Abstract
Telomere length has emerged as a potential biomarker of cellular aging and has been implicated in various psychiatric disorders, including depression. However, recent studies investigating the association between depression and telomere length have yielded inconsistent findings. The objective of this study is to systematically review the current literature to evaluate the correlation between depression and telomere length, while also examining the influence of potential moderators such as age, gender, assessment techniques, tissue resources, and depression assessment protocols on this association. We systematically included studies measuring telomere length in individuals meeting clinical or rating scale thresholds for Major Depressive Disorder (MDD), employing a thorough search strategy across PubMed, Embase, PsycINFO, and Google Scholar. Using a structured data abstraction form, studies were meticulously assessed for inclusion or exclusion based on predetermined criteria. Analysis involved standardized mean differences within a random effects model framework, allowing for a comprehensive examination of the association between depression and telomere length while accounting for heterogeneity across studies. Following the meticulous screening of titles, abstracts, and full texts, a total of 71 articles meeting our inclusion criteria were identified and included in the meta-analysis. Our analysis revealed a significant association between depression and telomere length, with a Cohen’s d effect size of -0.354 (p-value < 0.0001, I2 = 80%). Subgroup analysis uncovered significant influences of various factors on the relationship between depression and telomere length, including depression assessment tools, measurement scales, telomere measurement techniques, source tissue, and the presence of comorbid medical conditions. Moreover, multivariable meta-regression showed that age, depression measurement technique, and telomere measurement technique significantly impacted this association. Our findings underscore the complexity of the relationship between depression and telomere length and emphasize the importance of considering multiple factors when interpreting study outcomes. Further studies are necessary to elucidate the potential causality underlying this association and to explore the bidirectional connection between depression severity and telomere shortening.
Similar content being viewed by others
Introduction
Depression, a widespread mental disorder affecting a significant segment of the global population, is estimated to impact 5% of adults worldwide1. Major Depressive Disorder (MDD) characterized by its profound effects on various aspects of an individual’s life, including their quality of life, cognitive functions, and behavioral patterns2. The symptoms of MDD are far-reaching, leading to a pervasive sense of despondency and a notable decline in engagement with activities that were once-enjoyed3.
Beyond its psychological manifestations, research has unveiled a compelling association between MDD and inflammation4,5. This link adds another dimension to our understanding of depression, shedding light on the complex relationship between mental and physical health. Furthermore, studies suggest a bidirectional correlation6, emphasizing the complex nature of this relationship. Researchers have described a biological mechanism wherein depression arises through inflammatory dysregulation, contributing to various physical comorbidities7,8,9. Chronic systemic inflammation, which occurs when acute inflammatory mechanisms fail to eliminate tissue injury is a well-established risk factor for many chronic diseases, including cardiovascular diseases, chronic respiratory diseases, neurodegenerative diseases, autoimmune diseases, metabolic diseases, and cancers10. Also, individuals with MDD exhibit higher rates of morbidity and death than the general population11,12,13. In this context, telomere biology, which is affected by the inflammatory environment, is one of the explanations for the extra morbidity and mortality that has attracted a lot of interest.
The genomic stability-preserving telomeres, whose lengths range from a few to 15 kilobases and shrink with each cell division, are composed of tandem TTAGGG repeats14. In most somatic tissues, telomere shortening closely correlated with age and is regulated by genetic and epigenetic control, cellular stress, and inflammation15. This sets the stage for exploring the potential implications of this process in the context of depression, which has been linked to inflammation and its effects on telomere length.
Telomerase is a ribonucleoprotein complex that plays a crucial role in elongating telomeres by providing the template sequence needed for telomeric repeats16. A feedback loop exists between telomere/telomerase dysfunction and inflammatory signaling17. This pro-inflammatory state not only contributes to telomere/telomerase dysfunction but can also accelerate telomere erosion17. Furthermore, telomere/telomerase-related genes can regulate the release of inflammatory cytokines, while inflammation itself can promote the transcription and expression of these genes, thereby reinforcing the cycle of inflammatory signaling18.
By focusing on therapies aimed at mitigating the influence of inflammation on telomere length in individuals with depression, there’s a possibility of uncovering new strategies for enhancing mental health and overall well-being. Therefore, the discussion about telomeres provides a scientific basis for considering inflammation-mediated telomere dynamics as a potential target for therapeutic interventions in depression, thus bridging the gap between basic scientific understanding and potential clinical applications.
Depression is associated with increased level of oxidative stress and a pro-inflammatory environment, both of which contribute to telomere shortening. Elevated levels of inflammatory markers, mainly IL-6, play a crucial role in this process by promoting cell turnover and replicative senescence, inducing oxidative stress, and regulating telomerase. Recent studies showed that chronic exposure to high levels of IL-6 and TNF-α can significantly accelerate of leukocyte telomere erosion19,20,21. Additionally, various inflammatory cytokines and signaling pathways, such as the NF-κB pathway, can affect telomere length and telomerase activity by regulating many components of the telomeres. Many studies have shown that TNF-α secretion can induce telomere shortening by activating transcription factor 7 (ATF7), which is phosphorylated by p38, leading to the release of ATF7 and telomerase from telomeres, ultimately contributing to telomere erosion22.
Additionally, depressed individuals showed an increased sensitivity to the telomere-shortening effects of oxidative and inflammatory stress. This sensitivity may be due to a lack of inflammation regulatory antioxidant and anti-inflammatory responses, such as reduced levels of IL-1023. Therefore, have a limited capacity to recover from damaged caused by oxidative or inflammatory changes24,25,26,27. Therefore, this interplay between oxidative and inflammatory stress in depression may form a vicious cycle, where telomere shortening leads to immunosenescence and impaired immune function, which then triggers further inflammation and oxidative stress.
A meta-analysis conducted by Ridout et al. in 2016 revealed a significant association between depression and telomere length28. Additionally, the analysis emphasized the impact of various factors such as the techniques used for telomere length measurement, the tissue sample source, and the presence of underlying medical conditions on this observed relationship. Additionally, two other meta-analysis conducted during the same period by Yen Lin et al.29, and Schutte and Malouff 201530 provided further insights on the topic concerning the comparison of leukocyte telomere length (LTL) between individuals with depression and control subjects.
In light of recent contributions to the field, including several new published studies, this systematic review aims to evaluate the association between depression and TL. Through a rigorous review of current literature, this review aims to compare individuals with clinically diagnosed or rating scale thresholds for MDD to those not depressed. Furthermore, the review seeks to elucidate the impact of potential confounders, such as age, gender, techniques, tissue resources, and depression assessment protocols on the studied association. Understanding this link could lead to more targeted treatments for depression and improved risk assessment tools. Additionally, it may inform public health initiatives and guide future research directions in both depression management and telomere biology.
Methods
Protocol
A review proposal was registered with the International Prospective Register of Systematic Reviews (PROSPERO, registration number CRD42023424043). This study was designed, conducted, and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement31.
Study eligibility criteria
Concerning inclusion criteria, all the studies:1 Involving humans participants with unipolar depression meeting clinical or rating scale criteria for MDD, and control groups not meeting these thresholds2, published in English and having full texts accessibility4, prospective and retrospective observational studies5, assessing depression based on ICD/DSMV criteria or utilizing standardized questionnaire for measuring depressive symptoms, and6 measuring telomere length using a valid protocol and specific tool, were included. Studies with incomplete information, review articles, and studies on bipolar depression were excluded.
Literature search
A comprehensive electronic search strategy was conducted in May 2023 across several databases, including PubMed, PsycINFO, Embase, and Google Scholar32. No restrictions were imposed on publication dates, ensuring an expansive scope of relevant literature. The search was performed by two independent reviewers (A.I. and K.J.). The search strategy was designed to encompass a range of terms related to Major Depressive Disorder (MDD), such as “depression,” “depressive disorder,” “major depressive disorder,” “emotional disorder,” “melancholia,” and “dysthymia.” The exploration of telomere-related concepts was also incorporated, incorporating terms like “telomeres,” “telomerase,” and “telomere length.” Notably, adaptations were made to the search terms for compatibility with other bibliographic databases, with the complete search strategy provided in the Supplementary material. Additionally, references within relevant articles and review papers in this area were searched to identify citations not indexed in the aforementioned databases. Subsequently, the titles and abstracts of retrieved studies were subject to initial screening by two independent reviewers (A.I. and K.J) to ascertain eligibility for potential inclusion in the meta-analysis. Studies that clearly did not meet the established inclusion criteria were excluded during this phase. In cases where discrepancies emerged regarding eligibility, a consensus was reached through mutual agreement.
