Abstract
Epigenetic modifications can influence the phenotypes of subsequent generations through intergenerational and transgenerational effects. The aim of the research was to assess the impact of epigenetic factors acting during embryonic development on the structure and cellular composition of lymphoid organs in three generations of chickens. Two groups of eggs were injected once (in the F1 generation) with a synbiotic (SYNs) or a synbiotic + choline (SYNCHs), while two other groups were injected in each successive generation (F2, F3—SYNr, SYNCHr). Synbiotic administration resulted in an increased cortex/medulla ratio of the thymus in the F1 but not in subsequent generations. In the spleen, an intergenerational effect (from F1 to F2) was found in the choline-supplemented (SYNCHs) groups but not in the SYNs groups. Not all changes observed in the F1 were evident in the F2 generation. No intergenerational effect was found in the cecal tonsil, and no transgenerational effects were observed in any of the tested organs. In ovo administration of synbiotics with choline may induce intergenerational phenotypic effects on specific immune organs. However, these effects either persisted through the first two generations or appeared solely in the F1 or F2 generations. Changes were evident in young birds but not in mature ones.
Similar content being viewed by others
Introduction
Epigenetic modifications are essential mechanisms that fine-tune gene expression in response to extracellular signals and environmental changes. These mechanisms are considered to involve covalent modifications to DNA, as well as gene expression and translation modifications by microRNAs, other noncoding RNAs, and nucleosome positioning or remodeling1. In vertebrates, critical epigenetic reprogramming events occur during early embryogenesis and germ cell development2.
One way nonDNA-based inheritance may occur is through the transfer of an altered parental microbiome3. A similar research trend has been observed following in ovo stimulation of the chicken embryo microbiome, leading to long-term postembryonic effects. When bioactive substances such as prebiotics, probiotics, or synbiotics are administered in ovo on the 12th day of incubation, they can directly influence the composition of the microbiota in chickens4,5,6,7, while also indirectly improving their physiological traits8,9,10, immunological traits11,12,13, intestinal development14, and performance traits4,15. The molecular mechanisms underlying these phenotypic effects have also been extensively studied16,17,18,19,20.
Scientific reports have shown that specific probiotics can modify the expression of selected miRNAs21,22,23,24. For instance, it was demonstrated that the administration of a probiotic containing Saccharomyces boulardii increased the expression of specific miRNAs, which in turn alleviated inflammation22. Similarly, delivery of Lactobacillus plantarum can reduce cecal inflammation in Salmonella Typhimurium-infected neonatal broiler chicks through the regulation of miRNA expression21.
Epigenetic mechanisms have been suggested to be responsible for silencing the expression of selected genes in the liver and spleen following early stimulation of the gut microbiota with specific bioactive substances25,26. Both of the two epigenetic mechanisms known to be responsible for gene silencing—DNA methylation and miRNA expression—were identified in genes silenced after in ovo administration of bioactive substances, such as prebiotics, probiotics, or synbiotics, in the chicken liver and spleen23,27. However, it has also been proved that the level of DNA methylation is dependent on tissue, genotype, and bioactive substance23,26,27,28.
Epigenetic inheritance refers to the concept that epigenetic modifications can influence not only the phenotypes of individuals directly exposed to specific factors but also those of their offspring and subsequent generations. This occurs through intergenerational and transgenerational mechanisms, which may involve epigenetic alterations during embryonic development or the transmission of epigenetic marks through gametes29,30. Epigenetic effects are broadly categorized into intergenerational and transgenerational inheritance. Intergenerational inheritance refers to the transmission of traits or phenotypes influenced by environmental factors to the next generation, often observed in cases where parental experiences affect offspring31. These parental effects are commonly regarded as examples of “context-dependent” epigenetic inheritance32. When an environmental stressor persists and continuously induces epigenetic changes across successive generations, it is referred to as a “context-dependent” multigenerational effect32. In contrast, transgenerational “germline-dependent” inheritance occurs when an organism’s germline is directly affected, leading to phenotypic modifications that persist across generations even in the absence of the original causative agent (i.e., the environmental stressor)33. Transgenerational transmission is characterized by the offspring (F1) passing the effects to descendants who have not been exposed to the stimulus, with altered phenotypes appearing in the second (F2) generation for male transmission or the third (F3) generation for female transmission29,32,34,35. Such changes may result from an environmental factor directly affecting a gestating embryo and the already-formed oocytes within a female embryo in mammals31,36.
The effectiveness of transmitting epigenetic modifications to subsequent generations depends on the timing of the environmental factor’s interaction with the organism’s epigenome. It has been indicated that, initially, global DNA demethylation followed by de novo methylation occurs during the migration of primordial germ cells (PGCs) toward their final destination in the gonads37. A second similar process occurs after fertilization38. These two periods in the life of birds represent specific windows of methylome sensitivity to environmental factors30, during which modifications are more easily induced than at other life stages39. This is why environmental stimuli during these two periods have a more profound impact on the animal phenotype compared to similar factors encountered later in life34. Moreover, intense somatic cell divisions occurring during the perinatal period facilitate the transfer and preservation of epigenetic patterns in all subsequent generations of cells derived from those exposed to modifying factors34. Furthermore, since the epigenome regulates gene expression, these epigenetic modifications can directly influence developmental processes33.
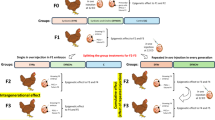
The aim of our research was to assess the impact of epigenetic factors acting during embryonic development on the structure and cellular composition of selected lymphoid organs in chickens. The addition of the choline as primary methyl group donor, was also tested. The experiment covered three generations of birds, allowing for the description of intergenerational and transgenerational effects. Moreover, the novelty of this work lies in the use of an in ovo chicken model to test the hypothesis of intergenerational and transgenerational inheritance of the influence of epigenetic modulators in birds. Additionally, this experimental design was innovative in its approach. In parallel, we established two experimental groups: one consisting of birds that received a single in ovo injection in the F1 generation and another comprising individuals subjected to repeated in ovo injections across successive generations. This setup enabled us to investigate both germline-dependent and context-dependent mechanisms of inheritance.
