Abstract
In this international collaboration, we examined whether survival in metastatic uveal melanoma is associated with primary tumor size at initial diagnosis. A total of 128 and 205 patients with American Joint Committee on Cancer (AJCC) stage IV disease were included from the Netherlands and Sweden, respectively. Across both cohorts, patients whose tumors had a largest basal diameter ≥ 16 mm or a higher AJCC stage presented with a greater number of hepatic metastases and showed shorter Kaplan-Meier overall survival from the time of metastatic detection. However, this association did not persist after adjustment for the number of hepatic metastases in multivariate Cox regression and a Markov multi-state model. These findings suggest that primary tumor size is associated with survival in metastatic disease and the burden of metastatic lesions at detection, indicating a potential survival benefit of primary tumor treatment. Nevertheless, caution is warranted when interpreting these results, as they differ from some previous reports.
Similar content being viewed by others
Introduction
Uveal melanoma is the most common primary intraocular malignancy in adults, characterized by its high propensity for developing fatal metastases1 The long-term incidence of metastatic death approaches 50%, with fewer improvements in prognosis compared to cutaneous melanoma2,3.
Primary tumor size and anatomic extent are critical factors in the American Joint Committee on Cancer (AJCC) staging system, which strongly predicts the time to metastatic disease4,5 In 2014, Damato and colleagues demonstrated that among those who succumb to their disease, the median survival time from primary tumor treatment to death was 4.6 years for patients in stage I, compared to only 2.7 years for patients in stage III6 Thus, a shorter metastasis-free interval is associated with poorer overall survival outcomes7,8,9,10.
However, the shorter time to metastasis and death in patients with more advanced stages might simply reflect lead-time bias, with larger tumors having had more time to grow and metastasize11 In this scenario, the treatment of large and small tumors might occur at different points on the same timeline, potentially resulting in similar dates of death despite different initial tumor sizes.
To address this issue, we propose evaluating the survival time from the detection of metastases to death. If this interval is shorter for patients whose primary tumors were larger at the time of treatment, it would suggest that the growth rate of metastases is associated with the size of the primary tumor. This would imply a survival benefit of treating the primary tumor at an earlier stage. In turn, this would clarify the potential benefit of treating uveal melanoma as soon as possible after diagnosis.
However, previous studies have not identified an association between primary tumor diameter at initial diagnosis and an increased risk or hazard ratio for death among patients who develop metastases7,8,12,13 The largest study to date, which did not include time-to-event survival analyses, included 330 patients with metastatic choroidal and ciliary body melanomas12 While this study confirmed a relationship between a shorter metastasis-free interval and reduced overall survival, it found no differences in AJCC stage distribution among patients with varying overall survival times.
In the present study, we analyze data from 333 metastatic patients treated at two European centers. Our aim is to determine whether primary tumor size influences survival after the onset of metastatic disease.
Results
Descriptive statistics
Of the 333 included patients with metastatic disease, 176 (53%) were female. The mean age at the time of diagnosis of the primary tumor was 61 years (SD 12 years). The mean largest basal diameter (LBD) was 13.2 mm (SD 3.5 mm), the mean tumor thickness was 6.7 mm (SD 3.0 mm), and the mean tumor volume was 609 mm³ (SD 493 mm³). Further details on the included patients and tumors are provided in Table 1.
All patients underwent baseline radiological examinations. In the Netherlands, abdominal ultrasound and chest x-ray were the first-choice imaging modalities. When these were inconclusive, additional imaging with computed tomography (CT), or magnetic resonance imaging (MRI) was performed. In Sweden, CT of the thorax and abdomen was the first-choice imaging modality. Of the 333 patients, 11 (3%) had metastases at initial presentation. Among these 11 patients, 4 (36%) had large primary tumors (LBD ≥ 16 mm). Two hundred and twenty-two patients (67%) underwent regular (biannual) liver ultrasonography for at least five years after primary tumor treatment. Among those with large primary tumors (LBD ≥ 16 mm), 48 of 76 (63%) received regular surveillance, compared with 174 of 245 (71%) for smaller tumors (LBD < 16 mm; chi‐square P = 0.20). Patients in surveillance did not present with a greater number of hepatic metastases at the initial diagnosis of metastatic disease (chi‐square test for trend P = 0.26). However, those with large primary tumors (LBD ≥ 16 mm) or more advanced AJCC stage had significantly more hepatic metastases upon radiological detection (chi‐square test for trend P = 0.01 and P < 0.001, respectively, Tables 2 and 3).
