Abstract
Fascioliasis, a significant foodborne parasitic disease, is transmitted via Lymnaea natalensis snails. This study evaluated the molluscicidal activities of Senna alata ethanolic extracts and its green-synthesized silver nanoparticles (AgNPs) against adult L. natalensis and its ova. AgNPs were synthesized using S. alata leaf extract, confirmed by UV-visible spectroscopy with an absorption peak at 362 nm. Characterization via XRD revealed crystalline peaks at 2θ values of 25.50°, 30.50°, and 38.00°, while SEM showed irregularly shaped particles averaging 20.32 nm with an elemental composition of 47.04% silver. FTIR identified key functional groups, including aromatic C–H stretching (3016.39 and 3098.26 cm−1) and O–H stretching of phenolics. Molluscicidal assays demonstrated that AgNPs achieved 100% snail mortality within 40 min at 0.167 mg/mL, significantly outperforming crude extracts (60% mortality at 5 mg/mL after 24 h; p < 0.05). The green-synthesized molluscicide AgNPs halted embryonic development at the blastula stage at all concentrations, whereas crude extracts showed minimal effects. Oxidative stress markers in snails exposed to AgNPs indicated elevated lipid peroxidation (p < 0.01) and catalase activity (p < 0.05), alongside reduced superoxide dismutase activity (p < 0.05). SDS-PAGE analysis revealed altered protein expression, with new bands at 17–95 kDa in AgNP-exposed snails. These findings highlight S. alata-AgNPs as a potent, eco-friendly molluscicide, effective at low concentrations. Further studies on environmental safety and field applications are recommended.
Similar content being viewed by others
Introduction
Since 2006, WHO has estimated the Global Burden of Disease (GBD) for foodborne trematodiases (FBTs) using disability-adjusted life years (DALYs)1. A 2023 scoping review found FBTs in 42% (93/224) of countries, with 26 being co-endemic2. Global Burden of Disease (GBD) 2019 reported 3.35 million cases and a DALY rate of 9.473. Among the FBTs, fascioliasis, caused by Fasciola hepatica and Fasciola gigantica, (liver fluke), has been reported between 1980 and 2022 in 26 African countries; with F. hepatica being restricted to 12 countries and F. gigantica documented in 24 African countries4. This neglected zoonotic disease is transmitted when definitive hosts (e.g., livestock, humans) ingest freshwater vegetation or water contaminated with Fasciola metacercariae5. The parasite’s lifecycle critically depends on Lymnaea snails as intermediate hosts, where miracidia develop into sporocysts, rediae, and finally cercariae. These cercariae are released into aquatic environments and encyst as metacercariae on vegetation5,6. In sub-Saharan Africa, L. natalensis serves as the primary intermediate host for F. gigantica, driving endemic transmission in both human and veterinary populations6,7.
Despite the availability of triclabendazole as an effective chemotherapeutic treatment, concerns about drug resistance necessitate alternative control measures8. Molluscicides have historically played a crucial role in controlling Lymnaea snail populations to mitigate the spread of fascioliasis. However, synthetic molluscicides such as niclosamide, metaldehyde, and copper sulphate pose environmental and ecological risks, prompting the need for natural plant-based alternatives9,10.
The green synthesis of metallic nanoparticles, particularly silver nanoparticles (AgNPs), has emerged as an eco-friendly alternative to traditional chemical and physical methods, leveraging plant extracts rich in phytochemicals. These biomolecules act as reducing and stabilizing agents, facilitating the conversion of Ag+ ions to Ag⁰ nanoparticles while minimizing toxic by-products11,12. For instance, Citrus limon and C. sinensis extracts have been used to synthesize AgNPs of 21 nm and 13 nm, respectively, demonstrating size-dependent antibacterial efficacy against Klebsiella pneumoniae13. Similarly, Azadirachta indica leaf extract produced spherical AgNPs (7.5 nm) with notable antimicrobial activity, attributed to oxidative stress induction and glutathione-S-transferase inhibition in Staphylococcus aureus13. The mechanism involves secondary metabolites like quercetin and polyphenols, which reduce Ag+ ions and cap the nanoparticles, enhancing stability and bioactivity14. Green-synthesized AgNPs exhibit superior advantages, including cost-effectiveness, scalability, and reduced environmental impact, while their diverse applications span antimicrobial, antiparasitic, and molluscicidal activities10,12,13,14,15. This approach aligns with sustainable nanotechnology, offering a viable solution to combat drug-resistant pathogens and the parasite intermediate host like L. natalensis.
Large-scale green synthesis of nanoparticles using plant extracts is increasingly viable for industrial applications due to its cost-effectiveness, scalability, and eco-friendliness. Studies demonstrate successful AgNP production using plant extract with methods adaptable to industrial scales13,15,16,17,18. Plant-based synthesis avoids expensive reagents and energy-intensive processes, reducing costs, while batch techniques like boiling and filtration enable scalability14. The approach eliminates toxic solvents, aligns with circular economy principles, and enhances nanoparticle stability via phytochemical capping13. Challenges include metabolite variability affecting standardization14, purification complexities, and the need for optimized reaction conditions13.
Several Nigerian plants have shown potential as molluscicidal agents. For instance, crude extracts of Annona squamosa, Alstonia congensis, Calotropis procera, and Morinda lucida, as well as nanoparticles derived from some of them, have demonstrated molluscicidal activities15,19,20. Significantly, these plant-based nanoparticles exhibited enhanced activity compared to the original crude extracts15. One such promising plant is S. alata, a medicinal species renowned for its bioactive compounds, including flavonoids, anthraquinones, and phenolics, which exhibit antimicrobial properties21. Although native to the Americas, S. alata is now naturalized and widely distributed across tropical regions, including West Africa from Senegal to Nigeria22. In these areas, it occurs as a common spontaneous or cultivated plant in villages, wastelands, forest clearings, and residential areas. Within Nigeria, it inhabits both rainforest and savanna ecosystems, spanning southern and northern regions23,24.
