Abstract
Vulvovaginal candidiasis (VVC) represents a universal health hazard that contributes to significant morbidity in HIV-positive women. Antifungal resistance is a worldwide increasing health problem that reduces treatment options while increasing treatment costs. The purpose of this study was to determine the prevalence of VVC, identify associated risk factors, characterize the distribution of Candida species, and assess their antifungal susceptibility patterns among HIV-positive women attending Mbarara City Health Centre IV. A laboratory-based, cross-sectional study design was conducted on 146 high vaginal swabs collected from HIV- positive women aged 18 years and above attending routine HIV care that presented with signs and symptoms of vulvovaginal candidiasis. These were subjected to microscopy and culture on Sabouraud Dextrose Agar. Candida isolates were identified by gram stain, germ tube, CHROM agar™, and Analytical profile index (API® Candida) tests. Susceptibility to fluconazole, clotrimazole, voriconazole, amphotericin B, miconazole, and nystatin by the disc diffusion method on Mueller Hinton agar supplemented with 2%w/v glucose and 0.5 µg/ml methylene blue dye. Out of the 146 high vaginal swabs collected, 71(49%) were colonized with Candida species with 43(62%) being Candida albicans while 26 (37.6%) were non-Candida albicans. Candida species were susceptible to amphotericin B (68/71, 95.7%) and resistant to fluconazole and clotrimazole (33/71, 45%). Elevated blood glucose levels were significantly associated with vulvovaginal candidiasis (p = 0.018; odds ratio = 2.93). Candida albicans is the leading cause of VVC, with a higher prevalence than non-Candida albicans. It also demonstrates that amphotericin B and nystatin are the most effective antifungal medications. Furthermore, diabetes is associated with VVC compared to other studied factors. We recommend the use of nystatin for the management of vulvovaginal candidiasis among HIV adult women.
Similar content being viewed by others
Introduction
Vulvovaginal candidiasis (VVC) is a symptomatic vaginitis caused by infection with Candida yeast1. It is the most prevalent and common fungal infection affecting women worldwide; it is estimated to afflict approximately 75% of all women at least once in their lifetime2. Nearly 8% of women globally suffer from recurrent VVC3 yet cases of reoccurrences are sporadic and have been reported in HIV-positive women4.
Vulvovaginal candidiasis presentation among women can occur in various forms irrespective of their HIV serostatus2. It usually first presents in a mild form as a thick white discharge, itching, burning, irritation, pain during sex, pain, or discomfort when urinating and can go on to a severe form including redness, swelling, and cracks in the wall of the vagina5. However, HIV-positive women are more susceptible to vulvovaginal candidiasis than HIV-negative women due to immunosuppression associated with HIV infection4.
Recurrent VVC is linked to pain, loss of self-esteem, reduced work performance, discomfort, interference with sexual activity, mental anguish, and significant direct and indirect financial burdens6. About 138 million women worldwide suffer from recurrent vvc each year. The economic impact of decreased productivity due to vvc in high-income nations could reach around US$14.39 billion annually by 2030. The financial effect in low- to middle-income countries is projected to be twice as high as that in high-income countries such as the United States of America3.
The common antifungal regimens employed in the Ugandan setting include the azoles like fluconazole and itraconazole. Although there are many effective antifungal drugs both topical and oral that are used for treating vulvovaginal candidiasis, reduced susceptibility of some vulvovaginal candida isolates to some antifungal agents has been reported in some countries in the world involving yeasts isolated from high vaginal swabs7,8,9.
Understanding the prevalence of vulvovaginal candidiasis, associated factors, and antifungal susceptibility patterns of Candida species in HIV-positive women is key in guiding appropriate antifungal therapy selection.
Methods
Study area
This study employed a cross-sectional design that included HIV-positive women aged 18 years and above who received regular HIV care at the Mbarara City Health Centre IV and consented to participate in the study.
Ethical consideration
Ethical approval was granted by the Research Ethics Committee of Mbarara University of Science and Technology (MUST-2023-1284), and administrative clearance was obtained from the Mbarara City Health Officer. Prior to enrollment, written informed consent was obtained from all participants. Confidentiality and privacy were strictly maintained in accordance with standard clinical protocols. Participants retained the right to withdraw at any time without compromising their medical care. Study supervisors ensured compliance with ethical guidelines, safeguarding patient rights and dignity throughout all procedures.
