Abstract
Studies of in situ plant response and adaptation to complex environmental stresses, are crucial for understanding the mechanisms of formation and functioning of ecosystems of anthropogenically transformed habitats. We study short- and long-term responses of photosynthetic apparatus (PSA) and anti-oxidant capacity to complex abiotic stresses of common plants Calamagrostis epigejos and Solidago gigantea in semi-natural (C) and heavy metal contaminated habitats (LZ). We found significant differences in leaf pigment content between both plant species growing on LZ plots and their respective C populations. The average values of leaf chlorophyll indexes were 27% lower in the LZ populations of both species and significantly lower in Sg plants in comparison to Ce ones. The average values of the anthocyanin index in CeLZ and SgLZ populations were significantly higher (by 18%) than in their respective controls. In both Ce and Sg plants occurring on LZ plots, the average leaf flavonol indexes were higher than on their controls by 31% and 15% and this index was significantly higher in SgLZ population than CeLZ and CeC plants (by 34% and 54%, respectively). Both Ce and Sg populations growing on LZ plots showed significantly lower photosynthetic rate (A), transpiration rate (E) and stomatal conductance (gs) in comparison to controls. On the other hand, a significantly higher photosynthetic rate was detected in SgLZ than in CeLZ populations. The catalase activities were significantly higher than recorded in Sg than in Ce tissues, irrespective of the plot type. They were also higher in LZ populations than those in controls for both species. Moreover, the H2O2 content in Sg tissues was significantly higher than those in Ce. Hydrogen peroxide content in CeLZ and SgLZ were respectively 39% (non-significant) and 57% higher, compared to their controls. The reverse pattern was found in the case of MDA, whose concentration was significantly higher in the leaves of Ce population compared to the control population. The average MDA concentration in CeLZ populations was 17% higher than in the CeC. In the case of Sg no significant differences were found. Mechanisms of plant species adaptation to industrial areas are crucial for species selection and planning effective reclamation of them. The analysis of chlorophyll fluorescence induction curves as well as well as the results of JIP test revealed the decreased of Fj value despite positive ΔK–band in SgLZ and CeLZ plants suggesting the increased rate of electron transfer from QA to QB at the acceptor side of PSII, thus a high quantity of P680+ and/or effective quenching by exogenous molecules. The increase in the I–P part of the induction curve typically attributed to the reduction of electron transporters (ferredoxin, intermediary acceptors, and NADP) of the PSI acceptor side was observed in both SgC and SgLZ but not in CeLZ populations. These changes demonstrate species-specific effects on electron transport during the light phase of photosynthesis under complex environmental stress. Our results show that Sg and Ce individuals developed a range of structural and functional adaptations to protect PSA against complex environmental stresses (possible combination of heavy metals, water deficiency, temperature, nutrient deficiency and salinity). Both species from LZ plots could tolerate high levels of Cd, Zn and Pb in leaf tissues. Therefore they can be potential candidates for use in phytoremediation of HM contaminated areas. However, further long-term field and experimental research on plant traits response and adaptation to complex environmental stresses on industrial habitats are needed.
Similar content being viewed by others
Introduction
Mining and processing of zinc and lead ores appeared to be a severe environmental and social problem1,2,3,4. This heavy industry contributes to significant changes in land relief, water relations, air pollution, and soil degradation and therefore to deterioration of health and quality of life for residents of nearby settlements. Because of storing a huge amount of waste, novel ecosystems of aboveground, flat flotation pools or artificially created ponds for flotation processes are formed. The wastes, stored in the flotation settling tanks are characterized by unfavourable but often unique physical and chemical properties and unfavourable air–water relations and structure that influence soil-forming processes. The resulting substrate is often nutrient-poor and has a higher pH and elevated concentrations of heavy metals (further referred to as HM). The plants that spontaneously colonize such sites, apart from HM stress, must often face a harsh environment in which, they are exposed to periodic water deficiency, heat waves and high-temperature periods, or salinity5.
For optimal growth and development in a given environmental conditions, plant species need a specific combination of resources, such as temperature, humidity, light spectrum and intensity, water, and mineral nutrients6,7. Any deviation from those optimal states can be considered environmental stress. In natural conditions, multiple fluctuating environmental stresses such as low or high temperature, low or high irradiation, drought or waterlogging or suboptimal mineral nutrients status in the soil operate together forcing plants to develop the complex short-term and long-term responses: acclimations to adaptations8,9,10,11. For example, our previous research on wastelands of Carboniferous gangue confirmed that species response was mainly shaped by a combination of temperature, water deficiency and salinity8.
Environmental stresses affect the plant in a variety of ways and at different levels of organization12. Drought is one of the most important and prevalent stress factors for plants, especially in arid and semiarid environments as well as anthropogenically changed urban areas13. Water deficit in plants is shaped mainly by low rainfall, salinity, high and low temperatures, and high intensity of light. In many cases, there is enough water in the soil, but plants cannot uptake it. The resulted osmotic stress can lead to an impairment of amino acid synthesis, protein metabolism, and the dark reaction of photosynthesis or respiration and therefore can result in a breakdown of the osmotic system of cells6,14. High temperature and heat weaves reduce anti-oxidant enzyme activity, further enhancing membrane damage and leading to an imbalance between photosynthesis and respiration15. On the other hand, salinity stress causes various physiological changes (interruption of membranes, nutrient imbalance, impairs ability to detoxify reactive oxygen species, differences in the anti-oxidant enzymes activity, and decreased in photosynthetic activity). Inhibition of the uptake of K+ ions, an essential element for growth and development, results in lower productivity and may even lead to plant death16.
Heavy metals belong to the natural components of the environment, but they become a pollutant when their natural content is exceeded17. The industry is considered by far the most important source of pollution of urban soils18 as well as accidents, dust emissions, improper waste handling, and increased automotive emissions. Metals from anthropogenic sources are characterised by higher solubility than metals of lithogenic origin19,20. Soil pH affects the mobility of metals in the soil environment. In neutral and alkaline soils, the solubility of heavy metals is usually low and increases with decreasing pH of the soil. In acidic soils, heavy metals are released from the solid phase and move to the soil solution21,22. The acid and very acid reaction of contaminated soils increases the mobility of heavy metals and their participation in the biogeochemical cycle23. Plants reduce the mobility and bioavailability of HM through sorption, sequestration in the vacuole, and precipitation (secretion of chelating agents in soil such as histidine, organic acids, and complex reactions)24,25. Toxicity of heavy metals at the molecular level is expressed, among others, on their interaction with thiol functional groups of several enzymes, including antioxidant enzymes, inhibition or interference with the function of, e.g. DNA polymerase, RNA polymerase, ATPase, malic acid synthesis enzymes and many others26. The toxicity of trace metals (Cu, Cr, Ni, Cd, Co, Pb, Zn) results from the fact that they substitute specific metals in protein structures and other biomolecules (e.g. Mg in chlorophyll, Ca in calmodulin)17. They are also characterised by preferential binding to O, N, and S of many molecules, as well as by their interactions with functional groups, e.g. with phosphate ADP and ATP or with carboxyl groups. Consequently, this disrupts the functioning (including inactivation) of bio-molecules, and negatively affects protein metabolism, Ca homeostasis, photosynthetic pigment biosynthesis or heme biosynthesis. Toxic metals cause closure of stomata, decrease in the number and volume of chloroplasts, hydrolysis of chlorophyll, decrease in ATP production, or increase in cell wall stiffness. Metals present in the cell in toxic amounts modify cell ultrastructure27,28. Trace metals (Zn, Pb, Cd, Cu, Hg, Cr, Co, Ni, Be) also affect cell nuclei and cell division: they cause a decrease in mitotic activity and disruption of cytokinesis, DNA and RNA damage, decrease in transcriptional activity, chromatin condensation, chromosome aberrations, destruction of nuclear envelope and decrease in nuclei volume.
Previous studies confirmed that plants spontaneously colonizing carboniferous spoil heaps developed several short- and long-term morphological (growth form, canopy height, leaf length and width, leaf colour, type of root system, increase in the number of root hairs), anatomical (e.g., changes in the structure of palisade and spongy soft tissues, in the structure of the root system), phenological, physiological, biochemical and genetic acclimatizations/adaptations to cope with environmental stresses29,30,31. Some adaptations to xeric conditions and strong winds are connected with shortening and reducing the number of shoots30. Plants colonizing brownfield sites can be tolerant of high concentrations of heavy metals in the soil through reduced transport to the aboveground parts and synthesis of some compounds such as metallothioneins and phytochelatins32 and various mechanisms, including metal immobilization, sequestration, and activation of repair systems33,34.
Multiple stress factors like drought, salt, and heavy metals share a common consequence in plants, they lead to the excessive production of reactive oxygen species (ROS), which in turn causes oxidative stress. While the sources of stress differ (water limitation for drought, ion toxicity for salt, and toxic elements for heavy metals), the physiological and biochemical impacts on plants tend to converge, particularly in terms of ROS generation and antioxidant responses35,36. Plants deal with oxidative stress via an endogenous defensive mechanism consisting of enzymatic (i.e. superoxide dismutase, SOD; catalase, CAT; ascorbate peroxidase, APX; glutathione reductase, glutathione peroxidase, GPX; and nonenzymatic (ascorbic acid, AsA; glutathione, GSH; phenolic acids; alkaloids; flavonoids; carotenoids; α-tocopherol; nonprotein amino acids; etc.) antioxidants37,38,39. In plant cells, the antioxidant defence system and ROS accumulation uphold a steady-state balance. Maintaining an optimum ROS level in the cell enables proper redox biology reactions and the regulation of numerous processes essential for plants such as growth and development40. The balance between ROS production and ROS scavenging maintains this intermediate level. However, during stress conditions, overgeneration of ROS demolishes the equilibrium and causes cellular damage, leading to programmed cell death (PCD) as well as decreasing plant productivity41. Overproduction of ROS in the presence of complex environmental stresses is primarily generated in key cellular organelles like chloroplasts42. However, ROS are also produced in other areas, such as membrane-bound exocellular enzymes involved in redox reactions during photosynthetic electron transport42.
Photosynthesis is one of the most stress-sensitive physiological processes in plants43. Water deficiency, salinity, high temperature, and elevated heavy metal content in the soil all have a greater influence on the photosynthetic capacity of plants44. Chlorophyll a fluorescence, in addition to biochemical and gas exchange methods, is confirmed as a reliable, technique for the assessment of electron transport and related photosynthetic processes45. For a quick assessment of the photosynthetic function in a high number of samples in the field conditions, a non-destructive analysis of fast chlorophyll transient was developed44,46. This method is based on the high-frequency recording of chlorophyll fluorescence (ChlF) emitted by dark pre-adapted leaf samples during the pulses of strong actinic light by a fluorometer. The fast fluorescence kinetics obtained in this way provide information on the structural and functional state of the photosystems, mainly PSII47,48. The mathematical model called the JIP test, describes this polyphasic transient and enables the calculation of parameters, quantum yields, and probabilities which give insight into PSII and PSI functions45,49,50. Numerous studies have demonstrated the usefulness of the JIP test in uncovering the differences in the chlorophyll fluorescence transient among plant genotypes, varieties, or mutants under controlled51 and field conditions8,52,53,54. Moreover, strong changes in ChlF transients have been observed in plants under different types of environmental stress such as high and low temperature55,56,57, drought58,59, nutrient deficiency60,61, salinity62,63 and heavy metals64,65. Moreover, ChlF is used in ecological52,53, climate change66,67 and vegetation research68,69.