Although S. alata is traditionally used for multiple ailments and its extracts have demonstrated significant pharmacological activities (antibacterial, antifungal, antiviral, anticancer, antidiabetic, antilipogenic, etc.)25, and mosquitocidal (larvicidal) effects26, the molluscicidal efficacy of both its crude extracts and green-synthesized nanoparticles derived from the plant has not been explored. Based on reports linking snail mortality to chemical-induced oxidative stress and protein profile alterations27,28, we hypothesized that S. alata extracts and their green-synthesized nanoparticles act via similar mechanisms. We tested this by evaluating oxidative stress markers and protein expression in exposed L. natalensis. We selected L. natalensis for molluscicidal evaluation due to its established role as the key intermediate host for F. gigantica in sub-Saharan Africa, where fascioliasis represents a major health and veterinary challenge. This species was also the most abundant snail found at our study site, and our findings may provide insights applicable to other trematode-transmitting snail species.
This study evaluates the molluscicidal activities of ethanolic extracts of S. alata and its green-synthesized AgNPs on adult L. natalensis and its eggs. The study explores the efficacy of these treatments by assessing mortality rates, antioxidant enzyme activity, and protein expression patterns in exposed snails. The findings aim to contribute to the development of alternative, eco-friendly molluscicidal agents for controlling fascioliasis in endemic regions.
Materials and methods
Plant collection and identification
S. alata leaves were selected for molluscicidal testing based on the established bioactivity against mosquito vectors as reported by Karthiyayni et al.26,29. Fresh leaves of S. alata were collected within the Ondo city metropolis, Ondo State, Nigeria. The leaves were washed and air-dried at room temperature. Fresh samples were subsequently taken to the University of Benin Herbarium, where they were identified and authenticated by Dr. Emmanuel I. Aigbokhan. The voucher specimen was deposited and linked to an existing voucher number (UBH-S556).
Preparation of extract
The dried leaves were pulverised into a fine powder using a commercial grinding machine. The powdered material (400 g) was soaked in 80% of absolute ethanol (Molychem, India) in glass jars and shaken manually for 7 days. Eighty percentage (80%) ethanol was chosen for S. alata extraction due to its proven efficiency in extracting molluscicidal bioactive compounds, offering optimal safety and evaporative properties19. Its compatibility with green synthesis facilitated silver ion reduction and nanoparticle stabilization15as confirmed by UV–vis spectroscopy. After this period, the mixture was filtered using muslin cloth, and the solvent was evaporated under reduced pressure using a rotary evaporator at 40 °C. The resulting extract was freeze-dried and stored at 4 °C until further use.
Habitat and collection of snails
Adult L. natalensis snails were collected from a man-made drainage at Oduduwa Street in Ondo city, located at coordinates 7° 04′ 50.4″ N, 49° 37′ 6″ E. The site is situated behind a church and a few kilometres from Adeyemi Federal University of Education, Ondo, Nigeria. The snails were transported to the Seeding Laboratory at the University of Medical Sciences, Ondo, using aerated transparent containers.
Snail identification
L. natalensis is a freshwater snail characterized by a right-coiled (dextral) shell with an elongated ovoid shape, typically black or brown. The aperture height covers approximately 3/4 of the shell height, with a thin, sharp lip and a closed umbilicus. The shell dimensions range from 5.75 to 7 mm in width and 4.6–19.2 mm in height. The snails were identified by an experienced malacologist in the Department of Biosciences and Biotechnology, University of Medical Sciences, Ondo. Before experimentation, the snails were acclimatized for at least 24 h.
Laboratory maintenance and acclimatization of snails
In the laboratory, the snails were housed in a plastic container covered with nets. They were fed daily with dried blanched pawpaw (Carica papaya) leaves, and the water in the containers was replaced every 24–48 h. The snails were allowed to lay eggs, and the eggs were collected for ovicidal assays before the snails were exposed to nanoparticles.
Synthesis of silver nanoparticles (AgNPs)
Sixty milligrams (60 mg) of S. alata extract was dissolved in 30 ml of distilled water and filtered three times to obtain a clear filtrate. Ten millilitres of 10 mM AgNO3 solution (AgNO3, 99.8% purity; Molychem, India) was added to 4 ml of the filtrate in a beaker, making a 7.14 mM final concentration of AgNO3 solution. The mixture was stirred continuously at 70–80 °C for 1 h12. The reduction of Ag+ to Ag⁰ was indicated by a colour change from colourless to dark brown. The formation of AgNPs was confirmed using UV-visible spectroscopy. The AgNP suspension was centrifuged at 8000 rpm for 20 min, and the pellets were washed three times with distilled water to remove residual silver ions and plant extract. The purified AgNPs were then lyophilized.
Characterization of AgNPs
X-ray diffraction (XRD)
XRD analysis was performed to determine the crystalline nature of the AgNPs using a Shimadzu XDS 2400 H diffractometer with Cu Kα radiation (λ = 1.5405 Å). The instrument was operated at 40 kV and 30 mA, with a graphite monochromator and an automatic divergence slit. Samples were placed in a Lucite holder on the goniometer, and XRD patterns were recorded in the 2θ range of 0°–60° with a step size of 0.017° and a counting time of 14 s per step.
Fourier transform infrared spectroscopy (FTIR)
FTIR analysis was conducted to identify functional groups present in the AgNPs. Lyophilized AgNPs were mixed with potassium bromide (KBr) in a 1:100 ratio and pressed into tablets. The spectra were recorded using a Varian 660 MidIR Dual MCT/DTGS spectrometer with an ATR detector, covering a frequency range of 4000–500 cm−1 at a resolution of 4 cm−1 and 200 scans per sample.
