Abstract
This study considers the impact of commercial honey on human health and analyzes the levels of elements linked to different honey kinds. The study analyzed the levels of various elements in 13 different honey samples. The level ranges for the elements were as follows: Cd (0.002–0.186 ppm), Cr (0.053–0.107 ppm), Mn (0.071–0.622 ppm), Fe (6.973–9.820 ppm), Zn (2.044–6.883 ppm), Sr (0.172–1.63 ppm), and Pb (0.030–2.566 ppm). An analysis of variance (ANOVA) was performed utilizing the Tukey simultaneous test at a 95% confidence level. A homogeneity of variances test was performed using various comparison intervals for the standard deviation, with the alpha level of 0.05 for the elements Cd, Cr, Mn, Fe, Zn, Sr, and Pb. Tukey’s (95%) and Fisher’s pairwise comparison methods were employed. The statistical analysis was performed using Minitab Version 22.1 software, with a significance level of p < 0.05. Variations in the elements of honey samples were noted based on the producing process. This study aimed to safeguard human health by quantifying the element levels present in honey. The results of this study indicate that honey-producing commercial organizations have to carefully evaluate hive site selection.
Similar content being viewed by others
Introduction
Honey, recognized as a nutrient-rich material with antibacterial properties1,2,3, facilitates the treatment of various disorders by enhancing the immune system and providing energy1,4. Furthermore, honey has various medical advantages, having been employed for thousands of years in the areas of dermatological and gastrointestinal health3. In contaminated regions, agricultural pesticides and insecticides may contaminate honey with heavy metals. The accumulation of heavy metals induces severe health conditions in the body. This research analyzed the mineral and heavy metal content of honey concerning human health. This study also encompasses the assessment of elemental concentrations in honey and evaluates food safety concerning human health. The objective is to ascertain the elemental concentrations in commercial local honey products to demonstrate that human health remains unaffected.
The novelty of this study is situated within the comprehensive element analysis of honey from Aksaray province’s Hasan Mountain in the Cappadocia region, alongside commercial honey from the Cappadocia region and other provinces, with critical evaluations regarding health, underscoring its originality. This study serves as a tangible illustration of comparing the region-specific honey with other commercial varieties.
This study briefly covers beekeeping history. Honey has been used in food and medicinal since prehistory5. Honey, an ancient sweetener, has been used for centuries, including consumption, wound healing, and sale. Its presence in cave paintings in Spain demonstrates its long history for at least 10,000 years6. Ancient Egyptians and Middle Easterners used honey for sweetening cakes, burying dead, prolonging life, healing illnesses, and replacing gold taxes in the Roman Empire6. Beekeeping has been practiced since the Stone Age, with Middle Eastern countries being genetic hotspots, as evidenced by inscriptions in Anatolia from the Hittite period (1300 BC)7. Beekeeping in Turkey ranks second-most globally with 5 million colonies, after China, and fifth-most globally with 73,929 tons, with China leading with 41.77 kg per colony8.
Turkey ranks second globally, with Argentina, Ukraine, and the US following9. Turkey’s honey production increased nearly from 95 thousand tons in 2013 to 114 thousand tons in 2023 data from the Turk State Institute of Statistics (TUIK). As per the 2023 data, Turkey possesses 9,224,881 hives, with a total honey production of 114,886 tons10. Beekeeping in Turkey is a longstanding socio-economic activity7,11. The geographical and climatic characteristics of Turkey create an optimal setting for beekeeping and honey production11. Our country’s honey production is limited, and we rarely produce bee products beyond honey. Increase honey output, diversify bee production, and use honeybees in agriculture to maximize our potential.
The public and private sectors in Turkey have not institutionalized beekeeping, despite its output potential7. China and Argentina use less honey per capita than industrialized nations, Italy, France, and Great Britain. In the US, Canada, and Australia, average consumption is between 0.6 and 0.8 kg5.
Honey is a natural product made from plant nectars, plant components, or insect secretions and stored in the honeycomb, according to the Turkish Food Codex12. Honey comes from plant nectars, plant components, or plant-sucking insects, according to the Codex Alimentarius and the EU. Bees collect honey, modify it, reduce water content, store it in honeycombs, and mature it13. The Turkish Food Codex Honey Communique 2012/28,366 divides honeys by origin into floral and secretory honeys. Flower honey is made from plant nectar, while secretion honey is from plant tissues or insects12. Honeycomb honey is made from comb cells, squeezed honey, extra virgin honey, pressed honey, and filtered honey. Pure beeswax, combs, or bees can be used to make honeycomb honey, which can be infused, processed, boiled, or filtered to reduce pollen12.