Visual inspection showed that the log-minus-log survival curves were parallel and did not cross, suggesting that the proportional hazards assumption was adequately met for the AJCC stage covariate (Supplementary Figure S1).
Prognostic implication of primary tumor size for survival in metastatic disease
Among the 333 included patients, AJCC stage at the time of primary tumor diagnosis was significantly associated with survival after the detection of metastases. The median Kaplan-Meier survival estimates after the first radiological detection of metastases were as follows: Stage I, 1.43 years (95% CI, 0.80–2.15); Stage IIA, 1.08 years (95% CI, 0.88–1.48); Stage IIB, 0.99 years (95% CI, 0.72–1.34); Stage IIIA, 0.40 years (95% CI, 0.30–0.75); Stage IIIB, 0.86 years (95% CI, 0.54–not determinable); Stage IIIC, 0.08 years (95% CI, not determinable); and Stage IV, 0.41 years (95% CI, 0.27–not determinable). The 1year survival rates by AJCC stage were as follows: Stage I, 58.9% (95% CI, 44.5–78.0); Stage IIA, 52.7% (95% CI, 44.0–63.2); Stage IIB, 49.5% (95% CI, 40.8–60.0); Stage IIIA, 29.3% (95% CI, 18.2–47.1); Stage IIIB, 46.2% (95% CI, 25.7–83.0); and Stage IV, 27.3% (95% CI, 10.4–71.6). Both patients initially diagnosed with Stage IIIC disease died within a year of metastasis detection.
In both cohorts, patients with primary tumors having an LBD < 16 mm demonstrated longer survival after metastasis detection (log-rank P ≤ 0.04, Fig. 1). Similarly, when stratified by AJCC stage, a significant trend was observed in both cohorts (log-rank P ≤ 0.04, Fig. 2).
Kaplan–Meier survival curves for 321 patients with metastatic uveal melanoma, stratified by the largest basal diameter (LBD) of the primary tumor. Twelve patients without LBD data were excluded. (A) Rotterdam (the Netherlands) cohort, (B) Stockholm (Sweden) cohort, and (C) combined cohorts. In both cohorts, patients with LBD < 16 mm at the time of treatment had significantly longer survival after metastasis detection than those with LBD ≥ 16 mm. P values were corrected using the Holm–Bonferroni method. AJCC, American Joint Committee on Cancer.
Kaplan–Meier survival curves for 222 patients with metastatic uveal melanoma, categorized by AJCC stage at the time of primary tumor diagnosis. Eleven patients already in stage IV at diagnosis were excluded. (A) Rotterdam cohort, (B) Stockholm cohort, and (C) combined cohorts. P values were corrected using the Holm–Bonferroni method. AJCC, American Joint Committee on Cancer.
Multivariate Cox regression analysis, with age at diagnosis included as a covariate, confirmed that AJCC stage at primary tumor diagnosis independently predicted mortality following the detection of metastases (Table 4).
Furthermore, we constructed a Cox model spanning the entire period from primary tumor diagnosis to death or last follow-up, incorporating the number of hepatic metastases detected after the metastasis-free interval as a time-varying covariate. The analysis revealed that the number of hepatic metastases was significantly associated with an increased hazard of death (hazard ratio (HR) 9.41, 95% CI, 6.34–13.97, P < 0.001). In contrast, the prognostic significance of AJCC stage at primary tumor diagnosis was not retained (HR 1.05, 95% CI, 0.87–1.27, P = 0.61, Table 5), suggesting that the impact of primary tumor size on survival after metastatic detection is related to the number of metastatic lesions.
