Abstract
The study aimed to explore the potential use of coal-fired power plant bottom ashes in Pleurotus ostreatus cultivation using spent coffee grounds. The study analyzed five compositions of growth substrate for mushrooms: pure coffee grounds (I) as a control sample; coffee grounds substrate with the addition of 1% (II); 5% (III); 10% (IV) bottom ash; and bottom ash alone (V). The study revealed that compared to the control sample (I), the addition of 1% bottom ash (II) did not affect the time of mycelium growth but slowed fruiting body growth by 4 days. With 5% addition (III), mycelium growth slowed by 6 days, and fruiting body growth by 7 days. At 10% (IV), growth was completely inhibited. Compared to sample (I), fruiting bodies grown on sample (II) had higher phosphorus, copper, and zinc accumulation, while chromium, nickel, and lead levels were lower in fruiting bodies grown on samples (II) and (III). Additionally, fruiting bodies grown on samples (II) and (III) contained less iron, silicon, selenium, aluminum, calcium, and magnesium. The results presented in the article regarding the levels of contamination in the cultivated mushrooms and in the substrate after cultivation, indicate the potential for their further management.
Similar content being viewed by others
Introduction
The Pleurotus species is the second most widely grown and disseminated edible mushroom genus after Agaricus species1. Due to its versatility, it may be grown on agro-industrial, food, and cellulose waste materials, replacing industrial substrates, which provide the necessary conditions for fungal growth2,3. Given these characteristics, scientists continue to explore the potential of P. ostreatus for waste management in a closed-loop system. The "closed-loop" concept in this study demonstrates how waste can be transformed into valuable resources, creating a system where materials are reused multiple times, thereby minimizing negative environmental impact. In coffee-producing countries, spent coffee grounds are already being used as suitable substrates for cultivating Pleurotus ostreatus, offering an innovative approach to recycling spent coffee grounds (SCG)4. Despite the observed reduction in mycelium growth rate, researchers have explored the incorporating spent coffee grounds into the substrates of Pleurotus pulmonarius and Pleurotus floridanus, noting that highest concentration of SCG used (89% w/w) did not support mycelial growth of both Pleurotus mushrooms5. Nevertheless, the growth rate inhibition does not pose a complete barrier to recycling coffee grounds in the production of Pleurotus ostreatus. This is due to environmental awareness and the reevaluation of the benefits of SCG recycling, considering the ongoing global coffee production6,7. Utilizing SCG as a substrate in the production of Pleurotus ostreatus ensures waste utilization as resources in a new production process, contributing to environmental pollution reduction through closed-loop utilization8.
In addition to its enormous adaptability, Pleurotus ostreatus mushrooms possess the ability to bioaccumulate, including the accumulation of heavy metals9,10. Green Biotechnology, as a component of a closed-loop economy, allows the use of Pleurotus ostreatus mushrooms for mycoremediation of contaminated soil and water. It also offers opportunities for biofermentation, providing substantial benefits for the natural environment and improving living conditions in impoverished countries grappling with hunger and malnutrition11. P. ostreatus also exhibits antioxidant, anti-cancer, anti-inflammatory, anti-hypercholesterolemic, antiviral, and immunostimulatory properties, making it a valuable food source12.
The disposal and accumulation of coal ashes (FA and BA) in the environment is also a concern, particularly as it pertains to electricity generation13. Bottom ash is a byproduct that contains high concentrations of heavy metals and salts. Environmental risk assessments of coal ash storage for the selected power plant were made in a study14 where the risks of ash disposal and its environmental impact are described in detail. Therefore, its safe disposal is essential to mitigate environmental concerns, by using this waste as recyclable materials. One viable option for managing bottom ash is its use in agriculture as a soil amendment15. Bottom ash (BA) has shown potential as a soil additive, with over 100,000 tons annually stored in Portugal meeting PTE limits for soil fertilizers in some countries. In16 researchers found that adding BA to mining soils increased soil water pH and enhanced macro and microelement solubilization. In17 researchers explored using BA from biomass combustion in forestry. In18 researchers demonstrated BA’s benefits as a fertilizer, improving soil texture, yields, and crop quality. Paper19 noted BA’s ability to improve low-quality soils by enhancing pH and nutrient composition. In20 researchers reported increased soil pH and nutrient levels with BA and humus addition. Another paper21 showed BA’s positive effects on seedbed conditions for Acacia mangium cultivation. Notably, BA enhances soil water retention18.
Bottom ash has potential uses in agriculture and forestry. In article17 scientists studied its use from woody biomass power plants in forestry. In18 researchers found that bottom ash as fertilizer improved soil texture, water and air conditions, and crop yields. It can raise soil pH, K, Ca, Mg, and total N content. In19 they noted its benefits for low-quality soils. Paper20 reported increased soil pH and nutrient levels with bottom ash and humus addition. Article21 showed improved seedbed conditions for Acacia mangium cultivation. Notably, bottom ash enhances soil water retention18. While some increase in soil Cd and Co was observed, crop concentrations remained unchanged22.
Clearly, exploring the potential use of agro-industrial waste like bottom ash and coffee grounds as substrates for cultivating P. ostreatus represents an exciting and promising avenue for development. It encourages a deeper exploration of this subject and warrants further investigation. The use of these additives allows for assessing their recycling capabilities and the accumulation of harmful compounds found in bottom ashes while simultaneously reducing environmental pollution. Furthermore, the research results can be compared with the findings of previous experiments conducted by23, who examined the bioremediation potential of Pleurotus ostreatus cultivated using a substrate enriched with fly ash. A comparative analysis will help identify any differences in their applications and enable the selection of suitable additives beneficial for both cultivation and the natural environment.