Data extraction
Data extraction was carried out, adhering to a predefined structured form, by independent reviewers. The inter-rater reliability was high (> 95%). Data extraction variables included study design, participant sociodemographic characteristics (age, percentage of male subjects, and presence of comorbid chronic medical conditions), depression measurement scale employed, and measures of depression severity, percentage of patients under anti-depressant treatment and mean duration of illness, telomere measurement technique and tissue source, mean (SD) telomere length for depressed and comparison subjects if available, in addition to bibliographic information. In articles where the telomere length was adjusted for other covariates, we reported the adjusted results. The authors of individual studies were contacted for supplementary information when original papers lacked the necessary data for analysis. Simon et al., 200633 furnished telomere length data for control subjects, individuals with mood disorders, and those meeting criteria for MDD. Specifically, telomere length data from MDD subjects (n = 15) and controls (n = 44) were utilized to calculate effect sizes for inclusion in this meta-analysis. Karabatsiakis et al., 201434 organized telomere length findings from the same subjects into various groups based on tissue or cell subpopulations. The outcomes for each group were standardized into mean differences and then aggregated to establish a standardized telomere length for comparison with other studies35. A similar methodology was employed to group white matter oligodendrocytes in the investigation conducted by Szebeni et al., 201436.
Assessment of study quality
To assess the potential bias in the included studies, a comprehensive evaluation was conducted independently by the Newcastle-Ottawa Scale (NOS)37, a well-established tool designed for case-control, cross-sectional, and cohort studies. This scale uses a star system with a maximum of nine stars to systematically evaluate a study across three key domains: the selection of participants, comparability of study groups, and the ascertainment of outcomes of interest. Studies with NOS scores 0–3, 4–6, and 7–9 were considered as low, moderate, and high quality, respectively.
Telomere measurement techniques
Different methods have been used to measure telomere length, including quantitative fluorescence in situ hybridization (qFISH), Southern blot, and quantitative PCR (qPCR). qFISH, it is a technique that utilizes fluorescently labeled peptide nucleic acid (PNA) probes that bind with high affinity to DNA oligonucleotide probes, allowing for high-resolution detection of telomere length at specific chromosome ends Image analysis software then captures and measures the fluorescent intensity provided, which correlates to telomere length38. However, on the other hand, Southern blot technique measures both canonical and non-canonical components of telomeres, measuring the length of terminal restriction fragments (TRF) to present telomere length39,40. In contrast, the qPCR method only measures the canonical component of telomeres, quantifying telomere length (T) relative to a single copy gene signal (S), and expresses it as a T/S ratio for to determine the average telomere length41.
Statistical analysis
Data was converted into standardized mean differences (SMDs) using the effect size calculator42 and reported as Cohen’s d43. The SMD is the mean difference in telomere length between the depressed and non-depressed groups divided by the pooled standard deviation of the distribution of the score used in the study. This results in a unit-less effect size measure that is comparable to other studies using similar measures of outcome. By convention, effect sizes of 0.2, 0.4, and 0.8 are considered small, medium, and large, respectively43 If only correlations (r) or odds ratios (OR) were reported, they were converted to Cohen’s d using the following equations d=\(\:2r/\sqrt{1-r^2}\) or d = log(OR)\(\:\sqrt{3}\:\)/ π35.
All statistical analyses were conducted using the Comprehensive Meta-Analysis Software (Version 2.2.064 Biostat, Englewood, New Jersey). The standard meta-analysis function, employing a random effects model44, was utilized for the analyses. To ascertain the presence of heterogeneity and the potential sources of variability in the effect sizes, several methods were employed. The Q statistics, its associated p-value, and the I2 statistic were examined. The I2 statistic represents the proportion of total variation attributable to heterogeneity rather than random error, with higher values indicating greater heterogeneity. The identification of heterogeneity suggests the presence of systematic differences among the included studies. Given the significant heterogeneity observed during initial analysis using a fixed effects model, a random effects model was employed. To assess the possibility of publication bias, a funnel plot was examined in relation to outcome measures. Furthermore, quantification of publication bias was executed using Egger’s regression intercept45. Meta-regression was used for analyzing the continuous moderators of mean age, gender distribution (percent male gender in a study), mean duration illness, and percentage of anti-depressants used). For categorical moderators, subgroup analyses were conducted using a continuous random effects model. The subgroup analysis was done based on the following:1 the telomere measurement techniques that were used (southern blot, polymerase chain reaction (PCR), or fluorescent in situ hybridization (FISH)…)2, tissue source from where the DNA was extracted3, presence of medical comorbidities (cancer, cardiovascular diseases…), and4 scale that was used to assess depression if they utilized a clinical interview of self-report method for assessment. A multivariate meta-regression was performed using variables found to be statistically significant moderators. In terms of statistical significance, two-sided p-values less than 0.05 were considered indicative of statistical significance.
Results
Study selection
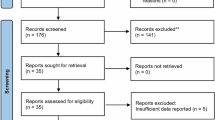
Following a systematic literature search, a total of 828 articles were initially identified. After removing duplicates, 608 articles remained, which were then screened for eligibility by two independent authors. Upon full-text examination of the remaining 120 articles, 48 articles were excluded because 33 articles were not related to our topic, one article assessed the risk of developing depression and anxiety, 10 articles could not obtain their full text or only their abstract was published, 2 articles were review articles, 2 articles were duplicate, and one article is published in French (Fig. 1). Liu et al., 201446 provided information for both depressed and control cohorts, distinguishing between subjects with and without diabetes, treated as distinct datasets in the meta-analysis, labeled as Liu et al. (2014). A similar strategy was utilized in the investigation conducted by Rius-Ottenheim et al., 201247, where data for two distinct groups were presented. Finally, this meta-analysis included 71 original papers33,34,36,]46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113with 2 papers contained two independent data leading to 73 data sets.
Study characteristics
Sociodemographic and clinical characteristics of all data sets are summarized in eTable 2 (supplementary material). We have a total 524,397 number of participants, including (n = 127,832) patients with depression, and (n = 396,565) controls. Among our included articles, 54 articles had a high quality with a very low risk of bias, and 18 articles had a moderate quality based on the score of New-castle Ottawa scale. Summary of quality assessment of risk of bias among selection, comparability and exposure domain in each article are summarized in etable 3 (supplementary material).
Overall meta-analysis
Figure 2 showed that the random effects model revealed an association between depression and TL, showing a significant decrease in TL in patients with depression with an SMD of -0.362 (95% CI − 0.438 to − 0.286, p < 0.0001). Sensitivity analyses revealed that no one study affected the overall significance of the results. After the inspection of the funnel plot (supplementary material efigure1) showed asymmetry in the combined effect size. Egger’s regression intercept was − 2.86 (95% CI -4.97 to -0.757, t = 2.708, two-tailed p = 0.008), suggesting the presence of publication bias. Also, a significant Heterogeneity was detected in the primary meta-analysis (q = 5799.534, tau2 = 0.088, I2 = 88%). Therefore, the next analysis was done to examine whether the heterogeneity resulted from age, gender distribution, duration of illness, or techniques that were used to measure the telomere length.
Results of a meta-analysis of all included studies showing the standardized mean difference effect sizes reporting Cohen’s d (x-axis) used to compare telomere length and depression using the random effects model. The square represents the weighted effect size and the lines the 95% confidence intervals. The triangle indicates overall effect size and 95%CI.
