Abstract
The present study aimed to investigate the influence of the applied fertilizer (agricultural digestate) on antioxidative properties and antimicrobial activity of leaves of hemp ‘Futura75’ cultivar, valuable in the nutritional process. An attempt was also made to establish correlations between the antioxidative properties and antimicrobial activity of the leaves and fertilizer doses. The total polyphenol content (TPC) and free radical scavenging activity were determined by means of the spectrophotometric method, whereas the polyphenol profile was identified with the high-performance liquid chromatography (HPLC). Microbiological analyses were conducted using the following test strains: Staphylococcus aureus, Pseudomonas aeruginosa, Candida albicans, and Aspergillus niger. The highest contents of polyphenols and flavonoids and the highest DPPH radical scavenging activity were determined in the hemp leaves at the early growth phase (I harvest date) after digestate application in a dose of 30 L (i.e., 35.84 mg·GAE·g−1 DW, 59.30 mg QE·g−1 DW and 78.36mmol Trolox·kg−1, respectively). The applied digestate affected also the antimicrobial activity of hemp leaves when applied in doses of 30 L [18–37%] and 50 L [15–30%], causing a tangible increase in the growth inhibition zones of, particularly, Candida albicans in the leaves harvested in terms I, II and III.
Similar content being viewed by others
Introduction
An increasingly common procedure deployed throughout Europe in the agricultural sector is the use of fermentation products as valuable organic fertilizers, as they effectively reduce nitrogen losses compared to mineral fertilizers, when applied in the same doses1. It is presumed that restoring the components accumulated in the material from the biogas plant to the soil is necessary not only from an economic perspective, but also from the standpoint of maintaining and restoring the ecological homeostasis2. The use of such fertilizer can exert multi-faceted effects on changes in the mobility of nutrients in the soil and the capability of plants for their uptake from the environment3. A properly prepared substrate for plant cultivation will provide the necessary nutrients as well as macroelements and microelements, affecting the rate of plant development and crop yield size.
A key factor in ensuring healthy growth, abundant flowering and excellent quality of the final product – hemp, is to ensure balanced growth conditions through tailored fertilization.
In recent years, the area devoted to hemp cultivation in the EU has increased significantly from 20,540 hectares (ha) in 2015 to 33,020 ha in 2022. Within the same period, hemp production increased from 97,130 tons to 179,020 tons (an increase of 84.3%). In recent years, there has also been growing interest in research into the potential use of industrial hemp in food and nutraceuticals4. Industrial hemp is defined as an annual herb of the genus Cannabis5, whose flowers, seeds, stems, leaves and roots contain less than 0.2–0.3% of tetrahydrocannabinol (THC). This THC level has led to a diverse range of products that can be legally produced from this plant and registered6. Hemp leaves, seeds and oil are used in the industry due to their content of bioactive substances, which exhibit multi-faceted biological properties7, like antioxidative and phytochemical activities. The main factors influencing antioxidative activity include the presence of phenolic or polyphenolic compounds, whereas stability of these compounds depends on the processing conditions. Polyphenols and flavonoids of hemp elicit a wide range of health benefits, which makes this plant even more valuable from the perspective of natural medicine. In addition to providing color pigmentation as well as affecting aroma and taste, they are also implicated in protective effects against diseases such as diabetes, cancer and osteoporosis8,9. Furthermore, some of them can be effective in preventing viral hepatitis, malaria, autoimmune diseases and inflammatory diseases by inhibiting kinase8.
Antioxidative activity of polyphenolic compounds is driven by their free radical scavenging capability and iron reducing potential. Polyphenols represent one of the most important groups of compounds present in C. sativa, like flavonoids, phenol amides and lignan amides, which are specific metabolites of this plant. In addition, they serve various roles in plant defense mechanisms, particularly through their action as antioxidants, which prevents the generation of reactive oxygen species (ROS)10,11. In the case of humans, polyphenols elicit health-promoting benefits due to their cardioprotective and chemoprotective effects12,13, as well as exhibit sedating, antispasmodic, analgesic and many other properties, including prevention of migraine, asthma, and hemorrhoids, and counteracting the symptoms of photophobia10,14.
In addition, the presence of numerous active components in the hemp plant may increase the heart rate, reduce blood pressure and stimulate appetite14. According to literature data, leaves of industrial hemp contain 89 g kg-1 of ethanol extract (EE), 212 g kg-1 of ash and 447 g kg-1 of neutral dietary fiber (NDF)15,16.
As Brugman et al.17 claimed, the overuse of xenobiotics in agricultural crops, observed along with the emergence of antibiotic- and pesticide-resistant strains in agriculture and medicine, may affect the ability of the host to properly interact with the microbiota. According to Langezaal et al.,18, a higher cannabidiol to tetrahydrocannabidiol ratio results in a stronger antimicrobial effect. Compared to the number of studies on human microbiota, there is a dearth of research addressing agricultural cultivation of hemp. This may, presumably, be due to the fact that the microbiota of agricultural microorganisms (fungi, bacteria) is influenced by plant species and genotypes, plant development stages, soil type and environmental conditions. Their interactions with plants as hosts may lead to beneficial, neutral or adverse effects on plant chemical composition19. Fungi found in hemp tissues as surface entities (epiphytes) or internal endophytes are capable of producing toxic secondary metabolites (mycotoxins), several of which are pathogenic to humans.
The current situation and the lack of systematic studies on the multi-faceted properties of hemp leaves have prompted us to investigate the effect of using natural fertilizer (agricultural digestate) on their chemical (antioxidative) and microbiological properties. The study also entailed determinations of the proximate chemical composition (protein, ash, water); contents of amino acids and phytochemicals; the composition of total polyphenols and flavonoids as well as overall antioxidant activity (DPPH•) of hemp leaves. In the final stage of the study, the antimicrobial activity of hemp leaf extracts was examined as affected by digestate dose applied in order to determine their effect on bacteria from the group of Staphylococcus aureus, Pseudomonas aeruginosa, Candida albicans and Aspergillus niger.
Materials and methods
Plant material
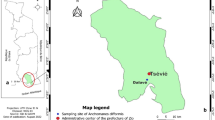
The studied material consisted of leaves of Cannabis sativa L. of ‘Futura75’ cultivar. Seeds of the cultivar were purchased from Polishemp Sp. z o.o. company by the Experimental Station in Czesławice, belonging to the University of Life Sciences in Lublin, Poland (51.304595 22.252620 - DMS: 51º 18’ 16.542” N 22º 15’ 9.432” E). The cultivar’s characteristics were as follows: type – monoecious, growing period – 140 days, height – 250–350 cm, oil content − 28–30%, fiber content – 30–35%, seed yield per 1 ha – 700–900 kg, biomass per 1 ha – 10-12t, and 1,000 seed weight – 16–18 g, as declared by the producer on the label.
Seeds were sown in the first decade of May and harvested in the third decade of September 2022. Plant leaves (n = 7 in three replications) were sampled from each plot on which 4 fertilization variants were applied. The samples were collected in the four last months of hemp growth, with the first sample collection date in the mid-June (BBCH 5), the second date in mid-July (BBCH 6), the third date in mid-August (BBCH 7), and the fourth date in mid-September (BBCH 8).
Scheme of the field experiment
The field experiment entailed 4 variants differing in digestate dose:
-
I variant – total digestate dose of 25,000 Lha−1 (I dose – 12,500Lha−1, II – 12,500 Lha−1);
-
II variant – total digestate dose 37,500 Lha−1 (I dose – 18,750 Lha−1, II – 18,750 Lha−1);
-
III variant – total digestate dose 50,000 Lha−1 (I dose – 25,000 Lha−1, II – 25,000 Lha−1);
-
IV variant – control plot not fertilized with the digestate.
The above doses of the digestate were established based on the analysis of soil samples. To make description of results easier, individual experimental variants were denoted as follows: control – for the control variant, 20 L for the digestate dose of 25,000 Lha− 1, 30 L – for the digestate dose of 37,500 Lha− 1, and 50 L – for the digestate dose of 50,000 Lha− 1.
Fertilization with the digestate was performed in the spring at two stages of leaf development:
-
the first dose at the BBCH 15 stage (the fifth pair of leaves – folded leaves),
-
the second dose at the BBCH 19 stage (the ninth pair of leaves – folded leaves).
The digestate was sourced from a biogas producing plant in Piaski (Lublin Province, Poland). It was produced from the following components: maize silage (70%), sugar beet pulp (15%), fruit pomace (5%), dairy waste (5%), and manure (5%).
