Abstract
This study aims to investigate the efficacy of intranasally transplanted neural stem cells (NSCs) in anosmia-induced mice. Twenty-four male mice were included in the study. A food-finding test (FFT) was performed beforehand to confirm that all mice had normal olfactory functions. The mice were then randomly divided into two groups (Groups 1 and 2) and anosmia was induced by 3-methylindole (3-MI). In the first week following the 3-MI injection (week 1), mice in Group 1 received intranasal saline, while mice in Group 2 received intranasal NSCs. The NSCs were prepared from green fluorescent protein (GFP)-transgenic mice. Olfactory function was evaluated through weekly FFTs. One week after NSC or saline administration (week 2), half of the mice in each group were randomly selected and sacrificed for histological examination. The remaining mice were sacrificed for histological examination in week 4. At week 4, the olfactory epithelium of all mice was also examined by polymerase chain reaction (PCR) to detect GFP + cells. The NSC group showed significantly shorter FFT times compared to the saline group. Epithelial damage scores, indicating greater injury, were higher in the saline group, whereas the NSC group exhibited greater epithelial thickness. Immunohistochemical analysis revealed a significantly higher number of olfactory marker protein-expressing cells in the NSC group. GFP + cells were observed in olfactory bulb (OB) and olfactory epithelium (OE) in the NSC group. PCR confirmed the presence of the GFP gene in the OE of NSC-transplanted mice. Intranasal NSCs promoted functional recovery and GFP + cell integration, supporting further research into their therapeutic potential for olfactory dysfunction.
Similar content being viewed by others
Introduction
Anosmia is the loss of the sense of smell, which can be either partial or complete. It may result from various causes, such as viral infections, neurological disorders, head trauma, or exposure to certain chemicals1. Odor molecules are detected by the axons of olfactory neurons in the nose, creating a signal pathway towards the olfactory cortex. Basal cells located within the olfactory epithelium (OE) are essential for replacing olfactory receptor neurons in cases of damage, thus facilitating regeneration of the OE2,3. These basal cells consist of two distinct types: globose basal cells and horizontal basal cells. In addition to these, mesenchymal stem cells located within the olfactory mucosa and olfactory ensheathing cells have also been identified as potential contributors to olfactory regeneration4,5. However, the specific roles of these different stem cell populations in the regeneration of the olfactory nerves remain unclear. It is still uncertain whether all these cell types actively participate in the process or whether certain subpopulations play a more dominant role. This uncertainty, along with the incomplete understanding of the pathophysiological mechanisms underlying anosmia and hyposmia, complicates the establishment of a definitive treatment algorithm for anosmia1.
Stem cell therapies have shown promise in various neurodegenerative diseases, and for this reason, they are being explored as potential treatments for olfactory disorders, which can be considered a form of neural degeneration1,6. Among potential regenerative strategies, neural stem cells (NSCs) have gained increasing attention due to their potential to support neural repair. NSCs can secrete neurotrophic factors, secretome, modulate inflammation, and differentiate into neural lineages, making them promising candidates for olfactory regeneration7,8,9,10,11.
The green fluorescent protein (GFP) transgenic mice model developed by Okabe et al. has enabled direct tracking of transplanted cells in vivo12. In this study, we used GFP-labeled NSCs to monitor integration into olfactory tissue. We hypothesized that intranasal NSCs would enhance olfactory regeneration and function in a 3-MI–induced anosmia mouse model. This study uniquely combines intranasal delivery with behavioral and molecular assessments.