Codex Alimentarius (2001) categorizes honeys into four types based on their origin: blossom honey, secretory honey, monofloral honey, and polyfloral honey13. Flower honey is derived from floral nectars, while secretory honey is made by bees collecting insects’ excretions. Bees in Central and Eastern Europe produce honey secretion, valued in Slovenia. Monofloral honey, derived from plants like clover, acacia, linden, and sunflower, is more expensive than polyfloral honey, which comes from multiple plant sources13. This study studied heavy metal components in commercial honey.
In recent years, environmental contamination has threatened human health. Heavy metals are poisonous and persistent, making them one of the most dangerous environmental pollutants. Numerous studies have shown that heavy metals affect honey14,15,16,17,18. Heavy metals are defined as metals with densities over 5 g/cm³19. A study on mitigating and preventing environmental contamination involves the utilization of biological indicators. Through pollutant accumulation, biological indicators reveal environmental contamination levels. Honey is a good biomonitoring tool for bee habitats20,21. Bees may go 4 km and cover 50 m2, interacting with air, soil, and water to produce honey with heavy metals. Quality control and nutritional value are important due to environmental degradation. Lead (Pb) is toxic at high metal concentrations, and some countries limit it to from 1 mg/kg to 0.215 mg/kg21.
Honey contains macro and trace elements needed for human nutrition. They can be dangerous if they exceed safety limits22. Honey is made from minerals that plants spontaneously absorb from the soil. Honey has 0.04–0.20% minerals. As the mineral concentration of honey escalates, its hue intensifies. Reports indicate the presence of 27 distinct components in honeys sourced from nine different countries23. This research seeks to alleviate the negative health effects of honey production while concurrently advancing environmental sustainability. This study aimed to safeguard human health by quantifying the amounts of elements in honey. This research improves food safety and encourages commercial honey producers to thoroughly evaluate the environmental impacts of hive site selection.
Materials and methods
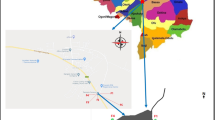
Physical, sensory, chemical, and microbiological aspects influence honey quality. The primary criterion for honey classification is its physical qualities, as they are quantifiable and yield valuable evaluation data24. This research gathered 13 samples of commercial honey. During spring, the markets in Aksaray city provided the commodities to the laboratory in their original packaging.
Materials
The study recommended polyethylene and polypropylene over glass and metal due to element contamination, pressure, and temperature resistance, as indicated in the CEM MARS 5Xpress microwave decode system guide. Specifically, polyethylene and polypropylene materials were employed for these experiments due to their resistance to the pressure and temperature conditions of the experimental apparatus. Moreover, this study incorporated an enhanced methodology for analyzing honey elements. The glassware and plastic apparatus employed were subjected to a 24-hour detergent soak, followed by a thorough rinse with tap water. The materials were then immersed in a 5% nitric acid solution for an additional 24 h. Finally, they were cleansed with ultra-pure water and oven-dried at 55 ˚C.
During the preparation of standards and samples during the study, all dilutions were carried out using ultrapure water. 65% Nitric acid (Merck 100456, Darmstadt, Germany) was used in the preparation of solutions, standards and samples. 30% Hydrogen peroxide (Merck 108600, Darmstadt, Germany) was used in the preparation of the samples. The experiment utilized high-purity argon gas (99.995% purity) from Linde AG. Additionally, a multi-element solution standard (High-Purity, ICP-MS-68B-A, 48 Component ICP-MS Standard at 100 µg/mL Solution A, North Charleston, USA) was employed as the commercial standard.
Furthermore, the elements chrome (Cr), cadmium (Cd), manganese (Mn), iron (Fe), Pb, strontium (Sr), and zinc (Zn) were analyzed using Inductively Coupled Plasma - Mass Spectrometry (ICP-MS). The ICP-MS apparatus (Thermo-Fisher Scientific Mass Spectrometer Model X Series II, Bremen, Germany) detected the elements Cr, Fe, Cd, Zn, Pb, Mn, and Sr in honey by introducing argon gas into a high-temperature plasma, which disrupted molecular bonds and ionized atoms.
Microwave defrosting system and conditions
The CEM MARS 5Xpress (Cem Corporation, Mars 5, U.S.A.) microwave system was utilized to conduct an efficient and effective thawing process. The Teflon tubes used in the microwave defrosting system were specifically the CEM MARS 5Xpress tubes with XP-1500 Plus Teflon vessel code. These tubes are designed to withstand high pressure (1500 psi) and temperature (240 ˚C) conditions.
