Abstract
Corneal alkali burns persist as a significant challenge in our field, often leading to a prolonged treatment course with various sight-threatening problems. This work, of utmost importance, aimed to apply the photo-tissue bonding technique (PTB) to weld the amniotic membrane (AM) to the corneal surface versus an amniotic membrane graft (AMG) and explore its safety in saving corneal protein against alkali burn.
Methods Twenty-seven rabbits with an induced corneal ulcer using 1 mol/L NaOH solution. Nine rabbits were used as an ulcer group without treatment, and the rest (n = 18) were subjected to two treatment protocols with AM. The first was attaching the AM to the corneal ulcer through photo-tissue bonding using 532 nm and rose Bengal stain as a photosynthesizing agent. The second was using cyanoacrylate glue as a tissue adhesive. The corneal total protein (TP), refractive index (RI), DNA fragmentation, and oxidative stress index (OSI) were evaluated. Results: The cornea’s TP showed a significant decrease (p˂0.001) immediately, 1 week, and 2 weeks after ulcer induction (-58.9%, -64.4%, and − 72.6%, respectively). The treatment with AM PTB showed improvement immediately (-45.2%, p˂0.001), after one week (-27.4%, p˂0.01), and after two weeks (-14.38%, p˂0.05). Moreover, the treatment with AMG showed improvement after the same periods with percentage changes of -52.05%, (p˂0.001), -41.8% (p˂0.001), and − 32.2%, (p˂0.01) with respect to the control. Moreover, RI of corneal protein showed improvement after two weeks of treatment with AM PTB (3%, p˃0.05) and AMG (7%, p˃0.05), respectively. The corneal protein DNA base pairs improved 88.49% for AM PTB and 82.35% for the AMG group. The oxidative stress was shifted towards an antioxidative state in AM PTB (-3.9%, P > 0.05) and the AMG group (15.9%, P < 0.05). Conclusion: The AM PTB technique used in corneal ulcers showed promising improvement in total corneal proteins, refractive index, DNA fragmentation, and OSI than AMG using cyanoacrylate glue. These results strongly support the use of AM PTB for ophthalmic purposes, suggesting its potential to enhance clinical research and practice for patients with corneal ulcers and ocular surface diseases.
Similar content being viewed by others
Corneal protein repair after amniotic membrane photo-tissue bonding versus amniotic membrane graft in treatment of corneal ulcer (An experimental study).
Background
Corneal alkali burn causes intractable keratitis, eruption formation, epithelial breakdown, inflammatory cell infiltration, stromal cell death, and endothelial dysfunction. The amniotic membrane (AM) is frequently used in cornea and sclera surgeries as a temporary patch or a reconstructive graft1. Amniotic membrane transplantation (AMT) covers persistent epithelial defects, pterygium surgery, and ocular surface reconstruction in stem cell deficiency. The AM comprises a single layer of epithelial cells attached to a basement membrane that lies over a stromal layer containing primarily types I and III collagen, proteoglycans, and fibroblasts. Currently, suturing the amnion to the cornea is a time-consuming process requiring high skills to place hair-fine sutures. In addition, suturing may injure the eye and act as a foreign body, leading to persistent inflammation, infection, and granuloma1,2.
The suture-less attachment of AM to the cornea, with its rapid formation of an immediate water-tight seal without damage to the cornea, is a reassuring sign of its efficiency3. Tissue adhesives, such as synthetic glue, are gaining approval in ophthalmology due to their potential to reduce the complications associated with current surgical methods. Cyanoacrylate glue, a strong tissue adhesive with high tensile strength, rapidly polymerizes upon contact with any fluid but has been associated with cytotoxicity4.
AM photo-tissue bonding (PTB) means sealing the AM over a sterile corneal ulcer using a light-activated technology that produces an immediate seal between tissue surfaces without additional glues or proteins5,6,7. A significant advantage of PTB is the minimal scarring and fibrosis produced compared to AM suturing8,9.
The Food and Drug Administration (FDA) approved Rose Bengal dye (RB) to diagnose ocular surface diseases. RB dye was used to generate singlet oxygen (1O2), which has been suggested to mediate the formation of protein − protein bonds10,11. Photosensitized protein crosslinking prevents deleterious changes to tissue in response to physiological pressures12,13. In addition, RB photosensitization makes tissue less sensitive to inflammation and is identified as photochemical tissue passivation14.