Results
Morphometrical analysis of central immune organs
The effect of administering synbiotic PoultryStar® solUS (Biomin GmbH, Herzogenburg, Austria; further referred to as PS) and synbiotic PS combined with choline in the thymus was assessed based on the C/M diameter ratio. On D14 of the F1 generation, a significant increase (P < 0.05) in this coefficient was observed in the SYNs group, along with an upward trend (P = 0.053) in the SYNCHs group (Fig. 1). However, no significant differences in the C/M ratio were observed between the examined groups in the F2 and F3 generations.
Cortex-to-medulla ratio in the thymus of chickens on day 14 after hatching. The control group (C) and groups treated in ovo with synbiotic (SYNs) or synbiotic combined with choline (SYNCHs) in the first generation, as well as those treated in the first and each subsequent generation with synbiotic (SYNr) or synbiotic combined with choline (SYNCHr), are shown. F1–F3 represent generations. A significant difference compared to C is indicated (*P < 0.05).
The C/M ratios in the bursa of Fabricius are presented in Fig. 2. The administration of bioactive compounds had no effect on the bursa of Fabricius in either the F1 generation or the subsequent generations.
Cortex-to-medulla ratio in the bursa of Fabricius of chickens on day 14 after hatching. The control group (C) and groups treated in ovo with synbiotic (SYNs) or synbiotic combined with choline (SYNCHs) in the first generation, as well as those treated in the first and each subsequent generation with synbiotic (SYNr) or synbiotic combined with choline (SYNCHr), are shown. F1–F3 represent generations. No significant differences compared to C were found.
An analogous analysis of the C/M ratio at 21 weeks of age was, in many cases, not possible due to significant involution of both the thymus and the bursa. In the remaining cases, no significant differences were found between the studied groups (data not shown).
To test the influence of environmental factors on the examined parameter, intergenerational comparisons of control groups were also performed. In the thymus, the C/M ratio differed significantly (P < 0.05) between all generations, with the lowest value observed in the F1 generation and the highest in the F2 generation (Fig. 3). In the bursa, the C/M ratio also showed a significant difference between the F1 and F2 generations, again reaching its lowest value in the F1 generation and the highest in the F2 generation (Fig. 3). It should be emphasized that while a natural effect of the rearing season on the C/M ratio was observed, all treated groups within each generation were compared to the same control group to ensure the validity of results within each individual generation.
Cell composition of peripheral immune organs
The influence of epigenetic factors on peripheral lymphoid organs was examined by analyzing the proportion of Bu-1+ cells (B lymphocytes), CD4+ cells (Th lymphocytes), and CD8+ cells (cytotoxic T lymphocytes and NK cells). Among the peripheral lymphoid organs, the spleen and CT were selected for analysis.
In the spleen of the F1 generation on day 14, a significant decrease in the number of CD8+ cells was observed in the SYNs group, which had previously received only the synbiotic, and in the SYNCHs group, which had received the synbiotic combined with choline during embryonic development (Fig. 4). However, this effect did not persist until the 21st week after hatching, at which point the proportion of all analyzed cell types showed no differences between the groups.
Area occupied by antigen-positive cells (mean ± SD) in the spleen of chickens from the control group (C) and groups treated in ovo with synbiotic (SYNs) or synbiotic combined with choline (SYNCHs) in the first generation, as well as those treated in the first and each subsequent generation with synbiotic (SYNr) or synbiotic combined with choline (SYNCHr). F1–F3 represent generations. Significant differences compared to C are indicated (*P < 0.05, **P < 0.01).
In the F2 generation on day 14, a significant decrease in the number of CD8+ cells was again observed in the SYNCHs group, which was also accompanied by a reduction in the number of Bu-1 positive cells (Fig. 4). By the 21st week after hatching, in the SYNCHs group, similar to the F1 generation, no differences in the number of immune cells were observed compared to the control. Interestingly, in the mature F2 SYNs group, a significant increase in the number of CD4+ cells was observed. The birds in this group had received a single PS injection in the F1 generation and were then reared without further interventions. In the F3 generation, the influence of epigenetic modulators on the number of spleen lymphocytes was not apparent, both on day 14 and at week 21 after hatching (Fig. 4).
In the spleen of the first generation, a significant increase in the CD4+/CD8+ cell ratio was observed in both the SYNs and SYNCHs groups, primarily due to a decrease in the percentage of CD8+ cells (Fig. 5). However, this effect was no longer present in chickens of this generation by week 21, nor was it observed in subsequent generations at either analyzed time point.
CD4+/CD8+ cell ratio in the spleen of chickens from the control group (C) and groups treated in ovo with synbiotic (SYNs) or synbiotic combined with choline (SYNCHs) in the first generation, as well as those treated in the first and each subsequent generation with synbiotic (SYNr) or synbiotic combined with choline (SYNCHr). F1–F3 represent generations. Significant differences compared to C are indicated (*P < 0.05, **P < 0.01).
Morphometric measurements of the germinal center surface area were also conducted in the spleen (data not shown). No statistically significant differences were observed between the control and in ovo-treated groups at any generation or time point.
In the CT of the F1 generation, the administration of bioactive substances had no effect on the number of lymphocytes, except for an increase in the number of CD8+ cells in the SYNCHs group at week 21 after hatching (Fig. 6). However, in the F2 generation, an increase in the number of CD4+ lymphocytes was observed in the SYNCHr group on day 14, along with an increase in the number of Bu-1+ lymphocytes in the SYNr and SYNCHr groups at week 21 after hatching (Fig. 6). These changes were only evident in individuals that received bioactive substances prenatally and whose parental generation (F1) had also received the same substances. In contrast, no significant changes were found in individuals from the SYNs and SYNCHs groups, which were only the offspring of treated birds.
Area occupied by antigen-positive cells (mean ± SD) in the cecal tonsil of chickens from the control group (C) and groups treated in ovo with synbiotic (SYNs) or synbiotic combined with choline (SYNCHs) in the first generation, as well as those treated in the first and each subsequent generation with synbiotic (SYNr) or synbiotic combined with choline (SYNCHr). F1–F3 represent generations. Significant differences compared to C are indicated (*P < 0.05, **P < 0.01).