The main objectives of the study are to investigate the potential use of bottom ash from coal-fired power plants in the cultivation of Pleurotus ostreatus on coffee grounds and to evaluate the accumulation of heavy elements in mushrooms grown on a substrate with added bottom ash. Potential applications of the research results include the use of bottom ash as an additive to the substrate in edible mushroom cultivation, especially at low concentrations (1–5%). It also offers possibilities for using mixtures of coffee grounds and bottom ash as soil additives in conventional cultivation, particularly for poor or degraded soils. This method can be utilized as an element of a circular economy, combining the recycling of industrial waste (bottom ash) and food waste (coffee grounds). There is also potential application in the bioremediation of contaminated soils, leveraging the ability of mushrooms to accumulate heavy metals, as well as the use of post-mushroom cultivation substrate as fertilizer or soil additive due to its enrichment with nutrients.
These studies open up new possibilities in sustainable waste management and food production, while simultaneously contributing to solving problems related to environmental pollution.
Materials and methods
This study employed a completely randomized design (CRD) to investigate the potential use of bottom ash from coal-fired power plants in the cultivation of Pleurotus ostreatus using spent coffee grounds as a substrate. Five treatments were prepared with different substrate compositions (Table 1).
Each treatment was replicated three times, resulting in a total of 15 experimental units. The substrate mixtures were placed in plastic bags and inoculated with P. ostreatus mycelium. The bags were randomly arranged in a controlled environment chamber maintained at 20 °C with 50% humidity. After four days, the bags were perforated to ensure air circulation. Throughout the cultivation process, the following data were collected:
-
1.
Mycelium growth rate
-
2.
Time to fruiting body development
-
3.
Environmental conditions (temperature and humidity)
-
4.
Growth and size of mycelium and mushrooms over time
-
5.
Elemental composition of raw materials, substrates, mycelium, and fruiting bodies
Analytical techniques used for elemental analysis included:
-
1.
ICP-OES for metals, metalloids, silicon, and phosphorus
-
2.
Flame emission photometry for macronutrients (Ca, Mg, Na, K)
-
3.
CHNS elemental analysis
Statistical analysis was performed to evaluate the effects of different bottom ash concentrations on P. ostreatus cultivation and the accumulation of heavy metals and other elements in the mushrooms and post-cultivation substrates.
One-way analysis of variance (ANOVA) was conducted using the average values of elements (As, Cd, Co, Cr, Cu, Fe, Mo, Ni, P, Pb, Si, Zn, N, C, H, S) obtained from the three replicates for each sample type: mycelium, fruiting bodies, and post-cultivation substrate. The significance level was set at α = 0.05. Duncan’s test was performed for post-hoc comparisons to determine which treatments were significantly different from each other (Appendix 1).
The statistical analysis aimed to identify significant differences in elemental composition among the different treatments and sample types, as well as to evaluate the effectiveness of the applied methods compared to the control.
Sample preparation
Five different substrate compositions were prepared for inoculation with Pleurotus ostreatus mycelium. The substrates were composed with the following ratios: I. sample: 300g of coffee grounds II. sample: 300g of coffee grounds, 20g of straw, 3g of bottom ash, III. sample: 300g of coffee, 20g of straw, and 15g of bottom ash, IV. sample: 300g of coffee, 20g of straw, and 30g of bottom ash, V. sample: 100g of bottom ash. The blends were placed into plastic bags and inoculated with mycelium, which was carried into the bags on a layer of wheat grains serving as a carrier. Inoculation trials were done on each mixture three times. These 50 percent humidity substrate bags were placed in a 20 °C chamber. Airflow and separation were ensured by spacing the bags at least 10 cm apart. 4 perforations with dimensions of 1 cm were done in each bags, four days after mycelium inoculation, to provide air circulation. Total mycelium overgrowth was observed in mixes I and II after 14 days. Mix III achieved full overgrowth at 20 days. The bag temperature was monitored during mycelium growth to maintain it below 28 °C. Elevated temperatures may kill mycelium. The substrate turned pale yellow as mycelium colonised it. To promote mushroom development, the temperature was decreased to 18 °C. Periodically ventilating the cultivation chamber removed excess CO2.
The room was in a muted light with 12 h of daily natural light. Fruiting bodies began developing in the perforations and partly under the protective coverings on the fifth, ninth, and twelfth days subsequent to reducing the substrate temperatures for types I, II, and III. To accommodate the mushroom fruiting bodies without causing harm, we delicately widened the holes in our bags to 2 cm. As depicted in Fig. 1, the harvesting process commenced once the cap margins of the largest specimens began to flatten, which is in line with Cornell Small Farms Program guidelines24. Following harvesting, mushroom samples underwent drying and crushing for laboratory analysis.
Investigation approach
The mushroom and substrate samples were tested for metals (Al, Cd, Cr, Cu, Fe, Mn, Mo, Ni, Pb, Zn), metalloids (As, Se), Si, and P using the ICP-OES method (ICPE-9800, Shimadzu). After mineralization in an inorganic acid mixture per PN-EN 16170: 2017-02. Additionally, macronutrients (Ca, Mg, Na, K) were measured using flame emission photometry (BWB-XP, BWB Technologies UK LTD) in compliance with PN-ISO 9964-3 and PN-R-04024:1997. Also, elemental analysis was done using a CHNS analyzer (Elementar Analysesysteme Gmbh, Hanau, Germany) according to25,26. The growth of mycelium and mushrooms was also assessed by measuring their growth and size over time. The cultivated mushrooms from sample 1 reached a height of 63 mm, from sample 2 a height of 52 mm, and from sample 3 only 30 mm. Diameter of the fruiting bodies cap from sample 1 was 75 mm, from sample 2 was 68 mm, and from sample 3 was 34 mm.
