Abstract
This study aimed to evaluate the diagnostic accuracy of ultrasonography in assessing the response of clipped axillary lymph nodes to neoadjuvant chemotherapy. Between February 2022 and September 2023, 43 patients who underwent axillary lymph node marking for targeted axillary dissection were retrospectively analyzed. Ultrasonography parameters such as the number, size, shape, cortical thickness, hilum status, and treatment response of the clipped lymph node were assessed. Post-surgery pathology results served as the gold standard. Ultrasonography revealed 70% complete and 30% partial response, while pathology results showed 51% complete response, 9% micro-metastases, and 40% macro-metastases. The diagnostic accuracy of ultrasonography was 81.4%, with 61.9% sensitivity and 100% specificity. A significant correlation was found between clipped node response in ultrasound and pathology. Additionally, a notable association was observed between clipped node response on ultrasonography, molecular subtype of the breast mass, and the mass’s response to NAC. Assessing the treatment response of clipped lymph nodes with preoperative ultrasound, followed by surgical excision using needle wire localization, can be a viable alternative to axillary dissection, offering low false-negative rates.
Similar content being viewed by others
Introduction
Neoadjuvant chemotherapy (NAC) is used in the treatment of operable and inoperable breast cancer patients1. NAC administered to patients with locally advanced breast cancer plays an important role in reducing tumor stage, decreasing tumor size, and treating axillary lymph node metastases2,3. Axillary lymph node metastasis has an important role in clinical staging and prognosis4. The complete response rates in patients have increased with new chemotherapy drugs. Studies have demonstrated that placing a marker within the breast mass for tumor localization in patients undergoing NAC and planned breast-conserving surgery is both useful and safe5,6. Depending on the response to NAC, either breast-conserving treatment or modified radical mastectomy is planned. Sentinel lymph node biopsy (SLNB) or axillary lymph node dissection (ALND) is performed based on the response of the axillary lymph nodes, followed by adjuvant chemotherapy, radiotherapy, and hormone therapy combinations tailored to the patient’s histopathology and receptor status3. The primary goals of axillary lymph node dissection in breast cancer include locally controlling the disease, accurately staging the disease for appropriate systemic and radiotherapeutic treatment, and improving overall survival7. A 40–50% axillary complete response rate is reported with NAC4,7.
In breast cancer patients scheduled for neoadjuvant chemotherapy, it is common practice to place a metallic marker (clip) within the mass and metastatic axillary lymph node under US guidance before treatment. This clip aids the radiologist, surgeon, and pathologist in the post-treatment period, ensuring appropriate marking and removal of the tumor bed, adjacent breast tissue, and metastatic axillary lymph node for evaluation. Disposable automatic marking needles under US guidance are utilized in this clip marking procedure.
In recent years, extensive and radical axillary surgery has been replaced by minimal axillary surgery using SLNB8. The adequacy of sentinel lymph node biopsy instead of axillary dissection in appropriate patients was also investigated in the ACOSOG Z0011 and AMAROS clinical trails7,8. ALND is a significant cause of morbidity in breast cancer patients9. With the rise in pathological complete response [pCR] rates in the axillary lymph nodes due to targeted therapy, the necessity of ALND in such cases is being questioned9. Therefore, placing a clip on the axillary lymph node at the time of initial diagnosis, particularly in patients with 1–2 metastatic lymph nodes in the axilla, is crucial for locating this lymph node in the post-treatment period and facilitating minimal axillary surgery. The response of axillary lymph node metastasis, as well as the response of the breast mass, affects the surgical treatment to be applied10. The false negative rate of SLNB in showing residual disease after NAC was reported to be 10–14%, and this rate can be reduced by the addition of targeted axillary dissection in appropriate patients9,11. Targeted axillary dissection (TAD) involves marking the malignantly diagnosed axillary lymph node before NAC and removing it during surgery, showing promising identification rates and false negative rates (FNR)9,11,12,13. Recent studies indicate that TAD can reduce false negative rates and and improve axillary nodal staging in node-positive patients following NAC7,14.
