Abstract
A systematic review of the effects of resistance training on neuromuscular adaptations related to maximal strength enhances our understanding of the mechanisms and dose–response relationships involved. This evidence supports the scientific application of maximal strength to improve athletic performance in practice. Methods: We retrieved literature from CNKI, PubMed, and Web of Science databases. We utilised Review Manager 5.4.1 software to construct forest plots and assess methodological quality based on the standards outlined in the Cochrane Handbook for Systematic Reviews of Interventions (version 5.1.0). Review Manager 5.3 was employed to analyse the outcome measures of the included studies. Given that the outcome measures were continuous variables, we chose the standardised mean difference (SMD) as the effect size metric for statistical analysis. We used Stata-SE 18.0 to conduct publication bias analysis. Results: Twenty studies examined the relationship between resistance training and maximal skeletal muscle strength, revealing low heterogeneity (I2 = 17%, P = 0.24). The results indicated an SMD of 0.77 (95% CI 0.57–0.98, P < 0.05), demonstrating a significant effect. The publication bias test yielded t = 4.12 (P < 0.05). Fifty-four studies examined the relationship between resistance training and peak torque, revealing moderate heterogeneity (I2 = 48%, P < 0.05). The results indicated an SMD of 0.77 (95% CI 0.62–0.93, P < 0.05), demonstrating a significant difference. The publication bias test yielded t = 6.69 (P < 0.05). Eighteen studies examined the relationship between resistance training and the percentages of Type I, Type IIa, and Type IIx muscle fibres, revealing significant moderate heterogeneity (I2 = 85%, P < 0.05). The results indicated an SMD of 0.14 (95% CI − 0.45–0.74, P = 0.63). The publication bias test yielded z = 3.62 (P < 0.05). Forty-one studies examined the relationship between resistance training and muscle thickness, revealing no heterogeneity (I2 = 0%, P = 0.80). The results indicated that resistance training programs significantly increase participants’ skeletal muscle thickness (SMD = 0.55, 95% CI 0.41–0.69, P < 0.05). The publication bias test yielded z = 2.09 (P < 0.05). Ten studies examined the relationship between resistance training and pennation angle, revealing moderate heterogeneity (I2 = 54%, P < 0.05). The results indicated an SMD of 0.36 (95% CI − 0.02–0.74, P = 0.06). The publication bias test yielded z = − 2.71 (P < 0.05). Twenty-eight studies examined the relationship between resistance training and EMG, revealing moderate heterogeneity (I2 = 58%, P < 0.05). The results indicated that resistance training programs significantly increase participants’ squat strength (SMD = 0.54, 95% CI 0.26–0.81, P < 0.05). The publication bias test yielded z = 5.62 (P < 0.05). Maximal resistance training enhances maximum strength and peak torque in bench presses and squats. Muscle adaptations include increased agonist muscle thickness, a higher proportion of Type I and Type IIa fibres, a reduction in Type IIx fibres, and an increase in pennation angle. Neural adaptations are reflected in heightened EMG amplitude, though the effect size varies with muscle contraction type and training experience. Physiologically, maximal resistance training activates satellite cells and the mTOR signalling pathway, contributing to muscle repair, hypertrophy, and strength improvement.
Similar content being viewed by others
Introduction
Resistance training is an effective exercise intervention widely used in athletes’ training and promoting health among the general population. The primary objective of resistance training is to enhance maximum muscle strength and functional performance through a systematic approach. Recently, many studies have examined the effects of resistance training on neuromuscular adaptations in developing maximum muscle strength, revealing the physiological mechanisms that enhance muscle strength. These mechanisms include structural changes in muscle fibres and adaptive responses in the nervous system. Neuromuscular adaptation refers to the physiological changes due to the interaction between the nervous and muscular systems during strength training. Resistance training can remodel the nervous and muscular systems, allowing muscles to exert strength more effectively under high-intensity loads1,2. However, current research indicates variability in training methods, models, and subjects regarding maximum strength development. Some studies suggest different muscle contraction forms correlate with motor unit activation patterns3. These factors may result in variations in training outcomes observed in muscles during exercise4,5. However, the mechanisms by which resistance training induces neuromuscular adaptations for maximum strength and the dose–response relationships that contribute to these adaptations remain poorly understood. Therefore, a systematic meta-analysis integrating existing literature will comprehensively understand how resistance training influences neuromuscular adaptations and the associated dose–response relationships, addressing current research gaps. Research on neuromuscular adaptations related to developing maximum strength through strength training has significant theoretical and practical implications, offering valuable scientific guidance and a foundation for future research and practical applications.
Research methods
Search strategy
Search terms: (“Strength training” OR “Strength” OR “Resistance training” OR “Strengthening programs” OR “Progressive strength training” OR “Resistance exercise” OR “Weight lifting” OR “Weight exercise” OR “Strength exercise” OR “Weight training” OR “Intensive strength training”) AND (“Neuroadaptation” OR “Neuromuscular adaptations” OR “Neuromuscular function” OR "Muscle adaptation”) 2000-01-01/2024-9-02. Form China National Knowledge Infrastructure, Wan fang Database, PubMed, Web of Science, Scopus, EBSCOhost (SPORT Discus) databases. See Appendix 1 for specific search strategies.
Selection criteria
The present meta-analysis selected studies that adhered to the PICOS framework. This standardized methodology encompasses five key components for systematic review and meta-analysis: population (P), interventions (I), comparators (C), outcomes (O), and study design (S)6. Two independent evaluators (YZ and XRZ) conducted a comprehensive assessment of potentially relevant studies according to the established inclusion and exclusion criteria. This process involved reviewing the titles, abstracts, and full texts of the studies to determine their eligibility for inclusion in the meta-analysis. If the two authors (YZ and XRZ) disagreed on the inclusion or exclusion of an article, a third author (KGS) was consulted to resolve the disagreement.
Data extraction
The data extraction process was carried out by the first reviewer (SS) using a standardized form developed in Microsoft Excel. The second reviewer (HCM) then independently verified all the extracted data. In cases where discrepancies arose between the two reviewers, the study information was rechecked to resolve any differences. From each included study, the following information was extracted: (1) general publication details (e.g., author name, year of publication); (2) participant details (e.g., age, sample size, sex); (3) intervention specifics (e.g., duration, frequency); and (4) outcome measures related to the indicators of neuromuscular adaptation.
Literature inclusion and exclusion criteria
Entry criteria
(1) Studies must employ a randomised controlled design. The research subjects and methods must clearly define the experimental or control groups, ensuring uniform distribution of samples by age, sex, etc., with random and parallel interventions; (2) Study subjects should include three populations: well-trained competitive athletes, fitness enthusiasts with general training experience, and healthy adults without training experience; (3) The study design must involve pure resistance training, with detailed descriptions of training type, load volume, intensity, frequency, and cycle; 4.The results section must include detailed data analysis tables showing pre- and post-training changes in physiological and biochemical indicators between experimental and control groups, along with statistical tests.
Exclusion criteria
(1) Studies with independent variables not involving resistance training; (2) Studies combining resistance training with pharmacological or nutritional interventions; (3) Studies combining resistance training with psychological interventions. (4) Studies involving minors. (5) Animal studies; (6) Studies combining resistance training with other interventions (e.g., blood flow restriction, electrical stimulation, oxygen restriction, or hypoxic environments); (7) Grey literature (e.g., conference papers, theses, and unpublished studies); (8) Studies involving participants with diseases or injuries. (9) Studies involving older adults; (10) Studies focusing on explosive Strength or endurance training; (11) Non-randomized controlled trials.
Methodological quality evaluation
The methodological quality assessment is conducted using Review Manager 5.4.1 software, following the quality assessment criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (version 5.1.0)7. The assessment criteria comprise seven evaluation indicators: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases. The overall evaluation of the risk of bias in the included literature results in three judgment categories: low risk of bias, unclear risk of bias, and high risk of bias. Study quality is classified into three levels based on the number of fulfilled evaluation criteria: Grade A for ≥ 4, Grade B for 2–3, and Grade C for ≤ 1. The methodological quality assessment is performed on the 24 included studies.
Statistical processing
Review Manager 5.3 was used to analyse the outcome measures of the included studies. Given that the outcome measures were continuous variables, we chose the standardised mean difference (SMD) as the effect size metric for statistical analysis. We used the SMD statistic to evaluate effect size, where SMD < 0.2 indicates a very small or negligible effect size, 0.2 ≤ SMD < 0.5 indicates a small effect size, 0.5 ≤ SMD < 0.8 indicates a moderate effect size, SMD ≥ 0.8 indicates a large effect size8. We employed the I2 statistic to test for heterogeneity, with I2 < 40% indicating low heterogeneity, 40% ≤ I2 ≤ 70% indicating moderate heterogeneity, and I2 > 70% indicating high heterogeneity9. We applied fixed-effect models for analyses with no or low heterogeneity and random-effect models for analyses with moderate to high heterogeneity. We conducted subgroup analyses for studies exhibiting substantial heterogeneity9. We used Stata-SE 18.0 for publication bias analysis, setting the significance level at P < 0.05.
