Abstract
Urinary tract infections (UTIs) are among the most common infectious diseases in both hospital and community settings, leading to increased morbidity and mortality, particularly in patients with DM due to impaired immune function and hyperglycemia. The rise of antimicrobial resistance (AMR) further complicates the treatment of bacterial uropathogens, presenting a significant public health challenge. Thus, this study aimed to assess the magnitude, antimicrobial resistance patterns, and risk factors of bacterial UTIs in DM patients at Bule Hora University Teaching Hospital, Southern Ethiopia, 2023. An institution-based cross-sectional study was conducted among 249 DM patients, selected via systematic random sampling, from July 1 to August 30, 2023. Sociodemographic and clinical data were gathered using a pretested structured questionnaire. Midstream urine samples were collected for bacterial culture and identification following standard microbiological procedures, with antimicrobial susceptibility testing performed using the Kirby-Bauer disk diffusion method on Mueller–Hinton agar. Data were entered into EpiData version 4.6 and analyzed using IBM SPSS Statistics 17. Frequencies and percentages were used to summarize the data, while logistic regression models were utilized to establish the relationship between dependent and independent variables. A P-value ≤ 0.05 at a 95% confidence interval was considered statistically significant. The prevalence of bacterial UTIs among DM patients was found to be 15.7%. E. coli was the most common bacterial isolate, accounting for 15/39 (38.5%) of cases, followed by CONS (17.9%) and Klebsiella spp. (15.4%). Gram-negative isolates exhibited high resistance to ampicillin (93.1%) and ampicillin-clavulanic acid (79.3%), while all Gram-positive bacteria were resistant to ampicillin (100%). Furthermore, 17/39 (43.6%) of the isolates were identified as multidrug-resistant. Being female (AOR 2.132, 95% CI 1.044–4.379), a previous history of UTI (AOR 2.520, 95% CI 1.219–5.209), and a duration of DM ≥ 5 years (AOR 2.30, 95% CI 1.11–4.786) were significantly associated with an increased risk of UTI. This study found a 15.7% prevalence of bacterial UTIs among DM patients, with being female, having a history of UTI, and having longer DM duration as significant risk factors. High resistance to common antimicrobial agents was also observed. Given these results, it is crucial to support the selection of empirical treatments with regular antimicrobial susceptibility testing in the study area to ensure effective management and combat rising resistance.
Similar content being viewed by others
Introduction
Bacterial infection of the kidney, bladder, ureter, or urethra can result in urinary tract infections (UTIs)1. Cloudy or foul-smelling urine, frequent urination, and painful urination are common symptoms2. The most common cause of UTIs is the growth of bacteria from the digestive system that ascend to the urethral opening3. Sexual activity and physical characteristics, such as a shorter urethra, lack of prostatic secretion, and a higher risk of urinary canal contamination, make women more likely than men to get UTIs4. Each year, UTIs affect approximately 150 million people worldwide5.
Diabetes mellitus (DM) is a metabolic disorder marked by chronic hyperglycaemia brought on by deficiencies in the action or production of insulin6. According to the International Diabetes Federation, 451 million adults worldwide had diabetes in 2017, and by 2045, that figure is predicted to increase to 693 million. Of these people, 79% live in middle- and low-income nations7. A patient’s health may be greatly impacted by several co-morbidities linked to DM, such as cardiovascular disease, renal impairment, and neuropathy8. Over time, high blood sugar levels harm nerves and blood vessels, raising the risk of consequences, including heart disease, high blood pressure, and poor circulation9.
Diabetes mellitus (DM) is commonly associated with UTIs due to factors such as immune dysfunction, poor metabolic control, elevated glycosylated hemoglobin (HbA1c), and impaired bladder emptying from autonomic neuropathy10,11. The high blood sugar levels in DM patients create an environment that promotes bacterial growth, increasing the risk of UTIs12. Neuropathy, urinary retention, and poor circulation in DM patients also increase UTI risk and recurrence13. While UTIs are primarily caused by bacterial uropathogens in DM patients, fungi can occasionally be involved14. The prevalence of UTIs among DM patients varies significantly across regions15. Studies report a prevalence of 29.82% in Romania16, 11% in India17, 51.3% in hospitals in Egypt18, and 15.97% in Ethiopia19. Common bacterial culprits in diabetic UTIs include Escherichia coli, Klebsiella pneumoniae, Proteus spp., group B Streptococcus, coagulase-negative Staphylococci (CONS), Staphylococcus aureus, Enterobacter spp., and Pseudomonas aeruginosa20,21.
In addition to having poorly controlled blood glucose, DM patients with UTIs also have concomitant conditions such as neuropathy, hypertension, and chronic renal disease, all of which impair immunity and raise the risk of infection22. High blood sugar impairs neutrophil function, which impedes the body’s ability to fight off infections and encourages the formation of uropathogens23. On the other hand, better glycemic control and fewer comorbidities are typically manifested in DM patients who do not have UTIs24. Additionally, multidrug-resistant organisms are more likely to produce UTIs in DM patients, which makes treatment more difficult and worsens outcomes25.
Urine culture is the most reliable diagnostic tool for diagnosing and monitoring UTIs26, yet it is not routinely performed during diabetic follow-ups. In developing countries, including Ethiopia, UTIs are often managed empirically, which can contribute to the emergence and spread of antimicrobial-resistant strains, a major cause of treatment failure in DM patients with UTIs27. Moreover, the inappropriate use of antibiotics significantly contributes to the increasing resistance of bacterial uropathogens to commonly prescribed antimicrobials28.