Lastly, a Markov multi-state model was used to evaluate transition dynamics from the initial primary tumor diagnosis to metastasis detection and eventually death. As anticipated, AJCC stage at the time of primary tumor diagnosis significantly influenced the transition from non‐metastatic to metastatic status: each one‐stage increase corresponded to a 37% higher hazard of developing metastases (HR 1.37, 95% CI, 1.09–1.72, P = 0.006). For the transition from metastatic disease diagnosis to death, however, AJCC stage lost its significance when the number of hepatic metastases was included as a covariate (Table 6).
Discussion
In this study, we found that patients treated for larger primary tumors presented with a greater number of hepatic metastases, even though they did not have more frequent radiologically visible lesions at baseline examinations and underwent radiological surveillance at the same intervals as patients with smaller primary tumors. Furthermore, once metastases were detected, patients with larger primary tumors died sooner than those with smaller tumors. These findings bridge the gap between earlier studies linking larger primary tumor size with shorter time to metastasis on one hand, and between shorter time to metastasis and shorter post-metastasis survival on the other12,14,15,16.
One plausible explanation for the increased number of metastases from larger primary tumors could be that these metastases grow more rapidly, potentially explaining their association with quicker demise. However, caution is warranted, as our findings differ from some previous studies. In 2000, Eskelin and colleagues observed no linear relationship between primary tumor volume and the estimated doubling time of metastases in 37 patients17 This underscores the need for further investigations in external, ideally larger, cohorts.
If confirmed, these observations are highly relevant for understanding how primary tumor treatment affects survival. Tumor cells are believed to disseminate early, as supported by the detection of circulating tumor cells and micrometastases in multiple organs of patients who died from unrelated causes18,19,20 Meanwhile, interventions such as enucleation or plaque brachytherapy prevent tumors from reaching higher AJCC stages than they would have in the absence of treatment. It is well-established both that lower stage tumors are associated with decreased mortality, and that the proportion of tumors with aggressive traits such as monosomy 3 and gain of 8q increases with increasing tumor size4,15,21 A reasonable interpretation is that a patient’s metastatic risk is largely determined when the tumor is still very small; nevertheless, by diagnosing and treating the tumor promptly, we may avert the additional risk conferred by continued tumor growth.
We acknowledge the validity of theories suggesting that a patient’s lifespan from diagnosis to death from metastases could be determined by micrometastases and genetic traits established at an early stage19,22,23 The longer interval between primary tumor diagnosis and metastatic death for smaller tumors might occur because these tumors are detected earlier in their progression. In contrast, larger tumors may have been growing undetected for a longer period, which could explain the shorter time from diagnosis to death for patients with larger tumors, as well as the observation of a greater number of metastases upon the initial detection of metastatic disease24 However, this reasoning, which assumes that all metastases grow at similar rates irrespective of primary tumor size and that any differences in survival time are merely a result of lead time bias, does not fully explain why patients with metastatic disease experience shorter survival when their primary tumor was diagnosed at a more advanced stage. Indeed, a recent meta-analysis indicates that tumor doubling time decreases as primary tumor grow larger25.
An alternative explanation is therefore that metastases originating from larger, more advanced primary tumors tend to grow more rapidly and are more immediately life-threatening than metastases from smaller, less advanced primary tumors. Thus, our findings suggest that a potential survival benefit of primary tumor treatment is to prevent the tumor from advancing to a higher AJCC stage and acquiring more malignant genetic traits, thereby reducing the aggressiveness and growth rate of the metastases it seeds.