Utilizing ICP-MS device
ICP-MS uses argon gas to dissolve molecular bonds and ionize atoms in a high-temperature plasma. The sample input system delivers the solution to the nebulizer and spray chamber. The sample becomes fog due to argon’s rapid velocity. Smaller droplets get to the argon plasma, while bigger ones are wasted. The plasma evaporates and ionizes the sample at temperatures reaching 10,000 K. Ions are introduced into the high vacuum environment from atmospheric pressure via sampler and skimmer cones. Ion lenses concentrate the ion stream on the quadrupole to direct it to the mass filter. Mass spectrometry analyzes ions’ mass-to-charge ratios to determine their amount.
Preparation of sample and thawing process
The study used microwave thawing equipment to prepare honey samples for analysis under different temperature, power, and time parameters utilizing the CEM MARS 5Xpress approach25. 1 g of honey samples was weighed in microwave device tubes and 9 ml of 65% HNO3 and 1 mL of 30% H2O2 were added. To avoid contamination, 9 ml of 65% HNO3 and 1 mL of 30% H2O2 were added to make a blank sample. Microwave dissolving dissolved all samples. Table 1 lists microwave defrosting system parameters. Table 1 shows the microwave defrosting technique using a 120 ˚C temperature for 20 min in the first step, 15 min in the second stage, and 20 min in the final stage. During temperature, the microwave runs at 1600 W.
Preparation of standards and calibration
Calibration used 1000 mg/L standards. The master stock standard solution was made. With this solution, intermediate stock solutions were made. Standards were made with 2% nitric acid. The primary stock standards are provided as percentages of water-acid solutions and based on 1000 ppb. Table 2 shows the calibration of the standards, including water-acid solution percentages and 1,000 ppb determinations.
Analysis of samples by ICP-MS
The appropriate operational parameters were determined for analysis first. Pre-prepared calibration standards were used for calibrations. The device was calibrated by entering the blank sample, then the standards, starting with the lowest concentration. Based on concentration-matched areas, calibration curves were created. Precision is used to build calibration curves. ICP-MS working settings for Cr, Fe, Cd, Zn, Pb, Mn, and Sr metal analysis are shown in Table 3. Under these conditions, Cr, Fe, Cd, Zn, Pb, Mn, and Sr were measured. The main elements were determined using an ICP-MS with the operating conditions in Table 3.
Statistical data analysis application
The rate of components in honey samples was measured using Microsoft Windows Excel 2021, Minitab (2024, Version 22.1), and SPSS Statistics for Windows, Version 17.0 (SPSS IBM Inc., North Castle, NY, USA). Studies with three or more replicates had their mean values statistically analyzed. Through statistical analysis of collected and created scenario data, this study proved its reliability and mathematical modeling. Cr, Fe, Cd, Zn, Pb, Mn, and Sr were measured in 13 samples. Each sample was triplicated, producing 273 measurements. ANOVA was used to measure Cr, Mn, Fe, Zn, Sr, Cd, and Pb. The honey sample study was statistically analyzed using SPSS (17.0) and Minitab (22.1). A statistical method was used to assess the experimental data’s quality, ensuring the study’s reliability and precision, which is essential for comparable research. ANOVA was performed using the Tukey simultaneous test at 95% confidence. The test for variance homogeneity used multiple standard deviation comparison intervals and 0.05 alpha. Minitab Version 22.1 used Tukey (95%) and Fisher pairwise comparisons. The statistical analysis utilized the ANOVA with a significance level of p < 0.05.
Results and discussions
This study summarizes a comprehensive heavy metal analysis of 13 commercial honey products. The analyses include statistical methods and result error margins. Table 4 shows standard deviation, standard error, mean element concentrations, and ranges for 13 Aksaray commercial honey samples. Minitab Version 22.1 was used for a one-way ANOVA with Tukey simultaneous (95%) test for equal variances (α = 0.05 for Cr, Mn, Fe, Sr, Cd, and Pb). Fisher pairwise comparison methods, utilizing a significance criterion of p < 0.05.
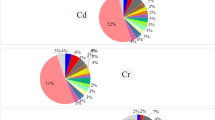
This study presents all aspects from Fig. 1. Cr, Mn, Fe, Zn, Sr, Cd, and Pb were calculated from honey sample element quantities (ppm) with standard error. Honey samples had different element levels. The findings were compared to research studies in the literature.
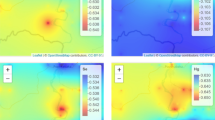
Figure 2 displays the experimentally determined element concentration in honey samples (ppm). The 6th commercial honey sample had the highest elements concentration, while the 5th sample had the lowest. The 2nd, 5th, and 7th honey samples had lower heavy metal levels than the 6th, 8th, and 9th. ICP-MS was also used to determine heavy metal concentration calibration curves.