Sub-group analysis of the association between telomere length and depression
Concerning depression measurement, studies that utilized self-report technique as a measure of depression showed a larger significant effect of depression on telomere length (d = -0.496, 95% CI- 0.736 to -0.255, p < 0.001) than studies measuring depression using interview-based technique (d = -0.234, 95% CI -0.324 to -0.145, p < 0.001), with a significant difference between these groups (p < 0.0001), as showed Table 1. The assessment of depression was done using different tools in each technique. In studies employing interview-based methods, those using the Structured Clinical Interview of DSM-IV (SCID) to assess depression showed a significantly larger effect of depression on telomere length (d=-0.782, 95% CI -1.163 to -0.4, p < 0.001) compared to other methods utilized. However, concerning self-reports instruments; none of the scales that were used indicated any significant effect (Table 1).
The majority of Studies used qPCR technique to measure telomere length, and both southern blot and qPCR techniques showed a significant effect of depression on telomere length, with a larger effect for southern blot technique (qPCR d=-0.290, 95% CI -0.363 to -0.218, p < 0.001 vs. Southern blot d=-1.131, 95% CI --2.249 to -0.013, p = 0.047). Conversely, FISH did not show a significant decrease in telomere length associated with depression (Table 1). Leukocytes were the main major source that was used, showed the most significant association between depression on telomere length (d = -0.418, 95% CI -0.523 to -0.314, p < 0.0001 (Table 1). Three studies used the brain as a tissue source and also showed a significant effect of depression telomere length of brain cells (d=-0.384, 95% CI -0.686 to -0.082, p = 0.013), however, saliva cells had a small non-significant effect size (d=-0.056, 95% CI -0.218 to 0.105, p = 0.492).
Regarding comorbid medical conditions, the difference between groups was statistically significant (p < 0.001). Groups with medical comorbidities showed a higher effect of depression on telomere length (d=-0.403, 95% CI -0.746 to -0.061, p < 0.001) compared to groups without any medical comorbidity (d=-0.324, 95% CI -0.403 to -0.244, p < 0.001). Notably, among groups with medical comorbidities, only patient with cancer showed a significant effect size (d=-0.614, 05% CI -1.101 to -0.126, p-0.014). Considering the potential influence of medical and psychiatric disorders, as well as various depression assessment methods, on telomere length physiology, we conducted a subgroup analysis to compare telomere length (TL) between patients diagnosed with MDD according to DSM (Diagnostic and Statistical Manual of Mental Disorders) criteria and without any medical comorbidity, against healthy controls. This analysis drew data from 23 studies employing a case-control design33,36,48,49,54,56,58,62,63,65,79,80,84,85,91,94,95,98,99,101,102,107,109. The analysis revealed a significant decrease in telomere length in MDD patients (d= -0.486, 95% CI = -0.685 to -0.288, p < 0.001) (Fig. 3). However, significant heterogeneity persisted among the studies (I2 = 94%). Sensitivity analysis indicated that the significant difference in telomere length was not influenced by any single study.
Meta-regression analysis of the association between telomere length and depression
Table 2 showed a highly significant linear relationship between the mean age of study participants and the effect size (B = 0.0101, 95% CI 0.0006 to 0.00156, p = 0.037). However, gender didn’t significantly affect the effect size (B = 0.0016, 95% CI -0.0012; 0.0044, p = 0.2602), nor did the duration of illness (B = 0.027, 95% CI -0.057 to 0.0112, p = 0.522), or the percentage of antidepressant use (B=-0.0012, 95% CI -0.0013 to 0.0105, p = 0.836).
Multivariable meta-regression analysis of the association between telomere length and depression
All the moderators that were significant in the sub-groups and meta-regression analysis were included in the multivariable meta-regression. Table 3 showed that age, telomere measurement technique as qPCR, and depression measurement technique remained significant, but source tissue and presence of comorbidities were no longer significant. A test of the model suggests that the overall effect size is related to the included variables (Q = 25.05, p < 0.001) and that the proportion of between-study variance explained by the model was low (R2 = 0.00).
Discussion
The objective of this study was to systematically review the current literature to evaluate the correlation between depression and telomere length, while also examining the influence of potential confounders such as age, gender, assessment techniques, tissue resources, and depression assessment protocols on this association. Our findings revealed a significant association between depression and telomere length shortening. Subgroup analysis unveiled significant influences of various factors on this relationship, including depression assessment tools, measurement scales, telomere measurement techniques, tissue sources, and comorbid medical conditions. Further analysis through multivariable meta-regression highlighted the significant impact of age, depression measurement technique, and telomere measurement technique on this association. Our findings emphasize the correlation between depression and telomere dynamics and highlight the importance of considering multiple factors in understanding this relationship.
Our study identified a significant relationship between depression and the reduction of telomere length, characterized by a moderate effect size (Cohen’s d = -0.354, p < 0.0001). This finding is consistent with the main results of previously published meta-analyses, although there are variations in effect sizes. Specifically, our effect size was slightly larger than that reported by Ridout et al. in 2016 (Cohen’s d = -0.34, p < 0.0001) and Schutte and Malouff in 2015 (r = -0.14, p < 0.0001), both of which shared similar inclusion criteria with our study28,30. However, Yen Lin et al. in 2010 reported a higher Hedges’ g value (g = -0.42, p < 0.0001), yet their analysis only included case-control studies29. Despite these differences, our findings add to the growing body of evidence supporting the association between depression and TL, underscoring the relevance of this relationship across diverse study populations and methodologies.
MDD has often been described as a syndrome of “premature aging”114. Although the exact causality of this connection remains unclear, numerous proposed mechanisms aim to elucidate this connection some have demonstrated decreased levels of telomerase and reduced expression of telomerase reverse transcriptase (Tert) in depression models, causing telomere shortening115,116. Others, meta-analyses, have revealed a correlation between depression and an inflammatory profile, including elevated levels of pro-inflammatory markers such as c-reactive protein and Interleukin 6117. Furthermore inflammation, associated with heightened immune cell replication and increased production of free radicals and oxidative stress, presents another potential mechanism inversely correlated with telomerase activity and TL due to inefficient repair mechanisms during oxidative stress26,118. Other potential mechanism implicated in accelerating telomere shortening involves inflammatory and stress responses induced by dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis and altered neuroendocrine stress response119,120, compounded by the release of pro-inflammatory cytokines, can both elevate cortisol levels. Elevated cortisol, acting via glucocorticoid receptors, is associated with decreased telomerase activity and shortened TL121,122. Finally, mitochondrial alterations leading to reduced ATP production, release of mitochondrial DNA and decreased activity of enzymes involved in the respiratory chain is commonly observed in psychiatric disorders in particular depression123, . Although the precise mechanisms is still unknown, a significant positive association has been established between LTL and mitochondrial DNA copy number in young and middle-aged adults124.
Subgroup analysis was conducted to investigate potential sources of heterogeneity within the study. Various factors were examined as potential moderators, including the measurement techniques for depression, TL measurement techniques and tissue sources, patient characteristics such as age, gender, and comorbid chronic medical conditions, as well as the study designs of the included articles.
Regarding depression measurement, both interview and self-report techniques were employed to assess depression. Our findings, supported by subgroup analyses and multivariable meta-regression, underscore the significant role of depression measurement as a moderator in the relationship between depression and telomere length. Our results indicate that both self-report and interview methodologies significantly influence the association between depression and telomere shortening. Particularly noteworthy is the amplified effect observed in studies employing self-report measures, suggesting a heightened impact compared to interview-based assessments. While previous research, exemplified by Ridout et al. (2016), has emphasized the superiority of interview-based techniques in revealing a stronger association between depression and telomere length (TL)28, the findings by Schutte and Malouff (2015) did not observe any significant impact of depression measurement techniques on telomere shortening30. However, our study challenges this notion. The observed disparity in results can be attributed to several factors. Potential over-reporting of depression symptoms in self-report assessments may stem from individuals’ reluctance to acknowledge or discuss sensitive topics due to societal stigma or denial. Moreover, state-dependent memory and memory deficits could lead to an over-expression of symptoms, further complicating self-reported measures125. Additionally, individuals experiencing severe depression may struggle to accurately assess and communicate the severity of their condition, particularly when relying solely on self-report measures111,112. It is worth noting that our results for the study employing interview technique revealed a Hedges’ g of -0.33, p = 0.007, surpassing the figure reported by Yen Lin et al. (2016), highlighting the robustness of our findings. Our findings shed light on the nuances of depression assessment within the context of telomere length (TL) association. Particularly noteworthy is the significant impact observed within the interview category, where the SCID emerged as the most influential. Furthermore, our analysis comparing TL among patients diagnosed with MDD through DSM criteria-based interviews revealed a notably greater effect size. These findings underscore the importance of considering specific diagnostic criteria and assessment methodologies when evaluating the relationship between MDD and TL. Moving forward, it is imperative for future research to prioritize the use of gold standard interview-based techniques, particularly SCID, in assessing depression109. By standardizing assessment methodologies and emphasizing the utilization of validated diagnostic tools such as SCID, researchers can enhance the reliability and comparability of findings across studies. This approach not only ensures consistency in depression assessment but also facilitates a deeper understanding of the association between depression and TL dynamics.
