Abstract
Substantial uncertainties remain regarding the establishment of an optimal examined lymph node (ELN) count for the comprehensive management of resectable colorectal cancer (CRC). The correlation of the ELN count with cancer-specific survival (CSS) and benefit from adjuvant chemotherapy (AC) in CRC was investigated using a large database, and the minimal threshold for ELN count in LN-negative patients was determined. The data on stage I to III CRC available in the SEER database (2010–2015) were analyzed to determine the correlation of ELN count with CSS and the benefit of AC using multivariable models. The series of odds ratios (ORs) and hazard ratios (HRs) were fitted using the join-point regression analysis. External validation was performed using the data of patients with stage I to III CRC (2004 to 2009) available in the SEER database. Among LN-negative patients, both cohorts indicated that an increase in the ELN count (≤ 18) led to incremental enhancements in CSS, while no additional improvement in CSS was noted beyond an ELN count of 18. Notably, the efficacy of AC diminished gradually as the ELN count increased. Moreover, post-AC CSS was impaired when the ELN count exceeded 18 (serial HRs > 1), a trend that was accentuated with a higher ELN count. Among the LN-positive patients, two cohorts exhibited proportional increases in ELNs, from one positive LN (PLN) to 20 PLN disease, and incremental benefit from AC, as the ELN count increased (serial HRs < 1). The present study recommends an ELN threshold of 18 when evaluating the quality of prognostic stratification and guiding AC for LN-negative cases. A higher ELN count would be associated with further accurate PLN detection and incremental benefit from AC in LN-positive diseases.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) ranks third among all cancers in terms of incidence and second in terms of mortality, with approximately 1.9 million new cases and 935,000 associated deaths recorded worldwide every year, accounting for nearly 10% of all cancer cases and deaths1. Approximately 75% of CRC patients are eligible for radical surgery upon diagnosis2, and postoperative management is primarily guided by histopathological evaluation3,4. Combined adjuvant chemotherapy (AC) comprising the use of fluorouracil and oxaliplatin is recommended for patients with positive lymph nodes (PLNs) or those without LN metastasis along with pT4, low differentiation, < 12 LNs, obstruction/perforation, and vascular and lymphatic invasion3,4. Nonetheless, the 5-year recurrence rate among LN-positive patients remained between 50% and 60%5, and despite a better prognosis noted for LN-negative patients, 25–30% of the patients experience recurrent disease5. It is reported that AC significantly reduces the risk of recurrence and mortality in LN-positive patients6. However, the absolute benefit of AC for LN-negative patients is limited, just 4%7. Therefore, numerous studies have been actively exploring factors that influence survival and the benefits of AC in potentially curable non-metastatic CRC, particularly in LN-negative patients. It has been found that the status of regional LN is one of the most important prognostic factors8.
Recent decades have witnessed a significant focus on the impact of involvement of LN as a key determinant of prognosis and treatment strategy in patients with resectable CRC9. Consequently, during radical surgery, surgeons strive to remove as many regional LNs as possible to eliminate the chances of residual PLNs. Therefore, the current clinical guidelines stipulate a minimum number of 12 examined LNs (ELNs), but this cutoff value has long been questioned and challenged due to the unclear potential relationship between this threshold and patient survival benefits. This indicate that the current clinical guidelines and practices are inadequate, leading to numerous studies conducting challenging investigations on this issue. However, none of these studies have so far reached a consensus on a specific ELN threshold and have rather reported a wide range of ELN thresholds (between 6 and 40)10,11,12. Such studies have, therefore, raised numerous concerns regarding their inconclusive results, such as the arbitrary selection of the ELN threshold, univariate analyses conducted without accounting for the other prognostic factors, and unconvincing outcomes from non-aggregated data analyses. Consequently, determining the ELN count threshold remains a debatable topic, particularly in regard to LN-negative patients, particularly in regard to LN-negative patients.