Evaluation of the results of the elements
Honey was tested for Cr, Mn, Fe, Zn, Sr, Cd, and Pb, and the results were compared to literature and standards. The following metal concentrations in honey meals were assessed for human health.
In Fig. 3, Cr concentration was determined as 0.053–0.107 ppm. Among the samples, the lowest average Cr value was found in the Honey 7 sample with 0.053 ± 0.042 ppm. The highest Cr average value was determined in the Honey 9 sample with 0.107 ± 0.020 ppm. Cr concentration in honey is expressed as 0.0039–0.038 ppm11, 0.12–0.55 ppm26, 0.1–0.36 ppm27, 0.0938–2.6442 ppm28 and 0.18–0.24 [ppm]29 in other literature studies. The Cr concentration in the study was measured at 0.053–0.107 ppm. The findings were deemed consistent with prior research.
In Fig. 3, the Mn concentration was measured at 0.071–0.622 ppm. Among the honey samples, the Honey 9 sample exhibited the lowest mean manganese (Mn) value at 0.071 ± 0.003 ppm. The Honey 3 sample demonstrated the highest mean Mn value at 0.622 ± 0.04 ppm. In other literature studies on Mn concentration in honey, the values determined are as follows: 0.32–4.56 ppm11, 0.18–2.15 ppm26, 0.13–5.61 ppm27, 0.014–1.78 ppm28, 0.09–0.41 ppm29, 1.12–5.59 ppm30, and 0.11-4.00 ppm31. Mn is a prevalent element found on Earth. The European Union has established a daily consumption value of 300 mg32. The Mn concentration in this study ranges from 0.071 to 0.622 ppm, and the results align with the literature.
In Fig. 3, Fe concentration was determined as 6.973–9.820 ppm. Compared to the honey samples, the lowest Fe mean value was found in the Honey 5 sample with 6.973 ± 0.508 ppm. The highest Fe mean value was found in the Honey 6 sample with 9.820 ± 0.383 ppm. Other literature studies on Fe concentration in honey are as follows: 1.8–10.2 ppm11, 0.67–3.39 ppm26, 7.40-16.03 ppm27, 0.57–8.74 ppm28, 2.135–20.04 ppm30, 0.18–2.80 ppm31, 1.85–8.87 ppm33, and 0.40-52.51 ppm34. Metal elements like Fe are abundant on Earth. Normal human physiology needs it. Fe boosts protein and enzyme function in human health. Insufficient Fe intake limits cell oxygenation. The upshot is fatigue, poor work performance, and lowered immunity. The EU requires 15 mg of Fe daily. Some people need more Fe. Children under four, pregnant women, teens, and chronic blood loss patients need more Fe32. These values exceed the European Union Codex and Standards Regulation (2009)35 for Fe. However, Fe elements are within the FAO/WHO Codex Alimentarius Commission (2000)36 limits. Honey 6 had the greatest Fe levels, 6.973–9.820 ppm.
In Fig. 3, Zn concentration was determined as 2.044–6.883 ppm. Compared to honey samples, the lowest Zn mean value was found in Honey 12 sample with 2.044 ± 0.442 ppm. The highest Zn mean value was found in Honey 8 sample with 6.883 ± 0.013 ppm. In other related literature studies, the Zn concentration in honey is as follows: 1.1–12.7 ppm11, 0.20–2.46 ppm26, 1.25–15.73 ppm27, 1.11–5.35 ppm28, 0.16–2.96 ppm29, 1.49–6.41 ppm30, 0.50–1.10 ppm31, 0.16–4.48 ppm33, 0.18–19.10 ppm34, 0.28–11.60 ppm37, 2.2–11.00 ppm38, and 1.00-1.1039. Zn is essential to life. Cell structure and functional integrity depend on this property40. Zn also promotes healthy childhood, adolescent, and pregnant growth. This stimulates approximately 100 enzymes that aid metabolism. EU guidelines prescribe 15 mg daily. Honey 8 had the highest Zn levels, 2.044–6.883 ppm. The honey 8 sample exceeds the EU Codex and Standards Regulation (2009) Zn limits35, while other honey samples are within allowed limits. The FAO/WHO Codex Alimentarius Commission (2000) Zn limits are met36. An average 100 g of honey contains 2–5 ppm Zn, according to the FAO41. The FAO/WHO Codex Alimentarius Commission (2000)36 set the honey Zn limit at 10 ppm. Nectars and fruit juices can include up to 5 ppm of Zn under the EU Codex and Standards Regulation (2009)35. This experiment’s concentration data is similar. Comparing the Zn concentration (2.044–6.883 ppm) to the literature shows positive and below-average results.