Multiple techniques are available for measuring TL, each with its strengths and limitations. In our analysis, we found that both Southern blot and qPCR demonstrated a significant association between depression and shortened TL, with Southern blot showing a higher impact with studies using this technique. This contrasts with a previous meta-analysis that suggested FISH and Southern blotting had a greater impact compared to qPCR28,29,30. This discrepancy can be attributed to the widespread use of qPCR-based methods, which are cost-effective and less time-consuming in the majority of included studies. Southern blot analysis and the FISH method have specific limitations that make them less suitable for epidemiological and population studies. For instance, the FISH technique requires fresh blood and is not suitable for solid tissues or preserved samples, making it more costly and time-consuming126. While the qPCR technique focuses solely on evaluating the canonical aspect of telomeres to measure TL signals (T), It is then standardized in relation to a single-copy gene signal (S) within the same sample, resulting in a T/S ratio that provides an indication of the average TL per genome127. Southern blotting measures both the canonical (strictly TTAGGG repeats) and non-canonical components of telomeres to determine the length of terminal restriction fragments (TRF) and present telomere length39. Previous papers have shown that both Southern blot and qPCR demonstrate good reliability and high reproducible results in measuring telomere length39. However, southern blot showed less measurement error compared to qPCR39,128. Therefore, despite being more laborious and time-consuming technically, Southern blot is recommended for future research assessing the association of depression with telomere length due to its higher measurement reliability than qPCR. Also, large-scale epidemiological studies that compare Southern blot analysis to qPCR, and potentially other methods, would greatly enhance our understanding of telomere length physiology.
Leukocyte emerged as the primary tissue source for TL assessment in the majority of the included articles, and our subgroup analysis demonstrated that they had the most robust effect on the association between depression and TL, consistent with findings from previously published meta-analyses28,29. Immune cells, such as leukocytes, hold particular significance in telomere research for several reasons. Firstly, their accessibility and the ease of blood collection make them a convenient tissue source for studying telomere dynamics. Furthermore, immune cells are exposed to both internal and external factors circulating throughout the body, which can profoundly influence telomere physiology129. Moreover, the connection between psychological processes and the neurological and immunological systems is intertwined with immune cells. This interplay underscores the importance of immune cell research in elucidating the biological mechanisms underlying the relationship between depression and TL dynamics. Given the pivotal role of immune cells in telomere research and their relevance to the biological mechanisms underlying depression, future studies should continue to prioritize the investigation of immune cell telomere dynamics. Additionally, expanding research efforts to explore the interactions between psychological factors, immune function, and telomere biology could yield further insights into the complex relationship between depression and TL. This interdisciplinary approach holds promise for advancing our understanding of depression’s etiology and identifying novel therapeutic targets for intervention.
Comorbid conditions, such as diabetes, high blood pressure, and cancer, have been documented to exert a significant impact on telomere length (TL), as evidenced by previous studies130,131. In our subgroup analysis, we observed a significant difference between patients with and without chronic medical conditions, with a larger effect size noted in the group with chronic comorbidities, aligning with the findings of Ridout et al. (2016)28. Particularly noteworthy was our observation that patients with cancer exhibited the strongest association compared to all other conditions. Chronic comorbidities are often associated with a persistent stress environment, triggering the release of glucocorticoids. This hormonal response can lead to increased mitochondrial activity and the production of Reactive Oxygen Species (ROS), ultimately inhibiting telomerase activity and contributing to TL shortening129. Chronic systemic inflammation is a common background to various chronic diseases (diabetes, hypertension, cancer, and depression), all of which were associated with telomere erosion. Persistent inflammation leads to increased oxidative stress and immune system activation, both contribute to telomere shortening and telomerase dysregulation132,133. In diabetes and hypertension, elevated systemic inflammation and oxidative stress accelerate telomere shortening, that also contribute for worsening disease progression and severity10. Similarly, in cancers, particularly those with an inflammatory component or exposure to known carcinogens, the resulting oxidative stress promotes telomere erosion, potentially driving genomic instability and tumor development134. ROS define this link between chronic inflammation and cancer135. Additionally, recent studies indicate that individuals with shorter telomeres have a higher risk of developing carcinomas, suggesting a bidirectional mechanistic link between telomere length and cancer susceptibility136. Moreover, exposure to smoking and inflammation-related factors, associated with cancer, accelerates telomere erosion137. This effect results from the interplay between genetic predisposition and environmental factors, making individuals more prone to rapid telomere degradation. These findings underscore the complex interplay between chronic medical conditions, stress physiology, and telomere dynamics. These insights underscore the importance of holistic approaches to healthcare, emphasizing the management of comorbidities and stress reduction strategies to mitigate the impact on TL health and overall well-being.
Our results revealed a positive impact of increasing age on the association between depression and telomere attrition, a finding that diverges from the conclusions of three previous meta-analyses which failed to detect a significant effect of age28,29,30. This result may be attributed to the inherent cellular process of natural telomere shortening that accompanies advancing age, with an estimated rate of 24.8–27.7 base pairs annually138,139.
Our study did not find a significant effect of gender on the correlation between MDD and TL, consistent with previous meta-analyses that also did not support the presence of this effect28,29. This lack of significance could be attributed to the heterogeneity of our sample, which included both pre- and post-menopausal women, as well as the variability in TL measurement techniques utilized. While some prior investigations have suggested a connection between gender and TL, with females typically exhibiting longer TL than males possibly due to the potential activation of telomerase by estrogen via the promoter region140,141,142, our findings did not align with this hypothesis. It’s worth noting that women beyond their reproductive years may be more susceptible to TL shortening than men, as the protective effect from estrogen diminishes. Moreover, previous meta-analyses have noted variations in the gender effect depending on the measurement methods employed, with Southern blot showing a significant impact while neither real-time PCR nor Flow-FISH did140. This underscores the importance of considering the nuances of TL measurement techniques when investigating the gender effect on TL. Therefore, future prospective studies are essential to comprehensively investigate the pathophysiology of the gender effect on TL among patients with depression. Employing various measurement techniques will be crucial in achieving a more nuanced understanding of the relationship between gender, depression, and TL dynamics.
Our subgroups didn’t show a difference related to the use of antidepressants, a factor that has not been thoroughly investigated in previous meta-analyses28,29,30. However, research on the impact of antidepressants suggests that telomerase activity may be influenced by psychotropic medications, though the precise direction of induced changes and their correlation with clinical efficacy remain subjects requiring further investigation143. Additionally, our analysis did not uncover a significant association between the duration of illness and telomere shortening, consistent with the findings of Yen Lin et al. (2016). The concept of a “dose-response” relationship, where longer exposure to illness leads to accelerated TL shortening, remains suggestive but mixed in the literature. For instance, Shalev et al. (2014) demonstrated a potential dose-response trend, indicating that the duration of “internalizing disorders,” such as MDD, could predict LTL at age 3893. However, other studies have failed to substantiate this “dose-response” hypothesis58,63.Our subgroup analysis revealed that the effect of depression on TL was particularly robust in studies utilizing a case-control design. This observation may be interpreted in the context of the study design itself, as case-control studies often yield computed mean differences in TL between groups, rather than the associations typically observed in cohort and cross-sectional study designs. Furthermore, the matching process inherent in case-control designs contributes to less heterogeneity and imposes stricter inclusion criteria, potentially resulting in clearer and more pronounced effects. However, it’s essential to acknowledge that previous literature has indicated that the inclusion of case-control studies may lead to inflated estimates of the association144,145. Therefore, while case-control studies offer valuable insights into the association between depression and TL, caution should be exercised when interpreting their results and their findings should be corroborated by other study designs for a comprehensive understanding of the relationship.