Prior to use, the digestate was analyzed for the concentrations of macroelements and heavy metals (Table 1). The pH value of the digestate used for hemp cultivation ranged from 6.5 to 7.00 and was similar to that of cattle slurry (pH = 7.90). We used natural bovine manure.
Digestate analysis
Concentrations of macroelements, heavy metals, nitrogen and pH in the digestate were analyzed according to the Polish Standard PN-EN ISO/IEC 17,025: 200520, and respective results are presented in (Tables 1 and 2).
Proximate composition of hemp leaves
Contents of water, ash, protein, crude fat, and raw fiber of hemp leaves were determined following standard AOAC methods21. Mean values of three determinations were converted into % of dry matter content.
Determination of the content of non-flavonoid phenols (NFE)
The total polyphenol (TP) content was determined using the Folin-Ciocalteu (F.C.) reagent following methodology posited by Singleton & Rossi22 and expressed in mg of gallic acid equivalents (GAE) per g of dry matter (DM). In turn, the content of non-flavonoid phenols (NFE) was determined using the F.C. reagent after removing flavonoid phenols (FP) by means of formaldehyde, following the method by Kramling & Singleton23 and expressed in µg GAE·g-1 DM. The FP content was computed as a difference between TP and NFP contents and expressed as µg GAE·g-1 DM.
Extraction of polyphenols
The extraction of polyphenols was conducted according to the procedure described by Calzolari et al.,24 with some minor modifications. Sample of hemp leaves (100 mg) were poured with 15 mL of methanol, and the mixture was vortexed for 5 min and then sonicated in the dark at a temperature of 4ºC for 30 min. Afterwards, the mixture was centrifuged at 4,000×g at 4 °C for 10 min, using an OHAUS FC5513 Frontier Micro centrifuge (OHAUS Europe GmbH, Switzerland). The resulting supernatants were combined, filtered through a 0.2 μm syringe filter (26 mm, RC membrane, Phenomenex, CastelMaggiore, Italy), and poured into a chromatographic vial.
Determination of the polyphenol profile with the HPLC method
The analysis of polyphenolic compounds was performed following the method described by Wang et al.25 with some minor modifications. Polyphenols were separated by means of a Flexar chromatograph with a UV-Vis detector (Perkin-Elmer, USA), using a Synergi Fusion RP-80 A column 200 × 4.6 mm (4 μm) Phenomenex (Torrance, USA), thermostated at a temp. of. 30ºC. An 80% solution of acetonitrile in 4.5% formic acid (reagent A) and 2.5% acetic acid (reagent B) was used as the eluent, at a flow rate of 1mL min− 1, according to the following gradient: reagent A concentration was increased linearly from 0 to 15% since minute 0 to minute 7, then to 20% since minute 8 to minute 15, and up to 100% since minute 16 till the end of analysis. After 10 min of column elution at 100% concentration of reagent A and 0% concentration of reagent B, the concentration of solution A was reduced to 0% in order to stabilize the column within 10 min, until successive sample injection. During the analysis, the solutions were degassed in a Merc apparatus. Sample absorbance was registered at: λ = 320 nm for phenolic acids, λ = 340 nm for flavones, λ = 360 nm for flavonols, and λ = 520 nm for anthocyanins. The compounds were identified based on their spectra within the range of 200 to 600 nm and comparison of their retention times with those of respective standards. Results are provided in mg100g− 1 sample dry matter.
Determination of total phenol content
The total phenol content was determined following the Folin-Ciocalteu method described by Singleton and Rossi22, with some minor modifications. A portion of extract (125µL) was mixed with 500µL of deionized water, and then 125µL of the Folin-Ciocalteu reagent were added to the mixture, which was next incubated at room temperature for 6 min. Then, 1.25mL of a 7.5% sodium carbonate solution and 1mL of deionized water were added to the mixture, which was next incubated in the dark for 90 min. Afterwards, sample absorbance was measured at a wavelength of 760 nm using a UV-2600i Spectrophotometer (Shimadzu, Japan). Results of determinations are expressed in mg GAE·g− 1 sample.
Determination of total flavonoid content
The total content of flavonoids in extracts was determined according to the procedure described by Zhishen at al.,26, with some minor modifications. A portion of extract (1mL) was mixed with 4mL of distilled water and 0.3mL of a 5% NaNO2 solution. After 5 min, 0.3mL of a 10% AlCl3 H2O solution was added to the mixture followed by 2mL of 1 M NaOH and 2.4mL of distilled water added after 1 min. The resulting mixture was thoroughly mixed, and absorbance was measured at a wavelength of 510 nm using a UV2600i spectrophotometer (Shimadzu, Japan). Results of determinations are expressed in mg QE·g− 1.
Determination of antioxidative properties in the DPPH assay
The free-radical scavenging activity was determined in the leaf extract with the Brand-Williams et al.,27 method, after some minor modifications. In order to obtain a stock solution of the DPPH radical, a DPPH standard (4 mg in 10mL) was diluted with methanol until its absorbance measured at a wavelength of 517 nm reached 0.90 (± 0.01). Afterwards, 200µL of the hemp leaf extract were added to 1mL of a DPPH• stock solution. The resulting sample was thoroughly mixed, and absorbance drop was measured after 10 min at a wavelength of 517 nm, using a UV-2600i spectrophotometer (Shimadzu, Japan). Results of determinations are expressed in mmol of Trolox equivalents kg− 1 sample.
Amino acid analysis
The content of amino acids in hemp leaves was determined using an AAA-500 amino acid analyzer (INGOS, Czechia). The analytical procedure was consistent with apparatus producer guidelines. The sample was hydrolyzed in 6 M HCl at a temp. of 110 °C for 24 h. After cooling, filtration and rinsing, the hydrolysate was evaporated in a vacuum evaporator, and the dry residue was dissolved in a buffer (pH = 2.2). The prepared sample was used for analysis with the ninhydrin method, with buffers having pH of 2.6, 3.0, 4.25, and 7.9. The ninhydrin solution was buffered using a buffer with pH 5.5. Analyses were conducted using a 370 mm long column filled with Ostion ANB INGOS ionex (Czechia). Column temperature was 55–74 °C, and reactor temperature was 120 °C. To determine sulfur amino acids, methionine and cysteine, oxidative hydrolysis was performed using a mixture of formic acid and hydrogen peroxide (9:1), at a temp. 110 °C, for 24 h. The sample was cooled and further handled as during acidic hydrolysis, under the following conditions: buffers with pH 2.6 and 3.0, column temperature of 60 °C, and reactor temperature of 120 °C. Amino acids were analyzed in three replications.
Antimicrobial activity of extracts from hemp leaves
The following strains were used as test microorganisms: Staphylococcus aureus ATCC® 33,591, Pseudomonas aeruginosa WDCM 00024 ATCC® 10,145, Candida albicans ATCC® 2091, and Aspergillus niger ATCC® 6275. All strains were purchased at Pol-Aura Odczynniki Chemiczne company (Poland). Antibacterial activity of the extracts was determined with the disk-diffusion method28,29. The bacterial strains from a 24-hour culture, in the number of 1.0·105 cfu·mL− 1, were sown in the amount of 0.1mL on Petri dishes for a submerged culture and poured with a Mueller-Hinton culture medium. Fungi were grown at 25 °C on potato dextrose agar (PDA) (OXOID, Hampshire, England). The inoculated medium was incubated at a temperature of 25˚C for 24 h in the case of yeast and for three to five days in the case of Aspergillus niger. Once the medium had solidified, sterile disks (6 mm in diameter) were placed on its surface. Filter paper discs containing 5 mg gentamicin were used as positive controls. Then, 20µL of each hemp leaf extract were injected onto the disks, and distilled water was injected on one disk as a control. The dishes were incubated at 37 ± 1 °C for 24 h (bacteria) or 5 days (fungi). Growth inhibition zones were measured using a caliper Verniera. Results are provided, after subtracting disk diameter and growth inhibition zone for the control samples, in millimeters. Determinations were conducted in 3 replications.
Statistical analysis
Statistical analysis aimed to determine the effect of 2 factors, i.e., harvest date of hemp leaves and dose of the biofertilizer, on the contents of: total polyphenols, flavonoids, DPPH, and polyphenol fractions, and the antimicrobial activity of the hemp leaf extract. The study results were analyzed using Statistica 13 software from StatSoft. One-way analysis of variance (ANOVA) was performed in order to determine the significance of the impact that the respective factors had on the analyzed values. The significance of differences between the mean values was established with Tukey’s test. The adopted significance level was α = 0.05. All the tests and analyses were conducted in 5 replications. In addition, a Pearson correlation analysis was conducted to check for correlations between contents of the analyzed variables.
