Abstract
Humans constantly explore the visual environment through saccades, bringing relevant visual stimuli to the center of the gaze. Before the eyes begin to move, visual attention is directed to the intended saccade target. As a consequence of this presaccadic shift of attention (PSA), visual perception is enhanced at the future gaze position. PSA has been investigated in a variety of saccade amplitudes, from microsaccades to locations that exceed the oculomotor range. Interestingly, recent studies have shown that PSA effects on visual perception are not equally distributed around the visual field. However, it remains unknown whether the magnitude of presaccadic perceptual enhancement varies with the amplitude of the saccades. Here, we measured contrast sensitivity thresholds during saccade planning in a two-alternative forced-choice (2AFC) discrimination task in human observers. Filtered pink noise (1/f) patches, presented at four eccentricities scaled in size according to the cortical magnification factor were used as visual targets. This method was adopted to mitigate well-known eccentricity effects on perception, thereby enabling us to explore the effects associated to saccade amplitudes. First, our results show that saccade preparation enhanced contrast sensitivity in all tested eccentricities. Importantly, we found that this presaccadic perceptual enhancement was not modulated by the amplitude of the saccades. These findings suggest that presaccadic attention operates consistently across different saccade amplitudes, enhancing visual processing at intended gaze positions regardless of saccade size.
Similar content being viewed by others
Introduction
Many animals-including humans-employ eye movements to actively sample the visual environment1. In most cases, this scanning is carried out by saccadic eye movements that constantly and abruptly change the visual input to the brain. This poses a great challenge for visual processing, as most of the visual sensory areas are retinotopically organized. Perhaps not surprisingly, several perceptual effects have been observed around the onset of saccadic eye movements2,3. One of the most robust effects is the presaccadic shift of attention (PSA)4,5,6, observed by the enhancement of perceptual processing of saccade-targeted objects.
Since PSA was first observed, studies have described several effects of presaccadic attention on visual perception (for reviews, see6,7). For instance, PSA was shown to enhance both contrast sensitivity8 and spatial resolution9 in the periphery of the visual field. Similar effects have been observed in the neural response of many visual areas10. However, recent studies have shown that PSA effects are not equally distributed across the visual field8,11. PSA effects have been observed in a wide variety of stimuli eccentricities5,8,9,12,13,14,15,16,17,18, from microsaccades19,20 (20 arcmin) to eccentricities that exceed the oculomotor range (45 dva)21. In the seminal work by Deubel and Schneider (1996)5, PSA was tested in three eccentricities (3.91, 5 and 6.09 dva). Even though the difference in eccentricity values was arguably too small to test for potential differences in PSA effects, there seems to be a reduction in discrimination performance with eccentricity (especially comparing the 3.91 dva versus 6.09 dva conditions). However, since in their study the visual targets had a fixed size (0.52 and 1.05 dva for width and height, respectively), overall performance decayed with eccentricity, hindering any attempt to interpret their results in terms of saccade amplitude.
Eccentricity effects on visual perception have also been investigated in conditions where attention is spatially allocated without concurrent eye movements- i.e., covert attention22,23,24,25,26,27,28,29. Similar to Deubel and Schneider’s results, visual performance tends to decrease for both endogenous (top-down) and exogenous (bottom-up) covert attention across eccentricities when stimuli sizes are fixed25,26,30. However, when stimuli sizes are scaled according to the cortical magnification factor (M-scaled), these results change. The effects of eccentricity on visual performance are strongly attenuated for endogenous-but not exogenous-attention26,29,30. Nevertheless, it is not possible to deduce the impact of eccentricity on PSA from studies of covert attention, as these processes appear to rely on distinct computations16.
Successful saccadic eye movements rely on a large network of brain areas31. Areas such as frontal eye fields (FEF) and superior colliculus (SC) are known for being involved in the planning of saccades32,33,34. These areas are topographically organized, and microstimulation of specific sites produces saccadic movements of fixed directions and amplitudes35,36. Correspondingly, the firing rates of motor and visuomotor cells in these areas increase prior to saccade execution to locations relative to the fixation position32,37. This pattern of activity is referred to as movement fields. Interestingly, FEF and SC are interconnected with several visual sensory areas38,39,40,41. Microstimulation and pharmacological manipulations of FEF induced attention-like effects in visual areas (V4) and behavior42,43,44. A recent study has shown that transcranial magnetic stimulation (TMS) of FEF during saccade preparation modulates PSA effects45. For these reasons, both areas have been associated with PSA6,10.
Given the close relationship between oculomotor planning and visual perception, here we asked whether presaccadic attentional shifts are modulated by the size of the planned saccades. Since the direction and amplitude of saccades seem to be determined by the topographic location and not the rate of neural activity in oculomotor areas32,46, we expected that PSA effects would be uniform across eccentricities. Our results confirmed this prediction. We found that saccade preparation enhanced contrast sensitivity in all tested eccentricities, and that this enhancement was not modulated by saccade amplitude.