This work aimed to evaluate the improvement of corneal protein after treating corneal ulcers. Fortunately, all used materials, including AM, RB, and diode green laser, are FDA-approved for ophthalmic uses. This FDA approval provides a strong foundation for the safety and efficacy of the procedure. Therefore, AM PTB was applied using RB and green laser (532 nm) to weld the AM to the corneal ulcer and compared with the AMG using cyanoacrylate glue as a tissue adhesive. The corneal protein concentration, refractive index measurements, oxidative stress index, and DNA damage analysis were evaluated immediately, one week, and two weeks later.
Results
Total corneal protein concentration
The corneal protein concentration of the ulcerated cornea and after the treatment with AM PTB and AMG is illustrated in Fig. 1. The total protein for the control was 146 ± 8 mg/g tissue wet wt and significantly decreased after induction of ulcers to 60 ± 6 mg/g tissue wet wt (p˂0.001), 52 ± 6 mg/g tissue wet wt (p˂0.001) and 40 ± 5 mg/g tissue wet wt (p˂0.001) when measured immediately, after one week and two weeks with percentage changes of −58.9%, −64.4% and − 72.6% compared with the control, respectively.Treatment of ulcers with AM PTB showed gradual improvement with values of 80 ± 6 mg/g tissue wet wt (p˂0.001), 106 ± 8 mg/g tissue wet wt (p˂0.01), and 125 ± 5 mg/g tissue wet wt (p˂0.05) with percentage changes of −45,2%, −27.4% and − 14.38% when measured immediately, after one week and two weeks respectively, compared with the control. Moreover, the treatment of the ulcer with AMG using cyanoacrylate glue showed gradual improvement after the same periods of the previous group with values of 70 ± 7 mg/g tissue wet wt (p˂0.001), 85 ± 4 mg/g tissue wet wt (p˂0.001), and 99 ± 5 mg/g tissue wet wt (p˂0.01) with percentage changes of −52.05%, −41.8% and − 32.2% with respect to the control.
The refractive index for corneal proteins (RI)
The RI of corneal protein for the control, ulcerated, treated corneas with AM PTB and AMG are shown in Fig. 2. The refractive index for the sample of the control cornea was 1.3245, which showed a significant decrease immediately after induction of corneal ulcer and after one week and two weeks. The RI was 1.3224, 1.3220, and 1.3218, with percentage changes of 16%, 19%, and 20% (p˂0.01) with respect to the control. Furthermore, treatment with AM PTB showed improvement and non-significant changes in the corneal protein refractive index with values of 1.3230, 1.3238, and 1.3240 with percentage changes of 11%, 5%, and 3% (p˃0.05), respectively, when compared with the control. Moreover, the corneas treated with AMG using cyanoacrylate glue also showed improvement in the RI of corneal protein when measured immediately, one week, and two weeks with values of 1.3228, 1.3231, and 1.3235, respectively. Consequently, the percentage change compared with the control was 12%, 10%, and 7% after the three estimated periods.
DNA fragmentation for corneal protein
The fragmentation of DNA of corneal protein was analyzed using 0.1% agarose gel electrophoresis (Fig. 3). The first lane represents the DNA markers ranging from 100 to 1000 BP (Base Pair). The second lane represents the control group in one band with 1064 BP. The third lane represents the ulcerated cornea with four fragmented bands (1151 BP, 346 BP, 256 BP, and 141 BP). The fourth lane represents the corneas treated with AMG, isolated, and immediately analyzed, which showed four fragments (1137 BP, 300 BP, 237 BP, and 152 BP). The DNA fragmentation was increased after one week (lane 5), as shown in Table 1, with values of 372 BP, 292 BP, 256 BP, 225 BP, and 200 BP. In addition, after two weeks (lane 6), there was an improvement in 82.35% of DNA but still more fragmentation in the rest of the DNA. After treatment with AM PTB (immediately), three fragments appeared in the agarose gel with 1124 BP, 636 BP, and 469 BP. The first band (1164 BP) showed noticeable improvement after two weeks, with a percentage of 88. 49% (lane 9), respectively.
Agarose gel electrophoresis for corneal protein represents the DNA fragmentation for the control, ulcerated and treated with AM PTB and AMG after different periods. AMG: Amniotic membrane graft, PTB: Photo-tissue bonding, (0): immediately. The samples were derived from the same experiment, and the gels/blots were processed in parallel. Original blots/gels are presented in Supplementary figure S1.
Evaluation of oxidative stress index (OSI).