Publication bias analysis
In this study, Begg’s test was uniformly applied as the primary method to detect potential publication bias. Begg’s test evaluates the correlation between effect sizes and their standard errors using Kendall’s rank correlation coefficient to identify significant small-study effects, which may indicate publication bias. Significant publication bias was considered present if Begg’s test yielded P < 0.05; otherwise, P > 0.05 indicated no significant bias10.
Results
Study selection
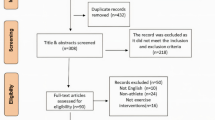
A preliminary search of the database generated 3312 documents. After deleting duplicates, the number of unique records is reduced to 2177. After screening the title, abstract and full text, 24 studies remained. See Fig. 1.
Study characteristics
A detailed overview of participant characteristics and the resistance training programs used in the included studies in Table 1. This study included a total of 587 participants. Among them, 24 studies exclusively assessed male subjects (n = 471), while 9 focused solely on female subjects (n = 164)11,12,13,14,15,16,17,18,19. Additionally, 8 studies included both male and female athletes11,12,14,15,17,18,19,20. Regarding resistance training experience, 7 studies investigated participants with prior resistance training experience21,22,23,24,25,26,27, whereas 17 studies focused on those without resistance training experience11,12,13,14,15,16,17,18,19,20,28,29,30,31,32,33,34. The duration of training interventions ranged from 4 to 14 weeks. Specifically, one study implemented a 7-week training program34, and another conducted a 14-week intervention16. Two studies employed a 10-week program24,31, three studies implemented a 4-week program18,20,29, four studies used a 6-week protocol14,17,21, seven studies adopted an 8-week training program12,15,22,25,27,30,33, and six studies conducted a 12-week intervention11,13,19,23,26,32. Regarding muscle contraction types, concentric and eccentric contractions were applied in 22 studies11,14,21,22,28, while isokinetic and isometric contractions were each utilized in study10,35. Regarding targeted muscle groups, 10 studies focused on upper limb resistance training18,20,23,24,26,29,30,31,32,33, whereas 17 studies investigated lower limb resistance training11,12,13,14,15,16,17,19,21,22,23,25,26,27,28,29,34.
Adaptations of skeletal muscle strength performance to strength training
Meta-analysis of the effects of resistance training on maximal skeletal muscle strength
Twenty studies have reported on the relationship between resistance training and maximum muscle strength23,24,26,30,32,33,34. The heterogeneity analysis showed low heterogeneity (I2 = 17%, P = 0.24). The meta-analysis forest plot is shown in Fig. 2; therefore, a fixed-effects model was employed for the meta-analysis. The results demonstrate that resistance training programs significantly increase participants’ maximum strength [SMD = 0.77, 95% CI (0.57, 0.98), P < 0.05]. The publication bias test results (z = 4.12, P < 0.05) were statistically significant, indicating the presence of publication bias.
Eight studies investigated the relationship between resistance training and squat strength benefits23,26,33,34. The heterogeneity analysis showed low heterogeneity (I2 = 39%, P = 0.12); therefore, a fixed-effects model was applied in the meta-analysis. The results demonstrate that resistance training programs significantly enhance participants’ squat strength [SMD = 1.14, 95% CI (0.79, 1.49), P < 0.05], showing a significant effect. Publication bias is not assessed due to insufficient studies (fewer than 10)8. Twelve studies investigated the relationship between resistance training and bench press strength24,30,32,33. The heterogeneity analysis showed low heterogeneity (I2 = 0%, P = 0.93); therefore, a fixed-effects model was applied for the meta-analysis. The results demonstrate that resistance training programs significantly enhance participants’ bench press strength [SMD = 0.6, 95% CI (0.35, 0.84), P < 0.05], showing a significant effect. The publication bias test results (z = 1.83, P = 0.0671) failed to reach statistical significance, suggesting no substantial evidence of publication bias. The meta-analysis forest plot is shown in Fig. 3.
This study investigates the relationship between resistance training and maximum strength benefits for subjects with prior resistance training experience. Eight studies examined the relationship between resistance training and maximum strength in subjects with prior resistance training experience23,24,26. The heterogeneity analysis revealed moderate heterogeneity (I2 = 50%, P = 0.05), so a random-effects model was applied for the meta-analysis. The results demonstrate that resistance training programs significantly increase maximum strength in subjects with prior resistance training experience [SMD = 1.09, 95% CI (0.62, 1.56), P < 0.05]. After excluding the fourth study, heterogeneity significantly decreased (I2 = 7%, P = 0.37). Although most existing literature has concentrated on comparing training volumes between 1 to 4 sets, research on volumes exceeding four sets, especially within the context of resistance training participants, remains relatively scarce36,37,38. Therefore, adopting excessively high training volumes (e.g., 8 sets) may exceed the adaptive capacity of many individuals, resulting in more significant variation in neuromuscular adaptations between individuals23. Notably, in individuals with extensive resistance training experience, such high training volumes may exacerbate adaptation differences. Given the variability in individuals’ physical conditions, training backgrounds, and recovery capabilities, excessively high training volumes may prevent some individuals from adapting effectively, leading to more significant neurological discrepancies39. This disparity could further amplify inconsistencies in strength improvements and increase heterogeneity within groups, thereby impacting the stability and consistency of subgroup analysis results. Therefore, excluding training groups using 8 sets in subgroup analyses of experienced trainees helps reduce confounding due to differences in training volume, ensuring more consistent and comparable results. After exclusion, the result was [SMD = 0.89, 95% CI (0.55, 1.23), P < 0.05], showing a significant effect. Since fewer than 10 studies were included, publication bias was not assessed. The meta-analysis forest plot is shown in Fig. 4.
Twelve studies examined the relationship between resistance training and maximum strength in subjects without prior resistance training experience30,32,33,34. The heterogeneity analysis showed low heterogeneity (I2 = 0%, P = 0.89); thus, a fixed-effects model was applied for the meta-analysis. The results demonstrate that resistance training programs significantly increase maximum strength in subjects without prior resistance training experience [SMD = 0.64, 95% CI (0.38, 0.89), P < 0.0001]. The publication bias test result (z = 2.00, P < 0.05) was statistically significant, suggesting the presence of publication bias.
Meta-analysis of the effects of resistance training on peak torque
Fifty-four studies examined the relationship between resistance training and peak torque13,14,18,20,25,27,29. The meta-analysis results showed moderate heterogeneity (I2 = 48%, P < 0.05), warranting a random-effects model. The results indicated a significant effect with SMD = 0.77, 95% CI (0.62, 0.93), P < 0.05. The meta-analysis forest plot is presented in Fig. 5. Studies on the effects of resistance training on peak torque exhibited moderate heterogeneity. The publication bias test (z = 6.69, P < 0.001) was statistically significant, suggesting the presence of publication bias. Subgroup analysis indicated that the source of heterogeneity could stem from test sites and testing methods.
To further explore the relationship between resistance training and peak torque at different test sites, 24 studies examined the relationship between resistance training and lower limb peak torque13,14,25,27. Heterogeneity analysis revealed statistically significant moderate heterogeneity (I2 = 44%, P < 0.05), warranting a random-effects model. The results demonstrated that resistance training programs significantly increase lower limb peak torque [SMD = 1.06, 95% CI (0.82, 1.29), P < 0.00001]. The publication bias test (z = 5.08, P < 0.05) was statistically significant, suggesting the presence of publication bias.
Thirty studies examined the relationship between resistance training and upper limb peak torque18,20,27. Heterogeneity analysis revealed statistically significant low heterogeneity (I2 = 35%, P = 0.03), leading to the use of a fixed-effects model. The results demonstrated that resistance training programs significantly increase upper limb peak torque [SMD = 0.54, 95% CI (0.40, 0.69), P < 0.05]. The publication bias test (z = 4.25, P < 0.05) was statistically significant, suggesting the presence of publication bias.
For upper limb peak torque, the intensity ranged from 45 to 85% of 1RM, with repetitions ranging from 8 to failure, mainly focusing on 12 repetitions. The number of sets ranged from 2 to 5, primarily 5 sets. The duration ranged from 8 to 12 weeks, primarily 8 weeks, with training frequency ranging from 2 to 3 sessions per week, mainly 3 sessions. All of these ranges can yield training benefits.
