The smaller particle size of AgNP-MWCNTs contributed to a more uniform dispersion across the hydrogel network, increasing the surface area and capillary forces and encouraging water absorption56.
The water retention capacity of these hydrogels over 10 days was also measured, as displayed in Fig. 7b. The retention capacity reduced gradually over time, with results ranging from 87.4 to 93.1% after 7 days. This retention was attributed to the presence of glycerol in hydrogels, which trapped water molecules and slowed down evaporation57. The guar gum hydrogel displayed the lowest water-retention capacity due to the presence of excessive free water in its network, leading to faster evaporation. However, the addition of f-MWCNTs and AgNP-MWCNTs increased denser hydrogel structures, serving as barrier layers that effectively reduced water evaporation from the network.
Relative resistance and gauge factor properties of hydrogel
Flexible hydrogel sensors possess the remarkable capacity to convert mechanical deformations into electrical signals, whether in the form of current or capacitance. When stretched or compressed, the hydrogels experience deformation in their internal conductive networks, leading to resistance or electrical conductivity changes. The guar gum hydrogel sensors displayed electrical conductivity owing to the presence of borate ions, which significantly improved the conductivity of the hydrogel. As shown in Fig. 8a, the guar gum hydrogel sensor containing 20wt% borax exhibited the highest sensitivity, with a gauge factor (GF) of 3.304 at a strain of 180% and a linearity (R2) of 0.9925. However, an increase in borax content up to 30wt% significantly lessened the sensitivity, attributed to the formation of uneven networks and aggregates of borax inside the hydrogel matrix.
Figure 8b shows that the hydrogel sensor filled with 0.9wt% f-MWCNTs exhibited the highest GF of 3.7 at a strain of 208.6% and the R2 of 0.9860. Other contents of f-MWCNTs filled in the hydrogel showed GFs in the range of 2.1 to 2.9, with R2 values between 0.9740 and 0.9931. At lower filler concentrations, the f-MWCNTs were uniformly distributed across the hydrogel network, creating effective conductive pathways and reducing the initial resistance by percolation theory58. However, at high filler concentrations, the sensitivity dropped due to the densification of the conductive network. The low-density conductive network was less susceptible to degradation under strain, leading to a more dramatic shift in electrical resistance. At lower strains, the f-MWCNTs established stable connections, but as stretching increased, the average distance between them widened, increasing electrical resistance due to the tunnelling effect and the contact resistance between f-MWCNTs59.
In Fig. 8c, the hydrogel sensor filled with 0.9wt% AgNP-MWCNTs exhibited the highest GF of 6.58 at a strain of 228.5% and the R2 of 0.9991. The inclusion of AgNP-MWCNTs at this percentage enhanced conductivity by enabling AgNPs to act as bridges between neighbouring MWCNTs, improving conductive pathways. However, adding 1.2wt% AgNP-MWCNTs resulted in a lower GF due to agglomeration within the hydrogel network. The R2 slightly increased, which might be attributed to the presence of a small amount of free carbon nanotubes in the hydrogel matrix60.
The real-time responses of the guar gum hydrogel strain sensors to various strains are shown in Figure S2(a-b). These strain sensors reliably and consistently detected accurate electrical signals under both minor strains of 1%, 5%, 10%, 15%, and 20%, as well as large strains of 30%, 60%, and 90%. As tensile strain increased, the sensors responded effectively, as indicated by the increasing ΔR/R0 values. Furthermore, this material exhibited remarkable durability and repeatability throughout 1500 stretching-releasing cycles, as depicted in Figure S2(c).
The strain-sensing capacities of the hydrogel sensor filled with 0.9 wt% f-MWCNTs were evaluated, as shown in Figure S3(a-b). The results showed that this material reliably detected different tensile strain levels. The consistency of the ΔR/R0 curves in Figure S3(c) indicates strong sensing repeatability. As strain increased, the resistance shifted more significantly, leading to higher ΔR/R0 values and a critical change in the conductive path. This material demonstrated excellent repeatability with minimal variation, suggesting strong anti-fatigue properties that could allow long-term usage.
Similarly, the hydrogel sensor filled with 0.9 wt% AgNP-MWCNTs indicated effective strain-sensing capacities, as shown in Figure S4(a-b). A higher applied strain caused a more significant change in resistance and a larger shift in the conductive path, raising the ΔR/R0 values. After 3000 tensile loading-unloading cycles at 50% strain, the sensor exhibited a steady ΔR/R0 response, as shown in Figure S4(c), demonstrating high durability and repeatability over the long-term stretching-releasing process.