We selected two species representing different functional groups, commonly occurring and spontaneously colonizing lead and zinc spoil heaps as well as seminatural habitats: perennial C3 grass Calamagrostis epigejos (L.) Roth and perennial C3 forb Solidago gigantea Aiton. Both species can form extensive and almost monodominant patches 70,71.
Calamagrostis epigejos (Ce) is a tall monocot belonging to the Poaceae family. It has a broad circumboreal distribution in the temperate zone of Eurasia. A distinct variety is found in southern and eastern Africa72,73. It reproduces both generatively and vegetatively forming the rhizomes up to 100 cm. Ce usually grows on open, sunny, with moderate moisture, and organic matter sites74. It occurs in a variety of habitats: forests75, thickets, clearings, meadows, xerothermic grasslands74, as well as on ruderal and post-industrial sites (spoil heaps, sandpits, quarries)75. Solidago gigantea Aiton (Sg) is a herbaceous perennial species belonging to the Asteraceae family. Its native distribution range is within non-arctic North America east of the Rocky Mountains, however, the species is invasive throughout Europe and Asia70. It reproduces both generatively by enormous quantities of tiny seeds and vegetatively by rhizomes up to 90 cm in length70. The species has broad tolerance to a range of environmental conditions (i.e. soil moisture, light, nutrient contents, temperature, pH, soil texture)70. In North America, Sg grows mainly on fertile, moist soils, such as river valleys, humid forests and thickets, and meadows. In Europe, it occurs in similar habitats but also in drier, anthropogenic habitats with less fertile soils (e.g. roadsides, fallows, forest edges, post-industrial habitats)76.
In many studies reactions of these species to abiotic stresses were reported (e.g. drought, heat, nutrient deficiency, heavy metals)77,78,79,80,81. The effect of toxic metals (Zn, Pb, Cd) on photosynthetic electron transport has also been demonstrated82. Our previous research focused on the response of species of the Solidago gigantea and Calamagrostis epigejos to complex environmental stresses acting on Carboniferous waste rock dumps. They showed that both species have different mechanisms of the stress response (changes in PSI, pigment content, the content of stress markers and enzymes), but are effective enough to allow them to survive in unfavourable conditions (drought, salinity, temperature) and maintain dense cover. However, to our knowledge, there are no such comprehensive studies on the impact of complex environmental stress in lead and zinc flotation tailings on the PSA of Ce and Sg in natural conditions. In this work, we study short- (leaf gas exchange parameters, chlorophyll fluorescence parameters, oxidative stress markers) and long-term (leaf metal content, leaf pigment content) responses of photosynthetic apparatus (PSA) to complex abiotic stresses (possible combination of drought, temperature, heavy metals, salinity and nutrient deficiency). We hypothesize, that these reactions are species-specific and would differ between grasslands and novel ecosystems of post-industrial sites (lead and zinc flotation tailings). They can be detected in photosynthetic apparatus status, as measured by chlorophyll fluorescence, leaf pigment content, gas exchange and activity of oxidative stress markers.
Materials and methods
Site description
For a study area, we have chosen “Żabie Doły” in Bytom-region situated in the Silesian Upland (southern Poland, 50°20′21′′ N, 18°57′7′′ E). The exploitation of lead and zinc ores started here in the 12 century at a depth of 50–100 m. The wastelands (tailing pools, ruderal lands) occupied an area of 145 ha. The excavated ores were subjected to enrichment by flotations, and wastes were stored on aboveground table settling tanks. The area of the settling pool reached 10 ha. In the Bytom area, the galman and sphalerite ores are particularly abundant. The “galmans” consist of a mixture of Zn- and Pb-carbonates, dolomite, cerussite, Pb-rich aragonite, goethite, hemimorphite, and galena83. Nonsulfide Zn ores occurred in the Bytom trough (Nowy Dwór, Orzeł Biały, Waryński, Marchlewski, Dąbrówka mines). The waste material consists of finely ground fragments of dolomite, limestone, clay and gravel. The substrate has unfavourable air-water relations, is poor in nutrients, alkaline, and saline, and is susceptible to water and wind erosion. It contains up to 8.0% of Zn, 1.5% of Pb, and Cd in content, usually over 100 mg kg−184. The object can be treated as a model for studying the formation and dynamics of novel ecosystems of HM-polluted areas.
The control plots were located in the northern part of the Silesian Upland (The Garb Tarnogórski mesoregion) on the territory of Świerklaniec municipality (50°26′26′′ N, 18°57′38′′ E) (Supplementary Table S1). The vegetation in this area is of natural and seminatural origin. The control plots were established in wet and moderately wet meadows localized inside mid-forests glades. These meadows are regularly managed by mowing.
The Silesian Upland lies within the temperate climate zone. The mean annual rainfall is 600–800 mm, the monthly minimum temperature is in January −2 °C, while the maximum is in July 14–16 °C. The cloud cover ranges from 60 to 80%85. The detailed temperature and precipitation data for the study area are provided in Supplementary Figure S1).
Vegetation and soil sampling
Native grass species Calamagrostis epigejos (L.) Roth (Ce) and alien herb Solidago gigantea Aiton. (Sg) occur commonly and abundantly on a variety of urban and post-industrial wastelands. Frequently, they play the role of dominants and form stable plant communities, particularly in further stages of succession86,87,88.
In July 2021, 40 4 × 4 m2 sample plots were established in two habitat types: 20 in the post-industrial sites (lead and zinc flotation tailings) and 20 in grasslands (control plots) to compare the response of native grass versus alien forb to complex environmental stresses in natural condition. On each type of habitat, 10 plots were located in Calamagrostis epigejos-dominated plots and 10 for Solidago gigantea-dominated vegetation (i.e. we chose patches with an abundance of Ce and Sg at least 60% of coverage in the study plots) (Supplementary Table S1). They were further marked as CeLZ, SgLZ in case of patches occurring on the lead and zinc flotation tailings, and CeC, SgC for plots that were established in grasslands (control plots). The study plots were established on flat or slightly inclined terrain, in order to make the plots as comparable as possible. The detailed description of sample plots with Calamagrostis epigejos and Solidago gigantea was included in Supplementary Table S1. The percentage cover of each vascular plant species was estimated according to the following scale: 1, 2, 5, 10%, and then at 10% intervals89. One composite substrate sample per sample plot was taken from three rhizosphere zones of Sg or Ce individuals at a depth of 10 cm. Next, soil samples were placed into plastic bags and transported to the laboratory for detailed physico-chemical analyses. Moisture was determined in situ using ML3 ThetaKit soil moisture portable sensor (Delta-T Devices, United Kingdom)90.
Substrate and plant physicochemical analyses
After the soil/substrate samples had been transported to the laboratory they were air-dried to constant weight. Initially, all soil samples were sieved through 2 mm mesh in order to remove skeletal parts. In the case of soil nitrogen total and heavy metal content analyses, we used 0.25 mm mesh. Following physicochemical analyses were performed as substrate/soil pH, in 1 M KCl (potential acidity) and water suspension (pH in H2O) (actual acidity), electrical conductivity (EC) (substrate to solution ratio 1:2.5), measured after 24 h of equilibration; substrate/soil organic carbon content (SOC) determined by the Tiurin method modified by Simakov and total N (TN) by the Kjeldahl method. The content of available forms of phosphorus (P2O5) was estimated according to the Polish Norm PN-R-04023:1996 based on the Egner–Riehm method. The concentration of available Mg was measured by spectrometric analysis90. To determine heavy metals (HMs) concentration in both substrate and plant material was acid-digested in a microwave-assisted wet digestion system using 65% HNO3 and 30% H2O2, 4:1, v/v) (ETHOS 1, Milestone, Sorisole, Italy). The concentration of bioavailable forms of HM was analysed in the extracts (soil, 0.1% CaCl2) and digests (plant, soil) using flame atomic absorption spectrophotometry (iCE 3500 FAAS, Thermo Fisher Scientific, Waltham, MA, USA).
Parameters of oxidative stress
The 5 individuals per sample plot of Ce or Sg were collected at each type of habitat and transported immediately to the laboratory. Next, 20 mature leaves per sample plot were collected for further analyses. 100 leaf samples per habitat type were analyzed in total. The level of lipid peroxidation was determined by the thiobarbituric acid (TBA) reaction according to the methodology described by91 with minor modifications. Fresh leaf tissues (200 mg) were homogenized in 4 ml 0.25% TBA in 10% trichloroacetic acid (TCA). The homogenate was incubated in a water bath at 95 °C for 30 min and then cooled in an ice bath. After centrifugation at 10,000 g for 10 min, the absorbance of the supernatant was measured at 532 nm and corrected by subtracting the non-specific absorbance at 600 nm. The concentration of malondialdehyde (MDA) was calculated using an extinction coefficient of 155 mM−1 cm−1. To determine hydrogen peroxide concentration fresh leaf tissues (200 mg) were homogenized in 1.5 ml of 0.1% TCA. The homogenate was centrifuged at 12,000 g for 15 min and 0.5 ml of the supernatant was added to 0.5 ml of 10 mM potassium phosphate buffer (pH 7.0) and 1 ml of 1 M potassium iodide (KI). The absorbance of the supernatant was measured at 390 nm, and the content of H2O2 was obtained using a standard curve91.
Fresh leaf tissues (200 mg) were homogenized in 1.5 ml of 10 mM potassium phosphate buffer (pH 7.0) to determine the catalase activity. The homogenate was centrifuged at 12,000 g for 20 min. The supernatant (20 µl) was added to 2 ml of 10 mM peroxide prepared in potassium phosphate buffer. Catalase activity was determined spectrophotometrically by monitoring the changes in absorbance caused by H2O2 reduction at 240 nm91.
The content of chlorophyll a fluorescence and pigment index
We used Dualex Scientific + leaf clip (Force-A, France) sensor for non-destructive measurement in situ chlorophyll content, flavonol index, and anthocyanins index in leaves92,93. Dualex measures the flavonols and anthocyanins content of the leaves’ epidermis thanks to a differential ratio of chlorophyll fluorescence. The content of them is given in relative absorbance units from 0 to 3 for flavonols and 0 to 1.5 for anthocyanins. The measurement of the chlorophyll content is based on the transmittance at two specific wavelengths. One wavelength is in the far-red region , which is absorbed by chlorophyll, while the other is in the near-infrared region, serving as a reference. The chlorophyll is given in μg cm-2.