These methods are commonly used in analytical chemistry for determining the elemental composition of samples. ICP-OES is particularly useful for trace element analysis, flame emission photometry is effective for alkali and alkaline earth metals, and CHNS analysis provides information about the organic composition of the samples. In the study, 3 independent experimental replications were conducted.
Mineral processing method
The approach of27 mineralized mushroom and substrate biomass samples in HNO3, H2O2, and HF. 9 mL of 65% HNO3 was added to 0.5 ± 0.001 g of the sample and left in a Teflon vessel covered with a lid for 12h for initial mineralization. Then, 1 mL of 30% H2O2 was added and slowly heated at 40 °C for 1h, observing the turbulence of mineralization. After this time, the temperature was raised to 100 °C and mineralized for the next 1h. If necessary, the acid (HNO3) in the vessel was was topped up to a volume of 10 mL. At the end of mineralization, 1 mL of 40% HF was added and heated for 0.5 h. After mineralization, the vessel was cooled and rinsed with water. The mineralizate was transferred to volumetric flasks (50 mL) and topped up with water.
Bottom ash samples were mineralized in aqua regia, i.e. 10 mL of a mixture of 35% HCl and 65% HNO3 in a 3:1 ratio was added to the sample and left in a Teflon vessel covered with a lid for 12 h. The mineralization methodology described above was then followed. The mineralizate was quantitatively transferred with water to volumetric flasks (PP, 50 mL). The mineralizates were analyzed for the content of elements. All reagents were pure for analysis (p.a.) (Chempur, WarChem).
Quality management
Certified Reference Material 0C6A.K1.5N.L5 'Solution of 32 components’ (CPAChem LTD) and the control standard 'ICP–multielement standard XI’ HC394644 (Merck) were used to analyze the elements using the ICP-OES method (used to test for metals (Al, Cd, Cr, Cu, Fe, Mn, Mo, Ni, Pb, Zn), metalloids (As, Se), Si, and P). In order to assess the quality of mineralization and analysis, certified reference material ‘Lichen’ (trace elements)' (IRMM, No. BCR-482) was used. The recovery range for metals and metalloids was 93–111%. Additionally, to control the quality of elemental analysis, the certified reference material ‘AlfaAlfa B2273’ (Cert. No. 41505) was used. The quality management methods used are crucial to ensure the accuracy, precision, and reliability of the ICP-OES analysis results. They helped to validate the analytical procedure and ensure the confidence of the data obtained from the study.
Risk assessment
The uncertainty analysis included two categories: u(A) for sample repeatability and u(B) for alpha-alpha 43221. These uncertainties involve C = 43.73%, H = 6.05%, N = 3.01%, and S = 0.27%. The CERTIFIED REFERENCE MATERIAL BCR® – 482 was used to analyse metal samples using the BWB-XP photoelectric flame photometer from BWB Technologies UK LTD. In28 uncertainty propagation approach was used to analyse Y_i’s uncertainties. The Joint Committee for Guides in Metrology (2011) recommends this procedure. The formula for estimating u(Y_i) is as follows:
In this context, u(X_i) signifies the standard uncertainties associated with the measured variables (X_i). The determination of the expanded uncertainty U(Y_i) assumed a coverage factor k of 2, corresponding to a 95% confidence level. The calculation is expressed as follows:
The documented acceptable margin of error for the tested samples is also provided.
Results and discussions
Ingredients utilized in creating mixtures
Coffee grounds, straw, and bottom ash were mixed to grow Pleurotus ostreatus. The elemental makeup is shown in Table 5. Among the materials utilized, the highest concentration of elements, including C, H, N, and S (as shown in Table 5), is predominantly present in coffee grounds (C = 53%; H = 6.74%; N = 2.32%; S = 0.17%), followed by straw (51.22%). In contrast, bottom ash exhibits a significantly lower content of these elements at just 6.13%. Pleurotus species rely on carbon and nitrogen as their primary nutritional sources, in addition to inorganic compounds. Furthermore, maintaining a higher carbon content relative to nitrogen content is crucial, as emphasized by29.
Mycelium and fruiting bodies of mushrooms cultivated on substrates I, II, and III
Despite inoculation of the mixtures No. IV (300 g grounds, 20 g straw, 30 g bottom ash) and V (bottom ash) with mycelium, mycelium and fungal fruiting bodies were not grown. The fungal fruiting bodies grown on substrates I, II and III (Fig. 2) had a higher content of carbon (42.16–43.06% on average), hydrogen (5.54–5.79% on average) and nitrogen (3.68–5.91% on average) than mycelium (carbon average: 39.81–41.50%, hydrogen average: 4.70–4.80%, nitrogen average: 2.81–3.31%). However, in the case of sulfur, the highest content was obtained for mycelium from substrate I (0.82%) and the lowest for fungal fruiting bodies from substrate III (0.3%) despite the fact that the content of this element found in substrate III was the highest of the three substrates tested and amounted to 0.41% (Table 2).