In general, there is a limited availability of thorough information concerning the burden of UTIs, trends in antimicrobial susceptibility, and the contributing factors to these infections among DM patients in southern Ethiopia, especially in the study area. This study seeks to fill this information gap by providing in-depth data on the prevalence of UTIs, the resistance profiles of urinary bacterial pathogens to frequently used antibiotics, and the various determinants influencing these infections among DM patients. The findings will improve the understanding of local patterns of antimicrobial resistance, inform treatment guidelines, and help develop targeted strategies to lower UTI incidence and improve patient care in the region. Therefore, this study aimed to evaluate the prevalence of bacterial UTIs and identify the causative pathogens, examine their antimicrobial susceptibility patterns, and explore the risk factors among DM patients at Bule Hora University Teaching Hospital (BHUTH), southern Ethiopia.
Method
Study area, design, and period
An institution-based cross-sectional study was carried out between July 1 and August 30, 2023, at the BHUTH in the southern Ethiopian region of Oromia. With a catchment population of nearly 1.3 million, the hospital is the largest in the West Guji Zone29, and it functions as a center for clinical services, teaching, and training.
Population
The source population consisted of all DM patients visiting BHUTH. The study population included all adult DM patients, regardless of whether they exhibited symptoms of urinary tract infection, who were admitted to the hospital during the study period and met the inclusion criteria.
Sample size determination and sampling techniques
The sample size for this study was determined using the single population proportion formula, with a prior study in Ethiopia reporting a prevalence of 10.5%30, a 95% confidence level (Z = 1.96), and a 4% margin of error applied. After accounting for a 10% non-response rate, the final sample size was calculated to be 249.
All the study participants were selected through systematic random sampling. To determine the sampling interval (k), the total number of DM patients visiting the hospital’s diabetics center (980) was divided by the required sample size for the study. The formula used was k = N/n, where N represents the total number of DM patients (980) and n is the required sample size. The calculation resulted in k 980/249, which is approximately 3.9. Therefore, every fourth eligible DM patient was selected for enrollment and interview.
Variables
In this study, the magnitude of bacterial UTIs was considered the dependent variable, while sociodemographic and clinical factors, including gender, age, BMI, residence, marital status, educational background, history of hypertension, and previous history of UTI were treated as predictor variables.
Eligibility criteria
The study included all adult DM patients aged 18 and above, regardless of whether they exhibited symptoms of UTI, who visited BHUTH for follow-up or new diagnoses during the study period. Exclusions were made for DM patients who had taken antibacterial medications in the two weeks prior, pregnant DM patients, and those with a known history of UTI.
Data collection and laboratory methods
Data collection
Data collectors underwent a two-day training session conducted by the principal investigator, which covered urine sample collection, sample processing, colony characterization, and antimicrobial susceptibility testing before data collection. Sociodemographic and clinical data were gathered using a structured, pre-tested questionnaire through face-to-face interviews, supplemented by a review of patient medical records31.
Laboratory methods
Urine sample collection
Diabetes mellitus (DM) patients were instructed to provide a 10–15 ml sample of freshly voided midstream urine, collected in a sterile, dry, wide-neck, leakproof, screw-capped container. The container was to be clearly labeled with a unique sample number, as well as the date and time of collection.
Urine culture, isolation, and identification
Urine samples, thoroughly mixed, were inoculated onto Cysteine-Lactose Electrolyte Deficient (CLED) medium, MacConkey agar, and blood agar using a sterile, calibrated wire loop with a 1-µl capacity. The media were incubated aerobically at 35–37 °C for 24 h. The following day, bacterial growth was assessed on each medium, and a total colony count was performed to identify significant bacteriuria. A culture with ≥ 100 colonies of a single bacterial type (≥ 105 cfu/mL) was considered significant. To identify Gram-negative isolates, a series of biochemical tests, including indole production, Triple Sugar Iron (TSI) test, hydrogen sulfide and gas production, oxidase test, citrate utilization, urease test, and motility test, were conducted. For Gram-positive bacteria, coagulase and catalase slide tests were performed32.
Antimicrobial susceptibility testing
The antimicrobial susceptibility patterns of the isolates against locally available antimicrobials were assessed using the agar disk diffusion method (Kirby-Bauer method) on Mueller–Hinton agar (MHA, Oxoid, England), following the Clinical Laboratory Standards Institute (CLSI) guidelines. Three to five pure colonies were selected, diluted in 5 mL of sterile normal saline (0.85% NaCl), and mixed to form a homogeneous suspension. The resulting turbidity was then adjusted to match the 0.5 McFarland standard.
A sterile cotton swab was immersed in the suspension, rotated against the tube wall to remove excess fluid, and then evenly streaked onto the surface of Mueller–Hinton agar. The plates were left at room temperature to dry for 3–5 min33. Antibiotic discs were then placed on the agar using sterile forceps, and the plates were incubated for 16 to 18 h at 37 °C. The diameter of the zone of inhibition around each disc was measured to the nearest millimeter using a ruler, and the isolates were classified as sensitive, intermediate, or resistant based on the CLSI standard table34. The selection of antimicrobials was guided by the CLSI guidelines and the availability of drugs locally. Susceptibility testing was conducted using the following antimicrobial agents: Ciprofloxacin (5 µg), Ceftriaxone (30 µg), Ampicillin (10 µg), Amoxicillin-clavulanic acid (30 µg), Vancomycin (30 µg), Penicillin (10 units), Norfloxacin (10 µg), Nitrofurantoin (30 µg), and Gentamicin (10 µg)34.
