Abstract
In this study, waste tires (WTs) were chosen to prepare an adsorbent material for carbon dioxide (CO2) capture to reduce their huge amount of waste. To improve the CO2 selectivity, the WT powder was amine-modified using tetraethylenepentamine (TEPA) or polyethyleneimine at different loading levels [2.5, 5, 10, and 15% (w/w)]. The optimum condition to develop a high-performance CO2 adsorbent material was found to be the use of 10% (w/w) TEPA-modified WT (WT10T) with a CO2 flow rate of 70 mL min−1 under ambient temperature and atmospheric pressure. Based on the kinetic study of WT10T, Avrami’s model fitted well with the experimental data, suggesting both physisorption and chemisorption of the CO2. When desorbed at 60 °C under vacuum pressure, the WT10T showed a reusability of more than 10 successive adsorption cycles. Additionally, the adsorbed CO2 in WT10T could be directly converted to value-added chemicals or fuels, such as ethylene, methane, carbon monoxide, and hydrogen, via an electrochemical reaction. Accordingly, this study shows a challenging way on how to both utilize waste materials as adsorbents and reduce the WT level from rubber industries.
Similar content being viewed by others
Introduction
The increase in greenhouse gas (GHG) emissions, especially carbon dioxide (CO2), in the atmosphere leads to global climate change, which has become a serious issue worldwide today1. Excessive GHGs in the atmosphere are responsible for various environmental problems, such as a rise in the sea water level, an increase in the earth’s temperature, and an increasing number of ocean storms, floods, etc2. The 26th UN Climate Change Conference of the Parties (COP26) resulted in an agreement that requires countries to decrease their GHG emissions by reducing the use of fossil fuels and replacing it with clean energy by 20303,4. Currently, there are many useful methods to reduce or capture GHG emissions, such as renewable technologies5,6, energy saving technologies7, and carbon capture, utilization, and storage (CCUS)8,9,10. Of these, CCUS is one of the interesting technologies and is based on CO2 adsorption by various solid adsorbents, such as activated carbon11,12,13, zeolites14,15,16, sepiolite17, heteroatom-doped porous carbons18,19, porous organic polymers20, metal-organic frameworks (MOFs)21, reduced graphene oxide22, and recycled waste materials23,24,25,26. These strategies are also compatible with the Bio-Circular-Green Economic Model (BCG)27 to create economic and environmental sustainability and to reduce the waste from many industries28. However, after CO2 adsorption in the adsorbent material, it is economically and environmentally important to make value-added products from the CO2. The possible ways used to convert CO2 into valuable hydrocarbon (HC) compounds include thermochemical, photocatalytic, and electrochemical conversions29,30,31. These concepts are mainly focused on this study, as they are the part of the challenge of how to utilize waste materials as adsorbents and how to convert CO2 to value-added chemicals.
In this study, the recycling of waste tires (WTs) was used to develop an adsorbent for CO2 capture, since WTs are a non-biodegradable material, due to their complex structure after vulcanization, and the annual global production of WTs is currently around 1.4 billion pieces every year32. At present, WTs are mainly disposed of by the traditional methods of landfill, which requires a large space, or burning, which accumulates toxic air pollution33. Therefore, the use of recyclable WTs as an adsorbent is beneficial for reducing the huge amount of their waste.
Several researchers have focused on the transformation of WTs for use in various applications. For instance, WTs have been pyrolyzed and chemically modified to produce a magnetic pyrochar adsorbent for wastewater treatment34,35,36,37. Pyrolytic WT products have been prepared via impregnation as a catalyst for producing hydrogen (H2) and carbon nanotubes38,39,40,41,42,43. Pyrochar from WT pyrolysis was treated by chemical activation to prepare an electrode for energy storage44,45,46,47. Finally, activated carbon from WT pyrolysis was formed by physical and chemical activation as a solid adsorbent for CO2 capture48,49,50.
However, these techniques prepared the obtained materials through a thermal process that consumes a high amount of energy, and the process is complicated. To solve such problems, modifying or functionalizing a WT adsorbent with amines on its surface benefits the easy preparation of a solid adsorbent. Natural rubber (NR) latex foam with amine-modified silica51,52 or modified NR with amine-modified silica particles was found to enhance the CO2 adsorption53. Therefore, this study focused on amine-modified WTs. It is interesting to note that the WTs used in this study consisted of some additives, such as carbon black or silica, which could enhance the efficiency of CO2 adsorption54,55. In addition, the presence of vulcanization in WTs could improve the thermal properties of the material56. Although, amine-modified WTs have been studied previously as an absorbent in wastewater treatment57, their application for CO2 capture has not been demonstrated yet.
To reduce the emission level of GHGs, CO2 can also be converted into valuable HC compounds or fuel, such as methane (CH4), ethane (C2H6), carbon monoxide (CO), methanol, and ethanol. There are three possible routes for CO2 conversion to higher value products as follows: photocatalytic conversion, electrochemical reduction, and thermochemical conversion58,59,60,61,62,63,64. In this study, the electrochemical reduction of CO2 was examined in the conversion of CO2 to more reduced chemical species using electrical energy. The electrochemical reduction can support renewable energy exploitation to create an artificial carbon-neutral cycle. Also, it can be easily carried out in aqueous media at room temperature and atmospheric pressure.
Copper (II) oxide (CuO) and copper (I) oxide (Cu2O) are both used as catalysts in the conversion of CO2 due to their unique properties that facilitate certain chemical reactions. Their application in the CO2 conversion processes is motivated by their redox properties, high surface area, ability to chemisorb CO2 molecules, presence of active sites, involvement in catalytic cycles, and capability to control the selectivity65,66,67,68. These catalysts play a crucial role in developing sustainable methods for utilizing CO2 as a feedstock to produce valuable chemicals and fuels.
Accordingly, the aim of this study was to modify WT samples as an adsorbent material for CO2 capture. The WT samples were directly impregnated with tetraethylenepentamine (TEPA) or polyethyleneimine (PEI) to enhance the CO2 adsorption capacity. A series of adsorbents prepared with different amine types (PEI or TEPA) and amine loading levels [0–15% (w/w)] was investigated for their CO2 capture under ambient temperature and atmospheric pressure. The adsorption kinetics were explored by fitting three adsorption kinetic models (pseudo 1 st order, pseudo 2nd order, and Avramis’ models). The reusability of the selected TEPA-modified WT was evaluated over 10 successive adsorption-desorption cycles, while for practical application, the CO2 captured in the TEPA-modified WT was converted to value-added HCs via the electrochemical CO2 reduction reaction (CO2RR) using a CuO: Cu₂O catalyst with a 1:2 molar ratio.