Electrical conductivity of hydrogel nanocomposites
The electrical conductivity characteristics of the guar gum hydrogel, the hydrogel filled with f-MWCNTs, and the hydrogel filled with AgNP-MWCNTs were characterised. Figure 9a illustrates the effect of incorporating f-MWCNTs and AgNP-MWCNTs on their electrical conductivity of the hydrogels. The electrical conductivity of the guar gum hydrogel, with different amounts of borax, ranged from 0.74 ± 0.06 to 0.88 ± 0.05 S m− 1. Borax, a main component of hydrogels, serves as a transport ion source. The dissociation of Na+ and [B(OH)4]− ions considerably increased the conductivity of the hydrogel.
Incorporating f-MWCNTs further improved into the conductivity of the hydrogel, with values ranging from 1.14 ± 0.03 to 1.71 ± 0.04 S m− 1. This improvement can be attributed to the f-MWCNTs creating efficient conductive pathways within the hydrogel due to their uniform dispersion. However, at 0.9wt% f-MWCNTs, the free space of hydrogel became inadequate for further dispersion, resulting in agglomeration and a subsequent decrease in conductivity.
The hydrogel filled with AgNP-MWCNTs showed a significant increase in conductivity, ranging from 2.10 ± 0.04 to 3.05 ± 0.02 S m− 1. At higher concentrations, AgNP-MWCNTs formed a network structure that facilitated electron transport, resulting in a significant increase in conductivity. The electrical conductivity values of all hydrogel samples are summarised in Table S2.
Practical demonstration as the Wearable Motion Sensor
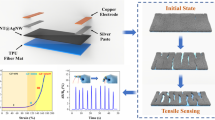
The AgNP-MWCNT hydrogel sensors were utilised as wearable sensors to monitor a range of human physiological activities. When physically attached to various body regions, these hydrogel sensors enable real-time detection of diverse human motions. Figure 9b illustrates the practical utilisation of hydrogel strain sensors for tracking human motions.
The AgNP-MWCNT strain sensors were used to monitor finger movements, as shown in Fig. 9c. The relative resistance rose with each successive finger bending angle of 30°, 60°, and 90°. The result showed that the relative resistance increased in response to slight increases in the bending angle of the fingers of the demonstrator. In addition, the hydrogel sensor was affixed to the wrist to detect up-and-down movements, exhibiting exceptional electrical stability and sensing capabilities. The resistance of the sensor varied with wrist movement, providing prompt and reliable responses, as shown in Fig. 9d.
Furthermore, the hydrogel could detect subtle movement associated with word pronunciation. Figure 9e,f demonstrate that this hydrogel sensor exhibited significant changes in relative resistance signals when pronouncing the words “Apple” and “Laptop”, respectively. The ability to detect movement and facial expressions is crucial for the future development of non-invasive diagnosis technologies61. Figure 9g, the application of the developed material for attachment to various movable body parts is presented. The hydrogel sensors adhere directly to the skin, causing no irritation.
Plot of (a) electrical conductivity of the guar gum hydrogel with different borax content (black), the hydrogel filled with different f-MWCNTs contents (red), and the hydrogel filled with different AgNP-MWCNTs content (blue). (b) Schematic of practical demonstration: pronunciation, finger angle measurement, and wrist angle measurement. Relative resistance of (c) finger bent at three distinct angles (30°, 60° and 90°) and (d) wrist up and down movement. Relative resistance of opening and closing mouth of word (e) “apple” and (f) “laptop”. (g) Photos present practical use of movable body parts.
Self-healing properties of hydrogel
The inherent capacity of hydrogel to undergo self-repair suggests that it can regain its original elasticity and flexibility after damage62. To evaluate self-healing, the properties of the hydrogels before and after damage were investigated63. Hydrogel samples were cut into two parts and promptly fused to create a single thin hydrogel layer. At the cross-sectional area, the borate bonds were severed, releasing free diol molecules and borate ions. These components facilitated the regeneration of nascent borate bonds, which served as the healing agents in the hydrogel64,65. Furthermore, f-MWCNTs possess dynamic characteristics that enable the re-establishment of physical connections between polymer chains, promoting self-healing. The high surface area and electrical conductivity of f-MWCNTs enhanced charge transfer and self-healing processes by promoting chemical rearrangement and reconnection of broken polymer chains. The self-healing ability of hydrogel is further enhanced by the synergistic effect of AgNP-MWCNTs, which formed a network within the hydrogel, strengthening its structural integrity. The surface of AgNP-MWCNTs also facilitates additional crosslinking sites, enhancing the efficiency and speed of the self-healing process.