Data quality assurance
The structured questionnaire, initially prepared in English, was translated into Afan Oromo by a language expert and then back-translated into English by a second expert. A pre-test of the questionnaire was conducted on 5% of adult DM patients attending Kerach Primary Hospital, Ethiopia, before the actual data collection, with corrections made based on the feedback from the pre-test. Data collectors underwent three days of training (two days before and one day after the pre-test) on data collection techniques, urine sample collection, sample processing, colony characterization, and antimicrobial susceptibility testing. The sterility of the paper culture medium was confirmed by incubating 35% of the batch at 37 °C overnight34. For the biochemical tests, the medium was inoculated with bacterial species known to produce either positive or negative reactions. Reference strains of E. coli (ATCC 259) and P. aeruginosa (ATCC 27853) were used to validate the performance of the culture media and antimicrobial discs. All microbiological results were carefully documented and subsequently rechecked by an experienced microbiologist.
Data analysis and interpretations
The collected data were coded and entered using EPI-Data version 4.6, then exported to IBM SPSS Statistics 17 for analysis. The data were organized, summarized, and presented using descriptive statistics. Variables with a P-value < 0.25 in the bivariate analysis were further examined using multivariate logistic regression to identify independently associated factors. A variable with a P-value < 0.05 and a 95% confidence interval in the multivariate logistic regression was considered statistically significant.
Ethical consideration
This study adhered to both national and international ethical guidelines. It was approved by the Institutional Health Research Ethics Review Committee of Bule Hora University (IHRERC/2023), and data collection permission was granted by the hospital director of BHUTH. All procedures followed the necessary ethical standards and the Declaration of Helsinki. The objectives, potential risks, and benefits were explained, and informed consent was obtained from all the study participants, who were also informed of their right to withdraw from the study at any time without consequence. Participants were assured that their data and samples would be kept confidential using codes and would be used solely for the study.
Results
Socio-demographic and clinical characteristics of the study participants
The study involved 249 DM patients, both symptomatic and asymptomatic for UTI. Of the participants, 144 (57.8%) were male, and 105 (42.2%) were female. Ages ranged from 18 to 89 years, with a mean age of 46.9 years. The largest age group, comprising 36.9% of participants, was between 35 and 51 years. Regarding marital status, 147 (59%) were married. More than half of the participants (139, 54.8%) lived in urban areas, and 54.6% had no formal education. Additionally, 82.3% were unemployed (Table 1). The clinical characteristics of the study participants are also presented in Table 1. Among the 249 study participants, 200 (80.3%) did not show signs or symptoms of UTIs during data collection, and 182 (73.1%) had no history of previous UTIs. The majority were diagnosed with type II DM (153, 61.4%). More than half of the participants (134, 53.8%) had been living with DM for five years or less. Additionally, 131 (56.2%) participants had fasting blood glucose levels exceeding 126 mg/dl.
Types of bacterial uropathogens isolated from diabetic patients
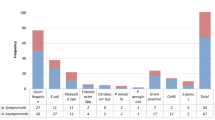
The overall prevalence of bacterial urinary tract infections among DM patients was 15.7% (95% CI: 11.1, 20.2). Of these, 11 (22.4%) were symptomatic, and 28 (14%) were from asymptomatic patients. The prevalence of UTIs was 21.9% in females and 11.1% in males. A higher prevalence of UTIs, 18.8%, was observed in the age group 52–68 years (Table 2).
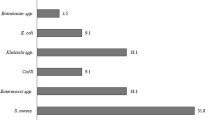
A total of 39 bacterial uropathogens were isolated from 249 DM patients tested for UTIs. Of these, gram-negative bacteria were more common, comprising 29 (74.4%) isolates, while gram-positive bacteria accounted for 10 (25%). Six bacterial species were identified, with E. coli being the most prevalent, accounting for 15 isolates (38.5%) isolates. This was followed by CONS (7, 17.9%), Klebsiella species (6, 15.4%), P. aeruginosa (4, 10.3%), Proteus species (3, 7.7%), S. aureus (3, 7.7%), and Enterobacter (1, 2.6%) (Fig. 1).
Antimicrobial susceptibility profiles of Gram-negative isolates
The Gram-negative isolates demonstrated strong sensitivity to Nitrofurantoin (82.14%), Gentamicin (78.57%), and Ceftriaxone (65.5%). In contrast, notable resistance was observed to Ampicillin (93.1%), Amoxicillin-clavulanic acid (79.3%), and Tetracycline (75.6%). Among the Gram-negative bacteria, E. coli exhibited the highest sensitivity to Nitrofurantoin (93.3%, n = 14), followed by Ciprofloxacin (73.3%, n = 11), Gentamicin (73.3%, n = 11), and Ceftriaxone (66.7%, n = 10). However, resistance was particularly high to Ampicillin (100%) and Amoxicillin-clavulanic acid (80%, n = 12).