Materials and methods
Materials
The WTs, mainly unusable tires from automobiles, were provided as a powder with an average particle size of 250 μm from the Union Commercial Development Co., Ltd, Thailand. The TEPA (189.3 g/mol), PEI (800 g/mol), Cu2O (98% purity analytical grade), and CuO (AR grade) were purchased from Sigma-Aldrich Co., Ltd. Absolute ethanol (99.99%), potassium bicarbonate (KHCO3; AR grade), and isopropyl alcohol (AR grade) were from Qrec Chemical Ltd., New Zealand. Super C65 nano carbon black conductive, used as a conductive additive in electrodes, was from MSE supplies™, USA. Ethylene glycol (AR grade) was purchased from KEMAUS Chemical, Australia. All chemicals were used as received without further purification.
Preparation of the amine-modified WT samples
The WT powders were modified by amines (TEPA or PEI) using wet impregnation. The selected amine was dissolved in 50 mL ethanol with stirring at ambient temperature (about 30 °C) for 30 min. Then, 30 g of WT powder was mixed with the amine solution by stirring at ambient temperature for 3 h. Finally, the slurry was then dried in an oven at 60 °C for 48 h. To optimize the amine loading level, amine loadings were varied at 0, 2.5, 5, 10, and 15% (w/w, based on 100 g WT). The amine-modified WT samples were coded WTxP or WTxT for PEI and TEPA modification, respectively, where x represents the amine % (w/w) loading level. The amine efficiency of the amine-doped composite that showed the maximum CO2 adsorption capacity was calculated based on previous work by Sakwa-Novak et al.69 In this study, the amine efficiency of WT10T and WT10P was approximately 0.1 and 0.08, respectively.
Characterization of the amine-modified WT samples
The WT samples before and after amine modification were characterized by nitrogen (N2) adsorption-desorption Micromeritics, Asap 2020, USA) to determine the Brunauer-Emmett-Teller (BET) surface area (SBET) and total pore volume (Vp). The samples were degassed under a N2 flux for 2 h at 120 °C. Scanning electron microscopy (SEM; JEOL, JSM-6480LV, USA) at an accelerating voltage of 15 kV was used to investigate the WT morphology before and after amine modification. The samples were placed on a stub and then coated with gold before testing. The elemental analysis was examined using a CHN/S analyzer (Perkin Elmer, PE-2400, USA), while the composition of the samples was evaluated using Fourier-transformed infrared spectroscopy (FTIR, Thermo Ficher, Nicolet iS5, USA). To confirm the CO2 adsorption-desorption in the adsorbent, Raman spectroscopy (DXR Raman microscope, Thermo Scientific, USA) was examined and recorded from 100 to 4000 cm−1.
The powder rubber samples (0.4 g) contained in tea bag were immersed in 100 mL of toluene for 72 h. After that the surface liquid was rapidly removed by blotting with filter paper. The samples were dried in an oven at 80 °C until at constant weight. The swelling ratio (Q), sol-gel fraction, and crosslink density were calculated according to Eqs. (1)–(4), respectively70,71.
where W1 is the initial sample weight, W2 is the sample weight after swelling, W3 is the dried sample weight after swelling, vr is the volume fraction of the rubber network in the swollen gel, vo is the molar volume of the toluene (106.2 cm3/mol)70, and χ is a rubber-solvent interaction parameter (0.393)71. The three parameters—swelling ratio, sol-gel fraction, and crosslink density—are studied to evaluate the properties of WT, specifically before and after modification. These parameters help in understanding the effects of amine incorporation on the tires’ properties, providing insight into how the amine modification influences the material’s structural and functional characteristics.
CO2 adsorption-desorption
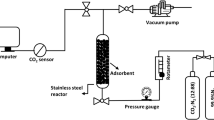
The CO2 adsorption-desorption of the unmodified and amine-modified WT samples was performed in a stainless-steel reactor as shown in Fig. 1. The dried sample (3 g) was loaded into the reactor and then heated under vacuum pressure at 60 °C under a N2 flow rate of 300 mL/min for 20 min to release existing bound gases. After that, the reactor was cooled down to room temperature, and then the gas was switched to CO2 [12% and 99% (v/v)] at a flow rate of 70 mL/min at ambient temperature and atmospheric pressure. The CO2 amount in the effluent was detected using a CO2 sensor (CM-40401, United States of America). The CO2 adsorption capacity was then calculated based on the difference between the CO2 adsorption breakthrough curve areas. The CO2 desorption of each sample was performed by the combination of vacuum and temperature swing adsorption (VTSA) process. The adsorbent after CO2 adsorption was heated at 60 °C under reduced pressure with a N2 flow rate of 100 mL/min and then cooled to ambience temperature. Finally, the sample was subjected to 10 sequential repeated CO2 adsorption-desorption cycles to evaluate its reusability.
Kinetic study
The pseudo 1 st order, pseudo 2nd order, and Avrami’s kinetic models were used to fit the experimental adsorption kinetic data to investigate the adsorption mechanism of the adsorbent using Eqs. (5)–(7), respectively72,
$$Pseudo \, 1^{st} \, order: \:{q}_{t}=\:{q}_{e}\left[1-\text{exp}\left(-{k}_{1}t\right)\right]$$(5)$$Pseudo \, 2^{nd} order: \:{q}_{t}=\:\frac{{k}_{2}{q}_{e}^{2}}{1+{k}_{2}{q}_{e}t}t$$(6)$$Avrami{'}s \, model: \:{q}_{t}=\:{q}_{e}\left[1-{\text{exp}\left(-{k}_{A}t\right)}^{n}\right]$$(7)
where \(\:t\) is the adsorption time (min), \(\:{q}_{e}\) is the adsorption capacity at equilibrium (mg\(\cdot\)g−1), \(\:{q}_{t}\) is the adsorption capacity at time \(\:t\) (min), \(\:{k}_{1}\) is the pseudo 1 st order rate constant (min− 1), \(\:{k}_{2}\) is the pseudo 2nd order rate constant (g\(\cdot\)mg−1\(\cdot\)min− 1), \(\:{k}_{A}\) (min− 1) is the Avrami’s rate constant, and \(\:n\) is the order of the kinetic equation.
CO2 conversion
The catalyst for the CO2 electrochemical conversion by the CO2RR was prepared as a 1:2 mol ratio of CuO: Cu2O on carbon paper. The electrochemical test was performed using a three-electrode cell on a H-cell station. A platinum plate of 1 × 2 cm in size was employed as the counter electrode and Ag/AgCl (in 3 M sodium chloride solution) soaked in a luggin capillary was used as the reference electrode. The cathode and anode compartments were separated by a piece of proton exchange membrane (Nafion 117, Dupont) to prevent re-oxidation of the products. The 0.1 M KHCO3 electrolyte solution was purged with CO2 [12% and 99% (v/v)] for 10 min before applying a reduction potential of −1.7 V (vs. Ag/AgCl). The gaseous products after the chronoamperometric electrolysis were detected by gas chromatography equipped with a thermal conductivity detector (GC-TCD) using helium as the carrier gas. The overall summary of this research is shown in Fig. 2.