Sample size
According to Mbarara City Council HCIV’s Open Medical Records System (OpenMRS), which was used to determine the sample size of participants, 10 HIV-positive women on average report VVC signs and symptoms each week (MCC Weekly Reports, 2023). Therefore, the sample size to a finite population using the formula n1 = N × n/ (n + N-1) where N is the finite population and n1 is the corrected sampled size. We adjusted for a 10% attrition rate (131/0.1). Therefore, we enrolled 144 participants in this study.
Data collection
Quantitative data were collected through structured questionnaires and medical record reviews. A systematic questionnaire conducted by an interviewer was utilized to obtain data about the participants’ socio-demographics of participants and the variables associated with vulvovaginal candidiasis.
Sample collection, culture and identification
Following a clinical examination by the midwife, vulvovaginal samples were collected with the patient in lithotomy position. The vulva was swabbed front-to-back using sterile saline-soaked gauze swabs. A sterile cotton swab was then inserted 20–30 mm into the vaginal canal and rotated gently for sample collection. Two vaginal swabs were taken; one for gram staining and the other for culture and sensitivity testing.
The labelled samples were transported to the Microbiology laboratory of Mbarara university of science and technology immediately after collection. In the laboratory, the swabs were inoculated on Sabouraud Dextrose Agar (SDA) at 37 °C for 48 h10. A wet mount preparation using saline and gram staining was performed to examine microscopically for budding yeast cells. Species identification was achieved through germ tube test, using Candida API strips, and subculturing on CHROMagar10,11.
Antifungal susceptibility testing was performed using the disc diffusion method on Mueller Hinton Agar supplemented with 2% glucose and methylene blue12. The antifungal agents tested included Fluconazole (25 µg), Voriconazole (1 µg), Nystatin (100 IU), Clotrimazole (50 µg), Miconazole (10 µg), and Amphotericin B (2.5 µg/mL). Plates were incubated at 37 °C for 24–48 h, allowing detection of slower-growing species like Candida glabrata and Candida krusei10. C. albicans ATCC 90,028, C. parapsilosis ATCC 22,019 and, C. krusei ATCC 6258 were used as control strains.
Every tenth sample was evaluated at the Epi-Centre laboratory Mbarara), a level three authorized laboratory in Mbarara, for external quality control.
Data analysis
Questionnaires were checked for completeness on the same day of the data collection. Data was entered in Excel, cleaned, coded, and backed up. Data were exported to STATA V17 for analysis.
The baseline characteristics of participants were summarized using mean with SD, median with IQR, or proportions as deemed appropriate. Descriptive statistics were used to summarize the demographic, behavioral, and clinical characteristics of the study population. The prevalence of VVC was estimated with confidence intervals. Logistic regression analysis was performed to identify factors associated with VVC adjusting for potential confounders.
Results
Social demographic characteristics of study participants
In this study, 420 HIV positive women at least 18 years old were screened of which 150 were eligible. Of the eligible participants, 146 participated in the study and 4 declined to participate. Growth was observed in 86 participants of which 71 were Candida species and 15 were other organisms and not Candida species therefore excluded from the study (Table 1).
Prevalence of VVC among HIV-positive women at Mbarara city health centre IV
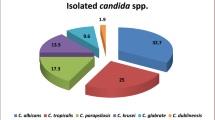
Of the 71 culture-positive cases, Candida species were isolated from 49% of HIV-positive women presenting with vulvovaginal candidiasis symptoms at Mbarara City Council health centre IV ART clinic. Candida albicans accounted for the majority of isolates (60.5%, n = 43), while non-albicans species constituted 39.4% (n = 28) of cases (Table 2, Fig. 1).
Antifungal susceptibility patterns
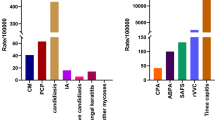
Antifungal susceptibility patterns of the isolated candida species among HIV-positive women at Mbarara City Health Centre IV. Candida species showed the highest susceptibility to polyenes (amphotericin B and nystatin) than to selected azoles as shown in Table 3.