Eleven studies investigated the relationship between peak torque and upper limb concentric testing methods18,20,27. The analysis showed low heterogeneity (I2 = 0%, P = 0.45), leading to using a fixed-effects model for the meta-analysis. Results indicated that concentric contraction testing of upper limb muscles significantly affected peak torque [SMD = 0.69, 95% CI (0.44, 0.93), P < 0.05]. The publication bias test (z = 1.16, P = 0.2458) was not statistically significant, suggesting no publication bias.
Ten studies examined the relationship between peak torque and lower limb concentric testing methods13,14,27. The heterogeneity analysis showed moderate heterogeneity (I2 = 39%, P = 0.10), prompting the use of a fixed-effects model. Results revealed that concentric contraction testing of lower limb muscles significantly influenced peak torque [SMD = 1.18, 95% CI (0.84, 1.52), P < 0.05]. Though the effect was not statistically significant. The publication bias test (z = 6.81, P < 0.05) was statistically significant, indicating potential publication bias.
Ten studies examined the relationship between peak torque and upper limb eccentric testing methods18,27. The heterogeneity analysis showed low heterogeneity (I2 = 0%, P = 0.78), leading to the use of a fixed-effects model. Results indicated that eccentric contraction testing of upper limb muscles significantly affected peak torque [SMD = 0.52, 95% CI (0.27, 0.77), P < 0.05]. The publication bias test (z = 1.67, P = 0.0959) was not statistically significant, suggesting no publication bias.
Six studies explored the relationship between peak torque and lower limb eccentric testing methods14,27. The heterogeneity analysis showed statistically significant moderate heterogeneity (I2 = 49%, P = 0.08), leading to using a random-effects model. Results showed that eccentric contraction testing of lower limb muscles influenced peak torque [SMD = 1.57, 95% CI (1.05, 2.10), P < 0.05]. The results remained statistically significant. Due to the limited number of studies, publication bias was not assessed.
Nine studies examined the relationship between peak torque and upper limb isometric testing methods18. The analysis indicated low heterogeneity (I2 = 0%, P = 1.00), leading to using a fixed-effects model for the meta-analysis. Results showed that isometric contraction testing of upper limb muscles affected peak torque [SMD = 0.19, 95% CI (− 0.08, 0.46), P = 0.17], though the effect was not statistically significant. Due to the limited number of studies, publication bias was not assessed.
Eight studies examined the relationship between peak torque and lower limb isometric testing methods25. The heterogeneity analysis showed low heterogeneity (I2 = 0%, P = 0.90), leading to using a fixed-effects model for the meta-analysis. Results indicated that isometric contraction testing of lower limb muscles significantly affected peak torque [SMD = 0.55, 95% CI (0.25, 0.85), P < 0.05]. Due to the limited number of studies, publication bias was not assessed.
Muscle physiology adaptations to strength training
Meta-analysis of the effects of resistance training on muscle fibres composition
Eighteen studies examined the relationship between resistance training and muscle fibres percentages15,19,31. Meta-analysis revealed high heterogeneity between studies (I2 = 85%, P < 0.05), leading to the use of a random-effects model. The meta-analysis showed no significant effect [SMD = 0.14, 95% CI (− 0.45, 0.74), P = 0.63]. The publication bias test (z = 0.53, P = 0.5959) was not statistically significant, indicating no publication bias. Studies on the effects of resistance training on muscle fibre percentages exhibited moderate heterogeneity. Subgroup analysis suggested that muscle fibre types may be the source of heterogeneity.
The relationship between resistance training and muscle fibre types (Type I, Type IIa, Type IIx) was investigated. The meta-analysis forest plot is shown in Fig. 6. Six studies have reported the relationship between Type I muscle fibres percentage15,19,31. Heterogeneity analysis showed low heterogeneity (I2 = 0%, P = 0.47), so a fixed-effects model was applied in the meta-analysis. The results indicated that resistance training programs increased Type I muscle fibre percentage [SMD = 0.31, 95% CI (− 0.03, 0.65), P = 0.08], although the effect was insignificant. Publication bias was not assessed due to the limited number of studies.
Six studies examined the relationship with Type IIa muscle fibres percentage15,19,31. Heterogeneity analysis revealed low heterogeneity (I2 = 15%, P = 0.32), leading to using a fixed-effects model in the meta-analysis. The results showed that resistance training programs increased Type IIa muscle fibres percentage [SMD = 0.05, 95% CI (− 0.29, 0.39), P = 0.76], with no significant effect. Publication bias was not assessed due to the limited number of studies (fewer than 10). Six studies reported the relationship with Type IIx muscle fibres percentage15,19,31. Heterogeneity analysis indicated low heterogeneity (I2 = 30%, P = 0.21), leading to using a fixed-effects model in the meta-analysis. The results showed that resistance training programs reduced Type IIx muscle fibres percentage [SMD = − 0.52, 95% CI (− 0.96, − 0.17), P = 0.76], though this effect was insignificant. Publication bias was not assessed due to the limited number of studies.
Meta-analysis of resistance training and muscle thickness
Forty studies investigated the relationship between resistance training and muscle thickness. Heterogeneity analysis showed medium heterogeneity (I2 = 53%, P < 0.05), leading to using a Radom-effects model for meta-analysis11,12,14,16,18,22,26,28,29,30,33. The results indicated that resistance training significantly increased skeletal muscle thickness in participants [SMD = 0.28, 95% CI (0.20, 0.69), P < 0.05]. The publication bias test (z = 2.09, P < 0.05) was statistically significant, indicating the presence of publication bias.
To further explore the effect of resistance training on different agonist muscles, subgroup analyses were conducted for the biceps brachii, triceps brachii, pectoralis major, rectus femoris, and vastus lateralis. The forest plot from the meta-analysis is shown in Fig. 7.
Seventeen studies examined the relationship between resistance training and biceps brachii thickness11,18,26,29,33. Heterogeneity analysis showed low heterogeneity (I2 = 0%, P = 0.99), leading to using a fixed-effects model for meta-analysis. The results indicated a significant increase in biceps brachii thickness following resistance training [SMD = 0.61, 95% CI (0.40, 0.83), P < 0.05]. The publication bias test (z = 1.57, P = 0.117) was not statistically significant, indicating no publication bias.
Six studies examined the relationship between resistance training and triceps brachii thickness11,22,26. Heterogeneity analysis showed low heterogeneity (I2 = 0%, P = 0.64), leading to using a fixed-effects model for meta-analysis. The results indicated a significant increase in triceps brachii thickness following resistance training [SMD = 0.65, 95% CI (0.27, 1.03), P < 0.05]. Since there were fewer than 10 studies, publication bias was not assessed.
Six studies examined the relationship between resistance training and pectoralis primary thickness11,30. Heterogeneity analysis showed low heterogeneity (I2 = 0%, P = 0.43), leading to using a fixed-effects model for meta-analysis. The results indicated a significant increase in pectoralis primary thickness following resistance training [SMD = 1.39, 95% CI (0.1.01, 1.77), P < 0.05]. Since there were fewer than 10 studies, publication bias was not assessed.
Six studies examined the relationship between resistance training and vastus lateralis thickness12,16,33. Heterogeneity analysis showed moderate heterogeneity (I2 = 46%, P = 0.10), leading to using a random-effects model for meta-analysis. The results indicated no significant increase in vastus lateralis thickness following resistance training [SMD = 0.07, 95% CI (− 0.31, 0.45), P = 0.72]. Since there were fewer than 10 studies, publication bias was not assessed.
Six studies examined the relationship between resistance training and rectus femoris thickness14,26,28. Heterogeneity analysis showed moderate heterogeneity (I2 = 54%, P = 0.10), leading to using a random-effects model for meta-analysis. The results indicated no significant increase in rectus femoris thickness following resistance training [SMD = 0.61, 95% CI (0.26, 0.97), P = 0.75]. Since there were fewer than 10 studies, publication bias was not assessed.
Meta-analysis of the effect of resistance training on pennation angle
Ten studies examined the relationship between resistance training and pennation angle12,17,28. Heterogeneity analysis showed moderate heterogeneity (I2 = 54%, P < 0.05), leading to using a random-effects model for the meta-analysis. The results suggested that resistance training may increase the pennation angle of skeletal muscles [SMD = 0.36, 95% CI (− 0.02, 0.74), P = 0.06], but the effect was not statistically significant. The forest plot from the meta-analysis is shown in Fig. 8. The publication bias test (z = − 2.71, P < 0.05) was statistically significant, indicating the presence of publication bias.
Neural adaptations to strength training
Meta-analysis of the effect of resistance training on electromyographic (EMG) activity
Twenty-eight studies examined the relationship between resistance training and EMG. Heterogeneity analysis indicated moderate heterogeneity (I2 = 58%, P < 0.05), leading to using a random effects model for the meta-analysis13,14,21,25. The results showed that resistance training programs significantly improved squat strength [SMD = 0.54, 95% CI (0.26, 0.81), P < 0.05]. The publication bias test (z = 5.62, P < 0.05) was statistically significant, indicating the presence of publication bias.