Results and discussion
Characterization of the amine-modified WT
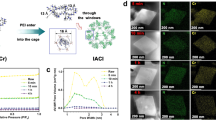
The FTIR spectra of the WT before and after amine modification are shown in Fig. 3. The peak located at 2914 cm−1 corresponded to the aliphatic C–H unsymmetrical stretching vibration of CH3– groups and the peak at 2847 cm−1 was attributed to the aliphatic C–H symmetrical stretching vibration of –CH2– groups71,73,74. These results indicated that the WT used in this study may contain both synthetic and natural rubbers. The peak at 1536 cm−1 was assigned to the C-S bond from the sulfur crosslinks formed during the WT production75. After the WT was modified by TEPA or PEI, a broad peak at 3210 cm−1 was evident, which represented the N-H symmetric and asymmetric stretching vibration of amine groups76,77. The N-H bending of the secondary amine [-N(R)H] was represented at 1650 cm−1, while the asymmetric and symmetric bending of primary amine (-NH2) were observed at 1560 and 1430 cm−1, respectively76,78,79. It is interesting to note that the peak intensity of N-H stretching and bending for WT10P was clearly higher than that for WT10T due to the high numbers of amine active sites in the PEI structure. In addition, based on the elemental analysis (Table 1), the nitrogen contents of WT10P were higher than those of WT10T. According to these results, the WT was successfully modified by TEPA or PEI.
However, the peak at 1536 cm−1 representing the C–S bond in both the WT10T and WT10P was markedly reduced or disappeared, while the sulfur content in WT10T (2.72%) and WT10P (2.44%) was decreased compared to that in the WT (3.09%) due to the devulcanization by amines80,81. Table 2 displays the swelling ratio, sol-gel fraction, and crosslink density of WT without and with various amine modifications. The swelling ratio of WT10T (4.11) and WT10P (3.91) was higher than that of unmodified WT (3.56). The sol fraction of WT10T and WT10P tended to be increased and the crosslink density of WT10T (23.30 × 10−3 mol/cm3) and WT10P (23.55 × 10−3 mol/cm3) was lower than that of WT (27.32 × 10−3 mol/cm3). These results confirmed that the amines (TEPA and PEI) could break up the sulfur bonds in rubber vulcanizates, resulting in a decreased level of C–S bonds. Accordingly, TEPA and PEI serve not only as modifiers of WT but also as devulcanizing agents.
The morphology of the WT before and after amine modification is shown as SEM images in Fig. 4. The WT surface contained some small pores and had a SBET and total Vp of 2.02 m2∙g−1 and.
5.80 × 10−4 cm3∙g⁻¹, respectively (Table 1). After amine modification, the modified WT surface became rougher and lost its elasticity, resulting in an increased SBET and total Vp. Although, the total Vp of WT10T and WT10P was increased after amine modification, their values seemed to be low when compared to those previously reported for other solid adsorbent materials, i.e., MOFs, carbon materials, silica, and zeolite82,83,84,85. Accordingly, the adsorbent materials prepared in this study exhibited the characteristics of a non-porous material.
Effect of the amine type, amine loading level, and CO2 concentration on the CO2 adsorption
The WT powder was modified by two amine types (TEPA and PEI) at varying amine loading levels [2.5–15% (w/w)] to form the CO2 adsorbents, which were then compared under the same operating condition of an ambient temperature and atmospheric pressure. The breakthrough curve of the adsorbents (Fig. 5) reveals distinct adsorption behaviors. When comparing amine types, the TEPA-modified WTs showed a low sensitivity of breakthrough behavior with the amine loading level, while the PEI-modified WTs exhibited a high sensitivity in their breakthrough capacity with the amine loading level. This can be explained by the structural effects of amine polymers on adsorbent performance. Branched amine polymers may obstruct CO2 adsorption compared to that of linear amine polymers. The breakthrough curves of WTs with different amine loadings (Fig. 5a and b) corresponded with the CO2 adsorption capacity of the amine-modified WT with various amine loadings (Fig. 5c). The CO2 adsorption capacity of the unmodified WT was about 7.10 mg/g of adsorbent, while the CO2 adsorption capacity of the WT modified with TEPA or PEI increased with increasing amine loading levels up to 5% or 10% (w/w) for PEI and TEPA, respectively. With an amine loading in the range of 2.5–5% (w/w), the CO2 adsorption capacity of the PEI-modified WT increased more than that for the TEPA-modified WT. This is presumably due to the difference in chemical structure of both amines, where PEI is an organic polyamine polymer with a network structure, while TEPA is a linear structure. Therefore, there are more basic sites in PEI than in TEPA to bind the CO2 molecules86.
Increasing the amine loading level to 10% (w/w) decreased the CO2 adsorption capacity of WT10P slightly, while that for WT10T was still increased compared to that with a 5% (w/w) loading level. This suggested that a high concentration of the high molecular weight PEI made the PEI-modified WT sample stickier, resulting in a decreased surface area and high CO2 diffusion resistance, whilst this behavior was not observed in the case of WT10T. At a 15% (w/w) amine loading level, the CO2 adsorption capacities of the WT15T and, especially, WT15P decreased markedly compared to those at 10% (w/w) due to the adsorbent surface becoming sticky. The morphology of the amine-modified adsorbents with various amine loading levels are represented in supplementary information (Figs. S1 and S2).
The FTIR spectral analysis was used to ascertain the nature of WT10T and WT10P before and after CO2 adsorption as well as CO2 desorption (Fig. 6). Before adsorption, the peaks of each adsorbent were located at 3210 cm−1, assigned to the N-H symmetric and asymmetric stretching vibration of amine groups. The peaks located at 1650 and 1560 cm−1 were attributed to the N-H bending of the secondary and primary amines, respectively. After CO2 adsorption, the peak located at 1535 cm−1, which represented the asymmetric stretching vibration of the carboxylate anion (COO−) of ammonium carbamate, was observed in both WT10T and WT10P87,88,89,90,91. This indicated that the CO2 molecules could be chemisorbed on the amine-impregnated WT. However, the interaction between CO2 molecules and amino groups was destroyed after the CO2 desorption under regeneration using the VTSA process, resulting in the presence of N-H bending peaks at 1650 and 1560 cm−1, similar to the peaks of the sorbent materials before CO2 adsorption.
Additionally, the CO2 capture in the adsorbent materials could be confirmed by Raman spectroscopy (Fig. 7). The two distinct bands at 1360 and 1570 cm−1 represented the carbon black in WT92,93 and corresponded to the disordered carbon (D band) and the graphite (G band) structures, respectively93. Before CO2 adsorption (Fig. 7a), the intensity peak ratio (ID/IG = I1360/I1570) presented the same value (0.85) because the carbon black in all samples was the same content and did not adsorb or react with CO2 molecules. After CO2 adsorption (Fig. 7b), the peak intensity of the band at 1360 cm−1 seemed to be increased due to a band overlap of CO2 molecules at 1370 and 1245 cm−1 and D band of carbon black as previous research52,94. The I1360/I1570 ratio of WT10P and WT10T was then increased to 0.89 and 0.91, respectively. Whist, the I1360/I1570 of WT did not change because the small amount of CO2 molecules was absorbed. Accordingly, the CO2 molecules could be adsorbed more favorably on the amine-impregnated adsorbent materials compared to the neat WT.