Results
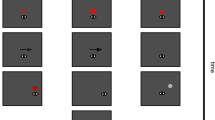
In this study, we investigated the effect of saccade amplitude on presaccadic attentional shifts. We developed a simple task to compare contrast sensitivity prior to saccades executed to targets presented at four eccentricities (Fig. 1A and B). Because we were interested in the effect of saccade amplitude, target sizes were corrected using a magnification factor to minimize the effect of visual eccentricity24,47 (see Stimuli and Task). Sixteen subjects performed a two-alternative forced choice (2AFC) discrimination task, in which they had to report the orientation (clockwise or counterclockwise) of a filtered pink noise target presented at the saccade (towards) or the opposite location (away). Discrimination performance was measured online using an adaptive psychometric procedure (Best PEST), targeting the contrast level for 80% orientation discrimination accuracy. Figure 1C–F shows an example of the staircase procedure applied to one subject in all sessions.
Task design and staircase result. (A) Trial example. Subjects were instructed to look at a fixation point (FP) for at least 500 ms at the beginning of each trial. Then, a stream of pink noise stimuli would be presented on both sides of the FP and remain on the screen until the end of the trial. After a random period of 500–900 ms, a saccadic cue (black line) would indicate the side where a saccade should be executed. After an SOA of \(\sim\)142 ms, a visual target (filtered pink noise) with a clockwise (CW) or counterclockwise (CCW) orientation would appear either on the left or right side of the screen for 50 ms. Subjects had to discriminate the orientation of the target at the end of the trial. (B) Stimuli eccentricities and sizes. (C–F) Example of the staircase procedure applied separately for each stimulus eccentricity. Target contrast is shown in function of the number of trials. Each staircase subplot shows the subject’s performance for cue towards (red) and away (gray) from the target. Filled and open dots represent correct and incorrect discrimination responses, respectively. On the right, the mean and standard error (SEM) for each condition are shown. The mean was calculated using the means of the posterior distribution given by the staircase procedure.
Saccade amplitude does not affect presaccadic attention
To investigate the effect of saccade amplitude on contrast sensitivity, we performed a two-way repeated-measures ANOVA on individual threshold values. As expected, this analysis showed that contrast thresholds were lower when saccades were planned towards the target than in the away condition (Fig. 2, F(1,15) = 81.94, p < 0.0001, partial \({\eta ^2}\) = 0.845). Our analysis also revealed a main effect of stimulus eccentricity (F(3,45) = 9.339, p = 0.0005, partial \({\eta ^2}\) = 0.384). Post-hoc comparisons showed that targets presented at intermediary eccentricities led to lower contrast thresholds when compared to the nearest eccentricity (4.1–6.02 dva, pbonf = 0.0001; and 4.1–8.6 dva, pbonf = 0.001). Importantly, we found no interaction between the cue-target relation (Towards and Away) and the target eccentricity condition (Fig. 3A; F(3,45) = 0.03, p = 0.99, partial \({\eta ^2}\) = 0.002). To quantify the evidence for the null effect of saccade amplitude on presaccadic attention, we performed a Bayesian repeated-measures ANOVA. This analysis was conducted on the presaccadic perceptual gain (Towards-Away) across all eccentricities (Fig. 3B). The resulting Bayes factor (BF01 = 22.691) indicated strong evidence in favor of the null hypothesis. Finally, as an exploratory analysis, we carried out post hoc comparisons using Bayesian t-tests. All comparisons yielded moderate to strong evidence supporting the null hypothesis (BFs01 > 3.767). These results support the interpretation that presaccadic shifts of attention are not modulated by the amplitude of the saccade being prepared to the target location.
Effect of presaccadic attention on contrast sensitivity. (A) Target contrast as a function of cue-target relation: Towards and Away conditions are shown in red and gray, respectively. The black dot on each boxplot represents the median and the whiskers represent the upper and lower value. The colored clouds represent the density distribution given by the dots, which represent subjects’ performance individually. The asterisk indicates the statistical significance (p < 0.0001). (B) Bagplot for contrast sensitivity for towards vs away cue-target relation condition. The brown polygon ( bag ) contains 50% of the data (black dots) whereas the fence, in a lighter brown color, contains the remaining non-outlier data. The median is indicated by a cross.
Presaccadic attention effects in all tested eccentricities. (A) Target contrast as a function of cue-target relation for each stimulus eccentricity: Towards and Away conditions are shown in red and gray, respectively. The black dot on each boxplot represents the median and the whiskers represent the upper and lower value. The colored clouds represent the density distribution given by the dots, which represent subjects’ performance individually. (B) Presaccadic perceptual gain (Towards-Away) across all eccentricities. Colored clouds represent the density distribution given by the dots separately for each eccentricity. The Bayes factor (BF01 = 22.691) indicates strong evidence in favor of the null hypothesis, suggesting no meaningful effect of saccade amplitude on contrast sensitivity.