Mean and SD values of TAC, TOC, and OSI levels for control and all treated groups were presented in Table 2. The data showed a significant elevation of TOC (114.3%, P < 0.001) and TAC (50.4%, P < 0.001) after corneal ulcer. The treatment with AM PTB showed gradual improvement in TOC and TAC after two weeks, reaching 10.7% and 11.8% with respect to the control. Alternatively, the OSI of the cornea showed non -significant changes (−3.9%, P > 0.05). Moreover, the treatment with AMG showed noticeable improvement in the TOC and TAC, with percentage changes of 32.1% and 13.9% values after two weeks, respectively. In the case of the corneal ulcer group, the data indicated shifting toward oxidative status (4.28 ± 0.3), and oxidative stress was very high compared with the control group (3.01 ± 0.2). After two weeks of AM PTB and AMG, the oxidative stress was shifted towards an antioxidative state with values of 2.98 ± 0.2 (−3.9%, P > 0.05) and 3.49 ± 0.2 (15.9%, P < 0.05), respectively.
Discussion
Our study, which utilized AM PTB on an ulcerated cornea with alkali burn, aimed to demonstrate the benefits of suture-less AM fixation to the cornea and collagen crosslinking through the photodynamic effect. Additionally, we applied the AMG technique using cyanoacrylate glue and compared the efficiency of both AM PTB and AMG in improving corneal protein. The implications of our findings are significant, as they provide valuable insights into the potential treatment of corneal ulcers, particularly in the context of alkali burns.
Previous studies used the AM PTB technique to weld or bond the edges of corneal wounds15. Other studies used RB and green laser-collagen crosslinking only without AM to treat a resistant infectious corneal ulcer16,17. Moreover, the usefulness of the fixation of AM using 2-octyl cyanoacrylate glue in treating experimental corneal burns with less vascularization and rapid epithelial healing was evaluated18.
Our study showed that the corneal protein concentration in the ulcerated group significantly decreased. This decrement suggests corneal melting because of the inflammatory reaction after the alkali burn. The primary mechanism of corneal melting in alkali burn is the presence of many polymorphonuclear leukocyte infiltration, collagenase, and matrix metalloproteinase, leading to the continuous digestion of corneal collagen19. In contrast, there was a gradual increase in the corneal proteins in the group treated with AM PTB and the group treated with AMG using cyanoacrylate glue. The improvement was significantly higher in the group treated with AM PTB than in the AMG group. The better improvement in the group of AM PTB may be due to the anti-inflammatory effect of the AM in addition to the photodynamic impact that makes the tissue less sensitive to inflammation, which is called tissue passivation12,13. Moreover, treatment with AM PTB reflects the increase in corneal resistance to collagenase. It prevents and delays corneal melting and can decrease collagen fibers’ damage and infiltration of inflammatory cells in the corneal tissues20. Furthermore, the resistance of collagen to enzymatic digestion by collagenase in corneas treated by collagen cross-linking using RB and green laser was reported previously21. This meets our results of the better improvement of corneal protein in the AM PTB group than the AMG group and may be due to the diffusion of RB into the stroma affecting more corneal proteins16. RB induces protein crosslinks without phototoxicity to keratocytes by a mechanism concerning 1O2. Further, RB forms two complexes with amnion stromal collagen, that bonding needs oxygen, and singlet oxygen mediates protein cross-linking22.
The corneal protein RI is crucial for characterizing proteins and complexes. This parameter is also essential, particularly for understanding the optical properties of eye tissues23. Empirical measurements of the corneal refractive index are limited, and the cornea’s layers exhibit varying refractive indices due to differing physiological properties24. Measurements of the RI of corneal protein extracts can significantly reflect the overall corneal RI from multiple structural changes rather than a single layer. In our study, the progressive decrease in RI in the ulcerated group due to reduced soluble protein in the cornea reflects improper remodeling and disordered protein structure. Furthermore, improving the RI after treatment revealed the reformation of a proper and ordered protein structure. In addition, this improvement is returned to the decrease in inflammation and melting of the cornea as reported by Soeken et al., which showed that sealing of the cornea by AM PTB could occur with minimal inflammation or secondary effects15.
In animal models, therapies that restore corneal transparency are evaluated using chemical and biophysical techniques. Advanced methods, such as DNA fragmentation analysis, enhance the assessment of these therapies, aiding their transition to clinical trials. Agarose gel electrophoresis outlines a method for analyzing DNA fragmentation linked to apoptosis. Key characteristics of apoptosis include DNA fragmentation, which produces a ladder pattern on the gel. While the method effectively assesses DNA fragmentation qualitatively, it allows quantitative analysis using the Gel Pro analyzer 4.5. Overall, this assay is valuable for both qualitative and quantitative evaluation of apoptosis, as shown in Table 1. Compared to the ulcerated corneas, the AMG and AM PTB-treated corneas had a less inflammatory response and effectively suppressed apoptosis (Table 1). Human amniotic epithelial cells improve the function of human corneal epithelial cells by reducing apoptosis. This is achieved through several mechanisms: decreasing reactive oxygen species (ROS) production, maintaining higher membrane potential, lowering p53 protein expression, and increasing survivin protein levels. Additionally, high expression of the PAX6 gene in corneal epithelial cells enhances cell proliferation and inhibits apoptosis, contributing to better preservation of corneal epithelial characteristics25,26.