Results and discussion
Proximate chemical analysis
Fresh hemp leaves represent a fine source of nutrients, including strong antioxidants, protein, crude fibers and ash. Table 3 presents results of proximate chemical analysis of hemp leaves, including contents of crude protein, fiber, ash, water and total phenols (NFE).
In the case of the control samples, the protein content of hemp leaves showed an ascending trend, i.e., increased in the subsequent harvest dates (II, III, IV) compared to harvest date I (22.80%) by 4.72% (23.88%), 6.60% (24.30%), and 7.85% (24.59%), respectively. The leaves from the first harvest date had a higher protein content in all digestate treatment variants compared to the control sample. In the case of harvest dates II, III, and IV, the protein content of the leaves tended to decrease at each digestate dose applied compared to the respective control.
The crude fiber content determined for the control leaves from the first harvest date was at 18.89%. In each subsequent harvest date, its value increased in both the control samples and the samples from all digestate application variants. The fiber content of the control leaves increased by 1.19, 0.38 and 2.82%. After soil amendment with the digestate, the determined fiber contents differed depending on digestate dose, with the greatest increase noted in the leaves from harvest date II and reaching 1.62% for digestate dose of 20 L, 2.97% for the 30 L dose, and 3.62% for the 50 L dose, compared to the control sample.
The mean total phenol content (NFE) of hemp leaves from the control variants ranged from 19.17 to 19.30% and showed a descending trend compared to the first harvest date (-2.44%, -1.81% and − 1.16%). In the experimental variants with digestate application in various doses, the greatest decreases in NFE content were determined in the leaves harvested in terms III and IV. Considering particular digestate doses, the total phenol content of the leaves from harvest date III was lower by 0.46% (20 L), 0.42% (30 L) and 0.41% (50 L) compared to the NFE content of the control leaves. In the case of harvest date IV, the respective decreases were at 0.41, 0.62 and 0.62%.
The crude ash content (Table 3) of the control hemp leaves ranged from 11.06 to 11.72% and compared to the control samples from the harvest date I showed a descending trend in the subsequent harvest dates. In harvest date II, a decrease in the crude fiber content of the leaves reached 4.60% compared to the control sample harvested in term I. In the leaves from the subsequent harvest dates (III and IV), the crude fiber content decreased by decrease 5.26 and 5.60%, respectively. Decreases were also noted in the ash content of the leaves harvested in term I upon digestate application in doses of 30 L (-3.86%) and 50 L (-4.26%).
Along with hemp plant development, the content of water (Table 3) in their leaves was observed to decrease. For the four harvest dates in control variants, it varied from 6.67 to 6.83%, and was found to decrease by 0.97, 0.19 and 3.56% in the leaves harvested in terms II, III and IV, compared to those from harvest date I.
In the case of experimental variants with various doses of the digestate, the water content of the leaves increased upon digestate application in doses of 20 L and 30 L for the leaves from harvest date II as well as upon digestate application in a dose of 30 L for the leaves harvested in term IV, compared to the control sample, i.e., by 2.21 and 1.72% (harvest date II) and by 1.45% (harvest date IV).
The Pearson correlation analysis demonstrated strong positive correlations between harvest date and digestate dose for the crude protein content (0.657) and the total phenol content (NFE) (0.774). A similar correlation was found for water content of the hemp leaves (0.533). In turn, harvest date and digestate dose were weakly corrected with crude fiber content (0.347) and crude ash content (0.386) of the h hemp leaves.
Analysis of amino acid content of hemp leaves
Table 4 presents results of amino acid content determination in hemp leaves. The alanine content of the control leaves ranged from 3.90 to 3.97 (g 100 g− 1 protein), with its highest content determined in the control leaves harvested in date I. In the leaves from the other harvest dates, the content of this amino acid was lower by 1.93, 1.43 and 1.51%. After digestate application, alanine content increased in hemp leaves along with plant growth (in leaves from the subsequent harvest dates), compared to the control variant, except for the leaves from the variant with 50 L digestate harvested in terms I and III, in which this amino acid content was lower by 0.67 and 3.32% than in the control leaves.
The content of aspartic acid (Table 4) determined in the control samples reached 8.30, 8.37, 8.42 and 8.54 (g 100 g− 1 protein). These results show an increase in its content in the subsequent harvest dates of leaves, i.e., by 0.76, 2.06 and 2.89%, compared to that determined in the leaves from harvest date I. Soil amendment with digestate resulted in a decrease in aspartic acid content of the leaves harvested in term II (1.71, 0.12, 1.79%) and IV (3.71, 2.50, 4.17%) for each dose variant. In the harvest dates I and III, a decrease in this amino acid content was found in the leaves from experimental variants with 20 and 50 L digestate application, namely by 0.12 and 0.40% as well as 3.21 and 0.87%, respectively.
The content of arginine in the control leaves harvested in the subsequent terms reached 4.21, 4.27, 4.30 and 4.31 (g 100 g− 1 protein) (Table 4). These results indicate that arginine content increased (+ 1.27, + 2.06, + 2.26%) along with plant development. After digestate application, its content decreased in the leaves harvested in all analyzed terms from the variants with 20 and 50 L digestate, i.e. by 0.79 and 0.63% (harvest date I), by 1.48 and 3.05% (harvest date II), by 0.47 and 4.19% (harvest date III) and by 1.31 and 7.73% (harvest date IV), compared to the control.
The content of cystine in the control hemp leaves (Table 4) ranged from 0.75 to 0.79 (g 100 g− 1 protein), with values higher by 4.41 and 1.32% determined in the leaves harvested in terms III and IV, compared to the control leaves harvested in term I. Digestate application during plant growth contributed to its content decrease by 0.88% in the leaves from the 50 L variant harvested in term I. In harvest date II, cystine content decrease was noted in the leaves from the variants with digestate application in doses of 30 and 50 L, i.e., by 4.87 and 2.65%, compared to the control leaves. In turn, in the case of leaves harvested in term IV, digestate application caused cystine content to decrease in all experimental variants by 5.22, 9.57 and 3.48% compared to the control.
The content of glutamic acid varied in the control samples between 10.04 and 10.52 (g 100 g− 1 protein) (Table 4). The results obtained indicate that its content increased in the subsequent harvest dates by 1.30, 1.56 and 4.78%. Digestate application contributed to its content decrease in the leaves harvested in the subsequent terms. In those from harvest dates II and III, its content decreased by 1.34 and 2.20% and by 0.85 and 3.14% in the experimental variants with 20 and 50 L digestate doses, compared to the control sample, whereas in those from harvest date IV, it decreased upon soil amendment with all digestate doses, i.e., by 1.17, 0.10 and 9.10%, compared to the control.
The content of glycine (Table 4) in the harvested control leaves ranged from 2.81 to 2.92 (g 100 g− 1 protein). The results obtained show that its content increased by 3.92, 3.56 and 3.44% compared to the control sample from harvest date I. After digestate application, glycine content increased only in the leaves harvested in term I. In the remaining harvest dates, its content decreased after digestate application in 20, 30 and 50 L doses by 0.34, 2.51 and 3.20% in the leaves from harvest date II; by 1.15, 5.62 and 7.45% in those from harvest date III as well as by 1.95, 9.59 and 13.66% in the leaves from harvest date IV, respectively.
The content of histidine in the control hemp leaves ranged from 2.25 to 2.42 (g 100 g− 1 protein) (Table 4). The results achieved show its content to increase in the consecutive harvest dates by 2.96, 3.11 and 7.25% compared to the harvest date I of the control sample. Digestate application resulted in a decreased content of this amino acid in leaves from all experimental variants (20, 30 and 50 L), except for the leaves from the 30 L digestate variant harvested in term III, in which histidine content increased slightly (by 0.57%) compared to the control sample.
The content of isoleucine determined in the control leaves varied between 3.27 and 3.47 (g 100 g− 1 protein), and was observed to increase in the leaves from the subsequent harvest dates by 1.43, 5.91 and 4.07% compared to the leaves from the control variant harvested in term I. Soil amendment with all three tested doses of the digestate (20, 30, 50 L) contributed to an increase in the content of this amino acid in the leaves harvested in term I, whereas digestate treatment at 20 L in the leaves from the consecutive harvest terms (II, III, IV). Isoleucine contents were higher by 1.32% (20, 30 L) and 2.14% (50 L) as well as by 0.80% and 1.15% in leaves from harvest date III and by 2.74% in those from harvest date IV, compared to the control.