Our research significantly contributes to the existing body of evidence by incorporating a broader array of articles, resulting in a substantially larger sample size compared to three previously conducted meta-analyses. Furthermore, our study explores uncharted territories by investigating the influence of antidepressant usage, various comorbidities, depression assessment methodologies, and diverse study designs on the relationship between depression and telomere length (TL). These aspects represent novel avenues of exploration that were overlooked in previous analyses, as detailed in eTable 1 of the supplementary materials. However, despite these strengths, our meta-analysis is not without limitations. Firstly, the inclusion of studies with varied research designs, such as cross-sectional, case-control, and prospective studies, restricts our ability to investigate a potential bidirectional relationship between depression and TL comprehensively. Secondly, despite conducting meticulous meta-regression and sensitivity analyses, substantial heterogeneity persisted in several outcomes. Additionally, the limited number of studies within certain moderator categories restricted our ability to effectively detect differences based on these factors. Thirdly, publication bias may be present due to the selective reporting of significant results, potentially skewing the overall findings. Finally, many publications included in our analysis originated from secondary analyses of studies initially designed for different purposes. These studies often utilized stored blood samples to measure TL, introducing variability due to differences in storage conditions or extraction methods. Therefore, it is essential to interpret the outcomes of our meta-analysis with consideration given to these potential sources of variability and bias.
Despite these limitations, our study represents a significant contribution to the field of depression and TL biology, offering novel insights and implications for both research and clinical practice. By incorporating a diverse range of articles and examining various moderators, we deepen our understanding of the complex interplay between depression and TL. Our findings highlight the need to consider individual characteristics, such as age, comorbidities, and depression assessment methodologies, when assessing TL in depressed individuals. This knowledge can inform personalized treatment strategies and aid in the identification of potential biomarkers for psychiatric disorders. Furthermore, our study identifies gaps in previous research and underscores the importance of standardized methodologies in future investigations. Ultimately, by elucidating the biological mechanisms underlying depression and its association with TL, our research contributes to improving outcomes and advancing public health initiatives aimed at addressing depression and its related health consequences.
Conclusion
In conclusion, our study offers valuable insights into the complex relationship between depression and TL, shedding light on previously unexplored avenues and providing implications for both research and clinical practice. Our study highlights the potential utility of TL as a biomarker for psychiatric disorders and emphasizes the importance of personalized treatment strategies tailored to patient-specific factors. Ultimately, by advancing knowledge in this field, our research contributes to improving outcomes and informing public health initiatives aimed at addressing depression and its associated health implications. Our findings underscore the need for future research endeavors aimed at elucidating the bidirectional association between depression and TL.
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
Change history
18 August 2025
A Correction to this paper has been published: https://doi.org/10.1038/s41598-025-15646-w
References
Ferrari, A. J. et al. The epidemiological modelling of major depressive disorder: application for the global burden of disease study 2010. PLOS ONE. 8 (7), e69637 (2013).
Otte, C. et al. Major depressive disorder. Nat. Rev. Dis. Primer. 2, 16065 (2016).
Global Health Data. Exchange | GHDx [Internet]. [cited 2023 Oct 17]. Available from: https://ghdx.healthdata.org/
Colasanto, M., Madigan, S. & Korczak, D. J. Depression and inflammation among children and adolescents: A meta-analysis. J. Affect. Disord. 277, 940–948 (2020).
Howren, M. B., Lamkin, D. M. & Suls, J. Associations of depression with C-reactive protein, IL-1, and IL-6: a meta-analysis. Psychosom. Med. 71 (2), 171–186 (2009).
Beurel, E., Toups, M. & Nemeroff, C. B. The bidirectional relationship of depression and inflammation: double trouble. Neuron 107 (2), 234–256 (2020).
Glassman, A. H. & Shapiro, P. A. Depression and the course of coronary artery disease. Am. J. Psychiatry. 155 (1), 4–11 (1998).
Hare, D. L., Toukhsati, S. R., Johansson, P. & Jaarsma, T. Depression and cardiovascular disease: a clinical review. Eur. Heart J. 35 (21), 1365–1372 (2014).
Musselman, D. L., Evans, D. L. & Nemeroff, C. B. The relationship of depression to cardiovascular disease: epidemiology, biology, and treatment. Arch. Gen. Psychiatry. 55 (7), 580–592 (1998).
Huang, X. et al. The relationship between telomere length and aging-related diseases. Clin. Exp. Med. 25 (1), 72 (2025).
Baker, J., Young, B., Baker, J. & Young, B. W. 20 years later: Deliberate practice and the development of expertise in sport. International Review of Sport & Exercise Psychology, 7(1), 135–157. Int Rev Sport Exerc Psychol. 2014;7:135–57. (2014).
Zivin, K. et al. Early mortality and years of potential life lost among veterans affairs patients with depression. Psychiatr Serv. Wash. DC. 63 (8), 823–826 (2012).
Lou, P. et al. Interaction of depressive and anxiety symptoms on the mortality of patients with COPD: a preliminary study. COPD 11 (4), 444–450 (2014).
Blackburn, E. H., Greider, C. W. & Szostak, J. W. Telomeres and telomerase: the path from maize, Tetrahymena and yeast to human cancer and aging. Nat. Med. 12 (10), 1133–1138 (2006).
Ridout, S. J. et al. Telomeres, Early-Life stress and mental illness. Adv. Psychosom. Med. 34, 92–108 (2015).
Lee, J., Pellegrini, M. V. & Biochemistry Telomere And Telomerase. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 [cited 2025 Mar 26]. Available from: http://www.ncbi.nlm.nih.gov/books/NBK576429/
Jurk, D. et al. Chronic inflammation induces telomere dysfunction and accelerates ageing in mice. Nat. Commun. 5 (1), 4172 (2014).
Ghosh, A. et al. Telomerase directly regulates NF-κB-dependent transcription. Nat. Cell. Biol. 14 (12), 1270–1281 (2012).
Parish, S. T., Wu, J. E. & Effros, R. B. Modulation of T lymphocyte replicative senescence via TNF-{alpha} inhibition: role of caspase-3. J. Immunol. Baltim. Md. 1950. 182 (7), 4237–4243 (2009).
Jaiswal, M., LaRusso, N. F., Burgart, L. J. & Gores, G. J. Inflammatory cytokines induce DNA damage and inhibit DNA repair in cholangiocarcinoma cells by a nitric oxide-dependent mechanism. Cancer Res. 60 (1), 184–190 (2000).
Xu, D. et al. Interferon alpha down-regulates telomerase reverse transcriptase and telomerase activity in human malignant and nonmalignant hematopoietic cells. Blood 96 (13), 4313–4318 (2000).
Liu, S. et al. The regulatory feedback of inflammatory signaling and telomere/telomerase complex dysfunction in chronic inflammatory diseases. Exp. Gerontol. 174, 112132 (2023).
Dhabhar, F. S. et al. Low serum IL-10 concentrations and loss of regulatory association between IL-6 and IL-10 in adults with major depression. J. Psychiatr Res. 43 (11), 962–969 (2009).
Weinstein, A. A. et al. Neurohormonal and inflammatory hyper-responsiveness to acute mental stress in depression. Biol. Psychol. 84 (2), 228–234 (2010).
Sebastián, C. et al. Telomere shortening and oxidative stress in aged macrophages results in impaired STAT5a phosphorylation. J. Immunol. Baltim. Md. 1950. 183 (4), 2356–2364 (2009).
Tsirpanlis, G. et al. Serum oxidized low-density lipoprotein is inversely correlated to telomerase activity in peripheral blood mononuclear cells of haemodialysis patients. Nephrol. Carlton Vic. 11 (6), 506–509 (2006).
