Abstract
Up to 25-35% of patients with inflammatory bowel disease (IBD) suffer from anxiety or depression. Mood disorders are correlated with activated inflammatory response. However, changes of inflammation-related proteins in IBD patients with anxiety or depression disorders are still unclear. We aimed to depict the plasma proteomics characteristics of IBD patients with anxiety or depression. Adult patients diagnosed with IBD were prospectively enrolled, and the clinical data were obtained. The Hospital Anxiety and Depression Scale (HADS) was used to assess anxiety or depression levels. OLINK panel (Target 96 Inflammation) was used to quantify the plasma levels of inflammation-related proteins. Among the involved 142 IBD patients (median age 39.5, 42.96% female), 41 were comorbid with anxiety or depression symptoms. The levels of anxiety and depression symptoms in active phase group were significantly higher than those in quiescent group (P = 0.020). The anxiety and depression levels of IBD patients were positively correlated with fatigue levels (r = 0.713, P < 0.001), and negatively correlated with sleep quality (r = 0.499, P < 0.001) and quality of life (r =-0.692, P < 0.001). Plasma levels of 92 inflammation-related proteins were measured in 61 IBD patients. Up-regulated levels of fibroblast growth factor 23 (FGF-23) were found in IBD patients with anxiety or depression disorders, with an area under the curve (AUC) of 0.67(95%CI:0.53–0.81, P = 0.031). The plasma levels of C-C motif chemokine 20 (CCL20) and C-X-C motif chemokine 1 (CXCL1) were up-regulated in IBD patients with anxiety or depression, respectively, and the corresponding AUCs were 0.68 (95%CI:0.54–0.82, P = 0.036) and 0.70(95%CI:0.56–0.84, P = 0.017). Correlation analysis showed that the levels of anxiety and depression symptoms in IBD patients were negatively correlated with plasma Delta/Notch-like epidermal growth factor-related receptor (DNER) (r=-0.253, P = 0.047) and interleukin-8 (IL-8) (r=-0.275, P = 0.031) levels, and were positively correlated with the plasma levels of CXCL1 (r = 0.290, P = 0.022) and FGF-23 (r = 0.290, P = 0.022). In addition, negative correlation was found between plasma DNER levels and Mayo clinical scores in ulcerative colitis (UC) patients (r=-0.464, P = 0.001). Mood disorders are closely related to disease flare of IBD patients. The increasing levels of anxiety and depression in IBD patients are accompanied by graver fatigue, worse sleep quality and lower quality of life. Inflammation-related immune regulation is associated with the development of emotional disorders in IBD patients.
Similar content being viewed by others
Introduction
Inflammatory bowel disease (IBD) is a chronic recurrent intestinal disease, including ulcerative colitis (UC) and Crohn’s disease (CD)1. The existence of anxiety and depression is quite common in IBD patients. About 19.1–35.1% of IBD patients suffer from anxiety and 21.0–25.2% have comorbid depressive symptoms, which are significantly higher than those in general population2,3. The occurrence of anxiety and depression in IBD patients accelerates disease progression, increases drug dosage, and worsens the prognosis4,5,6.
The mechanisms of anxiety and depression in IBD patients may include changes of brain morphology, role of gut microbiota, and genetic factors2. The gut-brain axis plays a key role in the development of mood disorders in IBD patients by bidirectional regulation: gut inflammation aggravates psychiatric symptoms, on the contrary, mental symptoms aggravate the intestinal symptoms2,7. Evidence has suggested that immune pathways are evoked in the pathogenesis of many psychiatric diseases and cognitive dysfunction8. The occurrence of anxiety and depression disorders is proved to be correlated with the activated inflammatory response, and markers reflecting the intestinal inflammation such as C-reactive protein (CRP), interleukin (IL-6), and tumor necrosis factor-α (TNF-α), are found to increase in patients with mental disorders7.
Clarifying the mechanisms of anxiety and depression in IBD patients will help identify the pathophysiology and provide strategies for disease treatment. However, few studies have focused on the changes of inflammation-related proteins in IBD patients with anxiety or depression symptoms, and there is lack of research in reporting sensitive plasma markers to predict anxiety and depression disorders in IBD patients.