Klebsiella species exhibited the highest sensitivity to Nitrofurantoin (100%), Gentamicin (83.3%), and Ceftriaxone (66.7%). Conversely, the highest resistance was observed against Ampicillin (100%), Amoxicillin-clavulanic acid (100%), and Tetracycline (83.3%). Among the 3 (7.7%) Proteus isolates, Amoxicillin-clavulanic acid showed the highest sensitivity (100%, n = 3), followed by Ceftriaxone (66.7%, n = 2). Resistance was most prominent against Ampicillin (66.7%, n = 2) and Tetracycline (66.7%, n = 2).
For P. aeruginosa, the highest sensitivity was noted for Gentamicin (100%, n = 4), Ceftriaxone (75%, n = 3), and Ciprofloxacin (50%, n = 2). However, it demonstrated complete resistance to Ampicillin and Amoxicillin-clavulanic acid (100%, n = 4) and high resistance to Ampicillin (75%, n = 3) and Norfloxacin (75%, n = 3). Enterobacter isolates showed full sensitivity to Nitrofurantoin (100%), Ceftriaxone (100%), and Gentamicin (100%), with complete resistance to both Ampicillin and Amoxicillin-clavulanic acid (100%) (Table 3).
Antimicrobial susceptibility profile of Gram-positive isolates
Among the Gram-positive isolates, CONS were the most prevalent, exhibiting high sensitivity to Vancomycin (100%), Ceftriaxone (71.4%), Ciprofloxacin (57.14%), and Gentamicin (57.14%). However, these isolates showed complete resistance to Ampicillin (100%) and significant resistance to Penicillin (71.4%). S. aureus demonstrated the highest sensitivity to Ceftriaxone (66.7%, n = 2), Vancomycin (66.7%, n = 2), Nitrofurantoin (66.7%, n = 2), and Gentamicin (66.7%, n = 2). On the other hand, the highest resistance was noted against Ampicillin (100%, n = 3), Penicillin (66.7%, n = 2), and Tetracycline (66.7%, n = 2) (Table 4).
Multi-drug resistant (MDR) profile of bacterial isolates
In this study, out of 39 bacterial isolates, 17 (43.6%) exhibited resistance to at least one antibiotic from three or more categories. Among the Gram-negative bacterial isolates, E. coli (53.3%, n = 8), Klebsiella species (50%, n = 3), P. aeruginosa (25%, n = 4), and Proteus species (33.3%, n = 1), and among the Gram-positive bacterial isolates, CONS (42.86%, n = 3), S. aureus (33.3, n = 3), were identified as multi-drug resistant isolates.
Factors contributing to urinary tract infections in diabetic patients
In the bivariate logistic regression analysis, variables with a p-value < 0.25 were considered potential candidates for multivariate logistic regression. These included gender, marital status, lack of formal education, duration of DM, previous history of UTIs, current UTI symptoms, antibiotic usage, and type of DM. Among the variables included in the multivariate logistic regression analysis, being female, having a DM duration of more than five years, and having a history of previous UTIs were statistically significant factors associated with UTIs (p-value < 0.05). The study found that females were twice as likely to develop UTIs as males (AOR 2.132, 95% CI 1.044–4.379).
This study found that patients with a history of DM lasting more than five years were twice as likely to develop UTI compared to those with diabetes for less than five years (AOR 2.30, 95% CI 1.11–4.786). Additionally, UTIs were more common among DM patients with a previous history of UTIs (26.9%) compared to those without such a history (11.5%). Patients with a prior UTI history were nearly three times more likely to develop a UTI than those without (AOR 2.562, 95% CI 1.296–5.29) (Table 5).
Discussion
Diabetic patients are at an increased risk of various infections, particularly UTIs19. UTIs remain one of the most prevalent issues faced by both patients and clinicians, as the bacteria involved can reduce the effectiveness of broad-spectrum antibiotics, leading to significant therapeutic challenges and affecting patient outcomes35. This study found that the overall prevalence of bacterial urinary tract infections in diabetic patients was 15.7% (95% CI, 11.1–20.2). The results were similar to those of other studies conducted in Hawassa (13.8%)36, Harar (15.4%)37, Metu (16.7%)27, Nekemte (16.5%)31, Sudan (19.5%)21, and China (11.2%)38.
This finding was lower than those from studies conducted in Arba Minch (33.8%)39, Pakistan (51.25%)40, India (34%)41, and Malaysia (40.2%)42. However, it was slightly higher than the results from studies in Addis Ababa (9.8%)20, Iran (8.06%)43, and Romania (10.7%)44. These variations may be attributed to differences in study populations, sample sizes, geographical factors, personal hygiene practices, and the methods used for bacterial identification and isolation.
This study found that the prevalence of bacterial uropathogens was higher in diabetic patients with symptomatic UTIs compared to those with asymptomatic UTIs. The bacterial prevalence of symptomatic UTIs in this study (22.4%) was similar to findings in Hawassa (21.7%)36 and Sudan (17.1%)21, but lower than those reported in Arba Minch (64.1%)39, Nigeria (35%)45, and India (49.15%)17. These differences may be attributed to variations in study populations, settings, and periods across different regions.
In this study, a total of 39 bacterial uropathogens from seven species were isolated, with Gram-negative bacteria being more prevalent (29 isolates, 74.4%) compared to Gram-positive bacteria (10 isolates, 25.6%). This predominance of Gram-negative bacteria aligns with findings from studies in Kombolcha (40 isolates, 70.2%)46 and Dessie (28 isolates, 71.8%)47. However, the prevalence in this study was slightly higher than that reported in Arba Minch (30.6%)48, Gondar (57.1%)49, and India (51.9%)50. These differences may be attributed to geographical factors and potential temporal variations within populations.