Cross-linking is the formation of chemical bonds between two molecules, such as linking two DNA strands by covalent bonds. That makes the polymer chains bound together with less extent of movement27. They become more rigid, stronger, and more challenging as they are linked, like in our study using agarose gel electrophoresis (Fig. 3). The results revealed marked resistance of DNA to fragmentation after two weeks in both the AM PTB group (88.49%) and the AMG group (82.35%), respectively (Table 1). The improvement in the AM PTB group is revealed to the collagen crosslinking that occurs through the reaction of singlet oxygen with the surrounding amino acids, mainly histidine, to form covalent bonds across collagen fibers20. Oxidized histidine then reacts with specific amino acids, primarily lysine, to form protein-protein crosslinks28. This photochemical crosslinking can strengthen the corneal stroma, improve biomechanical rigidity, and increase corneal stiffness and resistance to collagenase digestion12,22. Further, the increased resistance to collagenase digestion is owed to changes in the tertiary structure of the collagen fibrils, which block the access of proteolytic enzymes to the cleavage sites through steric hindrance29. Steric hindrance means prevention or retardation of inter- or intramolecular interactions because of the spatial structure of a molecule. Likewise, using cyanoacrylate glue in the AMG group may prevent collagenase production with a less inflammatory response19,30.
The present study highlights the antioxidative and oxidative state of the corneas exposed to alkali burn and treated with AM PTB and AMG. In the corneal ulcer group, the OSI was shifted toward high oxidative status (4.28 ± 0.3) compared with the control group (3.01 ± 0.2). After two weeks of treatment, the OSI was shifted towards an antioxidative state for both groups, and the improvement was better in the AM PTB group (−3.9%, P > 0.05) than in the AMG group (15.9%, P< 0.05), with respect to the control. Reactive oxygen species (ROS) are produced via oxidative reactions during metabolic and physiological processes31. These ROS attack cellular components, destroying lipids, proteins, and DNA, and can initiate a chain of reactions resulting in cellular complications32,33. Besides, enzymatic and non-enzymatic antioxidative mechanisms have been developed to reduce ROS damage32,33. The endogenous and food-derived antioxidants represent the total antioxidant activity accumulated in tissues12. Oxidative stress results when oxidation products such as lipids and proteins increase and antioxidant enzymes and vitamins decrease34,35. Hence, OSI, the corneal TOC to TAC level ratio, indicates oxidative stress reflecting the redox balance between oxidation and antioxidation11,36. In the present study, we observed an elevation in the TOC (114.3%) and TAC (50.4%) and, alternatively, an elevation in the OSI (42.2%) after induction of corneal ulcer. The ulcer induces oxidative stress and stimulates the antioxidant defense system by elevating TAC. Furthermore, treatment with AM PTB and AMG appeared time-dependent as the results indicated gradual improvement of the cornea after the 1st and 2nd week of treatment. Excitingly, the percentage change in OSI reflects the degree of oxidative stress. Our results suggested that the oxidative/antioxidative balance shifts towards the antioxidative status in the AM PTB group (− 3.9%, P> 0.05), which is better than the AMG group. These results match the present results of DNA fragmentation in which the photochemical crosslinking occurred through AM PTB causes resistance to collagenase digestion, blocks the access of proteolytic enzymes to the cleavage sites through steric hindrance, prevents further damage, and gives the cell time to repair the defect29. This study identified the AM PTB treatment as a better choice than AMG because it improved the corneal protein, reduced DNA fragmentation, and returned the oxidative stress to a balanced state between the oxidant and antioxidant. Moreover, after the estimated periods, the healing of ulcers treated with AM PTB showed better corneal integrity than in the AMG group. The follow-up showed no significant differences between the two treatment groups. No scaring, no corneal perforation, minimal superficial punctate keratitis, and a relatively smooth corneal profile. However, there was non-significant corneal neovascularization in the AMG-treated groups after one day.
This study demonstrates that a light-activated technology using AM PTB that welds the AM to the corneal surface by forming molecular crosslinks between proteins is strong enough to seal penetrating eye wounds and requires short irradiation. The seal has the benefits of being suture-less and rapid, creating an instant water-tight seal and not triggering another injury to the cornea17.