These findings also underscore the potential survival benefits of timely primary tumor treatment in uveal melanoma26,27 While it is evident that treatment cannot be initiated for tumors that have not yet been detected, and that some observation for growth is necessary to differentiate between benign and malignant choroidal melanocytic lesions, once a diagnosis of melanoma is established, delaying treatment may be detrimental. Treatment should be administered as soon as practically possible after diagnosis to minimize the risk of tumor progression to a more advanced AJCC stage. It is also important to note that small tumors that grow slowly will naturally take longer to progress to a higher AJCC stage, implying that delaying treatment for these lesions may have minimal or no impact on prognosis compared to rapidly growing tumors25,28 Conversely, while larger tumors are generally associated with more aggressive behavior, some small tumors may possess aggressive genetic mutations, and some larger tumors may have characteristics associated with a more favorable prognosis15 This variability highlights the need for a nuanced approach to treatment decisions, considering both tumor size and its molecular behavior. Many institutions tailor their radiological surveillance programs based on perceived metastatic risk, determined by factors such as AJCC stage, chromosome 3 status, gene expression profiling, or other prognostic markers29 While it may seem logical to recommend more frequent radiological examinations for patients with markers of aggressive disease, and less frequent or no examinations for those with markers of lower metastatic risk (e.g., AJCC stage I, disomy 3, or EIF1AX mutation), it is important to note that, to date, there is no evidence that surveillance improves survival in uveal melanoma. Our observations support this, as there was no significant difference in the number of metastatic lesions at the presentation of metastatic disease between patients who did and did not undergo radiological surveillance.
The next few years may be pivotal in this regard, as new treatments for metastatic disease could potentially improve survival rates, thereby increasing the value of early detection of metastatic lesions.
Limitations
This study has several limitations beyond those already discussed. Firstly, the results are derived from retrospective observational cohorts, which inherently limits the ability to draw definitive conclusions about causality. The potential survival benefit of early treatment versus observation of small primary uveal melanomas has been debated within the field30 Most previous studies have not found a significant association between the stage or size of the primary tumor at initial diagnosis and survival in patients with metastatic disease, suggesting caution in interpreting such associations7,8,12,13 To truly determine whether primary tumor treatment confers a survival benefit in uveal melanoma, a large randomized clinical trial would be necessary, comparing outcomes between a treated group and an untreated group. However, such a trial would raise serious ethical concerns and is unlikely to ever be conducted. Additionally, the validity of our findings relies on the accuracy of the underlying data, which was obtained from medical charts, treatment records, and cause of death registries. Although efforts were made to cross-verify information from cause of death registries with medical records, the possibility of misclassification remains. Such errors could have influenced the survival outcomes reported in this study.
Thirdly, the assessment of the dimensions and anatomical extent of primary tumors and metastases varies by measurement method. For primary tumors, methods include fundus photography, ultrasonography, and gross pathological examination, while for metastases, ultrasound, MRI, CT, and PET/CT are used. Gross pathological examination, commonly performed on enucleated eyes, involves various techniques that may affect measurement consistency. Some pathologists measure the chord length of the tumor’s transillumination shadow with a caliper, while others use a flexible ruler to measure arc dimensions, potentially yielding different results, especially in larger tumors (> 10 mm). Measurements can also vary after the globe is opened, depending on sectioning and whether taken fresh or after formalin fixation, which causes tissue shrinkage—affecting thickness more than basal diameter. Due to these discrepancies, it is uncertain how well pathological measurements reflect the true tumor dimensions compared to clinical measurements obtained with current methods.
Fourth, patient age was included as a covariate in regression analyses, despite ongoing debate about its association with prognosis, especially when considering competing risks and other prognostic factors in multivariate analyses2,31 However, age-related factors, such as comorbidities and overall health status, may affect both disease progression and a patient’s ability to tolerate aggressive treatments for metastases. Additionally, age has been linked to aggressive genetic traits, such as BAP1 mutation and aggressive gene expression profiles32 Older patients may present with more advanced disease within the same stage category due to a longer potential duration of tumor growth, supporting our decision to include age as a covariate.