Control analyses on saccade parameters
One possible confound in our study could be raised if saccades had different latencies for the different eccentricities. It is known that PSA effects are strongest when the target is presented just before saccade onset48. Because we used a fixed cue-target interval, if PSA effects decayed with eccentricity, but saccade latency increased, for example, then one effect could be canceling the other (i.e., targets in the nearest eccentricities would be presented closer to saccade onset). We addressed this possible confound with two analyses. First, we performed a two-way repeated-measures ANOVA on saccadic reaction times (SRTs) across eccentricities, with cue-target relation (towards and away) and stimulus eccentricity as factors. This analysis did not reveal a significant effect of cue-target relation on SRT (F(1,15) = 2.3797, p = 0.14375, partial \({\eta ^2}\) = 0.137). There was a significant effect of Eccentricity on SRT (F(3,45) = 19.903, p < 0.0001, partial \({\eta ^2}\) = 0.570). Post-hoc analyses showed that SRT tends to decrease with eccentricity (Fig. 4A–D) [4.1–6.02 dva, permuted-p = 0.004; 4.1–8.6 dva, permuted-p < 0.0001; 4.1–12.5 dva, permuted-p < 0.0001; 6.02–8.6 dva, permuted-p < 0.0001; 6.02–12.5 dva, permuted-p < 0.0001; 8.6–12.5 dva, permuted-p = 0.005]. It is worth noting that this pattern of results appears to contrast with studies showing that–at least for medium-sized saccades–saccade latency increases with amplitude49,50. However, recent evidence indicates that this relation disappears when stimulus size is scaled according to the magnification factor observed in SC and V151. Importantly, we found no interaction between the factors (F(3,45) = 1.381, p = 0.26486, partial \({\eta ^2}\) = 0.084). To further rule out a potential confound of SRT on our main findings, we repeated the primary contrast sensitivity analysis using the SRT as a covariate in a two-way repeated-measures ANCOVA. For this analysis, first we fitted a linear regression on the SRT values for each eccentricity and subject. Then we used the slope values as a covariate to test if any of our contrast sensitivity results could be explained by a modulation of SRT (see Data Analysis subsection for details). As with the primary ANOVA, this analysis revealed a significant main effect of cue-target relation, F(1, 14) = 40.462, p < 0.001, partial \({\eta ^2}\) = 0.743), indicating robust differences between towards and away saccades, and no interactions between the factors (F(1, 14) = 0.470, p = 0.705, partial \({\eta ^2}\) = 0.032). These results confirm that the observed presaccadic modulation of contrast sensitivity is not explained by individual differences in SRT. Interestingly, the main effect of eccentricity on contrast sensitivity was no longer significant when SRT was included as a covariate (F(1, 14) = 1.203, p = 0.32, partial \({\eta ^2}\) = 0.079), suggesting that the effects of eccentricity on contrast sensitivity and SRT are not independent, and may be driven by a common underlying factor.
Another concern could be related to saccade accuracy. Even though it is known that PSA is associated with saccade planning and not execution5,52, and that it occurs even when saccade endpoints fall as far as 1.5° away from the edge of the target stimulus21, one could still argue that highly inaccurate saccades could affect our main results. We performed an analysis to investigate the effect of eccentricity on saccade accuracy. Saccade accuracy data were corrected for each eccentricity according to the cortical magnification factor. This analysis revealed that saccade accuracy was equivalent for both cue-target relation conditions (F(1,15) = 1.3491, p = 0.26, partial \({\eta ^2}\) = 0.083) and across all eccentricities (F(3,45) = 0.22468, p = 0.73, partial \({\eta ^2}\) = 0.015) (Fig. 4E–H). We found no interaction between the factors (F(3,45) = 0.43749, partial \({\eta ^2}\) = 0.028). As in the previous analysis, we also examined whether saccade accuracy influenced contrast sensitivity analysis by conducting an ANCOVA including the slope of saccade accuracy change across eccentricities as a covariate. Consistent with the primary ANOVA, this analysis revealed a significant main effect of cue-target relation (F(1, 14) = 88.251, p < 0.001, partial \({\eta ^2}\) = 0.863), eccentricity (F(1, 14) = 8.612, p < 0.001, partial \({\eta ^2}\) = 0.381) indicating robust differences between toward and away saccades. Crucially, no significant interactions between factors were observed (F(1, 14) = 0.068, p = 0.976, partial \({\eta ^2}\) = 0.067). These findings confirm that the presaccadic modulation of contrast sensitivity cannot be attributed to individual differences in saccade accuracy.
Saccade parameters results. (A–D) Histograms depicting saccade reaction time (SRT) for cue-target relation condition towards (red) and away (gray) from the target for each eccentricity. (E–H) Density distribution of saccade landing position for each stimulus eccentricity for all subjects. Dashed circles represent noise stimuli. Left Y-axis: vertical landing position; X-axis: horizontal landing position. Right Y-axis: Histogram depicting the number of trials as a function of saccade landing position for each stimulus eccentricity..
Discussion
Presaccadic shifts of attention have been observed using a wide range of stimulus eccentricities5,8,9,12,13,14,15,16,17,18. Given the intrinsic link between stimuli eccentricities and saccade amplitudes, we expected that visual attention could be similarly engaged for saccades of different sizes. However, until now, the evidence for this assumption could only be obtained by combining results from several independent studies. Therefore, here we directly investigated if presaccadic attentional effects on visual perception are modulated by saccade amplitudes. We employed filtered pink noise (1/f) patches as target stimuli, scaled in size according to the cortical magnification factor. This approach aimed to mitigate the well-known effects of stimulus eccentricity on contrast sensitivity, allowing us to examine the effects of saccade amplitude on presaccadic shift of attention (PSA). Our results indicated that saccade preparation enhanced contrast sensitivity in all tested eccentricities. Furthermore, we showed that this presaccadic perceptual enhancement was not modulated by the amplitude of the saccade. Control analyses suggested that this result could not be explained by differences in saccade statistics.