Findings of substrate post-cultivation
The substrates I–IV (Fig. 3) were depleted in carbon by an average of 11.91% after mushroom cultivation, with the largest decrease recorded for substrate I, i.e. 100% coffee grounds. The reductions for hydrogen were also achieved (by an average of 1.79%) with the highest reduction for coffee grounds, i.e. -2.73%. In the case of 100% bottom ash substrate after inoculation with mycelium (substrate V), there was a significant increase in carbon content compared to bottom sediments alone — from 0.35% to 17.18%, and an increase in hydrogen from 0.02% to 1.58%. This phenomenon may result from the processes carried out by the grafted mycelium during the cultivation period despite the lack of visible growth of mycelium and fungal fruiting bodies, and from the addition of water. The post-cultivation substrates II–V contained higher amounts of nitrogen, and a slight decrease in this element (0.06%) was obtained in substrate I. The sulfur content increased in substrates I–IV (by an average of 0.41%) with the largest increase of 1% recorded for substrate I. While for the bottom ash, a significant reduction in sulfur content from 5.74% to 1.61% was achieved. The enrichment of post-cultivation substrates with nitrogen (substrates II–V) and the increase in sulfur content (substrates I–V) gives them useful fertilizer properties. The use of bottom ash as an ingredient to improve soil properties has been part of the research of many authors18,20,22. They pointed out, among other things, to the increase in nitrogen content of soils fertilized with bottom ash. In a study presented by20, the use of bottom ash mixed with humus affected its nutrient content. Thus, it seems possible to achieve similar fertilizer properties by adding coffee grounds and straw. As reported in reference30, the deposition of metals in mushroom fruiting bodies is significant and can exceed their concentrations in the growth substrate. Sample values for cadmium (Cd), referenced to organizational guidelines and consumption norms, are presented in Table 3. Interesting results were obtained for Cd concentrations in fungal fruiting bodies. As previously mentioned, fruiting bodies were grown only for mixes I, II and III (Fig. 4). The Cd content for fruiting bodies grown on coffee ground substrate was the highest — as high as 0.316 mg/kg. A smaller content (approx. six times less) was observed for samples of P. ostreatus grown on substrate with 3 g of bottom ash (mix II), i.e. 0.063 mg/kg, and a similar amount of 0.05 mg/kg in fruiting bodies grown on substrate with 15 g of bottom ash (mix III). Referring to the latest European Commission guidelines ((EU) 2021/1323), for Pleurotus ostreatus grown on coffee grounds, the Cd value is exceeded twice. In the study31, the average cadmium content of mushrooms (Agaricus bisporus, Lentinula edodes and Pleurotus ostreatus) was 0.17 mg/kg. A similar value was obtained for both mycelium for mixes II and III, and for mushrooms and it amounted to: 0.05 mg/kg, while the Cd level for the sample grown on coffee grounds was 0.163 mg/kg, and thus approx. three times less than in the mushrooms.
When analyzing the raw materials (Table 2) used to prepare growth substrate mixtures for the presence of cadmium (Cd), variations indicating significantly different levels of the analyzed element within the sample areas can be observed. The highest concentration of cadmium (0.736 mg/kg) was observed in bottom ash samples (Fig. 5), which, when combined with fly ash and straw (0.05 mg/kg), clearly indicates the direction of transfer. It’s worth noting that the methodology employed allowed for the detection of elements at a level as low as 50 µg, which, in fact, was the lowest recorded value among all the analyzed groups. With such a detection limit, the results indicating differences in the chemistry of growth substrates before and after cultivation become of significant importance. The analysis of cadmium content showed declines in all groups. Confirmation of the accuracy of the statistical data processing method used can also be found in the observation of a positive correlation between the amount of bottom ash added and the Cd content in the post-cultivation substrate. Similarly, for chromium (Cr), values related to consumption are presented in Table 4.
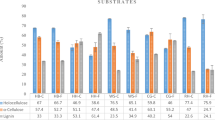
The bottom ash substrate samples had the greatest concentration of chromium (Cr) among the raw materials tested (68.89 mg/kg), whereas the coffee grounds and straw samples had similar concentrations of 2.29 and 3.33 mg/kg, respectively. This element was found in the highest concentration in the bottom ash substrate sample. A decrease in Cr was observed in all post-cultivation substrates containing bottom ash compared to the initial state for bottom ash alone. Another correlation emerged: as the content of bottom ash increased, so did the Cr content in the substrate after cultivation. Nonetheless, a decline in Cr content was noted in the substrates post-cultivation, where bottom ash was added and fungal fruiting bodies were cultivated, in contrast to those exclusively composed of coffee grounds. The Cr concentration in mushrooms grown solely on coffee grounds measured 21.21 mg/kg, whereas the mycelium on this substrate exhibited notably lower Cr content, at 15.61 mg/kg. The Cr content was low in the samples of fungal fruiting bodies grown on the substrates containing 3 g and 15 g of bottom ash: 3.86 mg/kg and 1.95 mg/kg, respectively, while the content of this element fund in the mycelium was higher, i.e.: 8.05 mg/kg (mix II) and 11.09 mg/kg (mix III). Calcium, magnesium, potassium, phosphorus as essential minerals, and trace elements such as copper, zinc, manganese, molybdenum, and iron found in mushrooms depict mushrooms as a valuable source of these nutrients38.