Fifth, we lacked data on the genetic or cytogenetic characteristics of both primary tumors and metastases. Additionally, data on the number of metastases were available for only 144 patients, and the size of these lesions was not documented. Information regarding the involvement of organs other than the liver at the time of first metastatic detection was also unavailable. The size and distribution of metastatic lesions are important prognostic factors in metastatic disease, and including these data in regression analyses might have altered the results. However, the number of hepatic metastases, which is associated with the presence of miliary metastases, also has substantial prognostic value7,24,33 Thus, we believe that the absence of data on the size of the largest metastatic lesion, and the potential involvement of other organs, likely had a limited impact on our findings.
Finally, the examined cohorts in our study were heterogeneous, with a median overall survival of 0.6 years in the Rotterdam cohort and 1.3 years in the Stockholm cohort. Nevertheless, it could be argued that the consistent association between larger primary tumor size and survival in metastatic disease—even under varying epidemiological conditions—supports the robustness of our findings.
Conclusions
This study found that patients treated for larger primary tumors presented with a greater number of hepatic metastases and, once metastases were detected, had shorter survival than those with smaller tumors. These findings suggest that treating the primary tumor, thereby preventing its progression to a more advanced AJCC stage, may offer a survival benefit for some patients, by potentially reducing the aggressiveness and growth rate of subsequent metastases.
Methods
Aim of the study
The aim of this study was to examine the prognostic implication of primary tumor size for survival in metastatic disease.
Patients and study design
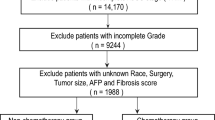
We collected data on all patients who developed metastatic disease after being diagnosed with primary uveal melanoma at either the Erasmus University Medical Center and the Rotterdam Eye Hospital, Rotterdam, The Netherlands, between 1993 and 2021, or the Ocular Oncology Service, St. Erik Eye Hospital, Stockholm, Sweden, between 1980 and 2021. A total of 626 patients met the following inclusion criteria:
Inclusion criteria:
-
1.
Data available in treatment registries at the respective institution.
-
2.
Clinically or histopathologically confirmed diagnosis of choroidal and/or ciliary body melanoma at the time of primary tumor diagnosis.
-
3.
Enrollment in a surveillance program with periodic liver examinations for a minimum of 5 years after primary tumor diagnosis (using contrast-enhanced ultrasound, computed tomography [CT], or magnetic resonance imaging [MRI]).
-
4.
Radiologically detected metastases in the liver and/or other organs.
-
5.
Availability of CT or MRI images.
Exclusion criteria:
-
1.
Iris melanoma (n = 0).
-
2.
Lack of recorded primary tumor thickness or LBD at the time of primary tumor diagnosis (n = 29).
-
3.
Unknown anatomical extent, specifically CBI or EXE, at the time of primary tumor diagnosis (n = 0).
-
4.
Unrecorded location of metastases (n = 51).
-
5.
Uncertain exact date of radiological detection of metastases, including cases where dates were not specified in referrals, medical notes, or radiological image files, or where the diagnosis was indeterminable due to unclear findings in one exam followed by an established diagnosis in a subsequent exam (n = 213).
After applying these criteria, 333 patients remained in the final cohort, of which 128 were from Rotterdam and 205 from Stockholm. The date of the first radiological detection of metastases was used as the date of metastasis. Data on the number of patients who underwent biopsy for histopathological confirmation of metastases were not available. Of the 128 patients from Rotterdam, 123 were included in a previous study, with metastases to the liver being observed in 96% of cases24 For the 123 patients in the Rotterdam sample, and for 21 of the patients in the Stockholm samle, data were available on the number of metastases present at the initial CT or MRI scan when the first metastases were observed.
The study was approved by the Swedish Ethical Review Authority (reference 2023-07537-02) and adhered to the tenets of the Declaration of Helsinki. Informed consent was waived by the Swedish Ethical Review Authority because the study relied on retrospective, pre-collected data. No sensitive information was shared between the institutions, and no new collection of identifiable information was conducted, including patients’ names, identification numbers, addresses, contact details, or photographs. No interventions, testing, or examinations were performed, and no analyses of biological tissues were conducted.