It has long been established that oculomotor areas, such as the frontal eye fields (FEF) and superior colliculus (SC), are involved in attentional selection53,54. Unsurprisingly, several studies have suggested that PSA also depends on the involvement of these areas42,43,55,56. Neural recording and stimulation studies in these areas have shown that they are topographically organized57. For example, stimulation of specific sites in FEF and SC will produce saccades with a fixed direction and amplitude relative to the current eye position35,36. Crucially, these saccade vectors are defined by the topographical location and not by the intensity of the stimulation or the firing rate.32,46,58. Therefore, the lack of interaction between saccade amplitude and presaccadic enhancement observed in our study aligns with the topographical organization of these brain areas.
It is well known that visual perception is not equally distributed across the visual field (for a review, see59). Instead, contrast sensitivity is much reduced in the upper vertical meridian47,60,61. Interestingly, recent studies have shown that PSA effects are unevenly allocated across the visual space8,11. These studies reveal a polar angle asymmetry, in which the contrast sensitivity enhancement before upward vertical saccades is highly diminished-or even extinguished. We did not find any asymmetry in the magnitude of contrast enhancement before horizontal saccades for stimuli at different eccentricities.
One interesting takeaway from our study is that PSA effects are probably not linked to motor training. Studies on the ecology of saccades have shown that the natural distribution of human saccade amplitudes is far from uniform62,63. We execute nearly twice as many 5° saccades than 10° ones in our daily activities62. If PSA was associated with the frequency of saccade amplitudes, then we would expect that our results followed the natural distribution. Instead, our results showed that PSA effects–at least for the eccentricities tested–seem to be uniformly distributed.
Evidence from psychophysical studies suggests that PSA is determined by the intention of the saccade goal and not the execution accuracy5,21,52,64. Given that saccades do not always land at the intended location–often undershooting the target–a large number of studies, starting from Deubel and Schneider’s (1996)5 seminal work, have proposed that despite these saccade errors, visual perception is always enhanced at the intended location. Wollenberg and colleagues (2018)52 tested perceptual enhancement prior to averaging saccades (i.e., when saccades land in between two closely located saccade targets), which allowed the dissociation of visual sensitivity modulations at the intended saccade goal versus at the endpoint of the saccade. Their results showed that, while accurate saccades toward one of the targets were associated with presaccadic enhancement, there was no attentional facilitation at the saccade endpoint of an averaging saccade. Another evidence comes from a study by Hanning and colleagues (2019)21 showing PSA effects to targets located outside the oculomotor range, even though, in these conditions, saccades always fell short of the target location. Our results agree with these proposals. One would expect that if PSA were related to the execution and not intention of the saccade, perceptual enhancement effects would decrease with saccade amplitude, as saccade accuracy tends to decline with increasing target eccentricity65,66. In our study, this was observed as an increase in the distribution (i.e., variability) of saccade endpoints with increasing amplitude (see Fig. 4E–H). Thus, despite comparable mean accuracy across eccentricities, the growing variance in endpoint location with saccade amplitude could, if PSA were linked to saccade execution, lead to diminished enhancement even when stimuli are appropriately magnified. Conversely, our results show that contrast sensitivity enhancement remained consistent prior to saccades ranging from 4.1 to 12.5° of visual angle, supporting the view that PSA is more closely tied to saccade intention than execution.
The majority of the evidence on the relationship between visual attention and target eccentricity comes from visual search studies23,24,67,68,69,70. These studies have shown that performance consistently decreases with the eccentricity of targets in a search array. However, this eccentricity effect seems to diminish when correcting the array stimuli for cortical magnification (30,69, cf.26), or when a pre-cue is presented indicating the target location68. More recently, using a classic Posner-like (covert) attentional task, Jigo and Carrasco (2020)28 have shown that a central cue is capable of enhancing the contrast sensitivity of targets presented at different eccentricities (0, 3, 6, and 12 dva), and across a wide range of spatial frequencies. Similarly, Hanning and Deubel71 have shown that visual performance is equated for 1/f stimuli when a pre-cue is presented.
Our work raises several interesting questions to be investigated in future studies. Here, we used a fixed cue-target SOA of \(\sim\)142 ms. This prevented us from investigating the time course of the perceptual enhancement before saccade onset. It is possible that differences in PSA for different saccade amplitudes might emerge when analyzing its temporal profile more carefully. In our study, we also limited the analysis of PSA effects to stimuli up to 12.5° in eccentricity. This was done for two main reasons. First, we wanted to avoid presenting stimuli near the blind spot, found around 15° in the temporal horizontal hemispace for each eye. Second, under natural, free-viewing conditions, head movements begin to contribute to gaze shifts for locations larger than 20° in both humans and non-human primates (for a review, see72). It is possible that PSA effects are attenuated for saccades larger than 20°, when head movements start being engaged. Finally, we have limited stimulus position to the horizontal meridian, as horizontal saccades are more common than vertical ones under natural behavior conditions62, and because–as described above–there are known differences in performance across the visual field8,11.
In conclusion, our study showed that PSA enhances contrast sensitivity equally before saccades of up to 12.5°. Moreover, our results suggest that any saccade amplitude within the range used here can be applied to future PSA studies without impairments to the phenomenon.