The obtained data highlighted the practical implications of using a sutureless AM PTB that resulted in better corneal protein healing than AMG using cyanoacrylate glue. The results indicate that using AM PTB offers an effective method for treating corneal wounds without requiring surgical skills and reducing suture-related complications. This finding is significant as it suggests that AM PTB can be recommended as a useful option in treating corneal ulcers and ocular surface diseases, such as pterygium excision, fornix reconstruction, and corneal melting, with excellent clinical outcomes. The study’s findings on the effectiveness of AM PTB in these clinical applications provide valuable insights for medical professionals. Additionally, this paper contributes not only to comparing the two techniques but also to the importance of the measured parameters such as protein content, refractive index, DNA fragmentation, and OSI levels as valuable markers for evaluating the effectiveness of the two treatment moods.
Methods
Experimental animals
Thirty-six female New Zealand rabbits (weight 2.0–2.5 kg) were obtained from the animal house of the Research Institute of Ophthalmology (RIO), Giza, Egypt. The rabbits were fed a standard laboratory diet during the experiment, free access to water at 22 ± 2 °C, 50–70% humidity, and a 12 h.-day–night cycle. The study was conducted in collaboration between the National Institute of Laser Enhanced Sciences, Cairo University (NILES), and the Research Institute of Ophthalmology, Giza, Egypt.
Induction of corneal ulcer
Adult female albino rabbits with intact corneas (n = 36) were divided into a control group of nine rabbits (n = 9), and the rest of the animals (n = 27) were generally anesthetized by intramuscular injection with 0.2 ml/kg xylaject® (xylazine hydrochloride 2%, ADWIA, Egypt)) and ketamine hydrochloride (0.6 ml/kg). In addition, Benoxinate hydrochloride (0.4%) eye drops (Benox® Eipico Co.,Cairo, Egypt.) was used for local anesthesia. A circular filter paper of 5.0 mm diameter was immersed in 1 mol/L sodium hydroxide solution (Sigma-Aldrich, St. Louis, MO, USA), centered on the animal’s right cornea for 30 s, and removed. The eye was irrigated immediately with a sterile saline solution for two minutes. The rabbits were divided equally into a corneal ulcers group (n = 9 eyes), a treated group with AM PTB (n = 9 eyes), and a treated group with AMG using cyanoacrylate glue (n = 9 eyes).
Preparation of the amnion patch
The ulcerated cornea surface was flushed with RB solution (1% wt/vol in PBS, Sigma-Aldrich, St. Louis, MO, USA)) for 2 min and then briefly washed with 0.9% saline solution. The sterile human AM patch (Bio Tissue Corporation, Miami, FL 33126, USA) was placed on filter paper with the stromal surface upward. RB solution (0.1% wt/vol in PBS) was placed on the AM stromal surface for 5 min. The dye-stained amnion patch was then cut to an appropriate size to cover the corneal ulcer with the epithelial basement membrane side facing up, and wrinkles were smoothed.
Bonding of the AM to the cornea
A diode-pumped solid-state laser was applied after the RB-stained amnion membrane was placed with its stromal surface in contact with the corneal ulcer (DPSS, HHs6-2, Tim e-relay, Elexperc, Egypt). The instrument was manufactured for research in the National Institute of Laser Enhanced Sciences (NILES), Cairo University laboratories. It emits a CW green light with a wavelength of 532 nm, irradiance of 0.25 W/cm2, and fluence of 100 J/cm2 through an optical fiber with a diameter of 4.0 mm.
Amniotic membrane grafting (AMG)
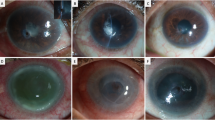
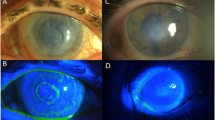
A lid speculum was used to expose the cornea and prevent gluing of the lids or the nictitating membrane. The ulcer was dried and debrided with a sterile cotton swab to remove loose or devitalized epithelial tissue. Cyanoacrylate glue (Monomeric n-butyl-2-cyanoacrylate, Histoacryl® B. Braun Surgical, SA, Carretera de Terrassa, Rubi, Spain) was drawn up into a 1-mL syringe through a 22-g needle. The needle was removed from the syringe, and the glue was pushed up to the flattened surface of the syringe tip and applied to cover the entire ulcerated area with a small amount extending beyond the ulcer edge. The amnion patch with an appropriate size was placed over the corneal ulcer with the epithelial basement membrane side facing up. Topical application of tobramycin/dexamethasone combination (Tobradex®, Alcon, USA.) was applied after finishing the procedure. Actual photographs of the various groups at different stages have been added as Supplementary figure S2.