Factors associated with symptomatic VVC among HIV-positive women at Mbarara City health centre IV
The factors that were assessed in this study were age, random blood glucose levels, education level, pregnancy status, TB disease, occupation, and marital status. The blood glucose levels of the participants showed a significant association (p = 0.018 and OR = 2.93) with infection with vulvovaginal candidiasis (see Table 4).
Discussion
Candida species can cause VVC in both HIV-positive and HIV-negative women. However, compared to HIV Negative women, HIV + women (with lower CD4 + T-cell counts) had a higher chance of acquiring VVC13. Effective management of vulvovaginal candidiasis (VVC) in HIV-positive women requires identification of causative Candida species, determination of their antifungal susceptibility patterns, and assessment of risk factors associated with VVC.
The observed 49% prevalence of vulvovaginal candidiasis (VVC) among HIV-positive women in our study aligns closely with the 50% reported by Merenstein et al.,14 in the US suggesting similar ART efficacy and diagnostic approaches between the populations. However, our prevalence was substantially higher than the 20% documented in Namibia15 reflecting differences in ART accessibility, regional Candida species distribution, or climatic conditions favoring fungal growth. Conversely, the markedly lower prevalence compared to Brazil’s 100%16 may indicate divergent study methodologies (e.g., inclusion of asymptomatic cases) or variations in HIV disease progression among cohorts. Notably, reduced VVC risk observed in women on combination ART underscores the critical role of sustained antiretroviral therapy in mitigating opportunistic infections. These disparities highlight the interplay of immunological (e.g., CD4 + counts), therapeutic (e.g., ART adherence), and environmental (e.g., humidity) factors in shaping VVC epidemiology across global settings17,18,19.
Proper identification of Candida species is important to aid in proper management of vaginal candidiasis. C. albicans is still the predominant species and this finding is consistent with many other studies7,13,20,21. However, some studies have reported a high prevalence of non-Candida albicans in vulvovaginal candidiasis22,23. The shifting epidemiology of vulvovaginal candidiasis, characterized by increasing non-albicans Candida (NAC) species prevalence alongside decreasing but still dominant C. albicans cases, likely results from multiple factors such as widespread use of broad-spectrum antibiotics that disrupt vaginal microbiota, increased availability and often indiscriminate use of antifungal agents (both over-the-counter and prescribed), and host immune suppression. While C. albicans remains the most prevalent species23.
In our study, factors such as pregnancy, occupation, education levels, and marital status were not associated with VVC among HIV positive women. however, the diabetic status of HIV-positive women in our study was found to be associated with VVC (p = 0.018) which agrees with the study of Keran et al.,24 who concluded that 8.8% had positive culture of Candida species. Hyperglycemia increases glucose concentration in vaginal secretions and tissues, thereby enhancing yeast adhesion and growth.This is because high blood sugar levels decrease neutrophil migration thereby weakening their chemotactic and phagocytic activity25.
In this study, Candida species showed the highest susceptibility to Amphotericin B except C.guilliermondii which showed intermediate susceptibility. The results are similar to the results of Kan et al.,26 that reported susceptibility of 98% to Amphotericin B by Candida species. Susceptibility of NAC to nystatin and resistance among C.albicans was observed which is in consistence with other studies27,28. This may because these drugs are both fungicidal and fungistatic reducing the likelihood for Candida species to develop resistance. Similar investigations have found similar susceptibility patterns for Amphotericin B, which is less widely used to treat VVC. However, the findings from this present study differ from a study by Khan, 201829 that reported a resistance of 41.6% in Candida albicans.
The high resistance of candida species to azoles in our current study is consistent with the outcomes of clotrimazole and fluconazole susceptibility patterns conducted by Sathi et al.30 and Mukasa et al.20 respectively. This may be attributed to the ‘over the counter’ antifungal vaginal pessaries and creams containing clotrimazole and fluconazole thus there is higher exposure of the organism and likely development of resistance.