AC is inherently a double-edged sword: On one hand, it effectively eliminates rapidly dividing cancer cells; on the other hand, it suppresses bone marrow function and the antitumor immune system, thereby reducing the body’s overall antitumor immune capacity13. Based on this duality, we propose the following hypotheses: (1) Therapeutic Benefit: When the cancer-killing effect of AC predominates over its immunosuppressive effects, patients are likely to benefit from AC. (2) Neutral Impact: When the cancer-killing effect of AC is comparable to its suppressive effect on the antitumor immune system, patients may not derive significant clinical benefit from AC. (3) Adverse Outcome: When the immunosuppressive effect of AC outweighs its antitumor efficacy, it may adversely affect patient prognosis.
Previous studies have demonstrated that LN-negative patients exhibit detectable levels of antitumor CD8+ T cells in both LNs and peripheral blood, whereas LN-positive patients show a compromised ability to generate these antitumor CD8+ T cells in these compartments14. Based on these findings, we propose the following hypotheses: (1) Low ELN Counts in LN-Negative Patients: In LN-negative patients with low ELN counts, systemic antitumor immune capacity remains relatively low (excluding cases where positive or micrometastatic LNs were missed due to technical limitations in pathological sampling)15,16. In such patients, the cancer-killing effects of AC would likely predominate, thereby conferring clinical benefit. (2) High ELN Counts in LN-Negative Patients: When ELN counts are high, patients are likely to possess enhanced systemic antitumor immunity15,16. In this scenario, AC’s immunosuppressive effects on antitumor immune mechanisms might outweigh its therapeutic benefits, potentially rendering AC ineffective or even detrimental. (3) LN-Positive CRC Patients: For LN-positive patients, where regional LN metastasis has already disrupted the generation of antitumor CD8+ T cells in both LNs and peripheral blood, AC may provide universal clinical benefit regardless of immune status. For instance, an international multicenter large cohort study suggested that the Consensus ImmunoScore, derived from CD3 + and CD8 + T cells within the primary tumor, effectively predicts the response to AC in stage III CRC patients17. In summary, the relationship between ELN and the benefits of AC for resectable CRC remains unclear, particularly in lymph LN-negative patients.
In this context, the present study involved investigating the intricate correlation of ELN dissection with cancer-specific survival (CSS), the efficacy of AC, and the detection of PLNs in patients with resectable CRC. The investigation was conducted using the data from two extensive public cohorts comprising consecutive cases documented over a 12-year period. The study aimed to provide substantial macro-level evidence for guiding and refining the treatment approaches and future research endeavors for the management of resectable CRC.
Materials and methods
Patient population
The Surveillance, Epidemiology, and End Results (SEER) database, which comprises the data of nearly 35% of the US population documented across 18 cancer registries, was used in the present study to determine the effect of ELN count on CSS and AC benefit in resected CRC. The SEER*Stat version 8.4.1 software was employed to extract the information on the CRC cases published in the SEER public access database between 2004 and 2015. The cohort from the 2010–2015 period, categorized according to the seventh edition TNM classification, constituted the training set, while the cohort from the 2004–2009 period, classified according to the sixth edition AJCC Cancer Staging Manual, formed the validation set. The patients in the training set were consistently reviewed and staged following the eighth edition AJCC Cancer Staging Manual. In contrast, the patients in the validation set were not restaged due to a lack of sufficient data such as tumor deposits.
Patient selection
The eligible participants included those who had undergone surgical resection for primary CRC at stage I to III and had at least one ELN. The individuals with missing ELN/PLN count data, stage IV disease, incomplete surgery, neoadjuvant radiotherapy and chemotherapy, overall survival of less than three months, death due to other causes, and unknown AC or CSS status were not eligible for inclusion in the present study. This study was reported in line with the STROCSS criteria18.
Study variables
Right-sided CRC included tumors in the cecum, ascending colon, hepatic flexure, and transverse colon. Left-sided CRC included tumors in the splenic flexure, descending colon, sigmoid colon, and rectum. The definition and calculation of the log odds of positive lymph nodes (LODDS) are described in the literature19. The PLN ratio was the ratio of PLNs to ELNs.
