Abstract
The present study aimed to investigate the physiochemical responses of Thymus vulgaris L. to salinity stress and was arranged in a two-factorial experiment based on a completely randomized design (CRD) with three replications under floating hydroponic conditions. The effect of salinity stress at three levels of 0, 100, and 200 mM NaCl for seven different time courses of 0, 6, 12, 24, 48, 72, 96, and 120 h on 3 months garden thyme plant (4 months after seed cultivation and 3 months after transplanting in hydroponic system) was investigated. The results showed that different concentrations of NaCl and different times of salinity stress had no significant effect on the fresh mass, while photosynthetic pigment content and DPPH (2,2-Diphenyl-1-picrylhydrazyl) radical scavenging activity were significantly decreased under high concentrations of sodium chloride and 72–120 h after salt stresses. In addition, the increase of salinity stress (100 and 200 mM) in 72–120 h treatment significantly increased compatible osmolytes, such as proline content. On the other hand, the activity of non-enzymatic antioxidants, such as phenolics and flavonoids under salt stress treatment with concentrations of 100 and 200 mM sodium chloride in the floating hydroponic system during 72–120 h showed significant increases. In general, it can be concluded that high concentrations of sodium chloride in floating hydroponic systems can cause significant adverse effects on the physical and biochemical properties of the garden thyme plant.
Similar content being viewed by others
Introduction
Salinity is one of the major abiotic stresses that affect plant growth and productivity1. Approximately 1125 million hectares of land around the world are affected by salinity stress2. The morphological, physiological, biochemical, and metabolic characteristics of plants are affected by salt stress3,4,5. Additionally, salt stress leads to oxidative stress through increasing reactive oxygen species (ROS), such as hydrogen peroxide (H2O2), superoxide (O2·−), and hydroxyl radicals (OH−)6,7,8,9.
In various ways, plants can protect the defense barriers against salinity stress from the destruction of cells and tissues, including morphological, physiological, biochemical, and molecular responses10,11. In addition, it has been determined that enzymatic and non-enzymatic antioxidants are made against the excessive production of ROS caused by the accumulation of salt around the root and inside the plant structure, and on the other hand, membrane lipid peroxidation can be the cause of salinity tolerance in plants12. Antioxidants such as phenolic compounds play a role in ROS scavengers under salt stress13. One of the other defense ways of plants against biotic and abiotic stresses is the release of volatile organic molecules14. Researchers revealed that high concentrations of salt stress lead to the accumulation of proline, total soluble sugar, and total soluble protein. Also, under NaCl stress, the levels of antioxidants, including phenolics and flavonoids increase15.
The Thymus genus is mainly found in the Mediterranean, Asia, Southern Europe, and North Africa. A wide difference in the essential oil composition and morphological characteristics of different thyme ecotypes has been observed, but the most important feature of all these ecotypes is their moderate smell and sometimes balsamic and spicy taste16. The economic importance of this plant is increasing in North America, Europe, and North Africa17,18,19. Garden thyme (Thymus vulgaris L.) is one of the most important members of the Lamiaceae and is widely spread in different regions of the Mediterranean and Asia. It has been widely used medicinally since ancient times20. Garden thyme, like other medicinal plants, is used in the chemical, cosmetic, food, perfumery, and pharmaceutical industries because it contains aromatic substances and essential oils. In addition, thyme essential oil is recognized as one of the top ten most widely utilized essential oils globally for food preservation purposes16. Also, large companies producing herbal medicines have realized that due to their anti-inflammatory and expectorant properties, they widely produce thyme syrups along with honey, ginger, etc. There are also many reports that these herbal medicines are used to treat coughs caused by viral infections, such as COVID-1921,22 and SARS-CoV-223, which have spread in recent years.
Salt stress negatively affects the growth, physiological, biochemical, and essential characteristics of medicinal plants, especially garden thyme. Previous studies showed that the high salinity of the nutrient solution decreased photosynthetic pigments in different plants24,25,26,27,28. Also, an increase in EC and pH of the nutrient solution decreased growth characteristics, relative water content, and the photosynthetic apparatus by disrupting chlorophyll metabolism, inducing oxidative stress, and prompting plastid degradation in different plants29,30,31,32,33,34,35,36,37,38.
Despite its economic and medicinal importance, the physiological and biochemical responses of T. vulgaris to salinity stress in hydroponic systems remain underexplored. Thus, the present study aimed to investigate the physiological and biochemical actions of garden thyme under salt stress at different time crouse. Ultimately, it will be determined whether the timing of salt stress application in a hydroponic system has an impact on the physiological and biochemical traits of garden thyme.
Materials and methods
Plant material, growth conditions, and salt treatments
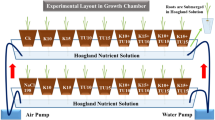
The experiment was conducted in the growth chamber and research laboratories of medicinal plants and the Horticulture Department of Arak University, Iran in 2024. Seeds of garden thyme obtained from Pharmasaat Co. of Germany were planted in a seed tray filled with perlite medium. After 1 month, the seedlings were planted in small plastic pots with perlite medium. These small pots were placed in the holes of the floating systems. After transferring the plants to the floating systems, the nutrient solution of Hoagland and Arnon’s39 (EC: 2.5 ds m−1, pH: 7) was used. The nutrient solution of Hoagland and Arnon’s contained: 5 mM KNO3, 5 mM Ca(NO3)2, 2 mM MgSO4, 1 mM KH2PO4, 7 μM MnCl2, 0.7 μM ZnSO4, 0.8 μM CuSO4, 0.8 μM Na2MoO4, 25 μM Fe-EDDHA, and 2 μM H3BO3. The nutrient solution (pH 6.7 ± 0.1, EC 2.1) was renewed every week. Deionized water was used for nutrient preparation. The floating system consisted of 63 square plastic containers measuring 25 × 30 × 30 cm3, each floating on a styrofoam measuring 25 × 30 × 30 cm3, and four plants planted in each container. All culture containers were connected to the air pump (HAILA, model: ACO-388 D) through holes and the nutrient solution was continuously aerated. After transplanting, the effect of salt stress of NaCl (Merck Co., Germany) in three levels (0, 100, and 200 mM NaCl) and eight time-course of salt stress (0, 6, 12, 24, 48, 72, 96, and 120 h) were studied on garden thyme based on prior studies. Acetic acid (CH3COOH) and sodium hydroxide (NaOH) were used to control and adjust the pH of the nutrient solution.