Materials and methods
This study was conducted in full compliance with the guidelines outlined in the National Institutes of Health’s Guide for the Care and Use of Laboratory Animals. The protocol was approved by the Local Ethics Board for Animal Experiments and supported by the Scientific Research Projects Coordination Unit, Gazi University (number: G.Ü.ET-21.070). A total of 24 healthy male mice (aged 8–10 weeks and weighing 18–22 g, Izmir Biomedicine and Genome Center, Turkey) were included in the study. The food-finding test (FFT) was used to confirm that all mice had intact olfactory function. NSCs were prepared from transgenic C57BL/6-Tg(CAG-EGFP)1Osb/J mice (Jackson Laboratory, USA) that ubiquitously express GFP. The mice were randomly assigned to two groups (n = 12 per group). An intraperitoneal injection of 300 µg/g 3-methylindole (3-MI) was administered to induce anosmia, which was confirmed with the FFT one-week post-injection. Weekly FFTs were subsequently performed to assess olfactory function. Half of the mice in each group were randomly selected and sacrificed for histological examination in the second week following 3-MI injection (week 2), while the remaining mice were sacrificed in week 4.
Food-finding test
An FFT was designed based on analyses of similar tests conducted in previous studies13,14,15,16,17. Mice were food-deprived for 24 h and then placed at the starting point of a T-shaped maze, with one corner containing a piece of cheese (2 × 1 cm) to which they had been previously acclimated. The maze was covered with sawdust, which was changed before each test. Each mouse was tested five times per FFT. All tests were conducted in a quiet environment at the same location and recorded with a video camera. Mice that could not find the cheese within 180 s were considered to have “failed”. Mice failing three out of five attempts in an FFT were categorized as “unsuccessful”. For each FFT, the minimum and maximum times were excluded to reduce outliers, and the average of the remaining three tests was taken. The average food finding time for failed mice was recorded as 180 s.
Preparation of stem cells
NSCs were isolated from the OBs of 8-week-old GFP-transgenic mice using previously published protocols8,18. Briefly, dissected OBs were mechanically teased apart and then enzymatically dissociated in the presence of 0.25% trypsin (Sigma, St Louis, MO) for 10 min at 37 °C. The digested tissue was mixed with Hank’s Balanced Salt Solution (Invitrogen, San Diego, CA). Next, the slurry was filtered through a 40- µm nylon mesh (Millipore, Billerica, MA, USA) to obtain individual cells. The cells were then washed via centrifugation at 500xg for 10 min. The isolated cells were cultured in CO2 incubators (Sanyo, Japan) at 37 °C and 5% CO2 in DMEM/F-12 culture medium (Invitrogen, USA) supplemented with 2% FBS (Sigma, USA), 2 mM L-glutamine (Sigma, USA), and penicillin/streptomycin solution (Sigma, USA). The culture medium also contained N-2 (R&D Systems, Minneapolis, USA), 20 ng/ml epidermal growth factor (R&D Systems, Minneapolis, MN), and 20 ng/ml basic fibroblast growth factor (R&D Systems, Minneapolis, USA). Finally, floating cells were transferred to fresh wells after 24 h. Neurospheres appeared in cultures within 7–10 days (Fig. 1a–b).
Administration of stem cells
One week after the 3-MI injection (week 1), 5 × 105 NSCs in 50 µL of phosphate-buffered saline (PBS) were transplanted transnasally into 12 mildly anesthetized mice (Group 2). The cells were administered in 5-µL drops into each nostril using a previously validated method8,9. The same volume of PBS was administered to the remaining 12 mice (Group 1) using the same method.
Evaluation of neural stem cells engraftment
All the animals were euthanized with a high dose of anesthesia (at least 200 mg/kg ketamine, 10 mg/kg xylazine). After decapitation, each head was bisected sagittally, and a randomly selected side was dissected under a microscope using microsurgical instruments to isolate the olfactory mucosa and OB from surrounding structures to examine transplanted GFP + stem cells without staining. The other side was preserved in formaldehyde for histological examination. All the histological assessments were conducted by blinded investigators. To analyze stem cell engraftment, 5-µm sections were obtained at −20 °C using a cryostat device (Leica CM1900, Germany) and then examined with an inverted immunofluorescence microscope (Leica DMI4000B, Germany).