Statistical analyses
The χ2 test was conducted to compare the differences for categorical variables. A t-test was conducted to determine the differences in the case of continuous variables. The Cox proportional hazard regression model was adopted to determine the effect of ELN count on CSS and the benefit of AC and then adjusted according to the other significant prognostic factors such as sex, age, marital status, grade, T stage, carcinoembryonic antigen (CEA) level, and peripheral nerve invasion. Assuming that a higher ELN represented a greater opportunity to identify PLNs, a binary logistic regression model was established to determine the correlation between the ELN count and PLN detection (with one PLN used as the reference) after adjusting for other potential confounders. The ‘survival’, ‘survminer’, and ‘rms’ modules in the R package were employed to conduct the above analyses. In addition, survival curves were generated based on the results of the Kaplan-Meier analysis and compared using a log-rank test.
Join-point regression analysis
The join-point regression model, alternatively referred to as piecewise regression, broken line regression, or multi-phase regression, was introduced by Kim in 200020. The model involves partitioning a long trend line into several segments, each of which is characterized by continuous linearity. The segmentation points in the joinpoint regression model are determined based on a hypothesis using the Z-test, which improves the segmentation and results in a better fit. In the present study, the join-point regression analysis was performed to analyze the dynamic trend of ELN count.
Fitting of curves and determination of breakpoints
The join-point regression analysis was performed using the NCI Joinpoint Regression software (version 4.8.0.1; Information Management Services Inc.) to obtain the hazard ratios (HRs) for CSS and the odds ratios (ORs) for PLN detection, based on which each ELN or PLN count was compared with one ELN or PLN reference, and the HRs of the AC vs. non-AC in each ELN or PLN subgroup. In addition, the dynamic trend of ELN count was determined using the join-point regression analysis. The software was configured to fit the breakpoints with a maximum of five join points for a series of HRs or ORs.
Results
Patient characteristics
A total of 107,071 patients in the training set and 70,750 patients in the validation set fulfilled the eligibility criteria and were included in the present study. The baseline characteristics of each cohort are listed in Table 1. The distribution of ELN count differed between the two cohorts; the training set had a larger ELN count (median, 17; interquartile range, 13 to 23) compared to the validation set (median, 15; interquartile range, 10 to 21).
ELN break point analysis for the patients with LN-negative CRC
Figure 1 depicts the fitting curves and the corresponding structural breakpoints for the HRs of CSS in LN-negative CRC. After adjusting for the confounding factors such as sex, age, marital status, T stage, grade, CEA level, and peripheral nerve invasion, all obtained breakpoints were essentially in agreement with one another (Fig. 1A and F; Table S1 and S2). According to the current TNM classification system, a threshold of 18 ELNs was generated based on the training set (≤ 18 ELNs, average HR value of change [AHRC] = − 0.0565). The internal validation sets from both cohorts also demonstrated that an ELN count of 18 was the best for determining patient survival, independent of the tumor site (Fig. 1B, C, E and F; Table S1 and S2).
Prognostic implication when using 18 ELNs as the breakpoint in LN-negative CRC
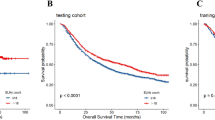
The LN-negative patients were divided into three groups based on their ELN counts – <18 ELN group, 18 ELN group, and > 18 ELN group. The Kaplan-Meier analysis revealed a significant difference in CSS between the < 18 ELN group and the remaining two groups (Fig. 2A and B: training set, both P < 0.001; validation set, both P < 0.001), while no statistical difference was noted in the CSS between the 18 ELN group and the > 18 ELN group (Fig. 2A and B, all P > 0.05). Subsequently, a multivariate analysis was conducted using the 18 ELN group as a reference, which revealed that the CSS of serial < 18 ELN subgroups was significantly worse (Table S3, almost all P < 0.05), while the CSS of serial > 18 ELN subgroups did not exhibit statistically significant differences (Table S3, almost all P > 0.05). These results demonstrated that an ELN count of 18 was the optimal breakpoint for a distinct prognosis.
Dynamic trends of insufficient ELN count in LN-negative CRC
Further analysis revealed that among the LN-negative patients, 55.45% of the patients in the training set had less than 18 ELNs (Fig. 3A), while 64.68% of the patients in the validation set had less than 18 ELNs (Fig. 3B). This led to substantially impacted patient survival. The dynamic trend of ELN count was evaluated based on the data documented from 2004 to 2015, and it was observed that the insufficient ELN count decreased at − 3.43% per year, significantly during the 2004–2009 period, following which it gradually decreased from 2009 to 2015 at − 0.75% per year (Fig. 3C).