In this study, E. coli (38.5%) was the most predominant organism, which aligns with findings from studies in Hawassa (33.3%)36, Kombolcha (33.3%)46, Harar (47.5%)37, as well as studies from other countries like Uganda (41.9%)51, Nigeria (39.6%)45, and India (25.6%)13. However, this contrasts with a study from Nekemte, Ethiopia, where the most commonly isolated pathogen was CoNS (24.2%)31. This difference may be due to E. coli being a common inhabitant of the gastrointestinal tract, from where it ascends to the urinary tract, aided by virulence factors such as toxins, fimbriae, and pili that facilitate colonization and invasion of the urinary epithelium. Additionally, the spectrum of infections caused by E. coli may vary between hospital settings and communities52.
Coagulase-negative staphylococcus (CoNS) (17.9%) was the second most predominant isolate after E. coli. This finding is consistent with studies conducted in Addis Ababa (18.9%)26, Harar (15%)37, Dilla (20.8%)14, and India (18.9%)52. However, it contradicts studies from Arba Minch39 and Sudan21, where Klebsiella species was identified as the second most common isolate. These differences may be attributed to variations in methods used to identify bacterial species and geographical factors.
In this study, Gram-negative isolates exhibited high susceptibility to Nitrofurantoin (82.14%), Gentamicin (78.57%), and Ceftriaxone (65.5%). The strong susceptibility to Nitrofurantoin and Gentamicin aligns with findings from similar studies conducted in Hawassa (Nitrofurantoin 94.1%, Gentamicin 64.7%)30 and India (Nitrofurantoin 80.8%, Gentamicin 76.8%)13. However, high resistance rates were observed to Ampicillin (93.1%) and Tetracycline (75.6%). These resistance patterns were comparable to a previous study from Metu, which reported resistance rates of Ampicillin (80%) and Tetracycline (68%)27. Such resistance may be linked to self-medication practices, misuse of antibiotics, their easy availability and low cost, and the lack of up-to-date guidelines for medication selection.
Among the Gram-negative isolates, E. coli demonstrated high sensitivity to Nitrofurantoin (93.3%), Ciprofloxacin (73.3%), and Gentamicin (73.3%). These results are consistent with a study from Hawassa, Ethiopia, which reported 100% sensitivity to Nitrofurantoin, 58.3% to Gentamicin, and 75% to Ciprofloxacin30. However, in this study, E. coli exhibited complete resistance to Ampicillin (100%) and significant resistance to Amoxicillin-Clavulanic acid (80%).
This finding is supported by a study from Dilla, Ethiopia, which reported 100% resistance to Ampicillin and 85.7% resistance to Amoxicillin-Clavulanic acid14. However, it contradicts a study from Sudan, where resistance to Ampicillin was lower (27.3%)21. In this study, Gram-positive bacteria also exhibited high resistance to Ampicillin (100%), consistent with a study from Gondar University Hospital, Ethiopia, which found similar resistance levels49. This resistance could be attributed to the use of medications without prescriptions and the indiscriminate use of commonly prescribed drugs, contributing to an increase in resistance.
Among Gram-positive isolates, CoNS demonstrated sensitivity to Vancomycin (100%) and Ciprofloxacin (85.7%), consistent with a study from Dilla, Ethiopia, which reported 100% sensitivity to Vancomycin and 80% to Ciprofloxacin14. The current study found that 17 bacterial isolates (43.6%) exhibited multiple drug resistance, a finding similar to a study conducted in Debre Tabor, which reported 56.7%53. However, the resistance rate in our study was lower than those observed in Metu (87.2%)27 and Harar (92.5%)37. This discrepancy could be attributed to easier access to medications outside of treatment centers, leading to the indiscriminate use of antibiotics without prescriptions36. The rise in multiple antibiotic-resistant bacteria is accelerating over time, driven by factors such as inadequate infection control, the increasing frequency of infections, limited antibiotic susceptibility testing, and the lack of updated drug selection guidelines.
The current study found that women with diabetes were 2.13 times more likely to develop UTIs than men, consistent with studies from Arba Minch39, Metu27, and Pakistan40. This may be due to anatomical factors such as a shorter urethra and proximity to the perirectal area, which facilitate pathogen colonization, as well as the lack of prostatic secretion in females4. Additionally, diabetic patients with a history of diabetes longer than five years were twice as likely to develop UTIs compared to those with a shorter duration, which aligns with findings from Wollo47, Addis Ababa20, and Romania44. However, this contradicts studies from Iran (61) and Addis Ababa (16), which found no significant association between diabetes duration and UTIs. Longer DM duration may increase complications such as neuropathy, catheterization, and frequent hospitalizations, all contributing to a higher UTI prevalence54.
This study found that diabetic patients with a history of UTI had a significantly higher likelihood of developing another UTI compared to those without a prior history. This is consistent with a study conducted in Addis Ababa, Ethiopia20, but contradicts findings from Hawassa, Ethiopia36, where patients without a history of UTI were more likely to develop one. This discrepancy may be due to infection relapse from inadequate treatment, the high sugar concentration in diabetic urine providing a conducive environment for bacterial growth, or recall bias55.