In week 4, mice in Groups 1 and 2 were examined using PCR to detect the presence of GFP in the olfactory epithelium. Genomic DNA was extracted from olfactory tissue using a commercially available kit following the manufacturer’s instructions (Qiagen, USA). Briefly, 5 mg of tissue was cut into small pieces and placed in an Eppendorf tube containing proteinase K buffer provided by the manufacturer. The tissue was incubated at 56 °C overnight. After the washing process, genomic DNA was placed in a column with binding buffer, then eluted with elution buffer by centrifugation. PCR was performed using a PCR kit (Taq PCR Master Mix Kit, Qiagen, USA) following standard protocols. GFP-specific primers were used (Table 1). PCR products were visualized on 1% agarose gels using a gel imaging system (iBright Imaging System, ThermoFisher, USA).
Histological examination
For histomorphological evaluation, the samples were initially fixed in a 10% neutral formaldehyde solution for 72 h, then placed in De Castro solution for decalcification. Following decalcification, samples were washed in running tap water and dehydrated through an alcohol series (50%, 70%, 80%, 90%, and 100% respectively). Paraffin blocks of 4–5 μm thickness were prepared, and sections were stained with hematoxylin-eosin. A light microscope (Leica DM4000, Germany) was used for evaluations. Loss of cilia, vacuolization, and inflammatory cell infiltration were each scored on a scale from 0 to 3 (0: no damage, 1: mild [sparse involvement], 2: moderate [focal dense clusters], 3: severe [widespread dense involvement]). The total histological injury score was calculated as the sum of these three parameters, with an overall maximum score of 917. The thickness of the OE was measured at three points (two peripheral and one central), and the average of these three values was calculated.
Immunohistochemical analyses were also performed. The samples were kept in xylol twice, each time for 15 min. They were then immersed in decreasing concentrations of ethanol solutions for 10 min each (100%, 96%, 90%, 80%, and 70% respectively). Later, the samples were soaked twice in distilled water for 5 min each time. Next, tissues were circumscribed with PAP-Pen in a humid environment and washed with PBS three times for 3 min each time. Treated with serum blocking solution for 10 min, the samples were incubated overnight with OMP primary antibodies (Biorbyt, UK, Cat: 473753) at + 4 °C. Following the incubation, the samples, washed again with PBS, were treated with a 3% hydrogen peroxide solution for 15 min to inhibit endogenous peroxidase activity. After re-washing the samples with PBS, a biotinylated secondary antibody (Genemed Biotechnologies, USA, Cat: 54003) was applied. Then, the samples were washed once again with PBS, subjected to chromogen containing diaminobenzidine, and left until a visible immunoreaction occurred. Next, they were re-washed with PBS and stained with Mayer’s Hematoxylin as a background. Dehydrated through an alcohol series, the samples were sealed with a lamella using Entellan after being kept in xylol. All the samples were evaluated by capturing photographs using the Leica DM4000 (Germany) computer-assisted imaging system and analyzed with the Leica Qwin V3 software. OMP binding was assessed through cell counts in seven independent fields selected for each slide.
Statistical analysis
Statistical Package for the Social Sciences for Windows, version 26. 0 (SPSS Inc., Chicago, IL) was used for statistical analysis. In terms of the descriptive statistics, frequencies and percentages were calculated for categorical variables and mean, median, standard deviation, minimum, and maximum values for numerical variables. Mann-Whitney U test was used for comparisons of FFT times, histological thickness measurements, the number of OMP + cells, and total epithelial damage scores between groups. Chi-square or Fisher’s Exact test was applied for categorical variables such as success rates in FFT and categorical histological parameters (loss of cilia, vacuolization, inflammatory cell infiltration). Wilcoxon test was used for within-group comparisons over time. A p-value less than 0.05 was considered significant.
Results
Food-finding test
The first food-finding test (FFT 1) was conducted beforehand to assess the olfactory functions of all the mice, while the second test (FFT 2) aimed to demonstrate the induction of anosmia with 3-MI. The remaining three tests were performed weekly to monitor olfactory function recovery. Following 3-MI injection, FFT 2 (in week 1) confirmed the induction of anosmia in both groups. All FFT results are summarized in Table 2 and Table 3. There was no statistically significant difference between the groups in FFT 1 and FFT 2.