Statistical analyses
Statistical significance was defined as P < 0.05, and all P values were two-sided. Holm-Bonferroni corrections were applied to all reported P values. Kaplan-Meier survival curves were generated, and multivariate and time-varying Cox regression analyses were performed using the survival and survminer packages in R (version 4.4.1, The R Foundation for Statistical Computing, Vienna, Austria). Survival distributions across AJCC stages at primary tumor diagnosis were compared using the log-rank test for trend. Additionally, patients were categorized based on whether the largest basal diameter of the primary tumor was larger or smaller than 16 mm. This threshold aligns with the definition of large tumors in the Collaborative Ocular Melanoma Study (COMS) and represents a common cutoff where enucleation is often favored over eye-preserving treatments such as plaque brachytherapy34 To examine the transition dynamics from primary tumor diagnosis to metastasis detection and subsequent death, a Markov multi-state model was constructed using the mstate and survival packages in R. The proportional hazards assumption was assessed by inspecting log-minus-log survival curves; the assumption was considered satisfied if the curves were parallel and did not cross. Tumor volume was estimated using a formula consistent with previously described methods, where LBD represents the largest basal tumor diameter:35,36
Data availability
Data, including the Rotterdam and Stockholm samples, are available upon reasonable request from the corresponding author, subject to approval from the Swedish Ethical Review Authority.
References
Lane, A. M., Kim, I. K. & Gragoudas, E. S. Survival rates in patients after treatment for metastasis from uveal melanoma. JAMA Ophthalmol. 136, 981. https://doi.org/10.1001/jamaophthalmol.2018.2466 (2018).
Stalhammar, G. Comprehensive causes of death in uveal melanoma: Mortality in 1530 consecutively diagnosed patients followed until death. JNCI Cancer Spectr. 7. https://doi.org/10.1093/jncics/pkad097 (2023).
Rantala, E. S., Hernberg, M. M., Piperno-Neumann, S., Grossniklaus, H. E. & Kivela, T. T. Metastatic uveal melanoma: the final frontier. Prog Retin Eye Res. 90, 101041. https://doi.org/10.1016/j.preteyeres.2022.101041 (2022).
Kujala, E. et al. Staging of ciliary body and choroidal melanomas based on anatomic extent. J. Clin. Oncol. 31, 2825–2831. https://doi.org/10.1200/JCO.2012.45.2771 (2013).
Simpson, E. R. et al. International validation of the American joint committee on Cancer’s 7th edition classification of uveal melanoma. JAMA Ophthalmol. 133, 376–383. https://doi.org/10.1001/jamaophthalmol.2014.5395 (2015).
Damato, B. E., Heimann, H., Kalirai, H. & Coupland, S. E. Age, survival predictors, and metastatic death in patients with choroidal melanoma: tentative evidence of a therapeutic effect on survival. JAMA Ophthalmol. 132, 605–613. https://doi.org/10.1001/jamaophthalmol.2014.77 (2014).
Mariani, P. et al. Development of a prognostic nomogram for liver metastasis of uveal melanoma patients selected by liver MRI. Cancers (Basel) 11. https://doi.org/10.3390/cancers11060863 (2019).
Valpione, S. et al. Development and external validation of a prognostic nomogram for metastatic uveal melanoma. PLoS One. 10, e0120181. https://doi.org/10.1371/journal.pone.0120181 (2015).
Eskelin, S., Pyrhonen, S., Hahka-Kemppinen, M., Tuomaala, S. & Kivela, T. A prognostic model and staging for metastatic uveal melanoma. Cancer 97, 465–475. https://doi.org/10.1002/cncr.11113 (2003).
Rietschel, P. et al. Variates of survival in metastatic uveal melanoma. J. Clin. Oncol. 23, 8076–8080. https://doi.org/10.1200/JCO.2005.02.6534 (2005).
Damato, B. & Coupland, S. E. A reappraisal of the significance of largest basal diameter of posterior uveal melanoma. Eye (Lond). 23, 2152–2160. https://doi.org/10.1038/eye.2009.235-cme (2009). quiz 2161 – 2152.