In the F3 generation, similar to the spleen, no changes in the number of immune cells were observed in the CT in any of the experimental groups compared to the control groups (Fig. 6).
Discussion
In poultry practice, the delivery of bioactive compounds, including synbiotics, on day 12 of embryonic development (E12) is referred to as in ovo stimulation, in contrast to in ovo feeding, which occurs around E17–186,40,41. However, the prebiotic and probiotic components in the synbiotic preparation are not available to the host at the same time. Due to their high solubility in water and relatively small size, prebiotics can penetrate the outer and inner shell membranes, stimulating the development of innate microflora in the developing intestinal tract20. In contrast, probiotics are most likely ingested by the embryo on E19, only after the chick’s beak mechanically breaks the inner shell membrane at the start of hatching20.
In the context of avian immune system development, the administration of bioactive substances on E12 occurred at a time when hematopoietic cell precursors were already present in both the thymus and the bursa of Fabricius. The epithelial thymus rudiment undergoes three waves of colonization by progenitors, each lasting 1–2 days: the first begins on E6, the second on E12, and the third starts around E18 and continues just after hatching42. The first wave derives from para-aortic foci, while the subsequent two waves come from the bone marrow43. In the bursa, the first B-cell precursors, which derive from para-aortic foci, enter the organ between E10 and E1544,45, and by E12, the first IgM+ B cells are already present46,47. In the present research, the influence of synbiotic PS and synbiotic PS combined with choline on the C/M ratio in the thymus and bursa of Fabricius was studied. In ovo synbiotic administration led to an increase in the C/M ratio in the thymus of F1 chickens on day 14. On the other hand, the effect of synbiotic administration was not observed in the bursa. In both analyzed organs, no significant differences in the C/M ratio were observed in subsequent generations, suggesting the loss of the phenotype observed in F1. Moreover, comparisons of the C/M ratio in the control groups across the three consecutive generations showed a significant increase in this ratio in the F2 generation compared to the F1 generation (Fig. 3). It should be noted that our experiment involved three consecutive bird generations, reflecting the natural changes that occur between generations raised in different seasons of the year. It is standard practice in poultry breeding for subsequent generations of birds to be raised in different seasons. The seasonal effect cannot be completely excluded in poultry breeding, particularly regarding semi-intensive management systems and more primitive, extensive poultry breeds. It is also worth noting that while a natural seasonal effect on the C/M ratio was observed in the control groups of our study, the administered bioactive compounds did not influence this phenotype within each separate generation. The C/M ratio in both organs followed the same trend, either increasing or decreasing within a given generation. Therefore, it can be assumed that different environmental factors affecting the birds’ immune system in each generation led to changes in the C/M ratio in both examined organs. The increase in this ratio in the thymus may result from the increased proliferation of young thymocytes in the cortex of this organ. In contrast, in the bursa of Fabricius, the cortex of the follicles gradually increases its surface area starting on the first day after hatching. This process depends on contact with environmental antigens in the intestinal lumen48 or even those absorbed directly from the surface of the cloaca (cloacal lips)49,50. One of the most likely environmental factors contributing to the significant changes in the central immune organs was the different seasons of the year, and therefore the composition of environmental antigens when the material was collected. The mechanisms by which environmental factors caused the observed phenotypic changes is a separate issue. It is possible that at least some of the agents are capable of epigenetically modifying progenitor cells in the central lymphoid organs.
Two different preparations were used in the experiment: one containing only the synbiotic PS (SYN) and the other containing the synbiotic PS supplemented with choline (SYNCH). Dietary choline is a primary methyl group donor that influences the formation of epigenetic marks, such as DNA and histone methylation. Previous studies in mice have indicated that DNA methylation changes in developing embryos can result in clearly visible and stable phenotypes in offspring51. Further studies confirmed that DNA and histone methylases are directly influenced by the availability of diet-derived methyl groups (from choline/betaine, methylfolate, or methionine), as these serve as precursors for the universal methyl donor S-adenosylmethionine52, which is required for the methylation of cytosine in DNA and lysine in histones. In our study, choline supplementation did not enhance the epigenetic effect on the C/M ratio in central lymphoid organs. We assume that the addition of choline to the in ovo injected synbiotic preparation did not provide a sufficiently strong stimulus to induce significant changes in the C/M ratio of the central immune organs.
The impact of bioactive compounds on secondary lymphoid organs was assessed in the spleen and CTs. The spleen is an organ that responds to blood-derived antigens, while the CTs primarily react to gut-derived stimuli. As demonstrated in our previous study, in ovo delivered synbiotics can influence the cellular composition in both of these organs in chickens11,53.
An intergenerational effect was observed in the SYNCHs group in the spleen on D14 (Fig. 4). The decreased number of CD8+ cells present in the F1 generation persisted into the F2 generation. In the F1 generation on D14, a reduced number of CD8+ cells was also noted in the SYNs group, but this effect was not significant in the F2 generation. These results suggests that the addition of choline to the preparation promoted the manifestation of effects of epigenetic modulators in the subsequent generation. However, it should be noted that readministration of the SYNCHs in each subsequent generation (F2 and F3, D14-SYNCHr groups) did not induce the epigenetic effect (e.g., F3-D14-SYNCHr). In the F3 generation, no changes in the number of lymphocytes were found between the groups receiving bioactive substances and the control group. Therefore, it can be concluded that in the context of the examined spleen cells, intergenerational but not transgenerational inheritance was observed.
The effect of in ovo administration of bioactive substances on spleen cell populations was generally not detected 21 weeks after hatching in the F1, F2, and F3 generations, with the only exception being an increase in CD4+ cells in the F2 SYNs group. Therefore, it can be assumed that the effects of epigenetic modulators are more clearly visible in younger individuals, while the cross-talk between the bird’s immune system and other environmental factors, along with the organism’s constant striving to maintain homeostasis, leads to the blurring of the effects at a later age. Moreover, the manifestation of a specific trait resulting from epigenetic mechanisms may be age-dependent. The theory of epigenetic dynamics suggests that epigenetic changes should be considered time-related, with a modified phenotype potentially undergoing “wash-in” and “washout” not only across but also within generations29, as a reaction to current environmental conditions and associated adaptation mechanisms54.