We aimed to describe the plasma proteomics characteristics of IBD patients with anxiety or depression, and to reveal the potential immunological mechanisms of emotional disorders in IBD patients from a new prospect (Fig. 1).
Materials and methods
Study design and participants
Patients with confirmed diagnosis of IBD based on clinical, endoscopic and pathological findings treated at Beijing Friendship Hospital, Capital Medical University from February 2023 to September 2023 were enrolled in this study if the following inclusion criteria were met: (1) ≥ 18 years old; (2) years of education ≥ 6; (3) with completed clinical data; (4) signed the informed consent for the study. Patients with current or past serious mental illness, such as schizophrenia, brain tumors, or disorders of consciousness, were excluded.
Clinical data collection and psychiatric symptoms assessment
Gender, age, and IBD related information containing age of onset, duration of disease, disease activity (UC patients assessed by Mayo clinical score; CD patients assessed by CDAI score) were collected. An active phase was defined as Mayo stool frequency score > 1 or rectal bleeding score > 0 for UC patients and CDAI score ≥ 150 for CD patients. The anxiety and depression symptoms were assessed by the Hospital Anxiety and Depression Scale (HADS), a widely used 14 questions (7 for anxiety and 7 for depression) self-reported scale for anxiety and depressive symptoms in hospital non-psychiatric patients9. The scale was divided into anxiety and depression subscales. A subscale score of ≥ 8 was regarded as the presence of anxiety or depression symptoms. The degree of fatigue was assessed by Multidimensional Fatigue Inventory (MFI)10 with higher scores indicating greater fatigue. The Pittsburgh Sleep Quality Index (PSQI) was used to evaluate the sleep quality of IBD patients11, and a higher PSQI score represented poorer sleep quality. Quality of Life (QoL) was assessed using the Inflammatory Bowel Disease Questionnaire (IBDQ)12, which could be divided into four dimensions, including bowel symptoms (IBDQ-B), systemic symptoms (IBDQ-S), emotional well-being (IBDQ-E) and social functioning (IBDQ-SF), with a higher score indicating a better QoL.
Blood sampling and measurement of plasma inflammation-related proteins
Peripheral blood samples were collected in vacutainer tubes containing the chelator ethylenediaminetetraacetic acid (EDTA) from IBD patients within 2 weeks of data collection and scale assessment. Blood samples were centrifuged at 1800 g for 10 min and the plasma layer was collected and stored at −80 ° C for future use. Plasma samples from each participant were analysed on the OLINK inflammation panel (Uppsala, Sweden), which contains 92 inflammation-related cytokines, chemokines or growth factors. The relative quantification of each protein was presented as normalized protein expression (NPX) values and can be used for further statistical analysis.
Statistical analysis
Statistical analyses were performed using SPSS 26.0. Categorical variables were presented by absolute counts (n) and relative frequencies (%). Normally distributed continuous variables were presented by mean ± standard deviation, and non-normally distributed data were presented by median and interquartile range. Unpaired Student’s t-test or Mann–Whitney U test was used to detect differences between groups, as appropriate. Receiver operating characteristic (ROC) curve analysis was performed to evaluate the ability of inflammation-related proteins in identifying anxiety or depression symptoms in IBD patients. Spearman correlation analysis was used to assess the associations between variables. A P value of less than 0.05 was considered statistically significant.
Ethical consideration
This study was performed in line with the principles of the Declaration of Helsinki and the protocol was approved by the Ethics Committee of Beijing Friendship Hospital, Capital Medical University (No. 2022-P2-292-02).
Results
Demographic and clinical characteristics of the enrolled patients
A total of 142 IBD patients were included in this study with median age of 39.5 (34.00–57.00) years, and 61 (42.96%) were female. The median age of IBD onset was 34.00 (26.00–49.00) and the median disease duration was 47.50 months (24.75–101.00). Among the 142 participants, 130 (91.55%) were UC patients, and 55 (38.73%) had active disease at the time of enrollment (Table 1). Patients were assigned to groups with anxiety or depression (A/D) (n = 41) or without A/D (n = 101). There were no statistical differences between patients with A/D and patients without A/D in gender, age, disease duration, and onset age of IBD. We found significant differences between IBD patients with A/D and without A/D in MFI, PSQI and IBDQ scores (P < 0.05).