In Fig. 3, the Sr concentration was measured at 0.172–1.63 ppm. In comparison to the honey samples, the Honey 2 sample exhibited the lowest mean value of Sr at 0.172 ± 0.08 ppm. The Honey 9 sample displayed the highest mean value at 1.63 ± 0.078 ppm. The concentration of Sr in honey varies from 0.20 to 1.25 ppm27, and from 0.03 to 1.45 ppm34 in other pertinent literature studies.
In Fig. 3, Cd concentration was obtained as 0.002–0.186 ppm in 13 different honey samples. According to the analysis results, the lowest Cd average value was found in the Honey 7 sample with 0.002 ± 0.025 ppm. The highest Cd average value was detected in the Honey 12 sample with 0.186 ± 0.006 ppm. Cd concentration in honey is as follows in other literature studies: 0.0009–0.0179 ppm11, 0.01–0.45 ppm26, 0.01–0.12 ppm27, 0.0905–0.3023 ppm28, 0.08–0.37 ppm29, 0.001–0.066 ppm30, 0.03–0.21 ppm31, 0.012–0.027 ppm37, 0.11–0.18 ppm38, and 0.007–0.021 ppm42. Honey 12 and Honey 6 samples had maximum Cd concentrations of 0.002–0.186 ppm. While Honey 6 and Honey 12 exceed the maximum Cd levels imposed by the European Union Codex and Standards Regulation (2009)35, other honey samples meet the limitations at 0.05 ppm.
In Fig. 3, Pb concentration was determined as 0.030–2.566 ppm. Compared to honey samples, the lowest Pb mean value was found in the Honey 7 sample with 0.030 ± 0.069 ppm. The highest Pb mean value was found in the Honey 12 sample with 2.566 ± 0.039 ppm. Other literature studies on Pb concentration in honey are as follows: 0.0084–0.106 ppm11, 0.003–0.04 ppm26, 0.06–2.02 ppm27, 0.0456-1.60 ppm28, 0.001–0.064 ppm29, 0.015–0.610 ppm30, 0.013–0.019 ppm31, 0.024–0.163 ppm37, 0.1–0.85 ppm38, and 0.062–0.541 ppm39. The FAO/WHO Codex Alimentarius Commission (2000) set the maximum Pb value in honey at 1 ppm. The European Union Codex and Standards Regulation (2009)35 limits Pb to 0.05 ppm in nectars and fruit juices. Pb is the metal that causes the most ecological damage from human activity. Pb levels in humans are estimated at 125–200 mg. Normal physiological systems excrete 1–2 mg of Pb. Most people only consume 300–400 mg daily. Modern human bones contain 500 to 1,000 times more Pb than ancient ones, according to bone studies. Persistent Pb exposure reduces sperm count and morphology. WHO classifies Pb as a Class 2 carcinogen43. Pb concentrations ranged from 0.030 to 2.566 ppm, with Honey 12 and Honey 6 samples having the highest amounts. Honey 7 and Honey 13 meet EU Codex and Standards Regulation (2009) Pb limits, but other honey samples surpass them35. Except for Honey 6 and Honey 12, all Pb samples meet FAO/WHO Codex Alimentarius Commission (2000) standards36.
Table 5 depicts honey values from 13 honey samples for each element, including those exceeding international organization limits (FAO/WHO/EUCodexs/FDA). These fundamental traits are considered components of acceptable daily intake (ADI).
The 2010–2020 honeybee pesticide and heavy metal research shows that environmental issues must be addressed to protect habitats and biodiversity44. Nanosensing, biosensing, electrochemical sensors, recognition molecules, and hybrid material methods must be improved to overcome this problem45,46.
The Honey 6, Honey 8 and Honey 12 samples had Pb, Cd, and Zn contents above the limits, but Cr, Mn, Fe, and Sr were within limits. The variation in values may be attributable to the proximity of the hives to contamination sources, but it demonstrates that honey producers should look at where to extract honey. In addition, this study focuses on the selection and conditions of honey bee hive locations, providing recommendations for improved site choices. The results indicate that beehives should be situated at a distance from populated areas, cultivated regions, major transportation routes, industrial zones, and other potential sources of environmental pollutants.
This study evaluated 13 commercial honey products, which were marketed in Aksaray province, situated in the Cappadocia region. Element levels were analyzed experimentally by obtaining 3 replicate samples from 13 commercial honey products. Subsequent research should concentrate on analyzing commercially available honey products across various local regions to gather data on the levels of elements of honey within the region under study. Honey, a widely consumed natural product, can be assessed from nutritional and toxicological perspectives.