A subgroup analysis explored the effects of resistance training on upper and lower limb EMG, separating muscles into upper and lower limbs. Eighteen studies examined the relationship with upper limb EMG14,25. Heterogeneity analysis showed low heterogeneity (I2 = 3%, P = 0.42), so a fixed-effects model was applied. The results showed a significant effect [SMD = 0.97, 95% CI (0.74, 1.20), P < 0.05]. The publication bias test (z = 3.97, P < 0.001) was statistically significant, indicating the presence of publication bias.
Ten studies examined the relationship with lower limb EMG13,21. Heterogeneity analysis showed low heterogeneity (I2 = 30%, P = 0.17), so a fixed effects model was applied. The results showed a significant effect [SMD = 0.92, 95% CI (0.61, 1.24), P < 0.05]. The publication bias test (z = 3.97, P < 0.001) was statistically significant, indicating the presence of publication bias. Figure 9 shows the forest plots for the effects of resistance training on upper and lower limb EMG.
Discussion
Moderator and subgroup analyses of resistance training effects on maximal strength
According to the results of our meta-analysis, training aimed at enhancing participants’ maximal strength typically employs intensity levels ranging from 60 to 85% of 1RM. The repetition range is generally between 3 and failure, with a primary focus on 10–12 repetitions per set. The number of sets varies from 1 to 8, with the most common range being 3–4. Training durations generally span 6–8 weeks, with a frequency of 1–5 sessions per week, typically 2–3 times. This training protocol has resulted in moderate to substantial improvements in maximal strength. The analysis results indicate that resistance training led to a moderate improvement in maximal muscle strength (SMD = 0.77). The strength assessment methods employed included the one-repetition maximum (1RM) squat test, knee extension test, elbow extension test, and bench press test.
The equipment included the Smith machine, barbells, free weights, and close-grip seated presses. Subgroup analysis was conducted to explore the effects of resistance training on maximal strength in the upper and lower limbs. The results revealed that the strength improvements in the upper limbs were less pronounced than those in the lower limbs, with a significant difference between groups (P < 0.05). Additionally, subgroup analysis was performed to assess the effects of resistance training on participants with and without prior resistance training experience. This study found that participants with resistance training experience significantly improved maximal strength more than those without such knowledge. However, the difference between groups was insignificant (P = 0.24). These results are consistent with previous research, which indicates that resistance training can moderately enhance maximal muscle strength, but responses to the same training stimulus vary across muscle groups. One study comparing single-set and multiple-set resistance training for improvements in upper and lower limb strength and muscle mass found that the increase in lower limb strength was significantly more significant than in the upper limbs, with statistical differences between the two40. This suggests that the lower limbs may be more sensitive to resistance training due to the larger muscle group volume and higher proportion of load-bearing fibres. A similar trend was observed in athletes as well. A previous study on elite male handball players reported that after 8 weeks of heavy resistance training, strength improvements differed between the upper and lower limbs, with the lower limbs showing greater enhancement, further confirming the region-specific nature of muscle adaptation41. In another study, personalized stimulus for different muscle groups is crucial in training programs due to the differences in muscle group responses to training loads and their physiological foundations.
Furthermore, regarding the impact of prior resistance training experience on strength improvements, studies have noted that individuals with previous experience typically experience more significant absolute gains in maximal strength than those with no such experience40. Moreover, research also shows that adaptations in lower limb strength are more pronounced within the resistance training group. In contrast, the impact on upper limbs is less affected by prior resistance training history42.
Moderator and subgroup analyses of resistance training effects on peak torque
The results of this meta-analysis indicate that for upper limb peak torque, the intensity ranged from 45 to 85% of 1RM, with repetitions ranging from 8 to failure, primarily focusing on 12 repetitions. The number of sets ranged from 2 to 5, with 5 being the most used. The duration ranged from 8 to 12 weeks, primarily 8 weeks, with the training frequency ranging from 2 to 3 sessions per week, mainly 3 sessions. This protocol resulted in a moderate to large improvement in the peak torque of the muscles. For lower limb peak torque, the intensity ranged from 30 to 100% of 1RM, with repetitions ranging from 6 to 12 and sets ranging from 2 to 4. The duration ranged from 4 to 8 weeks, primarily 4 weeks. The training frequency ranged from 3 to 5 sessions per week, with 3 being the most common. This protocol also led to moderate to significant improvements in the peak torque of the muscles. The analysis revealed a moderate improvement in peak torque following resistance training (SMD = 0.77). The strength assessment methods employed included concentric contraction, eccentric contraction, isometric contraction, and isokinetic contraction, with a contraction speed of 0.52 rad/s. The equipment included the Smith machine, barbells, free weights, and close-grip seated presses. Subgroup analysis was conducted to investigate the impact of resistance training on peak torque improvement for both upper and lower limbs. The results showed that the improvement in peak torque for the upper limbs was less than that for the lower limbs, with a significant difference between the groups (P < 0.001). Further subgroup analysis examined the effects of concentric contraction on peak torque, revealing that the improvement in upper limb peak torque was lower than that in the lower limbs, with a significant difference between the groups (P < 0.05).
Additionally, subgroup analysis examined the effects of eccentric contraction on peak torque improvement, showing that the improvement in upper limb peak torque was lower than that in the lower limbs, with a significant difference between the groups (P < 0.05). Subgroup analysis was also conducted to assess the effects of isometric contraction on peak torque improvement, and the results showed that the improvement in upper limb peak torque was lower than that in the lower limbs. However, this difference was not statistically significant (P = 0.08). These results are consistent with previous studies, which have shown that when total training volume is matched. However, upper and lower limb muscles experience increased strength and volume. Region-specific responses demonstrate that the lower limbs generate greater torque than the upper limbs during concentric and eccentric contractions43,44. In isometric contraction tests, both upper and lower limbs exhibited neuromuscular adaptations due to resistance training, but the lower limbs showed more significant changes in peak torque45,46.
Moderator and subgroup analyses of resistance training effects on muscle fibres composition
According to the results of our meta-analysis, the intensity required for muscle fibres adaptation for maximal strength typically ranges from 60 to 90% of 1RM, with 3 to 10 repetitions, 2–4 sets, throughout 8 to 12 weeks, and a frequency of 2–3 sessions per week, predominantly 3 times per week. This training protocol can lead to negligible to minor improvements. The analysis revealed that resistance training resulted in a minimal improvement in muscle fibres composition (SMD = 0.14). The assessment method used for muscle fibres composition was the 6 mm Bergstrom percutaneous biopsy needle. Subgroup analysis was conducted to investigate the effects of resistance training on the percentage changes in different muscle fibres types. The results showed that, after resistance training, the percentage of type I muscle fibres was higher than that of type IIa, which was, in turn, higher than that of type IIx, with a statistically significant difference between groups (P < 0.05). Previous studies have demonstrated that resistance or strength training can induce adaptive changes in muscle fibres type composition, specifically characterized by a significant reduction in the proportion of type IIx fibres, accompanied by a shift toward type IIa and even type I fibres. In contrast, the proportion of type I fibres is maintained or slightly increased. This transformation in fibres composition reflects an adaptation of muscle tissue toward more oxidative and fatigue-resistant characteristics, contributing to improvements in maximal strength output47,48,49,50. This fibres remodeling process during training typically manifests as a hierarchical distribution of the type I > type IIa > type IIx fibres, which has been validated by changes in myosin heavy chain (MHC) expression, namely, an increase in MHC IIa expression and a decrease in MHC IIx expression. These prior findings support the results of our analysis47,48,49,50.
Moderator and subgroup analyses of resistance training effects on muscle thickness
According to the results of our meta-analysis, in resistance training protocols where load intensity is expressed as a percentage of maximal voluntary contraction (MVC), the training intensity predominantly ranged from 60 to 100% MVC, with repetitions mostly between 6 and 10, sets ranging from 1 to 5, a frequency of 2 to 5 sessions per week, and training duration varying from 4 to 14 weeks. In contrast, protocols using repetition maximum (RM) to express load intensity mainly focused on a range of 70% to 80% 1RM (approximately 8–12 RM), with repetitions between 6 and 12, sets typically between 3 and 6, a frequency of 1 to 3 sessions per week, and a training duration primarily concentrated between 8 and 12 weeks. These protocols led to a small effect size improvement in the overall muscle thickness of both upper and lower limbs. The analysis showed a small improvement following resistance training (SMD = 0.28). The assessment methods for muscle thickness included B-mode ultrasonography (probe frequency range: 2.5–7.5 MHz) and high-resolution magnetic resonance imaging (repetition time: 520 ms; echo time: 20 ms).