The results indicated that Group 2 had statistically better success rates in FFT 3 (p < 0.05), while no statistically significant difference was observed in FFT 4 (p = 0.08) and FFT 5 (p = 0.242). Although Group 2 showed better mean FFT times at weeks 3 and 4 (FFT 4 and FFT 5), the differences did not reach statistical significance, which may be attributed to the limited sample size (n = 6 per group at week 4), potentially reducing the statistical power. When comparing average food-finding times in the groups, no statistically significant differences were observed in FFT 1 and FFT 2. However, in FFT 3, FFT 4, and FFT 5, the average finding times of Group 2 were significantly shorter compared to those of Group 1 (p < 0.05).
Detection of neural stem cells in olfactory tissues
The OE and OBs of the NSC-transplanted subjects were examined using an inverted fluorescence microscope. Whole-mount tissue imaging of the OE and OB revealed multiple conglomerates of green fluorescent neural stem cells. (Fig. 2). To prove that transferred NSCs incorporated themselves into the olfactory tissues and restored the olfactory function, we assessed the presence of GFP gene in OE of mice that received NSCs from GFP transgenic mice in week 4. Genomic DNA was isolated from the OE of both control and NSCs transplanted mice, and PCR analysis was performed. The GFP gene existed in NSC transplanted animals whereas no GFP amplification was observed in the control group (Fig. 3). This evidence demonstrated that transferred NSCs incorporated themselves into the olfactory tissue of the anosmic mice and restored the sense of smell.
Histological examinations
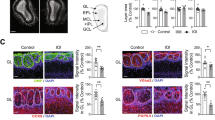
The epithelial thickness in Group 2 (stem cell transplantation group) was statistically greater in week 2 and week 4 compared to Group 1 (p < 0.05). The mean epithelial thickness was 12.44 ± 0.96 μm vs. 28.94 ± 4.4 μm in week 2 and 12.39 ± 0.88 μm vs. 41.89 ± 5.24 μm in week 4 for Group 1 and 2 respectively.
Histomorphologically, inflammatory cell infiltration, epithelial vacuolization, and cilia damage were scored in the OE of the subjects. The findings are summarized in Table 4. Mann-Whitney U test was used for comparisons of total epithelial damage scores between groups. Categorical histological parameters were evaluated using Fisher’s Exact or Chi-square tests. Histomorphological analysis revealed that epithelial damage was more pronounced in the control group (Fig. 4). The number of OMP + cells was significantly higher in Group 2 (Table 5). The significantly higher number of OMP + cells in the NSC group suggests enhanced neuronal maturation within the regenerating olfactory epithelium.
Images of histological evaluations (a) Olfactory epithelium of the group 1 at second week (Hematoxylin-eosin, 20x) (b) Olfactory epithelium of the group 2 at second week (Hematoxylin-eosin) (c) Group 1- OMP + cells at 4 weeks (d) Group 2 – OMP + cells at 4 weeks]: Epithelial thickness Black arrow: Duct of the gland ⬥: Vacuolization ❖: Inflammatory cell infiltration ★: Vascularization ←: Loss of cilia RE: Respiratory epithelium.
Discussion
Our study demonstrated that NSC transplantation contributes to the regeneration of the olfactory epithelium and enhances functional recovery in an anosmia-induced mouse model. Histological analysis confirmed the presence of GFP + cells in the OE and OB, suggesting successful integration of transplanted NSCs. Additionally, PCR results indicated the presence of these cells at week 4, supporting their potential role in long-term epithelial repair.
Cell therapies are anticipated to play an effective role in disease treatment in the coming decade. Stem cell treatments have proved to be efficacious particularly in diseases caused by nerve damage6,19,20,21. This effect seems to occur primarily through their paracrine mechanisms21,22,23. While paracrine effects, including secretion of neurotrophic and angiogenic factors, are proposed mechanisms, these effects remain likely but unconfirmed within the current experimental model. Key NSC-derived factors include neurotrophic factors like brain-derived neurotrophic factor and glial cell line-derived neurotrophic factor, which support neuronal survival, differentiation, and synaptic plasticity. Additionally, NSCs produce cytokines and growth factors, such as vascular endothelial growth factor and transforming growth factor-beta, which modulate inflammation and promote tissue repair24.