Haendeler, J. et al. Antioxidants inhibit nuclear export of telomerase reverse transcriptase and delay replicative senescence of endothelial cells. Circ. Res. 94 (6), 768–775 (2004).
Ridout, K. K., Ridout, S. J., Price, L. H., Sen, S. & Tyrka, A. R. Depression and telomere length: A meta-analysis. J. Affect. Disord. 191, 237–247 (2016).
Lin, P. Y., Huang, Y. C. & Hung, C. F. Shortened telomere length in patients with depression: A meta-analytic study. J. Psychiatr Res. 76, 84–93 (2016).
Schutte, N. S. & Malouff, J. M. The association between depression and leukocyte telomere length: a meta-analysis. Depress. Anxiety. 32 (4), 229–238 (2015).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, n71 (2021).
Bramer, W. M., Rethlefsen, M. L., Kleijnen, J. & Franco, O. H. Optimal database combinations for literature searches in systematic reviews: a prospective exploratory study. Syst. Rev. 6 (1), 245 (2017).
Simon, N. M. et al. Telomere shortening and mood disorders: preliminary support for a chronic stress model of accelerated aging. Biol. Psychiatry. 60 (5), 432–435 (2006).
Karabatsiakis, A., Kolassa, I. T., Kolassa, S., Rudolph, K. L. & Dietrich, D. E. Telomere shortening in leukocyte subpopulations in depression. BMC Psychiatry. 14, 192 (2014).
Borenstein, M., Hedges, L. V., Higgins, J. P. & Rothstein, H. R. Introduction To meta-analysis (Wiley, 2021).
Szebeni, A. et al. Shortened telomere length in white matter oligodendrocytes in major depression: potential role of oxidative stress. Int. J. Neuropsychopharmacol. 17 (10), 1579–1589 (2014).
Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 25 (9), 603–605 (2010).
Joksic, G., Joksic, I., Filipović, J. & Liehr, T. Telomere Length Measurement by FISH. In: Liehr T, editor. Fluorescence In Situ Hybridization (FISH): Application Guide [Internet]. Berlin, Heidelberg: Springer; 2017 [cited 2025 Mar 24]. pp. 147–52. Available from: https://doi.org/10.1007/978-3-662-52959-1_14
Aviv, A. et al. Impartial comparative analysis of measurement of leukocyte telomere length/dna content by Southern blots and qPCR. Nucleic Acids Res. 39 (20), e134 (2011).
Elbers, C. C. et al. Comparison between Southern blots and qPCR analysis of leukocyte telomere length in the health ABC study. J. Gerontol. Biol. Sci. Med. Sci. 69 (5), 527–531 (2014).
Dl, J. L. S, K E, S D. Telomere length measurement by qPCR - Summary of critical factors and recommendations for assay design. Psychoneuroendocrinology [Internet]. 2019 Jan [cited 2025 Mar 24];99. Available from: https://pubmed.ncbi.nlm.nih.gov/30343983/
Effect Size Calculator [Internet]. [cited 2023 Oct 17]. Available from: https://www.campbellcollaboration.org/escalc/html/EffectSizeCalculator-Home.php
Cohen, J. Statistical Power Analysis for the Behavioral Sciences 2nd edn 567 (Routledge, 1988).
DerSimonian, R. & Laird, N. Meta-analysis in clinical trials. Control Clin. Trials. 7 (3), 177–188 (1986).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315 (7109), 629–634 (1997).
Liu, Z., Zhang, J., Yan, J., Wang, Y. & Li, Y. Leucocyte telomere shortening in relation to newly diagnosed type 2 diabetic patients with depression. Oxid. Med. Cell. Longev. 2014, 673959 (2014).
Rius-Ottenheim, N. et al. Telomere length and mental Well-Being in elderly men from the Netherlands and Greece. Behav. Genet. 42 (2), 278–286 (2012).
Birkenæs, V. et al. Telomeres are shorter and associated with number of suicide attempts in affective disorders. J. Affect. Disord. 295, 1032–1039 (2021).
Boeck, C. et al. Targeting the association between telomere length and immuno-cellular bioenergetics in female patients with major depressive disorder. Sci. Rep. 8 (1), 9419 (2018).
Cai, N. et al. Molecular signatures of major depression. Curr. Biol. 25 (9), 1146–1156 (2015).
Carroll, J. E., Olmstead, R., Haque, R. & Irwin, M. R. Accelerated mononuclear cell telomere attrition in breast cancer survivors with depression history: A 2-year longitudinal cohort study. Cancer 128 (16), 3109–3119 (2022).
Chae, D. H. et al. Discrimination, mental health, and leukocyte telomere length among African American men. Psychoneuroendocrinology 63, 10–16 (2016).
Chang, S. C. et al. Prospective association of depression and phobic anxiety with changes in telomere lengths over 11 years. Depress. Anxiety. 35 (5), 431–439 (2018).
Edwards, A. C. et al. CHRONICITY OF DEPRESSION AND MOLECULAR MARKERS IN A LARGE SAMPLE OF HAN CHINESE WOMEN. Depress. Anxiety. 33 (11), 1048–1054 (2016).
Epel, E. S. et al. Wandering Minds and aging cells. Clin. Psychol. Sci. 1 (1), 75–83 (2013).
Fair, B. et al. Telomere length is inversely correlated with urinary stress hormone levels in healthy controls but not in un-medicated depressed individuals-preliminary findings. J. Psychosom. Res. 99, 177–180 (2017).
Falci, C. et al. Immune senescence and cancer in elderly patients: results from an exploratory study. Exp. Gerontol. 48 (12), 1436–1442 (2013).
Garcia-Rizo, C. et al. Abnormal glucose tolerance, white blood cell count, and telomere length in newly diagnosed, antidepressant-naïve patients with depression. Brain Behav. Immun. 28, 49–53 (2013).
Garland, S. N. et al. A nested Case-Controlled comparison of telomere length and psychological functioning in breast Cancer survivors with and without insomnia symptoms. Rejuvenation Res. 17 (5), 453–457 (2014).
Georgin-Lavialle, S. et al. Leukocyte telomere length in mastocytosis: correlations with depression and perceived stress. Brain Behav. Immun. 35, 51–57 (2014).
Gillis, J. C. et al. The relation of telomere length at midlife to subsequent 20-year depression trajectories among women. Depress. Anxiety. 36 (6), 565–575 (2019).
Gotlib, I. H. et al. Telomere length and cortisol reactivity in children of depressed mothers. Mol. Psychiatry. 20 (5), 615–620 (2015).
Hartmann, N., Boehner, M., Groenen, F. & Kalb, R. Telomere length of patients with major depression is shortened but independent from therapy and severity of the disease. Depress. Anxiety. 27 (12), 1111–1116 (2010).
Hassett, A. L. et al. Pain is associated with short leukocyte telomere length in women with fibromyalgia. J. Pain. 13 (10), 959–969 (2012).
Henje Blom, E. et al. Peripheral telomere length and hippocampal volume in adolescents with major depressive disorder. Transl Psychiatry. 5 (11), e676 (2015).
Hoen, P. W. et al. Association between anxiety but not depressive disorders and leukocyte telomere length after 2 years of follow-up in a population-based sample. Psychol. Med. 43 (4), 689–697 (2013).
Hoen, P. W. et al. Depression and leukocyte telomere length in patients with coronary heart disease: data from the heart and soul study. Psychosom. Med. 73 (7), 541–547 (2011).
Humphreys, K. L., Sisk, L. M., Manczak, E. M., Lin, J. & Gotlib, I. H. Depressive symptoms predict change in telomere length and mitochondrial DNA copy number across adolescence. J. Am. Acad. Child. Adolesc. Psychiatry. 59 (12), 1364–1370e2 (2020).
Huzen, J. et al. Telomere length and psychological well-being in patients with chronic heart failure. Age Ageing. 39 (2), 223–227 (2010).
Isehunwa, O. O. et al. Depression, religiosity, and telomere length in the study on stress, spirituality, and health (SSSH). Int. J. Ment Health Addict. 20 (3), 1465–1484 (2022).