Chlorophyll a fluorescence measurements were performed on the same leaves using Plant Efficiency Analyses (Pocket PEA fluorimeter, Hansatech Ltd. Pemtney, UK). Before analyses, each leaf was dark-adapted for 30 min using leaf clips. On 20 plants from each population the ChlF was measured in four periods of the day (8:00–10:00; 11:00–12:00, 14:00–16:00, 17:00–19:00 h), to take into account the short-term diurnal responses of plants to environmental stresses. For induction of fluorescence red actinic light was used (wavelength at peak 650 nm; spectral line half-width 22 nm) with the intensity of 3500 μmol m−2 s−1, and 1 s of transient fluorescence was recorded50,58,94,95,96,97. The fluorescence signal was collected with a maximum frequency of 105 points s−1 (each 10 μs) within 0–0.3 ms, after which the frequency of recording gradually decreased, collecting a total of 118 points within 1 s.
The FO level was measured as the fluorescence at 50 μs. The collected data were used for the calculation of basic parameters, while the fluorescence intensities determined at O—50 μs, J—2 ms, I—30 ms and maximum fluorescence, P ~ 300 ms (FM) were used for the calculation of the OJIP test parameters, related to specific (per reaction center) and phenomenological energy fluxes (per excited tissue cross-section; definitions of ChlF parameters are provided in Supplementary Table S2)98,99.
Gas-exchange parameters
A portable gas exchange infrared analyser (TARGAS—1, PP Systems, Amesbury, USA) was used to measure in situ three gas exchange parameters: net photosynthetic rate (A), stomatal conductance (gs), and transpiration rate (E) in fully developed leaves. The light intensity was set to 1500 μmol m−2 s−1, the air flow supply—to 250 cc min−1, the sample flow—to 150 cc min−1 and CO2 concentration in the chamber fluctuated around 400 ppm. Measurements were performed for each leaf after a three-minute acclimatization in the chamber (Sitko et al. 2019).
Statistical analysis
We used ANOVA and Tukey’s post-hoc test to detect the differences in physicochemical substrate parameters, chlorophyll fluorescence, pigments, oxidative stress markers, and gas-exchange parameters between plots with a dominance of Ce or Sg on flotation tailings (CeLZ, SgLZ) versus control plots CeC, SgC). The normality of the variables was examined with the Shapiro–Wilk test and transformed where needed. Analyses were conducted in Statistica 13.1.100. Principal component analysis (PCA) was used to summarize relationships between the possible environmental stressors (HM, high temperature, moisture, salinity, nutrient deficiency), pH(KCl), C/N, selected ChlF parameters, leaf pigment composition, and oxidative stress parameters in the two study species, growing on lead and zinc flotation tailings and control sites. Before the analysis, the data were standardised. The groups of individuals, related to different species, growing on lead and zinc flotation tailings and control sites were marked in the ordination diagram with 95% confidence interval ellipses. The analyses were performed in the R v. 4.0.3101, packages vegan102 and the results were visualised in ggplot2103.
Results
Physico-chemical substrate and plant parameters
The substrate in the CeLZ and SgLZ plots has on average, significantly higher pH (in H2O and KCl), electrical conductivity (EC), Ca2+, Mg2+, MgO, Na+ in comparison to control plots (Table 1). Moreover, on these plots significantly higher HM contents such as Cd, Pb, and Zn in total and their bioavailable forms were detected, than in grasslands (CeC and SgC, Table 2). The substrate HM total content in LZ plots exceeds those found on grasslands by 22–26, 21–38, and 27–43 times in the case of Cd, Pb and Zn respectively (Table 1). Additionally, the average HM content absorbed by plant tissue differed significantly between CeLZ and SgLZ and their respective C plots by 1.7–2 times in the case of Cd and Zn and 2–6 times in the case of Pb. On the other hand, lower average substrate moisture was found on LZ plots. The substrate content of available phosphorus (P2O5) was lower in the Ce-dominated plots and did not differ significantly between C and LZ plots in both species (Table 1).
Gas-exchange parameters
The Ce and Sg populations from LZ plots showed significant differences in photosynthetic gas-exchange parameters in comparison to C plots. They showed a lower photosynthetic rate, A (by 58.7% and 32.2% in the case of Ce and Sg, respectively). Moreover, we found significantly higher photosynthetic rate in Sg vs. Ce populations on LZ plots and no differences between C plots (Fig. 1A). Stomatal conductance (gs) of Ce and Sg plants from LZ populations was significantly lower, compared to control populations by 57% and 64% respectively. Moreover, gs was on average 23% lower in Ce in comparison to Sg on LZ plots and 35% lower on C plots (Fig. 1B). Similar pattern to A was found in transpiration rate: plants of CeLZ and SgLZ populations showed significantly lower E (60% and 58% respectively) as C populations (Fig. 1C).
Chlorophyll a fluorescence and pigment analysis
We found significant differences in leaf pigment content between both plant species growing on LZ plots and their respective C populations (Fig. 2). The average values of anthocyanin index in CeLZ and SgLZ populations were significantly higher (by 18%) than in their respective controls (Fig. 2A). On the other hand, the chlorophyll index was significantly lower in Sg plants in comparison to Ce ones (Fig. 2B). Moreover, in the LZ populations of both species, the average values of chlorophyll indexes were 27% lower compared to controls. In both Ce and Sg plants occurring on LZ plots, the average leaf flavonol indexes were higher than on their controls by 31% and 15%. The flavonol index in SgLZ population was significantly higher, than CeLZ and CeC plants (by 34% and 54%, respectively) (Fig. 2C).
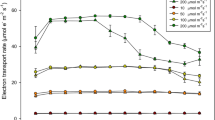
The differences in pigment indices and gas exchange parameters compared among control and lead–zinc spoil heaps in Ce and Sg populations were reflected in the state of light-dependent photosynthetic processes analysed by JIP test. The comparison of raw chlorophyll a fluorescence kinetics between populations (Fig. 3A–F, Table 3), revealed both species- and population-specific differences of amplitude, shape, and dynamic in time of the curves which reflected differences in acclimation of the studied species to complex environmental stresses (CES). The significantly lower maximum fluorescence value (FM) recorded in LZ in comparison to C populations is probably related to differences in structural leaf tissue characteristics, which influences actinic light absorption, reflection and reabsorption, decreased leaf chlorophyll content as a result of exposition to CES, and presence of chlorophyll fluorescence quenchers like carotenoids. On the other hand, decreasing FO on LZ plots indicates the lowering energy trapping efficiency of PSII (Fig. 3A, Table 3). There are significant differences in most chlorophyll fluorescence parameters between LZ and C populations of both species despite TRO/RC (Table 3). Moreover, tFM was significantly longer in CeC and CeLZ, but not in Sg populations. The Vj and VI values are significantly higher in both LZ populations, which could be related to the high availability of chlorophyll fluorescence quenchers or the formation of inactive PSII reaction centers as a result of the destruction of the oxygen-evolving complex (OEC).
The average (mean ± SE) chlorophyll fluorescence (ChlF) transients in 30 min dark-adapted leaves of Calamagrostis epigejos and Solidago gigantea from control (CeC, SgC) and lead and zinc spoil heaps (CeLZ, SgLZ) populations (A). Differential average ChlF curves (LZ-control) normalised between O–P (B) and O–K, O–J, J–I and I–P points (C–F). The ChlF transients were plotted on logarithmic scale from FO (50 μs) to 1 s. The time points important for the calculation of JIP test, marked on X-axis, were related to: O–fluorescence intensity recorded at FO (50 μs), L—at 150 μs, K—at 300 μs J–2 ms, I—at 30 ms, P—maximum fluorescence intensity (FM) at ca. 300 ms.
For a complete analysis of differences between the Ce and Sg plants in LZ and C populations, the OJIP curves were double normalized between O and P points (Fig. 3B). In the next step, differential curves for all populations were constructed by subtracting the controls, CeC. Moreover, the differential curves were constructed within different parts of induction, separately for ΔL, ΔK, ΔH and ΔG–bands by subtracting normalized fluorescence values between O–K, O–J, J–I and I–P respectively (Fig. 3C–F). The analysis of fluorescence transients confirmed small, but significant differences in the light-dependent O–K phase (negative ΔL—band in SgLZ and positive but insignificant in CeLZ, both at 15 µs). This points out more connectivity in PSII reaction centres in SgLZ to the rest of the population. Moreover, positive ΔK, ΔH and ΔG—bands were detected in both LZ plots, but in the case of CeLZ they are much less pronounced (Fig. 3E–F). Additionally, the times of the extremes for these bands are shifted among SgLZ and CeLZ plots (K: 30–80 µs, I: 7–15 ms and G: 120–150 ms, Fig. 3D–F). Interestingly, ΔK, ΔH and ΔG-bands are also visible on the SgC plot, possibly related to inter-specific differences in photosynthetic apparatus response. The increase in the I–P part of the induction curve was observed in both Ce, but not in Sg populations (Fig. 3F).
The analysis of the shapes of raw chlorophyll fluorescence kinetics within populations of both species (ChlF kinetics 1–4 on Fig. 4, Supplementary Tables S3–S6), revealed a wide range of physiological responses to CES, with an initial strong decrease of both FO and FM parameters in the midday with subsequent (not full) recovery afternoon. The comparison of differential curves constructed by subtracting fluorescence kinetics recorded in the morning period (8:00–10:00 am) from the subsequent ones, revealed steady-growing amplitudes of ΔK and ΔJ-bands in SgC and both LZ populations (Fig. 5). Initially, pronounced and positive, then negative ΔI-band and negative ΔG-band were detected in CeLZ, while the reverse pattern was found in the SgLZ.
Differential average ChlF curves (LZ-control) normalised between O–P points. The time changes of average chlorophyll fluorescence (ChlF) transients 30 min dark-adapted leaves of Calamagrostis epigejos and Solidago gigantea from control (CeC, SgC) and lead and zinc spoil heaps (CeLZ, SgLZ) populations. The differential curves were constructed by subtracting the morning (8:00–10:00 h).
The differences in ChlF induction curves in time were also confirmed by changes in average values of measured and calculated fluorescence parameters (Table 3). In LZ populations, an increase of average values ChlF parameters was found: tFM (in SgLZ), specific energy fluxes indices per one reaction center: trapping, TRO/RC, electron transport, EtO/RC, reduction of electron acceptors of PSI, ReO/RC, dissipation energy, DiO/RC, quantum yields (φDo γRC, δRo, φRo) and performance up to the reduction of PSI end electron acceptors (PITOT). In the same populations a decrease in time in the number of reaction centres per excited cross-section, RC/CSO and RC/ABS were observed (Table 3).