Rabbits were euthanized with a high dose of ketamine hydrochloride and Xylazine hydrochloride (5 times the dose used in anesthesia). Three rabbits from each group were sacrificed immediately, one week, and two weeks after treatment, and the eyes were enucleated. The corneas were carefully isolated, weighed, and homogenized using a cell homogenizer (type Tübingen 7400, Germany) in a 10-fold volume of 20 mM ice-cold Tris-HCl buffer, pH 7.4. The homogenate was centrifuged for 20 min at 15,000 rpm (Awel centrifuge MS 20, France), and the supernatant was withdrawn.
Measurement of the corneal protein concentration and the refractive index
According to Lowry et al.37, the supernatant was used to determine the corneal protein concentration. The corneal protein samples were measured at 750 nm using a spectrophotometer (Evolution 600 PC UV‑Vis; Thermos Fisher Scientific, Madison, WI). The corneal protein refractive index was measured using the Abbe refractometer attached to a temperature control unit at 37 ℃±0.02.
Corneal DNA fragmentation
According to Dash et al.38, DNA fragmentation was evaluated using agarose gel electrophoresis. 20 µl of the DNA markers and corneal samples were loaded on 1% agarose gel wells containing 0.5 mg/ml of ethidium bromide in TBE buffer (Tris/Borate/EDTA, pH 8.0). The electrophoresis technique was run using an agarose gel electrophoresis device (Model Horizon 58, Gibco BRL, USA). The separation was viewed using a gel documentation system (Bio-Rad™) and analyzed with Gel Pro analyzer 4.5 software.
Oxidative stress index (OSI)
The total oxidative concentration (TOC) of the cornea samples was determined using a colorimetric method39. The color intensity, at 510 nm, is related to the total oxidative molecules in the sample and is expressed in terms of mM/L. The total antioxidative concentration (TAC) in the supernatant of corneal samples (mM/L) was obtained using a colorimetric method at 505 nm40. The reaction was performed through an enzymatic reaction between the amount of antioxidants in the cornea and a defined amount of exogenously provided hydrogen peroxide (H2O2). The ratio of the total oxidative concentration (TOC) to the total antioxidative concentration (TAC) was accepted as the oxidative stress index (OSI)41. All reagents used were purchased from BioDiagnostic Laboratories Inc., Brooklyn, New York, USA.
Statistical analysis
Student’s t-test for unpaired data was used to compare between groups; significance was set at P ≤ 0.05. The TAC, TOC, and OSI results were explained as the mean ± standard deviation.
Data availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- AM:
-
Amniotic membrane
- PTB:
-
Photo tissue bonding
- AMG:
-
Amniotic membrane grafting
- RB:
-
Rose Bengal
- TP:
-
Total Protein
- RI:
-
Refractive Index
- TAC:
-
Total Antioxidative Concentration
- TOC:
-
Total Oxidative Concentration
- OSI:
-
Oxidative Stress Index
- ROS:
-
Reactive Oxygen Species
References
Dua, H. S., Gomes, J. A., King, A. J. & Maharajan, V. S. The amniotic membrane in ophthalmology. Surv. Ophthalmol. 49, 51–77. https://doi.org/10.1016/j.survophthal.2003.10.004 (2004).
Christo, C. G., van Rooij, J., Geerards, A. J., Remeijer, L. & Beekhuis, W. H. Suture-related complications following keratoplasty: a 5-year retrospective study. Cornea 20, 816–819. https://doi.org/10.1097/00003226-200111000-00008 (2001).
Lotfy, N. M., Al Rashidi, S. & Hagras, S. M. Clinical outcomes of vacuum dehydrated amniotic membrane (Omnigen) mounted on contact lens (Omnilenz) in eyes with acute chemical eye injuries. Graefe’s Arch. Clin. Exp. Ophthalmol. 261, 3541–3547. https://doi.org/10.1007/s00417-023-06151-9 (2023).
Ahmad, M. S. Z., Baba, M., Pagano, L., Romano, V. & Kaye, S. B. Use of dried amniotic membrane with glue to manage a corneal perforation. Eye 36, 894–895. https://doi.org/10.1038/s41433-021-01558- (2022).
Johnson, T. S. et al. Photochemical tissue bonding: a promising technique for peripheral nerve repair. J. Surg. Res. 143, 224–229. https://doi.org/10.1016/j.jss.2007.01.028 (2007).
Henry, F. P. et al. Improving electrophysiologic and histological outcomes by photochemically sealing amnion to the peripheral nerve repair site. Surgery 145, 313–321. https://doi.org/10.1016/j.surg.2008.11.005 (2009).
O’Neill, A. C. et al. Photochemical sealing improves outcome following peripheral neurorrhaphy. J. Surg. Res. 151, 33–39. https://doi.org/10.1016/j.jss.2008.01.025 (2009).