Subgroup analysis in this study explored the effects of resistance training on the thickness of different muscles. Results indicated that post-training increases in the thickness of the pectoralis major > triceps brachii > biceps brachii, and rectus femoris > vastus lateralis, with statistically significant differences between groups (P < 0.05). Previous studies have demonstrated that maximal strength training promotes muscle hypertrophy by imposing high mechanical tension, thereby increasing muscle cross-sectional area51,52. Among upper limb muscle groups, compound strength training exercises such as the bench press primarily activate the pectoralis major, the central agonist in pushing movements and are subjected to substantial mechanical load and contraction amplitude. Consequently, the hypertrophic response of the pectoralis major is significantly greater than that of other synergistic muscles.
Simultaneously, although subjected to less mechanical tension than the pectoralis major, the triceps brachii, as a synergist in extension during pushing actions, still participates actively in movement execution, leading to a notable hypertrophic effect. In contrast, the biceps brachii, which mainly contributes to pulling and auxiliary movements, is less activated during pushing-type training, resulting in comparatively limited hypertrophic adaptation53,54. For lower limb muscles, multi-joint compound exercises such as squats can significantly trigger the entire quadriceps group; however, functional differentiation among the muscle heads leads to variability in hypertrophic responses. For example, the rectus femoris possesses unique anatomical and physiological advantages in coordinating knee extension. Under maximal strength training conditions, its hypertrophic reaction is often more significant than the vastus lateralis. This can be partially attributed to the rectus femoris being a biarticular muscle crossing both the hip and knee joints. It requires it to undertake more complex roles in stability control and force transmission during training, making it more prone to adaptive enlargement under high mechanical tension55,56.
Moderator and subgroup analyses of resistance training effects on pennation angle
According to the findings of our meta-analysis, studies employing training intensity expressed as a percentage of maximal voluntary contraction (MVC) predominantly utilized load intensities ranging from 60 to 100% MVC, with 5–10 repetitions per set, 3–5 sets per session, a training frequency of 2 to 3 sessions per week, and intervention durations spanning 6 to 8 weeks. In contrast, training protocols quantifying intensity by repetition maximum (RM) primarily applied loads corresponding to 70% to 80% of one-repetition maximum (approximately equivalent to 8–12 RM)57, with 3 to 5 repetitions per set, performed twice weekly over 6 weeks. Such training parameters were found to elicit a moderate effect on enhancements in muscle pennation angle. Quantitative synthesis revealed a small but statistically significant effect size in the pennation angle following resistance training (SMD = 0.36). Muscle architectural assessments were conducted using B-mode ultrasonography. Previous literature has demonstrated that increases in muscle thickness are often accompanied by corresponding increases in pennation angle among adults, suggesting that resistance training induces muscular hypertrophy and facilitates the reorganization of intramuscular architecture58. Comparative analyses between resistance-trained individuals and untrained controls have corroborated these findings, with trained participants exhibiting significantly more significant increases in pennation angle59. Moreover, existing evidence suggests that changes in pennation angle frequently occur in tandem with alterations in muscle thickness and other architectural variables, such as muscle fascicle length. The extent to which pennation angle modifications contribute to maximal strength improvements appears to be muscle-specific and may vary based on the nature of the resistance training stimulus58,59,60. Consequently, changes in pennation angle should be interpreted as part of a multifactorial adaptation process rather than in isolation.
Moderator and subgroup analyses of resistance training effects on muscle activation (RMS values)
According to the results of the meta-analysis, studies employing training intensity based on the percentage of maximal voluntary contraction (MVC) typically involved 1 to 5 sets of 6 to 15 repetitions, with a training frequency of 2–3 sessions per week and an intervention duration ranging from 6 to 12 weeks. In resistance training protocols where the intensity was expressed in terms of repetition maximum (RM), training loads generally ranged from 60 to 80% of one-repetition maximum (1RM), with 6–12 repetitions per set, 2–5 sets per session, and training frequencies of 2 to 3 times per week over a 6–12-week period. The analysis demonstrated a small effect size for changes in the root mean square (RMS) muscle activity values following resistance training (SMD = 0.49). RMS values were assessed using surface electromyography sensors, specifically the Bagnoli 16 EMG system, with a sampling rate of 1,000 Hz and a band-pass filter set between 10 and 500 Hz in this study. Subgroup analyses were conducted to examine the effects of resistance training on RMS values in upper versus lower limb muscles. Results indicated that the improvement in RMS values was more significant in upper limb muscles than in lower limb muscles; however, the difference was not statistically significant (P = 0.81). Previous studies have reported that upper limb muscles exhibit higher levels of electrophysiological activation during resistance training, which may be attributed to differences in muscle fiber composition, neural drive, and the anatomical and physiological characteristics of movement patterns61. Furthermore, earlier research has explored bilateral and ipsilateral coupling effects in muscle activation across upper and lower limbs, revealing that voluntary contractions of upper limb muscles during specific motor tasks are often accompanied by enhanced neural activation and muscle recruitment. This phenomenon may help explain the more pronounced increases in RMS values observed in upper limb muscles following resistance training62.
Physiological mechanisms of strength training in enhancing maximal skeletal muscle strength
Strength training affects muscle adaptation by altering factors like muscle thickness, pennation angle, and muscle fibre composition, with skeletal muscle fibres classified into three types: Type I, Type IIa, and Type IIx. Increasing maximal strength involves various physiological adaptations, such as greater muscle thickness, changes in pennation angle, and shifts in muscle fibre types. The following sections will detail how these adaptations influence maximal strength development, supported by research explaining the underlying physiological mechanisms.
Increase in muscle thickness: One of the most prominent adaptations to strength training is muscle hypertrophy, which increases muscle thickness. Strength training induces micro-damage in muscle fibres, activating satellite cells that fuse with existing fibres, increasing their cross-sectional area63. Research shows that muscle cross-sectional area positively correlates with maximal strength, and increased muscle thickness directly enhances strength performance64,65.
Strength training also activates the mTOR (mammalian target of rapamycin) signalling pathway, which is crucial for protein synthesis and muscle growth. mTOR activation promotes the expression of muscle synthesis-related genes66,67, enhancing hypertrophy and increasing maximal strength. Changes in pennation angle: The pennation angle, or the angle between muscle fibres and tendons, affects muscle lever arm and contraction efficiency68. While the increase in pennation angle after strength training is minimal, it still has some impact on strength output. A larger pennation angle allows more muscle fibres to contract simultaneously, increasing muscle force output and overall strength capacity69. However, pennation angle adaptation is relatively tiny because significant changes require long-term training. Studies show that pennation angle adaptations are not evident in early training stages. As training progresses, adaptations mainly focus on shifts in muscle fibre types and increases in muscle thickness70.
Shifts in muscle fibre types: Strength training significantly influences muscle fibre types, a key factor in developing maximal strength. After strength training, Type I fibres increase, Type IIa fibres slightly increase, and Type IIx fibres significantly decrease. These changes reflect muscle adaptations to high-intensity training, enhancing strength output capacity71,72. The increase in Type I fibres, known for their endurance and durability, helps athletes exert strength more effectively during high-intensity training. Strength training activates growth factors like IGF-1 (insulin-like growth factor), promoting muscle growth and repair and increasing the number and function of Type I fibres73. Additionally, strength training boosts mitochondrial content in muscles, enhancing aerobic metabolism. Given Type I fibbers’ high aerobic capacity, strength training improves mitochondrial function to meet their energy demands, supporting their growth and function74.
Another crucial physiological mechanism is the transformation of muscle fibre types. Type IIa fibres, which are fast-twitch by nature, are characterised by high strength output and endurance. During strength training, Type IIb fibres can partially convert to Type IIa fibres to meet training demands. Factors like training intensity, frequency, and duration influence this conversion. Research shows that systematic strength training significantly boosts Type IIb-to-Type IIa conversion, increasing the proportion of Type IIa fibres75. The significant decrease in Type IIx fibres likely occurs due to their susceptibility to fatigue in high-intensity training, leading the body to retain fibres with better endurance and strength. Type IIx fibres primarily contribute to short-term, high-intensity bursts of power but have poor fatigue resistance. Hence, during maximal strength adaptation, the body retains Type I and Type IIa fibres to enhance overall strength performance39,76. Strength training promotes maximal strength development by increasing muscle thickness, adjusting the pennation angle, and altering muscle fibre types.
In the adaptation process of maximal strength training, the root mean square (RMS) value of skeletal muscle electromyographic signals is commonly used as a key electrophysiological indicator to assess muscle electrical activity intensity and neural drive levels57. A significant increase in RMS values generally indicates an enhanced number of active motor units, improved neural drive efficiency, and greater control by the central nervous system39,48.