It should also be noted that not all effects of the administered substances observed in the F1 generation were detectable in the F2 generation. Specifically, the significant increase in the CD4+/CD8+ cell ratio noted in the F1 generation on D14 in both the SYNs and SYNCHs groups was not observed in subsequent generations. It is important to note that the threshold for detecting significant changes may influence the study of transgenerational epigenetic inheritance29. Epigenetic effects may gradually fade across successive generations, rather than suddenly disappearing between two generations29. However, a fading phenotype might not be observed in successive generations due to the sensitivity of the detection and statistical methods applied.
The influence of synbiotics on immune-related gene expression in the CT has been previously reported55,56,57. Studies have shown that prebiotics affect the mitochondrial expression of genes associated with respiratory chain proteins and energy metabolism in the intestinal mucosa and CTs of broiler chickens. This suggests that by regulating the microbiota, we can directly influence bacterial metabolites, which, in turn, affect gut epithelial and immune cells, impacting their metabolism, epigenetic modifications, and gene expression58,59.
In the CTs of the present studies, the administered bioactive substances did not significantly affect the populations of immune cells tested on D14 in the F1 generation. However, they significantly increased the number of CD4+ lymphocytes in the F2 generation in the SYNCHr group at the same time point (Fig. 6). Therefore, as seen in the spleen, the addition of choline resulted in more pronounced changes in immune cell populations compared to synbiotics alone. The question arises as to why the effect of synbiotic + choline administration was not visible earlier than in the F2 generation. It can be hypothesized that the injected substances induced subtle changes in the epigenome in the F1 generation, but only the repeated administration in the F2 generation triggered the phenotypic effect. However, this effect was not observed in the F3 generation. Moreover, increases in the number of certain lymphocyte populations at week 21 after hatching, which were not reflected on D14 or in previous generations, were observed.
Numerous studies have demonstrated the intergenerational effects of environmental changes in birds30,3660–65. To our knowledge, only Leroux et al.66 have provided evidence of epigenetic transgenerational inheritance in these species. Previous research has indicated that epigenetic modifications programmed during embryogenesis are largely maintained throughout development and, therefore, are less susceptible to environmental changes later in life33. In the study by Leroux et al.66, the epigenetic factor genistein was administered to fertilized eggs before incubation. It can, therefore, be assumed that the transgenerational effect results not only from the properties of the factor but also from its early impact during embryonic development. Although the in ovo stimulation method used in the present study allowed the administration of bioactive substances on the 12th day of incubation, no transgenerational effects on immune system organs were observed. This may be due to the individual ability of the GP chickens to adapt to environmental conditions. This breed, unlike broilers, is characterized by a naturally higher adaptive capacity of the immune system to environmental stimuli67,68,69, which may also explain the limited intergenerational and transgenerational effects of synbiotic administration. Moreover, GP chickens can be classified as a breed of outbred nature. At first glance, this may be regarded as a drawback due to their higher genetic variability. However, studies in rodent models have shown that inbred lines may be less susceptible to epigenetic transgenerational phenomena70. Therefore, the outbred nature of GP chickens appears to be more advantageous than disadvantageous in the context of intergenerational and transgenerational epigenetic inheritance studies. Indeed, the advantage of outbred lines over inbred ones is that they are genetically more similar to wild species, providing more information about what can be observed in nature30.
It should be noted that the literature on intergenerational and transgenerational epigenetic inheritance primarily focuses on the negative effects of environmental factors on animals. Many recent articles have discussed the impact of maternal stress (such as food deprivation, physical restraint, social isolation, and corticosterone administration) on offspring phenotypes62,71. Some studies also describe the effects of dietary deficiencies72 on subsequent generations. Additionally, it has been shown that parental age influences the lifetime reproductive success of individuals73. Specifically, sparrow females whose mothers were older and males with older fathers had fewer recruits in the following years compared to sparrows with younger parents. The cause of at least some of these effects is suspected to involve epigenetic factors. In contrast, the current research focused on the impact of potentially positive factors, such as synbiotics and choline, on the development and condition of lymphoid organs. Therefore, it may be suggested that the influence of factors shaping the immune system in chickens is not a strong enough stimulus for the organism to demonstrate a clear, intergenerational and transgenerational effect.
It should be pointed out that, according to the theory of “dynamic” phenomena in epigenetics29, epigenetic changes should be considered dynamic across generations. It has been suggested that intergenerational and intragenerational epigenetic phenotypic modifications may change in nonlinear patterns over time29. We believe that we observed the dynamics of epigenetic changes in our current three-generational studies. The dynamic perspective suggests that epigenetic changes should be viewed as gradual, time-related alterations rather than simply “digital” (on–off) changes29. Thus, even if a modified trait is not evident in one generation, it should not be regarded as a discontinued epigenetic alteration between consecutive generations. Time-related, dynamic epigenetic alterations may be subject to “washout” and “wash-in” effects within and across generations, reflecting the organism’s response to environmental conditions. This may be due to unexplored additive effects resulting from environmental stressors that wax, wane, and then wax again across multiple generations29, which we hypothesize may have occurred in our current study.
In conclusion, in ovo administration of synbiotics, either alone or in combination with choline, may induce an intergenerational effect observed in the F2 generation. A transgenerational effect was not clearly observed in any of the organs studied. We hypothesize that the phenotypic modifications observed in immune organs may be of epigenetic origin and could have a dynamic nature due to age and the interaction of the organism with environmental conditions.
Materials and methods
Ethical consideration
The study was conducted in accordance with the guidelines established by the Local Ethical Committee for Animal Experiments in Bydgoszcz, Poland (Approval No. 15/2022, issued on April 20, 2022), as well as Directive 2010/63/EU and Regulation (EU) 2019/1010. The animal experimental protocols were approved by the above-mentioned Ethical Committee. Welfare monitoring was implemented throughout the study. The birds were housed under standard environmental conditions on a poultry farm, with rearing conducted by qualified personnel. A veterinarian at the facility oversaw animal welfare. The research adhered to the ethical principles of the 3Rs (Replacement, Reduction, and Refinement).