In animal studies, disease induction is necessary prior to testing a therapeutic agent. For the model of anosmia, we selected the commonly used agent 3-MI16,25,26,27,28, administering a maximal intraperitoneal dose that induces olfactory loss without causing pneumotoxic effects16. The primary mechanism is the damage caused by the metabolism of 3-MI by mixed-function oxidases, which are present in the olfactory epithelium of the nasal cavity. Since stem cells in the damaged olfactory epithelium are also affected, fibrosis and metaplasia occur subsequently. Although partial regeneration is observed over time in the olfactory epithelium, it is still considered a good olfactory dysfunction model26,27. Stem cells are known to home to injury sites, and in this case, the target region is the olfactory tissue damaged by 3-MI29.To mitigate the entrapment of systemically administered stem cells in the lungs, which reduces the number of cells reaching the target site, we opted for transnasal application in our study30. Treatment effectiveness was assessed using behavioral, histomorphological, and immunohistochemical analyses. Additionally, the transplanted stem cells were derived from GFP + mice to facilitate investigation of their integration into the tissue.
Frequently used in the literature to evaluate olfactory function, FFTs showed that mice administered stem cells performed better in the present study. In the stem cell group, the epithelial damage score was lower, with increased epithelial thickness and a higher expression of OMP + cells. These findings suggest that NSCs may be effective in treatment. When comparing Group 1 samples from weeks 2 and 4, a reduction in cilia loss was observed by the 4 th week, along with a regression in vacuolization, indicating a limited degree of epithelial regeneration. However, the lack of significant differences in OMP levels between the 2nd and 4 th-week samples suggests no substantial difference in neuroepithelial maturation at these time points. As OMP is a key marker of mature olfactory sensory neurons (OSNs), the higher OMP expression in the NSC-transplanted group implies a potential positive impact of stem cells on neuroepithelial maturation31. During week 4 immunofluorescence microscopy, a higher density of GFP + cells was observed compared to week 2, indicating potential settlement and proliferation of stem cells in the OE.
Lee et al. reported that in anosmia-induced mice, NSC transplantation resulted in higher survival rates, increased OMP expression, and improved FFT performance at week 4 post-injection8. Similarly, our study showed higher OMP expression in the NSC group; however, survival rates were comparable between groups, with no unexpected deaths. Kurtenbach et al., in contrast, transplanted globose basal cells intranasally, demonstrating that these cells formed axonal structures and enhanced olfactory function compared to controls9. However, their study utilized a congenitally ciliopathic OSN mouse model, whereas ours employed a chemically induced anosmia model. In contrast, NSCs provide exogenous regeneration, unlike endogenous basal cells. Furthermore, our histological comparisons with a control group—considering the differing survival outcomes reported in Lee et al.‘s study—and our analysis of stem cell impact through olfactory bulb examinations represent key contributions to the literature.
Although autofluorescent cellular proteins exhibit lower intensity, they may still be present in the host mouse and potentially mimic GFP. To eliminate this confounding factor and obtain more objective data in the present study, the GFP gene’s presence in the olfactory epithelium was confirmed through PCR analysis. GFP + structures were observed in the olfactory bulb. While Kurtenbach et al. demonstrated that these structures are axons of olfactory neurons, our lack of advanced histological techniques limits our ability to determine whether these structures are axons or stem cells that migrated directly to this area via the cribriform plate, which marks a limitation of our study.