The European Food Safety Authority (EFSA) recommends Adequate Intake (AI) copper consumption39. Copper intake for adults over 18 years old varies by European country, ranging from 1.15 to 2.07 μg/day40. A vegetarian diet increases copper intake, which affects the average intake. The average adult copper intake was 1.15–2.07 mg/day37. Copper content in research raw materials was highest in bottom ash (52.88 mg/kg), followed by coffee grounds (18.16 mg/kg) and straw (6.17 mg/kg). This element was found in the post-cultivation substrate at 27.23–40.49 mg/kg, and the higher the bottom ash additive quantity, the higher the fraction. All samples had similar mycelium copper content (40.9–44.08 mg/kg). The copper content of the fungal fruiting bodies varied: mix I mushrooms had 45.37 mg/kg copper, mix II mushrooms had 71.27 mg/kg, and mix III mushrooms had 32.07 mg/kg. According to41, Pleurotus ostreatus had 8.72 mg/kg Cu. The average copper content of oyster mushroom was 2.70 mg/kg (dry weight)30. The EFSA indicates that elevated consumption levels of nickel, chromium, manganese, chlorine, phosphorus, iron, tin, potassium, silicon, sodium, and vanadium do not pose health risks. There were fluctuations in the concentration of nickel among the raw materials studied. Most was 40.93 mg/kg in bottom ash, followed by 2.99 mg/kg in coffee grinds and 6.05 mg/kg in straw. After mushroom development, the substrate had 11.24 to 67.51 mg/kg nickel. Interestingly, bottom ash addition did not affect nickel content in the post-cultivation substrate. Mycelium nickel content ranged from 12.97 to 17.32 mg/kg among samples. However, fungal fruiting bodies have more nickel variation. After growing mushrooms on mix I, mix II, and mix III, the nickel concentration was 28.63, 7.6, and 3.35 mg/kg, respectively.
According to41, Pleurotus ostreatus had a nickel concentration of 1.16 mg/kg, ranging from 0.33 to 4.44 mg/kg. Nickel was detected at 0.248 mg/kg (dry weight) in champignon mushroom (Agaricus bisporus) fruiting bodies by42. Adamiak et al.43 investigated the heavy metal content of selected forest mushroom species from the Wysoczyzna Siedlecka region in 2013. Brown bay bolete mushrooms had an average nickel concentration of 1.66 mg/kg (dry weight), ranging from 1.44 to 2.14. Regulation44 limits lead in mushrooms, including Pleurotus ostreatus, to 0.03 mg/kg (fresh weight). The available information regarding the lead content in edible mushrooms lacks consistency. Adamiak et al.43 and Spodniewska et al.45 reported lead concentrations within permissible limits set by national regulations46 and EU regulations36 and47 for all edible mushrooms they examined. However, specific varieties such as chanterelles, birch bolete, porcino, and brown bay bolets could contain lead levels reaching 0.3 mg/kg (fresh weight) under these established criteria. In48 researchers found lead concentration above threshold values in parasol mushrooms from Poniatowa, Lublin voivodeship. Lead was highest in bottom ash at 81.26 mg/kg in this study’s raw material samples. Coffee grounds had 3.85 mg/kg lead, while straw had 6.39 mg/kg. The substrate’s lead concentration ranged from 9.06 mg/kg to 64.15 mg/kg after mushroom culture. Note that coffee ground substrate has 22.54 mg/kg lead after cultivation. Bottom ash incorporation correlated with post-cultivation substrate lead levels. Bottom ash increases substrate lead content. Mushroom lead levels varied too. The lead concentration of mix I mushrooms was 29.34 mg/kg, while mix II mushrooms had 6.35 mg/kg. Lead concentration in mix III mushrooms was 2.88 mg/kg. Lead levels in mycelium ranged from 16.92 to 20.25 mg/kg. According to43 study on heavy metals in Wysoczyzna Siedlecka forest mushroom species, brown bay bolete mushrooms had an average lead concentration of 0.234 mg/kg (dry weight), ranging from 0.142 to 0.357. Zinc is crucial for more than 300 enzymes, encompassing those involved in protein production37. It plays a vital role in the synthesis of chemicals that regulate plant growth. Mushrooms serve as a source of this essential nutrient38. On average, European adults intake between 8.0 to 14.0 mg of zinc daily40. The Recommended Dietary Allowance (RDA) stands at 11 mg for men and 8 mg for women37 to meet the body’s zinc needs. Top zinc content in test mixture raw materials was 123.3 mg/kg in bottom ash. Coffee ground contains 10.78 mg/kg zinc, while straw had 16.02 mg/kg. After mushroom development, the substrate had 15.82–69.02 mg/kg zinc. After cultivation, coffee ground substrate contained 27.33 mg/kg zinc. Higher bottom ash fractions increased zinc levels in the post-cultivation substrate. Zinc levels varied in farmed mushrooms. Mix I mushrooms had 96.76 mg/kg, while mix II mushrooms had 103.06 mg/kg. Mix III mushrooms had 59.68 mg/kg zinc. Zinc levels in mycelium ranged from 43.85 to 64.08 mg/kg. The typical amount of zinc found in brown bay boletes (Xerocomus badius), white and brown champignons (Agaricus bisporus), and oyster mushroom (Pleurotus ostreatus) was reported by41: 60.6 mg/kg (dry weight) for brown bay bolete, 29.8 mg/kg for white champignons, 31.3 mg/kg for brown champignons, and 25.7 mg/kg per oyster mushroom. Kalembasa et al.42 study on trace elements and heavy metals in champignon mushroom (Agaricus bisporus) fruiting bodies showed zinc at 48.5 mg/kg (d.w.), ranging from 35.0 to 59.8 mg/kg. Adamiak et al.43 study on heavy metals in selected forest mushroom species from Wysoczyzna Siedlecka found that brown bay bolete mushrooms had an average zinc content of 26 mg/kg (d.w.), ranging from 12.1 to 13.1 mg/kg.