Birds
The experiment was conducted on Green-legged Partridgelike (GP) chickens, an old, native, slow-growing breed known for its hardiness and low susceptibility to unfavorable conditions, and well-developed maternal traits. Birds of this breed are well adapted to extensive farming and exhibit strong disease resistance. Moreover, chickens have minimal environmental and nutritional demands. Unlike broiler breeds, there has been no selective breeding within this GP population67,68,69. Previous research suggests GP as an excellent model for academic and scientific research aimed, for example, at understanding the inheritance pattern and interaction pathways of genes underlying admixed phenotypes74. In addition, GP breed is thought to be a remarkable avian model for investigating the genetic background of immune responses, especially taking into consideration the potentially elevated levels of immunity noted in these birds75,76,77, as well as for studying the possibility of immune stimulation in chickens57. These traits make GP chickens an attractive research model for studies related to the immune system. It should be pointed out that both outbred and inbred lines have advantages to develop the epigenetic line. The inbred lines, e.g. broilers, allow for an objective interpretation of the epigenomic analyses due to low genetic variability within the line. On the other hand, outbred lines, e.g. GP, may be more susceptible to epigenetic changes and allow to observe transgenerational effects. Moreover, outbred lines show greater genetic similarity to wild species what makes them valuable model for studying natural complex biological patterns and phenomena30.
Experimental design
The experiment involved three consecutive generations of GP chickens. Three hundred eggs were used per each treatment and control group per each generation. Fertilized eggs obtained from F0 GP hens, were incubated under standard conditions at a commercial hatchery in Wągrowiec, Poland. On day 12 of embryonic development, after candling, bioactive compounds formulations—synbiotic PoultryStar® solUS (Biomin GmbH, Herzogenburg, Austria; ) at a dosage of 2 mg/embryo and its combination with choline (Sigma Alrich, Sain Louis, MA, USA, cat. no. C7527) at a dosage of 0.25 mg/embryo—suspended in physiological saline (0.9% NaCl) were delivered into the air cell of eggs with viable embryos via manual injection. Two experiments—one testing different choline sources and dosages, and another examining various dosage combinations of synbiotic PS and choline—were described in our previous manuscript78.
Three experimental groups were established in F1: (1) the synbiotic group (SYNs) was injected with a single dose of synbiotic PS (2 mg/embryo) suspended in 0.2 mL of physiological saline (0.9% NaCl); (2) the synbiotic + choline group (SYNCHs) was injected with a single dose of synbiotic PS (2 mg/embryo) combined with choline (0.25 mg/embryo) suspended in 0.2 mL of 0.9% NaCl; (3) the control group (C) was injected with 0.2 mL of 0.9% NaCl. After injection, the hole was sealed with nontoxic glue to prevent embryo contamination and moisture loss. Eggs were then incubated until hatching. The same injection protocol, using the same bioactive substances and dosages, was continued for three consecutive generations. In generations F2 and F3, on day 12 of incubation, groups SYNs and SYNCHs were additionally divided into two subgroups: (A) single injections in F1 (SYNs, SYNCHs) with no repeated injections in F2 and F3; (B) repeated injection scheme in each successive generation (SYNr, SYNCHr).
After hatching, all chickens in each generation were reared on the same local poultry farm under semi-intensive conditions in floor pens with bedding made of chopped wheat straw, enriched with perches. Two replicates were established for each group (control and treated) in each generation, with 30 birds per replicate. Indoor environmental conditions were managed according to breed-specific requirements. During colder seasons, the ambient temperature was maintained at 16–18 °C. Lighting conditions combined natural daylight from facility windows with supplemental artificial lighting. During the growth phase, a 12-h light and 12-h dark cycle (12:12) was implemented. As the birds reached reproductive maturity, the photoperiod was progressively increased to 16–17 h of light per day. All birds in each generation received the same commercial diet, free from antibiotics, probiotics, and prebiotics, purchased from a feed company (Golpasz, De Heus, Golub-Dobrzyń, Poland), with constant access to fresh water. Laying hens were provided with feed prepared directly on the farm, consisting of 75% winter wheat and 25% concentrate for laying hens from De Heus Polska (Manufacturer’s code: 1957-HD660 × 00 S-W00). The study is reported in accordance with ARRIVE guidelines.
Sample collection and processing
From each generation (F1–F3), on the 14th day (D14) and the 21st week (W21) after hatching, eight randomly selected chickens from each group were subjected to necropsy. Tissue samples from the thymus, bursa of Fabricius, spleen, and cecal tonsil (CT) were collected. The tissues were fixed in 4% neutral-buffered formaldehyde, embedded in paraffin using standard procedures, and sectioned into 5 μm thick slices. Additionally, spleen and CT samples were fixed in 4% phosphate-buffered paraformaldehyde (pH 7.4) for 1 h, washed in 0.1 M phosphate buffer, and infiltrated with buffered 30% sucrose. The samples were then frozen using a cryostat (CryoStar NX50, Epredia, Kalamazoo, MI, USA) and sectioned into 10 μm serial slices. The sections were air-dried overnight and stored frozen until further immunohistochemical staining.
H&E staining
Paraffin-embedded sections of each tissue were stained with Mayer’s hematoxylin (Roth GmbH, Karlsruhe, Germany) and eosin (Poch S.A., Gliwice, Poland) using the H&E staining method. The stained sections were then examined and photographed under a Nikon Eclipse Ni light microscope (Nikon, Melville, NY, USA) equipped with a video camera.
Immunohistochemical staining
In the cryosections, endogenous peroxidase activity was initially quenched using a 3% hydrogen peroxide solution. To prevent nonspecific binding, the sections were then preincubated with Antibody Diluent with Background Reducing Component (Agilent, Santa Clara, CA, USA) for 20 min. Monoclonal mouse antibodies (Southern Biotech, Birmingham, AL, USA) specific to chicken antigens—Bu-1 (also known as the chicken B cell marker chB6) (clone AV20, 1:500), CD4 (clone CT-4, 1:200), and CD8α (clone CT-8, 1:200)—were applied to the serial sections, with PBS serving as a control. The slides were incubated for 1 h at room temperature. Antibody visualization was performed using the EnVision FLEX+ (Dako/Agilent) detection system with 3,3ʹ-diaminobenzidine (DAB) chromogen, following the manufacturer’s instructions. Finally, the sections were dehydrated through a graded series of alcohols and embedded in Euparal (Roth).