Functional LN dissection results in impaired benefit from AC in LN-negative CRC
The efficacy of AC for LN-negative CRC was analyzed for the serial ELN subgroups of the training set. After excluding the other potentially significant prognostic factors, it was observed that with the increase in the ELN count, the HR value (AC vs. non-AC) also increased gradually, suggesting that the benefit of AC gradually decreased with the increase in the ELN count (Fig. 4A, serial HRs < 1, AHRC = 0.0081). Importantly, as the ELN count continued to increase, AC would result in impaired CSS (Fig. 4A, serial HRs > 1, AHRC = 0.0081). Further analysis of the three categories of patients revealed that the < 18 ELN group (Table 2, HR = 1.00, 95% CI [0.91–1.11], P = 0.96) and the 18 ELN group (Table 2, HR = 0.93, 95% CI [0.65–1.33], P = 0.69) received no benefit from AC, while the CSS of AC in the > 18 ELN group was significantly worse (Table 2, HR = 1.17, 95% CI [1.04–1.33], P = 0.01). These results suggested that an ELN count of less than 18 could be related to PLN or micrometastatic LN residue, thereby leading to no CSS damage. On the other hand, when the ELN count is greater than 18, it could often imply that the functional LN has been cleaned and damaged its anti-tumor function, resulting in impaired CSS.
Fitting curves of serial HRs for CSS (AC vs. non-AC) based on the join-point regression analysis. (A) All LN-negative patients in the training set. (B) Patients with the T1–2N0 diseases in the training set. (C) Patients with the T1–2N0 diseases in the validation set. Blue line, fitting curve; Red line, HR = 1.0.
In order to further verify that functional LN dissection is associated with impaired benefit after AC, T1–2N0 patients with a low probability of LN metastasis were specially selected from both training and validation sets for the verification of this hypothesis. The analysis results revealed that with an increase in the ELN count, the impaired CSS after AC became increasingly evident (Fig. 4B and C: training set, serial HRs > 1, AHRC = 0.0045; validation set, serial HRs > 1, AHRC = 0.0408), demonstrating that functional LN dissection is closely related to impaired CSS after AC.
Rising trends in excessive ELN count challenge the AC strategy for LN-negative CRC
As stated above, the > 18 ELN group and the 18 ELN group did not exhibit a statistical difference in CSS, although the benefit from AC suggests that unnecessary functional LN dissection should be avoided in clinical practice. According to the dynamic trend of ELN count observed in the data from the SEER database, the excessive ELN count increased significantly at 3.04% per year during 2004–2008 and then gradually increased from 2008 to 2015 at 1.03% per year (Fig. 3C). According to the current clinical guidelines, it is reasonable not to recommend ELNs ≥ 12 for postoperative AC, although the recommendation, according to the present study, would be to adjust the break point to an ELN count of 18.
However, it is of concern that in the > 18 ELN group’s stage II CRC data, 47.64% (7987/16765) and 11.84% (1092/9221) of the patients in the training set and validation set, respectively, continued to have at least one high-risk factor recommended for postoperative AC. Indeed, these percentages should be higher as information on certain high-risk factors, such as lymph vascular invasion, is missing from the SEER database, particularly for the validation set. This suggests that the simultaneous existence of various factors should be comprehensively considered to develop an optimal AC strategy for LN-negative patients.
ELNs improve the detection of PLNs and do not affect the benefit from AC in LN-positive CRC
The analysis of the distribution of PLN count in the data of both training and validation sets revealed that the percentage of PLN count decreased rapidly at first and then declined slowly, becoming almost negligible for PLNs > 20 (Fig. 5A and B). Therefore, only 1 ~ 20 PLNs were selected for subsequent analyses.