Comparison of anxiety or depression levels between patients with active and quiescent IBD
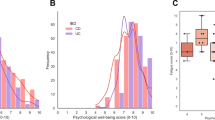
According to the disease activity status, participants were divided into active phase and quiescent phase. The scores of HADS-A (5.00 vs. 3.00, P = 0.036), HADS-D (4.00 vs. 3.00, P = 0.020) and HADS (9.00 vs. 5.00, P = 0.020) in patients with active IBD were significantly higher than those in the quiescent group (Fig. 2), indicating that active IBD is associated with more severe symptoms of anxiety and depression.
Correlation between HADS scores and clinical characteristics in IBD patients
Spearman correlation analysis showed that the HADS scores of IBD patients were positively correlated with MFI scores (r = 0.713, P < 0.0001) and PSQI scores (r = 0.499, P < 0.0001), and were negatively correlated with IBDQ scores (total IBDQ score (r =−0.692, P < 0.0001), IBDQ-B scores (r=−0.515, P < 0.0001), IBDQ-S scores (r=−0.626, P < 0.0001), IBDQ-E scores (r=−0.768, P < 0.0001), IBDQ-SF scores (r=−0.540, P < 0.0001)) (Fig. 3). These results suggested that higher levels of anxiety and depression in IBD patients were accompanied with higher levels of fatigue, worse sleep quality, and decreased quality of life.
Spearman correlation analysis between HADS scores and clinical characteristics in IBD patients. (A) Correlation between total HADS scores and MFI scores; (B) Correlation between total HADS scores and PSQI scores; (C) Correlation between total HADS scores and IBDQ scores (Total IBDQ score, IBDQ-B score, IBDQ-S score, IBDQ-E score, and IBDQ-SF score). Spots in red circle: patients with active IBD; Spots in blue triangle: patients with quiescent IBD.
Identification of plasma inflammation-related proteins specific to anxiety or depression in IBD patients
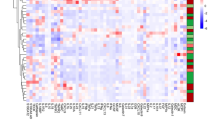
Plasma expression levels of 92 inflammation-related proteins were evaluated in 61 IBD patients (Table 2). The level of plasma fibroblast growth factor (FGF)−23 was significantly higher in IBD patients with A/D than in those without mood disorders (P = 0.031), and the area under curve (AUC) was 0.67 (95%CI:0.53–0.81) (Fig. 4A). In addition, compared with patients without anxiety, the plasma levels of C-C motif chemokine 20 (CCL20) (P = 0.036) and FGF-23 (P = 0.011) in IBD patients with anxiety were significantly up-regulated. The AUCs of CCL20 and FGF-23 for identifying IBD patients with anxiety were 0.68 (95%CI: 0.54–0.82) and 0.72 (95%CI: 0.57–0.86), respectively. Among IBD patients with depression, we found up-regulated plasma levels of C-X-C motif chemokine 1 (CXCL1) (P = 0.017) and FGF-23 (P = 0.045), compared with patients without depression. The AUCs of CXCL1 and FGF-23 for distinguishing IBD patients with depression were 0.70 (95%CI: 0.56–0.84) and 0.76 (95% CI:0.51–0.81), respectively (Fig. 4B-C). A subgroup analysis was further conducted among patients in both the active and quiescent phases of the disease. Within the active phase group, patients with mood disorders demonstrated significantly elevated plasma levels of cystatin 5 (CST5), axis inhibition protein 1 (AXIN1), FGF-23, sulfotransferase 1 A1 (ST1A1) and reduced plasma levels of urokinase-type plasminogen activator (uPA), delta/notch-like epidermal growth factor-related receptor (DNER), leukemia inhibitory factor receptor (LIF-R), and tumor necrosis factor-like weak inducer of apoptosis (TWEAK), compared to those without mood disorders (P < 0.05). In the quiescent phase group, IBD patients with mood disorders exhibited significantly decreased plasma levels of monocyte chemotactic protein 2 (MCP2) and monocyte chemotactic protein 3 (MCP3) compared to those without (P < 0.05) (Supplementary Fig. 1).
Specific plasma inflammation-related proteins between IBD patients with and without mood disorders. Mann–Whitney test and ROC curve of specific plasma proteins in differentiating: (A) IBD patients with or without anxiety or depression; (B) IBD patients with or without anxiety; (C) IBD patients with or without depression. *P < 0.05.