The CO2 adsorption capacity of WT10T at ambient (30 °C), 45 °C, and 60 °C is summarized in Fig. 8d. When the temperature increased during the CO2 adsorption, the CO2 adsorption of WT10T tended to be reduced. This was due to the exothermic nature of the adsorption reaction95,96, where increasing the temperature decreased the adsorption rate. As a result, the time for CO2 to flow through the adsorbent material, and the absorption level was reduced. This behavior is different from the previous study that used NR filled with N-[(3-trimethoxysilyl)propyl]ethylenediamine and N-[(3-trimethoxysilyl)propyl]diethylenetriamine)52. This may be due to the different amine chemical structure, rubber nature, and modified silica particles.
Overall, WT10T had the highest CO2 adsorption capacity (10.41 ± 0.24 mg∙g⁻¹ of adsorbent) at an ambient temperature and atmospheric pressure. Thus, this sample was then chosen for further study of kinetics, regeneration, and CO2RR. It is interesting to note that the effect of CO₂ concentration on Faradaic efficiency (FE) is significant. As the CO₂ concentration increased from 12% (v/v) to 99% (v/v), the CO₂ adsorption capacity of the WT10T adsorbent notably increased, from 10.41 ± 0.243 to 59.98 ± 1.577 mg∙g⁻¹ under ambient temperature and atmospheric pressure (Table 3). This enhancement in CO₂ adsorption was accompanied by an increase in FE, with both CH₄ and C₂H₄ detected as gas products. The increase in FE with higher CO₂ concentration can be attributed to more efficient interactions between the adsorbent and CO₂ molecules at higher concentrations, leading to better electrochemical conversion. Therefore, factors such as amine type, amine loading level, and CO₂ concentration play important roles in influencing both the CO₂ adsorption capacity and the Faradaic efficiency of the amine-modified WT adsorbent.
Here in this study, the comparison of CO2 adsorption capacity for different adsorbent materials is summarized in Table 4. The observed variation in the CO2 adsorption capacity was due to the difference in specific properties and testing conditions of each sorbent material under a similar condition, WT10T could be considered as a potential alternative solid sorbent material for CO2 capture. It has a high CO2 adsorption capacity, is easy to prepare, low-cost, low energy requirement, and is environmentally friendly. In addition, the advantage of using WTs to make an absorbent material is that it reduces the current large-scale amount of rubber waste.
Kinetic adsorption. Kinetic modeling is an important step in evaluating the CO2 adsorption mechanism of an adsorbent material. The experimental CO2 adsorption capacities of the WT10T sample were fitted to each kinetic model (Fig. 8a and c), with the results summarized in Table 5. The pseudo 1 st order and pseudo 2nd order models were clearly not suitable for describing the adsorption kinetics of WT10T due to their low R2 value, whereas Avrami’s model fitted well with the adsorption experimental data (R2 > 0.97 with the lowest root mean square error and mean square error values). Thus, CO2 could interact with WT10T by both physisorption with the rubbery matrix and by chemisorption with the amine groups, in agreement with the Avrami’s model order (na) being in the range of 0.82–1.27100.
Regeneration of adsorbent materials
To evaluate the stability of the amine-modified adsorbent materials, the adsorption-desorption cycle of WT and WT10T was investigated over 10 sequential cycles using the VTSA process. The relationship between the CO2 adsorption capacity and the cycle number for both WT and WT10T are shown in Fig. 9. The CO2 adsorption capacity of the unmodified WT decreased by 5.72% in the 2nd cycle compared to the 1 st cycle. This is because the heterogeneity in sorbent’s active site was treated during the first regeneration cycle. Thereafter, the CO2 adsorption capacity then remained almost constant until the 6th cycle, which can be explained by the stability of the active component in the sorbent with the employed regeneration method. Finally, the CO2 adsorption capacity gradually declined again by 13.55% and 16.47% at the 7th and 10th cycle (compared to the 1 st cycle), respectively. This decline was due to the loss of active components in the sorbent material, as evidenced by CHNS results. Thus, the WT adsorbent material showed a poor regeneration stability. In contrast, the CO2 adsorption capacity of WT10T seemed to be stable over all 10 cycles, decreasing by less than 3%. Accordingly, the regeneration stability of WT10T was better than that of WT, which reflected that the amine sites on WT10T are not volatile and do not show oxidative degradation during the regeneration process101,102. This result was also supported by the CHNS analysis, where the nitrogen contents of WT10T before and after regeneration seemed to be constant (Table 1).
Electrochemical CO2RR
To study the catalytic activity and the effect of the CO2 concentration on the catalytic performance of the CO2RR using a 1:2 mol ratio CuO: Cu2O on carbon paper catalyst, the CO2 was supplied to the CO2RR electrochemical cell at 12% and 99% (v/v). The resulting GC-TCD chromatogram of the CO2RR is shown in Fig. 10. With 12% (v/v) CO2, the CO2RR gave CO and H2 at a 0.15% and 5.99% Faradaic efficiency (FE), respectively. The relatively low FE values might come from the low CO2 concentration in the electrolyte solution. When the CO2 concentration was increased to 99% (v/v), the CO2RR produced CO, H2, CH4, and ethylene (C2H4) at a 25.12%, 26.84%, 0.78%, and 5.14% FE, respectively. These observations indicated that to obtain HC products from the electrochemical CO2RR, a high concentration of CO2 needs to be released from the adsorption process. It is important to know that the experiment in this study was conducted as a batch scale, which is a limitation for CO2 conversion. Adjusting the methodology to a continuous system and increasing the CO2 concentration will enhance the efficiency of CO2 conversion into valuable products using CuO and Cu2O catalysts.
Conclusion
In this study, different amine-modified WT samples were successfully prepared using TEPA and PEI, and then developed as an adsorbent for CO2 capture. The prepared adsorbents exhibited a non-porous material nature. Based on the FTIR and CHNS analyses, the specific amine spectrum was found on the WT surface and the nitrogen content was increased after modification. The optimum condition to obtain the highest CO2 adsorption capacity of the modified WT under a CO2 flow rate of 70 mL min−1, ambient temperature, and atmospheric pressure was modification of the WT using 10% (w/w) TEPA to give a CO2 adsorption capacity of 10.41 mg g−1 of adsorbent. The kinetic adsorption of WT10T fitted well with Avrami’s model, supporting both physical and chemical adsorptions. Finally, WT10T had a high stability in regeneration over at least 10 cycles. Accordingly, WT10T is an alternative CO₂ adsorbent that could potentially be used as a sorbent material for CO₂ capture. The CO2 present in the adsorbent material could undergo direct conversion to CO, H2, CH4, and C2H4 through CO2RR when CuO/Cu2O on carbon paper served as the catalyst. Moreover, elevated CO2 levels corresponded to a higher FE in HC product generation. This research can be related to the concept of BCG and CCUS and shows one way of how to potentially utilize waste materials as adsorbents and reduce the waste from rubber industries.
Data availability
The dataset generated and/or analyzed during the current study are available from the corresponding authors on reasonable request.