Scanning electron microscopy (SEM) and energy dispersive X-ray spectroscopy (SEM-EDX)
SEM-EDX analysis was performed using a Hitachi SU 3500 scanning microscope to examine the morphology and elemental composition of the AgNPs. Samples were prepared by cleaning, stabilizing, rinsing, dehydrating, drying, mounting, and coating with a 20 nm gold layer to enhance conductivity. EDX analysis was conducted to detect elements with atomic numbers > 4, including light elements such as carbon, nitrogen, and oxygen.
Preparation of Senna alata leaf extract and silver nanoparticles (AgNPs).
A 100 mg of semisolid S. alata leaf extract was dissolved in 20 mL of distilled water using a magnetic stirrer to obtain a 5.0 mg/mL stock solution. Serial dilutions were prepared to obtain working concentrations of 5.0, 2.5, 1.25, 0.63, and 0.313 mg/mL. Similarly, the working concentrations of S. alata-AgNPs (SA AgNPs) were prepared from the stock solution, followed by serial dilutions to obtain concentrations of 2.5, 1.25, 0.625, 0.3125, and 0.1625 mg/mL. Distilled water served as the control in all assays.
Molluscicidal testing
Adult L. natalensis snails were exposed to treatments in accordance with the previously reported molluscicide testing protocol30. After acclimatization, adult snails were exposed to varying concentrations of S. alata extracts (5.0, 2.5, 1.25, 0.63, and 0.313 mg/mL) and AgNPs (2.5, 1.25, 0.625, 0.3125, and 0.1625 mg/mL) to assess molluscicidal activity. Five adult snails were placed in 5 mL of each concentration, with mortality assessed after 24 h for plant extracts and within 5 to 40 min for AgNPs. The experiment was conducted in duplicate, with distilled water as the negative control. Suspected dead snails were transferred to fresh distilled water to confirm irreversible immobilization (no movement after prodding with a blunt probe). Concentrations were selected based on preliminary dose-response assays. The divergent concentration ranges for crude extracts (5.0–0.313 mg/mL) and AgNPs (2.5–0.1625 mg/mL) reflect the 30-fold higher potency of AgNPs, as established in preliminary screenings. Mortality was calculated as:
Molluscicidal Activity of S. alata extracts and AgNPs on L. natalensis egg masses
Egg masses were laid on a clear polythene bag lining the snail culture container, carefully removed, and placed in distilled water with the surface facing the water to promote embryonic development. To evaluate the impact of S. alata ethanolic extracts and AgNPs on embryonic development, 2–3 egg masses (each containing 8–10 embryos) were exposed to extract concentrations of 5.0, 2.5, 1.25, 0.63, and 0.313 mg/mL and AgNP concentrations of 2.5, 1.25, 0.63, 0.313, and 0.163 mg/mL for 6 days. Embryo development was assessed via photomicrography at ×40 magnification.
Snail tissue processing and homogenization
The soft tissues of exposed and control snails were extracted by crushing the shells using two slides. The tissues were weighed (1 g tissue per 9 ml of 0.1 M phosphate buffer saline) and homogenized using a glass Dounce homogenizer. The homogenates were centrifuged at 3000 rpm for 25 min, and the supernatants were stored at − 20 °C.
Protein Estimation using the BCA method
Protein concentrations in the homogenized snail tissues exposed to sub-lethal concentrations of SA-AgNPs were determined using the Bicinchoninic Acid (BCA) assay. This method relies on the reduction of Cu2+ to Cu+ by proteins, which then react with BCA to form a purple complex. Protein estimation was determined using BCA kit (E-BC-K318-M, Elabscience, China) according to the manufacturer’s instructions. Absorbance was measured at 562 nm using a microplate reader.
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE)
SDS-PAGE was conducted on snail samples post-exposure following a modified protocol by Oyeyemi et al.18. Adult Lymnaea snails were exposed to S. alata-based AgNPs for 25 min to prevent mortality and obtain homogenates for analysis. Briefly, 12% SDS-PAGE gels were prepared, with 10 µg of crushed snail samples loaded per well in duplicate for Coomassie staining. Protein separation was performed using the Servicebio SVE-2 Vertical Electrophoresis System, initially at 60 V, then increased to 120 V. The gel was stained with an adequate amount of Coomassie stain and shaken overnight for band visualization.
Antioxidant enzyme assay using snail homogenate
Superoxide dismutase (SOD) activity
SOD activity was measured using the Misra and Fridovich method31. A 100 µL aliquot of the diluted sample was mixed with 1250 µL of 0.05 M carbonate buffer (pH 10.2), followed by the addition of 30 µL freshly prepared adrenaline. Absorbance was recorded immediately at 450 nm and monitored every 30 s for 150 s.
Lipid peroxidation
Lipid peroxidation levels were determined by quantifying thiobarbituric acid reactive substances (TBARS) using the Rice-Evans et al. Method32. This assay relies on the reaction of 2-thiobarbituric acid (TBA) with malondialdehyde (MDA), a lipid peroxidation byproduct, forming a pink chromophore that absorbs at 532 nm. A 0.2 mL sample was mixed with 1.6 mL TRIS-KCl buffer, followed by 0.5 mL of 30% TCA and 0.5 mL of 0.75% TBA. The mixture was heated at 80 °C for 45 min, cooled in ice, and centrifuged at 3000 rpm for 15 min. The absorbance of the clear pink supernatant was measured at 532 nm against a distilled water blank.
Catalase (CAT) activity
Catalase activity was assessed using Claiborne’s method33. In a 96-well microplate, 50 µL hydrogen peroxide (H₂O₂) was added in triplicate, followed by 90 µL phosphate buffer and 10 µL sample. A distilled water blank (10 µL) was also included in triplicate. The mixture was inverted to mix and placed in a microplate reader, with absorbance changes measured at 240 nm every 15 s for 150 s.