In cases of axillary complete response, performing TAD and removing the clipped lymph node eliminates the need for extensive axillary dissection, reducing patient morbidity and improving quality of life12,15. The most appropriate imaging method for evaluating the axillary region is ultrasound (US)8,16. The presence of the lymph node clipped before treatment can be easily assessed with US, and comments can be made regarding its response to treatment4,8. By examining characteristics such as shape, cortex thickness, short axis-long axis ratio, presence of hilus loss, and cortex echogenicity of the lymph node, information can be provided about treatment response based on whether the lymph node exhibits pathological morphology4,8.
In this study, our aim was to assess the diagnostic accuracy of ultrasonography in determining the response of axillary metastatic lymph nodes, previously marked with a clip before surgery, to neoadjuvant chemotherapy.
Materials and methods
This retrospective study was approved by the Ege University Faculty of Medicine Medical Research Ethics Committee, waiving the requirement for patient informed consent. The ethics committee approval decision number is 23-11T/6. It was conducted in accordance with the Declaration of Helsinki and relevant guidelines and regulations. Between February 2022 and September 2023, axillary lymph node positive breast cancer patients who had undergone targeted axillary dissection with clipped node were retrospectively evaluated.
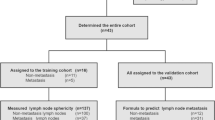
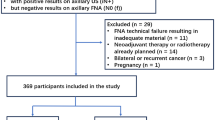
A total of 43 patients who underwent TAD, with available radiological imaging and pathology results, were included in the study. Five patients with incomplete radiological imaging and without surgical pathology results were excluded from the study (Fig. 1).
The radiological images were retrospectively reexamined using a picture archiving and communication system (Sectra IDS7 Workstation, Sectra AB, Sweden). US images were obtained with 18L6HD probe (Siemens Healthineers, Acuson S2000). Mammography (MG) images were performed with routine craniocaudal (CC) and mediolateral-oblique (MLO) positions (Digital Mammography System 2D/3D, Selenia Dimensions, Hologic). MRI scans were performed using 1.5-Tesla (Siemens Healthineers, Magnetom Amira & Symphony, Erlangen, Germany) and 3-Tesla (Siemens Healthineers, Magnetom Verio, Erlangen, Germany) MRI units.
From the radiological imaging (US, MG, MRI) conducted before systemic treatment, the distribution and diameter of the breast mass, axillary lymph node status, as well as the tumor histological type and molecular subtype were evaluated based on the biopsy pathology results. In multifocal and multicentric tumors, the diameter of the largest lesion was measured and recorded. Before and after NAC, US was used to assess the number of metastatic axillary lymph nodes, the size of the clipped lymph node, as well as its shape, cortical thickness, long-short axis ratio, cortex echogenicity and hilum status.
Ultrasonography findings were used to determine the response of the axillary lymph node to the NAC. If the clipped node was not seen on US, it was considered a complete response. If the clipped node had decrease in size but had a thick cortex (> 2 mm), was round in shape, or had hilum loss, it was considered a partial response. If it had not decrease in size or had enlarged, it was considered a non-response.
Post-surgery pathology results were used to record responses to treatment in the breast mass, the size of any remaining tumor if present, the size of metastatic foci in the axilla, and the number of metastatic lymph nodes.
Axillary marking method
A clip was placed on the malignant breast mass and the axillary lymph node in the same session under ultrasound guidance. We used sterile placement needle (18 gauge, 15 cm) with preloaded 99.99% Pure Gold® marker (1 mm × 4 mm) (Geotek® medical, Ankara, Turkiye) for clip marking procedure.
Clipped nodes observed after NAC were evaluated for axillary treatment response using ultrasound (Fig. 2).