Iron (Fe) (Tables 5 and 6) uptake by mushrooms was inhibited by bottom ash in the growing substrate. Post-cultivation substrate iron increased due to bottom ash’s high iron content. Compared to coffee grounds and straw, bottom sediments have somewhat more structural phosphorus (P). In the coffee ground and bottom ash substrates, both mycelium and mushrooms exhibited the highest phosphorus levels, particularly in mix II mushrooms, showcasing the highest concentration. Post-cultivation substrate No. II, combined with mycelium in bottom ashes, showcased the highest manganese (Mn) levels, albeit only reaching half the initial bottom ash concentration pre-cultivation. While silicon (Si) primarily resided in bottom ashes, its impact on substrate chemistry was minimal, yet it persisted in substantial quantities within the mycelium. Conversely, an escalation in bottom ash within the mix hindered the mushrooms’ absorption of silicon. Selenium, a trace element, varied from 5 mg/kg in bottom ash to 0.12 mg/kg in straw. However, bottom ash had almost 80,000 mg/kg aluminium. Similar aluminium values of 13.6 to 17.1 mg/kg showed that mushrooms grown on bottom ash substrates could not bioremediate aluminium. Mixture No. II had more than 1,100 mg/kg more aluminium than Mixture No. I in the post-cultivation substrate. Growth substrate No. 2 mycelium has 236 mg/kg sodium (Na), while straw had 77 mg/kg. Mycelium-inoculated post-cultivation bottom ash substrate had 38 mg/kg more sodium than pure bottom ash. Potassium (K) was collected by mushrooms on substrate No. II at 4041 mg/kg, similar to coffee ground substrate (4025 mg/kg). From 356 mg/kg for bottom ash to 527 mg/kg for substrate No. 2, the five post-cultivation substrates had similar potassium concentration. More bottom ash in the substrate lowered mushroom potassium bioaccumulation. However, 1% bottom ash increased mushroom potassium content. The bottom ashes contain almost 23,000 mg/kg of calcium (Ca), yet adding them to the growing substrate lowered mushroom calcium concentration compared to coffee grounds. Only substrates 2 and 3 mycelium had high calcium content. Calcium concentration in post-cultivation substrates exceeded that of coffee powder substrate by 800 mg/kg only with at least 10% bottom ash. Post-cultivation substrates with less bottom ash had less calcium. Inoculation mycelium with bottom ash reduced calcium (Ca) levels by over twofold. Mg uptake by mycelium and mushrooms on bottom ash substrates was similar. Lower magnesium content was found in the post-cultivation substrate with bottom ash than in coffee grounds alone. The post-cultivation substrate did not exceed the magnesium level of coffee grinds.
Conclusions
The study reveals promising potential for the management of both cultivated mushrooms and post-cultivation substrate. Mushrooms grown on substrates with 1–5% bottom ash addition demonstrated lower levels of chromium, nickel, and lead compared to those grown solely on coffee grounds. Notably, mushrooms cultivated on 1% bottom ash substrate showed higher accumulation of beneficial elements such as phosphorus, copper, and zinc, while maintaining similar levels of manganese, sodium, and potassium across all samples. These findings suggest that mushrooms grown with small amounts of bottom ash could potentially be safe for consumption, though further testing is necessary to confirm this.
Regarding the post-cultivation substrate, the addition of bottom ash increased levels of certain nutrients like phosphorus and potassium. The substrate with 1% bottom ash exhibited lower heavy metal content compared to the coffee ground substrate alone and improved moisture retention properties. These characteristics indicate that the post-cultivation substrates, particularly those with low percentages of bottom ash, could potentially serve as soil amendments or fertilizers, especially for depleted or light soils.
The authors emphasize the need for additional research and trials before confirming the feasibility of using these materials on a larger scale, particularly for human consumption or agricultural use. The optimal rate of bottom ash addition appears to be between 1 and 5%, balancing mushroom growth and minimizing heavy metal accumulation. The study provides a promising foundation for exploring circular economy approaches in mushroom cultivation and waste management, showcasing the diverse potential applications for post-cultivation substrates. However, further safety and efficacy assessments are required before implementing these findings on a broader scale.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Melanouri, E.-M., Dedousi, M. & Diamantopoulou, P. Cultivating Pleurotus ostreatus and Pleurotus eryngii mushroom strains on agro-industrial residues in solid-state fermentation. Part I: Screening for growth, endoglucanase, laccase and biomass production in the colonization phase. Carbon Resour. Convers. 5, 61–70 (2022).
Bellettini, M. B. et al. Factors affecting mushroom Pleurotus spp. Saudi J. Biol. Sci. 26, 633–646 (2019).
Sujiyama, S.-I. & Ueno, H. Performance of wood-rotting fungi-based enzymes on enzymic saccharification of rice straw. J. Sci. Food Agric. 93, 2841–2848 (2013).
Carrasco-Cabrera, C. P., Bell, T. L. & Kertesz, M. A. Caffeine metabolism during cultivation of oyster mushroom (Pleurotus ostreatus) with spent coffee grounds. Appl. Microbiol. Biotechnol. 103, 5831–5841. https://doi.org/10.1007/s00253-019-09883-z (2019).
Chai, W. Y., Krishnan, U. G., Sabaratnam, V. & Tan, J. B. Assessment of coffee waste in formulation of substrate for oyster mushrooms Pleurotus pulmonarius and Pleurotus floridanus. Future Foods 4, 100075. https://doi.org/10.1016/j.fufo.2021.100075 (2021).
Adams, M. R. & Dougan, J. Waste products. In Coffee (eds Clarke, R. J. & Macrae, R.) 257–291 (Springer, 1987).
Bharat Helkar, P. & Sahoo, A. Review: Food industry by-products used as a functional food ingredients. Int. J. Waste Resour. 6, 1000248 (2016).
Djekic, I. et al. Review on environmental models in the food chain-current status and future perspectives. J. Clean. Prod. 176, 1012–1025 (2018).