Correlation between HADS score and expression of plasma inflammation-related proteins
We further explored the correlation between HADS scores and plasma levels of inflammation-related proteins in IBD patients. The plasma levels of DNER were negatively correlated with HADS (r=−0.253, P = 0.047) and HADS-D (r=−0.293, P = 0.021) scores. Negative correlations were found between the plasma interleukin-8 (IL-8) levels and HADS (r=−0.275, P = 0.031), HADS-A (r=−0.255, P = 0.046) and HADS-D (r=−0.282, P = 0.026) scores, separately. In addition, we found a positive correlation between the plasma CXCL1 levels and HADS-D scores (r = 0.290, P = 0.022). Plasma FGF-23 levels were positively correlated with HADS (r = 0.290, P = 0.022) and HADS-A (r = 0.309, P = 0.015) scores (Fig. 5).
Correlation between plasma emotion-related proteins levels and disease activity in IBD patients
To figure out whether the previously found emotion-related plasma proteins were related to the intestinal inflammation in IBD patients, we analyzed the correlation between plasma levels of five proteins (IL-8, CXCL1, DNER, CCL20, FGF-23) and Mayo clinical scores in UC patients (n = 50). Results showed that significantly negative correlation was found between plasma DNER levels and Mayo clinical scores in UC patients (r=−0.464, P = 0.001) (Fig. 6). A correlation analysis was conducted between the peripheral blood CRP levels and differential protein levels in the enrolled patients. The results showed a significant positive correlation between plasma CXCL1 levels and blood CRP levels in IBD patients (Spearman r = 0.423, P = 0.028) (Supplementary Fig. 2). Additionally, we conducted a correlation analysis between the Ulcerative Colitis Endoscopic Index of Severity (UCEIS) scores and differential protein levels in UC patients who underwent colonoscopy during the study period. A significant positive correlation was found between plasma CXCL1 levels and UCEIS scores in UC patients (Spearman r = 0.483, P = 0.004), while plasma DNER levels showed a significant negative correlation with UCEIS scores (Spearman r=−0.390, P = 0.023) (Supplementary Fig. 3).
Discussion
This study characterized the clinical features of IBD patients with anxiety or depression and assessed their plasma inflammation-related proteins using OLINK technology. We found that active phase IBD patients exhibited significantly higher anxiety and depression symptoms compared to those in quiescent phase. These symptoms correlated with increased fatigue, poorer sleep quality, and diminished quality of life. Additionally, we identified specific plasma proteins that are predictive of mood disorders in IBD. Anxiety and depression levels negatively correlated with plasma DNER and IL-8, while positively correlating with CXCL1 and FGF-23. Notably, plasma DNER levels showed a significant negative correlation with UC activity, suggesting its potential role in the immune mechanisms underlying emotional disorders in IBD patients.
Several studies have reported close relationships between psychological factors and disease course in IBD patients13,14. A systematic review and meta-analysis of 12 longitudinal follow-up studies confirmed the bidirectional effect of anxiety or depression symptoms and the course of disease7. The study concluded that IBD patients with depression or anxiety had a significantly higher risk of therapy escalation, hospitalization, and emergency attendance. On the other hand, clinically active disease at baseline was found to be associated with future development of anxiety or depression. Our study showed that IBD patients in active phase were suffering from a significantly higher burden of anxiety and depression disorders than those in quiescent phase.
A considerable proportion of IBD patients have sleep disorders, which would negatively impact health and work productivity in the long term15. Our study found that higher anxiety and depression levels were associated with poorer sleep quality, aligning with previous research. The prevalence of fatigue in IBD patients is much higher than that in healthy individuals16, with over 80% of active phase patients and 40% of quiescent phase patients. We observed a significant positive correlation between fatigue levels and HADS scores in our IBD cohort, indicating a close link between mood disorders and fatigue symptoms. IBDQ is the most commonly used instrument for measuring disease-specific QoL in IBD patients17. Our research revealed that IBD patients with anxiety and depression experienced a decline in QoL across four aspects: bowel symptoms, systemic symptoms, emotional function, and social function.