After NAC, the responses of the clipped node, which could be seen with US, were evaluated with the parameters described above, and these clipped lymph nodes were marked with a needle wire under US guidance. Since the clipped lymph nodes that responded completely after the treatment were not visible on US, they were marked with a needle wire under CT guidance and excised surgically (Fig. 3). Specimen mammography was performed on all clipped lymph nodes marked under US and CT guidance, and it was confirmed that the clipped lymph node was removed in the perioperative period. Importantly, no instances of clip migration were observed in any of the clipped lymph nodes.
Statistical analyses
Pathology results were considered the gold standard, and statistical analyses for axillary lymph node response by ultrasound were conducted using the SPSS version 25 software package.
The normality of the data distribution was assessed using the Kolmogorov–Smirnov test and Shapiro–Wilk test. Descriptive statistics were presented as mean ± standard deviation for normally distributed data, and as median (min–max) for non-normally distributed data. Categorical data were described using frequency and percentage. Statistical comparisons between groups were performed using the Kruskal–Wallis test and Mann–Whitney U test. Analysis of categorical data was conducted using the chi-square test, Fisher’s exact test, and Fisher–Freeman–Halton test. The Spearman rank correlation coefficient was used to examine the relationships between variables. The significance level for all statistical analyses was set at α = 0.05.
Results
The mean age of the 43 patients in this study was 52.42 years, with a range of 20 to 77 years (standard deviation: 12.866). Breast masses were more frequently located on the left side (54%), with the upper outer quadrant being the most common site (58%). Of the masses, 56% were single focal, while 44% were multifocal and multicentric. The most prevalent histological type was invasive ductal carcinoma (77%). Regarding molecular subtypes, 56% were luminal, 33% were HER2-positive, and 11% were triple-negative breast cancer (TNBC) (Table 1). Before NAC, 23 patients (54%) had 1 or 2 lymph nodes, while 20 patients (46%) had multiple pathological lymph nodes. The metastatic lymph node cortex thickness ranged from 3 to 17 mm, with a mean of 8 mm. Hilum loss was observed in 56% of these lymph nodes, and 54% exhibited a round shape (Table 1). The response of the breast mass to treatment after NAC was complete in 25 patients (58%) and partial in 18 patients. Among the complete responders, 40% were in the luminal disease group, while 60% were in the HER2-positive and triple-negative groups. Notably, 18 out of 19 patients in the HER2-positive and TNBC molecular subtypes group achieved a pathological complete response, with all of these patients also achieving a pathological complete response in the axilla.
After NAC, the mean axillary lymph node cortex thickness measured on US was 1.8 mm. Pathological evaluation revealed a mean of 1.7 metastatic lymph nodes, with the mean diameter of the metastatic focus being 6.3 mm.
In the post-NAC US evaluation, 30 patients showed no lymph nodes or clips with pathological morphology in the axilla. Among them, there was a 70% complete response and 30% partial response in the axilla (Table 2).
Before NAC, 23 patients had 1–2 metastatic lymph nodes, while 20 had 3 or more pathological lymph nodes. After NAC, TAD, and SLNB+/-non-sentinel lymph node (non-SLN), were performed on all patients, regardless of axillary treatment response on US. ALND was performed on 14 patients who had pathological axillary lymph nodes on the preoperative US evaluation, and metastases were detected in TAD and SLNB/non-SLN during frozen section analysis (Table 3). A specimen radiograph was taken during operation, confirming the removal of the clips. Patients who underwent ALND were those exhibiting a partial response. In the pathological evaluation of the clipped lymph nodes, a complete response was observed in 51% of cases, with 40% showing macrometastasis and 9% showing micrometastasis (Table 2).
Regarding the evaluation of axillary lymph node response with US, the sensitivity was 61.9%, specificity was 100%, positive predictive value (PPV) was 100%, negative predictive value (NPV) was 73.3%, false negative rate (FNR) was 38.1%, and accuracy was 81.4%.
There were eight patients whose US axillary responses resulted in false-negative results. Five of these patients had multiple pathological lymph nodes before treatment, and TAD, SLNB/non-SLN, and ALND were performed on them. Metastasis was observed in three of these patients during ALND, while in the other two, metastasis was found only in the lymph node removed by TAD.