Growth chamber condition was set at a temperature of 25/15 °C (day/night) and a light condition of 16/8 h (day/night) with same photosynthetically active radiation (PAR) 250 ± 10 μmol m−2 s−1 and humidity 55–65% (Fig. 1). The EC and pH were monitored with portable pH meter (Fisherbrand™ accumet™ AP115 Portable pH Meter Kit) and portable EC meter (HI9033 EC Meter, Setare Arsh Aria company), respectively, daily during the cultivation process. After 3 months of transplanting the seedlings to the hydroponic system, the physiological and biochemical properties of garden thyme were measured. The formal identification of the specimens was performed by Dr. Hossein Salehi-Arjmand. A herbarium voucher specimen with sediment number TV-3321 has been donated to the public available herbarium of the Faculty of Agriculture and Natural Resources of Arak University, Iran.
Measurement of physiological and biochemical properties
Photosynthetic pigments
For the measurement of photosynthetic pigments, the method of Porra40 was used. In this method, the random sampling of 0.25 g of leaf mass of three mature leaves of each plant and three replications were selected and then extracted with 80% acetone and centrifuged at 350 rpm for 10 min. The extraction of chlorophyll from the selected leaves was read using a spectrophotometer (model T80 UV/VIS PG Instruments Ltd) at wavelengths of 646.6 and 663.6 nm. Lichtenthaler41 method was used to assay of total carotenoid at the wavelength of 470 nm.
OD: read absorbance V: the volume of acetone used W: sample mass (g).
The relative water content
To measure the relative water content of the leaves, the method of González and González-Vilar42 was used. For this purpose, after applying the salt stress, the youngest fully developed leaves were randomly sampled from the point of growth of the plants. After sampling, the fresh weight of the leaves was immediately measured with a digital scale with an accuracy of 0.0001. Then, the leaves were immersed in distilled water for 24 h at room temperature and low light to calculate the saturation weight. After this period, the samples were quickly and accurately measured with dry paper towels and their saturated weight. Then, the leaves were dried in an oven at 75 °C for 48 h and weighed again, and the relative water content of the leaves was calculated through the following equation:
where FW: fresh weight, DY: dry weight, and TW: turgid weight.
Electrolyte leakage
For the measurement of electrolyte leakage, the method of Shi et al.43 was used. In this method, from the leaves of the plants under stress treatment, 10 equal-sized square pieces were prepared separately from the young fully expanded leaves from three plants per three replicates and were placed in 50 mL glass vials, to remove electrolytes released during leaf disc excision rinsed with distilled water. Vials were then filled with 40 mL of distilled water and allowed to stand in the dark for 24 h at room temperature. The electrical conductivity (EC1) of the bathing solution was determined at the end of the incubation period. Vials were heated in a temperature-controlled water bath at 95 °C for 20 min and then cooled to room temperature and the electrical conductivity (EC2) was again measured.
where EC1: primary electrical conductivity and EC2: second electrical conductivity.
Proline content
The method of Paquin and Lechasseur44 was used to measure proline content with ninhydrin reagent. For this measurement, 0.50 g of fully developed leaves were homogenized in 5 mL of 95% ethanol in a Chinese mortar and then the resulting solution was transferred into a Falcon tube. After that, the prepared solution was centrifuged (Elektromag M 4815 PR) at 3500 rpm for 10 min. Then, for the determination of proline at a wavelength of 515 nm, a spectrophotometer (Thermomac UVS901) device was used. Standards were prepared with l-proline at: 0, 31.25, 62.5, 125, 250, and 500 (mg L−1) concentrations (y = 0.0196x + 0.0211; R2 = 0.9804).
Total phenolic compounds
To measure the total phenolic compound content, the method of Singleton and Rossi45 was used. The basis of work in this method is the reduction of Folin reagent by phenolic compounds in an alkaline environment and the creation of a blue complex that shows the maximum absorption at the wavelength of 765 nm with a spectrophotometer at a wavelength of 765 nm. The standard curve was prepared with gallic acid (y = 0.0256x + 0.085, R2 = 0.996).
Total flavonoid content
The method of Atanassova et al.46 was used to determine total flavonoid content. Aluminum chloride (AlCl3) was used for extraction, and quercetin was chosen as a standard. Briefly, to measure the total flavonoid content, 1 mg of the crude extract was diluted with 4 mL of water in a 10 mL volumetric flask. After that, 5% sodium nitrate (NaNO3) solution (0.3 mL) and then 10% aluminum chloride (0.3 mL), and 1.0 M NaOH (2 mL) were added to the flask. Then, 2.4 mL of water was added to the flask and mixed with high precision. All test steps were repeated three times for each sample. Finally, the absorbance of the mixture was read at a wavelength of 533 nm. Total flavonoid content values were determined as quercetin equivalent (QE) mg/g extract weight. Quercetin was used to draw the standard curve (y = 0.0194x + 0.0007, R2 = 0.993).