As a high-intensity, high-load training modality, maximal strength training induces multidimensional adaptations in the neuromuscular system. First, training promotes the recruitment of more high-threshold motor units, which primarily innervate fast-twitch muscle fibers (Type II fibers). These fibers have higher excitation thresholds and contraction capabilities, enabling them to quickly generate large amounts of tension, serving as the key neural foundation for maximal strength gains48. Second, training increases the discharge frequency of motor units, causing action potentials to be transmitted to muscle fibers faster, thereby enhancing muscle contractions’ stability and endurance50. Additionally, synchronization of discharge between motor units is improved, resulting in the coordinated activation of multiple muscle fibers and generating a greater resultant force within a given time, further enhancing overall muscle output51.
Moreover, regarding central regulatory mechanisms, training significantly enhances the excitability of the motor cortex, corticospinal tract, and spinal anterior horn neurons, allowing for more rapid and efficient transmission of neural impulses along the central-peripheral pathway. This increased excitability implies that the nervous system responds to training stimuli with greater sensitivity and precision, facilitating quicker and more extensive recruitment of target muscle groups to complete movements48. Therefore, the elevation of RMS values reflects increased muscle excitability and serves as a comprehensive indicator of neuromuscular adaptation, reflecting improved neural control, muscle functional integration, and synergistic efficiency of muscle groups following strength training54,55.
Conclusion
This meta-analysis aimed to assess the effects of resistance training on maximal strength, peak torque, muscle fibres composition, muscle thickness, pennation angle, and electromyographic signals (RMS values). The analysis results indicate that resistance training significantly affected most of the measured outcomes, particularly in maximal strength and peak torque, where resistance training significantly improved (SMD = 0.77). In comparing the upper and lower limbs, the improvements in the lower limbs were substantially more significant than those in the upper limbs, with statistical differences between the groups (P < 0.05).
Regarding changes in muscle fibres composition, after resistance training, the percentage of Type I muscle fibres increased, and the changes in Type I fibres were significantly more significant than those in Type IIa and Type IIx fibres, with statistically significant differences between the groups (P < 0.05). Although the overall effect on muscle thickness was relatively small (SMD = 0.28), resistance training had a more pronounced effect on the pectoralis major and rectus femoris (P < 0.05), suggesting that resistance training can effectively promote muscle hypertrophy in both the upper and lower limbs, particularly in the pectoralis major and rectus femoris.
Regarding electromyographic signals (RMS values), resistance training also showed a small effect size improvement (SMD = 0.49), with a substantially more significant improvement in RMS values for the upper limbs than the lower limbs. However, the inter-group difference was insignificant (P = 0.81). Additionally, the change in pennation angle (SMD = 0.36) indicates that resistance training promotes muscle structural adaptation.
Overall, this study provides strong evidence for the adaptive effects of resistance training on maximal strength, peak torque, muscle fibres composition, muscle thickness, pennation angle, and electromyographic signals, supporting resistance training as an effective intervention for enhancing athletic performance and muscle structure.
Limitations of the study
In this study, differences in training experience were considered a potential limiting factor. Although participants were categorised into three groups—those with no training experience, those with physical activity experience, and those with training experience—it is important to note that individuals within the “training experience” category may differ significantly. Specifically, training experience can be further subdivided into multiple levels, such as "highly experienced," "moderately experienced," and "slightly experienced." These different levels of training experience may have varying degrees of impact on training outcomes. For instance, individuals with “highly experienced” training backgrounds typically exhibit higher strength levels and better motor skills, and their physiological adaptation mechanisms may differ significantly from those of individuals with “slightly experienced” or “moderately experienced” backgrounds. Therefore, differences in the levels of training experience may influence research results concerning strength gains and neuromuscular adaptations to some extent.
To minimise the potential impact of training experience differences on the meta-analysis results, future studies should further refine the categorisation of training experience when grouping participants and ensure that different levels of training experience are described in sufficient detail. Researchers should specify participants’ strength levels or athletic grades to allow for a more precise assessment of the effect of training experience on training outcomes. Additionally, adopting a unified and standardised classification system for training experience will help improve the consistency and comparability of studies and enhance the external validity and interpretability of results.
In conclusion, future research should pay particular attention to the impact of training experience levels on training outcomes, ensuring that this factor is adequately considered in participant selection and research design. This will provide a stronger theoretical basis for the personalised design of training programs and further promote the precision and broad applicability of research in training.
Practical applications
This study focuses on resistance training interventions explicitly aimed at the development of maximal strength, revealing the heterogeneous adaptations between upper and lower limb muscle groups. The findings indicate that different muscle groups exhibit significantly distinct adaptive mechanisms in response to the same training loads. This suggests that training programs to enhance maximal strength should fully account for various muscle groups’ structural properties and functional roles. Training content, intensity, and periodization for the upper and lower limbs should be differentially regulated following athletes’ specific needs and individualized goals. These results align with the concept of specificity of adaptation77, emphasizing that maximal strength training should not adopt a one-size-fits-all model but rather pursue precise interventions based on divergent response mechanisms to improve training efficiency and outcome conversion.
Furthermore, the study found that resistance training targeting maximal strength can induce a mild increase in the proportion of type I muscle fibers. In contrast, its effect on transforming type IIa and IIx fibers remains limited. Although fiber-type transitions play a role in maximal strength adaptations, the effect sizes are relatively small. These adaptations are more closely related to improvements in muscular endurance and fatigue resistance rather than directly enhancing neuromuscular force production. Therefore, when designing strength-focused training interventions, the pre-existing muscle fiber composition of the athlete should be taken into account to match appropriate training loads and modalities, thereby increasing the specificity and precision of the intervention.
Theoretically, improvements in maximal strength are driven by the synergistic interaction between neural regulation mechanisms and morphological adaptations in muscle structure. In the early stages of training, strength gains are primarily attributed to neural adaptations, including reduced motor unit recruitment thresholds, increased firing rates, and enhanced synchronization of motor units. Muscle cross-sectional area (CSA) increases as training continues, and structural remodeling becomes dominant. Consequently, the design of training programs aimed at enhancing maximal strength should be based on the principle of neuro-muscular dual adaptation1, with scientifically structured periodization and loading strategies to ensure dynamic coordination and mutual reinforcement between neural stimuli and structural loading.
In terms of practical application, this study provides theoretical support and operational guidance for addressing a long-standing question in resistance training: effectively promoting maximal strength gains through evidence-based training methods. While training intensity and periodization remain foundational to program success, individual factors such as training history, sport-specific demands, and muscle group function also play critical roles in modulating adaptive outcomes. Therefore, it is recommended that coaches and athletes follow evidence-based training principles and draw on the empirical data presented in this study to construct highly individualized and task-oriented maximal strength programs. Avoiding standardized training templates can improve interventions’ specificity, adaptability, and effectiveness. Overall, this study enriches the theoretical framework of maximal strength training and provides actionable evidence to support optimized resistance training strategies focused on maximal strength development.
Data availability
The datasets generated and analysed during the current study are provided in Appendix 1. No additional data are available.
References
Sale, D. G. Neural adaptation to resistance training. Med. Sci. Sports Exerc.20(5 Suppl), S135–S145. https://doi.org/10.1249/00005768-198810001-00009 (1988).
Liu, Yu. et al. Biomechanical basis of muscle strength and muscle strength diagnosis. Taiwan Normal Univ. Sports Res.2, 151–179 (1996).
Semmler, J. G., Kornatz, K. W., Dinenno, D. V., Zhou, S. & Enoka, R. M. Motor unit synchronisation is enhanced during slow lengthening contractions of a hand muscle. J. Physiol.545(2), 681–695. https://doi.org/10.1113/jphysiol.2002.026948 (2002).
Duchateau, J. & Enoka, R. M. Neural control of shortening and lengthening contractions: Influence of task constraints. J. Physiol.586(24), 5853–5864. https://doi.org/10.1113/jphysiol.2008.160747 (2008).
Duclay, J. & Martin, A. Evoked H-reflex and V-wave responses during maximal isometric, concentric, and eccentric muscle contraction. J. Neurophysiol.94(5), 3555–3562. https://doi.org/10.1152/jn.00348.2005 (2005).
Hass, C. J., Garzarella, L., De Hoyos, D. & Pollock, M. L. Single versus multiple sets in long-term recreational weightlifters. Med Sci Sports Exerc32(1), 235–242 (2000).
Higgins, J. P., & Green, S. Cochrane handbook for systematic reviews of interventions[M]. Cochrane Collaboration: 4. (2011).