Statistical analysis
Data were recorded in Microsoft Excel 2010 and analyzed using SPSS version 20 (IBM Corp., Armonk, NY, USA) and Excel. Mean values and standard errors were computed for replicated data. One-way analysis of variance (ANOVA) was used to determine statistical significance between treatment groups, followed by Tukey’s test for multiple comparisons. A p-value < 0.05 was considered statistically significant.
Results
Synthesis of silver nanoparticles
The synthesis of silver nanoparticles (AgNPs) began upon adding S. alata ethanolic leaf extract to a 10 mM silver nitrate (AgNO₃) solution. The mixture’s colour transitioned progressively from colourless to dark-brown, indicating the formation of AgNPs. Further confirmation was obtained through UV-visible spectroscopy, which revealed an absorption peak at approximately 362 nm in the aqueous suspension of S. alata-synthesized silver nanoparticles.
Fourier transformed infrared spectroscopy (FTIR)
The FTIR spectrum of green-synthesized silver nanoparticles using S. alata shows absorption peaks at 3098.26, 3016.39, 2600.85, 1851.23, 1548.13, 1472.32, 900.26, 875.01, and 805.42 cm−1. Key bands at 3016.39 and 3098.26 cm−1 indicate aromatic C–H stretching, while 1472.32 cm⁻¹ confirms aromaticity. Overlapping bands suggest O-H stretching of phenolic compounds. Peaks at 805.42, 875.01, and 900.26 cm⁻¹ represent aromatic C–H out-of-plane bending of olefinic substituents (Fig. 1). No C=O bonding was detected.
X-ray diffraction (XRD) analysis
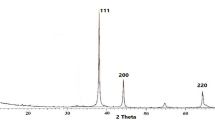
The XRD pattern of green-synthesized silver nanoparticles using S. alata (Fig. 2) displays distinct peaks at 2θ values of 25.50, 30.50, 32.00, 35.50, 38.00, 41.00, 44.00, 45.00, 51.50, and 57.00, corresponding to the (012), (104), (006), (110), (113), (202), (024), (018), (116), and (211) planes, respectively.
SEM/EDX analysis
SEM analysis revealed the surface topography of AgNPs with nanometer-scale resolution, while backscattered electrons imaging provided elemental contrast. The SEM image (Fig. 3) shows SA-AgNPs with an irregular, dense texture, large pores, and particles, averaging 20.32 nm in size. EDX confirmed silver (47.04%) as the major element, alongside carbon (20.71%), oxygen (28.53%), chlorine (1.31%), nitrogen (1.91%), copper (0.14%), and iron (0.36%). The AgNPs exhibited an optical absorption peak near 4 eV due to surface plasmon resonance.
Molluscicidal activities against adult snails
The molluscicidal activity of S. alata crude extracts is shown in Fig. 4. Lower concentrations (0.313–1.25 mg/ml) caused weak mortality (10–20%), while 5 mg/ml induced 60% mortality. Overall, there was no significant difference in snail mortality across different crude extract concentrations (p = 0.063). S. alata silver nanoparticles exhibited strong molluscicidal effects, causing snail mortality within 20 min at 0.63–2.5 mg/ml and within 30 min at 0.163–0.313 mg/ml. Complete mortality occurred within 40 min at all tested concentrations (Table 1).
Molluscicidal activities against L. natalensis ova
The crude extract of S. alata exhibited weak ovicidal activity, with the highest concentration (5 mg/ml) significantly inhibiting embryonic growth and preventing development beyond the blastula stage. Lower concentrations had minimal effect on the hippo stage, though some rupturing was observed at 2.5 mg/ml. In contrast, S. alata-mediated green-synthesized nanoparticles strongly inhibited embryonic development at all concentrations, halting growth at the blastula stage (Fig. 5).
Ovicidal activities of S. alata leaf extract (A–F) and its green-synthesized silver nanoparticles (AgNPs) (G–L) against L. natalensis eggs (Magnification x40): (A) 5 mg/ml; (B) 2.5 mg/ml; (C) 1.25 mg/ml; (D) 0.63 mg/ml; (E) 0.313 mg/ml; (F) Negative control; (G) 2.5 mg/ml; (H) 1.25 mg/ml; (I) 0.63 mg/ml; (J) 0.313 mg/ml; (K) 0.163 mg/ml; (L) Negative control for S. alata AgNPs.
Effects of plant extracts and green-synthesized nanoparticles on markers of oxidative stress
Table 2 presents antioxidant parameters in adult Lymnaea snails exposed to S. alata extract for 24 h. The level of LPO, a marker of oxidative stress, was significantly increased compared to the negative control group only at the 2.5 mg/ml (p < 0.05) of the extract-treated groups, whereas it was significantly (p < 0.01) increased across all SA AgNPs-treated groups. Similarly, the activity of SOD increased in the extract-treated groups (p > 0.05) control group while it was significantly decreased (p < 0.05) in the SA-AgNps treated groups. Catalase activity, however, was significantly decreased in the extract-treated groups while it was increased in the Np-treated groups (Tables 2 and 3).
Protein expression patterns in relation to exposure to plant extract and SA-AgNPs
Protein expression patterns were similar between S. alata-exposed snails and the negative control, though proteins at 55 kDa and 43 kDa were more pronounced in extract-treated snails. In contrast, snails exposed to S. alata-mediated nanoparticles expressed several proteins absent in the control, including those at 17–26, 34, 43, 55, 72, and 95 kDa (Fig. 6). Expression patterns remained consistent across all nanoparticle concentrations.