The content of leucine in the control leaves ranged from 7.15 to 7.31 (g 100 g− 1 protein) and was observed to decrease by 0.28% in the control leaves from harvest date II compared to the control leaves from harvest date I. In the successive harvest terms, its content increased by 0.46 and 2.00%, respectively. The results achieved for the individual digestate treatment variants showed, in most cases, a decrease in leucine content, except for the leaves from the 20 L digestate variant harvested in terms II, III and IV, in which leucine content was higher by 0.23, 0.83 and 0.73%, compared to the control sample.
The lysine content of the control leaves ranged from 3.79 to 3.98 (g 100 g− 1 protein) (Table 4) and was observed to increase along with plant development, with ultimate values higher by 0.70, 3.35 and 5.11% than in the control leaves harvested in term I. In most cases, digestate application contributed to an increase in the content of this amino acid in hemp leaves, except for digestate dose of 50 L which reduced lysine content of the leaves harvested in terms II, III and IV by 1.22, 1.36 and 6.62%, respectively, compared to the control samples.
The content of methionine in the control samples was at 0.83, 0.84, 0.88 and 0.91 (g 100 g− 1 protein), and showed an upward trend, increasing by 2.02, 6.05 and 9.68% compared to the control leaves harvested in term I. Digestate application in 20 and 50 L doses caused methionine content to decrease in the leaves harvested in term I, i.e., by 2.82 and 3.23%, respectively, compared to the control sample. In the harvest date II, a lower content of this amino acid was determined in the leaves from 30 to 50 L digestate variants (by 4.35 and 10.28%, respectively). In harvest date III, a decrease in methionine content of the leaves was noted only upon soil amendment with 50 L dose of the digestate (by 8.37% compared to the control). In the leaves from the last harvest term, methionine content decreased by 0.37 and 11.03% in the variants with 30 and 50 L doses of the digestate, compared to the control samples.
The content of proline in the control leaves reached 2.90 (harvest date I and II), 3.10 (harvest date III) and 3.25 (g 100 g− 1 protein) (harvest date IV), meaning an increase by 6.79 and 12.07% in the control leaves from the two last harvest terms. After digestate application, the content of proline decreased in leaves from all experimental variants and all harvest dates, compared to the control sample.
The content of phenylalanine in the subsequent control samples reached 3.90, 3.92, 3.90 and 3.95(g 100 g− 1 protein). After digestate application during plant growth, the content of this amino acid decreased in most leaf samples, except for those harvested in term I. The greatest decrease in its content was noted for the leaves from the harvest date IV. The achieved values were lower by 3.54, 5.06, and 7.17%, compared to the control sample.
The content of serine in the control samples of hemp leaves ranged from 3.19 to 3.12 (g 100 g− 1 protein) and showed an upward trend along with plant growth. After digestate application in the analyzed doses, its content increased in leaves from most variants, except for the those from the 50 L variant harvested in terms II, III and IV, in which it decreased by 3.39, 0 L.85 and 6.96%, respectively, compared to the control sample.
The content of threonine in the control hemp leaves ranged from 2.29 to 2.52 (g 100 g− 1 protein), and was found to increase in the subsequent harvest dates, by 3.05, 3.92 and 9.74%. Soil amendment with digestate contributed to both an increase and a decrease in the content of this amino acid in the harvested leaves. In the leaves harvested in term I, its content increased by 1.89, 0.58 and 0.44% in each digestate dose variant, respectively; in those from harvest term II – it increased by 2.26 and 0.14% in the leaves from 20 to 30 L digestate variants and decreased by 4.23% in those from the 50 L digestate variant, compared to the control sample.
The content of tyrosine in the control hemp leaves ranged from 3.11 to 3.21 (g 100 g− 1 protein) and showed an ascending trend (0.21, 2.68 and 3.11%) in the subsequent harvest dates (Table 4). After digestate application in the analyzed doses, tyrosine content was observed to both increase and decrease in hemp leaves. Its 0.21% decrease was noted in the leaves from harvest date I and 50 L digestate variant. In the leaves from harvest date II, its decrease was determined in the 30 and 50 L digestate variants (0.53 and 3.21%), in those from harvest date III – in the 50 L digestate variant (0.21%), and in those harvested in term IV – in the 30 and 50 L digestate variants (0.10 and 2.18%).
The content of valine ranged from 3.95 in the control hemp leaves harvested in terms I and II to 4.15 (g 100 g− 1 protein) in those from the harvest date IV (Table 4). It was higher compared to the leaves from harvest date I by 0.17% in the leaves from harvest date II, by 2.96% in those from harvest date III and by 5.07% in those from the last harvest term (IV). After digestate application, valine content decreased in the leaves from thee 30 and 50 L variants harvested in the three subsequent terms (II, III, IV). The determined values were lower by 2.36 and 0.42% (harvest date II), by 3.45 and 2.79% (harvest date III) well as by 6.11 and 5.55% (harvest date IV), compared to the control sample.
The Pearson correlation analysis showed no strong correlations between contents of the individual amino acids and leaf harvest dates and digestate doses.
Analysis of the contents of polyphenols and flavonoids and DPPH radical scavenging potential in hemp leaves
A lack of literature works providing the complete composition of the polyphenolic fractions of hemp leaves have prompted the analysis of their contents depending on soil amendment with various doses of the digestate. It is noteworthy that hemp plants contain a vast number of bioactive compounds, like e.g. polyphenols, exhibiting antioxidative properties. Among multiple compounds, these plants also contain a few antioxidants, like phenolic acids and flavonoids, as well as proteins, which represent valuable components to be used as food-enriching additives30. Antioxidative properties (PP) may protect human cells from oxidative damage induced by reactive oxygen species (ROS)31,32. Antioxidative mechanisms also contribute to the induction of other biological activities, like anti-inflammatory, anti-carcinogenic, anti-diabetic and anti-viral33. Flavonoids, and in particular – hemp flavonoids, can play the role of a geographic marker, indicating that leaves derived from various regions may differ in their content of phytochemicals34. The total content of flavonoids in leaves and flowers of hemp may account for 2.5% of their dry matter, whereas they are practically absent in hemp seeds and roots35.
Table 5 shows the content of polyphenols and flavonoids as well as the DPPH radical scavenging activity of hemp leaves. The content of polyphenols in the control hemp leaves was observed to decrease with plant growth (Table 5), by 15.06, 9.76 and 19.87% in the leaves from harvest dates II, III and IV compared to the control leaves harvested in term I [33.51 mg(GAE)g-1DW]. A descending trend was also observed after 50 L digestate treatment in the leaves harvested in terms I, II, III and IV, when their polyphenol content decreased by 10.11, 18.87, 1.78 and 4.54%, compared to the control sample. The greatest losses in polyphenols were observed in the leaves harvested in term II in all digestate treatment variants. These values determined for the subsequent digestate doses were lower by 14.06, 11.77 and 18.87% compared to the respective control sample.
The content of flavonoids in the control hemp leaves (Table 5) was also observed to decrease along with plant growth. Compared to the control sample from harvest date I, it decreased by 1.72% in the leaves from harvest date II, by 0.68% in those from harvest date III and by 3.68% in the leaves harvested in term IV. Decreased contents of flavonoids were also determined after 50 L digestate treatment in the leaves harvested in terms II, III and IV, i.e., by 0.71, 0.82 and 0.13% compared to the control sample.
Similar observations were made for the antioxidative activity of hemp leaf extracts determined in the DPPH assay (Table 5). A significant decrease was noted in the antioxidative properties of the control samples along with plant growth (68.99, 61.34, 63.38 and 59.41mmol Trolox kg-1). In the case of the 50 L digestate treatment, the antioxidative potential of the leaves harvested in the second, third and fourth term was lower by 1.55, 1.28 and 0.22% compared to the respective control samples. The remaining fertilizer treatment variants resulted in the increased antioxidative properties of hemp leaves.
The Pearson correlation analysis showed a moderate positive correlation between harvest date and digestate dose and the antioxidative properties and DPPH radical scavenging potential of hemp leaves (0.576).