Material and methods
Participants
Sixteen subjects participated in this study (10 females and 6 males; mean age 26.75). Except for three authors (L.Z.B, E.C.L, M.H.S.Z), all participants were naive to the purposes of the study. All participants had normal or corrected-to-normal vision, with 11 of them having dominance in the right eye. All participants gave informed written consent. The procedures for the study were approved by the local Ethics Research Committee of the University of São Paulo (approval number: 25333219.5.0000.5464), in accordance with the Declaration of Helsinki.
Apparatus
The experiment was carried out in a quiet, dimly lit room, where the participants sat comfortably in front of a computer screen and positioned their heads on a chin-rest. Stimuli were presented at a distance of 57 cm from the subjects’ eyes on a high-resolution screen (1920 x 1080, refresh rate 120 Hz, VIEWPixx/3D, VPixx Technologies, Canada) controlled using the Psychtoolbox toolbox73 in MATLAB74. Manual responses were recorded using a response box (ResponsePixx). Eye position was monitored at 1KHz sampling rate using the desktop mount EyeLink Plus 1000 (Eye Link Plus 1000 SR Research).
Stimuli and task
Each trial started with a fixation point on a gray background. The fixation point consisted of a white dot with a diameter of 0.3 dva, within a black circle with a diameter of 0.64 dva. After 500 ms of central fixation–within a 2.35 dva radius window–two visual streams (50 ms SOA) of pink noise stimuli were presented at the left and right sides of the fixation dot until the end of the trial. After a random interval between 500-900 ms, a central line (0.41 dva in length and 0.14 dva in width) pointed to the left or right side (75 ms duration), signaling a spatial cue. Subjects were instructed to execute a saccade as fast as possible to the stimuli location at the cued side. Then, following an interval of 67 ms (\(\sim\)142 ms cue-target SOA), an orientation-filtered pink noise patch (40° or −40° relative to vertical) was presented at the cued or uncued location for 50 ms. Subjects were instructed to discriminate in a nonspeeded manner the target’s orientation (clockwise or counterclockwise) by pressing a button on the response box. The saccadic cue was not predictive of the future target location (i.e., 50% validity).
Visual performance was tested at four different logarithmically spaced eccentricities (4.1, 6.02, 8.6, and 12.5 dva), and a magnification factor (M-scaling) was applied to the stimuli sizes (1.78, 2.2, 2.78, and 3.64 dva) (Fig. 1B). Using the magnification factor in combination with the pink noise stimuli ensured that visual performance would remain similar across all eccentricities30,47,71.
Procedures
Participants performed three sessions of 320 trials (960 trials in total). Each session consisted of 20 short blocks of 16 trials, with approximately 30–40 min of duration. At the beginning of each block, a message displayed on the screen indicated the stimulus eccentricity in that block. The eccentricity was randomized between blocks. Within the same block, trials were repeated if: the subjects’ eyes deviated from the fixation point before cue onset; saccade reaction times were faster than 190 ms or slower than 350 ms; saccades were executed to an uncued location; no saccade was detected during the whole trial. Subjects received visual feedback indicating unsuccessful saccade execution at the end of these trials (the fixation point turned yellow). Subjects were allowed to rest between blocks.
Before starting each experimental session, the participants performed a short training session similar to the main experiment. During training, in addition to the saccade feedback, participants also received a second feedback regarding the discrimination performance (the fixation point turned green for correct or red for incorrect responses). In the first experimental session, participants received detailed instructions before the training session.
Staircase procedure
A Best Pest staircase procedure75 was used to measure the contrast level for 80% correct discrimination responses in each condition, using Palamedes toolbox76. For each session, eight staircases were applied concurrently to derive the targets’ contrast level for each of the eight experimental conditions: Cue-target relation (Towards and Away) and Stimulus Eccentricity (4.1, 6.02, 8.6, and 12.5 dva). Each staircase condition consisted of 40 trials. The contrast threshold for each condition was derived from the mean of the posterior distribution. Then, before calculating the overall mean across all sessions for each condition, we excluded outlier data (1.56% of all staircase procedures) for which the mean of a specific staircase condition was higher than 0.5 log compared to the other two means in the same condition. Moreover, we also excluded the data from an entire session of a subject who initially misunderstood the instructions and took three times longer to complete the session. Running the same analysis but without excluding the outlier values yielded similar results than with exclusions.
Data analysis
The analysis of discrimination performance was based on the contrast level needed to achieve 80% correct discrimination in each condition (see staircase procedure above). In addition to discrimination thresholds, we also considered as dependent variables saccade reaction times (SRT) and saccade accuracy. Two-way repeated-measures ANOVAs were implemented for all dependent variables. Additionally, we implemented two-way repeated-measures ANCOVAs for contrast level, using SRTs and saccade accuracy as covariates. A linear regression analysis was performed on these covariates values for each eccentricity. The slope of each subject’s curve was then used as covariates. In the results section, p-values are shown with Greenhouse-Geisser correction due to sphericity violation. Bonferroni correction was applied to post-hoc analysis on discrimination performance. Since SRT and saccade accuracy did not show normal distribution, according to the Kolmogorov-Smirnov test, post-hoc analyses were run using a non-parametric permutation test77. Given that the stimuli sizes were corrected by the magnification factor, saccade accuracy data was corrected for each eccentricity by dividing the post-saccade position by the stimulus radius. We report effect sizes in terms of partial eta squared (partial \({\eta ^2}\) ) for ANOVAs and ANCOVAs. Bayesian ANOVAs and t-tests were used to report the strength of evidence for or against the null hypothesis. All Bayes Factors (BF) were calculated using a standard Cauchy prior (s = 0.707) for the alternative hypothesis. Reported BF01 values indicate how many times more evidence there is in favor of the null hypothesis compared with the alternative hypothesis. All data analyses were run on MATLAB (R2022b) and JASP (Version 0.19.3)[Computer software]78.