SDS-PAGE showing total protein bands of tissues of L. natalensis.. Mk- Marker. Lane 1: 5.0 mg/ml exposed to S. alata extract. Lane 2: 2.5 mg/ml exposed to S. alata extract. Lane 3: 1.25 mg/ml exposed to S. alata extract. Lane 4: 0.625 mg/ml exposed to S. alata extract. Lane 5: 0.3125 mg/ml exposed to S. alata extract. Lane 6: Control for S. alata extract. Lane 7: 0.024 mg/ml exposed to S. alata AgNPs. Lane 8: 0.012 mg/ml exposed to S. alata AgNPs. Lane 9: 0.006 mg/ml exposed to S. alata AgNPs. Lane 10: 0.003 mg/ml exposed to S. alata AgNPs.
Discussion
Plant-derived products have been widely recommended as promising alternatives for the development of molluscicidal agents19,34. Although plant-based formulations are generally regarded as environmentally sustainable with minimal toxicity risks, they are not entirely free from challenges commonly associated with conventional drugs, such as poor biodistribution, instability, and limited shelf life35. In many cases, achieving the desired therapeutic or molluscicidal effects requires significantly increasing the concentration of plant extracts, which may impact their practicality and efficiency36. These limitations highlight the need for innovative approaches, such as nanotechnology, to enhance the stability, potency, and targeted delivery of plant bioactivities, thereby improving their overall effectiveness as molluscicidal agents.
The primary phytochemicals in S. alata include saponins, phenolic acids, flavonoids, tannins, and alkaloids37. The presence of phenolic acids and O-H stretching in the plant extract used for green synthesis may contribute to synergistic effects, enhancing the nanoparticles’ overall properties and biological activity. The band at 900.26 cm−1 corresponds to the bending vibrations of –C-H groups38, indicating that the FTIR spectrum of S. alata-based AgNPs supports the role of S. alata phytochemicals in both the reduction of Ag⁺ ions and the stabilization of the green-synthesized nanoparticles39.
The well-defined and intense XRD peaks indicate the crystalline nature of the synthesized nanoparticles, confirming their structural integrity. Similar findings have been reported for green-synthesized Alstonia scholaris and A. congensis nanoparticles, as documented by Rajasekar et al.39 and Oyeyemi et al.15, respectively. Additionally, the presence of elements such as nitrogen (N), chlorine (Cl), oxygen (O), copper (Cu), and iron (Fe) in the nanoparticle formulation suggests their origin from the natural phytoconstituents of the plant utilized in the green synthesis process. Notably, Cl, Cu, and Fe serve as essential micronutrients for plant metabolism, further supporting their inclusion in the formulation. Moreover, the scanning electron microscopy (SEM) analysis reveals nanoparticle aggregation, which can be attributed to the inherent structural and electronic properties of the bioactive phytochemicals involved in the synthesis, as previously highlighted by Ahmed et al.40.
Our study revealed weak molluscicidal activities of S. alata ethanolic extract on the adult snails and the developing embryos, in contrast to the plant-mediated silver nanoparticles. The impressive molluscicidal and ovicidal properties of the green-synthesized nanoparticles compared to the parent plant have been earlier reported15. The green-synthesized nanoparticles offer a distinct advantage over the crude extract of the source plant, particularly in their efficacy at lower doses, which aligns with sustainable environmental practices and underscores the practicality of nanotechnology in eco-friendly applications15. The enhanced inhibitory effect of our green-synthesized formulation on embryonic development may be attributed to the improved bioavailability and targeted delivery of bioactive plant compounds, allowing more effective interaction with developing embryos encased within gelatinous egg masses41. The nanoscale transformation of the plant-derived material potentially alters its chemical composition or increases the abundance of specific functional groups, leading to changes in its physicochemical properties. In particular, modifications in lipophilicity could enhance the nanoparticle’s ability to penetrate egg membranes, thereby improving its bioactivity and overall effectiveness41.
At a significantly reduced concentration, more than 30 times lower than the green-synthesized nanoparticles (0.167 mg/ml), complete mortality (100%) of adult L. natalensis snails was achieved within just 40 min. In contrast, the highest tested concentration of the crude plant extract (5 mg/ml) was only able to eliminate 60% of the snails after a prolonged 24-hour exposure period. This remarkable and rapid molluscicidal action of the nanoparticles suggests an acute toxic effect, which is highly advantageous as it minimizes the likelihood of adaptive escape responses that often occur when snails are exposed to sub-lethal doses of plant extracts19,30. The nanoscale formulation may have enhanced the bioavailability and penetration efficiency of the active phytochemicals, facilitating a more immediate and potent interaction with the snail’s physiological systems. Additionally, the increased surface area-to-volume ratio of the nanoparticles could have accelerated their mode of action42, ensuring swift toxicity to the snails and reducing the chance of resistance development in targeted mollusk populations. This highlights the potential of nanotechnology as an effective and environmentally sustainable alternative to conventional plant-based molluscicides.
Analysis of SDS-PAGE protein banding patterns in the tissue-soluble protein extracts of L. natalensis revealed that snails exposed to green-synthesized nanoparticles exhibited a higher number of protein bands compared to the control group. This suggests that nanoparticle exposure may have induced notable changes in protein expression or metabolism. Similar observations have been reported in previous studies; for instance, Bakry et al.28 documented qualitative and quantitative alterations in the protein profiles of Helisoma duryi snails following exposure to deltamethrin and malathion. Additionally, Biomphalaria alexandrina snails subjected to a formulated mixture of lambda-cyhalothrin and acetamiprid insecticides displayed an increased number of unique protein bands relative to unexposed controls43.