Conclusions
This study validated that the several constituents of commercial honey confer health advantages. The element levels were assessed in 13 honey samples from Aksaray to evaluate their commercial quality. 13 commercial honey samples exhibited elements of Pb, Cd, and Zn exceeding permissible limits. The remaining elements satisfied the standards following the research. The health limit level of Honey 2 is the highest among commercially available honeys. The minimum quality criterion for commercial honey is Honey 6. The subsequent constituents in honey surpass the allowable thresholds set by regulations: Honey 8 Zn, Honey 6 Cd, Pb, and Honey 12 Cd and Pb. The closeness of honey-gathering colonies to sources of contamination accounts for the difference in value. In conclusion, apiarists should meticulously select hive locations for honey extraction. When situating beehives, it is essential to consider industrial zones, roadsides, closeness to major urban centers, and sources of agricultural contamination.
Pesticide-derived heavy metal ions increase pollution. Concerns about pollution in agricultural areas around beehives depend on insecticide choice. All these variables make honey production that doesn’t harm humans a major concern. This study suggests that commercial honey producers should carefully consider their production area. The study has major implications for environmental monitoring and honey production, encouraging honey producers to consider environmental issues in site selection.
This study is limited to 13 commercial honeys sold in the Cappadocia region. Future research may expand upon this investigation by evaluating the nutritional values and food safety of honeys consumed across a broader geographical area and employing more advanced analytical techniques. Ascertaining the element levels of other widely consumed foods, such as honey, is of significant importance in providing valuable insights for consumers in the future.
Data availability
“All data generated or analyzed during this study are included in the manuscript.”
Abbreviations
- BRR:
-
Best result rankings
- Cd:
-
Cadmium
- Cr:
-
Chrome
- cs:
-
Calibration of the standards
- FAO:
-
Food and agriculture organization
- Fe:
-
Ferro (iron)
- g:
-
Gram
- ICP-MS:
-
Inductively coupled plasma mass spectrometry
- L:
-
Liter
- min:
-
Minutes
- Mn:
-
Manganese
- Pb:
-
Lead
- ppb:
-
Parts per billion
- ppm:
-
Parts per million
- SPSS:
-
Statistical package for the social sciences
- Sr:
-
Strontium
- WHO:
-
World health organization
- WL:
-
Within limit
- \(\:T\) :
-
Temperature, [˚C]
- \(\:\dot{P}\) :
-
Power, [W]
- \(\:t\) :
-
Time, [min]
- \(\:\vartheta\:\) :
-
Standard volume, [μL, mL]
- \(\:\dot{m}\) :
-
Flow rate, [L/min]
- \(\:{n}_{p}\) :
-
Pump rate, [rpm]
- \(\:{\dot{P}}_{f}\) :
-
Forward power, [W]
- \(\:{\dot{P}}_{i}\) :
-
Ignition power, [W]
References
Molla, M. M. et al. Effect of honey and lemon juice on the physicochemical, nutritional, microbial and antioxidant properties of guava–pineapple jelly during storage periods. Discover Food. 2, 31. https://doi.org/10.1007/s44187-022-00033-5 (2022).
Basualdo, C., Sgroy, V., Finola, M. S. & Marioli, J. M. Comparison of the antibacterial activity of honey from different provenance against bacteria usually isolated from skin wounds. Vet. Microbiol. 124 (3–4), 375–381. https://doi.org/10.1016/j.vetmic.2007.04.039 (2007).
Mundo, M. A., Padilla-Zakour, O. I. & Worobo, R. W. Growth Inhibition of foodborne pathogens and food spoilage organisms by select Raw honeys. Int. J. Food Microbiol. 97 (1), 1–8. https://doi.org/10.1016/j.ijfoodmicro.2004.03.025 (2004).
Turkmen, N., Sari, F., Poyrazoglu, E. S. & Velioglu, Y. S. Effects of prolonged heating on antioxidant activity and colour of honey. Food Chem. 95 (4), 653–657. https://doi.org/10.1016/j.foodchem.2005.02.004 (2006).
Alvarez-Suarez, J. M., Tulipani, S., Romandini, S., Bertoli, E. & Battino, M. Contribution of honey in nutrition and human health: a review. Mediterranean J. Nutr. Metabolism. 3, 15–23. https://doi.org/10.1007/s12349-009-0051-6 (2010).
Nayik, G. A. et al. Honey: its history and religious significance: A review. Univers. J. Pharm. 03, 01, 5–8 (2014).