The question of whether a single NSC transplantation is sufficient for long-term recovery or if multiple interventions are necessary depends on several factors, including cell survival, integration, and disease progression. Chronic or progressive conditions may require periodic NSC transplantation to sustain therapeutic effects, particularly if the initial population diminishes over time due to attrition or immune response. In many cases of olfactory loss, the etiology often involves acute events such as trauma or infection, which are typically non-progressive. In these cases, a one-time autologous transplantation, which carries minimal rejection risk, could potentially be sufficient. However, despite the low rejection risk, challenges in NSC production from the individual complicate autologous transplants. This makes allogeneic transplants a viable alternative, though the higher likelihood of immune responses in allogeneic transplants could necessitate repeated applications. Moreover, limited data exist on how the etiology of olfactory dysfunction affects stem cell integration, making it challenging to predict the survival and functionality of transplanted NSCs in these contexts. Further research is required to understand the interactions between various causes of olfactory loss and stem cell therapies, particularly regarding cell integration and the potential need for repeated treatments. The challenges in producing NSCs and the potential benefit of repeated treatments suggest that MSCs could be considered as an alternative to NSCs, given the ease of repeated transplantations32,33. Although MSCs have shown promise in treating olfactory loss, there is currently no evidence that nasal mesenchymal cells belong to the OSN lineage9,22,23,34,35.
In this study, the effectiveness of NSCs in an anosmia-induced mouse model was evaluated. It should be noted that, as an animal model, this study may not fully replicate human anosmia. This study is limited by its small sample size and the use of single-dose stem cell administration. These factors may affect the generalizability of the results to broader clinical contexts. Additionally, rodents possess heightened olfactory capabilities and a larger proportion of olfactory receptor neurons compared to humans. A major limitation of this study is the lack of evaluation of the differentiation status of transplanted stem cells. However, cell therapies for olfactory loss can be applied non-invasively and conveniently through endoscopic methods. Therefore, positive results from preclinical studies will be promising for the potential transition to clinical trials.
Conclusion
In a mouse model of anosmia induced by intraperitoneal administration of 3-MI, intranasal NSCs enhanced functional recovery and promoted cell integration, supporting further studies on dosing strategies and long-term efficacy.
Data availability
The data supporting the findings of this study are included in this published article. Raw data generated and/or analysed during the current study are available from the corresponding author, upon reasonable request.
Change history
13 August 2025
A Correction to this paper has been published: https://doi.org/10.1038/s41598-025-14924-x
References
Whitcroft, K. L. et al. Position paper on olfactory dysfunction: 2023. Rhinology https://doi.org/10.4193/Rhin22.483 (2023).
Hadley, K., Orlandi, R. R. & Fong, K. J. Basic anatomy and physiology of olfaction and taste. Otolaryngol. Clin. North. Am. 37, 1115–1126. https://doi.org/10.1016/j.otc.2004.06.009 (2004).
Smith, T. D. & Bhatnagar, K. P. Anatomy of the olfactory system. Handb. Clin. Neurol. 164, 17–28. https://doi.org/10.1016/b978-0-444-63855-7.00002-2 (2019).
Au, E. & Roskams, A. J. Olfactory ensheathing cells of the lamina propria in vivo and in vitro. Glia 41, 224–236. https://doi.org/10.1002/glia.10160 (2003).
Alvites, R. D. et al. Rat olfactory mucosa mesenchymal stem/stromal cells (OM-MSCs): A characterization study. Int. J. Cell. Biol. 2020 (2938258). https://doi.org/10.1155/2020/2938258 (2020).
Kim, S. U. & de Vellis, J. Stem cell-based cell therapy in neurological diseases: a review. J. Neurosci. Res. 87, 2183–2200. https://doi.org/10.1002/jnr.22054 (2009).
Chen, X., Fang, H. & Schwob, J. E. Multipotency of purified, transplanted globose basal cells in olfactory epithelium. J. Comp. Neurol. 469, 457–474. https://doi.org/10.1002/cne.11031 (2004).
Lee, C. H. et al. Transplantation of neural stem cells in anosmic mice. Clin. Exp. Otorhinolaryngol. 3, 84–90. https://doi.org/10.3342/ceo.2010.3.2.84 (2010).
Kurtenbach, S. et al. Cell-Based therapy restores olfactory function in an inducible model of hyposmia. Stem Cell. Rep. 12, 1354–1365. https://doi.org/10.1016/j.stemcr.2019.05.001 (2019).