Fairbairn, N. G. et al. Light-activated sealing of acellular nerve allografts following nerve cap injury. J. Reconstr. Microsurg. 32, 421–430. https://doi.org/10.1055/s-0035-1571247 (2016).
Senthil-Kumar, P. et al. A light-activated amnion wrap strengthens colonic anastomosis and reduces peri-anastomotic adhesions. Lasers Surg. Med. 48, 530–537. https://doi.org/10.1002/lsm.22507 (2016).
Fernandes, J. R. et al. Prevention of capsular contracture with photochemical tissue passivation. Plast. Reconstr. Surg. 133 571 – 577. https://doi.org/10.1097/01.prs.0000438063.31043.79 (2014).
Ludvikova, L. et al. Photochemistry of rose bengal in water and acetonitrile: a comprehensive kinetic analysis. Phys. Chem. Chem. Phys. 18, 16266–16273. https://doi.org/10.1039/c6cp01710j (2016).
Zhu, H., Alt, C., Webb, R. H., Melki, S. & Kochevar, I. E. Corneal crosslinking with rose bengal and green light: efficacy and safety evaluation. Cornea 35, 1234–1241. https://doi.org/10.1097/ICO.0000000000000916 (2016).
Goldstone, R. N. et al. Photochemical tissue passivation attenuates AV fistula intimal hyperplasia. Ann. Surg. 267 (1), 183–188. https://doi.org/10.1097/SLA.0000000000002046 (2018).
Alarcon, E. I. et al. Rose Bengal binding to collagen and tissue photobonding. ACS Omega. 2, 6646–6657. https://doi.org/10.1021/acsomega.7b00675 (2017).
Soeken, T. A. et al. Sealing of corneal lacerations using photoactivated rose Bengal dye and amniotic membrane. Cornea 37 (2), 211–217. https://doi.org/10.1097/ICO.0000000000001389 (2018).
Martinez, J. D. et al. Rose Bengal photodynamic antimicrobial therapy: a pilot safety study. Cornea 40 (8), 1036–1043. https://doi.org/10.1097/ICO.0000000000002717 (2021).
Verter, E. E. et al. Light-initiated bonding of amniotic membrane to cornea. Invest. Ophthalmol. Vis. Sci. 52 (13), 9470–9477 (2011).
Zarei-Ghanavati, M. Rose Bengal-green light for collagen cross-linking. J. Ophthalmic Vis. Res. 12 (2), 241–242. https://doi.org/10.4103/jovr.jovr_70_17 (2017).
Abbaszadeh, M., Aldavood, S. J., Foroutan, A. R., Azizzadeh, M. & Selk, G. M. Effects of sutureless amniotic membrane patching with 2-Octyl cyanoacrylate (Dermabond) on experimental corneal alkali burn in dogs. Comp. Clin. Path. 19, 357–362. https://doi.org/10.1007/s00580-009-0877-9 (2010).
Mittal, M., Tidake, P. & Kumar, M. Emerging strategies in treating corneal alkali burns: a narrative review. Cureus 15 (10), e47662. https://doi.org/10.7759/cureus.47662 (2023). PMID: 38021904; PMCID: PMC10670556.
Gao, X. W., Zhao, X. D., Li, W. J., Zhou, X. & Liu, Y. Experimental study on the treatment of rabbit corneal melting after alkali burn with collagen cross-linking. Int. J. Ophthalmol. 5 (2), 147–150. https://doi.org/10.3980/j.issn.2222-3959.2012.02.06 (2012).
Wang, T. et al. Subacute effects of rose Bengal/Green light cross linking on rabbit thin corneal stability and safety. Lasers Surg. Med. 50 (4), 324–332. https://doi.org/10.1002/lsm.22762 (2018).
Zhao, H., Brown, P. H. & Schuck, P. On the distribution of protein refractive index increments. Biophys. J. 100, 2309–2317 (2011).
Patel, S., Marshall, J. & Fitzke, F. W. Refractive index of the human corneal epithelium and stroma. J. Refract. Surg. 11 (2), 100–141 (2013).
Long, Q. et al. A novel tissue-engineered corneal epithelium based on ultra-thin amniotic membrane and mesenchymal stem cells. Sci. Rep. 14 (1), 1–15. https://doi.org/10.1038/s41598-024-68219-8 (2024).
Sha, X., Liu, Z., Song, L., Wang, Z. & L., X. Human amniotic epithelial cell niche enhances the functional properties of human corneal endothelial cells via inhibiting P53-survivin-mitochondria axis. Exp. Eye Res. 116, 36–46 (2013).