Morphometry
The cortex-to-medulla (C/M) diameter ratio was measured in three lobules from each of the eight thymus samples. In the bursa of Fabricius, the cortex-to-medulla area ratio was measured in transverse sections of three individual follicles from each of the eight samples. These measurements were obtained from microphotographs taken at 40× or 100× magnification using NIS-Elements BR 5.41 imaging software (Nikon). The decision to use diameter rather than area to determine the cortex-to-medulla ratio in the thymus was based on previous observations that the medullas of adjacent lobules merge with each other79.
In the spleen and CT, the proportions of Bu-1+, CD4+, and CD8+ cells were estimated on immunohistochemically stained slides. The brown-colored area occupied by antigen-positive cells was measured within a 0.344 mm² area (200× magnification) using the NIS-Elements BR 5.41 program and expressed as a percentage of the field of view. For each individual spleen, the CD4+/CD8+ cell ratio was calculated, followed by the estimation of the group average. Moreover, in the spleen, the area of germinal centers was measured within a 1.376 mm² area (100× magnification) and expressed as a percentage of the field of view79. In the CT, the fields of view were consistently selected, beginning at the lamina propria mucosae and extending toward the organ’s lumen53. In all cases, a histologist ensured the elimination of artifacts.
Statistical analysis
Morphometric measurement data were analyzed using Statistica 13.3 software (StatSoft Polska Sp. z o.o., Kraków, Poland). To assess the significance of differences between groups, one-way ANOVA with posthoc Tukey tests was performed for data that met the criteria of normal distribution and homogeneity of variance. For data that did not meet these assumptions, the Kruskal–Wallis test (one-way ANOVA on ranks) was used. A significance level of P < 0.05 was indicated with a single asterisk, while P < 0.01 was marked with a double asterisk on the graphs.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Tuscher, J. J. & Day, J. J. Multigenerational epigenetic inheritance: one step forward, two generations back. Neurobiol. Dis. 132, 104591 (2019).
Wrenzycki, C. & Niemann, H. Epigenetic reprogramming in early embryonic development: effects of in-vitro production and somatic nuclear transfer. Reprod. Biomed. Online. 7, 649–656 (2003).
Fridmann-Sirkis, Y. et al. Delayed development induced by toxicity to the host can be inherited by a bacterial-dependent, transgenerational effect. Front. Genet. 5, 27 (2014).
Bednarczyk, M. et al. Influence of different prebiotics and mode of their administration on broiler chicken performance. Animal 10, 1271–1279 (2016).
Pedroso, A. A., Batal, A. B. & Lee, M. D. Effect of in Ovo administration of an adult-derived microbiota on establishment of the intestinal Microbiome in chickens. Am. J. Vet. Res. 77, 514–526 (2016).
Siwek, M. et al. Prebiotics and synbiotics - in Ovo delivery for improved lifespan condition in chicken. BMC Vet. Res. 14, 402 (2018).
Villaluenga, C. M., Wardeńska, M., Pilarski, R., Bednarczyk, M. & Gulewicz, K. Utilization of the chicken embryo model for assessment of biological activity of different oligosaccharides. Folia Biol. (Krakow). 52, 135–142 (2004).
Kolodziejski, P. A. et al. GLP1 and GIP are involved in the action of synbiotics in broiler chickens. J. Anim. Sci. Biotechnol. 9, 13 (2018).
Pruszynska-Oszmalek, E. et al. In Ovo injection of prebiotics and synbiotics affects the digestive potency of the pancreas in growing chickens. Poult. Sci. 94, 1909–1916 (2015).
Stadnicka, K. et al. Injection of raffinose family oligosaccharides at 12 days of egg incubation modulates the gut development and resistance to opportunistic pathogens in broiler chickens. Anim. (Basel). 10, 592 (2020).
Madej, J. P. & Bednarczyk, M. Effect of in ovo-delivered prebiotics and synbiotics on the morphology and specific immune cell composition in the gut-associated lymphoid tissue. Poult. Sci. 95, 19–29 (2016).
Stefaniak, T. et al. Impact of prebiotics and synbiotics administered in Ovo on the immune response against experimental antigens in chicken broilers. Animals 10, 643 (2020).
Stefaniak, T. et al. Selected prebiotics and synbiotics administered in Ovo can modify innate immunity in chicken broilers. BMC Vet. Res. 15, 105 (2019).
Sobolewska, A. et al. The influence of in Ovo injection with the prebiotic DiNovo® on the development of histomorphological parameters of the duodenum, body mass and productivity in large-scale poultry production conditions. J. Anim. Sci. Biotechnol. 8, 45 (2017).
Tavaniello, S. et al. Effect of galactooligosaccharides delivered in Ovo on meat quality traits of broiler chickens exposed to heat stress. Poult. Sci. 99, 612–619 (2020).
Dunislawska, A. et al. Metabolic gene expression in the muscle and blood parameters of broiler chickens stimulated in Ovo with synbiotics. Anim. (Basel). 10, 687 (2020).
Dunislawska, A., Slawinska, A., Bednarczyk, M. & Siwek, M. Transcriptome modulation by in Ovo delivered Lactobacillus synbiotics in a range of chicken tissues. Gene 698, 27–33 (2019).
Hu, Y. et al. In Ovo injection of betaine affects hepatic cholesterol metabolism through epigenetic gene regulation in newly hatched chicks. PLoS One. 10, e0122643 (2015).
Płowiec, A., Sławińska, A., Siwek, M. Z. & Bednarczyk, M. F. Effect of in Ovo administration of inulin and Lactococcus lactis on immune-related gene expression in broiler chickens. Am. J. Vet. Res. 76, 975–982 (2015).
Slawinska, A., Plowiec, A., Siwek, M., Jaroszewski, M. & Bednarczyk, M. Long-term transcriptomic effects of prebiotics and synbiotics delivered in Ovo in broiler chickens. PLoS One. 11, e0168899 (2016).