The observed increase in protein banding in our study could be attributed to various biological mechanisms. One possible explanation is that nanoparticle exposure induced genetic modifications, leading to alterations in DNA that subsequently influenced protein synthesis and expression patterns44. Alternatively, the appearance of additional protein bands may be due to the synthesis of stress-related proteins or the disruption of polypeptide metabolism as the snails attempt to counteract nanoparticle-induced physiological stress45. The shift in protein banding patterns highlights the potential molecular impact of nanoparticle exposure, suggesting that these nanomaterials may trigger adaptive or stress-responsive pathways within the exposed snails. Further studies on the functional roles of these altered proteins could provide deeper insights into the biochemical and molecular interactions between green-synthesized nanoparticles and aquatic organisms.
The elimination of reactive oxygen species (ROS), such as superoxide anion radicals, typically occurs through the enzymatic dismutation activity of superoxide dismutase (SOD), which converts them into hydrogen peroxide. In our study, exposure to green-synthesized nanoparticles significantly activated the antioxidant enzyme catalase, a crucial component of the cellular defense system against oxidative stress. This increase in catalase activity in L. natalensis aligns with findings from previous research, which reported a similar oxidative stress response in snails exposed to metal pollutants27. This suggests that the nanoparticles exert oxidative stress-inducing effects, prompting the snail’s defense mechanisms to upregulate catalase activity to mitigate potential damage.
Furthermore, the nanoparticle-induced oxidative stress was evident through elevated lipid peroxidation (LPO) levels and a concurrent reduction in SOD activity. This pattern indicates a disruption in the oxidative balance, likely driven by the enhanced generation of ROS. In contrast, snails exposed to the crude plant extract exhibited an increase in SOD activity, which may have been a compensatory response to LPO-induced oxidative stress46. The detoxification process of the plant extract within the snail’s system could have resulted in the generation of superoxide anion radicals as metabolic byproducts, further activating antioxidant pathways45. These findings highlight the potential of green-synthesized nanoparticles to modulate oxidative stress responses, underscoring their bioactivity and interaction with the physiological defense mechanisms of exposed organisms.
Our study demonstrates S. alata-AgNPs’ molluscicidal activity while highlighting key limitations. Nanoparticle characterization would benefit from dynamic light scattering (size distribution/polydispersity) and zeta potential (stability/surface charge) analyses. While FTIR characterized AgNPs, crude extract analysis would clarify phytochemical profiles. Mechanistically, despite observed oxidative stress, precise cellular interactions and individual phytochemical contributions require further study. Environmental assessments of nanoparticle persistence and non-target effects remain outstanding. While chemical molluscicide was not included as a positive control, mortality kinetics and oxidative stress data align with previous studies39,46. Future work will validate efficacy against niclosamide. These gaps present critical opportunities for future research to refine characterization, elucidate mechanisms, and evaluate ecological impacts, thus strengthening both scientific understanding and practical applications of plant-derived nanomolluscicides.
Conclusion
This study demonstrates the potent molluscicidal activities of green-synthesized S. alata-based silver nanoparticles (AgNPs) against L. natalensis, a key intermediate host of fascioliasis. Compared to crude S. alata extracts, the AgNPs exhibited significantly enhanced efficacy at lower concentrations, achieving complete mortality within a short exposure period. The nanoparticles also disrupted embryonic development and induced oxidative stress responses in snails, as evidenced by altered antioxidant enzyme activities and protein expression patterns. These findings underscore the potential of S. alata-mediated AgNPs as an eco-friendly alternative to synthetic molluscicides for controlling fascioliasis transmission. Further studies on field applicability, toxicity, and long-term environmental impact are warranted to advance their use in integrated parasite control strategies.
Data availability
Data associated with the study will be made available by the corresponding author on reasonable request.
References
Liu, L. et al. Global, regional and National disease burden of food-borne trematodiases: projections to 2030 based on the global burden of disease study 2021. Infect. Dis. Poverty. 13, 95. https://doi.org/10.1186/s40249-024-01265-6 (2024).
Tidman, R., Kanankege, K. S., Bangert, M. & Abela-Ridder, B. Global prevalence of four neglected foodborne trematodes targeted for control by WHO: a scoping review to highlight the gaps. PLoS Negl. Trop. Dis. 17 (3), e0011073. https://doi.org/10.1371/journal.pntd.0011073 (2023).
Global Burden of Disease Collaborative Network. Global Burden of Disease Study 2019 Results. Seattle, United States: Institute for Health Metrics and Evaluation (IHME), 2020 (2019). https://gbd2019.healthdata.org/gbdresults/ (accessed 9 Jun 2025).
Nukeri, S. et al. Potential hybridization of Fasciola hepatica and F. gigantica in Africa—A scoping review. Pathogens. 11, 1303. https://doi.org/10.3390/pathogens11111303 (2022).
Zhou, P., Chen, N., Zhang, R. L., Lin, R. Q. & Zhu, X. Q. Food-borne parasitic zoonoses in china: perspective for control. Trends Parasitol. 24 (4), 190–196. https://doi.org/10.1016/j.pt.2008.01.001 (2008).
Sabourin, E., Alda, P., Vázquez, A., Hurtrez-Boussès, S. & Vittecoq, M. Impact of human activities on fasciolosis transmission. Trends Parasitol. 34 (10), 891–903. https://doi.org/10.1016/j.pt.2018.08.004 (2018).
Mas-Coma, S., Bargues, M. D. & Valero, M. A. Fascioliasis and other plant-borne trematode zoonoses. Int. J. Parasitol. 35 (11–12), 1255–1278. https://doi.org/10.1016/j.ijpara.2005.07.010 (2005).
Olaechea, F., Lovera, V., Larroza, M., Raffo, F. & Cabrera, R. Resistance of Fasciola hepatica against triclabendazole in cattle in patagonia (Argentina). Vet. Parasitol. 178 (3–4), 364–366. https://doi.org/10.1016/j.vetpar.2010.12.047 (2011).