DPPH (2,2-Diphenyl-1-picrylhydrazyl) radical scavenging activity
The method of Barros et al.47 was used to measure radical scavenging activity in the plant tissue extracts. The absorbance at 517 nm was selected to determine the reduction of DPPH radicals. The radical scavenging activity was calculated as a percentage of DPPH discoloration using the following equation:
where A0 is the absorbance of the DPPH solution and A1 is the absorbance of the sample.
Statistical analysis
A two-factorial experiment based on a completely randomized design (CRD) with three replications was arranged for this study. The SAS software version 9.4 was used to analyze the obtained data (SAS Institute, Cary, NC, USA). To test the statistical analysis of data, a two-way ANOVA model was used. Also, to determine the specific differences between group means, Duncan’s multiple-range test was used as a post hoc test. Furthermore, differences were considered significant at a 0.05 probability level. Multivariate analysis of variance was performed using XLSTAT software (Addinsoft, New York, USA).
Results and discussion
Photosynthetic pigments
The results showed that the increase in salinity stress in garden thyme plants decreased photosynthetic pigment content. The concentration of 100 mM sodium chloride at 96 and 120 h significantly decreased photosynthetic pigments (Table 1). On the other hand, the results showed that the more the intensity of salinity stress and the duration of salinity stress increase, the less amount of chlorophyll remains in garden thyme plants and this can be caused by the destruction and loss of photosynthetic pigments and receptors. The reduction of light pigments decreases the production of carbohydrates in plants (Table 1). The maximum damage of photosynthetic pigments occurred under 200 mM sodium chloride and at 96 and 120 h of salt stress. Total chlorophyll content was also affected by varying levels of salinity stress and application times. When the salinity stress increased up to 100 mM compared with the control level, the plants had less decrease in chlorophyll content (Table 1). The results of this experiment showed that salinity stress with an intensity of 100 mM and more can cause a significant decrease in the carotenoid content, so treating with 100 and 200 mM sodium chloride concentrations for 96 and 72 h can generally cause a significant decrease in the carotenoid content and reduce photosynthesis in these plants. On the other hand, destroying the photosynthetic apparatus provides the basis for the production of reactive oxygen species, which are responsible for the deterioration of plant structures (Table 1). According to the researchers’ findings, the decrease in the chlorophyll content under salinity stress and the increase in the duration of the stress in hydroponic conditions is mainly due to the decrease of aminolevulinic acid synthase (ALA), an enzyme that catalyzes the synthesis of Daminolevulinic acid as the first common precursor in the biosynthesis of chlorophylls48. Also, scientists believe that in the early days of salinity stress, the contribution of chlorophyllase in chlorophyll degradation is moderate, but with the increase of salinity stress, other complementary pathways of chlorophyll degradation should be considered49. In this research, it was also found that increasing the salinity stress to 200 mM sodium chloride and applying the salinity stress for more than 48 h in the hydroponic environment caused a significant decrease in the content of photosynthetic pigments, which can be related to the destruction of chlorophylls with chlorophyllase enzyme. Salt stress can trigger ultrastructural changes, but in general, the main purpose of these changes is the destruction of chloroplast49. Gururani et al.50 found that salinity stress potentially affects the photosynthetic electron transport chain, resulting in increased production of reactive oxygen species (ROS) in cells. Under these conditions, oxidative light damage and light inhibition are created in plants. In the current research, it was also found that the increase in salinity stress (200 mM sodium chloride) and the duration of applying more stress (more than 48 h) caused a decrease in the content of photosynthetic pigments, which can be related to the destruction of the photosynthetic electron transport chain. Researchers believe that under severe salinity stress conditions, stomatal and non-stomatal limitations lead to a reduction in photosynthetic rate51. It has been reported that the increase in salinity stress in T. vulgaris and T. daenensis33 and various plant species of the Lamiaceae family (Mentha spicata L., Origanum dictamnus L., Origanum onites L.)28, caused a decrease in the content of photosynthetic pigments, which is consistent with the results of the present study. The decrease in chlorophyll content under salinity stress may be the result of ion absorption disorders with a simultaneous decrease in photosynthesis and respiration of medicinal plants28.
The relative water content (RWC)
The results showed that the relative water content of leaves was influenced by different concentrations of salinity stress and the duration of stress in hydroponic conditions. The high concentration of sodium chloride in the hydroponic environment and the increase in the stress time caused a significant decrease in the relative water content of the leaves so the use of a concentration of 100 mM sodium chloride for 96 and 120 h at a concentration of 200 mM sodium chloride caused a decrease in leaf relative water content. The reasons for this decrease can be the destruction of the roots and the inability to absorb and transfer water and nutrients to the aerial parts of the plants. Decreasing in relative water content causes the leaves to lose their moisture, and in this way, the leaves become dry, then, the absorption of light in the plants is also reduced, and the plants would be destroyed in the case of more treatment under stressful conditions (Table 1). Also, results showed that leaf RWC was lower in plants under high concentrations of sodium chloride (200 mM and severe salt stress) and longer exposure times to salinity stress (more than 48 h) than in control plants (treated with distilled water). The results of this study were consistent with the findings of Dalton et al.52. These researchers found that the sharp decreases in RWC could be due to the high concentration of Na+ in plants and on the other hand delaying the osmotic adjustment of the tolerance threshold of garden thyme plants. On the other hand, it seems that the relative content of leaf water is an important and efficient indicator for the water status and the adaptive trait of thyme plants to the surrounding environment, since it was related to a preadjustment in some plants tolerant to NaCl implementation53. Also, Emami Bistgani et al.33 found that exposure to 90 mM NaCl caused a significant decrease in the relative water content of leaves of T. vulgaris and T. daenensis, and in this way, damage to plant structures and osmotic stress occurs. In another study, it was found that the increase in salinity stress decreased the relative water content of the leaves of the rocket30. Since there is a correlation between the relative content of leaf water and the osmotic potential30, it can be stated that the osmotic potential in the conditions of salinity stress is considered a factor of osmotic adaptation against stress and also one of the defense strategies against abiotic stress, such as salinity and drought stress54. Under salt stress conditions, osmotic regulation involves net accumulation in a cell in response to stress conditions. In this condition, the osmotic potential decreases, but the water inside the cells is absorbed to maintain the turgor of the cells under stress conditions55.