The hydrogels demonstrated self-healing properties and electrical conductivity through the mobility of borate ions and charge transfer from conductive materials. Figure 10 shows the self-healing characteristics of the guar gum hydrogel and the hydrogels filled with f-MWCNTs and AgNP-MWCNTs, as evidenced by their connection to the LED indicator. First, the original hydrogel thin film displayed a stable and bright LED indication. Second, when the hydrogel thin films were cut into two parts, the LED light was turned off. Finally, upon rejoining the two parts, the LED indicator turned on and remained stable, demonstrating the solid electrical conductivity and self-healing ability of various materials.
Figure 11a,b show the optical microscopic analysis of various hydrogels after the self-healing process. The guar gum hydrogel demonstrated the ability to repair the cut area in 12 s. In contrast, the hydrogel filled with f-MWCNTs exhibited a faster repair time of 10 s, as displayed in Fig. 11c,d. The hydrogel filled with AgNP-MWCNTs shows the fastest self-healing capability, achieving repair in 8 s, as depicted in Fig. 11e,f. The incorporation of f-MWCNTs and AgNP-MWCNTs significantly enhanced the self-healing ability of the hydrogel. This enhancement results from the small-sized f-MWCNTs and AgNP-MWCNTs, efficiently filling the hydrogel network. These particles created additional sites for crosslinking by facilitating the formation of hydrogen bonds and promoting the recombination of borax and diol at the contact surface.
Moreover, after the self-healing process of various hydrogel films, their mechanical strength was investigated by a magnet weight-holding technique, as shown in Figure S5. The thin sample of guar gum hydrogel could support a weight of up to 135.60 g. In comparison, the hydrogel filled with f-MWCNTs film could withstand up to 215.30 g, while the hydrogel filled with AgNP-MWCNTs film demonstrated a weight capacity of 295.10 g before material failure occurred.
Optical microscopic images of (a,b) the guar gum hydrogel, (c,d) the hydrogel filled with f-MWCNTs, and (e,f) the hydrogel filled with AgNP-MWCNTs after cutting and self-healing. Self-healing efficiency of (g) guar gum hydrogel with different borax content, (h) the hydrogel filled with f-MWCNTs content, (i) the hydrogel filled with AgNP-MWCNTs content.
Figure 11g to i shows the self-healing efficiency of various hydrogel materials. Among them, the guar gum hydrogel with 20wt% borax exhibited the highest self-healing efficiency, achieving a tensile strength recovery of 64.8 ± 3.8% and an elongation at break recovery of 72.5 ± 2.1%. The hydrogel filled with f-MWCNTs demonstrated a greater self-healing efficiency, with a tensile strength recovery of 73.7 ± 4.9% and elongation at break recovery of 78.4 ± 2.9%. Notably, the hydrogel filled with AgNP-MWCNTs achieved the highest self-healing efficiency value, with a tensile strength recovery of 82.4 ± 2.1% and an elongation at break recovery of 83.2 ± 3.6%.
Conclusions
Hydrogels based on guar gum, filled with f-MWCNTs and AgNP-MWCNTs, were successfully synthesised via simple one-pot synthesis. The incorporation of AgNP-MWCNTs and f-MWCNTs significantly enhanced the strain-sensing capability of the hydrogel system due to increased conductivity. In particular, the hydrogel filled with AgNP-MWCNTs exhibited a sensitive response, which can be attributed to the synergistic interaction between AgNPs and MWCNTs, where AgNPs acted as a bridge connecting MWCNTs.
These advantageous properties facilitated the development of wearable hydrogel sensors that showed excellent sensitivity and stability, enduring up to 3000 tensile loading and unloading cycles at 50% strain. The practical demonstrations further highlighted the high sensitivity of the sensors to subtle movements, such as pronunciation and finger and wrist movements. Notably, the use of green design principles in this work offered a cost-effective and environmentally sustainable approach to developing high-performance soft materials for a variety of sensing applications, such as flexible electronics, artificial skins, and healthcare monitoring.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514. https://doi.org/10.1038/nature16521 (2016).