Jansen, R. et al. An integrative study of five biological clocks in somatic and mental health. eLife ;10. (2021).
Jordan, C. D. et al. Association of psychosocial factors with leukocyte telomere length among African Americans in the Jackson heart study. Stress Health J. Int. Soc. Investig Stress. 35 (2), 138–145 (2019).
Kazantseva, A. V. et al. Individual differences in relative telomere length in mentally healthy subjects: the effect of TERT gene polymorphism and urban residency. Russ J. Genet. 58 (9), 1135–1144 (2022).
Ladwig, K. H. et al. Posttraumatic stress disorder and not depression is associated with shorter leukocyte telomere length: findings from 3,000 participants in the population-based KORA F4 study. PloS One. 8 (7), e64762 (2013).
Lahav, Y., Avidor, S., Stein, J. Y., Zhou, X. & Solomon, Z. Telomere length and depression among Ex-Prisoners of war: the role of subjective age. J. Gerontol. Ser. B. 75 (1), 21–29 (2018).
Lee, K. A. et al. Telomere length is associated with sleep duration but not sleep quality in adults with human immunodeficiency virus. Sleep 37 (1), 157–166 (2014).
Lin, J. et al. Depressive symptoms and short telomere length are associated with increased mortality in bladder cancer patients. Cancer Epidemiol. Biomark. Prev. Publ Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 24 (2), 336–343 (2015).
Liu, J. J., Wei, Y. B., Forsell, Y. & Lavebratt, C. Stress, depressive status and telomere length: does social interaction and coping strategy play a mediating role? J. Affect. Disord. 222, 138–145 (2017).
Lung, F. W., Chen, N. C. & Shu, B. C. Genetic pathway of major depressive disorder in shortening telomeric length. Psychiatr Genet. 17 (3), 195–199 (2007).
Mendes-Silva, A. P. et al. Telomere shortening in late-life depression: A potential marker of depression severity. Brain Behav. 11 (8), e2255 (2021).
Mutz, J. & Lewis, C. M. Telomere Length Associations With Clinical Diagnosis, Age, and Polygenic Risk Scores for Anxiety Disorder, Depression, and Bipolar Disorder. Biol Psychiatry Glob Open Sci [Internet]. ; (2022). Available from: https://www.sciencedirect.com/science/article/pii/S266717432200101X
Needham, B. L. et al. Depression, anxiety and telomere length in young adults: evidence from the National health and nutrition examination survey. Mol. Psychiatry. 20 (4), 520–528 (2015).
Phillips, A. C. et al. Do symptoms of depression predict telomere length? Evidence from the West of Scotland twenty-07 study. Psychosom. Med. 75 (3), 288–296 (2013).
Pisanu, C. et al. Leukocyte telomere length is reduced in patients with major depressive disorder. Drug Dev. Res. 81 (3), 268–273 (2020).
Pisanu, C. et al. Investigating the role of leukocyte telomere length in Treatment-Resistant depression and in response to electroconvulsive therapy. J. Pers. Med. ;11(11). (2021).
Prabu, P. et al. Altered circulatory levels of miR-128, BDNF, cortisol and shortened telomeres in patients with type 2 diabetes and depression. Acta Diabetol. 57 (7), 799–807 (2020).
Puterman, E. et al. Multisystem resiliency moderates the major depression-telomere length association: findings from the heart and soul study. Brain Behav. Immun. 33, 65–73 (2013).
Ratanatharathorn, A. et al. Posttraumatic stress disorder, depression, and accelerated aging: leukocyte telomere length in the nurses’ health study II. Biol. Psychiatry Glob Open. Sci. 3 (3), 510–518 (2023).
Ryan, K. M. & McLoughlin, D. M. Telomere length in depression and association with therapeutic response to electroconvulsive therapy and cognitive side-effects. Psychol. Med. 50 (12), 2096–2106 (2020).
Savolainen, K. et al. History of mental disorders and leukocyte telomere length in late adulthood: the Helsinki birth cohort study (HBCS). J. Psychiatr Res. 46 (10), 1346–1353 (2012).
Schaakxs, R., Verhoeven, J. E., Oude Voshaar, R. C., Comijs, H. C. & Penninx, B. W. J. H. Leukocyte telomere length and late-life depression. Am. J. Geriatr. Psychiatry Off J. Am. Assoc. Geriatr. Psychiatry. 23 (4), 423–432 (2015).
Shaffer, J. A. et al. Depressive symptoms are not associated with leukocyte telomere length: findings from the Nova Scotia health survey (NSHS95), a population-based study. PloS One. 7 (10), e48318 (2012).
Shalev, I. et al. Internalizing disorders and leukocyte telomere erosion: a prospective study of depression, generalized anxiety disorder and post-traumatic stress disorder. Mol. Psychiatry. 19 (11), 1163–1170 (2014).
Simon, N. M. et al. Telomere length and telomerase in a well-characterized sample of individuals with major depressive disorder compared to controls. Psychoneuroendocrinology 58, 9–22 (2015).
Squassina, A. et al. Telomere attrition and inflammatory load in severe psychiatric disorders and in response to psychotropic medications. Neuropsychopharmacology 45 (13), 2229–2238 (2020).
Starnino, L., Busque, L., Tardif, J. C. & D’Antono, B. Psychological profiles in the prediction of leukocyte telomere length in healthy individuals. PloS One. 11 (10), e0165482 (2016).
Surtees, P. G. et al. Life stress, emotional health, and mean telomere length in the European prospective investigation into Cancer (EPIC)-Norfolk population study. J. Gerontol. Ser. A. 66A (11), 1152–1162 (2011).
Teyssier, J. R., Chauvet-Gelinier, J. C., Ragot, S. & Bonin, B. Up-regulation of leucocytes genes implicated in telomere dysfunction and cellular senescence correlates with depression and anxiety severity scores. PloS One. 7 (11), e49677 (2012).
Teyssier, J. R., Ragot, S., Chauvet-Gélinier, J. C., Trojak, B. & Bonin, B. Expression of oxidative stress-response genes is not activated in the prefrontal cortex of patients with depressive disorder. Psychiatry Res. 186 (2), 244–247 (2011).
Tyrka, A. R. et al. Alterations of mitochondrial DNA copy number and telomere length with early adversity and psychopathology. Biol. Psychiatry. 79 (2), 78–86 (2016).
Vance, M. C. et al. Prospective association between major depressive disorder and leukocyte telomere length over two years. Psychoneuroendocrinology 90, 157–164 (2018).
Verhoeven, J. E. et al. Major depressive disorder and accelerated cellular aging: results from a large psychiatric cohort study. Mol. Psychiatry. 19 (8), 895–901 (2014).
Verhoeven, J. E. et al. Depression, telomeres and mitochondrial DNA: between- and within-person associations from a 10-year longitudinal study. Mol. Psychiatry. 23 (4), 850–857 (2018).
Verhoeven, J. E., van Oppen, P., Révész, D., Wolkowitz, O. M. & Penninx, B. W. J. H. Depressive and anxiety disorders showing robust, but Non-Dynamic, 6-Year longitudinal association with short leukocyte telomere length. Am. J. Psychiatry. 173 (6), 617–624 (2016).
Vyas, C. M. et al. Pilot study of DNA methylation, molecular aging markers and measures of health and well-being in aging. Transl Psychiatry. 9 (1), 118 (2019).
Wei, Y. B. et al. hTERT genetic variation in depression. J. Affect. Disord. 189, 62–69 (2016).
Wikgren, M. et al. Short telomeres in depression and the general population are associated with a hypocortisolemic state. Biol. Psychiatry. 71 (4), 294–300 (2012).
Wium-Andersen, M. K., Ørsted, D. D., Rode, L., Bojesen, S. E. & Nordestgaard, B. G. Telomere length and depression: prospective cohort study and Mendelian randomisation study in 67 306 individuals. Br. J. Psychiatry J. Ment Sci. 210 (1), 31–38 (2017).