Electrical leakage
Electrical leakage was affected by different concentrations of salinity stress and stress application times (Table 2). It was also found that increasing the duration of salinity stress in the concentration of 100 mM sodium chloride caused an increase in electrical leakage from the leaves of the treated plants. Also, it was found that the use of severe stress of 200 mM sodium chloride caused more than 60% of electrical leakage after 24 h of applying stress to 120 h. High electrical leakage causes the destruction of plants because it causes the cell membrane to rupture, and the performance of the plants decreases if stress duration increases, the plants will dry and die. Researchers believe that high electrical leakage may be due to the accumulation of certain ions in the salt stress conditions, which causes nutritional imbalance in plant tissues and structures. For instance, Na+ has potentially toxic effects on membrane permeability because this element can be distributed within cells and between different tissues and organs, thereby increasing EL56. Increasing the Na+ in plants causes more severe damage to lipid peroxidation and membrane damage57. In addition, researchers believe that the increase in salinity stress levels in many plants causes an increase in the levels of cellular free radicals, which is largely related to damage to the membrane caused by electrical leakage58. Emami Bistgani et al.33 reported that the increase in salinity stress caused an increase in EL in T. vulgaris and T. daenensis, which is consistent with the results of the present research due to the salinity stress and the increase in the duration of the stress. High salt stress disrupts the electron transport chain, which leads to oxidative damage in plants. Excess energy produced during electrochemical reactions can be dissipated through the Mehler reaction, leading to excessive production of ROS (such as H2O2). H2O2 and EL are the damage markers associated with a series of free radical production reactions that can damage cellular structures and macromolecules, unbalance cellular redox potential, and reduce membrane fluidity, leading to electrolyte leakage and It dries quickly. Membranes are the most sensitive to oxidative stress and are a suitable indicator of stress tolerance59.
Proline content
Proline content was affected by experimental factors (Table 2). The results showed that increasing the concentration of sodium chloride at different times of stress caused an increase in the proline content. Increasing the stress period for more than 24 h in hydroponic conditions caused a significant increase in the proline content. The 96 and 120 h after salt stress led to the highest proline content in the treated plants. The increase of compatible osmolytes, such as proline in stressful conditions makes plants able to withstand stress and suffer less damage. On the other hand, these osmolytes reduce the production of reactive oxygen species in plants, and with the increase in salinity stress, these compatible osmolytes also increase to prevent damage to plants from harmful agents59. Researchers found that proline accumulation is related to salinity tolerance in plants, and the reason for this is the efficiency of these compounds in reducing oxidative stress60. In addition, proline has other different roles in plants, which can reduce leaf water potential, increase water absorption, and reduce transpiration to maintain cell turgor pressure61. With these interpretations, proline accumulation at higher salinity levels is different depending on the plant species34. Azeem et al.59 found that the increase in salinity stress increased the content of proline in various plant species of the Lamiaceae family (Mentha spicata L., Origanum dictamnus L., and Origanum onites L.)28, which is consistent with the results of the present investigation. Organic osmolyte accumulation effectively helped osmotic regulation, maintaining rigidity, and protecting cellular metabolism from salt toxicity62. As reported in different plant species, proline accumulation and soluble sugar content indicate their active involvement in osmotic regulation and salt tolerance of Moringa48,63. These compounds protect and stabilize enzymes and proteins, reduce oxidation of lipid bilayers, act as free radical absorbers and cellular redox balancers, provide sites for carbon and nitrogen storage, and play a role in regulating cytosolic pH64. Moreover, these compounds participate in stress signaling and modulating gene expression under stressful conditions65.
Phenol and flavonoid contents
The phenol content was affected by experimental factors. The increase in sodium chloride concentration at different times of application of salinity stress caused an increase in phenolic compounds (Table 2). Also, the maximum phenol content was observed under the treatment of 100 mM sodium chloride concentration and 72–120 h after salt stress. The content of phenolic compounds in the concentration of 200 mM was lower than the concentration of 100 mM sodium chloride. The results also showed that at the level of 200 mM salt stress and from 48 to 120 h after salt stress, the content of phenolic compounds increased significantly. The increase of phenolic compounds is a non-enzymatic antioxidant barrier against damage caused by reactive oxygen species and prevents cell destruction against damage caused by salt stress.
Different concentrations of sodium chloride and different times of salinity stress had a significant effect on the flavonoid content (Table 2). Increasing sodium chloride concentration caused an increase in flavonoid content. The application of salinity stress with concentrations of 100 and 200 mM sodium chloride after 72 h affected the biochemical characteristics of the plants, including phenolic compounds. Applying 100 and 200 mM salt stress after 72–120 h increased the content of phenolic compounds in garden thyme. The flavonoid content under 100 mM sodium was higher than the concentration of 200 mM sodium chloride. The increase in salinity stress decreases the tolerance of plants to stress, and in this way, fewer contents of antioxidant compounds are synthesized in plants, in case of an increase of more than 200 mM, the plant will face severe stress and eventually, it will turn into the death of the plant. The researchers reported that the increase in salinity stress caused an increase in phenolics and flavonoids in T. vulgaris and T. daenensis plants33,66, which agreed with the results of the present study. Moderate salinity stress through increasing the content of total phenolic compounds can cause normal tolerance to salinity in different plants67. It has been found that salt stress caused an increase in phenolics and flavonoids of red pepper68. Also, Awika and Rooney69 concluded that the amount of production of these compounds in plants depends on genetics and environment. Phenolics and flavonoids act as antioxidants in plants and protect the internal structures of plants against various environmental stresses. The consequence of abiotic stresses is the production of harmful ROS, which causes damage to plant cells and tissues70. It has been found that the main role of phenolics and flavonoids is to inhibit oxidative stress and scavenge the reactive oxygen species (ROS), such as reactive superoxide anion radical (O2·−), singlet oxygen (O2), hydrogen peroxide (H2O2), and hydroxyl radical (·OH)71,72. In addition, the increased activity of antioxidant enzymes and compounds, such as phenol and flavonoid indicate the integrated participation of enzymatic and non-enzymatic antioxidant components in the regulation of ROS under salt stress conditions in thyme plants under hydroponic conditions59.