Eye movements
In addition to the ’online’ eye movement analysis, we used a velocity-based saccade detection algorithm to detect saccades offline, which provides more detailed information regarding saccade parameters79. Saccades onset and offset were detected based on the current eye velocity at the point in which median eye velocity exceeded 5 SDs with a threshold duration of 8 ms. This analysis was used only for SRT and saccade accuracy data. A total of 497 (3,24%) trials were excluded.
Data availability
The datasets generated in this study can be acquired from the corresponding author on request.
References
Yarbus, A. L. Eye movements and vision (Springer, 2013).
Golomb, J. D. & Mazer, J. A. Visual remapping. Annual Rev. Vis. Sci. 7, 257–277 (2021).
Binda, P. & Morrone, M. C. Vision during saccadic eye movements. Annual Rev. Vis. Sci. 4, 193–213 (2018).
Kowler, E., Anderson, E., Dosher, B. & Blaser, E. The role of attention in the programming of saccades. Vision. Res. 35, 1897–1916 (1995).
Deubel, H. & Schneider, W. X. Saccade target selection and object recognition: Evidence for a common attentional mechanism. Vision. Res. 36, 1827–1837 (1996).
Li, H.-H., Hanning, N. M. & Carrasco, M. To look or not to look: dissociating presaccadic and covert spatial attention. Trends Neurosci. 44, 669–686 (2021).
Carrasco, M. How visual spatial attention alters perception. Cogn. Process. 19, 77–88 (2018).
Hanning, N. M., Himmelberg, M. M. & Carrasco, M. Presaccadic attention enhances contrast sensitivity, but not at the upper vertical meridian. iScience 25, 103851 (2022).
Kroell, L. M. & Rolfs, M. The peripheral sensitivity profile at the saccade target reshapes during saccade preparation. Cortex 139, 12–26 (2021).
Moore, T. & Zirnsak, M. Neural mechanisms of selective visual attention. Annu. Rev. Psychol. 68, 47–72 (2017).
Hanning, N. M., Himmelberg, M. M. & Carrasco, M. Presaccadic attention depends on eye movement direction and is related to v1 cortical magnification. J. Neurosci. 44, e1023232023 (2024).
Ouerfelli-Ethier, J., Bona, I. C., Fournet, R., Pisella, L. & Khan, A. Z. Pre-saccadic attention relies more on suppression than does covert attention. J. Vis. 23, 1–1 (2023).
Schneider, W. X. & Deubel, H. Visual Attention and Saccadic Eye Movements: Evidence for Obligatory and Selective Spatial Coupling. In Studies in Visual Information Processing, vol. 6, 317–324 (Elsevier, 1995).
Born, S., Mottet, I. & Kerzel, D. Presaccadic perceptual facilitation effects depend on saccade execution: Evidence from the stop-signal paradigm. J. Vis. 14, 7–7 (2014).
Kwak, Y., Hanning, N. M. & Carrasco, M. Presaccadic attention sharpens visual acuity. Sci. Rep. 13, 2981 (2023).
Li, H.-H., Pan, J. & Carrasco, M. Different computations underlie overt presaccadic and covert spatial attention. Nat. Hum. Behav. 5, 1418–1431 (2021).
Li, H.-H., Barbot, A. & Carrasco, M. Saccade preparation reshapes sensory tuning. Curr. Biol. 26, 1564–1570 (2016).
Rolfs, M. & Carrasco, M. Rapid simultaneous enhancement of visual sensitivity and perceived contrast during saccade preparation. J. Neurosci. 32, 13744–13752a (2012).
Shelchkova, N. & Poletti, M. Modulations of foveal vision associated with microsaccade preparation. Proc. Natl. Acad. Sci. 117, 11178–11183 (2020).
Guzhang, Y., Shelchkova, N., Clark, A. M. & Poletti, M. Ultra-fine resolution of pre-saccadic attention in the fovea. Curr. Biol. 34, 147–155 (2024).
Hanning, N. M., Szinte, M. & Deubel, H. Visual attention is not limited to the oculomotor range. Proc. Natl. Acad. Sci. 116, 9665–9670 (2019).
Carrasco, M., McElree, B., Denisova, K. & Giordano, A. M. Speed of visual processing increases with eccentricity. Nat. Neurosci. 6, 699–700 (2003).
Carrasco, M., Evert, D. L., Chang, I. & Katz, S. M. The eccentricity effect: Target eccentricity affects performance on conjunction searches. Percept. Psychophys. 57, 1241–1261 (1995).
Carrasco, M. & Frieder, K. S. Cortical magnification neutralizes the eccentricity effect in visual search. Vision. Res. 37, 63–82 (1997).