Rantala, E. S. et al. Determinants of Long-Term survival in metastatic choroidal and ciliary body melanoma. Am. J. Ophthalmol. 246, 258–272. https://doi.org/10.1016/j.ajo.2022.10.017 (2023).
Kodjikian, L. et al. Prognostic factors of liver metastases from uveal melanoma. Graefes Arch. Clin. Exp. Ophthalmol. 243, 985–993. https://doi.org/10.1007/s00417-005-1188-8 (2005).
Shields, C. L. et al. Conditional survival of uveal melanoma using the Cancer genome atlas (TCGA) classification (Simplified Version) in 1001 cases. Saudi J. Ophthalmol. 36, 308–314. https://doi.org/10.4103/sjopt.sjopt_69_21 (2022).
Stalhammar, G. et al. Improved staging of ciliary body and choroidal melanomas based on Estimation of tumor volume and competing risk analyses. Ophthalmology 131, 478–491. https://doi.org/10.1016/j.ophtha.2023.10.026 (2023).
Bansal, R., Sener, H., Ganguly, A., Shields, J. A. & Shields, C. L. Metastasis-free survival of uveal melanoma by tumour size category based on the Cancer genome atlas (TCGA) classification in 1001 cases. Clin. Exp. Ophthalmol. https://doi.org/10.1111/ceo.14446 (2024).
Eskelin, S., Pyrhönen, S., Summanen, P., Hahka-Kemppinen, M. & Kivelä, T. Tumor doubling times in metastatic malignant melanoma of the Uvea: tumor progression before and after treatment. Ophthalmology 107, 1443–1449. https://doi.org/10.1016/s0161-6420(00)00182-2 (2000).
Grisanti, S. et al. Detection of Circulating tumor cells in patients with small choroidal melanocytic lesions. Ophthalmology 130, 1290–1303. https://doi.org/10.1016/j.ophtha.2023.07.025 (2023).
Gill, V. T. et al. Multiorgan involvement of dormant uveal melanoma micrometastases in postmortem tissue from patients without coexisting macrometastases. Am. J. Clin. Pathol. 160, 160–174. https://doi.org/10.1093/ajcp/aqad029 (2023).
Eide, N. et al. The results of stricter inclusion criteria in an immunomagnetic detection study of micrometastatic cells in bone marrow of uveal melanoma Patients - Relevance for dormancy. Pathol. Oncol. Res. 25, 255–262. https://doi.org/10.1007/s12253-017-0355-7 (2019).
Shields, C. L. et al. American joint committee on Cancer classification of uveal melanoma (Anatomic Stage) predicts prognosis in 7,731 patients: the 2013 Zimmerman lecture. Ophthalmology 122, 1180–1186. https://doi.org/10.1016/j.ophtha.2015.01.026 (2015).
Field, M. G. et al. Punctuated evolution of canonical genomic aberrations in uveal melanoma. Nat. Commun. 9, 116. https://doi.org/10.1038/s41467-017-02428-w (2018).
Stalhammar, G. & Gill, V. T. Digital morphometry and cluster analysis identifies four types of melanocyte during uveal melanoma progression. Commun. Med. (Lond). 3, 60. https://doi.org/10.1038/s43856-023-00291-z (2023).
Yavuzyigitoglu, S. et al. Radiological patterns of uveal melanoma liver metastases in correlation to genetic status. Cancers (Basel) 13. https://doi.org/10.3390/cancers13215316 (2021).
Stalhammar, G., Hagstrom, A., Ermedahl Conradi, M. & Williams, P. A. Choroidal nevi and melanoma doubling times and implications for delays in treatment: A systematic review and meta-analysis. Surv. Ophthalmol. 70, 38–46. https://doi.org/10.1016/j.survophthal.2024.09.004 (2025).
Stalhammar, G. & Gill, V. T. The long-term prognosis of patients with untreated primary uveal melanoma: A systematic review and meta-analysis. Crit. Rev. Oncol. Hematol. 172, 103652. https://doi.org/10.1016/j.critrevonc.2022.103652 (2022).