Strength and limitation of the study
The strength of this study lies in its robust design, using a large sample of 249 DM patients from a single institution, allowing for an accurate assessment of bacterial UTI prevalence, antimicrobial resistance patterns, and associated risk factors. The use of standard microbiological procedures and antimicrobial susceptibility testing ensures reliable and valid results. However, a limitation of the study is its cross-sectional design, which prevents establishing causal relationships between risk factors and UTIs. Additionally, the study focused on a single hospital, which may limit the generalizability of the findings to other regions or populations.
Conclusion and recommendation
The prevalence of bacterial UTIs was 15.7%, with E. coli as the most common pathogen, followed by CoNS and Klebsiella. High antimicrobial resistance was noted, with 43.6% of isolates being multidrug-resistant. Gender, DM duration > 5 years, and a history of UTIs were key risk factors in DM patients. Empirical therapy should be guided by local bacterial profiles, with a focus on rational antibiotic use in the study area. Health education on UTIs and antibiotic use is essential. Restrictions on antibiotics like Ampicillin and Amoxicillin-clavulanic acid are recommended, with Nitrofurantoin, Gentamicin, Ceftriaxone, and Vancomycin as alternatives. Furthermore, a follow-up study involving DM patients is essential to gaining valuable insights into the progression of UTIs.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- AOR:
-
Adjusted odd ratio
- AST:
-
Antimicrobial susceptibility test
- ATCC:
-
American type culture collection
- BHUTH:
-
Bule Hora University Teaching Hospital
- BMI:
-
Body mass index
- CI:
-
Confidence interval
- CFU:
-
Colony forming units
- CLSI:
-
Clinical Laboratory Standards Institute
- CONS:
-
Coagulase negative Staphylococci
- DM:
-
Diabetes mellitus
- SPSS:
-
Statistical package for social science
- SOP:
-
Standard operating procedures
- UTIs:
-
Urinary tract infections
- WHO:
-
World Health Organization
References
Foxman, B. The epidemiology of urinary tract infection. Nat. Rev. Urol. 7(12), 653–660 (2010).
Tan, N. C. et al. Accuracy of urinary symptoms and urine microscopy in diagnosing urinary tract infection in women. Fam. Pract. 36(4), 417–424 (2019).
Addis, T., Mekonnen, Y., Ayenew, Z., Fentaw, S. & Biazin, H. Bacterial uropathogens and burden of antimicrobial resistance pattern in urine specimens referred to Ethiopian Public Health Institute. PLoS ONE 16(11), e0259602 (2021).
Kalal, B. S. & Nagaraj, S. Urinary tract infections: A retrospective, descriptive study of causative organisms and antimicrobial pattern of samples received for culture from a tertiary care setting. J. Germs 6(4), 132 (2016).
Öztürk, R. & Murt, A. Epidemiology of urological infections: A global burden. World J. Urol. 38(11), 2669–2679 (2020).
Cho, N. H. et al. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. J Diabetes Res. 138, 271–281 (2018).
Cho, N. H. et al. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diab. Res. Clin. Pract. 138, 271–281 (2018).
Athyros, V. G. et al. Diabetes and lipid metabolism. Hormones (Athens) 17(1), 61–67 (2018).
Poorolajal, J., Farbakhsh, F., Mahjub, H., Bidarafsh, A. & Babaee, E. How much excess body weight, blood sugar, or age can double the risk of hypertension?. Public Health 133, 14–18 (2016).
Alrwithey, F. A. et al. Urinary tract infection in patients with diabetes mellitus. J. Egypt. J. Hosp. Med. 69(3), 2133–2136 (2017).
Al-Tulaibawi, N. A. J. Prevalence and sensitivity of bacterial urinary tract infection among adult diabetic patients in Misan Province, Iraq. J. Pure Appl. Microbiol. 13(2), 847–853 (2019).
Jagadeesan, S., Tripathi, B. K., Patel, P. & Muthathal, S. Urinary tract infection and diabetes mellitus-etio-clinical profile and antibiogram: A North Indian perspective. J. Fam. Med. Prim. Care 11(5), 1902–1906 (2022).
Kande, S., Patro, S., Panigrahi, A., Khora, P. K. & Pattnaik, D. Prevalence of uropathogens and their antimicrobial resistance pattern among adult diabetic patients. Indian J. Public Health 65(3), 280–286 (2021).
Diriba, K., Awulachew, E. & Bizuneh, B. Identification of bacterial uropathogen and antimicrobial resistance patterns among patients with diabetic and hypertension attending Dilla University General Hospital, Dilla, Ethiopia. Infect. Drug Resist. 16, 4621–4633 (2023).
Gurjar, D., Mathur, A., Sai, R., Lakesar, A. & Saxena, P. Recent trends in the antimicrobial susceptibility patterns of urinary pathogens in type II diabetes mellitus. J. Int. J. Res. Med. Sci. 6, 1288–1291 (2018).
Zaha, D. C. et al. Prevalence of urinary tract infection and antimicrobial susceptibility among diabetic patients. Farmacia 68(2), 250–255 (2020).
Lature, L. H., Lature, M. L. & Pyadala, N. J. I. Assessment of urinary tract infections among type 2 diabetic patients in a rural teaching hospital. Sangareddy 7(1), 28–32 (2020).