Oxidative stress parameters
The catalase activities in Sg populations were significantly higher than recorded in Ce tissues, irrespective of the plot type (Fig. 6A). Catalase activity was higher in LZ populations than those in controls for both species. These values, however, were statistically insignificant. Moreover, the H2O2 content in Sg tissues was significantly higher than those in Ce. Hydrogen peroxide content in CeLZ and SgLZ were respectively 39% (non-significant) and 57% higher, compared to their respective controls (Fig. 6B). The reverse pattern was found in the case of MDA, whose concentration was significantly higher in leaves of Ce population compared to Sg population. The average MDA concentration in Ce leaves in LZ populations was 17% higher than in the controls. In the case of Sg no significant differences were found between LZ and C plots (Fig. 6C).
Discussion
Studies of in situ plant response and adaptation to complex environmental stresses, are crucial for understanding the mechanisms of formation and functioning of ecosystems of anthropogenically transformed habitats. In natural conditions, plants may experience any type of single, interactive, cumulative, hierarchical, and sequential effects of biotic and/or abiotic stressor(s)104. Moreover, novel ecosystems developing on brownfield sites are often governed by distinct ecological and environmental rules105. The species compositions that arise in the course of succession on post-industrial sites are frequently based on one or two most abundant species and some accompanying ones75,106. Plants colonizing these areas often develop a range of acclimatizations/adaptations to complex environmental stresses operating in these areas89,107,108,109,110 and possess different sets of functional traits as a result of habitat filtering111,112. This fact provides an opportunity to explore the assembly rules in plant communities that develop on post-industrial sites as a result of human activity as well as different ecosystem functions of such novel ecosystems113,114,115.
Abiotic parameters of the lead and zinc spoil heaps
Post-industrial sites are characterised by abiotic conditions that differ or in some cases are even extreme compared to natural or semi-natural sites. The role of abiotic parameters in the formation of vegetation, soil-forming or microbiological processes occurring on brownfield sites has been repeatably highlighted116,117,118,119. Sites associated with the extraction and processing of zinc and lead ores are characterised by elevated concentrations of heavy metals. In our studies, the Cd, Zn, Pb content in the substrate were several times higher than those on the control areas (on average 24, 35 and 29 times for Cd, Zn and Pb total and 2, 1.8 and 1.1 times in case of their bioavailable forms respectively). Moreover, on LZ plots they exceeded on average 6 times the permissible standards in Poland adopted for these areas (Dz.U. 2016 poz. 1395). This is not the case with the Cu content in the soil. Soil pH influences the growth and development of plants, determines the solubility of heavy metals and uptake of nutrients by plants, influences the rate and direction of biological and physico-chemical processes in the soil, and determines the state of equilibrium of sorption and desorption processes of hydrogen and metal cations in the soil120. The substrate pH(KCl) recorded in both LZ plots was close to neutral and significantly higher in comparison to controls, which was related to the presence of ground dolomite or limestone fragments in the waste material. Moreover, contents of exchangeable cations (Ca2+, Mg2+) as well as MgO were higher in LZ substrate in comparison to control plots. The neutral or alkaline reaction of the substrate means that these metals tend to form phosphates and carbonates barely released from the solid phase into the soil solution121.
The important factor, that determines the bioavailability of metals, is soil organic matter content. An increase in soil organic matter content is related to their lower bioavailability due to their strong binding with organic matter of most metals (including Cu, Zn, Pb and Mn) to plants. For example, Nadgórska-Socha et al.122 explained the relatively low bioavailability of the Zn, Pb, to a high content of organic matter (30%) in the substrate, which changes them into biologically inactive forms123. However, in our study, the content of organic matter in the soil of LZ plots was significantly lower than in the control ones.
The serious problem of many post-industrial sites is connected with a lack of water accessible to plants, particularly in long-term water shortage during summer months. It has an adverse effect on the development of permanent vegetation cover. Soil moisture content is a parameter of high variability relative to the site, season, age, soil depth, and reclamation management124. This is in line with our results, where the lower average substrate moisture was found on LZ plots of both species in comparison to control plots. Moreover, the soil moisture was lower in CeC vs. SgC (but not significantly) and significantly lower in CeLZ in comparison to SgC. This trend seems to be the outcome of differences in growth form as Sg develop a much denser leaf canopy.
In our previous studies8 we did not find significant differences in soil P2O5 content between Ce and Sg-dominated coal-mine spoil heaps plots in comparison to control ones, while such differences were species-specific. The same pattern was detected in the present study, which confirmed that Ce as the grass can tolerate lower P in the soil than forbs125. The combination of lower soil moisture and elevated soil HM content may lead to an imbalance in essential nutrient acquisition and/or displacement of metals in biomolecules. For example, cadmium competes with Zn and P126.
Gas exchange parameters, pigment indexes, chlorophyll fluorescence parameters and species response to complex abiotic stresses on LZ spoil heaps
The complex environmental stresses acting LZ spoil heaps resulted mainly from a combination of soil elevated HM content, water and nutrient deficiency, high temperature and salinity, and affected photosynthetic apparatus of Sg and Ce. In agreement with studies of other post-industrial wastelands have shown, the response of species or their populations to them can vary and be manifested in both short-term and long-term31.
Both Ce and Sg populations growing on LZ plots showed significantly lower photosynthetic rate (A), transpiration rate (E) and stomatal conductance (gs) in comparison to controls (Fig. 1.). On the other hand, on LZ plots, a significantly higher photosynthetic rate was detected in the perennial herb Sg than in perennial grass Ce. In our previous studies110, conducted on Ce and Sg populations on coal mine spoil heaps, the combination of drought, salinity and elevated temperature stress factors generally resulted in a similar pattern, but the decrease in net photosynthetic rate (A), transpiration rate (E) and stomatal conductance (gs) on LZ plots relative to controls were less pronounced. Moreover, on coal mine spoil heaps Ce showed a higher net photosynthetic rate on both studied habitats (coal mine spoil heaps and control plots) in comparison to Sg. The reduction of photosynthetic rate is a common response in plants exposed to HM. The toxic effect of Cd on photosynthetic rate was reported for agricultural plants: lettuce127, barley128, rice129, maize130, mustard131, Picris divaricata Vaniot132, and tobacco133. In natural conditions, plants developed a range of adaptations to elevated soil HM contents. In experimental research on Arabidopsis species, heavy metals (Zn, Cd) caused a considerable decrease in the photosynthetic rate (A) in Arabidopisis arenosa (L.) Lawalrée (38 and 49% of control for Cd and Zn, respectively). In control conditions, the transpiration rate (E) of A. arenosa was twice that of A. halleri (L.) O'Kane & Al-Shehbaz. Cd treatment caused a significant decrease in E for both species (19 and 30% of the control for A. arenosa and A. halleri, respectively). On the contrary, E significantly decreased during Zn treatment only in A. arenosa (35% of the control). In the control conditions, the stomatal conductance (gs) of A. arenosa was more than twice that of A. halleri. Changes in gs were both heavy metal and species-specific. In the case of Cd-treated plants, the decrease in gs was 12% of the control in A. arenosa and A. halleri (26% of the control). In Zn treated plants, a significant decrease in gs was observed only in A. arenosa (22% of the control).
The accumulation of heavy metals in plants on LZ plots might depend on stomatal conductance, transpiration rate, and water uptake and on the HM on air entering the stomata. They may affect the photosynthesis in plants134.
Heavy metals influence also water delivery to the shoot due to the inhibition of transpiration as they decrease the size of the leaves and the thickness of the lamina, reduce intercellular spaces, affect the density of stomata and decrease their aperture5.
We found significant differences in leaf pigment content between both plant species growing on LZ plots and their respective C populations (grasslands). Analysis of chlorophyll content was considered a powerful tool for determining plant tolerance to stress135,136,137. Chlorophyll plays a significant role in plant metabolism and any reduction of chlorophyll content is closely associated with plant growth138. The chlorophyll index was significantly lower in Sg plants in comparison to Ce ones (Fig. 2B). Moreover, in the LZ populations of both species, the average values of chlorophyll indexes were 27% lower compared to controls. Other studies also confirmed that the plants (eg. Arabidopsis halleri (L.) O'Kane & Al-Shehbaz, Viola tricolor L., Betula pendula Roth) growing on spoil heaps have a lower chlorophyll content than plants from reference habitats139,140,141.
The values of anthocyanins were higher in both plants growing in areas with elevated total zinc and lead content (by 18%) in comparison to non-polluted sites. Anthocyanins can be formed as a reaction to lots of adverse environmental conditions, e.g. chilling, freezing, nutrient shortage (N- and P-deficiency), and enhanced resistance to heavy metals contamination142,143,144,145. Moreover, the changes in plant pigments (due to increased anthocyanins concentration) may be the first indication of insufficient detoxification of metals resulting in the deregulation of plant physiology. In summer months, under strong light anthocyanins can serve as a useful optical filter diverting excessive high-energy quanta away from an already saturated photosynthetic electron transport chain144. Chloroplasts irradiated with light that has first passed through a red filter have been shown to generate fewer superoxide radicals, thereby reducing the propensity for structural damage to the photosystems.
In both Ce and Sg plants occurring on LZ plots, the average leaf flavonol indexes were higher than on their controls by 31% and 15%. Moreover, the flavonol index in SgLZ population was significantly higher, than CeLZ and CeC plants (by 34% and 54%, respectively). In contrast, the content of flavonols lowered in Arabidopsis arenosa and A. halleri after exposure to Cd and Zn, whereas the content of anthocyanins increased136. According to145 flavonols and anthocyanins play an important role in plant response to TME stress in hyperaccumulating and non-acumulating plant species through the enhancement of antioxidant capacity and/or metal chelation in plants.
The results analysis of fluorescence kinetics revealed both species-, population- and time-specific differences in shape and amplitude of the curves which suggested differences in antenna organization and energy fluxes at the donor and the acceptor side of PSII. The differences were visible in fluorescence rise at the O-K phase was light-dependent and provided information about antenna size and connectivity of PSII reaction centres (ΔL band)46. The comparison ΔL bands in LZ plants, indicated no changes in antenna connectivity in Ce, while and strong increase in connectivity in the Sg population. The O-J (“photochemical”) phase is related to the rate of closure of reaction centers and the balance between electron donation from OEC to P680 + and electron accept from QA, that is, state of OEC and manifested with ΔK-band within 200–300 μs on the OJIP curve47,146. ΔK is related to the processes on the donor side of PSII. An increase in its magnitude indicates an imbalance of the rate of photochemical charge separation in PSII RCs and the rate of electron donation from the oxygen-evolving complex (OEC) to RC44, and it may be related to a range of environmental stresses such as water, nutrient deficiency, salinity, high temperature and heavy metals147Interestingly, ΔK amplitude was smaller than recorded by us in Sg and Ce populations growing on coal-mine spoil heaps110.