Conclusions
This study demonstrates that vulvovaginal candidiasis (VVC) is a prevalent opportunistic infection among HIV-positive women at Mbarara City Health Centre IV, with Candida species identified in 49% of symptomatic cases. Candida albicans was the predominant isolate (60.5%), reinforcing its central role in VVC etiology in this population. Notably, polyenes specifically amphotericin B and nystatin exhibited the highest antifungal activity, suggesting their potential as first-line treatments. Furthermore, elevated blood glucose levels were significantly associated with VVC (p = 0.018), implicating hyperglycemia as a key modifiable risk factor. These findings underscore the need for targeted antifungal stewardship and glycemic control interventions to mitigate the burden of VVC in immunocompromised women.
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data set is not publicly available due to privacy or ethical restrictions.
References
Rosati, D., Bruno, M., Jaeger, M., Ten Oever, J. & Netea, M. G. Recurrent vulvovaginal candidiasis: an immunological perspective. Microorganisms. 8 (2), 144 (2020).
Willems, H. M., Ahmed, S. S., Liu, J., Xu, Z. & Peters, B. M. Vulvovaginal candidiasis: a current Understanding and burning questions. J. Fungi. 6 (1), 27 (2020).
Denning, D. W., Kneale, M., Sobel, J. D. & Rautemaa-Richardson, R. Global burden of recurrent vulvovaginal candidiasis: a systematic review. Lancet. Infect. Dis. 18 (11), e339–e47 (2018).
Lema, V. M. Recurrent vulvo-vaginal candidiasis: diagnostic and management challenges in a developing country context. Obstet. Gynecol. Int. J. 7 (5), 260 (2017).
Farr, A. et al. Guideline: vulvovaginal candidosis (AWMF 015/072, level S2k). Mycoses. 64 (6), 583–602 (2021).
Okonkwo, N. & Umeanaeto, P. Prevalence of vaginal candidiasis among pregnant women in Nnewi town of Anambra state, nigeria: A recent perspective. Theory Appl. Microbiol. Biotechnol. 3, 160–168 (2020).
Ocan, M. et al. Prevalence and antifungal susceptibility of vaginal Candida albicans among pregnant women attending Arua regional referral hospital, West nile region of Uganda. Acta Sci. Microbiol. 1 (6), 17–22 (2018).
Zarrinfar, H., Kord, Z. & Fata, A. High incidence of Azole resistance among Candida albicans and C. glabrata isolates in Northeastern Iran. Curr. Med. Mycol. 7 (3), 18 (2021).
Minooeianhaghighi, M. H., Sehatpour, M., Zarrinfar, H. & Sen, T. Recurrent vulvovaginal candidiasis: the causative agents, clinical signs and susceptibility to fluconazole in Gonabad city, Northeast Iran. Curr. Women’s Health Rev. 16 (1), 46–51 (2020).
Gowher, J., Karpagaselvi, K. & Baker, A. Differentiation of oral Candida species in chronic renal failure patients undergoing Hemodialysis. Oral Maxillofac. Pathol. J. 13(1). (2022).
Hemaid, A. S. S., Abdelghany, M. M. E. & Abdelghany, T. M. Isolation and identification of Candida spp. From immunocompromised patients. Bull. Natl. Res. Centre. 45, 1–8 (2021).
Petra, N. P. et al. Detection of mutated erg11 and fks1 genes among resistant Candida species isolated in pregnant women in mbarara, Uganda. Int. STD Res. Rev. 13 (2), 57–66 (2024).
Kamya Ramesh Swaminathan, D. M. D., Gerald, S. & Swathi, C. Prevalence of vulvovaginal candidiasis in the women of the reproductive age, in rural India. Diabetes. 7, 58 (2017).
Merenstein, D. et al. Colonization by Candida species of the oral and vaginal mucosa in HIV-infected and noninfected women. AIDS Res. Hum. Retroviruses. 29 (1), 30–34 (2013).
Djomand, G. et al. Prevalence and correlates of genital infections among newly diagnosed human immunodeficiency virus–Infected adults entering human immunodeficiency virus care in windhoek, Namibia. Sex. Transm. Dis. 43 (11), 698–705 (2016).
Alczuk, S. S. D. et al. Effect of highly active antiretroviral therapy on vaginal Candida Spp. isolation in HIV-infected compared to HIV-uninfected women. Revista do Instituto de Medicina Tropical de São Paulo. 57(2), 169–174 (2015).