Tanner-Smith, E. E., Durlak, J. A. & Marx, R. A. Empirically based mean effect size distributions for universal prevention programs targeting school-aged youth: A review of meta-analyses. Prev. Sci.: Off. J. Soc. Prev. Res.19(8), 1091–1101. https://doi.org/10.1007/s11121-018-0942-1 (2018).
Higgins, J. P., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ (Clin. Res. ed.)327(7414), 557–560. https://doi.org/10.1136/bmj.327.7414.557 (2003).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ (Clin. Res. Ed.)315(7109), 629–634. https://doi.org/10.1136/bmj.315.7109.629 (1997).
Abe, T., DeHoyos, D. V., Pollock, M. L. & Garzarella, L. Time course for strength and muscle thickness changes following upper and lower body resistance training in men and women. Eur. J. Appl. Physiol.81, 174–180 (2000).
Alegre, L. M., Ferri-Morales, A., Rodriguez-Casares, R. & Aguado, X. Effects of isometric training on the knee extensor moment–angle relationship and vastus lateralis muscle architecture. Eur. J. Appl. Physiol.114, 2437–2446 (2014).
Botton, C. E. et al. Neuromuscular adaptations to unilateral vs. bilateral strength training in women. J. Strength Condit. Res.30(7), 1924–1932 (2016).
Cadore, E. L. et al. Muscle conduction velocity, strength, neural activity, and morphological changes after eccentric and concentric training. Scand. J. Med. Sci. Sports24(5), e343–e352. https://doi.org/10.1111/sms.12186 (2014).
Hostler, D. et al. Skeletal muscle adaptations in elastic resistance-trained young men and women. Eur. J. Appl. Physiol.86, 112–118 (2001).
Ullrich, B. et al. Neuromuscular responses to 14 weeks of traditional and daily undulating resistance training. Int. J. Sports Med.36(07), 554–562 (2015).
Wilson, M. T. et al. Enhanced skeletal muscle contractile function and corticospinal excitability precede strength and architectural adaptations during lower-limb resistance training. Eur. J. Appl. Physiol.123(9), 1911–1928 (2023).
Yoshida, R., Murakami, Y., Kasahara, K. & Nakamura, M. Minimum intensity of daily six eccentric contractions to increase muscle strength and size. Scand. J. Med. Sci. Sportshttps://doi.org/10.1111/sms.14683 (2024).
Sterczala, A. J. et al. Skeletal muscle adaptations to high-intensity, low-volume concurrent resistance and interval training in recreationally active men and women. Physiol. Rep.12(6), e15953. https://doi.org/10.14814/phy2.15953 (2024).
Nuzzo, J. L., Barry, B. K., Jones, M. D., Gandevia, S. C. & Taylor, J. L. Effects of four weeks of strength training on the corticomotoneuronal pathway. Med Sci Sports Exerc49(11), 2286–2296 (2017).
Duan, Z. & Chen, P. Comparison of the IEMG amplitude of quadriceps and hamstring before and after 6-week dynamic resistance training in trained and untrained individuals. Chin. J. Sports Med.03, 251–254 (2009).
Brigatto, F. A. et al. Effect of resistance training frequency on neuromuscular performance and muscle morphology after 8 weeks in trained men. J. Strength Cond. Res.33(8), 2104–2116. https://doi.org/10.1519/JSC.0000000000002563 (2019).
Marshall, P. W., McEwen, M. & Robbins, D. W. Strength and neuromuscular adaptation following one, four, and eight sets of high intensity resistance exercise in trained males. Eur. J. Appl. Physiol.111, 3007–3016 (2011).
Martínez-Cava, A. et al. Bench press at full range of motion produces greater neuromuscular adaptations than partial executions after prolonged resistance training. J. Strength Cond. Res.36(1), 10–15 (2022).
Remaud, A., Cornu, C. & Guével, A. Neuromuscular adaptations to 8-week strength training: isotonic versus isokinetic mode. Eur. J. Appl. Physiol.108(1), 59–69 (2010).
Schoenfeld, B. J., Peterson, M. D., Ogborn, D., Contreras, B. & Sonmez, G. T. Effects of low-vs high-load resistance training on muscle strength and hypertrophy in well-trained men. J. Strength Cond. Res.29(10), 2954–2963 (2015).
Vuk, S., Damjan, B. & Ivankovic, M. Effects of short and long inter-set rest on maximal isokinetic strength at slow and fast angular velocities in trained young males after eight weeks of resistance training. Kinesiology56(1), 117–127 (2024).
Ema, R., Saito, I. & Akagi, R. Neuromuscular adaptations induced by adjacent joint training. Scand. J. Med. Sci. Sports28(3), 947–960 (2018).
Jenkins, N. D. et al. Neuromuscular adaptations after 2 and 4 weeks of 80% versus 30% 1 repetition maximum resistance training to failure. J. Strength Cond. Res.30, 2174–2185 (2016).
Chaves, S. F. N. et al. Effects of horizontal and incline bench press on neuromuscular adaptations in untrained young men. Int. J. Exer. Sci.13(6), 859–872. https://doi.org/10.70252/FDNB1158 (2020).
Paddon-Jones, D., Leveritt, M., Lonergan, A. & Abernethy, P. Adaptation to chronic eccentric exercise in humans: The influence of contraction velocity. Eur. J. Appl. Physiol.85, 466–471 (2001).
Ogasawara, R., Yasuda, T., Sakamaki, M., Ozaki, H. & Abe, T. Effects of periodic and continued resistance training on muscle CSA and strength in previously untrained men. Clin. Physiol. Funct. Imaging31(5), 399–404 (2011).
Zaroni, R. S. et al. High resistance-training frequency enhances muscle thickness in resistance-trained men. J. Strength Cond. Res.33(Suppl 1), S140–S151. https://doi.org/10.1519/JSC.0000000000002643 (2019).
Baruah, J., Autade, S. S. & Thapa, R. K. Effects of complex-contrast versus traditional resistance training on linear sprint, change-of-direction speed, jumps, and maximal strength of physically active males. Int. J. Kinesiol. Sports Sci.12(2), 1–10. https://doi.org/10.7575/aiac.ijkss.v.12n.2p.1 (2024).
Rønnestad, B. R. et al. Dissimilar effects of one- and three-set strength training on strength and muscle mass gains in upper and lower body in untrained subjects. J. Strength Cond. Res.21(1), 157–163. https://doi.org/10.1519/00124278-200702000-00028 (2007).
Ostrowski, K. J., Wilson, G. J., Weatherby, R., Murphy, P. W. & Lyttle, A. D. The effect of weight training volume on hormonal output and muscular size and function. J. Strength Cond. Res.11(3), 148–154 (1997).
Rhea, M. R., Alvar, B. A., Ball, S. D. & Burkett, L. N. Three sets of weight training superior to 1 set with equal intensity for eliciting strength. J. Strength Cond. Res.16(4), 525–529 (2002).
Hortobagyi, T. et al. Adaptive responses to muscle lengthening and shortening in humans. J. Appl. Physiol.80, 765–772 (1996).
Wilson, J. M. et al. The effects of endurance, strength, and power training on muscle fiber type shifting. J. Strength Cond. Res.26(6), 1724–1729. https://doi.org/10.1519/jsc.0b013e318234eb6f (2012).
Hermassi, S., Chelly, M. S., Tabka, Z., Shephard, R. J. & Chamari, K. Effects of 8-week in-season upper and lower limb heavy resistance training on the peak power, throwing velocity, and sprint performance of elite male handball players. J. Strength Cond. Res.25(9), 2424–2433. https://doi.org/10.1519/jsc.0b013e3182030edb (2011).
Bartolomei, S., Hoffman, J. R., Stout, J. R. & Merni, F. Effect of lower-body resistance training on upper-body strength adaptation in trained men. J. Strength Cond. Res.32(1), 13–18. https://doi.org/10.1519/jsc.0000000000001639 (2018).
Karagiannis, C. et al. The effects of upper limb exercise training on upper limb muscle strength in people with chronic obstructive pulmonary disease: a systematic review and meta-analysis of randomized controlled trials. Ther. Adv. Respir. Dis.17, 17534666231170812. https://doi.org/10.1177/17534666231170813 (2023).
Chalari, E., Methenitis, S., Arnaoutis, G., Stergiou, I. Ε., Kampouropoulou, C., Karampelas, E., & Nomikos, T. Different kinetics of oxidative stress and inflammatory markers after eccentric exercise in upper and lower limbs. In The 9th Conference of Biochemistry and Physiology of Exercise 17 (2019). https://doi.org/10.3390/proceedings2019025017
Boulton, D., Green, S., Macefield, V. G. & Taylor, C. E. A comparison of muscle sympathetic nerve activity to non-contracting muscle during isometric exercise in the upper and lower limbs. Front. Neurosci.https://doi.org/10.3389/fnins.2019.00341 (2019).