Chen, Q. et al. Involvement of MicroRNAs in probiotics-induced reduction of the cecal inflammation by Salmonella typhimurium. Front. Immunol. 8, 704 (2017).
Rodríguez-Nogales, A. et al. Intestinal anti-inflammatory effect of the probiotic Saccharomyces boulardii in DSS-induced colitis in mice: impact on MicroRNAs expression and gut microbiota composition. J. Nutr. Biochem. 61, 129–139 (2018).
Sikorska, M., Siwek, M., Slawinska, A. & Dunislawska, A. MiRNA profiling in the chicken liver under the influence of early microbiota stimulation with probiotic, prebiotic, and synbiotic. Genes (Basel). 12, 685 (2021).
Teng, Y. et al. Plant-derived Exosomal MicroRNAs shape the gut microbiota. Cell. Host Microbe. 24, 637–652e8 (2018).
Dunislawska, A., Slawinska, A., Siwek, M. & Bednarczyk, M. Epigenetic changes in poultry due to reprogramming of the gut microbiota. Anim. Front. 11, 74–82 (2021).
Dunislawska, A., Slawinska, A., Gryzinska, M. & Siwek, M. Interaction between early in Ovo stimulation of the gut microbiota and chicken host - splenic changes in gene expression and methylation. J. Anim. Sci. Biotechnol. 12, 73 (2021).
Dunislawska, A., Slawinska, A. & Siwek, M. Hepatic DNA methylation in response to early stimulation of microbiota with Lactobacillus synbiotics in broiler chickens. Genes (Basel). 11, 579 (2020).
Dunislawska, A., Gryzinska, M. & Siwek, M. Changes in the gene expression and methylation in chicken cecal tonsils after in Ovo administration of bioactive substances. Sci. Rep. 13, 19840 (2023).
Burggren, W. W. Dynamics of epigenetic phenomena: intergenerational and intragenerational phenotype ‘washout’. J. Exp. Biol. 218, 80–87 (2015).
Guerrero-Bosagna, C. et al. Transgenerational epigenetic inheritance in birds. Environ. Epigenet. 4, dvy008 (2018).
Perez, M. F. & Lehner, B. Intergenerational and transgenerational epigenetic inheritance in animals. Nat. Cell. Biol. 21, 143–151 (2019).
Burggren, W. W. & Crews, D. Epigenetics in comparative biology: why we should pay attention. Integr. Comp. Biol. 54, 7–20 (2014).
Skinner, M. K. Role of epigenetics in developmental biology and transgenerational inheritance. Birth Defects Res. C Embryo Today. 93, 51–55 (2011).
Bednarczyk, M., Dunislawska, A., Stadnicka, K. & Grochowska, E. Chicken embryo as a model in epigenetic research. Poult. Sci. 100, 101164 (2021).
Skinner, M. K. What is an epigenetic transgenerational phenotype? F3 or F2. Reprod. Toxicol. 25, 2–6 (2008).
Frésard, L. et al. Epigenetics and phenotypic variability: some interesting insights from birds. Genet. Sel. Evol. 45, 16 (2013).
Lees-Murdock, D. J. & Walsh, C. P. DNA methylation reprogramming in the germ line. Epigenetics 3, 5–13 (2008).
Reik, W., Dean, W. & Walter, J. Epigenetic reprogramming in mammalian development. Science 293, 1089–1093 (2001).
Feil, R. & Fraga, M. F. Epigenetics and the environment: emerging patterns and implications. Nat. Rev. Genet. 13, 97–109 (2012).
Gulewicz, K. & Bednarczyk, M. Sposób Stymulacji Korzystnego Profilu Bakteryjnego Wylężonych Piskląt (Polish Nb PL197726, 2008).
Uni, Z. & Farket, P. R. Enhancement of development of oviparous species by in ovo feeding. U.S. Patent No. 6,592,878. Washington, DC: U.S. Patent and Trademark Office. (2003).
Coltey, M. et al. Analysis of the first two waves of thymus homing stem cells and their T cell progeny in chick-quail chimeras. J. Exp. Med. 170, 543–557 (1989).
Yvernogeau, L., Nagy, N., Dunon, D., Robin, C. & Jaffredo, T. Development of the avian hematopoietic and immune systems. in Avian Immunology (eds Kaspers, B., Chat, K. A., Göbel, T. W. & Vervelde, L.) 45–69 (Academic, London, UK, (2022).
Houssaint, E. Cell lineage segregation during bursa of Fabricius ontogeny. J. Immunol. 138, 3626–3634 (1987).
Nagy, N., Magyar, A., Tóth, M. & Oláh, I. Origin of the bursal secretory dendritic cell. Anat. Embryol. (Berl). 208, 97–107 (2004).
McCormack, W. T., Tjoelker, L. W. & Thompson, C. B. Avian B-cell development: generation of an Immunoglobulin repertoire by gene conversion. Annu. Rev. Immunol. 9, 219–241 (1991).
Reynaud, C. A., Bertocci, B., Dahan, A. & Weill, J. C. Formation of the chicken B-cell repertoire: ontogenesis, regulation of Ig gene rearrangement, and diversification by gene conversion. Adv. Immunol. 57, 353–378 (1994).
Ratcliffe, M. J. H. Antibodies, Immunoglobulin genes and the bursa of Fabricius in chicken B cell development. Dev. Comp. Immunol. 30, 101–118 (2006).
Ekino, S. et al. Role of environmental antigen in the development of IgG + cells in the bursa of Fabricius. J. Immunol. 155, 4551–4558 (1995).
Ekino, S. et al. Suppression of immune response by isolation of the bursa of Fabricius from environmental stimuli. Aust J. Exp. Biol. Med. Sci. 58, 289–296 (1980).
Wolff, G. L., Kodell, R. L., Moore, S. R. & Cooney, C. A. Maternal epigenetics and Methyl supplements affect agouti gene expression in avy/a mice. FASEB J. 12, 949–957 (1998).
Zeisel, S. & Choline Other Methyl-Donors Epigenetics Nutrients 9, 445 (2017).