Arora, B., Tandon, R., Attri, P. & Bhatia, R. Chemical crosslinking: role in protein and peptide science. Curr. Protein Pept. Sci. 18 (9), 946–955. https://doi.org/10.2174/1389203717666160724202806 (2017).
Kochevar, I. E. & Redmond, R. W. Photosensitized production of singlet oxygen. Methods Enzymol. 319, 20–28. https://doi.org/10.1016/s0076-6879(00)19004-4 (2000).
Spoerl, E., Wollensak, G. & Seiler, T. Increased resistance of crosslinked cornea against enzymatic digestion. Curr. Eye Res. 29 (1), 35–40. https://doi.org/10.1080/02713680490513182 (2004).
de Almeida Manzano, R. P., Naufal, S. C., Hida, R. Y., Guarnieri, L. O. & Nishiwaki-Dantas, M. C. Antibacterial analysis in vitro of ethyl-cyano-acrylate against ocular pathogens. Cornea 25 (3), 350–351. https://doi.org/10.1097/01.ico.0000183490.16131.e3 (2006).
Erel, O. A novel automated method to measure total antioxidant response against potent free radical reactions. Clin. Biochem. 37, 112–119. https://doi.org/10.1016/j.clinbiochem.2003.10.014 (2004).
Harma, M., Harma, M. & Erel, O. Oxidative stress in women with preeclampsia. Am. J. Obstet. Gynecol. 192, 656–657. https://doi.org/10.1016/j.ajog.2004.07.094 (2005).
Yeni, E. et al. Comparison of oxidative/antioxidative status of penile corpus cavernosum blood and peripheral venous blood. Int. J. Impot. Res. 17, 19–22. https://doi.org/10.1038/sj.ijir.3901262 (2005).
Koksal, H. & Kurban, S. Total oxidant status, total antioxidant status, and paraoxonase and arylesterase activities during laparoscopic cholecystectomy. Clinics 65, 285–290. https://doi.org/10.1590/S1807-59322010000300008 (2010).
Abdelkawi, S. A. & Hassan, A. A. Evaluation of protein change and oxidative stress index after photodynamic therapy of corneal neovascularization. Gen. Physiol. Biophys. 31, 449–455. https://doi.org/10.4149/gpb_2012_045 (2012).
Rabus, M. et al. Plasma and tissue oxidative stress index in patients with rheumatic and degenerative heart valve disease. Arch. Turk. Soc. Cardiol. 36, 536–540 PMID: 19223719. (2008).
Lowry, O. H., Rosebrough, N. J., Farr, A. L. & Randall, R. J. Protein measurements with the Folin phenol reagent. J. Biol. Chem. 193, 256–275. https://doi.org/10.1016/S0021-9258(19)52451-6 (1951).
Dash, H. R., Shrivastava, P. & Das, S. Quantification of DNA by using agarose gel electrophoresis technique. In Principles and Practices of DNA Analysis: A Laboratory Manual for Forensic DNA Typing. New York, USA: Springer Protocols Handbooks, Springer Nature; 119–125. DOI: https://doi.org/10.1007/978-1-0716-0274-4_15. (2020).
Aebi, H. Catalase in vitro. Methods Enzymol. 105, 121–126. https://doi.org/10.1016/S0076-6879(84)05016-3 (1984).
Koracevic, D., Koracevic, G., Djordjevic, V., Andrejevic, S. & Cosic, V. Method for the measurement of antioxidant activity in human fluids. J. Clin. Pathol. 54, 356–361. https://doi.org/10.1136/jcp.54.5.356 (2001).
Demirbag, R. et al. Influence of oxidative stress on the development of collateral circulation in total coronary occlusions. Int. J. Cardiol. 116, 14–19. https://doi.org/10.1016/j.ijcard.2006.02.012 (2007).
Acknowledgements
Not applicable.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Open access funding.
Provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
S.A., S.M., A.A., D.F., M.E.: experiment design; S.A.: data collection, analysis and interpretation; S.A.: manuscript drafting, critical revision of the manuscript; S.M., S.A., A.A., D.F., M.E. read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical statement
The Institutional Animal Care and Use Committee, Cairo University (CU-IACUC) approved the study with approval no. 1/F/30/19. All experiments were performed in accordance with the relevant guidelines and regulations. All methods were conducted in compliance with the ARRIVE guidelines. We ensured animal welfare and minimalized animal suffering during the experiment.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mohammad, S., Abdelkawi, S., Ebrahim, M. et al. Corneal protein repair after amniotic membrane photo-tissue bonding versus amniotic membrane graft in the treatment of corneal ulcer (an experimental study). Sci Rep 14, 30561 (2024). https://doi.org/10.1038/s41598-024-81266-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-81266-5