Desouky, D. E., Gabr, H. M., El-Helbawy, M., Hathout, H. M. J. & Sciences, H. Urinary tract infection: Prevalence, risk factors, bacterial etiologies and antimicrobial resistance profile among Egyptian diabetic patients: Urinary tract infection: prevalence, risk factors, bacterial etiologies and antimicrobial resistance profile among Egyptians. Eur. J. Med. Health Sci. 2, 4 (2020).
Tegegne, K. D. et al. Prevalence of urinary tract infections and risk factors among diabetic patients in Ethiopia, a systematic review and meta-analysis. PLoS ONE 18(1), e0278028 (2023).
Yenehun Worku, G., Belete Alamneh, Y. & Erku, A. W. Prevalence of bacterial urinary tract infection and antimicrobial susceptibility patterns among diabetes mellitus patients attending Zewditu Memorial Hospital, Addis Ababa, Ethiopia. Infect. Drug Resist. 14, 1441–1454 (2021).
Hamdan, H. Z. et al. Urinary tract infections and antimicrobial sensitivity among diabetic patients at Khartoum, Sudan. J. Ann. Clin. Microbial. Antimicrob. 14(1), 1–6 (2015).
Geerlings, S. E. Clinical presentations and epidemiology of urinary tract infections. Microbiol. Spectrum 4, 5. https://doi.org/10.1128/microbiolspec.uti-0002-2012 (2016).
Dhar, A. K., Anitha, K. & Bhatt, S. Immune System and Diabetes Mellitus 19–47 (Elsevier, 2024).
W Thomsen R, Mor A. Diabetes and risk of community-acquired respiratory tract infections, urinary tract infections, and bacteremia (2012).
Madrazo, M. et al. Clinical impact of multidrug-resistant bacteria in older hospitalized patients with community-acquired urinary tract infection. BMC Infect. Dis. 21, 1–7 (2021).
Woldemariam, H. K. et al. Common uropathogens and their antibiotic susceptibility pattern among diabetic patients. BMC Infect Dis. 19(1), 43 (2019).
Gutema, T., Weldegebreal, F., Marami, D. & Teklemariam, Z. Prevalence, antimicrobial susceptibility pattern, and associated factors of urinary tract infections among adult diabetic patients at Metu Karl Heinz Referral Hospital, Southwest Ethiopia. Int. J. Microbiol. 2018, 7591259 (2018).
Okalla, C. E., Shu, E. Y., Etoga, M. E., Mengue, E. R. & Adiogo, D. Bacterial and fungal uropathogens in diabetic patients and their susceptibility patterns: Case study of two hospitals in Douala. Asian J. Microbiol. Biotechnol. 3(3), 79–89 (2018).
Aliyo, A. & Gemechu, T. Assessment of intestinal parasites and associated factors among HIV/AIDS patients on antiretroviral therapy at Bule Hora General Hospital, West Guji, Ethiopia. SAGE Open Med. 10, 20503121221124684 (2022).
Mohammed, A., Beyene, G. & Teshager, L. Urinary pathogenic bacterial profile, antibiogram of isolates and associated risk factors among diabetic patients in Hawassa town, southern Ethiopia: A cross-sectional study. Urol. Nephrol. Open Access J. 8(4), 84–91 (2020).
Kebamo, S., Dabso, R., Deressa, A. & Gebrie, M. Urinary tract infection: Bacterial etiologies, drug resistance profile and associated risk factors among diabetic patients attending Nekemte Referral Hospital, Ethiopia. J. Am. J. Curr. Microbiol. 5(1), 19–31 (2017).
Dimri, A. G., Chaudhary, S., Singh, D., Chauhan, A. & Aggarwal, M. Morphological and biochemical characterization of food borne gram-positive and gram-negative bacteria. Sci. Arch. 1(1), 16–23 (2020).
Patra, J. K. et al. Isolation, culture, and biochemical characterization of microbes. Pract. Guide Environ. Biotechnol. 6, 83–133 (2020).
Humphries, R., Bobenchik, A. M., Hindler, J. A. & Schuetz, A. N. Overview of changes to the clinical and laboratory standards institute performance standards for antimicrobial susceptibility testing, M100. J. Clin. Microbiol. 59, 12. https://doi.org/10.1128/jcm.00213-21 (2021).
Adler, A. & Katz, D. E. The continuing plague of extended-spectrum β-lactamase producing enterbacterales infections: An update. J. Infect. Dis. Clin. 34(4), 677–708 (2020).
Nigussie, D. & Amsalu, A. Prevalence of uropathogen and their antibiotic resistance pattern among diabetic patients. Turk. J. Urol. 43(1), 85–92 (2017).
Abate, D., Kabew, G., Urgessa, F. & Meaza, D. Bacterial etiologies, antimicrobial susceptibility patterns and associated risk factors of urinary tract infection among diabetic patients attending diabetic clinics in Harar, Eastern Ethiopia. East Afr. J. Health Biomed. Sci. 1(2), 11–20 (2017).
He, K., Hu, Y., Shi, J.-C., Zhu, Y.-Q. & Mao, X.-M. Prevalence, risk factors and microorganisms of urinary tract infections in patients with type 2 diabetes mellitus: A retrospective study in China. J. Therap. Clin. Risk Manag. 6, 403–408 (2018).
Mama, M. et al. Prevalence and associated factors of urinary tract infections among diabetic patients in Arba Minch Hospital, Arba Minch province, South Ethiopia. Turk. J. Urol. 45(1), 56 (2019).