Edholm, P., Strandberg, E. & Kadi, F. Lower limb explosive strength capacity in elderly women: Effects of resistance training and healthy diet. J. Appl. Physiol.123(1), 190–196. https://doi.org/10.1152/japplphysiol.00924.2016 (2017).
Eunsook, S., So, J. & Kang, S. Assessments of muscle fibre distribution by non-invasive methods. Int. J. Appl. Sports Sci.32, 66–74 (2020).
Mogi, Y. & Wakahara, T. Effects of growth on muscle architecture of knee extensors. J. Anat.241(3), 683–691. https://doi.org/10.1111/joa.13711 (2022).
Martinez-Canton, M. et al. Role of CaMKII and sarcolipin in muscle adaptations to strength training with different levels of fatigue in the set. Scand. J. Med. Sci. Sports31, 103–191 (2020).
Williamson, D. L., Gallagher, P. M., Carroll, C. C., Raue, U. & Trappe, S. Reduction in hybrid single muscle fiber proportions with resistance training in humans. J. Appl. Physiol.91(5), 1955–1961. https://doi.org/10.1152/jappl.2001.91.5.1955 (2001).
Abe, T. et al. Skeletal muscle size and circulating IGF-1 are increased after two weeks of twice daily “KAATSU” resistance training. Int. J. Kaatsu Train. Res.1, 6–12 (2005).
Krzysztofik, M., Wilk, M., Wojdała, G. & Gołaś, A. Maximizing muscle hypertrophy: a systematic review of advanced resistance training techniques and methods. Int. J. Environ. Res. Public Health16(24), 4897. https://doi.org/10.3390/ijerph16244897 (2019).
Ogasawara, R., Thiebaud, R. S., Loenneke, J. P., Loftin, M. & Abe, T. Time course for arm and chest muscle thickness changes following bench press training. Interv. Med. Appl. Sci.4(4), 217–220. https://doi.org/10.1556/imas.4.2012.4.7 (2012).
Ferreira, D. V., Gentil, P., Soares, S. & Bottaro, M. Recovery of pectoralis major and triceps brachii after bench press exercise. Muscle Nerve56(5), 963–967. https://doi.org/10.1002/mus.25541 (2017).
Bloomquist, K. et al. Effect of range of motion in heavy load squatting on muscle and tendon adaptations. Eur. J. Appl. Physiol.113(8), 2133–2142. https://doi.org/10.1007/s00421-013-2642-7 (2013).
McMahon, G., Morse, C. I., Burden, A., Winwood, K. & Onambele-Pearson, G. Impact of range of motion during ecologically valid resistance training protocols on muscle size, subcutaneous fat, and strength. J. Strength Cond. Res.28(1), 245–255. https://doi.org/10.1519/jsc.0b013e318297143a (2014).
Hill, M., Roberts, M., Price, M. & Kay, A. D. Effects of flywheel training with eccentric overload on standing balance, mobility, physical function, muscle thickness, and muscle quality in older adults. J. Strength Cond. Res.36(11), 3190–3199. https://doi.org/10.1519/jsc.0000000000004006 (2021).
Folland, J. P. & Williams, A. G. The adaptations to strength training: Morphological and neurological contributions to increased strength. Sports Med. (Auckland, N.Z.)37(2), 145–168. https://doi.org/10.2165/00007256-200737020-00004 (2007).
Fukutani, A. & Kurihara, T. Comparison of the muscle fascicle length between resistance-trained and untrained individuals: Cross-sectional observation. Springerplushttps://doi.org/10.1186/s40064-015-1133-1 (2015).
Aagaard, P. et al. A mechanism for increased contractile strength of human pennate muscle in response to strength training: changes in muscle architecture. J. Physiol.https://doi.org/10.1111/j.1469-7793.2001.t01-1-00613.x (2001).
Takarada, Y. et al. Effects of resistance exercise combined with moderate vascular occlusion on muscular function in humans. J. Appl. Physiol.88(6), 2097–2106. https://doi.org/10.1152/jappl.2000.88.6.2097 (2000).
Huang, H. J. & Ferris, D. P. Upper and lower limb muscle activation is bidirectionally and ipsilaterally coupled. Med. Sci. Sports Exer.41(9), 1778–1789. https://doi.org/10.1249/mss.0b013e31819f75a7 (2009).
Schoenfeld, B. J. The mechanisms of muscle hypertrophy and their application to resistance training. J. Strength Cond. Res.24(10), 2857–2872 (2010).
Chargé, S. B. & Rudnicki, M. A. Cellular and molecular regulation of muscle regeneration. Physiol. Rev.84(1), 209–238. https://doi.org/10.1152/physrev.00019.2003 (2004).
Pan, D., Liu, G. & Jin, Q. Effects of “slow training” movements at maximum repetitions on quadriceps strength endurance. J. Strength Cond. Sci.3(5), 86. https://doi.org/10.33142/jscs.v3i5.10193 (2024).
Pei, C. Research on physical training methods for professional ice hockey players. J. Strength Cond. Sci.2(6), 35. https://doi.org/10.33142/jscs.v2i6.7500 (2022).
Bodine, S. C. et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat. Cell Biol.3(11), 1014–1019. https://doi.org/10.1038/ncb1101-1014 (2001).
Zheng, L.-F., Chen, P.-J. & Xiao, W.-H. Signaling pathways controlling skeletal muscle mass. Acta Physiol. Sin.71(4), 671–679. https://doi.org/10.13294/j.aps.2019.0021 (2019).
Betts, J. A., Beelen, M., Stokes, K., Loon, L. J. & v.,. Endocrine responses during overnight recovery from exercise: impact of nutrition and relationships with muscle protein synthesis. Int J Sport Nutr Exerc. Metab.21(5), 398–409. https://doi.org/10.1123/ijsnem.21.5.398 (2011).
Zhang, Q. Muscle fibres types and athletic performance. Biol. Bull.7, 20–21 (1987).
Wang, Z. Effects of eccentric trapezius training on shoulder joint stability in volleyball players. Financ. Manag.2(6), 187. https://doi.org/10.26549/cjygl.v2i6.847 (2018).
Li, S., Wang, Y., Teng, Y., Wang, X. & Liang, S. Effects of core stability training on the whipping kick combination ability in Sanda athletes. J. Educ. Technol. Innov.https://doi.org/10.61414/jeti.v5i3.132 (2023).
Xu, Y., Qu, H., Chang, Y. & Duan, X. Application of metal-organic frameworks in gas pre-concentration, pre-separation and detection. Acta Chim. Sin.80(8), 1183. https://doi.org/10.6023/a22030134 (2022).
Cao, L., Huang, L. & Zhou, S. Research progress on the effects of insulin-like growth factor-I on skeletal muscle growth and repair. J. Tianjin Inst. Phys. Educ.3, 242–245 (2006).
Kovacheva, E., Hikim, A. P., Shen, R., Sinha, I. & Sinha-Hikim, I. Testosterone supplementation reverses sarcopenia in aging through regulation of myostatin, c-Jun NH2-terminal kinase, Notch, and Akt signaling pathways. Endocrinology151(2), 628–638 (2010).
Liu, J. & Zhou, P. A novel myoelectric pattern recognition strategy for hand function restoration after incomplete cervical spinal cord injury. IEEE Trans. Neural Syst. Rehabil. Eng.21(1), 96–103. https://doi.org/10.1109/tnsre.2012.2218832 (2013).
Bogdanis, G. C. Effects of physical activity and inactivity on muscle fatigue. Front. Physiol.https://doi.org/10.3389/fphys.2012.00142 (2012).
Coffey, V. G. & Hawley, J. A. Concurrent exercise training: Do opposites distract?. J. Physiol.595(9), 2883–2896. https://doi.org/10.1113/jp272270 (2016).
Funding
Public, commercial, or not-for-profit funding agencies have received no specific grant.
Author information
Authors and Affiliations
Contributions
W.C.R.: Manuscript writing and literature selection.; K.G.S.: Literature selection and study design. S.S., H.C.M., X.R.Z.: Literature analysis and table preparation; Y.Z.: Grammar checking and formatting.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rong, W., Geok, S.K., Samsudin, S. et al. Effects of strength training on neuromuscular adaptations in the development of maximal strength: a systematic review and meta-analysis. Sci Rep 15, 19315 (2025). https://doi.org/10.1038/s41598-025-03070-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-03070-z
Keywords
This article is cited by
-
Effects of mHealth interventions to prescribe resistance training: a systematic review and meta-analysis of randomized controlled trials
International Journal of Behavioral Nutrition and Physical Activity (2025)
-
Effect of resistance training on kinetic and kinematic indicators in jump athletes: a systematic review
BMC Sports Science, Medicine and Rehabilitation (2025)