Venugopal, D., Husain, K., Mustafa, S. & Sabeen, S. Epidemiology, risk factors and antimicrobial profile of vulvovaginal candidiasis (VVC): A study among women in the central region of Saudi Arabia. J. Med. Mycol. 31 (2), 101049 (2021).
Apalata, T., Carr, W. H., Sturm, W. A., Longo-Mbenza, B. & Moodley, P. Determinants of symptomatic vulvovaginal candidiasis among human immunodeficiency virus type 1 infected women in rural KwaZulu-Natal, South Africa. Infect. Dis. Obstet. Gynecol. 2014 (1), 387070 (2014).
Mtende, L. M., Kasujja, M. et al. Factors associated with vulvovaginal candidiasis and antifungal susceptibility patterns among nonpregnant women attending the gynecology clinic at Hoima regional referral hospital: A cross-sectional study. (2024).
Mukasa, K. J. et al. Antifungal susceptibility patterns of vulvovaginal Candida species among women attending antenatal clinic at Mbarara regional referral hospital, South Western Uganda. Br. Microbiol. Res. J. 5 (4), 322 (2015).
Mushi, M. F., Olum, R. & Bongomin, F. Prevalence, antifungal susceptibility and etiology of vulvovaginal candidiasis in sub–Saharan africa: A systematic review with meta-analysis and meta-regression. Med. Mycol. 60 (7), myac037 (2022).
Mulu, A. et al. Frequent detection of ‘azole’resistant Candida species among late presenting AIDS patients in Northwest Ethiopia. BMC Infect. Dis. 13, 1–10 (2013).
Makanjuola, O., Bongomin, F. & Fayemiwo, S. A. An update on the roles of non-albicans Candida species in vulvovaginitis. J. Fungi. 4 (4), 121 (2018).
Kiran, C. K., Kandati, J. & Ponugoti, M. Etiologic characterization of vulvovaginitis among females attending a tertiary care hospital: a one year study. Int. J. Reprod. Contracept. Obstet. Gynecol. 6, 2246 (2017).
Yano, J. et al. PL fidel current patient perspectives of vulvovaginal candidiasis: Incidence, symptoms, management and post-treatment outcomes. 19, 48. https://doi.org/10.1186/s12905-019-0748-8 (2019).
Kan, S. et al. In vitro antifungal activity of Azoles and other antifungal agents against pathogenic yeasts from vulvovaginal candidiasis in China. Mycopathologia. 188 (1), 99–109 (2023).
Shrestha, P., Pokharel, S. M. & Shrestha, A. Antifungal susceptibility pattern of Candida isolates causing vulvovaginitis in reproductive age women. Tribhuvan Univ. J. Microbiol. 7, 1–7 (2020).
Lomeli-Martinez, S. M. et al. In vitro Azole antifungals susceptibility of Candida spp. Isolates from HIV-infected patients with periodontitis. J. Med. Mycol. 32 (3), 101294 (2022).
Khan, M., Ahmed, J., Gul, A., Ikram, A. & Lalani, F. K. Antifungal susceptibility testing of vulvovaginal Candida species among women attending antenatal clinic in tertiary care hospitals of Peshawar. Infect. Drug Resist. 447–456 (2018).
Sathi, F. A. et al. Prevalence and antifungal susceptibility of clinically relevant Candida species, identification of Candida auris and Kodamaea ohmeri in Bangladesh. Trop. Med. Infect. Dis. 7 (9), 211 (2022).
Acknowledgements
We acknowledge the district health office for their cooperation during the data collection. We also appreciate the data collectors, supervisors, and study participants for their invaluable input to the success of this study.
Funding
The authors received no funding for this study.
Author information
Authors and Affiliations
Contributions
All authors contributed equally and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mbatudde, N., Nalumaga, P., Bazira, J. et al. Symptomatic vulvovaginal candidiasis and antifungal resistance in HIV-1 positive women at Mbarara city health centre IV. Sci Rep 15, 24123 (2025). https://doi.org/10.1038/s41598-025-09215-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-09215-4