Next, using one PLN group as the reference, a migration relationship was noted between ELNs and PLNs in LN-positive CRC; that is, the increase in the ELN count would gradually increase the detection rate of PLNs (Fig. 5C, serial ORs > 1; training set, average OR value of change [AORC] = 0.0023; validation set, AORC = 0.0026), indicating that ELNs formed the basis for PLN detection. Moreover, in both cohorts, successive increases in ELNs did not affect the efficacy of AC for LN-positive CRC (Fig. 5D, serial HRs < 1, training set, AHRC = − 0.0013; validation set, AHRC = 0.0000). Accordingly, a breakpoint for ELN count was not established for LN-positive CRC.
Serial PLNs predict a gradual increase in poor CSS and benefit from AC in LN-positive CRC
PLN count is the diagnostic and stratification criterion for the N stage. Therefore, the relationship between PLNs and CSS was investigated, and it was revealed that CSS progressively deteriorated with successive increases in PLNs (Fig. 6A, serial HRs > 1; training set, 1 ~ 13 PLNs: AHRC = − 0.0964; validation set, 1 ~ 20 PLNs: AHRC = − 0.0810), indicating that the current guideline of using 6 PLNs as the breakpoint is not ideal. In addition, increasing PLN count was associated with a continuously increasing benefit from AC (Fig. 6B, serial HRs < 1; training set, AHRC = − 0.0177; validation set, AHRC = − 0.0194), indicating that patients with a greater PLN count benefit more from AC, which demonstrated the feasibility of greater intensity AC for patients with a greater PLN count.
Discussion
In the field of research on resectable CRC, the debate regarding the establishment of an optimal threshold for ELN count has continued for over 30 years. However, despite multiple studies, no consensus has been reached so far due to various limitations, such as those related to the sample size of the study and the analytical methods used. Importantly, previous research has primarily been focused on the apprehension that insufficient ELNs could overshadow PLNs, while the influence of normally functioning negative LNs on patient systemic immunity, prognosis, and AC effectiveness has been disregarded. According to the present, for the LN-negative disease, an ELN count of 18 is the optimal threshold with no impact on patient survival, while excessive ELNs are associated with diminished benefits from AC. In the case of LN-positive disease, on the other hand, it was revealed that after adjusting for the other risk factors associated with LN involvement, increased ELN count could indicate a mitigated risk of undetected PLNs, potentially facilitating a further comprehensive eradication of residual disease and enhancing the long-term survival.
Since 1992, the guideline recommending a minimum of 12 retrieved ELNs in CRC has remained a topic of debate in the scientific community10,11,12. Unfortunately, previous studies have produced no consistent and reliable conclusions, mostly due to limitations related to the sample size and analysis methods. After accounting for the confounding factors, the present study conclusively demonstrated that an ELN count of less than 18 was revealed as a significant factor contributing to impaired CCS in LN-negative patients, while no substantial increase in CSS was noted beyond 18 ELNs. Therefore, it was speculated that an ELN count of 18 is the optimal threshold for determining the prognosis of LN-negative patients, with extensive data from both internal and external validation sets supporting this finding.
Current research demonstrates that tumor-drainingLNs serve as critical sites for tumor antigen presentation and antitumor immune activation, playing a pivotal role in antitumor immunity21. Therefore, ELN count reflects both the host’s antitumor immune status and distinct tumor biological characteristics (e.g., microsatellite instability patients typically exhibit higher ELN counts). Building on this evidence, we propose the following hypothesis: An elevated ELN count may indicate enhanced systemic antitumor immune competence, and increased tumor antigen expression/release capable of activating tumor-specific immune responses. This dual mechanism may underlie the observed association between higher ELN counts and improved patient prognosis.
AC inherently functions as a double-edged sword. we therefore speculate that in patients with compromised antitumor immune responses (e.g., ELN < 18), the cancer-killing effects of AC may outweigh its immunosuppressive effects on antitumor immunity. Conversely, in patients exhibiting robust antitumor immunity (e.g., ELN > 18), AC’s immunosuppressive effects might dominate over its tumoricidal activity. For instance, a recent preclinical model demonstrated that oxaliplatin induced liver damage and created an inhibitory microenvironment, resulting in a notable increase in CRC liver metastasis13. As expected, in LN-negative patients, the benefit of AC was observed to gradually decrease as the number of ELNs increased in the present study. Our study indicated for the first time that a higher ELN count may decrease or impair AC efficacy. However, the potential mechanism of ELN count affecting the efficacy of AC remains unclear. We speculate that, excluding cases where PLNs or micrometastatic LNs were missed due to technical limitations in pathological sampling, it may be related to the immunosuppressive effects of AC outweighing its cancer-killing effects.