Wolkowitz, O. M., Epel, E. S., Reus, V. I. & Mellon, S. H. Depression gets old fast: do stress and depression accelerate cell aging? Depress. Anxiety. 27 (4), 327–338 (2010).
Wolkowitz, O. M. et al. PBMC telomerase activity, but not leukocyte telomere length, correlates with hippocampal volume in major depression. Psychiatry Res. 232 (1), 58–64 (2015).
Yen, Y. C. & Lung, F. W. Older adults with higher income or marriage have longer telomeres. Age Ageing. 42 (2), 234–239 (2012).
Zhang, D., Cheng, L., Craig, D. W., Redman, M. & Liu, C. Cerebellar telomere length and psychiatric disorders. Behav. Genet. 40 (2), 250–254 (2010).
Zhao, Q. et al. Depressive symptoms are associated with leukocyte telomere length in American indians: findings from the strong heart family study. Aging 8 (11), 2961–2970 (2016).
Heuser, I. Depression, endocrinologically a syndrome of premature aging? Maturitas 41 (Suppl 1), S19–23 (2002).
Xie, X. et al. Major depressive disorder mediates accelerated aging in rats subjected to chronic mild stress. Behav. Brain Res. 329, 96–103 (2017).
Wei, Y. B., Backlund, L., Wegener, G., Mathé, A. A. & Lavebratt, C. Telomerase dysregulation in the hippocampus of a rat model of depression: normalization by lithium. Int. J. Neuropsychopharmacol. 18 (7), pyv002 (2015).
Osimo, E. F. et al. Inflammatory markers in depression: A meta-analysis of mean differences and variability in 5,166 patients and 5,083 controls. Brain Behav. Immun. 87, 901–909 (2020).
Houben, J. M. J., Moonen, H. J. J., van Schooten, F. J. & Hageman, G. J. Telomere length assessment: biomarker of chronic oxidative stress? Free Radic Biol. Med. 44 (3), 235–246 (2008).
Penninx, B. W. J. H. et al. Late-Life depressive symptoms are associated with both hyperactivity and hypoactivity of the Hypothalamo-Pituitary-Adrenal Axis. Am. J. Geriatr. Psychiatry. 15 (6), 522–529 (2007).
Vreeburg, S. A. et al. Major depressive disorder and Hypothalamic-Pituitary-Adrenal Axis activity: results from a large cohort study. Arch. Gen. Psychiatry. 66 (6), 617–626 (2009).
Otte, C. et al. A meta-analysis of cortisol response to challenge in human aging: importance of gender. Psychoneuroendocrinology 30 (1), 80–91 (2005).
Tomiyama, A. J. et al. Does cellular aging relate to patterns of allostasis? An e`xamination of basal and stress reactive HPA axis activity and telomere length. Physiol. Behav. 106 (1), 40–45 (2012).
Vaváková, M., Ďuračková, Z. & Trebatická, J. Markers of oxidative stress and neuroprogression in depression disorder. Oxid. Med. Cell. Longev. 2015, 898393 (2015).
Tyrka, A. R. et al. Association of telomere length and mitochondrial DNA copy number in a community sample of healthy adults. Exp. Gerontol. 66, 17–20 (2015).
Stuart, A. L. et al. Comparison of self-report and structured clinical interview in the identification of depression. Compr. Psychiatry. 55 (4), 866–869 (2014).
Lin, J., Smith, D. L., Esteves, K. & Drury, S. Telomere length measurement by qPCR - Summary of critical factors and recommendations for assay design. Psychoneuroendocrinology 99, 271–278 (2019).
Cawthon, R. M. Telomere measurement by quantitative PCR. Nucleic Acids Res. 30 (10), e47 (2002).
Elbers, C. C. et al. Comparison between Southern blots and qPCR analysis of leukocyte telomere length in the health ABC study. J. Gerontol. Ser. A. 69 (5), 527–531 (2014).
Lin, J. & Epel, E. Stress and telomere shortening: insights from cellular mechanisms. Ageing Res. Rev. 73, 101507 (2022).
Rizvi, S., Raza, S. T. & Mahdi, F. Telomere length variations in aging and age-related diseases. Curr. Aging Sci. 7 (3), 161–167 (2014).
Wentzensen, I. M., Mirabello, L., Pfeiffer, R. M. & Savage, S. A. The association of telomere length and cancer: a meta-analysis. Cancer Epidemiol. Biomark. Prev. Publ Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 20 (6), 1238–1250 (2011).
Cildir, G., Pant, H., Lopez, A. F. & Tergaonkar, V. The transcriptional program, functional heterogeneity, and clinical targeting of mast cells. J. Exp. Med. 214 (9), 2491–2506 (2017).
Scrivo, R., Vasile, M., Bartosiewicz, I. & Valesini, G. Inflammation as common soil of the multifactorial diseases. Autoimmun. Rev. 10 (7), 369–374 (2011).
Stindl, R. Tying it all together: telomeres, sexual size dimorphism and the gender gap in life expectancy. Med. Hypotheses. 62 (1), 151–154 (2004).
Ohshima, H. & Bartsch, H. Chronic infections and inflammatory processes as cancer risk factors: possible role of nitric oxide in carcinogenesis. Mutat. Res. 305 (2), 253–264 (1994).
Broberg, K., Björk, J., Paulsson, K., Höglund, M. & Albin, M. Constitutional short telomeres are strong genetic susceptibility markers for bladder cancer. Carcinogenesis 26 (7), 1263–1271 (2005).
Assavanopakun, P., Sapbamrer, R., Kumfu, S., Chattipakorn, N. & Chattipakorn, S. C. Effects of air pollution on telomere length: evidence from in vitro to clinical studies. Environ. Pollut. 312, 120096 (2022).
Valdes, A. M. et al. Obesity, cigarette smoking, and telomere length in women. Lancet Lond. Engl. 366 (9486), 662–664 (2005).
Brouilette, S., Singh, R. K., Thompson, J. R., Goodall, A. H. & Samani, N. J. White cell telomere length and risk of premature myocardial infarction. Arterioscler. Thromb. Vasc Biol. 23 (5), 842–846 (2003).
Gardner, M. et al. Gender and telomere length: systematic review and meta-analysis. Exp. Gerontol. 51, 15–27 (2014).
Needham, B. L. et al. A test of biological and behavioral explanations for gender differences in telomere length: the multi-ethnic study of atherosclerosis. Biodemography Soc. Biol. 60 (2), 156–173 (2014).
Lansdorp, P. M. Sex differences in telomere length, lifespan, and embryonic dyskerin levels. Aging Cell. 21 (5), e13614 (2022).
Monroy-Jaramillo, N., Dyukova, E. & Walss-Bass, C. Telomere length in psychiatric disorders: is it more than an ageing marker? World J. Biol. Psychiatry Off J. World Fed. Soc. Biol. Psychiatry. 19 (sup2), S2–20 (2018).
Lijmer, J. G. et al. Empirical evidence of Design-Related Bias in studies of diagnostic tests. JAMA 282 (11), 1061–1066 (1999).
Parker, L. A., Saez, N. G., Porta, M., Hernández-Aguado, I. & Lumbreras, B. The impact of including different study designs in meta-analyses of diagnostic accuracy studies. Eur. J. Epidemiol. 28 (9), 713–720 (2013).
Author information
Authors and Affiliations
Contributions
A.I.: Conceptualization, screening (titles and abstract) and retrieval of full text, data collection, statistical data analysis (meta-analysis and meta-regression), interpretation of results, writing – Original Draft Preparation, K.J.: screening (titles and abstract) and retrieval of full text, data collection, writing – Original Draft Preparation, J.K.: data collection, writing – Original Draft Preparation, Y.T.: data collection, writing – Original Draft Preparation, N.S.: Data collection, Writing – Original Draft Preparation, L.A.A.: project supervision and review of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: In the original version of this Article the author name Karilina Jaalouk appeared twice in the author list.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ismail, A., Jaalouk, K., Koteish, J. et al. Exploring the association between depression and telomere length: A systematic review and meta-analysis. Sci Rep 15, 22967 (2025). https://doi.org/10.1038/s41598-025-07076-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-07076-5