Table 6 presents the polyphenol content in the analyzed Cannabis sativa samples. The most often detected flavones include cannflavin A (CVA), cannflavin B (CVB), luteolin-7-O-glucoside, luteolin and apigenin. Cannflavin is a mixture of two prenylated derivatives of cannflavin A and B. The presence of cannflavins A and B (CVA and CVB) in hemp leaves is implicated in their anti-inflammatory properties and in their potential to provide omega-3 fatty acids in food or cosmetic products36,37. The application of products featuring anti-inflammatory properties that prevent autoimmune diseases, cardiovascular diseases and neoplasms is essential for the maintenance of human health38. The contents of cannflavin A and cannflavin B flavonoids in hemp differ in the morphological parts of plants. Pollastro et al.11 have demonstrated their contents at 6.0 and 0.8 mg kg− 1, respectively, in hemp leaves, whereas Izzo et al.13 at 61.8 and 84.5 mg kg− 1, respectively, in hemp inflorescences.
In the present study, the contents of cannflavin A (CVA) and B (CVB) determined in the control leaves harvested in term I reached 69.96 and 61.21 mg·kg− 1, respectively (Table 6), and were observed to increase in the control leaves from the subsequent harvest dates by 4.78, 6.05 and 8.81% in the case of cannflavin A and by 6.95, 9.27 and 5.49% in the case of cannflavin B, respectively. After fertilizer application, their contents decreased in leaves from all digestate treatment variants and harvest dates.
Hemp leaves are also rich sources of bioactive antioxidants, like luteolin and luteolin-7-O-glucoside. According to Ahmada-Qasema et al.39, the latter is the most stable polyphenol during in vitro digestion, which makes it one of the factors responsible for the bioactivity of most extracts from leaves, including hemp leaves.
Contents of luteolin and luteolin-7-O-glucoside determined in the control hemp leaves harvested in term I reached 9.82 and 7.19 mg·kg− 1, respectively (Table 6) and were similar to the values reported by McPartland et al.40. The luteolin content of the control samples followed a downward trend and was lower by 0.14, 0.88 and 3.36% in the leaves from harvest dates II, III and IV compared to those harvested in term I. The results achieved after digestate application showed a decrease in luteolin content of the leaves harvested from 30 to 50 L variants in term I by 0.07 and 1.76%, compared to the control sample. A similar observation was made for the leaves harvested in term II from the same digestate treatment variants (30 and 50 L), i.e., their luteolin content was 0.68 and 1.56% lower than in the control leaves from harvest date I.
The content of luteolin-7-O-glucoside determined in the control hemp leaves increased between harvest dates I and II and then followed a downward trend in the samples from harvest dates III and IV (-0.32 and − 0.88%, respectively). After digestate application, the content of this flavonoid increased in leaves from practically all harvest terms compared to the control samples, regardless of fertilizer dose. An exception was observed in the 50 L digestate variant, where luteolin-7-O-glucoside content decreased by 0.64% in the leaves from the harvest date II and by 0.19% in those from harvest date IV, compared to the respective control sample.
Quercetin (IQue) type flavonoids are abundant in medicinal and dietetic plants and show multi-faceted biological activities41. The cited authors showed IQue to exhibit the antiviral effects on influenza, Zika, herpes, Ebola, dengue, chickenpox and shingles42,43. The O-glycosides belonging to the group of aglycones found in hemp include quercetin, quercetin-3-glucoside, rutin and apigenin. They all are considered to be strong flavonoids present in various extracts from hemp. Their concentrations depend on the morphological part of the processed hemp plant (seeds or stems). According to Aloo et al.44, the contents of rutin and quercetin were higher in the extract from hemp seeds than in that from hemp stems. Their everyday intake may elicit health benefits, including boosted antioxidative potential, obesity prevention as well as anti-inflammatory and anti-diabetic effects. Differences in contents of these metabolites are presumed to be one of the causes of differences observed in the biological activity of hemp seeds and hemp stem extract44,45.
The results achieved regarding quercetin in the control hemp leaves showed it content to vary between 11.65 and 11.82 mg kg− 1 (Table 6), and increase in the leaves from harvest dates II and III by 0.23 and 0.90%, respectively, compared to the control leaves from the harvest date I. In turn, a 1.22% decrease in quercetin content was noted in the leaves harvested in term IV, compared to those from term I. In the case of digestate application, an upward trend was noted in all variants compared to the respective control samples. In leaves harvested in term I, the greatest increase in quercetin content was determined after 20 L digestate treatment, and the value achieved was higher by 7.63% compared to the control sample. In the leaves from the second harvest term, quercetin content increased upon digestate treatments in doses of 20 and 30 L by 9.03 and 7.62%, respectively, compared to the control sample. In the leaves from harvest date III, the content of this metabolite was the highest in the 30 L digestate variant, being 7.51% higher than in the control sample. In the leaves from the last harvest date, a 6.95% increase in quercetin content was noted in the 30 L digestate variant, compared to the control sample.
Quercetin-3-glucoside content of hemp leaves decreased along with plant growth (Table 6). Its values determined in the control samples ranged from 33.19 to 33.64 mg kg− 1. An increase in the content of this compound was noted in the leaves from the harvest date II; however, it was small and higher by only 0.03% compared the control sample from harvest date I. After digestate application, quercetin-3-glucoside content decreased in hemp leaves in most analyzed experimental variants.
Pharmacological efficacy of rutin, associated with its various properties, like anti-inflammatory and antioxidative activity, has become extremely important for the pharmaceutical industry, leading to many drugs registered worldwide that contain this valuable compound46,47. A study conducted by Kaur & Muthuraman48, has shown positive outcomes of rutin administration in the treatment of renovascular hypertension caused by renal ischemia. To the best of authors’ knowledge, there has been no reports to date on rutin content of hemp leaves. In the present study, the content of rutin determined in the control hemp leaves was observed to increase insignificantly regardless of the harvest date of leaves (15.69-15.94 mg kg− 1). The content of this metabolite in the leaves from the subsequent harvest terms was 0.65, 1.57 and 0.47% higher than in the control leaves from harvest date I. Fertilizer application during hemp plant growth contributed, in most cases, to rutin content increase in the leaves, except for the leaves from the 50 L digestate variant harvested in terms II, III and IV, which contained 1.10, 0.04 and 1.08% less rutin, respectively, than their control counterparts.
The content of aglycone in the form of apigenin in hemp makes it one of the most important plants with strong antifungal, antiparasitic, antioxidative, as well as anxiety-, depression- and pain-relieving properties. This compound is also used in the fight against cancer and Alzheimer’s disease. A 2019 study on the therapeutic potential of apigenin found that it had stronger antioxidative properties than other flavonoids49. In turn, an experiment with rats performed by Huang et al.50 demonstrated that apigenin inhibited oxidative stress by regulating selected pathways in the liver. Optimal doses of apigenin and other flavonoids in daily supplementation are assumed to range from 50 mg to 600 mg. A study conducted by Jin et al.51 with different morphological parts of hemp plants showed the highest contents of flavonoids in leaves (0.34–0.44%) and no presence of these compounds in roots. The total content of flavonoids in cannabis leaves was estimated at ca. 1%40. The content of apigenin in the samples analyzed by McPartland et al.40 ranged from 0.03 to 0.07% in leaves and from 0.004 to 0.01% in inflorescence. In our study, the control leaves of hemp grown with various applied doses of the digestate contained from 2.71 (harvest date IV) to 2.82 mg kg− 1 (harvest date III) of apigenin (Table 6). These results point to a decreasing content of this flavonoid in the control samples of the leaves. A tangible increase in apigenin content was noted in the leaves harvested in term III, i.e., by 2.92% compared to the control samples from harvest date I. In all experimental variants of digestate application, apigenin content was found to decrease in the leaves from harvest date I by 0.12, 4.87 and 7.06% compared to the control sample. In the leaves harvested in terms II and III, a decrease was noted only after 50 L digestate treatment when apigenin content was 2.24 and 0.59% lower than in the control samples.
The presence of kaempferol compounds in hemp leaves boosts their pharmacological potential including their anti-inflammatory, anti-carcinogenic, neuroprotective and cardioprotective effects52,53. The content of kaempferol in the control samples of hemp leaves ranged from 1.59 (harvest date IV) to 2.10 mg kg− 1 (harvest date II) (Table 6). Lower kaempferol contents were determined in the control leaves harvested in terms III and IV, i.e., by 2.40 and 18.01%, respectively, than in those from term I, whereas in the control leaves from harvest date II, its content increased by 8.23%. In turn, digestate application did not cause any downward trend in the content of this compound in any of the experimental variants. The greatest increases in kaempferol content were noted in the leaves from 30 L digestate variants and reached 13.72 in harvest date I leaves, 17.27 in harvest date II leaves, 17.40 in harvest date III leaves and 19.04% in harvest date IV leaves, compared to the control.