Previous studies have demonstrated that LN-negative patients exhibit detectable levels of antitumor CD8+ T cells in both LNs and peripheral blood, whereas LN-positive patients show a compromised ability to generate these antitumor CD8 + T cells in these compartments14. Therefore, in LN-positive patients, where regional LN metastasis has already disrupted the generation of antitumor CD8 + T cells in both LNs and peripheral blood, AC can provide universal clinical benefit. This is consistent with our observations in LN-positive patients.
The current standard treatment for LN-negative CRC advocates radical surgical resection with regional LN dissection. The findings of the present study indicated an increasing trend of excessive LN dissection. Given that surgical methods and the extent of LN dissection for CRC have become highly standardized, the increasing number of ELNs can be attributed to enhanced compliance with these surgical standards and the meticulous efforts of pathologists in tissue sampling. According to the clinical guidelines, AC is deemed necessary for LN-negative cases with T4, low differentiation, obstruction/perforation, and vascular and lymphatic invasion3,4. However, this therapeutic strategy was not beneficial for patient survival22. The present study may partly explain the lack of benefit from AC for stage II CRC patients, highlighting the requirement of comprehensively considering multiple high-risk and protective factors when formulating AC strategies.
LN metastasis is a crucial independent prognostic factor significantly impacting the survival of patients with CRC23. The present study identified a direct positive correlation between the retrieval of ELNs and PLNs in LN-positive patients. This correlation provided a foundation for the detection of PLNs. Importantly, it was revealed that the retrieval of ELNs does not impact the efficacy of AC. According to these findings, a specific threshold for ELNs could not be established for the LN-positive patients. Furthermore, the PLN ratio and the LODDS are superior prognostic stratification indices for the LN-positive disease (Figure S1A-S1B). The present study also revealed a continuous linear relationship between PLNs and CSS, indicating that the current guideline of using 7 PLNs as a threshold for N staging is unreasonable. Similar findings have been reported in previous studies. For instance, in rectal cancer, patients with only one PLNs presented a 3–10% higher 5-year survival rate compared to those with 2 to 3 PLN, while patients with 4 to 6 PLNs exhibited a 5–20% higher 5-year survival rate compared to those with 7 or more PLNs24. Moreover, the benefit from AC demonstrated a linear correlation with increasing PLNs, indicating that indiscriminately administering AC for LN-positive patients is inappropriate and that the patients with a greater PLN count could require further intensive AC. In this context, the results of the IDEA study provide a certain level of evidence, according to which three months of AC suffices for low-risk LN-positive patients (T1–3N1), while the high-risk LN-positive patients (T4 or N2) should ideally receive six months of AC25.
As with all research, the present study also had certain limitations, such as its retrospective design and lack of validation from concurrent and prospective independent cohorts. However, the conclusions were drawn based on the data from a large public database, and are, therefore, highly reliable and reproducible. Second, although assessing the clinical relevance of additional LN testing necessitates further prospective studies focused on patient outcomes and prognosis, the present study yielded numerous novel findings, providing solid evidence for the concepts of ELN dissection and guidance for AC in patients with different LN statuses. Third, due to the absence of certain variable data in the SEER database (e.g., advancements in surgical techniques, inter-institutional variability, inherent imbalances in the distribution of prognostic factors, types of chemotherapy), our analysis could not fully adjust for all potential confounding factors. This incomplete adjustment may compromise the accuracy and interpretation of the findings. Fourth, given that the SEER database primarily comprises patient data from the US, the generalizability of our results to other geographic regions or healthcare systems remains uncertain. Therefore, we recommend that future multicenter studies incorporate international datasets and implement more comprehensive adjustments for potential confounders to enhance both the generalizability and reliability of research outcomes. Fifth, it is critical to note that for rectal cancer patients (T3, N+), the primary treatment modality involves preoperative radiotherapy or chemoradiotherapy. Therefore, caution should be exercised when directly applying these findings to this specific population, as their therapeutic regimens and prognostic profiles may substantially differ from those of colon cancer patients. Potential limitations may exist when generalizing these results to rectal cancer patients undergoing preoperative radiotherapy or chemoradiotherapy.