Faria, M. G. I. et al. Bioaccumulation of lithium (Li2CO3) in mycelia of the culinary-medicinal oyster mushroom, Pleurotus ostreatus (Agaricomycetes). Int. J. Med. Mushrooms 20(9), 901–907. https://doi.org/10.1615/IntJMedMushrooms.2018027343 (2018).
Bressa, G., Cima, L. & Costa, P. Bioaccumulation of Hg in the mushroom Pleurotus ostreatus. https://doi.org/10.1016/0147-6513(88)90020-6 (1988).
El-Ramady, H. et al. Green biotechnology of oyster mushroom (Pleurotus ostreatus L.): A sustainable strategy for myco-remediation and bio-fermentation. Sustainability 14, 3667. https://doi.org/10.3390/su14063667 (2022).
Melanouri, E.-M., Dedousi, M. & Diamantopoulou, P. Cultivating Pleurotus ostreatus and Pleurotus eryngii mushroom strains on agro-industrial residues in solid-state fermentation. Part II: Effect on productivity and quality of Carposomes. Carbon Resour. Convers. 5, 52–60 (2022).
Wang, N., Sun, X., Zhao, Q., Yang, Y. & Wang, P. Leachability and adverse effects of coal fly ash: A review. J. Hazard Mater. 396, 122725. https://doi.org/10.1016/j.jhazmat.2020.122725 (2020).
Lemly, A. D. Environmental hazard assessment of coal ash disposal at the proposed Rampal power plant. Hum. Ecol. Risk Assess. Int. J. 24(3), 627–641. https://doi.org/10.1080/10807039.2017.1395685 (2018).
Jayaranjan, M. L. D., van Hullebusch, E. D. & Annachhatre, A. P. Reuse options for coal fired power plant bottom ash and fly ash. Rev. Environ. Sci. Biotechnol. 13, 467–486. https://doi.org/10.1007/s11157-014-9336-4 (2014).
Cruz, N. C. et al. Ashes from fluidized bed combustion of residual forest biomass: Recycling to soil as a viable management option. Environ. Sci. Pollut. Res. 24, 14770–14781. https://doi.org/10.1007/s11356-017-9013-6 (2017).
Nurmesniemi, K., Manskinen, R. & Pöykiö, O. D. Forest fertilizer properties of the bottom ash and fly ashfrom a large-sized (115 MW) industrial power plant incinerating wood-based biomass residues. J. Univ. Chem. Technol. Metallur. 47(1), 43–52 (2012).
Wearing, C., Birch, C. & Nairn, J. An assessment of Tarong bottom ash for use on agricultural soils. Dev. Chem. Eng. Miner. Process. 12, 531–543. https://doi.org/10.1002/apj.5500120508 (2004).
Singh, S., Gond, D. P., Pal, A., Tewary, B. K. & Sinha, A. Performance of several crops grown in fly ash amended soil. In World of Coal Ash (WOCA) Conference; 2011 May 9–12, USA (2011).
Oklima, A. M., Iskandar, S. & Tjahyandari, S. D. Utilizing coal ash and humic substances as soil ameliorant on reclaimed post-mining land. J. Trop. Soils 19(3), 161–169 (2014).
Hamanaka A., Shimada H. & Ogata S. Utilization of coal ash for preparation of seedbed in disturbed land in Indonesian Open Cut Coal Mine. J. Pol. Miner. Eng. Soc. 109–116 (2016).
Agustini, R. Y. et al. The utilization of bottom ash and cow manure compost as soil ameliorant on acid mineral soil and its effects on mustard (Brassica juncea). J. Trop. Soils 22(2), 51–58. https://doi.org/10.5400/jts.15.2.95 (2017).
Hnydiuk-Stefan, A. et al. Accumulation of pollutants from fly ash in Pleurotus ostreatus and a substrate based on coffee grounds by elemental analysis using the ICP-OES method and photometric method. Environ. Sci. Pollut. Res. 30, 88197–88212. https://doi.org/10.1007/s11356-023-28751-y (2023).
Cornell Small Farms Program. https://smallfarms.cornell.edu/projects/mushrooms/harvest-to-market-guide/post-harvesting-handling/. Accessed 1 Oct 2023.
PN-G-04571. Solid Fuels—Determination of Carbon, Hydrogen and Nitrogen Content with Automatic Analyzers. Macro Method (Polish Committee for Standardization, 1998).
PN-G-04584. Solid Fuels—Determination of Total Sulphur and Ash Content with Automatic Analyzers (Polish Committee for Standardization, 2001).
Ostrowska, A., Gawliński, S. & Szczubiałka, Z. Methods of analysis and assessment of soil and plant properties (IOS, 1991) (in Polish).
Junga, R., Chudy, P. & Pospolita, J. Uncertainty estimation of the efficiency of small-scale boilers. Measurement 97, 186–194. https://doi.org/10.1016/j.measurement.2016.11.011 (2017).
Hoa, H. T., Wang, C. L. & Wang, C. H. The effects of different substrates on the growth, yield, and nutritional composition of two oyster mushrooms (Pleurotus ostreatus and Pleurotus cystidiosus). Mycobiology 43(4), 423–434. https://doi.org/10.5941/MYCO.2015.43.4.423 (2015).
Florczak, J., Chudy, J., Barasińska, M. & Karwowski, B. Wybrane składniki odżywcze grzybów dziko rosnących: uszaka bzowego (Hirneola auricula judae), boczniaka ostrogowatego (Pleurotus ostreatus), i zimówki aksamitnotrzonowej (Flammulina velutipes) [eng: Selected nutrients of wild mushrooms: lilac (Hirneola auricula judae), oyster mushroom (Pleurotus ostreatus), and winterworm (Flammulina velutipes)]. Bromat. Chem. Toksykol. 47(4), 876–882 (2014).