De Gioia, R. et al. Neural stem cell transplantation for neurodegenerative diseases. Int. J. Mol. Sci. 21 https://doi.org/10.3390/ijms21093103 (2020).
Dause, T. J., Denninger, J. K., Smith, B. M. & Kirby, E. D. The neural stem cell secretome across neurodevelopment. Exp. Neurol. 355, 114142. https://doi.org/10.1016/j.expneurol.2022.114142 (2022).
Okabe, M., Ikawa, M., Kominami, K., Nakanishi, T. & Nishimune, Y. Green mice’ as a source of ubiquitous green cells. FEBS Lett. 407, 313–319. https://doi.org/10.1016/s0014-5793(97)00313-x (1997).
Cho, H. J. et al. Newly developed method for mouse olfactory behavior tests using an automatic video tracking system. Auris Nasus Larynx. 45, 103–110. https://doi.org/10.1016/j.anl.2017.03.007 (2018).
Kim, B. Y. et al. The neuroplastic effect of olfactory training to the recovery of olfactory system in mouse model. Int. Forum Allergy Rhinol. 9, 715–723. https://doi.org/10.1002/alr.22320 (2019).
Kim, H. Y. et al. Effects of Statins on the recovery of olfactory function in a 3-methylindole-induced anosmia mouse model. Am. J. Rhinol Allergy. 26, e81–84. https://doi.org/10.2500/ajra.2012.26.3719 (2012).
Kim, J. W., Hong, S. L., Lee, C. H., Jeon, E. H. & Choi, A. R. Relationship between olfactory function and olfactory neuronal population in C57BL6 mice injected intraperitoneally with 3-methylindole. Otolaryngol. Head Neck Surg. 143, 837–842 (2010).
Yasak, A. G., Yigit, O., Araz Server, E., Durna Dastan, S. & Gul, M. The effectiveness of platelet-rich plasma in an anosmia-induced mice model. Laryngoscope 128, E157–e162. https://doi.org/10.1002/lary.27029 (2018).
Ahn, J. M. et al. Maintenance of regional difference in cellular composition of neurospheres derived from adult mouse olfactory bulb. Eur. Arch. Otorhinolaryngol. 265, 429–434. https://doi.org/10.1007/s00405-007-0487-6 (2008).
Andres, R. H., Choi, R., Steinberg, G. K. & Guzman, R. Potential of adult neural stem cells in stroke therapy. Regen Med. 3, 893–905. https://doi.org/10.2217/17460751.3.6.893 (2008).
Lindvall, O. & Kokaia, Z. Prospects of stem cell therapy for replacing dopamine neurons in Parkinson’s disease. Trends Pharmacol. Sci. 30, 260–267. https://doi.org/10.1016/j.tips.2009.03.001 (2009).
Cofano, F. et al. Mesenchymal stem cells for spinal cord injury: current options, limitations, and future of cell therapy. Int. J. Mol. Sci. 20 https://doi.org/10.3390/ijms20112698 (2019).
Jo, H., Jung, M., Seo, D. J. & Park, D. J. The effect of rat bone marrow derived mesenchymal stem cells transplantation for restoration of olfactory disorder. Biochem. Biophys. Res. Commun. 467, 395–399. https://doi.org/10.1016/j.bbrc.2015.09.142 (2015).
Kwon, J. W., Jo, H. G., Park, S. M., Ku, C. H. & Park, D. J. Engraftment and regenerative effects of bone marrow stromal cell transplantation on damaged rat olfactory mucosa. Eur. Arch. Otorhinolaryngol. 273, 2585–2590. https://doi.org/10.1007/s00405-016-3957-x (2016).
Zayed, M. A. et al. Stem-Cell-Based therapy: the celestial weapon against neurological disorders. Cells 11 https://doi.org/10.3390/cells11213476 (2022).
Durham, S. K. & Castleman, W. L. Pulmonary lesions induced by 3-methylindole in mice. Am. J. Pathol. 121, 128–137 (1985).