Stalhammar, G. Delays between uveal melanoma diagnosis and treatment increase the risk of metastatic death. Ophthalmology 131, 1094–1104. https://doi.org/10.1016/j.ophtha.2023.11.021 (2024).
Damato, B. et al. Deferral of treatment for small choroidal melanoma and the risk of metastasis: An investigation using the Liverpool uveal melanoma prognosticator online (LUMPO). Cancers (Basel) 16. https://doi.org/10.3390/cancers16081607 (2024).
Hagström, A., Witzenhausen, H. & Stålhammar, G. Tailoring surveillance imaging in uveal melanoma based on individual metastatic risk. Can. J. Ophthalmology/Journal Canadien D’ophtalmologie. https://doi.org/10.1016/j.jcjo.2024.07.014 (2024).
Damato, B. Does ocular treatment of uveal melanoma influence survival? Br. J. Cancer. 103, 285. https://doi.org/10.1038/sj.bjc.6605765 (2010).
Kujala, E., Mäkitie, T. & Kivelä, T. Very long-term prognosis of patients with malignant uveal melanoma. Invest. Ophthalmol. Vis. Sci. 44, 4651–4659 (2003).
Decatur, C. L. et al. Driver mutations in uveal melanoma: associations with gene expression profile and patient outcomes. JAMA Ophthalmol. 134, 728–733. https://doi.org/10.1001/jamaophthalmol.2016.0903 (2016).
Mariani, P. et al. Surgical management of liver metastases from uveal melanoma: 16 years’ experience at the Institut curie. Eur. J. Surg. Oncol. 35, 1192–1197. https://doi.org/10.1016/j.ejso.2009.02.016 (2009).
The Collaborative Ocular Melanoma. Study (COMS) randomized trial of pre-enucleation radiation of large choroidal melanoma II: initial mortality findings. COMS report 10. Am. J. Ophthalmol. 125, 779–796. https://doi.org/10.1016/s0002-9394(98)00039-7 (1998).
Richtig, E., Langmann, G., Mullner, K., Richtig, G. & Smolle, J. Calculated tumour volume as a prognostic parameter for survival in choroidal melanomas. Eye (Lond). 18, 619–623. https://doi.org/10.1038/sj.eye.6700720 (2004).
Uner, O. E., See, T. R. O., Szalai, E., Grossniklaus, H. E. & Stalhammar, G. Estimation of the timing of BAP1 mutation in uveal melanoma progression. Sci. Rep. 11, 8923. https://doi.org/10.1038/s41598-021-88390-6 (2021).
Funding
Open access funding provided by Karolinska Institute.
Support for this study was provided to Gustav Stålhammar from: (1) Region Stockholm (RS-2019-1138). (2) The Swedish Cancer Society (20 0798 Fk). (3) The Crown Princess Margareta Foundation for the Visually Impaired (2022-017). (4) Karolinska Institutet (2022 − 01671). (6) The Swedish Eye Foundation (2022-05-09). The sponsors or funding organizations had no role in the design or conduct of this study.
Author information
Authors and Affiliations
Contributions
Serdar Yavuzyigitoglu: Conceptualization, Methodology, Data curation, Investigation, Resources, Writing – Reviewing and Editing. Shiva Sabazade: Writing- Reviewing and Editing, Validation. Viktor Gill: Writing- Reviewing and Editing, Validation. Erwin Brosens: Supervision, Writing – Reviewing and Editing. Emine Kiliç: Methodology, Data curation, Project Administration. Gustav Stålhammar: Conceptualization, Methodology, Formal analysis, Investigation, Writing -Original Draft, Visualization, Supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yavuzyigitoglu, S., Sabazade, S., Gill, V. et al. Higher AJCC stage at primary tumor diagnosis may predict shorter survival in metastatic uveal melanoma. Sci Rep 15, 19557 (2025). https://doi.org/10.1038/s41598-025-03961-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-03961-1