In the remaining three patients with false-negative US responses, only one lymph node showed pathological morphology before treatment. In this removed lymph node, fibrotic changes secondary to treatment and a 0.3–0.4 cm metastatic focus were observed, so ALND was not performed (Table 4). Axillary radiotherapy was planned as complementary treatment for these patients.
In four patients with false-negative results, no pathological lymph nodes or clips were detected on axillary US, and these were evaluated as a complete response. However, micrometastasis was found in these lymph nodes in the pathology results, with metastatic focus diameters of 1–2 mm. In another four patients, the cortical thickness of the lymph nodes observed on ultrasound was less than 2 mm, with no suspicious morphological findings. In these four patients, macrometastasis was detected, with metastatic focus diameters of 7–8 mm. The inability to detect micrometastases is a known limitation of US.
In the statistical analysis; no significant relationship was observed among parameters such as patient age, lesion quadrant, lesion distribution, pathological subtype of the breast mass, pre-treatment pathological lymph node count, cortex thickness, and short axis/long axis ratio with the treatment response of the clipped node in US evaluation and pathology results (Table 1).
A significant association was identified between axillary pathological response and residual tumor size (p = 0.017). The cortical thickness of the clipped lymph node on post-treatment ultrasound, along with the short axis/long axis ratio of the lymph node, exhibited a correlation with the axillary response in pathology results (p < 0.0001). There was statistical concordance between the treatment response of the clipped lymph node on ultrasound and the lymph node response in pathology results (p < 0.0001). Significant differences were observed between the molecular subtype and axillary lymph node response both with US (p = 0.011) and pathology results (p = 0.0004) (Table 1). Significant relationships were noted between the treatment response of the breast mass and the axillary node response in both pathology result and US evaluations (p < 0.0001) (Table 5).
Discussion
Needle wire guided targeted axillary dissection appears to be a promising approach for minimal axillary surgery. In node-positive breast cancer patients who received NAC, TAD demonstrated an identification rates of 96%, with a reported FNR of 7%15.
In the study conducted by Lim et al., they achieved 100% accuracy in removing the clips by marking the skin with the Skin Mark Clipped Axillary Nodes Removal Technique (SMART) technique and using the UltraCor Twirl Marker11. In our study, the clips that responded completely to NAC and could not be visualized with US were marked with a needle wire under CT guidance, and all of them were successfully removed. There are a limited number of studies on this technique in the literature. In a study of five cases conducted by Trinh et al., it was reported that this method is easy and convenient for marking lymph nodes that cannot be detected by ultrasound after NAC17. In our study, axillary markers for 23 patients were placed under CT guidance. None of our patients developed complications related to the marking, and it provided a suitable guide for the surgeon during the operation. Limitations of this method include processing time and radiation exposure. Our total processing time with this method, including preparation, was approximately 10–15 min, which is similar to marking under ultrasound guidance. To minimize radiation exposure, performing low-dose CT scans and adjusting the processing area and section thickness can be considered as potential solutions.
In their study, Li and colleagues investigated the US parameters influencing the complete response in axillary lymph nodes after NAC4. They found no significant association with the lymph node short-axis ratio. Notably, they observed a 71% nodal complete response in patients receiving HER2-targeted therapy. Similarly, in our study, a nodal complete response was observed in all HER2-positive patients after treatment. Furthermore, akin to our findings, they identified a significant relationship between cortical thickness and axillary nodal complete response post-treatment4. In our study, no significant statistical differences were found between lymph node US findings before NAC and treatment response. We believe this may be related to the small number of patients and the retrospective nature of the image evaluation. However, a significant relationship was observed between the lymph node cortex thickness measured in the ultrasound performed after treatment and the diameter of the lymph node metastatic focus identified in pathology. Cortical thickness is one of the key parameters in the preoperative assessment. Hieken et al. found, in a study involving a group of 272 patients newly diagnosed with breast cancer, that MRI and PET-CT have limitations in evaluating axillary nodal response18. Compared to other imaging methods, US is a cost-effective, practical, noninvasive, and reliable approach for evaluating the axillary region.