Feng, J. & Spence, I. The effects of spatial endogenous pre-cueing across eccentricities. Front. Psychol. 8, 888 (2017).
Staugaard, C. F., Petersen, A. & Vangkilde, S. Eccentricity effects in vision and attention. Neuropsychologia 92, 69–78 (2016).
Barbot, A. & Carrasco, M. Attention modifies spatial resolution according to task demands. Psychol. Sci. 28, 285–296 (2017).
Jigo, M. & Carrasco, M. Differential impact of exogenous and endogenous attention on the contrast sensitivity function across eccentricity. J. Vis. 20, 11 (2020).
Casteau, S. & Smith, D. T. Covert attention beyond the range of eye-movements: Evidence for a dissociation between exogenous and endogenous orienting. Cortex 122, 170–186 (2020).
Carrasco, M. & Frieder, K. S. Cortical magnification neutralizes the eccentricity effect in visual search. Vision. Res. 37, 63–82 (1997).
Munoz, D. P. Commentary: saccadic eye movements: overview of neural circuitry. Prog. Brain Res. 140, 89–96 (2002).
Bruce, C. J. & Goldberg, M. E. Primate frontal eye fields. i. Single neurons discharging before saccades. J. Neurophysiol. 53, 603–635 (1985).
Wurtz, R. H. & Goldberg, M. E. Activity of superior colliculus in behaving monkey. 3. Cells discharging before eye movements. J. Neurophysiol. 35, 575–586 (1972).
Liu, Y., Yttri, E. A. & Snyder, L. H. Intention and attention: different functional roles for lipd and lipv. Nat. Neurosci. 13, 495–500 (2010).
Schiller, P. H. & Koerner, F. Discharge characteristics of single units in superior colliculus of the alert rhesus monkey. J. Neurophysiol. 34, 920–936 (1971).
Schiller, P. H. & Tehovnik, E. J. Neural mechanisms underlying target selection with saccadic eye movements. Prog. Brain Res. 149, 157–171 (2005).
Munoz, D. P. & Wurtz, R. H. Saccade-related activity in monkey superior colliculus. ii. spread of activity during saccades. J. Neurophysiol. 73, 2334–2348 (1995).
Stanton, G., Bruce, C. & Goldberg, M. Topography of projections to posterior cortical areas from the macaque frontal eye fields. J. Comparative Neurol. 353, 291–305 (1995).
Schall, J. D., Morel, A., King, D. J. & Bullier, J. Topography of visual cortex connections with frontal eye field in macaque: Convergence and segregation of processing streams. J. Neurosci. 15, 4464–4487 (1995).
Markov, N. T. et al. Weight consistency specifies regularities of macaque cortical networks. Cereb. Cortex 21, 1254–1272 (2011).
Cerkevich, C. M., Lyon, D. C., Balaram, P. & Kaas, J. H. Distribution of cortical neurons projecting to the superior colliculus in macaque monkeys. Eye Brain 6, 121–137 (2014).
Moore, T. & Fallah, M. Microstimulation of the frontal eye field and its effects on covert spatial attention. J. Neurophysiol. 91, 152–162 (2004).
Moore, T. & Armstrong, K. M. Selective gating of visual signals by microstimulation of frontal cortex. Nature 421, 370–373 (2003).
Noudoost, B. & Moore, T. Control of visual cortical signals by prefrontal dopamine. Nature 474, 372–375 (2011).
Hanning, N. M., Fernández, A. & Carrasco, M. Dissociable roles of human frontal eye fields and early visual cortex in presaccadic attention. Nat. Commun. 14, 5381 (2023).
Schiller, P. H. & Stryker, M. Single-unit recording and stimulation in superior colliculus of the alert rhesus monkey. J. Neurophysiol. 35, 915–924 (1972).
Jigo, M., Tavdy, D., Himmelberg, M. M. & Carrasco, M. Cortical magnification eliminates differences in contrast sensitivity across but not around the visual field. eLife 12, e84205 (2023).
Deubel, H. The time course of presaccadic attention shifts. Psychol. Res. 72, 630–640 (2008).
Kalesnykas, R. & Hallett, P. Retinal eccentricity and the latency of eye saccades. Vision. Res. 34, 517–531 (1994).
Hafed, Z. M. & Goffart, L. Gaze direction as equilibrium: more evidence from spatial and temporal aspects of small-saccade triggering in the rhesus macaque monkey. J. Neurophysiol. 123, 308–322 (2020).
Zhang, Y. & Fries, P. Eccentricity-dependent saccadic reaction time: The roles of foveal magnification and attentional orienting. bioRxiv 2023–08 (2023).
Wollenberg, L., Deubel, H. & Szinte, M. Visual attention is not deployed at the endpoint of averaging saccades. PLoS Biol. 16, e2006548 (2018).
Bogadhi, A. R., Bollimunta, A., Leopold, D. A. & Krauzlis, R. J. Brain regions modulated during covert visual attention in the macaque. Sci. Rep. 8, 15237 (2018).
Bollimunta, A., Bogadhi, A. R. & Krauzlis, R. J. Comparing frontal eye field and superior colliculus contributions to covert spatial attention. Nat. Commun. 9, 3553 (2018).