The shape of the ChlF induction curve (IC) from phase J to phase I corresponds to the reduction of the secondary electron acceptor QB, plastoquinone (PQ), cytochrome (Cyt b6f.), and plastocyanin94. The presence of the ΔH and ΔG bands in both LZ plots can be connected with damage to the plastoquinone pool and PSI end of electron acceptors such as Ferredoxin-NADP + Reductase (FNR). The decreased Fj value despite positive ΔK–band in SgLZ and CeLZ plants suggests the increased rate of electron transfer from QA to QB at the acceptor side of PSII, thus a high quantity of P680 + and/or effective quenching by exogenous molecules. Lastly, the increase in the I–P part of the induction curve is typically attributed to the reduction of electron transporters (ferredoxin, intermediary acceptors, and NADP) of the PSI acceptor side was observed in both SgC and SgLZ but not in CeLZ populations. These changes demonstrate species-specific effects on electron transport during the light phase of photosynthesis under CES.
The average values of maximum quantum yields of primary photochemical reactions (φPo = Fv/FM) in both Sg and Ce populations, ranged from 0.7 to 0.76. There were no differences between C and LZ plots and they were lower on CeLZ and higher on SgLZ plots in comparison to their controls. These values were typical for plants from populations that underwent complex environmental stresses and where the differences in size, age and physiological conditions occur like in case of Brachypodium pinnatum (L.) P.Beauv. growing on xerothermic grasslands53.
However, there are species-specific time-related differences in the response of PSA probably to an increase in light intensity and temperature with a subsequent decrease in humidity148. While in the CeC population, only the small dynamics of ChlF kinetics were observed, in the SgC and CeLZ we confirmed steadily growing amplitudes of ΔK and ΔJ-bands during the middle day hours, followed by a sharp increase rate of the entire electron transport chain up to the acceptor side of PSI. The last mechanism was not observed in SgLZ population, confirming a strong reduction of the whole ETR. In both species, φPo reached a value of 0.8 (in control populations) during the morning hours and dropped to 0.67–0.77.
Oxidative stress marker parameters in the response of species to abiotic stresses
Different anti-oxidative mechanisms can have evolved in plants in sites with elevated heavy metal contents that are dependent on both types of metal and a particular species136,149. In lipid peroxidation, malondialdehyde (MDA) is formed as a final product. It is used as an indicator of the intensity of oxidative stress and damage to membranes. The elevated level of MDA in plants exposed to metals is explained by electrolyte leakage, because of membrane damage149. In our studies, the concentration of MDA was significantly higher in leaves of Ce population growing on lead and zinc spoil heaps (by 17%) compared to the control population. Surprisingly, in the case of Sg no significant differences were found between LZ and C plots.
Metals can influence lipid peroxidation directly and indirectly. Cd—non redox active HM belongs to the latest group, which can inactivate antioxidant enzymes such as CAT or SOD150,151,152,153,154. The catalase activity was higher in Ce and Sg populations growing on lead and zinc spoil heaps (LZ) than those in controls for both species. Moreover, the values of enzymes in Sg tissue were significantly higher than those recorded in Ce tissues, irrespective of the plot type. Similarly, CAT activity in leaves of Viola tricolor L. plants exposed to heavy metal stress was at least twice as high as in the control material155. In Triticum aestivum L. activity of CAT increased significantly along with the increase of Cd concentration156. The addition of Mg supplementation lowered the toxic effect and restored CAT activity to normal levels suggesting a protective role of Mg under oxidative stress157. Large differences in catalase activity in the response to Cd or Zn treatment were detected between A. arenosa and A. halleri136. The activity of enzymes such as CAT involved in decreasing oxidative stress caused by the increase of Cd stress was confirmed in studies that deal with the response to stress caused by Cd in Allium sativum L.158; Tanacetum parthenium (L.) Sch.Bip.159; Hypericum perforatum L150.
Hydrogen peroxide (H2O2) takes part in host defence and oxidative biosynthetic reactions and at low levels can also function as a signalling agent, particularly in higher organisms160. H2O2 content in SgLZ tissues was significantly higher (57%) than those in CeLZ (39%). Hydrogen peroxide contents in CeLZ and SgLZ were respectively 39% (non-significant) and 57% higher, compared to their respective controls. This fact can be connected with a response to stress caused by elevated levels of HMs. Similar results we detected in our previous studies conducted on Sg and Ce populations growing on coal mine spoil heaps110. In laboratory (experimental) studies, Cd caused a similar and significant increase in the concentrations of hydrogen peroxide and MDA, while Zn treatment did not cause any change compared to the control conditions136. However, considering the contrasting accumulation of Cd and Zn in both species, A. arenosa can better cope with Cd-induced oxidative stress compared to A. halleri, while the opposite trend was observed for Zn.
The relationship between soil parameters on lead and zinc spoil heaps, ChlF parameters, pigment indexes, oxidative stress markers
Principal Component Analysis (PCA, Fig. 7A,B), performed on physico-chemical soil parameters, and Ce/Sg eco-physiological traits related to structure and functioning PSA (ChlF parameters, leaf pigment indexes, stress markers), confirmed the existence of complex environmental stresses on areas connected with lead and zinc processing. Such areas are susceptible to stress connected with elevated heavy metals content, nutrient and water deficiency, salinity, as well as high temperature. The first three PCA axes explained a 45 % variation in the data. The first PCA axis explains the 21% of variation and is positively correlated with total and bioavailable heavy metals content in the soils and plant leaves (Zn, Pb, Cd, Cu), soil pH, some basic cations (Mg), electrical conductivity (EC) while negatively with soil moisture. It separates CeLZ, SgLZ populations, growing on soil with higher HM content, higher soil pH, electrical conductivity (EC) and in drier places from populations of both species (SgC, CeC) growing in seminatural habitats. The flotation material is fine-grained, prone to over-dried and with neutral to alkaline reactions due to the presence of calcium or dolomite fragments, and higher electrical conductivity. In contrast, SgC and CeC populations in Świerklaniec occurred in remnants of wet meadows in sites with higher humidity. The variation in data explained by the second PCA axis (16.5%) is related within population differentiation in the efficiency of photosynthetic apparatus (Fig. 7A). The third axis, which explains 7.5% separates SgC populations growing in places with higher organic carbon (Corg), total nitrogen (Ntot), and available phosphorus (Pbio) content in the soil from CeLZ growing in nutrient-poorer habitats with higher bioavailable Zn in the soil. The Sg individuals growing on areas with elevated HM contents in the soil have structural changes in photosynthetic apparatus such as lower leaf chlorophyll, higher flavonoids and anthocyanins content. A reduction in chlorophyll content was reported in drought-stressed plants such as Populus davidiana (Dode) C.K.Schneid. by161. The chlorophyll content decreased to a significant level at higher water deficits in maize and wheat plants162. In experimental studies, the chlorophyll content in Allium schoenoprasum L. leaf decreased due to desiccation treatments163. Moreover, higher values of stress markers such as H2O2 in leaves were recorded in SgLZ individuals in comparison to CeLZ populations. In contrast, the CeLZ populations are characterized by higher values of Vj and ChlF parameters φRo and φDo, related to decreased QA to QB electron transport and PSI electron acceptors (Fig. 7). In leaves of CeLZ populations higher values of such stress markers as MDA were recorded in comparison to CeC and both Sg populations growing on lead and zinc spoil heaps and in semi-natural habitats.
PCA analysis shows correlations between physicochemical soil parameters, chlorophyll fluorescence parameters. The first and second components (A) and first and thrird components (B) are presented. Abbreviations: temp—temperature, Ntot—total nitrogen, EC—electrical conductivity, Chl—chlorophyll a, Flav—flavonol, Anth—anthocyanin, phi Eo—quantum yield for electron transport from QA—to plastoquinone; phiPo—maximum quantum yield of primary PSII photochemistry, psi Ro—quantum yield for reduction of end electron acceptors at the PSI acceptor side (RE); Vi—relative variable fluorescence at I-step (20 ms); Vj—relative variable fluorescence at J-step (2 ms); dVg/dto − slope of prompt fluorescence curve rise, between 50 and 100 µs.
Conclusions
We confirmed the complex environmental stresses acting on lead and zinc spoil heaps, resulting from a combination of soil elevated HM content, water and nutrient deficiency, high temperature and salinity trigger the habitat- and species- specific short- and long-term responses and structural changes of photosynthetic apparatus of Sg and Ce. The Sg individuals growing on areas with elevated HM contents in the soil have lower leaf chlorophyll, higher flavonoids and anthocyanins content and higher values ChlF parameters such as Vi. Moreover, higher values of stress markers such as H2O2 in leaves were recorded in SgLZ individuals in comparison to CeLZ populations. In contrast, the CeLZ populations are characterized by higher values of Vj and JIP parameters φRo and φDo, related to quantum yield of reduction in final electron acceptors of PSI per photon absorbed and quantum yield of heat dissipation. In leaves of Ce from LZ populations higher values of such stress markers as MDA were recorded in comparison to CeC and both Sg populations growing on lead and zinc spoil heaps and in semi-natural habitats.
Our research scheme and timing cover the combination of the most important environmental stressors operating in the short-term period in the studied area. We confirmed the application of wide-range indicators of environmental stresses allowed the detection of short- and long-term site- and species-specific responses of PSA of two species commonly occurring in HM-contaminated sites. Both species from LZ plots could tolerate high levels of Cd, Zn and Pb in leaf tissues. Therefore they can be potential candidates for use in the phytoremediation of HM-contaminated areas.
However, given the short period of the study and partially controlled experimental conditions, further long-term field and experimental research are needed, to give deeper insight into the regulatory mechanisms and adaptation strategy of native expansive and alien invasive species under climate change in the context of future management of post-industrial areas.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Pietrzykowski, M., Antonkiewicz, J., Gruba, P. & Pajak, M. Content of Zn, Cd and Pb in purple moor-grass in soils heavily contaminated with heavy metals around a zinc and lead ore tailing landfill. Open Chem. 16, 1143–1152 (2018).
Kicińska, A., Smreczak, B. & Jadczyszyn, J. Soil bioavailability of cadmium, lead, and zinc in the areas of Zn-Pb ore mining and processing (Bukowno, Olkusz). J. Ecol. Eng. 20, 84–92 (2019).
Wahsha, M. et al. Microbial enzymes as an early warning management tool for monitoring mining site soils. CATENA 148, 40–45 (2017).
Zhang, X. et al. Impacts of lead/zinc mining and smelting on the environment and human health in China. Environ. Monit. Assess. 184, 2261–2273 (2012).
Rucińska-Sobkowiak, R. Water relations in plants subjected to heavy metal stresses. Acta Physiol. Plant. https://doi.org/10.1007/s11738-016-2277-5 (2016).
Schulze, E.-D., Beck, E. & Müller-Hohenstein, K. Plant Ecology (Springer, Berlin, 2005).
Grime, J. P. The stress debate: symptom of impending synthesis?. Biol. J. Lin. Soc. 37, 3–17 (1989).
Kompała-Bąba, A. et al. Eco-physiological responses of Calamagrostis epigejos L (Roth) and Solidago gigantea Aition to complex environmental stresses in coal-mine spoil heaps. Land Degrad. Dev. ldr. https://doi.org/10.1002/ldr.4119 (2021).