According to Kim et al.54, the presence of kaempferol-3-O-glucoside in hemp leaves increases their known therapeutic properties. Its content in the control hemp leaves analyzed in our study ranged from 3.04 to 3.20 mg kg− 1, and was observed to decrease along with plant growth, except for the control leaves harvested in term I (+ 1.59%) (Table 6). The results obtained for the leaves from harvest dates II and III were lower by 1.27 and 3.49% from those determined in the control leaves from harvest date I. After digestate application in a 50 L dose, the content of kaempferol-3-O-glucoside decreased in leaves from all harvest dates by 1.59, 2.29, 0.54 and 1.31% compared to the respective control samples.
Other compounds exhibiting antioxidative properties and belonging to the group of phenolic acids include coffee tannic acid referred to as chlorogenic acid (CGA), p-coumaric acid and ferulic acid. Coffee tannic acid is a water-soluble compound synthesized during plant aerobic oxidation in the shikimic acid pathway. It exhibits high biological activity manifested mainly in its antioxidative, liver- and kidney-protecting, antibacterial, anticarcinogenic, glucose and lipid metabolism-regulating, anti-inflammatory, and nervous system-protecting properties25. Due to the content of physiologically-active substances beneficial to human health, it serves the following roles: blood pressure regulation, anti-septic effect, resistance to neoplasms, anti-inflammatory effect, anti-oxidative effect, etc55. Generally, CGA may form a protein-polyphenolic complex through hydrogen bonds with protein, thereby creating a thicker intermediate layer, inhibiting diffusion of free radicals and increasing antioxidative capacity56. In the cosmetic industry, it is used for skin whitening, moisturizing, sun protection and hair care because it can prevent collagen damage caused by reactive oxygen species and protect human skin from ultraviolet rays57. These compounds derived from hemp leaves represent a fine alternative for the current industry because plant residues are, in most cases, treated as harvest waste. As shown in literature data, their content varies between morphological parts of a plant, e.g., Izzo et al.13 demonstrated its content between 1.2 and 4.3 mg kg− 1 in inflorescences of various C. sativa cultivars.
In the present study (Table 6), caffeic acid content of the control hemp leaves ranged from 1.55 to 1.65 mg kg− 1. Its lowest value was determined for the control samples from harvest date III (1.55 mg kg− 1), being 4.52% lower compared to the control leaves from harvest date I. Analysis of the samples from experimental variants with digestate application demonstrated that, in most cases, the content of this acid increased compared to the control samples. A decrease in its content was noted only in the leaves from the variant with 50 L digestate, i.e., by 1.23% in those from harvest date I, by 3.29% in those from harvest date II and by 2.42% in those harvested in term IV.
The content of chlorogenic acid in the control leaves of industrial hemp varied between 16.44 and 16.94mk kg− 1. A decrease in its content was determined for the control leaves harvested in term IV (2.66%). In the experimental variants with the fertilizer, the content of this acid increased in most cases, regardless of digestate dose, except for the leaves from the 50 L digestate treatment harvested in terms I and II, in which chlorogenic acid content was 1.76 and 0.02% lower, respectively, than in the respective control samples.
Apart from the above-mentioned, noteworthy are also a few hydroxycinnamic acids, like p-coumaric acid and ferulic acid.
p-Coumaric acid is a representative phenylpropanoid compound ubiquitous in the plant kingdom58. This acid and its derivatives have been shown to exhibit antioxidative, antimicrobial and anti-inflammatory activities58,59. Their presence in food may alleviate alcohol hepatoxicity and protect skin against UV radiation59,60. p-Coumaric acid has multiple applications, e.g., as an active component of cosmetics61. Its content in cannabis leaves turned out to be relatively high compared to roots or flowers of this plant13. The results obtained in the present study allow concluding that its content was higher than in cannabis roots analyzed by Oh et al.62. In the control samples of hemp leaves, its content ranged from 8.08 to 8.24 mg kg− 1, with the lowest values noted for the leaves from harvest dates III and IV in which it was lower by 0.37 and 1.5% than in the leaves harvested in term I (Table 6). An insignificant increase in p-coumaric acid content (by 0.45%) was determined in the leaves harvested in term II. Digestate application contributed to an increase in this acid content in almost all cases. Its highest content was determined in the leaves from digestate treatment variants of 30 and 50 L harvested in terms II and IV and, i.e., by 3.20 and 3.28% in the leaves harvested in term II and by 2.39 and 2.85% in those harvested in term IV, respectively.
Ferulic acid contents determined in hemp leaves analyzed in the present study are comparable to those determined in the flowers of these plants13. In our study (Table 6), the content of ferulic acid in the control leaves ranged from 21.20 to 21.31 mg kg− 1. Compared to the first control sample (leaves from harvest date I), a 0.5% decrease in its content was noted in the leaves harvested in term IV. After digestate application, ferulic acid content was found to increase in most cases, except for the leaves harvested in term I from the 50 L digestate variant in which it was lower by 0.58%.
The content of catechins in the control hemp leaves varied between 97.64 and 98.17 mg kg− 1 and was higher than the values of 40.9 to 85.3 mg kg− 1 reported by Izzo et al.,13 for various cannabis cultivars. Catechin contents determined for the control leaves harvested in terms II and IV were lower by 0.28 and 0.01%, respectively, compared to the control leaves from the harvest date I. Digestate application in a dose of 50 L contributed to a decrease in the content of catechins in the leaves harvested in terms I and III by 0.45, 0.01 and 0.36% compared to the control sample.
The content of epicatechins in the analyzed control leaves of hemp ranged from 39.13 to 39.81 mg kg− 1 (Table 6). Its values determined for the leaves from harvest dates II, III and IV were higher by 0.18, 1.74 and 0.73%, respectively, than in the control leaves harvested in term I. Fertilizer application in all doses tested did not decrease epicatechin content of the leaves. The greatest increase, by 6.68 and 6.41% compared to the control sample, was noted in the leaves harvested in term II in the variants with 20 and 30 L of digestate.
The content of another flavonoid – naringenin, in the control hemp leaves ranged from 0.43 to 0.52 mg kg− 1 (Table 6). Its content did not decrease neither in the control leaves nor in those from the digestate treatment variants. In the study conducted by Izzo et al.,13, naringenin content ranged from 0.5 to 2.00 mg kg− 1 depending on cannabis cultivar and was comparable with the results reported in the present study.
The Pearson correlation analysis demonstrated a moderate positive correlation between harvest date and digestate dose and the content of quercetin-3-glucoside (0.694) in the analyzed hemp leaves.
Microbiological analysis of hemp leaves
Bacteria and fungi communities colonizing the roots, leaves, flowers and rhizosphere of C. sativa cultivars have been investigated and described in sparse literature works63,64. However, various data on the antibacterial potential of these plants have remained unconfirmed in spite of the fact they derive from scientific research. Considering dearth of investigations addressing microflora of C. sativa leaves, it was analyzed in the present study. Growing interest in natural bactericidal agents, particularly those of plant origin, have prompted studies aimed to search for them also in plants such as industrial hemp. The obtained plant extracts offer a promising and healthy solutions to the increasing resistance to antibiotics and may represent an alternative to synthetic compounds aimed to inhibit bacteria development65. According to a study conducted by Fu et al.66, an extract from industrial hemp leaves exerts a strong bacteriostatic effect on E. coli and S. aureus bacteria.
Table 7 presents an analysis of the antimicrobial activity of hemp leaves. The hemp leaf extracts prepared in our study were tested using selected bacterial and fungal strains, which enabled establishing their antibacterial and antifungal activities. Results of microbiological analyses indicate bacteria and fungi growth inhibition zones induced by ethanolic extracts from hemp leaves (20 mg mL− 1). The ethanolic extract from hemp leaves showed activity against all microorganisms used in the study (Table 7). Distinct activity was determined against fungi, but mainly against Staphylococcus aureus, Pseudomonas aeruginosa and Candida albicans. The activity values achieved in all harvest dates differed compared to the control samples. However, the harvest date itself had no significant effect on the antibacterial and antifungal activity of the extracts. In the case of the experimental variants with fertilizer application, the enhanced antimicrobial activity was observed upon soil amendment with 30 and 50 L digestate doses. These fertilizer doses resulted in a significant enlargement of growth inhibition zones, particularly for C. albicans in the case of extracts from hemp leaves harvested in terms I, II and III (85, 50% - harvest date I, 50% - harvest date II, and 20% - a harvest date III), compared to the respective control samples. In the case of leaves harvested in term IV, the increase in the antimicrobial activity of the extract was higher by 80 and 100% for S. aureus at all three digestate concentrations and by 100% for P. aeruginosa at 50 L digestate dose, compared to the control sample (Table 7).