Ahmad, S., Hussain, A., Khan, M. S. A. & Shakireen, N. Diabetes mellitus and urinary tract infection: Causative uropathogens, their antibiotic susceptibility pattern and the effects of glycemic status. Pak. J. Med. Sci. 36(7), 1550 (2020).
Bhullar, S., Harale, M. & Oomen, A. B. J. Study of prevalence of asymptomatic urinary tract infection in patients with uncontrolled diabetes mellitus. 7329–7336 (2023).
Shah, M. A. et al. Prevalence and associated factors of urinary tract infections among diabetic patients. Health Sci. J. 13(2), 1–5 (2019).
Nashtar, S. B., Hashim, I. & Hamdan, S. J. Assessment of antimicrobial susceptibility patterns in urine culture of patients with urinary tract infection attending Al-Kindy teaching hospital. Iraqi Med. J. 7, 1012–1020 (2018).
Chiţă, T. et al. Urinary tract infections in Romanian patients with diabetes: Prevalence, etiology, and risk factors. Ther. Clin. Risk Manag. 13, 1–7 (2017).
Clare, E. C., Juliet, A. O., Maureen, O. C., Romeo, A. J. & Ada, K. O. E. Urinary Tract infections and antimicrobial sensitivity patterns of uropathogens isolated from diabetic and non-diabetic patients attending some hospitals in Awka. Am. J. Microbiol. Res. 9(3), 83–91 (2021).
Oumer, O., Metaferia, Y. & Gebretsadik, D. Bacterial uropathogens, their associated factors, and antimicrobial susceptibility pattern among adult diabetic patients in two health centers at Kombolcha town, Northeastern Ethiopia. SAGE Open Med. 10, 20503121221139148 (2022).
Walelgn, B., Abdu, M. & Kumar, P. The occurrence of urinary tract infection and determinant factors among diabetic patients at Dessie Referral Hospital, South Wollo, Northeast Ethiopia. SAGE Open Med. 9, 20503121211060616 (2021).
Mitiku, A. et al. Magnitude and antimicrobial susceptibility profiles of Gram-Negative bacterial isolates among patients suspected of urinary tract infections in Arba Minch General Hospital, southern Ethiopia. J. PLoS One 17(12), e0279887 (2022).
Yismaw, G., Asrat, D., Woldeamanuel, Y. & Unakal, C. G. Urinary tract infection: Bacterial etiologies, drug resistance profile and associated risk factors in diabetic patients attending Gondar University Hospital, Gondar, Ethiopia. J. Eur. J. Exp. Biol. 2(4), 889–898 (2012).
Bharti, A. et al. Asymptomatic bacteriuria among the patients of type 2 diabetes mellitus. J. Fam. Med. Primary Care 8(2), 539 (2019).
Odoki, M. et al. Prevalence of bacterial urinary tract infections and associated factors among patients attending hospitals in Bushenyi district, Uganda. Int. J. Microbial. 2019, 424 (2019).
Kaur, P., Sachan, R. S. K., Karnwal, A. & Devgon, I. A review on clinical manifestation and treatment regimens of UTI in diabetic patients. Iran. J. Med. Microbiol. 16(2), 98–115 (2022).
Worku, S., Derbie, A., Sinishaw, M. A., Adem, Y. & Biadglegne, F. Prevalence of bacteriuria and antimicrobial susceptibility patterns among diabetic and nondiabetic patients attending at Debre Tabor Hospital, Northwest Ethiopia. Int. J. Microbiol. 2017(1), 5809494 (2017).
Fu, A. Z. et al. Risk characterization for urinary tract infections in subjects with newly diagnosed type 2 diabetes. J. Diab. Comp. 28(6), 805–810 (2014).
Ahmed, A. E., Abdelkarim, S., Zenida, M., Baiti, M. A. H., Alhazmi, A. A. Y., Alfaifi, B. A. H., et al. (eds.) Prevalence and associated risk factors of urinary tract infection among diabetic patients: A cross-sectional study. In Healthcare. (MDPI, 2023).
Acknowledgements
The authors would like to express their heartfelt gratitude to the study participants for their cooperation in providing valuable data and specimens. We also thank Bule Hora University for the opportunity to conduct the research and for granting ethical clearance. Special thanks go to the Department of Medical Laboratory Science at Bule Hora University and its staff for their exceptional support and collaboration throughout the project, as well as to Adama Regional Public Health Laboratory for their material and reagent assistance.
Author information
Authors and Affiliations
Contributions
M.E.G., G.B., K.B., and A.A.A. conceptualized the ideas, supervised data collection, conducted laboratory experiments, analyzed and interpreted the data, and wrote the manuscript. E.C., N.C., D.M.B., L.D., M.W., D.T., and B.B.T. contributed to the data analysis, conceived the structure and content of the manuscript, and manuscript writing. All authors were also responsible for acquiring the reagents and materials. All authors have approved the final manuscript and are accountable for ensuring the accuracy and integrity of all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gena, M.E., Bitew, G., Chane, E. et al. Bacterial profile and antimicrobial susceptibility pattern of urinary tract infection among diabetic patients at Bule Hora University Teaching Hospital, Southern Ethiopia. Sci Rep 15, 24112 (2025). https://doi.org/10.1038/s41598-025-05782-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-05782-8