The development of IBD is marked by systemic inflammation, activating immune cells and altering the expression of inflammation-related proteins18. Proinflammatory cytokines communicate with the brain through cellular, molecular, and neural pathways, altering neural activity19. Studies report altered inflammation markers in mood disorders. A recent study using the OLINK proteomics Platform identified 13 differentially expressed inflammation-related proteins in adolescents with depression versus healthy controls. Five proteins (CXCL6, CXCL11, IL-18, CXCL5, CCL4) were up-regulated, while eight (OSM, VEGF-A, TRAIL, TGF-α, TBFSF14, IL-10Rβ, Flt3L, HGF) were down-regulated in the depression group20. The flare of IBD activates the immune system, with immune cell-released inflammatory factors potentially contributing to the onset and progression of mood disorders in IBD patients21,22. However, the association between plasma inflammation-related proteins and anxiety or depression in IBD patients, and whether these protein changes were driven by intestinal inflammation, remained uncertain. Our study used OLINK proteomics to detect 92 inflammation-related proteins in plasma, identifying five closely linked to anxiety or depression in IBD patients.
DNER, a transmembrane protein, belongs to the non-classical Notch ligand family and binds to the Notch1 receptor23. DNER was first found to be highly expressed in Purkinje neurons and plays a key role in cerebellar development24. One study described the function and mechanism of DNER in macrophages in the context of chronic lung inflammation25. They discovered DNER localization in activated macrophages of COPD patients, regulating IFN-γ secretion through non-canonical Notch in pro-inflammatory macrophages. No prior studies have reported DNER’s role in gastroenterological inflammation or mood disorders. Our research revealed a negative correlation between plasma DNER levels and anxiety/depression in IBD patients, with a significant association also found between lower DNER levels and UC disease activity. We hypothesize that DNER may protect against gut inflammation in IBD and regulate mood disorders via the immune-mediated gut-brain axis. This is the first study to report DNER level changes in intestinal inflammation and mood disorder patients; however, further clinical and basic research is needed to elucidate DNER’s role in IBD and IBD-related emotional disorders. FGF-23 can be expressed in specific parts of the brain26 and has been shown to be associated with mood regulation27. No prior study has reported the relationship between FGF-23 and mood disorders in IBD patients. Our study first found significantly elevated plasma FGF-23 levels in IBD patients with anxiety or depression, which positively correlated with the severity of these mood disorders. The initial study on CCL20 in mood disorders found a positive correlation between plasma CCL20 levels and anxiety/depression scores in pregnant women28. Our study, possibly the second to report on CCL20 in mood disorders, aligns with these findings, revealing significantly elevated plasma CCL20 levels in IBD patients with anxiety disorders. CXCL1 has been identified in microglia, neurons, and oligodendrocyte progenitor cells29,30 and its level has been reported to alter in patients with depressive mood disorders31,32. However, the trend of plasma CXCL1 levels in mood disorder patients remains uncertain. Our findings revealed elevated plasma CXCL1 levels in IBD patients with depressive symptoms, positively correlating with depression scores. Consistent with previous findings that IL-8 was down-regulated in mood disorder patients33,34, we found a significant negative correlation between the levels of anxiety and depression and the plasma levels of IL-8 in IBD patients.
This study offers several notable advantages. Firstly, it pioneers the use of proteomics technology to explore anxiety and depression disorders in IBD patients, identifying five inflammation-related proteins that are closely associated with these disorders. Secondly, through multiple clinical scales, we comprehensively assessed IBD patients, examining the relationships between anxiety and depression symptoms, fatigue, sleep quality, and quality of life, thereby elucidating the clinical characteristics of IBD patients with these psychological comorbidities. This study lays a foundation for understanding the potential immunological mechanisms underlying these conditions in IBD patients. However, this study has several limitations. It is a single-center, cross-sectional investigation with a limited sample size, and it does not include a comparative analysis of outcomes before and after treatment. Also, the absence of multiple comparison corrections may increase the risk of Type I errors. Additionally, we did not consider the potential confounding influences of medication usage, dietary habits, and other variables on the results. Furthermore, our study population was primarily comprised of patients with ulcerative colitis due to the demographics of our clinical center. To address these gaps, larger-scale prospective cohort studies and mechanistic experiments are needed to investigate the relationship between plasma proteins and psychological comorbidities in IBD patients.