Coefficients of Pearson linear correlation reached 0.23 (S. aureus), 0.003 (P. aeruginiosa), 0.14 (C. albicans) and 0.07 (A. niger), indicating a weak correlation of the antimicrobial activity of extracts with the harvest date of leaves and digestate dose. To the best of our knowledge, antimicrobial effects may not be directly due to the antioxidative capacity of polyphenols or extracts, but microorganisms can influence antioxidative properties by, for example:
Production of antioxidants: certain microorganisms synthesize natural antioxidants such as enzymes, vitamins or phenolic compounds.
Biotransformation: microorganisms can modify chemical compounds into forms with more potent antioxidative properties.
Conclusions
The conducted study and the analysis of main components of hemp leaves demonstrated that digestate application contributed to a decrease in the total polyphenol content (−15.06, −9.76, −19.87%), flavonoid content (−1.72, −0.68, −3.68%) and antioxidative capacity measured in the DPPH assay (−11.08, −8.13, −13.87%) in the leaves depending on harvest date of the control sample and digestate dose. Similar observations were made for the contents of NFE (−2.44, −1.81, −3.16%) and ash (−4.60, −5.26, −5.60%) in leaves. A positive effect of the planned experiment was observed in crude fiber content (+ 0.19, + 0.38, + 2.82%) of the leaves from all variants. An increase was also noted in crude protein content of the control leaves (+ 4.72, + 6.59, + 7.85%) and in the contents of individual amino acids, which confirms the applicability of hemp leaves as additives rich in plant-derived protein in many industry branches. Extracts from hemp leaves exposed to bacterial and fungal strains demonstrated positive antibacterial and antifungal activity against S. aureus, P. aeruginosa and C. albicans strains in most experimental variants, particularly after soil amendment with the tested doses of the digestate.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Insam, H., Gómez-Brandón, M. & Ascher, J. Manure-based biogas fermentation residues—Friend or foe of soil fertility? Soil Biol. Biochem. 84, 1–14. https://doi.org/10.1016/j.soilbio.2015.02.006 (2015).
Verdi, L. et al. Does the use of digestate to replace mineral fertilizers have less emissions of N2O and NH3? Agric. For. Meteorol. 269–270, 112–118. https://doi.org/10.1016/j.agrformet.2019.02.004 (2019).
Berthrong, S. T., Buckley, D. H. & Drinkwater, L. E. Agricultural management and labile carbon additions affect soil microbial community structure and interact with carbon and nitrogen cycling. Microb. Ecol. 66, 158–170. https://doi.org/10.1007/s00248-013-0225-0 (2013).
EC. Agriculture and rural development. https://agriculture.ec.europa.eu/farming/crop-productions-and-plant-based-products/hemp_en (2023).
Russo, E. B. History of cannabis and its preparations in saga, science, and sobriquet. Chem. Biodivers. 4, 1614–1648. https://doi.org/10.1002/cbdv.200790144 (2007).
Adesina, I. S. Soil nutritional requirement and biological soil health implications of hemp cultivation in North Carolina (Doctoral dissertation North Carolina Agricultural and Technical State University, 2021).
Cushnie, T. T. & Lamb, A. J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents. 26, 343–356. https://doi.org/10.1016/j.ijantimicag.2005.09.002 (2005).
Wach, A., Pyrzyńska, K. & Biesaga, M. Quercetin content in some food and herbal samples. Food Chem. 100 (2), 699–704. https://doi.org/10.1016/j.foodchem.2005.10.028 (2007).
Kakudidi, E., Tugume, P., Asiimwe, S. & Anywar, G. Traditional modern health uses of Cannabis sativa L. In Africa and its phytochemical and Pharmacological profile. Cannabis Marijuana Healthc. 189–210. https://doi.org/10.1007/978-981-16-8822-5_10 (2022).
Mkpenie, V., Essien, E. & Udoh, I. Effect of extraction conditions on total polyphenol contents, antioxidant and antimicrobial activities of Cannabis sativa L. Electron. J. Environ. Agricultural Food Chem. 11, 300–307 (2012).
Pollastro, F., Minassi, A. & Fresu, L. G. Cannabis phenolics and their bioactivities. Curr. Med. Chem. 25 (10), 1160–1185 (2018).
Castaldo, L. et al. Red wine consum. cardiovasc. health molecules. 24 (19), 3626 https://doi.org/10.3390/molecules24193626 (2019).
Izzo, L. et al. Analysis of phenolic compounds in commercial Cannabis sativa L. Inflorescences using UHPLC-Q-Orbitrap HRMS. Molecules 25, 631. https://doi.org/10.3390/molecules25030631 (2020).
Farinon, B., Molinari, R., Costantini, L. & Merendino, N. The seed of industrial hemp (Cannabis sativa L.): nutritional quality and potential functionality for human health and nutrition. Nutrients 12 (7), 1935. https://doi.org/10.3390/nu12071935 (2020).
Karlsson, L., Finell, M. & Martinsson, K. Effects of increasing amounts of hempseed cake in the diet of dairy cows on the production and composition of milk. Animal 4, 1854–1860. https://doi.org/10.1017/S1751731110001254 (2010).
Kleinhenz, M. D. et al. Nutrient concentrations, digestibility, and cannabinoid concentrations of industrial hemp plant components. Appl. Anim. Sci. 36 (4), 489–494. https://doi.org/10.15232/aas.2020-02018 (2020).
Brugman, S. et al. H. M. A comparative review on microbiota manipulation: lessons from fish, plants, livestock, and human research. Front. Nutr. 5, 80 (2018).
Langezaal, C. R., Chandra, A. & Scheffer, J. J. C. Antimicrobial screening of essential oils and extracts of some Humulus lupulus L. Cultivars. Pharm. World Sci. 14 (6), 353–356. https://doi.org/10.1007/BF01970171 (1992).
Ranjith, A., Srilatha, C. M., Lekshmi, P. C. & Rameshbabu, N. Antiaflatoxigenic potential of essential oils of spices—a review. World Mycotoxin J. 14, 463–475. https://doi.org/10.3920/WMJ2020.2636 (2021).
PN-EN ISO & /IEC 17025. General requirements for the competence of testing and calibration laboratories (in Poland). (2005).
AOAC (Association of Official Analytical Chemists). Official Methods of Analysis of the Association of Official Analytical Chemistry. 13th Edition (Ed) Horwitz, W. 1141 (AOAC, 1980).
Singleton, V. L. & Rossi, J. A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 16, 144–158. https://doi.org/10.5344/ajev.1965.16.3.144 (1965).
Kramling, T. E. & Singleton, V. L. An estimate of the nonflavonoid phenols in wines. Am. Journal Enol. Viticulture. 20, 86–92 (1969).
Calzolari, D. et al. High added-value compounds from Cannabis threshing residues. Ind. Crops Prod. 108, 558–563 (2017).
Wang, L. et al. Peng C. The biological activity mechanism of chlorogenic acid and its applications in food industry. Review- Front. Nutr. 9, 943911. https://doi.org/10.3389/fnut.2022.943911 (2022).
Zhishen, J., Mengcheng, T. & Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 64 (4), 555–559. https://doi.org/10.1016/S0308-8146(98)00102-2 (1999).
Brand-Williams, W., Cuvelier, M. E. & Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 28, 25–30 (1995).
Bauer, A. W., Kirby, W. M. M., Sherries, J. C. & Turck, M. Antibiotic susceptibility testing by single 22. disk method. Am. J. Clin. Pathol. 45, 493–496 (1966).
Collins, C. H., Patricia, M. L., Microbiological & methods. 441–446 (1994).
Pojić, M. et al. Characterization of By-products originating from hemp oil processing. J. Sci. Food Agricultural. 62, 12436–12442. https://doi.org/10.1021/jf5044426 (2014).
Guzzetti, L. et al. Assessment of dietary bioactive phenolic compounds and agricultural sustainability of an African leafy vegetable Corchorus Olitorius L. Front. Nutrtions. 8, 667812 (2021).
Bosso, H., Barbalho, S. M., Goulart, R. D. A. & Otoboni, A. M. M. B. Green coffee: economic relevance and a systematic review of the effects on human health. Crit. Rev. Food Sci. Nutr. 61, 1–17.
Cheng, D. et al. Chlorogenic acid protects against aluminum toxicity via mapk/akt signaling pathway in murine RAW264.7 macrophages. J. Inorg. Biochem. 190, 113–120. https://doi.org/10.1016/j.jinorgbio.2018.11.001 (2019).