Oh, J. Y. et al. Intrinsically stretchable and healable semiconducting polymer for organic transistors. Nature 539, 411–415. https://doi.org/10.1038/nature20102 (2016).
Bae, J. et al. Fiber supercapacitors made of nanowire-fiber hybrid structures for wearable/flexible energy storage. Angew. Chem. Int. Ed. 50, 1683–1687. https://doi.org/10.1002/anie.201006062 (2011).
Liu, L., Yu, Y., Yan, C., Li, K. & Zheng, Z. Wearable energy-dense and power-dense supercapacitor yarns enabled by scalable graphene–metallic textile composite electrodes. Nat. Commun. 6, 7260. https://doi.org/10.1038/ncomms8260 (2015).
Chou, H. H. et al. A chameleon-inspired stretchable electronic skin with interactive colour changing controlled by tactile sensing. Nat. Commun. 6, 8011. https://doi.org/10.1038/ncomms9011 (2015).
Wang, S. et al. Skin electronics from scalable fabrication of an intrinsically stretchable transistor array. Nature 555, 83–88. https://doi.org/10.1038/nature25494 (2018).
Lee, J. et al. Room-temperature nanosoldering of a very long metal nanowire network by conducting‐polymer‐assisted joining for a flexible touch‐panel application. Adv. Funct. Mater. 23, 4171–4176. https://doi.org/10.1002/adfm.201203802 (2013).
Meng, B. et al. A transparent single-friction-surface triboelectric generator and self-powered touch sensor. Energy Environ. Sci. 6, 3235–3240. https://doi.org/10.1039/C3EE42311E (2013).
Ge, G. et al. A flexible pressure sensor based on rGO/polyaniline wrapped sponge with tunable sensitivity for human motion detection. Nanoscale 10, 10033–10040. https://doi.org/10.1039/C8NR02813C (2018).
Ryu, S. et al. Extremely elastic wearable carbon nanotube fiber strain sensor for monitoring of human motion. ACS nano. 9, 5929–5936. https://doi.org/10.1021/acsnano.5b00599 (2015).
Guo, Q. et al. Protein-inspired self-healable Ti3C2 MXenes/rubber-based supramolecular elastomer for intelligent sensing. ACS nano. 14, 2788–2797. https://doi.org/10.1021/acsnano.9b09802 (2020).
Ismail, Z., Idris, W., Abdullah, A. H. & W. F. & Graphene-based temperature, humidity, and strain sensor: a review on progress, characterization, and potential applications during Covid-19 pandemic. Sens. Int. 3, 100183. https://doi.org/10.1016/j.sintl.2022.100183 (2022).
Kim, J. H., Kim, S. R., Kil, H. J., Kim, Y. C. & Park, J. W. Highly conformable, transparent electrodes for epidermal electronics. Nano Lett. 18, 4531–4540. https://doi.org/10.1021/acs.nanolett.8b01743 (2018).
Cai, L. et al. Super-stretchable, transparent carbon nanotube-based capacitive strain sensors for human motion detection. Sci. Rep. 3, 3048. https://doi.org/10.1038/srep03048 (2013).
Matsalis, S., Paterakis, G., Koutroumanis, N., Anagnostopoulos, G. & Galiotis, C. Fabrication and performance of capacitive humidity and strain sensors that incorporate 3D-printed nanocomposite electrodes. Sens. Int. 5, 100272. https://doi.org/10.1016/j.sintl.2023.100272 (2024).
Markvicka, E. J., Bartlett, M. D., Huang, X. & Majidi, C. An autonomously electrically self-healing liquid metal–elastomer composite for robust soft-matter robotics and electronics. Nat. Mater. 17, 618–624. https://doi.org/10.1038/s41563-018-0084-7 (2018).
Song, D., Chen, X., Wang, M. & Xiao, X. Flexible sensors for mechatronic engineering education. Sens. Int. 4, 100236. https://doi.org/10.1016/j.sintl.2023.100236 (2023).
Xu, M., Qi, J., Li, F. & Zhang, Y. Highly stretchable strain sensors with reduced graphene oxide sensing liquids for wearable electronics. Nanoscale 10, 5264–5271. https://doi.org/10.1039/C7NR09022F (2018).