DPPH radical scavenging activity
The results showed that the content of DPPH radical scavenging activity was affected by different concentrations of sodium chloride and different times applying salinity stress (Table 2). Increasing salinity stress increased DPPH radical scavenging activity. Applying salinity stress at concentrations of 100 and 200 mM sodium chloride increased DPPH radical scavenging activity after 96 and 120 h after applying the stress. Also, it was found that the concentration of 200 mM sodium chloride after 24–120 h of salinity stress caused a significant increase in the DPPH radical scavenging activity of the garden thyme plant. Kaur et al.73 found that the DPPH radical scavenging activity of chickpea seedlings increased with increasing salinity stress, which agrees with the findings of the present research. It has been found that increasing the salinity stress up to 100 mM increased DPPH inhibition in the Kenaf (Hibiscus cannabinus L.) plant15. In the present study, the DPPH inhibition increased significantly with increasing salinity stress and the duration of stress (Table 2). Damage caused by free radicals is associated with many diseases and as a treatment, antioxidants have shown a strong effect against them59. Therefore, the increase in antioxidant activity and levels of bioactive phytochemicals can be used to increase the medicinal potential of garden thyme against biotic and abiotic stresses. Also, Azeem et al.59 found that the activity of antioxidant compounds in the Moringa medicinal plant increased with increasing salinity stress.
Correlation and multivariate analyses
The correlation plot (Table 3, Fig. 2) shows the correlations between fresh mass, physiological, and biochemical characters. There were positive and significant correlations between carotenoid, flavonoid, antioxidant, chlorophyll a, chlorophyll b, and total chlorophyll in the control level and 100 mM salinity stress. Also, the relative content of leaf water showed positive and significant correlations with the content of phenolic compounds and proline. Also, in these two salinity levels, the content of total chlorophyll showed positive and significant correlations with the content of chlorophylls a and b. At the level of 200 mM sodium chloride (severe stress), most of the physiological and biochemical traits showed positive and significant relationships with each other, but these traits did not show significant correlations with the fresh weight (Fig. 2). Moreover, heatmap could classify the treatments, where two distinguished clusters were obtained for the employed treatments. As shown in Fig. 2, fresh weight, electrical leakage, and relative water content of leaves were placed in one cluster in all salinity stress treatments and salinity application times (Fig. 3). Agglomerative hierarchical clustering (AHC) dendrogram showed that application of 100 mM sodium chloride for 72, 96, and 120 h and 200 mM salinity stress intensity for 48, 72, 96, and 120 h were placed in one cluster based on physiological and biochemical traits, while other salinity stress treatments and the durations of applying salt stress were placed in a cluster (Fig. 4).
The size and color intensity of the squares are proportional and consistent with the Pearson correlation coefficient at p < 0.05 in Thymus vulgaris. Red squares indicate positive and significant correlations, while blue squares indicate negative correlations. Pearson’s correlation coefficient for variables on the vertical and horizontal axis is on the correlation scale from − 1 to 1. *Also indicates values that are statistically different at p < 0.05.
The correlation finding is particularly important because proline content and electrolyte leakage are recognized as key biomarkers for assessing plant stress response mechanisms. Proline is an amino acid that protects against environmental stresses, such as drought, salinity, and cold. Electrolyte leakage, on the other hand, is a measure of cell membrane integrity, which can be compromised under stress conditions28,48.
The strong correlation (r = 0.74, p < 0.01) indicates a significant relationship between proline accumulation and cell membrane damage. This finding provides valuable insights into the ability of plants to respond to stress conditions. For instance, the relationship between high proline content and low electrolyte leakage helps us understand how plants adapt to stress and which conditions contribute to greater stress tolerance63.
The observed correlation coefficient between proline content and electrolyte leakage (r = 0.74) indicates a strong and statistically significant relationship between these two variables (p < 0.01). Proline is a crucial component of the osmoregulation mechanisms developed by plants in response to abiotic stress, playing key roles in maintaining osmotic balance at the cellular level, mitigating the damage caused by reactive oxygen species (ROS), and stabilizing protein structures. On the other hand, electrolyte leakage increases as a result of cell membrane damage under stress conditions and is considered an indicator of membrane integrity loss. In this context, the significant relationship between high proline content and increased electrolyte leakage is an important finding regarding the stress response mechanisms of plants28,63.
Such correlations play a critical role in agricultural applications by informing strategies for improving stress tolerance and plant breeding. For example, selecting genotypes with high proline accumulation or identifying appropriate cultivation methods may offer practical applications for enhancing plant resilience under stress conditions.