Kaya, F. Beekeeping structure and evaluation status in Agri Province. Ataturk Univ. J. Graduate School Social Sci. 12 (2), 35–55 (2008).
Kosoglu, M., Yucel, B., Saner, G. & Dogaroglu, M. Current situation analysis of beekeeping in Turkey. Hasad Hayvancilik J. 281, 52–61 (2008).
http://www.dunya.com/ekonomi (2020).
Turkish Statistical Institute (TUIK). Animal production statistical 2023. News Bull. 49681, (2024).
Tuzen, M., Silici, S., Mendil, D. & Soylak, M. Trace element levels in honeys from different regions of Turkey. Food Chem. 103, 325–330. https://doi.org/10.1016/j.foodchem.2006.07.053 (2007).
Republic of Turkish Official Gazette. Turk Codex Honey Declaration. (28366 repeating), 2–10. (2012).
Bradbear, N. Bees and their Role in Forest Live Hoods, non-wood Forest Products (Food and Agriculture Organization of the United Nations, 2009).
Grainger, M. N. C., Klaus, H., Hewitt, N., Gan, H. & French, A. D. Graphical discrimination of new Zealand honey from international honey using elemental analysis. Biol. Trace Elem. Res. 202, 754–764. https://doi.org/10.1007/s12011-023-03680-6 (2024).
Margaoan, R. et al. Environmental pollution effect on honey bees and their derived products: a comprehensive analysis. Environ. Sci. Pollut. Res. 2024 https://doi.org/10.1007/s11356-024-33754-4 (2024).
Knoll, S. & Cappai, M. G. Foraging activity of honey bees (Apis mellifera L., 1758) and exposure to cadmium: A review. Biol. Trace Elem. Res. 2024 https://doi.org/10.1007/s12011-024-04118-3 (2024).
Mustapha, S. et al. Honey as a sustainable indicator of heavy metals in tropical rainforest vegetation zone: an early warning monitoring approach. Int. J. Trop. Insect Sci. 43, 1263–1281. https://doi.org/10.1007/s42690-023-01038-y (2023).
Oroian, M. et al. Heavy metals profile in honey as a potential indicator of botanical and geographical origin. Int. J. Food Prop. 19 (8), 1825–1836. https://doi.org/10.1080/10942912.2015.1107578 (2016).
DuffusJ.H. Heavy metals a meaningless term? Pure Appl. Chem. 74 (5), 793–807. https://doi.org/10.1351/pac200274050793 (2002). IUPAC Technical Report.
Almedia-Silva, M. et al. Trace elements in wild and orchard honeys. Appl. Radiat. Isot. 69, 1592–1595. https://doi.org/10.1016/j.apradiso.2011.01.013 (2011).
Buldini, P. L., Cavalli, S., Mevoli, A. & Sharma, J. L. Ion chromatographic and voltammetric determination of heavy and transition metals in honey. Food Chem. 73 (4), 487–495. https://doi.org/10.1016/s0308-8146(01)00132-7 (2001).
Leblebici, Z. & Aksoy, A. Determination of heavy metals in honey samples from central Anatolia using plasma optical emission spectrofotometry (ICP-OES). Pol. J. Environ. Stud. 17 (4), 549–555 (2008).
Moniruzzaman, M., Chowdhury, M. A. Z., Abdur Rahman, M., Sulaiman, S. A. & Gan, S. H. Determination of mineral, trace element, and pesticide levels in honey samples originating from different regions of Malaysia compared to Manuka honey. BioMed. Res. Int. ID. 359890, 1–10. https://doi.org/10.1155/2014/359890 (2014).
Pridal, A. & Vorlova, L. Honey and its physical parameters. Czech J. Anim. Sci. 47 (10), 439–444 (2002).
Madejczyk, M. & Baralkiewicz, D. Characterization of Polish rape and honeydew honey according to their mineral contents using ICP-MS and F-AAS/AES. Anal. Chim. Acta. 617 (1–2), 11–17. https://doi.org/10.1016/j.aca.2008.01.038 (2008).
Vanhanen, L. P., Emmertz, A. & Savage, G. P. Mineral analysis of mono-floral new Zealand honey. Food Chem. 128, 236–240. https://doi.org/10.1016/j.foodchem.2011.02.064 (2011).
Yücel, Y. & Sultanoğlu, P. Characterization of Hatay honeys according to their multi-element analysis using ICP-OES combined with chemometrics. Food Chem. 140, 231–237. https://doi.org/10.1016/j.foodchem.2013.02.046 (2013).