Lin, L. et al. Dual conductive network enabled superhydrophobic and high performance strain sensors with outstanding electro-thermal performance and extremely high gauge factors. Chem. Eng. J. 385, 123391. https://doi.org/10.1016/j.cej.2019.123391 (2020).
Ying, W. B. et al. Waterproof, highly tough, and fast self-healing polyurethane for durable electronic skin. ACS Appl. Mater. Interfaces. 12, 11072–11083. https://doi.org/10.1021/acsami.0c00443 (2020).
Wang, Z., Cong, Y. & Fu, J. Stretchable and tough conductive hydrogels for flexible pressure and strain sensors. J. Mater. Chem. B. 8, 3437–3459. https://doi.org/10.1039/C9TB02570G (2020).
Singha, I. & Basu, A. Chitosan based injectable hydrogels for smart drug delivery applications. Sens. Int. 3, 100168. https://doi.org/10.1016/j.sintl.2022.100168 (2022).
López-Marcial, G. R. et al. Agarose-based hydrogels as suitable bioprinting materials for tissue engineering. ACS Biomaterials Sci. Eng. 4, 3610–3616. https://doi.org/10.1021/acsbiomaterials.8b00903 (2018).
Tang, L. et al. A super-tough ionic conductive hydrogel with anti-freezing, water retention, and self-regenerated properties for self-powered flexible sensor. Appl. Mater. Today. 32, 101820. https://doi.org/10.1016/j.apmt.2023.101820 (2023).
Lei, Z., Wang, Q., Sun, S., Zhu, W. & Wu, P. A bioinspired mineral hydrogel as a self-healable, mechanically adaptable ionic skin for highly sensitive pressure sensing. Adv. Mater. 29, 1700321. https://doi.org/10.1002/adma.201700321 (2017).
Zhang, H. et al. Dual physically cross-linked carboxymethyl cellulose-based hydrogel with high stretchability and toughness as sensitive strain sensors. Cellulose 27, 9975–9989. https://doi.org/10.1007/s10570-020-03463-5 (2020).
Wang, B. et al. Development of hierarchically constructed robust multifunctional hydrogels for sensitive strain sensors by using guar gum and polydopamine to encapsulate liquid metal droplets. Appl. Mater. Today. 35, 101961. https://doi.org/10.1016/j.apmt.2023.101961 (2023).
Pan, X. et al. Ultraflexible self-healing guar gum-glycerol hydrogel with injectable, antifreeze, and strain-sensitive properties. ACS Biomaterials Sci. Eng. 4, 3397–3404. https://doi.org/10.1021/acsbiomaterials.8b00657 (2018).
Wang, Y., Yang, M. & Zhao, Z. Facile fabrication of self-healing, injectable and antimicrobial cationic guar gum hydrogel dressings driven by hydrogen bonds. Carbohydr. Polym. 310, 120723 (2023).
Maroufi, L. Y., Tabibiazar, M., Ghorbani, M. & Jahanban-Esfahlan, A. Fabrication and characterization of novel antibacterial chitosan/dialdehyde guar gum hydrogels containing pomegranate peel extract for active food packaging application. Int. J. Biol. Macromol. 187, 179–188 (2021).
Bocchinfuso, G. et al. Guar gum and scleroglucan interactions with borax: experimental and theoretical studies of an unexpected similarity. J. Phys. Chem. B. 114, 13059–13068. https://doi.org/10.1021/jp105838t (2010).
Tanpichai, S., Phoothong, F. & Boonmahitthisud, A. Superabsorbent cellulose-based hydrogels cross-liked with borax. Sci. Rep. 12, 8920 (2022).
Williams, W. P., Quinn, P. J., Tsonev, L. I. & Koynova, R. D. The effects of glycerol on the phase behaviour of hydrated distearoylphosphatidylethanolamine and its possible relation to the mode of action of cryoprotectants. Biochim. et Biophys. Acta (BBA)-Biomembranes. 1062, 123–132. https://doi.org/10.1016/0005-2736(91)90383-J (1991).
Dashnau, J. L., Nucci, N. V., Sharp, K. A. & Vanderkooi, J. M. Hydrogen bonding and the cryoprotective properties of glycerol/water mixtures. J. Phys. Chem. B. 110, 13670–13677. https://doi.org/10.1021/jp0618680 (2006).