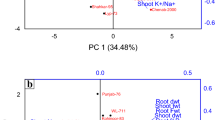
Principal component analysis (PCA)
The vector size shows the effect of each attribute and the direction of the vector depends on the values of PC1 and PC2. PCA summarizes the changes of 11 characteristics during three levels of salinity stress concentration A: no stress, B: 100 mM sodium chloride salt stress, and C: 200 mM sodium chloride salt stress. In non-stressed conditions, the PCA explained 69.8% of the total variation of the three levels of salinity stress (Fig. 5). This value was 64.8% and 79.94% for 100 and 200 mM sodium chloride salinity treatments, respectively (Fig. 5). Most of the variations were explained by the first component (PC1). Regardless of the direction of effect, flavonoids, carotenoids, and total chlorophyll in non-stress conditions, flavonoid, phenol, and antioxidant capacity at 100 mM salt stress, and carotenoids at 200 mM sodium chloride level have the largest contribution in the first main component (PC1). Relative water content and fresh weight at the control level, fresh weight, relative leaf water content, and electrical leakage at the level of 100 mM salinity stress, and total chlorophyll, relative leaf water content, and chlorophyll b have the largest effect in the second main component (PC2) (Fig. 5).
Conclusions
Salinity has harmful effects on the photosynthetic system of medical plants as it is known that salinity diminishes the photosynthesis rate by disrupting chlorophyll metabolism, inducing oxidative stress, and prompting plastid degradation. This decline can be attributed to the adverse effects of ion absorption disturbances on chloroplast development and the protein translation machinery within plastids. This research was designed to investigate salinity stress with different concentrations of sodium chloride and different times of applying salinity stress in garden thyme grown in the hydroponic system. The results showed that the increase of salinity stress in the hydroponic system in different periods of applying salinity stress can cause irreparable damage to the physiological and biochemical characteristics of garden thyme plants in hydroponic conditions. The increase in antioxidant activity and levels of bioactive phytochemicals can be used to increase the medicinal potential of garden thyme against biotic and abiotic stresses. Therefore, regulating the concentration of nutrients and optimizing the EC and pH of the nutrient solution in hydroponic systems are among the most important factors for growing medicinal plants, including garden thyme.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Kashif, M. H. et al. Comparative cytological and gene expression analysis reveals potential metabolic pathways and target genes responsive to salt stress in kenaf (Hibiscus cannabinus L.). J. Plant Growth Regul. 39, 1245–1260 (2020).
Liu, M. et al. Crop halophytism: An environmentally sustainable solution for global food security. Trends Plant Sci. 25(7), 630–634 (2020).
Li-ping, L. et al. Ameliorants improve saline–alkaline soils on a large scale in northern Jiangsu Province, China. Ecol. Eng. 81, 328–334 (2015).
Kumar, S. et al. Effect of salt stress on growth, physiological parameters, and ionic concentration of water dropwort (Oenanthe javanica) cultivars. Front. Plant Sci. 12, 660409 (2021).
Rahman, A. et al. Manganese-induced salt stress tolerance in rice seedlings: Regulation of ion homeostasis, antioxidant defense and glyoxalase systems. Physiol. Mol. Biol. Plants 22, 291–306 (2016).
Sahin, U. et al. Effects of individual and combined effects of salinity and drought on physiological, nutritional and biochemical properties of cabbage (Brassica oleracea var. capitata). Sci. Hortic. 240, 196–204 (2018).
Nxele, X., Klein, A. & Ndimba, B. Drought and salinity stress alters ROS accumulation, water retention, and osmolyte content in sorghum plants. S. Afr. J. Bot. 108, 261–266 (2017).
Rahman, M. M. et al. Ethanol positively modulates photosynthetic traits, antioxidant defense and osmoprotectant levels to enhance drought acclimatization in soybean. Antioxidants 11(3), 516 (2022).
De Rossi, S. et al. Investigating the drought and salinity effect on the redox components of Sulla coronaria (L.) Medik. Antioxidants 10(07), 1048 (2021).
Shahid, M. A. et al. Insights into the physiological and biochemical impacts of salt stress on plant growth and development. Agronomy 10(7), 938 (2020).
Chen, C. et al. Variations in physiology and multiple bioactive constituents under salt stress provide insight into the quality evaluation of Apocyni Veneti Folium. Int. J. Mol. Sci. 19(10), 3042 (2018).
Sachdev, S. et al. Abiotic stress and reactive oxygen species: Generation, signaling, and defense mechanisms. Antioxidants 10(2), 277 (2021).
Chen, W. et al. Comparative effects of osmotic-, salt-and alkali stress on growth, photosynthesis, and osmotic adjustment of cotton plants. Photosynthetica 49, 417–425 (2011).
Kobaisy, M. et al. Phytotoxic and fungitoxic activities of the essential oil of kenaf (Hibiscus cannabinus L.) leaves and its composition. J. Agric. Food Chem. 49(8), 3768–3771 (2001).
Birhanie, Z. M. et al. Salt stress induces changes in physiological characteristics, bioactive constituents, and antioxidants in Kenaf (Hibiscus cannabinus L.). Antioxidants 11(10), 2005 (2022).
Cutillas, A.-B. et al. Thyme essential oils from Spain: Aromatic profile ascertained by GC–MS, and their antioxidant, anti-lipoxygenase and antimicrobial activities. J. Food Drug Anal. 26(2), 529–544 (2018).
Letchamo, W. & Gosselin, A. Transpiration, essential oil glands, epicuticular wax and morphology of Thymus vulgaris are influenced by light intensity and water supply. J. Hortic. Sci. 71(1), 123–134 (1996).
Nickavar, B., Mojab, F. & Dolat-Abadi, R. Analysis of the essential oils of two Thymus species from Iran. Food Chem. 90(4), 609–611 (2005).
Pirbalouti, A. G., Hashemi, M. & Ghahfarokhi, F. T. Essential oil and chemical compositions of wild and cultivated Thymus daenensis Celak and Thymus vulgaris L. Ind. Crops Prod. 48, 43–48 (2013).