Noudoost, B., Clark, K. L. & Moore, T. A distinct contribution of the frontal eye field to the visual representation of saccadic targets. J. Neurosci. 34, 3687–3698 (2014).
Bisley, J. W. & Mirpour, K. The neural instantiation of a priority map. Curr. Opin. Psychol. 29, 108–112 (2019).
Sommer, M. A. & Wurtz, R. H. Composition and topographic organization of signals sent from the frontal eye field to the superior colliculus. J. Neurophysiol. 83, 1979–2001 (2000).
Ohayon, S., Grimaldi, P., Schweers, N. & Tsao, D. Y. Saccade modulation by optical and electrical stimulation in the macaque frontal eye field. J. Neurosci. 33, 16684–16697 (2013).
Strasburger, H., Rentschler, I. & Jüttner, M. Peripheral vision and pattern recognition: A review. J. Vis. 11, 13–13 (2011).
Abrams, J., Nizam, A. & Carrasco, M. Isoeccentric locations are not equivalent: The extent of the vertical meridian asymmetry. Vision. Res. 52, 70–78 (2012).
Rubin, N., Nakayama, K. & Shapley, R. Enhanced perception of illusory contours in the lower versus upper visual hemifields. Science 271, 651–653 (1996).
Foulsham, T., Teszka, R. & Kingstone, A. Saccade control in natural images is shaped by the information visible at fixation: Evidence from asymmetric gaze-contingent windows. Atten., Percept., Psychophys. 73, 266–283 (2011).
Irving, E. L. & Lillakas, L. Difference between vertical and horizontal saccades across the human lifespan. Exp. Eye Res. 183, 38–45 (2019).
Hanning, N. M. & Deubel, H. The effect of spatial structure on presaccadic attention costs and benefits assessed with dynamic 1/f noise. J. Neurophysiol. 127, 1586–1592 (2022).
Abrams, R. A., Meyer, D. E. & Kornblum, S. Speed and accuracy of saccadic eye movements: Characteristics of impulse variability in the oculomotor system. J. Exp. Psychol. Hum. Percept. Perform. 15, 529 (1989).
Guadron, L., van Opstal, A. J. & Goossens, J. Speed-accuracy tradeoffs influence the main sequence of saccadic eye movements. Sci. Rep. 12, 5262 (2022).
Wolfe, J. M., O’Neill, P. & Bennett, S. C. Why are there eccentricity effects in visual search? visual and attentional hypotheses. Perception Psychophys. 60, 140–156 (1998).
Carrasco, M. & Yeshurun, Y. The contribution of covert attention to the set-size and eccentricity effects in visual search. J. Exp. Psychol. Hum. Percept. Perform. 24, 673 (1998).
Carrasco, M., McLean, T. L., Katz, S. M. & Frieder, K. S. Feature asymmetries in visual search: Effects of display duration, target eccentricity, orientation and spatial frequency. Vision. Res. 38, 347–374 (1998).
Carrasco, M., Giordano, A. M. & McElree, B. Attention speeds processing across eccentricity: Feature and conjunction searches. Vision. Res. 46, 2028–2040 (2006).
Hanning, N. M. & Deubel, H. A dynamic 1/f noise protocol to assess visual attention without biasing perceptual processing. Behav. Res. Methods 55(5), 2583–2594 (2022).
Freedman, E. G. Coordination of the eyes and head during visual orienting. Exp. Brain Res. 190, 369–387 (2008).
Kleiner, M., Brainard, D. & Pelli, D. What’s new in psychtoolbox-3? (2007).
Inc., T. M. Matlab version: 9.13.0 (r2022b) (2022).
Pentland, A. Maximum-likelihood estimation-the best pest (1980).
Prins, N. & Kingdom, F. A. Applying the model-comparison approach to test specific research hypotheses in psychophysical research using the palamedes toolbox. Front. Psychol. 9, 1250 (2018).
Maris, E., Schoffelen, J.-M. & Fries, P. Nonparametric statistical testing of coherence differences. J. Neurosci. Methods 163, 161–175 (2007).
JASP Team. JASP (Version 0.19.3)[Computer software] (2025). https://jasp-stats.org/.
Engbert, R. & Mergenthaler, K. Microsaccades are triggered by low retinal image slip. Proc. Natl. Acad. Sci. 103, 7192–7197 (2006).
Acknowledgements
This work was supported by the São Paulo Research Foundation (FAPESP) and by IDOR/Pioneer Science Initiative (https://www.pioneerscience.org). We thank everyone who volunteered to participate in our experiment. We would also like to thank all our lab colleagues for all the suggestions given throughout the development of the study.
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology: G.R., L.Z.B., E.C.L., M.H.S.Z., G.M.M. Research: G.R., L.Z.B., E.C.L. Software: L.Z.B., G.M.M. Data collection: L.Z.B., M.H.S.Z. Data analysis: L.Z.B., E.C.L., G.R. Visualization: L.Z.B. Writing: L.Z.B., G.R. Manuscript Review: G.R., L.Z.B., E.C.L., G.M.M. Funding acquisition: G.R.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zimmermann Bortoluzzi, L., Carlos-Lima, E., Mueller de Melo, G. et al. Presaccadic attentional shifts are not modulated by saccade amplitude. Sci Rep 15, 27780 (2025). https://doi.org/10.1038/s41598-025-09338-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-09338-8