Liu, X. et al. A highly stretchable, sensing durability, transparent, and environmentally stable ion conducting hydrogel strain sensor built by interpenetrating Ca2+-SA and glycerol-PVA double physically cross-linked networks. Adv. Compos. Hybrid. Mater. 5, 1712–1729 (2022).
Prusty, R. K., Rathore, D. K. & Ray, B. C. CNT/polymer interface in polymeric composites and its sensitivity study at different environments. Adv. Colloid Interface Sci. 240, 77–106. https://doi.org/10.1016/j.cis.2016.12.008 (2017).
Fethi, A. Novel materials for electrochemical sensing platforms. Sens. Int. 1, 100035. https://doi.org/10.1016/j.sintl.2020.100035 (2020).
Buldum, A. & Lu, J. P. Contact resistance between carbon nanotubes. Phys. Rev. B. 63, 161403. https://doi.org/10.1103/PhysRevB.63.161403 (2001).
Ge, G. et al. Highly stretchable and autonomously healable epidermal sensor based on multi-functional hydrogel frameworks. J. Mater. Chem. A. 7, 5949–5956. https://doi.org/10.1039/C9TA00641A (2019).
Zhang, D. et al. From design to applications of stimuli-responsive hydrogel strain sensors. J. Mater. Chem. B. 8, 3171–3191. https://doi.org/10.1039/C9TB02692D (2020).
Zhou, L., Xin, Q., Lin, J., Liang, S. & Yang, G. A low-cost hydrogel with high conductivity and flexibility for pressure sensor and supercapacitor. Appl. Mater. Today. 34, 101907. https://doi.org/10.1016/j.apmt.2023.101907 (2023).
Rojas, J., Toro-Gonzalez, M., Molina-Higgins, M. & Castano, C. Facile radiolytic synthesis of ruthenium nanoparticles on graphene oxide and carbon nanotubes. Mater. Sci. Engineering: B. 205, 28–35. https://doi.org/10.1016/j.mseb.2015.12.005 (2016).
Zare, Y., Rhee, K. Y. & Hui, D. Influences of nanoparticles aggregation/agglomeration on the interfacial/interphase and tensile properties of nanocomposites. Compos. Part. B: Eng. 122, 41–46. https://doi.org/10.1016/j.compositesb.2017.04.008 (2017).
Niu, B., Yang, S., Tian, X. & Hua, T. Highly sensitive and stretchable fiber strain sensors empowered by synergetic conductive network of silver nanoparticles and carbon nanotubes. Appl. Mater. Today. 25, 101221. https://doi.org/10.1016/j.apmt.2021.101221 (2021).
Awasthi, S. & Pandey, S. K. Recent advances in smart hydrogels and carbonaceous nanoallotropes composites. Appl. Mater. Today. 36, 102058. https://doi.org/10.1016/j.apmt.2024.102058 (2024).
Shojaei, A. & Khasraghi, S. S. Composite Materials 307–357 (Elsevier, 2021).
Song, T. et al. Self-healing materials: a review of recent developments. ES Mater. Manuf. 14, 1–19. https://doi.org/10.30919/esmm5f465 (2021).
Pathan, N. & Shende, P. Strategic conceptualization and potential of self-healing polymers in biomedical field. Mater. Sci. Engineering: C. 125, 112099. https://doi.org/10.1016/j.msec.2021.112099 (2021).
Rammal, H. et al. Advances in biomedical applications of self-healing hydrogels. Mater. Chem. Front. 5, 4368–4400. https://doi.org/10.1039/D0QM01099E (2021).
Jun, L. Y. et al. Comparative study of acid functionalization of carbon nanotube via ultrasonic and reflux mechanism. J. Environ. Chem. Eng. 6, 5889–5896. https://doi.org/10.1016/j.jece.2018.09.008 (2018).
Verma, D. et al. Development of MWCNT decorated with green synthesized AgNps-based electrochemical sensor for highly sensitive detection of BPA. J. Appl. Electrochem. 51, 447–462. https://doi.org/10.1007/s10800-020-01511-3 (2021).
Makvandi, P. et al. Injectable hyaluronic acid-based antibacterial hydrogel adorned with biogenically synthesized AgNPs-decorated multi-walled carbon nanotubes. Prog. Biomater. 10, 77–89. https://doi.org/10.1007/s40204-021-00155-6 (2021).