Conclusion
The anxiety and depression symptoms in IBD patients are closely related to disease flare. The increasing levels of anxiety and depression in IBD patients are accompanied by graver fatigue, worse sleep quality and lower quality of life. Several inflammation-related proteins are significantly changed in plasma of IBD patients with anxiety and depression and are significantly correlated with the levels of anxiety and depression. Inflammation-related immune regulation is associated with the development of emotional disorders in IBD patients. A better understanding of the specific immunological mechanism related to mood disorders in IBD patients, which is critical to achieving effective approaches for the diagnosis and treatment of psychological comorbidities in IBD patients is needed.
Data availability
The data are not publicly available due to privacy or ethical restrictions. Underlying data can be shared upon reasonable request made to the corresponding author.
References
Kaplan, G. G. & Ng, S. C. Understanding and preventing the global increase of inflammatory bowel disease. Gastroenterology ;152. (2017).
Bisgaard, T. H. et al. Depression and anxiety in inflammatory bowel disease: epidemiology, mechanisms and treatment. Nat. Rev. Gastroenterol. Hepatol. 19, 717–726 (2022).
Barberio, B. et al. Prevalence of symptoms of anxiety and depression in patients with inflammatory bowel disease: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 6, 359–370 (2021).
Kim, S. et al. Depression and anxiety are associated with poor outcomes in patients with inflammatory bowel disease: A nationwide population-based cohort study in South Korea. Gen. Hosp. Psychiatry. 81, 68–75 (2023).
Sauk, J. S. et al. High perceived stress is associated with increased risk of ulcerative colitis clinical flares. clinical gastroenterology and hepatology: the official clinical practice. J. Am. Gastroenterological Association ;21. (2023).
Roderburg, C. et al. Association between inflammatory bowel disease and subsequent depression or anxiety disorders - A retrospective cohort study of 31,728 outpatients. J. Psychiatr. Res. 169, 231–237 (2024).
Fairbrass, K. M. et al. Bidirectional brain-gut axis effects influence mood and prognosis in IBD: a systematic review and meta-analysis. Gut 71, 1773–1780 (2022).
Müller, N. & Ackenheil, M. Psychoneuroimmunology and the cytokine action in the CNS: implications for psychiatric disorders. Prog. Neuro-psychopharmacol. Biol. Psychiatry ;22. (1998).
Zigmond, A. S. & Snaith, R. P. The hospital anxiety and depression scale. Acta Psychiatr Scand. 67, 361–370 (1983).
Smets, E. M. et al. The multidimensional fatigue inventory (MFI) psychometric qualities of an instrument to assess fatigue. J. Psychosom. Res. 39, 315–325 (1995).
Smyth, C. The Pittsburgh sleep quality index (PSQI). J. Gerontol. Nurs. 25, 10–11 (1999).
Guyatt, G. et al. A new measure of health status for clinical trials in inflammatory bowel disease. Gastroenterology 96, 804–810 (1989).
Jordi, S. B. U. et al. Depressive symptoms predict clinical recurrence of inflammatory bowel disease. Inflamm. Bowel Dis. 28, 560–571 (2022).
Narula, N. et al. Anxiety but not depression predicts poor outcomes in inflammatory bowel disease. Inflamm. Bowel Dis. 25, 1255–1261 (2019).
Barnes, A. et al. Systematic review and meta-analysis of sleep quality in inactive inflammatory bowel disease. JGH Open: Open. Access. J. Gastroenterol. Hepatol. 6, 738–744 (2022).
Holten, K. I. A. et al. Fatigue in patients with newly diagnosed inflammatory bowel disease: results from a prospective inception cohort, the IBSEN III study. J. Crohn’s Colitis. 17, 1781–1790 (2023).
Alrubaiy, L. et al. Systematic review of health-related quality of life measures for inflammatory bowel disease. J. Crohn’s Colitis. 9, 284–292 (2015).
Neurath, M. F. Cytokines in inflammatory bowel disease. Nat. Rev. Immunol. 14, 329–342 (2014).
Slavich, G. M. & Irwin, M. R. From stress to inflammation and major depressive disorder: a social signal transduction theory of depression. Psychol. Bull. 140, 774–815 (2014).
Yang, L. et al. Identification of plasma inflammatory markers of adolescent depression using the Olink proteomics platform. J. Inflamm. Res. 16, 4489–4501 (2023).