Calumpang, S. M. F., Medina, M. J. B., Tejada, A. W. & Medina, J. R. Environmental impact of two molluscicides: niclosamide and metaldehyde in a rice paddy ecosystem. Bull. Environ. Contam. Toxicol. 55, 494–501. https://doi.org/10.1007/BF00196027 (1995).
Stevens, M. M., Doran, G. & Mo, J. Efficacy and environmental fate of copper sulphate applied to Australian rice fields for control of the aquatic snail Isidorella newcombi. Crop Prot. 63, 48–56. https://doi.org/10.1016/j.cropro.2014.05.004 (2014).
Al-Dbass et al. Biogenic silver nanoparticles from two varieties of Agaricus bisporus and their antibacterial activity. Molecules. 27 (21), 7656. https://doi.org/10.3390/molecules27217656 (2022).
Okeke, I. J., Oyeyemi, O. T. & Morenikeji, O. A. Ovicidal and miracicidal activities of Calotropis procera and its green-synthesized nanotized derivative: a quest for new antifasciola agents. Acta Trop. 236, 106700. https://doi.org/10.1016/j.actatropica.2022.106700 (2022).
Bhat, R. S., Al-Dbass, A. M., Khayyat, A. I. A. & Al-Daihan, S. Utilizing biomolecule-rich citrus fruit waste as a medium for the eco-friendly Preparation of silver nanoparticles with antimicrobial properties. Inorganics. 12 (7), 180. https://doi.org/10.3390/inorganics12070180 (2024).
Daghestani, M. et al. Bactericidal and cytotoxic properties of green synthesized nanosilver using Rosmarinus officinalis leaves. Green. Process. Synth. 9 (1), 230–236. https://doi.org/10.1515/gps-2020-0025 (2020).
Oyeyemi, O. T., Babalola, B. M., Akinmulero, T. C., Adesida, P. A. & Oyeyemi, I. T. Molluscicidal activities of green-synthesized Alstonia congensis silver nanoparticles. Asian Pac. J. Trop. Biomed. 13 (4), 176–183. https://doi.org/10.4103/2221-1691.374234 (2023).
Almukaynizi, F. et al. Cytotoxicity of green-synthesized silver nanoparticles by Adansonia digitata fruit extract against HTC116 and SW480 human colon cancer cell lines. Green. Process. Synth. 11 (1), 411–422. https://doi.org/10.1515/gps-2022-0031 (2022).
Bhat, R. S., Almusallam, J., Al Daihan, S. & Al-Dbass, A. Biosynthesis of silver nanoparticles using Azadirachta indica leaves: characterisation and impact on Staphylococcus aureus growth and glutathione-S-transferase activity. IET Nanobiotechnol. 13, 498–502. https://doi.org/10.1049/iet-nbt.2018.5133 (2019).
Oyeyemi, O. T., Corsini, C. A., Gonçalves, G., Borges, W. C. & Grenfell, R. R. Q. Evaluation of immunodiagnostic potential of schistosomula crude antigen (SCA) in Schistosoma mansoni- infected human population. Sci. Rep. 11, 10530. https://doi.org/10.1038/s41598-021-89929-3 (2021).
Oyeyemi, O. T., Eneh, C. G. & Okeke, I. J. Molluscicidal activities of ethanolic extracts of Calotropis procera and Morinda Lucida against Lymnaea natalensis. Acta Biol. Szeged. 66 (1), 69–73. https://doi.org/10.14232/abs.2022.1.69-73 (2022).
Hussein, A. A. A. et al. The molluscicidal activity of green synthesized copper oxide-based Annona squamosa seed extract nanoparticles on the feeding behavior, biochemical, molecular, and immunohistochemical alterations of Biomphalaria Alexandrina snails. Biol. Trace Elem. Res. 202 (5), 2327–2337. https://doi.org/10.1007/s12011-023-03823-9 (2024).
Liu, A., Xu, L., Zou, Z. & Yang, S. Studies on chemical constituents from leaves of Cassia alata. Chin. J. Chin. Mater. Med. 34, 861–863 (2009).
Irvine, F. R. Woody Plants of Ghana with Special Reference To their Uses, 143–144 (Oxford University Press, 1961).
Adjanohoun, E. et al. Traditional medicine and pharmacopoeia. Contribution to ethnobotanical floristic studies in Western nigeria. nigeria. pub. Organization of African unity. Sci. Tech. Res., 420 (1991).
Elujoba, A. A. & Ogunti, E. O. Pharmacopoeial and biological standardization of Cassia alata and Cassia podocarpa with reference to Senna. Glimpses Plant Res. XI: 469 – 79 (1993).
Oladeji, O. S., Adelowo, F. E., Oluyori, A. P. & Bankole, D. T. Ethnobotanical description and biological activities of Senna alata. Evid. Based Complement. Alternat Med. 2020, 2580259. https://doi.org/10.1155/2020/2580259 (2020).
Edwin, U. P., Nyiutaha, I. G., Essien, A. E., Nnamdi, O. K. & Sunday, E. M. Larvicidal effect of aqueous and ethanolic extracts of Senna alata on Anopheles gambiae, Culex quinquefasciatus and Aedes aegypti. Pak. J. Pharm. Sci. 26 (3), 561–566 (2013).
Basopo, N. & Ngabaza, T. Pollutants snails, oxidative stress, organophosphates, metals. Adv. Biol. Chem. 5, 225–233. https://doi.org/10.4236/abc.2015.56019 (2015).
Bakry, F. A., Hasheesh, W. S. & Hamdi, S. A. H. Biological, biochemical, and molecular parameters of Helisoma duryi snails exposed to the pesticides malathion and deltamethrin. Pestic Biochem. Physiol. 101, 86–92. https://doi.org/10.1016/j.pestbp.2011.08.004 (2011).