Leblebici, Z. Determination of heavy metal pollution in some honey samples from Kayseri province, MSc Thesis, Erciyes University, Kayseri. (2006).
Flamminii, F., Consalvo, A., Cichelli, A. & Chiaudani, A. Assessing mineral content and heavy metal exposure in Abruzzo honey and bee pollen from different anthropic areas. Foods 13, 1930. https://doi.org/10.3390/foods13121930 (2024).
Gulec, M. Determination of the metal contents in honey samples, collected from some of the cities of East-Anatolian region in Turkey, Doctorate Thesis, Ankara University, Ankara. (2007).
Mara, A. et al. Elemental fingerprinting combined with machine learning techniques as a powerful tool for geographical discrimination of honeys from nearby regions. Foods 13 (2), 243. https://doi.org/10.3390/foods13020243 (2024).
https://food.ec.europa.eu. (2024).
Hameed, O. M., Shaker, O. M., Ben Slima, A. & Makni, M. Biochemical profiling and physicochemical and biological valorization of Iraqi honey: A comprehensive analysis. Molecules 29, 671. https://doi.org/10.3390/molecules29030671 (2024).
Hernandez, O. M., Fraga, J. M. G., Jimenez, A. I., Jimenez, F. & Arias, J. J. Characterization of honey from the Canary Islands: determination of the mineral content by atomic absorption spectrophotometry. Food Chem. 93, 449–458. https://doi.org/10.1016/j.foodchem.2004.10.036 (2005).
Anonymous, A. I. J. N. & European Fruit Juice Association., Regional Workshop On Fruit and Vegetable Processing in The EU, Practical consequences of implementation of EU legislation and Codex standards applicable to juices, Serbia, (2009).
Anonymous & Codex Alimentarius Commission Joint FAO/WHO. Proposed Draft Revised Regional Standard for Vinegar (Food Standards Programme Recommended European, 2000).
Vincevica-Gaile, Z., Klavins, M., Rudovica, V. & Viksna, A. Potentially toxic metals in honey from Latvia: Is there connection with botanical origin?. Recent Res. Environ. Energy Syst. Sustain. 158–163. (2012).
Demirezen, D. & Aksoy, A. Determination of heavy metals in bee honey using by inductively coupled plasma optical emission spectrometry (ICP-OES), G.U. J. Sci. 18 (4), 569–575 (2005).
Ragab, M. A. A., El-Yazbi, A. F. & El-Hawiet, A. Fast economic electrochemical assay for vitamins and heavy mineral components in honey samples of different botanical origin. Microchem. J. 155, 104770. https://doi.org/10.1016/j.microc.2020.104770 (2020).
Belgemen, T. & Akar The genes involved in zinc metabolism. J. Ankara Univ. Fac. Med., 57, 3, 161–166. (2004).
Chudzinska, M. & Baralkiewicz, D. Estimation of honey authenticity by multielements characteristics using inductively coupled plasma-mass spectrometry (ICP-MS) combined with chemometrics. Food Chem. Toxicol. 48, 284–290. https://doi.org/10.1016/j.fct.2009.10.011 (2010).
Kahvecioglu, O., Kartal, G., Guven, A. & Timur, S. Environmental effects of metals-I. J. Metall. 136, 47–53 (2003). [in Turkish].
Hrynko, I., Kaczyński, P. & Lozowick, B. A global study of pesticides in bees: quechers as a sample Preparation methodology for their analysis – Critical review and perspective. Sci. Total Environ. 792, 148385. https://doi.org/10.1016/j.scitotenv.2021.148385 (2021).
Báez, D. F. et al. Graphene-based sensors for small molecule determination in real samples. Microchem. J. 167, 106303. https://doi.org/10.1016/j.microc.2021.106303 (2021).
Manoj, D., Shanmugasundaram, S. & Anandharamakrishnan, C. Nanosensing and nanobiosensing: concepts, methods, and applications for quality evaluation of liquid foods. Food Control. 126, 108017. https://doi.org/10.1016/j.foodcont.2021.108017 (2021).
Acknowledgements
This study is funded by Aksaray University Scientific Research Projects Agency (Funding P. No BAP2015-062).
Funding
This research was supported by the following research projects: BAP 2015-062, funded by Aksaray University Scientific Research Projects Agency in Turkey.
Author information
Authors and Affiliations
Contributions
OOT and MA wrote the manuscript; they took an active part in experimental research. OOT and MA checked the manuscript and made statistical adjustments. OOT determined the elements.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Taner, O.O., Ardic, M. Comparing cappadocia honey samples for elements level. Sci Rep 15, 33767 (2025). https://doi.org/10.1038/s41598-025-00247-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-00247-4