Turk, M. A., Henk, W. G. & Flory, W. 3-Methylindole-induced nasal mucosal damage in mice. Vet. Pathol. 24, 400–403. https://doi.org/10.1177/030098588702400506 (1987).
Miller, M. A. & O’Bryan, M. A. Ultrastructural changes and olfactory deficits during 3-methylindole-induced olfactory mucosal necrosis and repair in mice. Ultrastruct Pathol. 27, 13–21. https://doi.org/10.1080/01913120309944 (2003).
Wiethoff, A. J., Harkema, J. R., Koretsky, A. P. & Brown, W. E. Identification of mucosal injury in the murine nasal airways by magnetic resonance imaging: site-specific lesions induced by 3-methylindole. Toxicol. Appl. Pharmacol. 175, 68–75. https://doi.org/10.1006/taap.2001.9235 (2001).
Smart, N. & Riley, P. R. The stem cell movement. Circ. Res. 102, 1155–1168. https://doi.org/10.1161/circresaha.108.175158 (2008).
Fischer, U. M. et al. Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect. Stem Cells Dev. 18, 683–692. https://doi.org/10.1089/scd.2008.0253 (2009).
Albeanu, D. F. et al. Olfactory marker protein (OMP) regulates formation and refinement of the olfactory glomerular map. Nat. Commun. 9, 5073. https://doi.org/10.1038/s41467-018-07544-9 (2018).
Carr, V. M. & Farbman, A. I. The dynamics of cell death in the olfactory epithelium. Exp. Neurol. 124, 308–314. https://doi.org/10.1006/exnr.1993.1201 (1993).
Gaun, V., Martens, J. R. & Schwob, J. E. Lifespan of mature olfactory sensory neurons varies with location in the mouse olfactory epithelium and age of the animal. J. Comp. Neurol. 530, 2238–2251. https://doi.org/10.1002/cne.25330 (2022).
Franceschini, V. et al. Transplanted human adipose tissue-derived stem cells engraft and induce regeneration in mice olfactory neuroepithelium in response to dichlobenil subministration. Chem. Senses. 39, 617–629. https://doi.org/10.1093/chemse/bju035 (2014).
Kim, Y. M. et al. Effects of systemic transplantation of adipose tissue-derived stem cells on olfactory epithelium regeneration. Laryngoscope 119, 993–999. https://doi.org/10.1002/lary.20187 (2009).
Funding
Supported by Gazi University Scientific Research Projects Coordination Unit (Project Code: 01/2020-39, ID: 6283).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. B.H.: Writing – original draft, Visualization, Software, Project administration, Methodology, Investigation, Formal analysis, Data curation, Conceptualization, A.C.: Writing – review & editing, Methodology, Project administration,, Visualization, Conceptualization, E.U.B.: Writing – review & editing, Methodology, Visualization, Data curation, Conceptualization, D.D.: Writing – review & editing, Methodology, Visualization, Data curation, Conceptualization, M.A.: Writing – review & editing, Visualization, Data curation, B.T.C.: Writing – review & editing, Visualization, Data curation, Formal analysis, N.O.: Writing – review & editing, Visualization, Data curation, Formal analysis, M.M.S.: Writing – review & editing, Methodology, Project administration,, Visualization, Conceptualization.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no funding, financial relationships, or conflicts of interest to report.
Ethical approval
The study was approved by Gazi University Local Ethics Committee for Animal Experiments on October 25, 2021 (number: G.Ü.ET-21.070). All procedures were performed in accordance with the ethical standards of the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health, and ARRIVE guidelines. No unexpected adverse events occurred.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained an error in the name of Emin Ümit Bagriacik, which was incorrectly given as Emin Ümit Bagariack, and Figures 3 and 4 were switched in the HTML version. Full information regarding the correction made can be found in the correction for this Article.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hazir, B., Ceylan, A., Bagriacik, E.Ü. et al. Effects of intranasal neural stem cells transplantation on olfactory epithelium regeneration in an anosmia-induced mouse model. Sci Rep 15, 17015 (2025). https://doi.org/10.1038/s41598-025-01358-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-01358-8