Conclusion
The present study recommends using an ELN count of 18 as a threshold for evaluating the quality prognostic stratification and guiding the AC for LN-negative diseases. A greater ELN count is associated with a further accurate detection of PLNs and sustained benefit from AC in LN-positive cases of the disease.
Data availability
The data in this study are accessible through the SEER database (https://seer.cancer.gov/data-software/) and do not contain confidential information.
Abbreviations
- CRC:
-
Colorectal cancer
- ELN:
-
Examined lymph node
- PLN:
-
Positive lymph node
- CSS:
-
Cancer-specific survival
- AC:
-
Adjuvant chemotherapy
- OR:
-
Odds ratio
- AORC:
-
Average OR value of change
- HR:
-
Hazard ratio
- AHRC:
-
Average HR value of change
- PNI:
-
Peripheral nerve invasion
- IQR:
-
Interquartile range.
- LODDS:
-
Log odds of positive lymph nodes
- CEA:
-
Carcino-embryonic antigen
References
Sung, H. et al. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249. https://doi.org/10.3322/caac.21660 (2021).
SiegelRL et al. Colorectal cancer statistics, 2020. CA Cancer J. Clin. 70, 145–164. https://doi.org/10.3322/caac.21601 (2020).
Argilés, G. et al. Localised colon cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 31, 1291–1305. https://doi.org/10.1016/j.annonc.2020.06.022 (2020).
Benson, A. B. et al. American society of clinical oncology recommendations on adjuvant chemotherapy for stage II colon cancer. J. Clin. Oncol. 22, 3408–3419. https://doi.org/10.1200/jco.2004.05.063 (2004).
Jemal, A. et al. Cancer statistics, 2006. CA Cancer J. Clin. 56, 106–130. https://doi.org/10.3322/canjclin.56.2.106 (2006).
Taieb J., Gallois C. Adjuvant Chemotherapy for Stage III Colon Cancer. Cancers. 12, 2679. https://doi.org/10.3390/cancers12092679 (2020).
Sargent, D. et al. Evidence for cure by adjuvant therapy in colon cancer: observations based on individual patient data from 20,898 patients on 18 randomized trials. J. Clin. Oncol. 27, 872–877. https://doi.org/10.1200/jco.2008.19.5362 (2009).
Chen, K., Collins, G., Wang, H. & Toh, J. W. T. Pathological features and prognostication in colorectal Cancer. Curr. Oncol. (Toronto Ont.). 28 (6), 5356–5383 (2021).
Resch, A. & Langner, C. Lymph node staging in colorectal cancer: old controversies and recent advances. World J. Gastroenterol. 19 (46), 8515–8526. https://doi.org/10.3748/wjg.v19.i46.8515 (2013).
McDonald, J. R., Renehan, A. G., O’Dwyer, S. T. & Haboubi, N. Y. Lymph node harvest in colon and rectal cancer: current considerations. World J. Gastrointest. Surg. 4, 9–19. https://doi.org/10.4240/wjgs.v4.i1.9 (2012).
Valsecchi, M. E., Leighton, J. Jr. & Tester, W. Modifiable factors that influence colon cancer lymph node sampling and examination. Clin. Colorectal Cancer. 9, 162–167. https://doi.org/10.3816/CCC.2010.n.022 (2010).
Noura, S. et al. Impact of metastatic lymph node ratio in node-positive colorectal cancer. World J. Gastrointest. Surg. 2, 70–77. https://doi.org/10.4240/wjgs.v2.i3.70 (2010).
Ma, Y., Guo, C., Wang, X., Wei, X. & Ma, J. Impact of chemotherapeutic agents on liver microenvironment: oxaliplatin create a pro-metastatic landscape. J. Exp. Clin. Cancer Res. 42, 237. https://doi.org/10.1186/s13046-023-02804-z (2023).