References
Dwivedi, Y. K. et al. Climate change and COP26: are digital technologies and information management part of the problem or the solution? An editorial reflection and call to action. Int. J. Inf. Manage. 63, 102456. https://doi.org/10.1016/j.ijinfomgt.2021.102456 (2022).
Ma, C., Bai, J., Hu, X., Jiang, Z. & Wang, L. Nitrogen-doped porous carbons from polyacrylonitrile fiber as effective CO2 adsorbents. J. Environ. Sci. 125, 533–543. https://doi.org/10.1016/j.jes.2022.03.016 (2023).
Castrejon-Campos, O. Evolution of clean energy technologies in mexico: A multi-perspective analysis. Energy Sustainable Dev. 67, 29–53. https://doi.org/10.1016/j.esd.2022.01.003 (2022).
UNFCCC. Report of the Conference of the Parties on its twenty-sixth session, held in Glasgow from 31 October to 13 November 2021. United Kingdom of Great Britain and Northern Ireland, (2021).
Han, X. et al. Comparative life cycle greenhouse gas emissions assessment of battery energy storage technologies for grid applications. J. Clean. Prod. 392, 136251. https://doi.org/10.1016/j.jclepro.2023.136251 (2023).
Kourkoumpas, D. S. et al. A review of key environmental and energy performance indicators for the case of renewable energy systems when integrated with storage solutions. Appl. Energy. 231, 380–398. https://doi.org/10.1016/j.apenergy.2018.09.043 (2018).
Ullah, A., Hashim, N. A. & Rabuni, M. F. Mohd junaidi, M. U. A review on methanol as a clean energy carrier: roles of zeolite in improving production efficiency. Energies 16 https://doi.org/10.3390/en16031482 (2023).
Wang, W. et al. Experimental investigation of stress corrosion on supercritical CO2 transportation pipelines against leakage for CCUS applications. Energy Rep. 9, 266–276. https://doi.org/10.1016/j.egyr.2022.11.179 (2023).
Leonzio, G., Bogle, I. D. L. & Ugo Foscolo, P. Life cycle assessment of a carbon capture utilization and storage supply chain in Italy and germany: comparison between carbon dioxide storage and utilization systems. Sustainable Energy Technol. Assess. 55, 102743. https://doi.org/10.1016/j.seta.2022.102743 (2023).
Gür, T. M. Carbon dioxide emissions, capture, storage and utilization: review of materials, processes and technologies. Prog Energy Combust. Sci. 89, 100965. https://doi.org/10.1016/j.pecs.2021.100965 (2022).
Guerrero Peña, G. D. J. et al. Carbon dioxide adsorbents from flame-made diesel soot nanoparticles. Sci. Total Environ. 859, 160140. https://doi.org/10.1016/j.scitotenv.2022.160140 (2023).
Nandi, R., Jha, M. K., Guchhait, S. K., Sutradhar, D. & Yadav, S. Impact of KOH activation on rice husk derived porous activated carbon for carbon capture at flue gas alike temperatures with high CO2/N2 selectivity. ACS Omega. 8, 4802–4812. https://doi.org/10.1021/acsomega.2c06955 (2023).
Malini, K., Selvakumar, D. & Kumar, N. S. Activated carbon from biomass: preparation, factors improving basicity and surface properties for enhanced CO2 capture capacity – A review. J CO2 Util. 67, 102318. https://doi.org/10.1016/j.jcou.2022.102318 (2023).
Indira, V. & Abhitha, K. Mesoporogen-free synthesis of hierarchical zeolite A for CO2 capture: effect of freeze drying on surface structure, porosity and particle size. Results Eng. 17, 100886. https://doi.org/10.1016/j.rineng.2023.100886 (2023).
Phuong, N. T. T. et al. Enhancement of CO2 adsorption by introducing mesopores into FAU zeolite using acid-base leaching. J. Appl. Sci. Eng. (Taiwan). 26, 793–800. https://doi.org/10.6180/jase.202306_26(6).0005 (2023).
Rahmah, W. et al. Small-pore zeolite and zeotype membranes for CO2 capture and sequestration – A review. J. Environ. Chem. Eng. 10, 108707. https://doi.org/10.1016/j.jece.2022.108707 (2022).
Liu, L. et al. Low-cost DETA impregnation of acid-activated sepiolite for CO2 capture. Chem. Eng. J. 353, 940–948. https://doi.org/10.1016/j.cej.2018.07.086 (2018).
Bai, J. et al. Synthesis and characterization of polyphenylene sulfide resin-derived S-doped porous carbons for efficient CO2 capture. Colloids Surf. A: Physicochem Eng. Asp. 674, 131916. https://doi.org/10.1016/j.colsurfa.2023.131916 (2023).
Bai, J. et al. Sulfur-doped porous carbon adsorbent: A promising solution for effective and selective CO2 capture. Chem. Eng. J. 479, 147667. https://doi.org/10.1016/j.cej.2023.147667 (2024).
Sang, Y. et al. N-rich porous organic polymers based on schiff base reaction for CO2 capture and mercury (II) adsorption. J. Colloid Interf Sci. 587, 121–130. https://doi.org/10.1016/j.jcis.2020.12.002 (2021).
Qi, S. C. et al. Active sites modulation with Runge–Gross theorem: CO2 capture by porphyrinic metal–organic frameworks at excited States. AIChE J. 69, e17994. https://doi.org/10.1002/aic.17994 (2023).
Roy, S. et al. Pores on pores: A novel approach to fabricate super adsorbents from used face masks for large CO2 capture and dye removal. Carbon 206, 422–433. https://doi.org/10.1016/j.carbon.2023.02.040 (2023).
Sanz-Pérez, E. S. et al. Reuse and recycling of amine-functionalized silica materials for CO2 adsorption. Chem. Eng. J. 308, 1021–1033. https://doi.org/10.1016/j.cej.2016.09.109 (2017).
Serafin, J., Sreńscek-Nazzal, J., Kamińska, A., Paszkiewicz, O. & Michalkiewicz, B. Management of surgical mask waste to activated carbons for CO2 capture. J CO2 Util. 59, 101970. https://doi.org/10.1016/j.jcou.2022.101970 (2022).
Igalavithana, A. D. et al. Gasification Biochar from Biowaste (food waste and wood waste) for effective CO2 adsorption. J. Hazard. Mater. 391, 121147. https://doi.org/10.1016/j.jhazmat.2019.121147 (2020).
Jin, C. et al. Sawdust wastes-derived porous carbons for CO2 adsorption. Part 1. Optimization Preparation via orthogonal experiment. Sep. Purif. Technol. 276, 119270. https://doi.org/10.1016/j.seppur.2021.119270 (2021).
Concil, B. Call for UK consultants: Foresight into bio-circular-green economic model (BCG) projectThailand,. (2021).
Mikunda, T. et al. Carbon capture and storage and the sustainable development goals. Int. J. Greenh. Gas Control. 108, 103318. https://doi.org/10.1016/j.ijggc.2021.103318 (2021).