Li, C. R. et al. LC-MS-based untargeted metabolomics reveals chemical differences of Cannabis leaves from different regions of China. Ind. Crops Prod. 176, 114411. https://doi.org/10.1016/j.indcrop.2021.114411 (2022).
Cui, Q., Li, J. & Yu, C. Extraction and purification of total flavonoids from Cannabis sativa fibers and their evaluation for antioxidant and antibacterial activities. Text. Res. J. 93 (1–2), 56–69. https://doi.org/10.1177/00405175221114929 (2023).
Minassi, A., Giana, A., Ech-Chahad, A. & Appendino, G. A regiodivergent synthesis of ring A C-prenylflavones. Org. Lett. 10, 2267–2270 (2008).
Barnett, S. E. et al. Evaluating the Microbiome of hemp. Phytobiomes J. 4, 351–363 (2020).
Furman, D. et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 25, 1822–1832 (2019).
Ahmad-Qasem, M. H. et al. J. V. Influence of olive leaf processing on the bioaccessibility of bioactive polyphenols. J. Agric. Food Chem. 62, 6190–6198 (2014).
McPartland, J. M. & Russo, E. B. Cannabis and Cannabis extracts: greater than the sum of their parts?? J. Cannabis Ther. 1, 103–132 (2001).
Agrawal, K., Blunden, G. & Jacob, C. Antiviral significance of Isoquercetin (Quercetin-3-O-Glucoside) with special reference to its Anti-Coronaviral potential. 19(1) (2024). https://doi.org/10.1177/1934578X231219560
Joshi, R. S. et al. Discovery of potential multi-target-directed ligands by targeting host-specific SARS-CoV-2 structurally conserved main protease. J. Biomol. Struct. Dyn. 39 (9), 3099–3114 (2021).
Hoffmann, M. et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 181 (2), 271–280e8 (2020).
Aloo, S. O., Kwame, F. O. & Oh, D. H. Identification of possible bioactive compounds and a comparative study on in vitro biological properties of whole hemp seed and stem. Food Bioscience. 51, 102329. https://doi.org/10.1016/j.fbio.2022.102329 (2023).
Rusmana, D. et al. Antioxidant activity of Phyllanthus niruri extract, Rutin and Quercetin. Indonesian Biomedical J. 9 (2), 84–90 (2017).
Abarikwu, S. D. et al. Rutin, an antioxidant flavonoid, induces glutathione and glutathione peroxidase activities to protect against ethanol effects in cadmium-induced oxidative stress in the testis of adult rats. Andrologia 49 (7), 1–12. https://doi.org/10.1111/and.12696 (2017).
Ganeshpurkar, A. & Saluja, A. The Pharmacological potential of Rutin. Saudi Pharm. J. 25 (2), 149–164. https://doi.org/10.1016/j.jsps.2016.04.025 (2016).
Kaur, S. & Muthuraman, A. Therapeutic evaluation of Rutin in two-kidney one-clip model of renovascular hypertension in rat. Life Sci. 150, 89–94. https://doi.org/10.1016/j.lfs.2016.02.080 (2016).
Salehi, S. et al. Martins, N. The therapeutic potential of apigenin. Int. J. Mol. Sci. 20 (6), 1305. https://doi.org/10.3390/ijms20061305 (2019).
Huang, C. S. et al. Protection by chrysin, apigenin, and Luteolin against oxidative stress is mediated by the Nrf2-dependent up-regulation of Heme Oxygenase 1 and glutamate cysteine ligase in rat primary hepatocytes. Arch. Toxicol. 87, 167–178. https://doi.org/10.1007/s00204-012-0913-4 (2013).
Jin, H-R., Lee, S. & Choi, S-J. Pharmacokinetics and protective effects of Tartary buckwheat flour extracts against Ethanol-Induced liver injury in rats. Antioxidants 9 (10), 913. https://doi.org/10.3390/antiox9100913 (2020).
Bainey, K. R. & Armstrong, P. W. Clinical perspectives on reperfusion injury in acute myocardial infarction. Am. Heart J. 167 (5), 637–645. https://doi.org/10.1016/j.ahj.2014.01.015 (2014).
Burmistrova, O., Quintana, J., Diaz, J. G. & Estevez, F. Astragalin heptaacetate-induced cell death in human leukemia cells is dependent on caspases and activates the MAPK pathway. Cancer Lett. 309 (1), 71–77. https://doi.org/10.1016/j.canlet.2011.05.018 (2011).
Kim, Y. H., Choi, Y. J. & Kang, M. K. Astragalin inhibits allergic inflammation and airway thickening in ovalbumin-challenged mice. J. Agric. Food Chem. 65 (4), 836–845. https://doi.org/10.1021/acs.jafc.6b05160 (2017).
Miao, M. & Xiang, L. Chapter Three - Pharmacological action and potential targets of chlorogenic acid. Adv. Pharmacol. 87, 71–88. https://doi.org/10.1016/bs.apha.2019.12.002 (2020).
Guo, X. et al. Effect of chlorogenic acid on the physicochemical and functional properties of coregonus peled myofibrillar protein through hydroxyl radical oxidation. Molecules 24, 3205. https://doi.org/10.3390/molecules24173205 (2019).
Castro, A. C. C. et al. Santos A. G. Green coffee seed residue: A sustainable source of antioxidant compounds. Food Chem. 246, 48–57. https://doi.org/10.1016/j.foodchem.2017.10.153 (2018).
Boz, H. p-Coumaric acid in cereals: presence, antioxidant and antimicrobial effects. Int. J. Food Sci. Technol. 50 (20215), 2323–2328. https://doi.org/10.1111/ijfs.12898 (2015).
Pei, K., Ou, J., Huang, J. & Ou, S. p-Coumaric acid and its conjugates: dietary sources, Pharmacokinetic properties and biological activities. J. Sci. Food Agricultural. 96, 2952–2962 (2016).
Lee, S. I. et al. Protective effect of Sasa quelpaertensis and p-coumaric acid on ethanol-induced hepatotoxicity in mice. J. Appl. Biol. Chem. 51, 148–154 (2008).
Boo, Y. C. p-Coumaric acid as an active ingredient in cosmetics: A review focusing on its antimelanogenic effects. Antioxidants 8, 275 (2019).
Oh, C. M. et al. Identification of p-Coumaric acid and Ethyl p-Coumarate as the main phenolic components of hemp (Cannabis sativa L.) roots. Molecules 27, 2781. https://doi.org/10.3390/molecules27092781 (2022).
Singh, B. K. & Trivedi, P. Microbiome and the future for food and nutrient security. Microb. Biotechnol. 10 (1), 50–53. https://doi.org/10.1111/1751-7915.12592 (2017).
Ahmed, B. & Hijri, M. Potential impacts of soil microbiota manipulation on secondary metabolites production in cannabis. J. Can. Res. 3 (25). https://doi.org/10.1186/s42238-021-00082-0 (2021).
Hayek, L. J., Emerson, J. M. & Gardner, A. M. A placebo-controlled trial of the effect of two preoperative baths or showers with chlorhexidine detergent on postoperative wound infection rates. J. Hosp. Infect. 10 (2), 165–172. https://doi.org/10.1016/0195-6701(87)90143-5 (1987).
Fu, Y., Zhu, S., Duan, S. & Liu, L. Bioassay-Guided Isolation and Identification of Antibacterial Components against Escherichia coli from Industrial Hemp Leaves. Separations 10, 35 https://doi.org/10.3390/separations10010035 (2023).
Funding
Open Access funding enabled and organized by University of Life Sciences in Lublin SUBB.WTR.19.042.
Author information
Authors and Affiliations
Contributions
M.K-G. = Performing the experiments, data collection M.K-G.; M.S. and M.K. = Performing the experiments, data collection, preparing the first draft M.K.-G.; M.S. and M.K. =Data collection; M.K.-G. and M.S. = Performing experience, M.K.-G. = Revising the first draft, wrote the main manuscript text and prepared figures, M.K.-G. = Supervisor, idea generation; revising the first draft, M.K.-G.; K.S. = Supervisor; idea generation, editing and revision. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kachel-Górecka, M., Stryjecka, M., Koszel, M. et al. Effect of agricultural digestate on antioxidative properties and antimicrobial activity of hemp (Cannabis sativa) leaves. Sci Rep 15, 22955 (2025). https://doi.org/10.1038/s41598-025-04565-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-04565-5