Anshori, I. et al. Functionalized multi-walled carbon nanotube/silver nanoparticle (f-MWCNT/AgNP) nanocomposites as non-enzymatic electrochemical biosensors for dopamine detection. Nanocomposites 7, 97–108. https://doi.org/10.1080/20550324.2021.1948242 (2021).
Thombare, N., Jha, U., Mishra, S. & Siddiqui, M. Borax cross-linked guar gum hydrogels as potential adsorbents for water purification. Carbohydr. Polym. 168, 274–281. https://doi.org/10.1016/j.carbpol.2017.03.086 (2017).
Luchsinger, W. W. & Stone, B. A. Linkage sequencing of oligosaccharides by their rates of alkaline degradation. Carbohydr. Res. 46, 1–8. https://doi.org/10.1016/S0008-6215(00)83525-9 (1976).
Yu, X. et al. Highly sensitive, weatherability strain and temperature sensors based on AgNPs@ CNT composite polyvinyl hydrogel. J. Mater. Chem. A. 10, 15000–15011. https://doi.org/10.1039/D2TA02559K (2022).
Ge, G. et al. Muscle-inspired self-healing hydrogels for strain and temperature sensor. ACS Nano. 14, 218–228. https://doi.org/10.1021/acsnano.9b07874 (2020).
Li, J. et al. Correlations between percolation threshold, dispersion state, and aspect ratio of carbon nanotubes. Adv. Funct. Mater. 17, 3207–3215. https://doi.org/10.1002/adfm.200700065 (2007).
Zha, J. W., Shehzad, K., Li, W. K. & Dang, Z. M. The effect of aspect ratio on the piezoresistive behavior of the multiwalled carbon nanotubes/thermoplastic elastomer nanocomposites. J. Appl. Phys. 113 https://doi.org/10.1063/1.4772747 (2013).
Xiang, D. et al. Synergistic effects of hybrid conductive nanofillers on the performance of 3D printed highly elastic strain sensors. Compos. Part A: Appl. Sci. Manufac. 129, 105730. https://doi.org/10.1016/j.compositesa.2019.105730 (2020).
Tolvanen, J., Hannu, J. & Jantunen, H. Stretchable and washable strain sensor based on cracking structure for human motion monitoring. Sci. Rep. 8, 13241. https://doi.org/10.1038/s41598-018-31628-7 (2018).
Dong, H. et al. Ultrasmall gold nanoparticles/carboxymethyl chitosan composite hydrogel: tough, restorable, biocompatible antimicrobial dressing for wound healing. Appl. Mater. Today. 38, 102206. https://doi.org/10.1016/j.apmt.2024.102206 (2024).
Wang, S. & Urban, M. W. Self-healing polymers. Nat. Reviews Mater. 5, 562–583. https://doi.org/10.1038/s41578-020-0202-4 (2020).
Cao, D., Lv, Y., Zhou, Q., Chen, Y. & Qian, X. Guar gum/gellan gum interpenetrating-network self-healing hydrogels for human motion detection. Eur. Polymer J. 151, 110371. https://doi.org/10.1016/j.eurpolymj.2021.110371 (2021).
Jafari, H. et al. An injectable, self-healing, 3D printable, double network co-enzymatically crosslinked hydrogel using marine poly- and oligo-saccharides for wound healing application. Appl. Mater. Today. 29, 101581. https://doi.org/10.1016/j.apmt.2022.101581 (2022).
Acknowledgements
This work is supported by the 90th Anniversary of Chulalongkorn University Scholarship under the Ratchadapisek Somphot Endowment Fund, National Research Council of Thailand (NRCT) and Chulalongkorn University (N42A660910), Thailand Science Research and Innovation Fund, Chulalongkorn University, and the Second Century Fund (C2F), Chulalongkorn University.
Author information
Authors and Affiliations
Contributions
Aamir Khan conducted experiments and prepared a draft manuscript. Nichakan Nichakonpong and Tawan Wongsalam discussed the results. Peerawat Prathumrat and Chutiwat Likitaporn edited final manuscript. Pornnapa Kasemsiri consulted the results and validated the manuscript. Manunya Okhawilai supervised the design of the project and edited the final manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Khan, A., Nichakornpong, N., Wongsalam, T. et al. Development of green synthesised AgNPs decorated on MWCNT modified guar gum-based self-healing hydrogel for strain sensors. Sci Rep 14, 29715 (2024). https://doi.org/10.1038/s41598-024-81085-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-81085-8