Mazari, S. A. et al. An overview of catalytic conversion of CO2 into fuels and chemicals using metal organic frameworks. Process. Saf. Environ. Prot. 149, 67–92. https://doi.org/10.1016/j.psep.2020.10.025 (2021).
Zhou, J., Liu, H. & Wang, H. Photothermal catalysis for CO2 conversion. Chin. Chem. Lett. 34, 107420. https://doi.org/10.1016/j.cclet.2022.04.018 (2023).
Yang, D., Li, S., He, S. & Zheng, Y. Can conversion of CO2 into fuels via electrochemical or thermochemical reduction be energy efficient and reduce emissions? Energy Convers. Manage. 273, 116425. https://doi.org/10.1016/j.enconman.2022.116425 (2022).
Xu, G. et al. Characteristics, kinetics, infrared analysis and process optimization of co-pyrolysis of waste tires and oily sludge. J. Environ. Manage. 316, 115278. https://doi.org/10.1016/j.jenvman.2022.115278 (2022).
Jimoda, L., Sulaymon, I., Alade, A. & Adebayo, G. Assessment of environmental impact of open burning of scrap tyres on ambient air quality. Int. J. Environ. Sci. Technol. 15, 1–8. https://doi.org/10.1007/s13762-017-1498-5 (2017).
Feizi, F., Reguyal, F., Antoniou, N., Zabaniotou, A. & Sarmah, A. K. Environmental remediation in circular economy: end of life tyre magnetic Pyrochars for adsorptive removal of pharmaceuticals from aqueous solution. Sci. Total Environ. 739, 139855. https://doi.org/10.1016/j.scitotenv.2020.139855 (2020).
Feizi, F., Sarmah, A. K., Rangsivek, R. & Gobindlal, K. Adsorptive removal of propranolol under fixed-bed column using magnetic tyre char: effects of wastewater effluent organic matter and ball milling. Environ. Pollut. 305, 119283. https://doi.org/10.1016/j.envpol.2022.119283 (2022).
Feizi, F., Sarmah, A. K. & Rangsivek, R. Adsorption of pharmaceuticals in a fixed-bed column using tyre-based activated carbon: experimental investigations and numerical modelling. J. Hazard. Mater. 417, 126010. https://doi.org/10.1016/j.jhazmat.2021.126010 (2021).
Ahmad, W. et al. Utilization of tires waste-derived magnetic-activated carbon for the removal of hexavalent chromium from wastewater. Materials 14, 34. https://doi.org/10.3390/ma14010034 (2021).
Li, W. et al. Catalysts evaluation for production of hydrogen gas and carbon nanotubes from the pyrolysis-catalysis of waste tyres. Int. J. Hydrogen Energy. 44, 19563–19572. https://doi.org/10.1016/j.ijhydene.2019.05.204 (2019).
Elbaba, I. F., Wu, C. & Williams, P. T. Hydrogen production from the pyrolysis–gasification of waste tyres with a nickel/cerium catalyst. Int. J. Hydrogen Energy. 36, 6628–6637. https://doi.org/10.1016/j.ijhydene.2011.02.135 (2011).
Elbaba, I. F. & Williams, P. T. High yield hydrogen from the pyrolysis–catalytic gasification of waste tyres with a nickel/dolomite catalyst. Fuel 106, 528–536. https://doi.org/10.1016/j.fuel.2012.12.067 (2013).
Zhang, Y., Tao, Y., Huang, J. & Williams, P. Influence of silica–alumina support ratio on H2 production and catalyst carbon deposition from the Ni-catalytic pyrolysis/reforming of waste tyres. Waste Manage. Res. 35, 0734242X1772220. https://doi.org/10.1177/0734242X17722207 (2017).
Zhang, Y. & Williams, P. T. Carbon nanotubes and hydrogen production from the pyrolysis catalysis or catalytic-steam reforming of waste tyres. J. Anal. Appl. Pyrol. 122, 490–501. https://doi.org/10.1016/j.jaap.2016.10.015 (2016).
Zhang, Y., Wu, C., Nahil, M. A. & Williams, P. Pyrolysis–catalytic reforming/gasification of waste tires for production of carbon nanotubes and hydrogen. Energy Fuels. 29, 3328–3334. https://doi.org/10.1021/acs.energyfuels.5b00408 (2015).
Hood, Z. et al. Conversion of waste tire rubber into high-value-added carbon supports for electrocatalysis. J. Electrochem. Soc. 165, H881–H888. https://doi.org/10.1149/2.1081813jes (2018).
Zhi, M. et al. Effects of pore structure on performance of an activated-carbon supercapacitor electrode recycled from scrap waste tires. ACS Sustainable Chem. Eng. 2, 1592–1598. https://doi.org/10.1021/sc500336h (2014).
Zhao, P., Han, Y., Dong, X., Zhang, C. & Liu, S. Application of activated carbons derived from scrap tires as electrode materials for supercapacitors. ECS J. Solid State Sci. Technol. 4, M35–M40. https://doi.org/10.1149/2.0271505jss (2015).
Naskar, A. et al. Tailored recovery of carbons from waste tires for enhanced performance as anodes in lithium-ion batteries. RSC Adv. 4, 38213-38221. https://doi.org/10.1039/C4RA03888F (2014).
Duduku, K., Anisuzzaman, S., Bono, Teo, B. & Rao, V. Carbon dioxide capture using waste tire based adsorbent. IOP Conf. Ser. : Mater. Sci. Eng. 606, 012006. https://doi.org/10.1088/1757-899X/606/1/012006 (2019).
Jankovská, Z., Večeř, M., Koutník, I. & Matějová, L. A case study of waste scrap tyre-derived carbon black tested for nitrogen, carbon dioxide, and cyclohexane adsorption. Molecules 25, 4445. https://doi.org/10.3390/molecules25194445 (2020).
Tang, X., Song, M., Lei, Y. & Wang, X. Comparison and application of different component municipal solid wastes based carbon on adsorption of carbon dioxide. Int. J. Green. Energy. 14, 135–140. https://doi.org/10.1080/15435075.2016.1253578 (2017).
Panploo, K., Chalermsinsuwan, B. & Poompradub, S. Natural rubber latex foam with particulate fillers for carbon dioxide adsorption and regeneration. RSC Adv. 9, 28916–28923. https://doi.org/10.1039/C9RA06000F (2019).
Panploo, K., Chalermsinsuwan, B. & Poompradub, S. Effect of amine types and temperature of a natural rubber based composite material on the carbon dioxide capture. Chem. Eng. J. 402, 125332. https://doi.org/10.1016/j.cej.2020.125332 (2020).
Tumnantong, D., Panploo, K., Chalermsinsuwan, B., Prasassarakich, P. & Poompradub, S. Carbon dioxide adsorption of diallylamine-modified natural rubber with modified silica particles. Express Polym. Lett. 15, 899–909. https://doi.org/10.3144/expresspolymlett.2021.72 (2021).