Gao, X. et al. Change of monocytes/macrophages in ulcerative colitis patients with symptoms of anxiety and depression. BMC Gastroenterol. 23, 67 (2023).
Tang, Y. et al. Crohn’s disease patients with depression exhibit alterations in monocyte/macrophage phenotype and increased Proinflammatory cytokine production. Dig. Dis. (Basel Switzerland). 38, 211–221 (2020).
D’Souza, B., Meloty-Kapella, L. & Weinmaster, G. Canonical and non-canonical Notch ligands. Curr. Top. Dev. Biol. ;92. (2010).
Eiraku, M. et al. Delta/notch-like epidermal growth factor (EGF)-related receptor, a novel EGF-like repeat-containing protein targeted to dendrites of developing and adult central nervous system neurons. J. Biol. Chem. 277, 25400–25407 (2002).
Ballester-López, C. et al. The Notch ligand DNER regulates macrophage IFNγ release in chronic obstructive pulmonary disease. EBioMedicine 43, 562–575 (2019).
Duce, J. A. et al. Gene profile analysis implicates Klotho as an important contributor to aging changes in brain white matter of the rhesus monkey. Glia 56, 106–117 (2008).
Ford-Perriss, M., Abud, H. & Murphy, M. Fibroblast growth factors in the developing central nervous system. Clin. Exp. Pharmacol. Physiol. 28, 493–503 (2001).
Camacho-Arroyo, I. et al. Chemokine profile in women with moderate to severe anxiety and depression during pregnancy. BMC Pregnancy Childbirth. 21, 807 (2021).
Horuk, R. et al. Expression of chemokine receptors by subsets of neurons in the central nervous system. Journal of Immunology (Baltimore, Md.: 1997;158:2882–2890. (1950).
Nguyen, D. & Stangel, M. Expression of the chemokine receptors CXCR1 and CXCR2 in rat oligodendroglial cells. Brain Res. Dev. Brain Res. 128, 77–81 (2001).
Bot, M. et al. Serum proteomic profiling of major depressive disorder. Translational Psychiatry. 5, e599 (2015).
Walss-Bass, C. et al. Inflammatory markers as predictors of depression and anxiety in adolescents: statistical model Building with component-wise gradient boosting. J. Affect. Disord. 234, 276–281 (2018).
Irwin, M. R. et al. Association of interleukin-8 and risk of incident and recurrent depression in long-term breast cancer survivors. Brain. Behav. Immun. 105, 131–138 (2022).
Shan, L-L. et al. Association of serum Interleukin-8 and serum amyloid A with anxiety symptoms in patients with cerebral small vessel disease. Front. Neurol. 13, 938655 (2022).
Acknowledgements
The authors would like to thank all participants for their time and interest in the study. All authors declare no conflicts of interest.
Funding
This project was supported by Beijing Nova Program (Z201100006820147), Beijing Science and Technology Program (Z211100002921028), Capital’s Funds for Health Improvement and Research (CFH2022-2-2025 and 2020-4-2085), and National Key Research and Development Plan (2022YFC3602104). The funding bodies had no role in the study design, data collection or analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
M.S.Z., P.L., H.Y.S. and J.W. all contributed to the design of the study. M.S.Z, C.S.E.W, S.Y.Z and H.Y.S. recruited the participants. M.S.Z., W.R.Z., Y.D., F.X., C.Y.X. and Z.W. collected data. M.S.Z., W.R.Z and Y.D. analyzed the data. M.S.Z. and W.R.Z drafted the manuscript. H.Y.S and J.W. critically revised the manuscript for important intellectual content. All authors have approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and clinical trial number
The study was approved by the Ethics Committee of Beijing Friendship Hospital, Capital Medical University (No. 2022-P2-292-02; Approval date 2022.10.8), and all methods were carried out by the principle embodied in the Declaration of Helsinki. This study has been registered on the medical research registration and filing information system (Clinical trial number MR-11-23-021147; Registration date 2023.7.4).
Consent to participate
All participants of this study provided written informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, M.S., Zhang, W.R., Dang, Y. et al. Plasma inflammation-related proteins associated with anxiety and depression disorders in IBD patients. Sci Rep 15, 18445 (2025). https://doi.org/10.1038/s41598-025-03543-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-03543-1