Mittler, R. Abiotic stress, the field environment and stress combination. Trends Plant Sci. 11, 15–19 (2006).
Suzuki, N., Rivero, R. M., Shulaev, V., Blumwald, E. & Mittler, R. Abiotic and biotic stress combinations. New Phytol 203, 32–43 (2014).
Sewelam, N., Oshima, Y., Mitsuda, N. & Ohme-Takagi, M. A step towards understanding plant responses to multiple environmental stresses: a genome-wide study: Plant responses to multiple abiotic stresses. Plant Cell Environ. 37, 2024–2035 (2014).
Tuteja, N. & Sopory, S. K. Chemical signaling under abiotic stress environment in plants. Plant Signal. Behav. 3, 525–536 (2008).
Seleiman, M. F. et al. Drought stress impacts on plants and different approaches to alleviate its adverse effects. Plants 10, 259 (2021).
Larcher, W. Physiological Plant Ecology: Ecophysiology and Stress Physiology of Functional Groups (Springer, Berlin, New York, 2003).
Haider, S. et al. Molecular mechanisms of plant tolerance to heat stress: current landscape and future perspectives. Plant Cell Rep 40, 2247–2271 (2021).
Hasanuzzaman, M. et al. Regulation of ascorbate-glutathione pathway in mitigating oxidative damage in plants under abiotic stress. Antioxidants 8, 384 (2019).
Kabata-Pendias, A. Trace Elements in Soils and Plants (CRC Press, 2011).
Guo, G., Wu, F., Xie, F. & Zhang, R. Spatial distribution and pollution assessment of heavy metals in urban soils from southwest China. J. Environ. Sci. 24, 410–418 (2012).
Toxic Metals in Soil-Plant Systems. (Wiley, Chichester, West Sussex, England ; New York, 1994)
Dmuchowski, W. & Bytnerowicz, A. Long-term (1992–2004) record of lead, cadmium, and zinc air contamination in Warsaw, Poland: Determination by chemical analysis of moss bags and leaves of Crimean linden. Environ. Pollut. 157, 3413–3421 (2009).
McBride, M. B. Soluble trace metals in alkaline stabilized sludge products. J. Environ. Qual. 27, 578–584 (1998).
Kicińska, A., Pomykała, R. & Izquierdo-Diaz, M. Changes in soil pH and mobility of heavy metals in contaminated soils. Eur. J. Soil Sci. 73, e13203 (2022).
Alloway, B. J. Sources of Heavy Metals and Metalloids in Soils. In Heavy Metals in Soils (ed. Alloway, B. J.) vol. 22 11–50 (Springer Netherlands, Dordrecht, 2013).
Göhre, V. & Paszkowski, U. Contribution of the arbuscular mycorrhizal symbiosis to heavy metal phytoremediation. Planta 223, 1115–1122 (2006).
Violante, A., Cozzolino, V., Perelomov, L., Caporale, A. G. & Pigna, M. Mobility and bioavailability of heavy metals and metalloids in soil environments. J. Soil Sci. Plant Nutr. 10, (2010).
Schutzendubel, A. Plant responses to abiotic stresses: Heavy metal-induced oxidative stress and protection by mycorrhization. J. Exp. Bot. 53, 1351–1365 (2002).
Hall, J. L. Cellular mechanisms for heavy metal detoxification and tolerance. J. Exp. Bot. 53, 1–11 (2002).
Krzesłowska, M. The cell wall in plant cell response to trace metals: Polysaccharide remodeling and its role in defense strategy. Acta Physiol. Plant 33, 35–51 (2011).
Viehweger, K. How plants cope with heavy metals. Bot. Stud. 55, 1–12 (2014).
Załęcka, R. & Wierzbicka, M. The adaptation of Dianthus carthusianorum L. (Caryophyllaceae) to growth on a zinc-lead heap in southern Poland. Plant Soil 246, 249–257 (2002).
Sytar, O., Ghosh, S., Malinska, H., Zivcak, M. & Brestic, M. Physiological and molecular mechanisms of metal accumulation in hyperaccumulator plants. Physiol. Plant. https://doi.org/10.1111/ppl.13285 (2020).
Sharma, R. et al. Responses of Phytochelatins and Metallothioneins in Alleviation of Heavy Metal Stress in Plants. In Plant Metal Interaction 263–283 (Elsevier, 2016). https://doi.org/10.1016/B978-0-12-803158-2.00010-2.
Koszelnik-Leszek, A. & Bielecki, K. Physiological responses of nonmetallicolous and serpentine Silene vulgaris ecotypes. Environ. Protect. Eng. 47, 5–12 (2021).
Chmielowska-Bąk, J. & Deckert, J. Plant recovery after metal stress—a review. Plants https://doi.org/10.3390/plants10030450 (2021).
Kostić, O. et al. Ecophysiological response of Populus alba L. to multiple stress factors during the revitalisation of coal fly ash lagoons at different stages of weathering. Front. Plant Sci. 14, 1337700 (2024).
Koim-Puchowska, B. et al. Can environmental stressors determine the condition of ecological plant groups?. Plants 13, 1550 (2024).
Uraguchi, S., Watanabe, I., Yoshitomi, A., Kiyono, M. & Kuno, K. Characteristics of cadmium accumulation and tolerance in novel Cd-accumulating crops, Avena strigosa and Crotalaria juncea. J. Exp. Bot. 57, 2955–2965 (2006).
Gill, S. S. & Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 48, 909–930 (2010).
Kaur, N., Kaur, J., Grewal, S. K. & Singh, I. Effect of heat stress on antioxidative defense system and its amelioration by heat acclimation and salicylic acid pre-treatments in three Pigeonpea genotypes. Ind. Jour. of Agricult. Bioch. 32, 106 (2019).
Mittler, R. ROS are good. Trends Plant Sci. 22, 11–19 (2017).
Raja, V., Majeed, U., Kang, H., Andrabi, K. I. & John, R. Abiotic stress: Interplay between ROS, hormones and MAPKs. Environ. Exp. Bot. 137, 142–157 (2017).
Sharma, A. et al. Photosynthetic response of plants under different abiotic stresses: A review. J. Plant Growth Regul. 39, 509–531 (2020).
Crafts-Brandner, S. J. & Salvucci, M. E. Sensitivity of photosynthesis in a C4 plant, maize, to heat stress. Plant Physiol. 129, 1773–1780 (2002).
Kalaji, H. M. et al. Chlorophyll a fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol. Plant. 38, 102 (2016).
Kalaji, H. M. et al. Frequently asked questions about chlorophyll fluorescence, the sequel. Photosynth. Res. 132, 13–66 (2017).
Tsimilli-Michael, M. & Strasser, R. J. The energy flux theory 35 years later: formulations and applications. Photosynth. Res. 117, 289–320 (2013).
Strasser, R. J., Tsimilli-Michael, M. Srivastava, A. & Srivastava, A. Analysis of the chlorophyll a fluorescence transient. In Papageorgiou GC, Govindjee (eds) Advances in photosynthesis and respiration. Chlorophyll a fluorescence: a signature of photosynthesis 321–362 (Kluwer Acad. Publ., Dordrecht, 2005).
Goltsev, V. N. et al. Variable chlorophyll fluorescence and its use for assessing physiological condition of plant photosynthetic apparatus. Russ. J. Plant Physiol. 63, 869–893 (2016).
Lazar, D. Modelling of light-induced chlorophyll a fluorescence rise (O-J-I-P transient) and changes in 820 nm-transmittance signal of photosynthesis. PHOTOSYNTHETICA 47, 483–498 (2009).
Stirbet, A. On the relation between the Kautsky effect (chlorophyll a fluorescence induction) and photosystem II: Basics and applications of the OJIP fluorescence transient. J. Photochem. Photobiol. B Biol. 104, 236–257 (2011).
Banks, J. M. Chlorophyll fluorescence as a tool to identify drought stress in Acer genotypes. Environ. Exp. Bot. 155, 118–127 (2018).
Bussotti, F. et al. Photosynthesis responses to ozone in young trees of three species with different sensitivities, in a 2-year open-top chamber experiment (Curno, Italy). Physiol. Plant. 130, 122–135 (2007).
Bąba, W., Kalaji, H. M., Kompała-Bąba, A. & Goltsev, V. Acclimatization of photosynthetic apparatus of tor grass (Brachypodium pinnatum) during expansion. PLOS ONE 11, e0156201 (2016).
Bussotti, F. & Pollastrini, M. Revisiting the concept of stress in forest trees at the time of global change and issues for stress monitoring. Plant Stress 2, 100013 (2021).
Alam, B., Nair, D. & Jacob, J. Low temperature stress modifies the photochemical efficiency of a tropical tree species Hevea brasiliensis: Effects of varying concentration of CO2 and photon flux density. Photosynthetica 43, 247–252 (2005).
Kalaji, H. M., Bosa, K., Koscielniak, J. & Hossain, Z. Chlorophyll a fluorescence-a useful tool for the early detection of temperature stress in spring barley (Hordeum vulgare L.). Omics- J. Integr. Biol. 15, 925–934 (2011).
Oukarroum, A., Goltsev, V. & Strasser, R. J. Temperature effects on pea plants probed by simultaneous measurements of the kinetics of prompt fluorescence, delayed fluorescence and modulated 820 nm reflection. PLOS ONE 8, e59433 (2013).
Goltsev, V. et al. Drought-induced modifications of photosynthetic electron transport in intact leaves: Analysis and use of neural networks as a tool for a rapid non-invasive estimation. Biochim. et Biophys. Acta (BBA) – Bioenerg. 1817, 1490–1498 (2012).
Dąbrowski, P. et al. Exploration of chlorophyll a fluorescence and plant gas exchange parameters as indicators of drought tolerance in perennial ryegrass. Sensors 19, 2736 (2019).
Kalaji, H. M. et al. Identification of nutrient deficiency in maize and tomato plants by in vivo chlorophyll a fluorescence measurements. Plant Physiol. Biochem. 81, 16–25 (2014).
Kalaji, H. M. et al. Chlorophyll fluorescence as a tool for nutrient status identification in rapeseed plants. Photosynth. Res. 136, 329–343 (2018).
Mehta, P., Kraslavsky, V., Bharti, S., Allakhverdiev, S. I. & Jajoo, A. Analysis of salt stress induced changes in photosystem II heterogeneity by prompt fluorescence and delayed fluorescence in wheat (Triticum aestivum) leaves. J. Photochem. Photobiol. B-Biol. 104, 308–313 (2011).
Kan, X. et al. Effects of salinity on photosynthesis in maize probed by prompt fluorescence, delayed fluorescence and P700 signals. Environ. Exp. Bot. 140, 56–64 (2017).
Azevedo, H., Pinto, C., Fernandes, J., Loureiro, S. & Santos, C. Cadmium effects on sunflower growth and photosynthesis. J. Plant Nutr. 28, 2211–2220 (2005).