Karthiyayni, N., Pravin, Y., Palanikumar, M., Mohanraj, R. S. & Sujatha, P. S. Mosquitocidal efficacies of the medicinal plant Senna alata methanolic leaf extract against dengue and Zika virus vector Aedes aegypti. Int. J. Pharm. Biol. Sci. 9 (3), 244–255. https://doi.org/10.22376/ijpbs.2018.9.3.b244-255 (2018).
Adenusi, A. A. & Odaibo, A. B. Laboratory assessment of molluscicidal activity of crude aqueous and ethanolic extracts of Dalbergia sissoo plant parts against Biomphalaria Pfeifferi. Travel Med. Infect. Dis. 6 (4), 219–227. https://doi.org/10.1016/j.tmaid.2007.12.004 (2008).
Misra, H. P. & Fridovich, I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 247 (10), 3170–3175 (1972).
Rice-Evans, C., Omorphos, S. & Baysal, E. Sickle cell membranes and oxidative damage. Biochem. J. 237 (1), 265 (1986).
Claiborne, A. Catalase activity. In CRC Handbook of Methods for Oxygen Radical Research (ed Greenwald, R. A.) 283–284 (CRC, 1985).
Salawu, O. T. & Odaibo, A. B. The molluscicidal effects of Hyptis suaveolens on different stages of Bulinus globosus in the laboratory. Afr. J. Biotechnol. 10 (50), 10241–10247. https://doi.org/10.5897/AJB10.415 (2011).
Gunasekaran, T., Haile, T., Nigusse, T. & Dhanaraju, M. D. Nanotechnology: an effective tool for enhancing bioavailability and bioactivity of phytomedicine. Asian Pac. J. Trop. Biomed. 4 (Suppl 1). https://doi.org/10.12980/APJTB.4.2014C980 (2014). S1-7.
Adetunji, V. O. & Salawu, O. T. Efficacy of ethanolic leaf extracts of Carica papaya and Terminalia catappa as molluscicides against the snail intermediate hosts of schistosomiasis. J. Med. Plants Res. 4 (22), 2348–2352. https://doi.org/10.5897/JMPR10.468 (2010).
Onyegeme-Okerenta, B. M., Nwosu, T. & Wegwu, M. O. Proximate and phytochemical composition of leaf extract of Senna alata (L.) Roxb. J. Pharmacogn. Phytochem. 6 (2), 320–3232 (2017).
Bagherzade, G., Tavakoli, M. M. & Namaei, M. H. Green synthesis of silver nanoparticles using aqueous extract of saffron (Crocus sativus L.) wastages and its antibacterial activity against six bacteria. Asian Pac. J. Trop. Biomed. 7 (3), 227–233. https://doi.org/10.1016/j.apjtb.2016.12.014 (2017).
Rajasekar, R., Samuel, M., Edison, T. N. J. I. & Raman, N. Sustainable synthesis of silver nanoparticles using Alstonia scholaris for enhanced catalytic degradation of methylene blue. J. Mol. Struct. 1246, 131208. https://doi.org/10.1016/j.molstruc.2021.131208 (2021).
Ahmed, S., Ahmad, M., Swami, B. L. & Ikram, S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: A green expertise. J. Adv. Res. 7 (1), 17–28. https://doi.org/10.1016/j.jare.2015.02.007 (2016).
Omobhude, M. E., Morenikeji, O. A. & Oyeyemi, O. T. Molluscicidal activities of curcumin-nisin polylactic acid nanoparticle on Biomphalaria Pfeifferi. PLoS Negl. Trop. Dis. 11 (8), e0005855. https://doi.org/10.1371/journal.pntd.0005855 (2017).
Bantz, C. et al. The surface properties of nanoparticles determine the agglomeration state and the size of the particles under physiological conditions. Beilstein J. Nanotechnol. 5, 1774–1786. https://doi.org/10.3762/bjnano.5.188 (2014).
Ibrahim, A. M., Sayed, S. S. M. & Shalash, I. R. A. Toxicological assessment of lambda-cyhalothrin and Acetamiprid insecticides formulated mixture on hatchability rate, histological aspects, and protein electrophoretic pattern of Biomphalaria Alexandrina (Ehrenberg, 1831) snails. Environ. Sci. Pollut. Res. 25, 32582–32590. https://doi.org/10.1007/s11356-018-3238-x (2018).
El-Sayed, K. A. Effect of the plant Cupressus macrocarpa (Cupressaceae) on some hematological and biochemical parameters of Biomphalaria Alexandrina snails. J. Egypt. Soc. Parasitol. 36, 911–924 (2006).
El-Deeb, F. A. A. et al. Biomarkers of oxidative stress in Biomphalaria Alexandrina snails for assessing the effects of certain inorganic fertilizers. Molluscan Res. 37, 289–294. https://doi.org/10.1080/13235818.2017.1357783 (2017).
El-Gendy, K. S., Radwan, M. A. & Gad, A. F. In vivo evaluation of oxidative stress biomarkers in the land snail, Theba pisana exposed to copper-based pesticides. Chemosphere. 77, 339–344. https://doi.org/10.1016/j.chemosphere.2009.07.015 (2009).
Funding
No funding was made available for the study.
Author information
Authors and Affiliations
Contributions
O.T.O. conceptualized this study, supervised the study, wrote the original draft, and revised and edited the final draft. O.T.O. and I.T.O. developed the methodology. E.A.F., T.O.O., S.A.K., O.S.B., and I.T.O. were involved in data collection. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Oyeyemi, O.T., Fasomoyin, E.A., Oriade, T.O. et al. Molluscicidal activities of Senna alata silver nanoparticles against adult and egg stages of Lymnaea natalensis. Sci Rep 15, 25746 (2025). https://doi.org/10.1038/s41598-025-11771-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-11771-8