Hu, X. et al. A review of N-functionalized solid adsorbents for post-combustion CO2 capture. Appl. Energy. 260, 114244. https://doi.org/10.1016/j.apenergy.2019.114244 (2020).
Güzel Kaya, G. Polyethylene glycol/silica and carbon black/silica xerogel composites as an adsorbent for CO2 capture. Turk. J. Chem. 45, 2013–2023. https://doi.org/10.3906/kim-2101-45 (2021).
Ramarad, S., Khalid, M., Ratnam, C. T., Luqman Chuah, A. & Rashmi, W. Waste tire rubber in polymer blends: A review on the evolution, properties and future. Prog Mater. Sci. 72, 100–140. https://doi.org/10.1016/j.pmatsci.2015.02.004 (2015).
Yang, G. X. & Jiang, H. Amino modification of Biochar for enhanced adsorption of copper ions from synthetic wastewater. Water Res. 48, 396 (2014).
Wang, Y., Chen, E. & Tang, J. Insight on reaction pathways of photocatalytic CO2 conversion. ACS Catal. 12, 7300–7316. https://doi.org/10.1021/acscatal.2c01012 (2022).
Ma, J. et al. A short review of catalysis for CO2 conversion. Catal. Today. 148, 221–231. https://doi.org/10.1016/j.cattod.2009.08.015 (2009).
Kumar, A., Aeshala, L. M. & Palai, T. Electrochemical reduction of CO2 to useful fuel: recent advances and prospects. J. Appl. Electrochem. 53, 1295–1319. https://doi.org/10.1007/s10800-023-01850-x (2023).
Garg, S. et al. Advances and challenges in electrochemical CO2 reduction processes: an engineering and design perspective looking beyond new catalyst materials. J. Mater. Chem. A. 8, 1511–1544. https://doi.org/10.1039/C9TA13298H (2020).
Liang, S., Huang, L., Gao, Y., Wang, Q. & Liu, B. Electrochemical reduction of CO2 to CO over transition metal/N-doped carbon catalysts: the active sites AndReaction mechanism. Adv. Sci. 8, 2102886. https://doi.org/10.1002/advs.202102886 (2021).
Parvez, A. M., Afzal, M. T., Hebb, T. G. V. & Schmid, M. Utilization of CO2 in thermochemical conversion of biomass for enhanced product properties: A review. J CO2 Util. 40, 101217. https://doi.org/10.1016/j.jcou.2020.101217 (2020).
Roy, S., Cherevotan, A. & Peter, S. C. Thermochemical CO2 hydrogenation to single carbon products: scientific and technological challenges. ACS Energy Lett. 3, 1938–1966. https://doi.org/10.1021/acsenergylett.8b00740 (2018).
Li, C. F., Guo, R. T., Zhang, Z. R., Wu, T. & Pan, W. G. Converting CO2 into value-added products by Cu2O-based catalysts: from photocatalysis, electrocatalysis to photoelectrocatalysis. Small 19, 2207875. https://doi.org/10.1002/smll.202207875 (2023).
Roy, A., Jadhav, H. S. & Gil Seo, J. Cu2O/CuO electrocatalyst for electrochemical reduction of carbon dioxide to methanol. Electroanalysis 33, 705–712. https://doi.org/10.1002/elan.202060265 (2021).
Nitopi, S. et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem. Rev. 119, 7610–7672. https://doi.org/10.1021/acs.chemrev.8b00705 (2019).
Ohya, S., Kaneco, S., Katsumata, H., Suzuki, T. & Ohta, K. Electrochemical reduction of CO2 in methanol with aid of CuO and Cu2O. Catal. Today. 148, 329–334. https://doi.org/10.1016/j.cattod.2009.07.077 (2009).
Sakwa-Novak, M. A., Tan, S. & Jones, C. W. Role of additives in composite pei/oxide CO2 adsorbents: enhancement in the amine efficiency of supported PEI by PEG in CO2 capture from simulated ambient air. ACS Appl. Mater. Interfaces. 7, 24748–24759. https://doi.org/10.1021/acsami.5b07545 (2015).
Tunlert, A., Prasassarakich, P. & Poompradub, S. Antidegradation and reinforcement effects of phenyltrimethoxysilane-or N-[3-(trimethoxysilyl) propyl] aniline-modified silica particles in natural rubber composites. Mate Chem. Phys. 173, 78–88. https://doi.org/10.1016/j.matchemphys.2016.01.041 (2016).
Kaewpetch, B., Prasongsuk, S. & Poompradub, S. Devulcanization of natural rubber vulcanizates by Bacillus cereus TISTR 2651. Express Polym. Lett. 13, 877–888. https://doi.org/10.3144/expresspolymlett.2019.76 (2019).
Wang, Y., Guo, T., Hu, X., Hao, J. & Guo, Q. Mechanism and kinetics of CO2 adsorption for TEPA- impregnated hierarchical mesoporous carbon in the presence of water vapor. Powder Technol. 368, 227–236. https://doi.org/10.1016/j.powtec.2020.04.062 (2020).
Wu, B. & Zhou, M. Recycling of waste tyre rubber into oil absorbent. Waste Manage. (New York N Y). 29, 355–359. https://doi.org/10.1016/j.wasman.2008.03.002 (2008).
Gorassini, A., Adami, G., Calvini, P. & Giacomello, A. ATR-FTIR characterization of old pressure sensitive adhesive tapes in historic papers. J. Cult. Herit. 21, 775–785. https://doi.org/10.1016/j.culher.2016.03.005 (2016).
Karabork, F., Pehlivan, E. & Akdemir, A. Characterization of styrene butadiene rubber and microwave devulcanized ground tire rubber composites. J. Polym. Eng. 34, 543–554. https://doi.org/10.1515/polyeng-2013-0330 (2014).
Yoosuk, B., Wongsanga, T. & Prasassarakich, P. CO2 and H2S binary sorption on polyamine modified fumed silica. Fuel 168, 47–53. https://doi.org/10.1016/j.fuel.2015.11.080 (2016).
Liu, F. et al. Facilely synthesized meso-macroporous polymer as support of poly(ethyleneimine) for highly efficient and selective capture of CO2. Chem. Eng. J. 314, 466–476. https://doi.org/10.1016/j.cej.2016.12.004 (2017).
Wang, Y. et al. Amine-functionalized mesoporous ZSM-5 zeolite adsorbents for carbon dioxide capture. Solid State Sci. 73, 27–35. https://doi.org/10.1016/j.solidstatesciences.2017.09.004 (2017).
Wang, W. et al. Sulfuric acid modified bentonite as the support of tetraethylenepentamine for CO2 capture. Energy Fuels. 27, 1538–1546. https://doi.org/10.1021/ef3021816 (2013).
Jiang, C., Zhang, Y., Ma, L., Zhou, L. & He, H. Tailoring the properties of ground tire rubber/high-density polyethylene blends by combining surface devulcanization and in-situ grafting technology. Mater. Chem. Phys. 220, 161–170. https://doi.org/10.1016/j.matchemphys.2018.08.040 (2018).