Baycu, G. et al. Cadmium-zinc accumulation and photosystem II responses of Noccaea caerulescens to Cd and Zn exposure. Environ. Sci. Pollut. Res. 24, 2840–2850 (2017).
Pflug, E. E. et al. Resilient leaf physiological response of European beech (Fagus sylvatica L.) to summer drought and drought release. Front. Plant Sci. 9, 187 (2018).
Jänicke, L. K. et al. OLCI-A/B tandem phase: evaluation of FLuorescence EXplorer (FLEX)-like radiances and estimation of systematic differences between OLCI-A and OLCI-FLEX. Atmos. Meas. Tech. 16, 3101–3121 (2023).
Porcar-Castell, A. et al. Chlorophyll a fluorescence illuminates a path connecting plant molecular biology to Earth-system science. Nat. Plants 7, 998–1009 (2021).
Rajewicz, P. A. et al. The photosynthetic response of spectral chlorophyll fluorescence differs across species and light environments in a boreal forest ecosystem. Agric. Forest Meteorol. 334, 109434 (2023).
Weber, E. & Jakobs, G. Biological flora of central Europe: Solidago gigantea Aiton. Flora 200, 109–118 (2005).
Mudrák, O., Doležal, J. & Frouz, J. Initial species composition predicts the progress in the spontaneous succession on post-mining sites. Ecol. Eng. 95, 665–670 (2016).
Hultén, E. & Fries, M. Atlas of Northern European vascular plants north of the tropic of cancer. (Koeltz, Königstein, 1986).
Pfab, M. F. & Victor, J. E. Threatened plants of Gauteng, South Africa. South Afr. J. Bot. 68, 370–375 (2002).
Rebele, F. & Lehmann, C. Biological Flora of Central Europe: Calamagrostis epigejos (L.) Roth. Flora 196, 325–344 (2001).
Kompała-Bąba, A. et al. Do the dominant plant species impact the substrate and vegetation composition of post-coal mining spoil heaps?. Ecol. Eng. 143, 105685 (2020).
Kompala-Bąba, A. & Bąba, W. The spontaneous succession in a sand-pit–the role of life history traits and species habitat preferences. Pol. J. Ecol. 61, 13–22 (2013).
Dambiec, M., Klink, A. & Polechońska, L. Concentration and translocation of trace metals in Solidago gigantea in urban areas: A potential bioindicator. Int. J. Environ. Sci. Technol. 19, 11729–11740 (2022).
Fiala, K., Tůma, I. & Holub, P. Effect of nitrogen addition and drought on above-ground biomass of expanding tall grasses Calamagrostis epigejos and Arrhenatherum elatius. Biologia 66, 275–281 (2011).
Lehmann, C. & Rebele, F. Evaluation of heavy metal tolerance in Calamagrostis epigejos and Elymus repens revealed copper tolerance in a copper smelter population of C. epigejos. Environ. Exp. Bot. 51, 199–213 (2004).
Verlinden, M., Van Kerkhove, A. & Nijs, I. Effects of experimental climate warming and associated soil drought on the competition between three highly invasive West European alien plant species and native counterparts. Plant Ecol. 214, 243–254 (2013).
Walczyk, A. M. & Hersch-Green, E. I. Do water and soil nutrient scarcities differentially impact the performance of diploid and tetraploid Solidago gigantea (Giant Goldenrod, Asteraceae)?. Plant Biol. J. 24, 1031–1042 (2022).
Kráľová, K., Masarovičová Elena & Jampílek, J. Plant Responses to Stress Induced by Toxic Metals and Their Nanoforms. In Handbook of Plant and Crop Stress, 479–508 (CRC Press, Boca Raton, 2019).
Coppola, V., Boni, M., Gilg, H. A. & Strzelska-Smakowska, B. Nonsulfide zinc deposits in the Silesia-Cracow district, Southern Poland. Miner. Deposita 44, 559–580 (2009).
Kompała-Bąba, A., Błońska, A. & Woźniak, G. Vegetation of ‘Żabie Doły’ area (Bytom) covering the wastelands of zinc-lead industry. Arch. Environ. Protect. 30, 59–76 (2004).
Skurczyńska M, Leśniok M (2008). Przyroda województwa śląskiego. Biblioteka CDPGŚ. http://przyroda.katowice.pl/pl/przyrodanieozywiona/klimat/127-klima [Access 10.06.2024].
Rostański, A. & Woźniak, G. Trawy (Poaceae) występujące spontanicznie na terenie nieużytków poprzemysłowych. in In L. Frey (Red.) Biologia traw. Fragmenta Floristica et Geobotanica Polonica. Supplementum 9. PAN. Kraków 31–42 (2007).
Woźniak, G. Problems of Calamagrostis epigejos synecology on post-industrial sites. In Frey L. (Ed.), Biology of grasses. W. Szafer Institute of Botany, Polish Academy of Science Krakow. 353–361 (2005).
Kompała-Bąba, A. & Bąba, W. Udział traw w zbiorowiskach roślinnych wykształcających się na obszarze wybranych kamieniołomów Wyżyny Śląskiej. Fragm. Flor. Geobot. Polonica 18, 359–374 (2011).
Woźniak, G. et al. Use of remote sensing to track postindustrial vegetation development. Land Degr. Dev. 32, 1426–1439 (2021).
Bednarek, R., Dziadowiec, H., Pokojska, U. & Prusinkiewicz, Z. Badania ekologiczno-gleboznawcze - Google Scholar. Wydawnictwo Naukowe PWN https://scholar.google.pl/scholar?hl=pl&as_sdt=0,5&cluster=15836461043494564398 (2011).
Bouazizi, H., Jouili, H., Geitmann, A. & El Ferjani, E. Copper toxicity in expanding leaves of Phaseolus vulgaris L.: Antioxidant enzyme response and nutrient element uptake. Ecotoxicol. Environ. Saf. 73, 1304–1308 (2010).
Cerovic, Z. G., Masdoumier, G., Ghozlen, N. B. & Latouche, G. A new optical leaf-clip meter for simultaneous non-destructive assessment of leaf chlorophyll and epidermal flavonoids. Physiol. Plant. 146, 251–260 (2012).
Julkunen-Tiitto, R. et al. Assessing the response of plant flavonoids to UV radiation: An overview of appropriate techniques. Phytochem. Rev. 14, 273–297 (2015).
Kalaji, H. M. et al. Frequently asked questions about in vivo chlorophyll fluorescence: practical issues. Photosynth. Res. 122, 121–158 (2014).
Strasser, R. & Tsimilli-Michael, M. Experimental approach to estimate in vivo the balance of the mechanisms for performance of light reactions, dark reactions, energetic cooperativity, non-Q(A) and non-Q (B)-reducing and non-oxygen-evolving PSII centers. Acta Physiol. Plant. 26, 94 (2004).
Tsimilli-Michael, M. & Strasser, R. J. Photosystem II adaptation to changing light conditions as a process leading to optimality/stability: a theoretical approachand a model using different types of conformational/state changes. Acta Physiol. Plant. 26, 176–177 (2004).
Stirbet, A., Lazár, D. & Kromdijk, J. Chlorophyll a fluorescence induction: Can just a one-second measurement be used to quantify abiotic stress responses?. Photosynthetica 56, 86–104 (2018).
Brestic, M., Zivcak, M., Kalaji, H. M., Carpentier, R. & Allakhverdiev, S. I. Photosystem II thermostability in situ: Environmentally induced acclimation and genotype-specific reactions in Triticum aestivum L. Plant Physiol. Biochem. 57, 93–105 (2012).
Strasser, R. J. & Stirbet, A. D. Estimation of the energetic connectivity of PS II centres in plants using the fluorescence rise O-J–I–P. Math. Comput. Simul. 56, 451–462 (2001).
TIBCO Software Inc. Statistica (data analysis software system), version 13. Preprint at (2017).
R Core Team. R: A Language and Environment for Statistical Computing. Preprint at (2020).
Oksanen, J. et al. Vegan: Community Ecology Package. (2019).
Wickham, H. Ggplot2: Elegant Graphics for Data Analysis. (Springer-Verlag New York, 2016).
Nawaz, M. et al. A review of plants strategies to resist biotic and abiotic environmental stressors. Sci. Total Environ. 900, 165832 (2023).
Hobbs, R. J., Higgs, E. S. & Hall, C. M. Defining Novel Ecosystems. in Novel Ecosystems (eds Hobbs, R. J., Higgs, E. S. & Hall, C. M.) 58–60 (Wiley, 2013). https://doi.org/10.1002/9781118354186.ch6.
Merwin, L., Umek, L. & Anastasio, A. E. Urban post-industrial landscapes have unrealized ecological potential. Restor. Ecol. 30, e13643 (2022).
Řehounková, K. & Prach, K. Life-history traits and habitat preferences of colonizing plant species in long-term spontaneous succession in abandoned gravel–sand pits. Basic Appl. Ecol. 11, 45–53 (2010).
Woźniak, G., Chmura, D., Tokarska-Guzik, B. & Sierka, E. Applicability of the concept of functional groups for analysis of spatiotemporal vegetation changes on manmade habitats. Pol. J. Environ. Stud. 20, 623–631 (2011).
Vereecken, H. et al. Modeling soil processes: Review, key challenges, and new perspectives. Vadose Zone J. 15, 1–57 (2016).
Kompała-Bąba, A. et al. Eco-physiological responses of Calamagrostis epigejos L (Roth) and Solidago gigantea Aition to complex environmental stresses in coal-mine spoil heaps. Land Degrad. Dev. 32, 5427–5442 (2021).
Planchuelo, G., Von Der Lippe, M. & Kowarik, I. Untangling the role of urban ecosystems as habitats for endangered plant species. Landsc. Urban Plan. 189, 320–334 (2019).
Planchuelo, G., Kowarik, I. & Von Der Lippe, M. Endangered plants in novel urban ecosystems are filtered by strategy type and dispersal syndrome, not by spatial dependence on natural remnants. Front. Ecol. Evol. 8, 18 (2020).
Gallagher, R. V., Beaumont, L. J., Hughes, L. & Leishman, M. R. Evidence for climatic niche and biome shifts between native and novel ranges in plant species introduced to Australia. J. Ecol. 98, 790–799 (2010).
Nassauer, J. I. & Raskin, J. Urban vacancy and land use legacies: A frontier for urban ecological research, design, and planning. Landsc. Urban Plan. 125, 245–253 (2014).
Botzat, A., Fischer, L. K. & Kowarik, I. Unexploited opportunities in understanding liveable and biodiverse cities A review on urban biodiversity perception and valuation. Glob. Environ. Change 39, 220–233 (2016).
Li, Z., Ma, Z., Van Der Kuijp, T. J., Yuan, Z. & Huang, L. A review of soil heavy metal pollution from mines in China: Pollution and health risk assessment. Sci.Total Environ. 468–469, 843–853 (2014).