Rahim, M. K. et al. Dynamic CD8(+) T cell responses to cancer immunotherapy in human regional lymph nodes are disrupted in metastatic lymph nodes. Cell 186, 1127–1143e1118. https://doi.org/10.1016/j.cell.2023.02.021 (2023).
Lal, N., Chan, D. K. H., Ng, M. E., Vermeulen, L. & Buczacki, S. J. A. Primary tumour immune response and lymph node yields in colon cancer. Br. J. Cancer. 126, 1178–1185. https://doi.org/10.1038/s41416-022-01700-1 (2022).
Lee, S. H. et al. Immune microenvironment and lymph node yield in colorectal cancer. Br. J. Cancer. 129, 917–924. https://doi.org/10.1038/s41416-023-02372-1 (2023).
Mlecnik, B. et al. Multicenter international society for immunotherapy of Cancer study of the consensus immunoscore for the prediction of survival and response to chemotherapy in stage III Colon Cancer. J. Clin. Oncol. 38, 3638–3651. https://doi.org/10.1200/JCO.19.03205 (2020).
Mathew, G. et al. STROCSS 2021: strengthening the reporting of cohort, cross-sectional and case-control studies in surgery. Int. J. Surg. 96, 106165. https://doi.org/10.1016/j.ijsu.2021.106165 (2021).
Li, T. et al. Prognostic implications of ENE and LODDS in relation to lymph node-positive colorectal cancer location. Transl. Oncol. 14, 101190. https://doi.org/10.1016/j.tranon.2021.101190 (2021).
Kim, H. J., Fay, M. P., Feuer, E. J. & Midthune, D. N. Permutation tests for joinpoint regression with applications to cancer rates. Stat. Med. 19, 335–351. https://doi.org/10.1002/(sici)1097-0258(20000215)19:33.0.co;2-z (2000).
Delclaux, I., Ventre, K. S., Jones, D. & Lund, A. W. The tumor-draining lymph node as a reservoir for systemic immune surveillance. Trends. Cancer https://doi.org/10.1016/j.immuni.2019.01.010 (2023).
O'Connor, E. S. et al. Adjuvant chemotherapy for stage II colon cancer with poor prognostic features. J. Clin. Oncol. 29, 3381–3388. https://doi.org/10.1200/JCO.2010.34.3426 (2011).
Compton, C. C. et al. Prognostic factors in colorectal cancer. College of American pathologists consensus statement 1999. Arch. Pathol. Lab. Med. 124, 979–994. https://doi.org/10.5858/2000-124-0979-pficc (2000).
Gunderson, L. L., Jessup, J. M., Sargent, D. J., Greene, F. L. & Stewart, A. Revised tumor and node categorization for rectal cancer based on surveillance, epidemiology, and end results and rectal pooled analysis outcomes. J. Clin. Oncol. 28, 256–263. https://doi.org/10.1200/jco.2009.23.9194 (2010).
Grothey, A. et al. Duration of adjuvant chemotherapy for stage III Colon cancer. N. Engl. J. Med. 378, 1177–1188. https://doi.org/10.1056/NEJMoa1713709 (2018).
Acknowledgements
The authors acknowledge each editor and reviewer for their profound insight in this study.
Funding
This work was supported by the Talent Introduction Fund of Qingdao Municipal Hospital (grant number NA to T.F.).
Author information
Authors and Affiliations
Contributions
T.F. designed and supervised the project. R.S.X., B.Y and J.C.F performed the data curation and analysis. R.S.X., B.Y. and T.F. analyzed and interpreted the results. R.S.X, B.Y., T.F. and J.W. drafted and reviewed the manuscript. R.S.X., J.F.S and J.F.W made detailed modifications to the manuscript during the revision process.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xiang, R., Yang, B., Sun, J. et al. Effect of examined lymph node count on precise cancer-specific survival and effectiveness of adjuvant chemotherapy in resected colorectal cancer. Sci Rep 15, 25856 (2025). https://doi.org/10.1038/s41598-025-03999-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-03999-1