Walvekar, R., Afiq, Z. M., Ramarad, S. & Khalid, M. Devulcanization of waste tire rubber using amine based solvents and ultrasonic energy. MATEC Web Conf. 152, 01007. https://doi.org/10.1051/matecconf/201815201007 (2018).
Kundu, N. & Sarkar, S. Porous organic frameworks for carbon dioxide capture and storage. J. Environ. Chem. Eng. 9, 105090. https://doi.org/10.1016/j.jece.2021.105090 (2021).
Sharma, A. et al. Carbon materials as CO2 adsorbents: A review. Environ. Chem. Lett. 19, 875–910. https://doi.org/10.1007/s10311-020-01153-z (2021).
Mathai Varghese, A. & Karanikolos, G. CO2 capture adsorbents functionalized by amine – bearing polymers: A review. Int. J. Greenh. Gas Control. 96, 103005. https://doi.org/10.1016/j.ijggc.2020.103005 (2020).
Azmi, A. A. & Aziz, M. A. A. Mesoporous adsorbent for CO2 capture application under mild condition: A review. J. Environ. Chem. Eng. 7, 103022. https://doi.org/10.1016/j.jece.2019.103022 (2019).
Cao, Y., Song, F., Zhao, Y. & Zhong, Q. Capture of carbon dioxide from flue gas on TEPA-grafted metal-organic framework Mg2(dobdc). J. Environ. Sci. 25, 2081–2087. https://doi.org/10.1016/S1001-0742(12)60267-8 (2013).
Lee, J. J. et al. Silica supported sterically hindered amines for CO2 capture. Langmuir 34, 12279–12292. https://doi.org/10.1021/acs.langmuir.8b02472 (2018).
Danon, A., Stair, P. C. & Weitz, E. FTIR study of CO2 adsorption on amine-grafted SBA-15: Elucidation of adsorbed species. J. Phys. Chem. C. 115, 11540–11549. https://doi.org/10.1021/jp200914v (2011).
Milella, F. & Mazzotti, M. Estimating speciation of aqueous ammonia solutions of ammonium bicarbonate: Application of least squares methods to infrared spectra. React. Chem. Eng., 4, 1284–1302. (2019). https://doi.org/10.1039/C9RE00137A. (2019).
Wilfong, W. C., Srikanth, C. S. & Chuang, S. S. C. In situ ATR and DRIFTS studies of the nature of adsorbed CO2 on tetraethylenepentamine films. ACS Appl. Mater. Interfaces. 6, 13617–13626. https://doi.org/10.1021/am5031006 (2014).
Yu, J. & Chuang, S. S. The role of water in CO2 capture by amine. Ind. Eng. Chem. Res. 56 6337 – 6347. https://doi.org/10.1021/acs.iecr.7b00715 (2017).
Pawlyta, M., Rouzaud, J. N. & Duber, S. Raman microspectroscopy characterization of carbon blacks: spectral analysis and structural information. Carbon 84, 479–490. https://doi.org/10.1016/j.carbon.2014.12.030 (2015).
Gomez-Hernandez, R., Panecatl-Bernal, Y. & Mendez-Rojas, M. A. High yield and simple one-step production of carbon black nanoparticles from waste tires. Heliyon 5, e02139. https://doi.org/10.1016/j.heliyon.2019.e02139 (2019).
Vitkin, V. et al. Raman laser spectrometer: application to 12C/13C isotope identification in CH4 and CO2 greenhouse gases. Appl. Sci. 10, 7473. https://doi.org/10.3390/app10217473 (2020).
Zhou, X., Yi, H., Tang, X., Deng, H. & Liu, H. Thermodynamics for the adsorption of SO. NO CO2 Flue Gas Activated Carbon Fiber Chem. Eng. J. 200–202, 399–404. https://doi.org/10.1016/j.cej.2012.06.013 (2012).
Raganati, F., Chirone, R. & Ammendola, P. CO. Capture Temp. Swing. Adsorption: Working Capacity as Affected Temp. CO2 Partial Press. Ind Eng. Chem. Res. 59, 3593–3605. https://doi.org/10.1021/acs.iecr.9b04901 (2020).
Chagas, J. A. O. et al. Characterization, and CO2 uptake of adsorbents prepared by hydrothermal carbonization of Chitosan. ACS Omega. 5, 29520–29529. https://doi.org/10.1021/acsomega.0c04470 (2020).
Wang, J., Adelodun, A. A., Oh, J. M. & Jo, Y. M. TEPA impregnation of electrospun carbon Nanofbers for enhanced low-level CO2 adsorption. Nano Convergence. 7, 7. https://doi.org/10.1186/s40580-020-0217-y (2020).
Pu, Q. et al. Systematic study of dynamic CO2 adsorption on activated carbons derived from different biomass. J. Alloys Compd. 887, 161406. https://doi.org/10.1016/j.jallcom.2021.161406 (2021).
Liu, Y. & Yu, X. Carbon dioxide adsorption properties and adsorption/desorption kinetics of amine-functionalized KIT-6. Appl. Energy. 211, 1080–1088. https://doi.org/10.1016/j.apenergy.2017.12.016 (2018).
Zhou, C. et al. Amine-functionalized mesoporous monolithic adsorbents for post-combustion carbon dioxide capture. Chem. Eng. J. 413, 127675. https://doi.org/10.1016/j.cej.2020.127675 (2021).
Yang, C., Xiong, Y., Chen, J., Jin, J. & Mi, J. Amine-functionalized micron-porous polymer foams with high CO2 adsorption efficiency and exceptional stability in PSA process. Chem. Eng. J. 420, 129555. https://doi.org/10.1016/j.cej.2021.129555 (2021).
Acknowledgements
This project is funded by National Research Council of Thailand (N42A650264) and Chulalongkorn University. The authors also acknowledge the Ratchadapisek Somphot Fund for Postdoctoral Fellowship Chulalongkorn University; Center of Excellence in Green Materials for Industrial Application; Thailand Science Research and Innovation Fund Chulalongkorn University and Hub of Talent: Sustainable Materials for Circular Economy, National Research Council of Thailand (NRCT).
Funding
National Research Council of Thailand (N42A650264), Chulalongkorn University, Ratchadapisek Somphot Fund for Postdoctoral Fellowship, Center of Excellence in Green Materials for Industrial Application and Thailand Science Research and Innovation Fund Chulalongkorn University.
Author information
Authors and Affiliations
Contributions
N. J.: Methodology, Formal analysis, Investigation, Visualization, Data curation, Writing-original draft. D.T.: Formal analysis, Investigation, Data curation, Writing – review & editing. J. L.: Data curation. P. P.: Data curation. P. T.: Data curation, Writing-review & editing. B. C.: Data curation, Writing-review & editing. S. P.: Supervision, Conceptualization, Validation, Resources, Funding acquisition, Writing – review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jaree, N., Tumnantong, D., Luangchaiyaporn, J. et al. Amine-modified waste tires for CO2 capture and resource utilization. Sci Rep 15, 29618 (2025). https://doi.org/10.1038/s41598-025-15313-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15313-0