In the study conducted by Pislar et al., it was noted that US, widely used in post-treatment axillary evaluation in patients with node-positive pre-treatment, led to overdiagnosis in 50% of cases. Sensitivity ranged from 37 to 100%, with specificity ranging from 69 to 92%. The evaluation of response with US emphasized cortical thickness, with a focus on perinodal involvement. Similarly, in our study, sensitivity was 61.9% and specificity was 100%. Pislar and colleagues also highlighted a connection between molecular subtypes and axillary response. In our study, axillary complete response rates were particularly high, especially in HER2-positive and TNBC patients, with assessments aligned with US findings19.
The primary limitations of this study include the small sample size and retrospective design. There are several studies in the literature that utilize artificial intelligence algorithms to predict the presence of metastasis in the axillary lymph node. For example, Zhou et al. conducted a study using axillary lymph node US images, while Sun et al. also focused on predicting axillary lymph node metastasis from US images of primary breast cancer20,21. Additionally, comparative studies involving larger patient cohorts, particularly those undergoing TAD, could assess alternative marking techniques. In the current era emphasizing minimal axillary surgery, pre-TAD evaluation of clipped lymph nodes with US holds significance in surgical planning. To obtain more detailed information on the impact of ultrasound evaluation before TAD on surgical therapeutic effectiveness, studies with long-term follow-up results should be conducted in a larger patient cohort.
Conclusion
The assessment of clipped lymph node treatment response with preoperative ultrasound, followed by excision using needle wire localization, can be used as a viable alternative method to axillary dissection with low false-negative rates.
Data availability
The data used in this study were entered into an Excel file and analyzed using the SPSS program. Information about the data used and/or analyzed during the current study can be obtained from the corresponding author upon reasonable request, to the extent permitted by the ethics committee of our university.
Abbreviations
- NAC:
-
Neoadjuvant chemotherapy
- SLNB:
-
Sentinel lymph node biopsy
- non-SLN:
-
Non-sentinel lymph node
- ALND:
-
Axillary lymph node dissection
- TAD:
-
Targeted axillary dissection
- FNR:
-
False negative rates
- US:
-
Ultrasound
- MG:
-
Mammography
- TNBC:
-
Triple negative breast cancer
References
Schulz-Wendtland, R. et al. Evaluation of a marker clip system in sonographically guided core needle biopsy for breast cancer localization before and after neoadjuvant chemotherapy. Geburtshilfe Frauenheilkunde 77, 169–175 (2017).
Boughey, J. C. et al. Tumor biology correlates with rates of breast-conserving surgery and pathologic complete response after neoadjuvant chemotherapy for breast cancer: Findings from the ACOSOG Z1071 (Alliance) prospective multicenter clinical trial. Ann. Surg. 260(4), 608–616 (2014).
Scheel, J. R. et al. MRI, clinical examination, and mammography for preoperative assessment of residual disease and pathologic complete response after neoadjuvant chemotherapy for breast cancer: ACRIN 6657 trial. Am. J. Roentgenol. 210(6), 1376–1385 (2018).
Li, Z., Tong, Y., Chen, X. & Shen, K. Accuracy of ultrasonographic changes during neoadjuvant chemotherapy to predict axillary lymph node response in clinical node-positive breast cancer patients. Front. Oncol. 12, 845823 (2022).
Oh, J. L., Nguyen, G. & Whitman, G. J. Placement of radiopaque clips for tumor localization in patients undergoing neoadjuvant chemotherapy and breast conservation therapy. Cancer 110, 2420–2427 (2007).
Dash, N., Chafin, S. H. & Johnson, R. R. Usefulness of tissue marker clips in patients undergoing neoadjuvant chemotherapy for breast cancer. Am. J. Roentgenol. 173, 911–917 (1999).