Hornok, L. Cultivation and Processing of Medicinal Plants (Wiley, 1992).
Nadi, A. et al. Thymus vulgaris, a natural pharmacy against COVID-19: A molecular review. J. Herb. Med. 38, 100635 (2023).
Sardari, S. et al. Therapeutic effect of thyme (Thymus vulgaris) essential oil on patients with covid19: A randomized clinical trial. J. Adv. Med. Biomed. Res. 29(133), 83–91 (2021).
Şakalar, Ç. & Ertürk, M. Inactivation of airborne SARS-CoV-2 by thyme volatile oil vapor phase. J. Virol. Methods 312, 114660 (2023).
Soufi, H. et al. The roles of light in a plant factory: Photosynthesis efficiency and gas exchange parameters of lettuce as a function of light spectra. Greenh. Plant Prod. J. 1(1), 1–26 (2024).
Badawy, E. et al. Effect of salinity stress and soil types on growth, photosynthetic pigments and essential oil of Artemisia annua L. Asian J. Crop Sci. 10, 40–47 (2018).
Zrig, A. et al. Influence of light intensity and salinity on growth and antioxidant machinery of Thymus vulgaris L. Indian J. Exp. Biol. New Delhi 58(5), 323–335 (2020).
Trifunović-Momčilov, M. et al. Changes in photosynthetic pigments content in non-transformed and AtCKX transgenic centaury (Centaurium erythraea Rafn) shoots grown under salt stress in vitro. Agronomy 11(10), 2056 (2021).
Stefanakis, M. K. et al. The effect of salinity and drought on the essential oil yield and quality of various plant species of the Lamiaceae family (Mentha spicata L., Origanum dictamnus L., Origanum onites L.). Horticulturae 10(3), 265 (2024).
Soufi, H. R. et al. Spectral composition of LED light differentially affects biomass, photosynthesis, nutrient profile, and foliar nitrate accumulation of lettuce grown under various replacement methods of nutrient solution. Food Sci. Nutr. 11(12), 8143–8162 (2023).
Hniličková, H. et al. Effects of salt stress on water status, photosynthesis and chlorophyll fluorescence of rocket. Plantt Soil Environ. 63, 362–367 (2017).
Soufi, H. R., Roosta, H. R. & Hamidpour, M. The plant growth, water and electricity consumption, and nutrients uptake are influenced by different light spectra and nutrition of lettuce. Sci. Rep. 13(1), 20766 (2023).
Soufi, H. R. et al. Manipulation of light spectrum is an effective tool to regulate biochemical traits and gene expression in lettuce under different replacement methods of nutrient solution. Sci. Rep. 13(1), 8600 (2023).
Bistgani, Z. E. et al. Effect of salinity stress on the physiological characteristics, phenolic compounds and antioxidant activity of Thymus vulgaris L. and Thymus daenensis Celak. Ind. Crops and Prod. 135, 311–320 (2019).
Suleiman, M. K. et al. Effects of salt stress on growth, proline and mineral content in native desert species. Sustainability 15(7), 6232 (2023).
Pungin, A. et al. Effect of salinity stress on phenolic compounds and antioxidant activity in halophytes Spergularia marina (L.) Griseb. and Glaux maritima L. cultured in vitro. Plants 12(9), 1905 (2023).
Nazari, M. et al. Deciphering the response of medicinal plants to abiotic stressors: A focus on drought and salinity. Plant Stress 10, 100255 (2023).
Zrig, A. et al. Potassium and calcium improve salt tolerance of Thymus vulgaris by activating the antioxidant systems. Sci. Hortic. 277, 109812 (2021).
Koźmińska, A. et al. Beet molasses enhance salinity tolerance in Thymus serpyllum—A study under greenhouse condition. Plants 10(9), 1819 (2021).
Hoagland, D. R. & Arnon, D. I. The Water-Culture Method for Growing Plants Without Soil/D.R. Hoagland, D. R. (Dennis Robert), 1884–1949 (University of California, College of Agriculture, Agricultural Experiment Station, Berkeley, Calif., 1938).
Porra, R. J., Thompson, W. A. & Kriedemann, P. E. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: Verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta (BBA( Bioenerg. 975, 384–394 (1989).
Lichtenthaler, H. K. [34] Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. In Methods in Enzymology 350–382 (Elsevier, 1987).
González, L. & González-Vilar, M. Determination of relative water content. In Handbook of Plant Ecophysiology Techniques 207–212 (Springer, 2001).
Shi, Q. et al. Effects of different treatments of salicylic acid on heat tolerance, chlorophyll fluorescence, and antioxidant enzyme activity in seedlings of Cucumis sativa L. Plant Growth Regul. 48, 127–135 (2006).
Paquin, R. & Lechasseur, P. Observations on a method of determining free proline in plant extracts. Can. J. Bot. 57(18), 1851–1854 (1979).
Singleton, V. L. & Rossi, J. A. Colorimetry of total phenolics with phosphomolybdic–phosphotungstic acid reagents. Am. J. Enol. Vitic. 16(3), 144–158 (1965).
Atanassova, M., Georgieva, S. & Ivancheva, K. Total phenolic and total flavonoid contents, antioxidant capacity and biological contaminants in medicinal herbs. J. Univ. Chem. Technol. Metall. 46(1), 81–88 (2011).
Barros, L., Baptista, P. & Ferreira, I. C. Effect of Lactarius piperatus fruiting body maturity stage on antioxidant activity measured by several biochemical assays. Food Chem. Toxicol. 45(9), 1731–1737 (2007).
Coêlho, M. R. et al. Salt tolerance of Calotropis procera begins with immediate regulation of aquaporin activity in the root system. Physiol. Mol. Biol. Plants 27, 457–468 (2021).