Madej, J. P. et al. Genotype-dependent development of cellular and humoral immunity in the spleen and cecal tonsils of chickens stimulated in Ovo with bioactive compounds. Poult. Sci. 99, 4343–4350 (2020).
Hanson, H. E. et al. Epigenetic potential affects immune gene expression in house sparrows. J. Exp. Biol. 224, jeb238451 (2021).
Bełdowska, A. et al. Impact of in Ovo administration of xylo- and mannooligosaccharides on broiler chicken gut health. Poult. Sci. 103, 104261 (2024).
Dunislawska, A. et al. Synbiotics for broiler chickens-In vitro design and evaluation of the influence on host and selected microbiota populations following in Ovo delivery. PLoS One. 12, e0168587 (2017).
Sławinska, A., Siwek, M. Z. & Bednarczyk, M. F. Effects of synbiotics injected in Ovo on regulation of immune-related gene expression in adult chickens. Am. J. Vet. Res. 75, 997–1003 (2014).
Jackson, D. N. & Theiss, A. L. Gut bacteria signaling to mitochondria in intestinal inflammation and cancer. Gut Microbes. 11, 285–304 (2020).
Rose, E. C., Odle, J., Blikslager, A. T. & Ziegler, A. L. Probiotics, prebiotics and epithelial tight junctions: a promising approach to modulate intestinal barrier function. Int. J. Mol. Sci. 22, 6729 (2021).
Berghof, T. V. L., Parmentier, H. K. & Lammers, A. Transgenerational epigenetic effects on innate immunity in broilers: an underestimated field to be explored? Poult. Sci. 92, 2904–2913 (2013).
Dixon, L. M., Sparks, N. H. C. & Rutherford, K. M. D. Early experiences matter: a review of the effects of prenatal environment on offspring characteristics in poultry. Poult. Sci. 95, 489–499 (2016).
Ericsson, M. et al. Long-term and transgenerational effects of stress experienced during different life phases in chickens (Gallus gallus). PLoS One. 11, e0153879 (2016).
Feeney, A., Nilsson, E. & Skinner, M. K. Epigenetics and transgenerational inheritance in domesticated farm animals. J. Anim. Sci. Biotechnol. 5, 48 (2014).
Jensen, P. Adding ‘epi-’ to behaviour genetics: implications for animal domestication. J. Exp. Biol. 218, 32–40 (2015).
Jensen, P. Transgenerational epigenetic effects on animal behaviour. Prog Biophys. Mol. Biol. 113, 447–454 (2013).
Leroux, S. et al. Embryonic environment and transgenerational effects in quail. Genet. Sel. Evol. 49, 14 (2017).
Calik, J. A profile of the laying hens covered by the gene-pool protection program in Poland. Zesz Nauk. PTIE I PTG Oddz W Rzeszowie. 11, 21–26 (2009). (In Polish).
Calik, J. & Krawczyk, J. The role and importance of local laying Hen breeds and basic breed protection program requirements. Wiad Zootech. XLIV, 28–33 (2006). (In Polish).
Kasperek, K., Drabik, K., Zięba, G. & Batkowska, J. The quality of eggs derived from Polbar and Greenleg Partridge hens - Polish Conservative breeds. Acta Sci. Pol. Zootechnica. 22, 53–62 (2023).
Guerrero-Bosagna, C. et al. Epigenetic transgenerational inheritance of Vinclozolin induced mouse adult onset disease and associated sperm epigenome biomarkers. Reprod. Toxicol. 34, 694–707 (2012).
Khan, N., Peters, R. A., Richardson, E. & Robert, K. A. Maternal corticosterone exposure has transgenerational effects on grand-offspring. Biol. Lett. 12, 20160627 (2016).
Brun, J. M. et al. Influence of grand-mother diet on offspring performances through the male line in muscovy Duck. BMC Genet. 16, 145 (2015).
Schroeder, J., Nakagawa, S., Rees, M., -E. Mannarelli, M. & Burke, T. Reduced fitness in progeny from old parents in a natural population. Proc. Natl. Acad. Sci. U S A. 112, 4021–4025 (2015).
Siwek, M. et al. Insights into the genetic history of Green-legged Partridgelike fowl: MtDNA and genome-wide SNP analysis. Anim. Genet. 44, 522–532 (2013).
Siwek, M. et al. Identification of the rate of chimerism of different tissues with microsatellite markers in chicken chimeras. Folia Biol. (Krakow). 58, 257–263 (2010).
Slawińska, A. et al. Quantitative trait loci associated with the humoral innate immune response in chickens were confirmed in a cross between Green-Legged Partridgelike and white leghorn. Poult. Sci. 90, 1909–1915 (2011).
Siwek, M., Szyda, J., Sławińska, A. & Bednarczyk, M. Detection of two QTL on chicken chromosome 14 for keyhole lymphet Haemocyanin. J. Appl. Genet. 53, 115–119 (2012).
Ibrahim, M., Bednarczyk, M., Stadnicka, K. & Grochowska, E. Inter- and transgenerational effects of in Ovo stimulation with bioactive compounds on cecal tonsils and cecal mucosa transcriptomes in a chicken model. Int. J. Mol. Sci. 26, 1174 (2025).
Madej, J. P., Stefaniak, T. & Bednarczyk, M. Effect of in ovo-delivered prebiotics and synbiotics on lymphoid-organs’ morphology in chickens. Poult. Sci. 94, 1209–1219 (2015).
Funding
This research was funded by the National Science Centre, Poland (Project No. 2020/37/B/NZ9/00497).
Author information
Authors and Affiliations
Contributions
J.M.: conceptualization, data curation, methodology, investigation, formal analysis, validation, visualization, writing—original draft; T.S.: data curation, writing—review and editing; K.S.: conceptualization, writing—review and editing; M.B.: supervision, funding acquisition, conceptualization, project administration; E.G.: conceptualization, project administration, writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Madej, J.P., Stefaniak, T., Stadnicka, K. et al. Effects of in ovo stimulation with potential epigenetic modulators on immune system phenotype across three generations in a chicken model. Sci Rep 15, 21989 (2025). https://doi.org/10.1038/s41598-025-06183-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-06183-7