Flores-Funes, D. et al. Feasibility and validation of the targeted axillary dissection technique in the axillary staging of breast cancer after neoadjuvant therapy: Definitive results. Surg. Oncol. 38, 101636 (2021).
Banys-Paluchowski, M. et al. Axillary ultrasound for prediction of response to neoadjuvant therapy in the context of surgical strategies to axillary dissection in primary breast cancer: A systematic review of the current literature. Arch. Gynecol. Obstet. 301, 341–353 (2020).
Song, Y. X. et al. Diagnostic accuracy of de-escalated surgical procedure in axilla for node-positive breast cancer patients treated with neoadjuvant systemic therapy: A systematic review and meta-analysis. Cancer Med. 11(22), 4085–4103 (2022).
Dorsi, C., Sickles, E. A., Mendelson, E. B. & Morris, E. A. Breast Imaging Reporting and Data System: ACR BI-RADS Breast İmaging Atlas 5th edn. (American College of Radiology, 2013).
Lim, G. H. et al. Initial results of a novel technique of clipped node localization in breast cancer patients postneoadjuvant chemotherapy: Skin mark clipped axillary nodes removal technique (SMART trial). Cancer Med. 9(6), 1978–1985 (2020).
Kanesalingam, K. et al. Targeted axillary dissection after neoadjuvant systemic therapy in patients with node-positive breast cancer. ANZ J. Surg. 90(3), 332–338 (2020).
Munck, F. et al. Comparing methods for targeted axillary dissection in breast cancer patients: A nationwide, retrospective study. Ann. Surg. Oncol. 30(11), 6361–6369 (2023).
Aragón-Sánchez, S. et al. Feasibility of targeted axillary dissection for de-escalation of surgical treatment after neoadjuvant chemotherapy in breast cancer. Surg. Oncol. 44, 101823 (2022).
Siso, C. et al. Feasibility and safety of targeted axillary dissection guided by intraoperative ultrasound after neoadjuvant treatment. Eur. J. Surg. Oncol. 49(10), 106938 (2023).
Skarping, I., Förnvik, D., Zackrisson, S., Borgquist, S. & Rydén, L. Predicting pathological axillary lymph node status with ultrasound following neoadjuvant therapy for breast cancer. Breast Cancer Res. Treat. 189(1), 131–144 (2021).
Trinh, L. et al. CT-guided wire localization for ınvolved axillary lymph nodes after neo-adjuvant chemotherapy in patients with ınitially node-positive breast cancer. Breast J. 22(4), 390–396 (2016).
Hieken, T. J., Boughey, J. C., Jones, K. N., Shah, S. S. & Glazebrook, K. N. Imaging response and residual metastatic axillary lymph node disease after neoadjuvant chemotherapy for primary breast cancer. Ann. Surg. Oncol. 20(10), 3199–3204 (2013).
Pislar, N. et al. Axillary ultrasound for predicting response to neoadjuvant treatment in breast cancer patients—A single institution experience. World J. Surg. Oncol. 21(1), 292 (2023).
Zhou, L. Q. et al. Lymph node metastasis prediction from primary breast cancer US ımages using deep learning. Radiology 294(1), 19–28 (2020).
Sun, S. et al. Deep learning prediction of axillary lymph node status using ultrasound images. Comput. Biol. Med. 143, 105250 (2022).
Author information
Authors and Affiliations
Contributions
O.A. and A.O. contributed to the study conception and design. O.A., B.G., and G.S. contributed to the investigation, methodology, formal analysis, data curation, and validation. O.A. wrote the original draft and contributed to the review and editing. A.O. performed supervision and review. The first draft of the manuscript was written by O.A., and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Corresponding author: Ozge Aslan.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Aslan, O., Oktay, A., Goktepe, B. et al. Ultrasonographic accuracy in evaluating response of clipped lymph nodes in targeted axillary dissection in breast cancer. Sci Rep 15, 611 (2025). https://doi.org/10.1038/s41598-024-84827-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-84827-w