Yamane, K., Taniguchi, M. & Miyake, H. Salinity-induced subcellular accumulation of H2O2 in leaves of rice. Protoplasma 249, 301–308 (2012).
Gururani, M. A., Mohanta, T. K. & Bae, H. Current understanding of the interplay between phytohormones and photosynthesis under environmental stress. Int. J. Mol. Sci. 16(8), 19055–19085 (2015).
Paranychianakis, N. & Chartzoulakis, K. Irrigation of Mediterranean crops with saline water: From physiology to management practices. Agric. Ecosyst. Environ. 106(2–3), 171–187 (2005).
Dalton, F., Maggio, A. & Piccinni, G. Assessing the effect of solar radiation on plant salt tolerance as defined by the static and dynamic indices. Plant Soil 229, 189–195 (2001).
Tátrai, Z. A. et al. Morphological and physiological plant responses to drought stress in Thymus citriodorus. Int. J. Agron. 2016(1), 4165750 (2016).
Hajlaoui, H. et al. Differential effects of salt stress on osmotic adjustment and solutes allocation on the basis of root and leaf tissue senescence of two silage maize (Zea mays L.) varieties. Ind. Crops Prod. 31(1), 122–130 (2010).
Pérez-Pérez, J. et al. Response to drought and salt stress of lemon ‘Fino 49’under field conditions: Water relations, osmotic adjustment and gas exchange. Sci. Hortic. 122(1), 83–90 (2009).
Tavakkoli, E., Rengasamy, P. & McDonald, G. K. High concentrations of Na+ and Cl− ions in soil solution have simultaneous detrimental effects on growth of faba bean under salinity stress. J. Exp. Bot. 61(15), 4449–4459 (2010).
Banu, M. N. A. et al. Proline and glycinebetaine induce antioxidant defense gene expression and suppress cell death in cultured tobacco cells under salt stress. J. Plant Physiol. 166(2), 146–156 (2009).
Al Busaidi, K. T. S. & Farag, K. M. The use of electrolyte leakage procedure in assessing heat and salt tolerance of Ruzaiz date palm (Phoenix dactylifera L.) cultivar regenerated by tissue culture and offshoots and treatments to alleviate the stressful injury. J. Hortic. For. 7(4), 104–111 (2015).
Azeem, M. et al. Salinity stress improves antioxidant potential by modulating physio-biochemical responses in Moringa oleifera Lam. Sci. Rep. 13(1), 2895 (2023).
Molinari, H. B. C. et al. Evaluation of the stress-inducible production of proline in transgenic sugarcane (Saccharum spp.): Osmotic adjustment, chlorophyll fluorescence and oxidative stress. Physiol. Plant. 130(2), 218–229 (2007).
Ashraf, M. & Harris, P. J. Potential biochemical indicators of salinity tolerance in plants. Plant Sci. 166(1), 3–16 (2004).
Munns, R. & Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 59(1), 651–681 (2008).
Sultana, R. et al. Coumarin-mediated growth regulations, antioxidant enzyme activities, and photosynthetic efficiency of Sorghum bicolor under saline conditions. Front. Plant Sci. 13, 799404 (2022).
El Moukhtari, A. et al. How does proline treatment promote salt stress tolerance during crop plant development?. Front. Plant Sci. 11, 1127 (2020).
Saleem, A. et al. Iron sulfate (FeSO4) improved physiological attributes and antioxidant capacity by reducing oxidative stress of Oryza sativa L. cultivars in alkaline soil. Sustainability 14(24), 16845 (2022).
Valifard, M. et al. Effects of salt stress on volatile compounds, total phenolic content and antioxidant activities of Salvia mirzayanii. S. Afr. J. Bot. 93, 92–97 (2014).
Salem, M. Z., Abdel-Megeed, A. & Ali, H. M. Stem wood and bark extracts of Delonix regia (Boj. Ex. Hook): Chemical analysis and antibacterial, antifungal, and antioxidant properties. BioResources 9(2), 2382–2395 (2014).
Navarro, J. M. et al. Changes in the contents of antioxidant compounds in pepper fruits at different ripening stages, as affected by salinity. Food Chem. 96(1), 66–73 (2006).
Awika, J. M., Rooney, L. W. & Waniska, R. D. Anthocyanins from black sorghum and their antioxidant properties. Food Chem. 90(1–2), 293–301 (2005).
Sharma, P. et al. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012(1), 217037 (2012).
Selmar, D. & Kleinwächter, M. Stress enhances the synthesis of secondary plant products: The impact of stress-related over-reduction on the accumulation of natural products. Plant Cell Physiol. 54(6), 817–826 (2013).
Shomali, A. et al. Diverse physiological roles of flavonoids in plant environmental stress responses and tolerance. Plants 11(22), 3158 (2022).
Kaur, N. et al. DPPH radical scavenging activity and contents of H2O2, malondialdehyde and proline in determining salinity tolerance in chickpea seedlings. Indian J. Biochem. Biophys. 51, 407–415 (2014).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. HS, MAK, and MA experimented and collected data. HS wrote the manuscript. AK and YT reviewed, edited, and revised the manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Statement specifying permissions
For this study, we acquired permission to study Thymus vulgaris issued by the Agricultural and Natural Resources Ministry of Iran.
Statement on experimental research and field studies on plants
The either cultivated or wild-growing plants sampled comply with relevant institutional, national, and international guidelines and domestic legislation of Iran.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Salehi-Arjmand, H., Akhoundi, M., Akramian, M. et al. Time course of physiological and biochemical responses of Thymus vulgaris to short-term salinity stress in hydroponics. Sci Rep 15, 16272 (2025). https://doi.org/10.1038/s41598-025-00768-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-00768-y