Kompała-Bąba, A. et al. Vegetation diversity on coal mine spoil heaps – how important is the texture of the soil substrate?. Biologia 74, 419–436 (2019).
Masoudian, M. S. et al. Stability and characterisation of spoil heaps in European surface lignite mines: A state-of-the-art review in light of new data. Environ Earth Sci 78, 505 (2019).
Spasić, M., Borůvka, L., Vacek, O., Drábek, O. & Tejnecký, V. Pedogenesis problems on reclaimed coal mining sites. Soil Water Res. 16, 137–150 (2021).
Buczkowski, R., Kondzielski, I. & Szymański, T. Metody Remediacji Gleb Zanieczyszczonych Metalami Ciężkimi. (Wydawnictwo Uniwersytetu Mikołaja Kopernika, Toruń, 2002).
Adamczyk-Szabela, D. & Wolf, W. M. The impact of soil pH on heavy metals uptake and photosynthesis efficiency in Melissa officinalis, Taraxacum officinalis, Ocimum basilicum. Molecules 27, 4671 (2022).
Nadgórska-Socha, A., Kandzora-Ciupa, M., Ciepał, R. & Walasek, K. Effects of Zn, Cd, Pb on physiological response of Silene vulgaris plants from selected populations. Pol. J. Environ. Stud. 20, 599–604 (2011).
Kabata-Pendias, A. Soil–plant transfer of trace elements—an environmental issue. Geoderma 122, 143–149 (2004).
Cejpek, J., Kuráž, V. & Frouz, J. Hydrological properties of soils in reclaimed and unreclaimed sites after brown-coal mining. Pol. J. Environ. Stud. 22, 645–652 (2013).
Moeneclaey, I. et al. Species ecological strategy and soil phosphorus supply interactively affect plant biomass and phosphorus concentration. Basic Appl. Ecol. 62, 1–11 (2022).
Mishra, J., Singh, R. & Arora, N. K. Alleviation of heavy metal stress in plants and remediation of soil by rhizosphere microorganisms. Front. Microbiol. 8, 1706 (2017).
Dias, M. C. et al. Cadmium toxicity affects photosynthesis and plant growth at different levels. Acta Physiol. Plant 35, 1281–1289 (2013).
Smýkalová, I. & Zámečníková, B. The relationship between salinity and cadmium stress in barley. Biol. plant. 46, 269–273 (2003).
He, J., Ren, Y., Zhu, C. & Jiang, D. Effects of cadmium stress on seed germination, seedling growth and seed amylase activities in rice (Oryza sativa). Rice Sci. 15, 319–325 (2008).
Akhtar, T. et al. Photosynthesis and growth response of maize (Zea mays L.) hybrids exposed to cadmium stress. Environ. Sci. Pollut. Res. 24, 5521–5529 (2017).
Per, T. S., Khan, S., Asgher, M., Bano, B. & Khan, N. A. Photosynthetic and growth responses of two mustard cultivars differing in phytocystatin activity under cadmium stress. Photosynthetica 54, 491–501 (2016).
Ying, R.-R. et al. Cadmium tolerance of carbon assimilation enzymes and chloroplast in Zn/Cd hyperaccumulator Picris divaricata. J. Plant Physiol. 167, 81–87 (2010).
Liu, H., Wang, H., Ma, Y., Wang, H. & Shi, Y. Role of transpiration and metabolism in translocation and accumulation of cadmium in tobacco plants (Nicotiana tabacum L.). Chemosphere 144, 1960–1965 (2016).
Guo, Z. et al. Effects of heavy metals on stomata in plants: A review. IJMS 24, 9302 (2023).
Li, R., Guo, P., Michael, B., Stefania, G. & Salvatore, C. Evaluation of chlorophyll content and fluorescence parameters as indicators of drought tolerance in barley. Agric. Sci. China 5, 751–757 (2006).
Szopiński, M. et al. Toxic EFfects of Cd and Zn on the photosynthetic apparatus of the Arabidopsis halleri and Arabidopsis arenosa pseudo-metallophytes. Front. Plant Sci. 10, 748 (2019).
Talebzadeh, F. & Valeo, C. Evaluating the effects of environmental stress on leaf chlorophyll content as an index for tree health. IOP Conf. Ser.: Earth Environ. Sci. 1006, 012007 (2022).
Morales, F. et al. Photosynthetic metabolism under stressful growth conditions as a bases for crop breeding and yield improvement. Plants 9, 88 (2020).
Sitko, K., Rusinowski, S., Kalaji, H. M., Szopiński, M. & Małkowski, E. Photosynthetic Efficiency as Bioindicator of Environmental Pressure in A. halleri. Plant Physiol. 175, 290–302 (2017).
Sitko, K. et al. Effect of drought and heavy metal contamination on growth and photosynthesis of silver birch trees growing on post-industrial heaps. Cells 11, 53 (2021).
Pilarska, M., Niewiadomska, E., Sychta, K. & Słomka, A. Differences in the functioning of photosynthetic electron transport between metallicolous and non-metallicolous populations of the pseudometallophyte Viola tricolor. J. Plant Physiol. 250, 153185 (2020).
Ernst, W. H. O. Evolution of metal hyperaccumulation and phytoremediation hype. New Phytol. 146, 357–358 (2000).
Gould, K. S. Nature’s Swiss Army knife: the diverse protective roles of anthocyanins in leaves. J. Biomed. Biotechnol. 2004, 314–320 (2004).
Nadgórska-Socha, A., Ciepał, R., Kandziora, M. & Kafel, A. Heavy metals bioaccumulation and physiological responses to heavy metal stress in populations of Silene vulgaris Moench (Garcke) from heavy metal contaminated sites. Ecol. Chem. Eng. 16, 389–397 (2009).
Corso, M. et al. Contrasting cadmium resistance strategies in two metallicolous populations of Arabidopsis halleri. New Phytol. 218, 283–297 (2018).
Lazar, D. The polyphasic chlorophyll a fluorescence rise measured under high intensity of exciting light. Funct. Plant Biol. 33, 9–30 (2006).
Srivastava, A., Guisse, B., Greppin, H. & Strasser, R. J. Regulation of antenna structure and electron transport in photosystem II of Pisum sativum under elevated temperature probed by the fast polyphasic chlorophyll a fluorescence transient: OKJIP. Biochim. Biophys. Acta-Bioenerg. 1320, 95–106 (1997).
Bussotti, F., Gerosa, G., Digrado, A. & Pollastrini, M. Selection of chlorophyll fluorescence parameters as indicators of photosynthetic efficiency in large scale plant ecological studies. Ecol. Indic. 108, 105686 (2020).
Małkowski, E., Sitko, K., Zieleźnik-Rusinowska, P., Gieroń, Ż. & Szopiński, M. Heavy Metal Toxicity: Physiological Implications of Metal Toxicity in Plants. In Plant Metallomics and Functional Omics (ed. Sablok, G.) 253–301 (Springer International Publishing, Cham, 2019). https://doi.org/10.1007/978-3-030-19103-0_10.
Dazy, M., Masfaraud, J.-F. & Férard, J.-F. Induction of oxidative stress biomarkers associated with heavy metal stress in Fontinalis antipyretica Hedw. Chemosphere 75, 297–302 (2009).
Juknys, R., Vitkauskaitė, G., Račaitė, M. & Venclovienė, J. The impacts of heavy metals on oxidative stress and growth of spring barley. Open Life Sci. 7, 299–306 (2012).
Anjum, N. A. et al. Biophysical and biochemical markers of metal/metalloid-impacts in salt marsh halophytes and their implications. Front. Environ. Sci. https://doi.org/10.3389/fenvs.2016.00024 (2016).
Benyó, D. et al. Physiological and molecular responses to heavy metal stresses suggest different detoxification mechanism of Populus deltoides and P. x canadensis. J. Plant Physiol. 201, 62–70 (2016).
Mwamba, T. M. et al. Interactive effects of cadmium and copper on metal accumulation, oxidative stress, and mineral composition in Brassica napus. Int. J. Environ. Sci. Technol. 13, 2163–2174 (2016).
Słomka, A., Libik-Konieczny, M., Kuta, E. & Miszalski, Z. Metalliferous and non-metalliferous populations of Viola tricolor represent similar mode of antioxidative response. J. Plant Physiol. 165, 1610–1619 (2008).
Rizvi, A., Khan, M. & S.,. Biotoxic impact of heavy metals on growth, oxidative stress and morphological changes in root structure of wheat (Triticum aestivum L.) and stress alleviation by Pseudomonas aeruginosa strain CPSB1. Chemosphere 185, 942–952 (2017).
Singh, V., Tripathi, B. N. & Sharma, V. Interaction of Mg with heavy metals (Cu, Cd) in T. aestivum with special reference to oxidative and proline metabolism. J. Plant Res. 129, 487–497 (2016).
Zhang, H., Jiang, Y., He, Z. & Ma, M. Cadmium accumulation and oxidative burst in garlic (Allium sativum). J. Plant Physiol. 162, 977–984 (2005).
Hojati, M. et al. Cadmium and copper induced changes in growth, oxidative metabolism and terpenoids of Tanacetum parthenium. Environ. Sci. Pollut. Res. 24, 12261–12272 (2017).
Stone, J. R. & Yang, S. Hydrogen peroxide: A signaling messenger. Antioxid. Redox Signal. 8, 243–270 (2006).
Zhang, X., Zang, R. & Li, C. Population differences in physiological and morphological adaptations of Populus davidiana seedlings in response to progressive drought stress. Plant Sci. 166, 791–797 (2004).
Nayyar, H. & Gupta, D. Differential sensitivity of C3 and C4 plants to water deficit stress: Association with oxidative stress and antioxidants. Environ. Exp. Bot. 58, 106–113 (2006).
Egert, M. & Tevini, M. Influence of drought on some physiological parameters symptomatic for oxidative stress in leaves of chives (Allium schoenoprasum). Environ. Exp. Bot. 48, 43–49 (2002).
Acknowledgements
We would like to thank the referees who provided helpful comments on our article. We are also grateful for students and technicians that helped us in both field and laboratory work. This study was partially founded by Institute of Biology, Biotechnology and Environmental Protection, University of Silesia in Katowice.
Author information
Authors and Affiliations
Contributions
W.Ba. and A.K.B. provided the idea and designed the experiment. W.Bi., E.S., A.K.B., W.Ba. collected the samples. W.Ba., A.K.B., W.Bi. and E.S. analyzed the soil and plant samples. W.Ba., A.K.B., and W.Bi. analyzed statistically the data set. W.Ba., A.K.B. wrote the first draft of the manuscript. All authors read the final draft.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bąba, W., Kompała-Bąba, A., Sierka, E. et al. Photosynthetic response of Solidago gigantea Aition and Calamagrostis epigejos L. (Roth) to complex environmental stress on heavy metal contaminated sites. Sci Rep 14, 31481 (2024). https://doi.org/10.1038/s41598-024-82952-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-82952-0