Gapinski, M. & Wozniak, W. Mikroelementy w uprawnych grzybach wielkoowocnikowych. Zesz. Probl. Postępów Nauk Rol. 434(2), 641–646 (1996).
(EU) 2021/1323, COMMISSION REGULATION of 10 August 2021 amending Regulation (EC) No 1881/2006 as regards maximum levels of cadmium in certain foodstuffs. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32021R1323.
Wojciechowska-Mazurek, M., Starska, K., Brulińska-Ostrowska, E., Karłowski, K. & Grudzińska, B. Ocena pobierania metali szkodliwych dla zdrowia z całodziennymi racjami pokarmowymi dzieci i młodzieży w wybranych województwach. Bromat. Chem. Toksykol. 267, 101–103 (2003).
Friberg L., Elinder C.G., Kjelstrom T. Cadmium. Enviromental Health Criteria 134 (World Health Organisation, 1992).
Jurkowska, K., Sawicka, E. & Piwowar, A. Chrom–pierwiastek już dobrze znany czy wciąż nieznany–dwa oblicza działania. Farm. Pol. 75(4), 208–218 (2019).
(EU) No 1169/2011 Regulation of the European Parliament and of the Council of 25 October 2011 on the provision of food information to consumers.
Jarosz, M., Rychlik, E., Stoś, K. & Charzewska, J. Normy żywienia dla populacji Polski i ich zastosowanie (EN: Nutrition standards for the Polish population and their application) 68–437. (Narodowy Instytut Zdrowia Publicznego-Państwowy Zakład Higieny, 2020).
Mattila, P. et al. Contents of vitamins, mineral elements, and some phenolic compounds in cultivated mushrooms. J. Agric. Food Chem. 49, 2343–2348 (2001).
Bost, M. et al. Dietary copper and human health: Current evidence and unresolved issues. J. Trace Elem. Med. Biol. 35, 107–115 (2016).
Agostoni, C., Bresson, J. L., Fairweather-Tait. S. et al. EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific Opinion on Dietary Reference Values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 8(3), 1461. https://doi.org/10.2903/j.efsa.2010.1461 (2010).
Kuziemska, B., Wysokiński, A., Jaremko, D., Popek, M. & Kożuchowska, M. Zawartość wybranych metali ciężkich w grzybach jadalnych (EN: Content of selected heavy metals in edible mushrooms). Inż. Ekol. 19(1) (2018).
Kalembasa, D., Becher, M. & Rzymowski, D. Wybrane pierwiastki śladowe oraz metale ciężkie w podłożu, okrywie i owocnikach pieczarki (Agaricus bisporus) (EN: Selected trace elements and heavy metals in the substrate, casing and fruiting bodies of mushrooms (Agaricus bisporus)). Ochrona Śr. Zasobów Nat. 52, 86–92 (2012).
Adamiak, E. A., Kalembasa, S. & Kuziemska, B. Zawartość metali ciężkich w wybranych gatunkach grzybów jadalnych (EN: The content of heavy metals in selected species of edible mushrooms). Acta Agrophys. 20(1), 7–16 (2013).
(EU) 2021/1317 Commission Regulation of 9 August 2021 amending Regulation (EC) No 1881/2006 as regards maximum levels for lead in certain foodstuffs.
Spodniewska, A., Barski, D. & Zasadowski, A. Zawartość kadmu i ołowiu w wybranych gatunkach grzybów pochodzących z województwa warmińsko-mazurskiego (EN: Cadmium and lead content in selected species of mushrooms from the Warmian-Masurian Voivodeship). Ochrona Śr. Zasobów Nat. 41, 135–141 (2009).
Journal of Laws 2003, no.37, item. 325 and 326, REGULATION OF THE MINISTER OF HEALTH of Poland of 13 January 2003 on the maximum levels of chemical and biological contaminants that may be present in food, food ingredients, permitted additives, processing aids or on the surface of food.
(EC) No 1881/2006 COMMISSION REGULATION (EC) No 1881/2006 of 19 December 2006 establishing maximum levels for certain contaminants in foodstuffs (Text with EEA relevance) (OJ L 364, 20.12.2006).
Falandysz, J., Gucia, M. & Mazur, A. Niektóre składniki mineralne i ich współczynnik biokoncentracji w czubajce kani (Macrolepiota procera) z okolic Poniatowej w woj. Lubelskim (EN: Some minerals and their bioconcentration factor in czubajce kani (Macrolepiota procera) from the vicinity of Poniatowa in the province Lublin). Bromatol. Chem. Toksykol. XL(3), 249–255 (2007).
Author information
Authors and Affiliations
Contributions
A.H.-S., J.B.K., D.B.M., Ł.B.: Experiments, investigations, writing and editing, funding acquistion. Ż.G., M.B., R.J., K.B., R.R.: Writing, review, editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The authors Anna Hnydiuk-Stefan, Jolanta Beata Królczyk, Dominika Barbara Matuszek, Łukasz Biłos, Żaneta Grzywacz, Marta Bożym, Robert Junga, Karol Bierczyński, Ritu Rai confirm that the experiments complied with relevant institutional, national, and international guidelines and legislation.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hnydiuk-Stefan, A., Królczyk, J.B., Matuszek, D.B. et al. Impact of bottom ash addition on Pleurotus ostreatus cultivation on coffee ground substrate. Sci Rep 14, 31890 (2024). https://doi.org/10.1038/s41598-024-83434-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-83434-z