Abstract
This study aimed to assess differences in fresh semen quality between mature and senile dogs, focusing on oxidative stress markers, including spermatozoa lipid peroxidation and enzymatic antioxidant activity. Furthermore, the ability of frozen-thawed spermatozoa from these age groups to bind to the zona pellucida was investigated. Forty clinically healthy dogs were categorized into two groups: mature (3 ± 0.2 years; n = 20) and senile (10 ± 0.2 years; n = 20). A total of 107 semen samples (2–3 ejaculates per dog) were analyzed for motilities and kinetic parameters, sperm concentration, total sperm count (TSC), acrosome and membrane integrity, morphological defects, DNA fragmentation, lipid peroxidation, and the enzymatic activities of superoxide dismutase, catalase, and glutathione peroxidase in spermatozoa and seminal plasma. A zona-binding assay was performed using frozen-thawed semen from mature (n = 10) and senile (n = 10) dogs. Mature dogs exhibited significantly higher total motility, sperm concentration, TSC, membrane integrity, and zona binding capacity compared to senile dogs (P < 0.05). Conversely, senile dogs had significantly higher proportions of proximal cytoplasmic droplets and tail defects (P < 0.05). However, lipid peroxidation, enzymatic antioxidant activities, and acrosome integrity did not differ significantly between the groups (P > 0.05). In conclusion, the observed decline in fresh semen quality with age in dogs does not appear to be associated with increased susceptibility to oxidative stress. Furthermore, frozen-thawed spermatozoa from mature dogs exhibited superior zona binding capacity, likely due to a greater proportion of progressively motile sperm.
Similar content being viewed by others
Introduction
Several factors, including biochemical influences, have been identified as contributors to decreased sperm quality and infertility in various studies1,2,3,4,5. While extensive research in men and bulls has examined the impact of age on established oxidative stress markers6,7,8this aspect remains relatively underexplored in dogs. Studies on mouse models and bulls have demonstrated a correlation between aging, increased sperm DNA damage, and heightened susceptibility to oxidative stress9,10,11. This vulnerability arises from a decline in key antioxidant enzymes, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), which play a crucial role in protecting the sperm nucleus from oxidative damage. For example, antioxidant enzymes such as peroxiredoxin, which are essential for maintaining DNA integrity, are also affected by aging6,12,13. Recent studies in rats further indicate that germ cells in older males exhibit impaired DNA repair mechanisms and a diminished capacity to respond to oxidative stress compared to younger males14,15.
Considerable evidence suggests that the lipid composition of the sperm membrane plays a crucial role in determining sperm sensitivity to cold, as well as its motility and overall viability1,6,16,17. Reactive oxygen species (ROS) are continuously generated in male reproductive tissues, primarily as byproducts of normal metabolic processes and during fertilization when sperm encounter elevated oxygen levels2,18. While low ROS levels can enhance sperm function by promoting hyperactivation, capacitation, and the acrosome reaction19excessive ROS levels are detrimental. They induce lipid peroxidation (LPO), particularly in the midpiece of the sperm cell, where mitochondria are concentrated20. This oxidative damage compromises sperm motility and viability by depleting intracellular ATP, damaging the axoneme, and causing morphological abnormalities. Consequently, robust antioxidant defense mechanisms within the sperm plasma membrane are essential for mitigating peroxidative damage and preventing sperm dysfunction6,21.
The use of assisted reproductive techniques, such as artificial insemination (AI) with fresh and frozen-thawed semen and the freezing, storage, and shipment of canine semen, has increased in recent years22,23. This trend is primarily driven by the need to exchange genetic material and preserve semen from valuable stud dogs. Given the relatively long estrous cycles in dogs and the challenges of accurately determining the fertile period, assessing the impact of aging on semen quality is crucial. Using semen from aging dogs may compromise reproductive efficiency. Predicting in vivo fertility requires integrating multiple in vitro sperm functional tests to improve accuracy24,25,26. A key evaluation in canine reproduction is assessing the ability of spermatozoa to bind to the zona pellucida. Damage to molecular binding structures, undetectable through routine morphological sperm assessments, can significantly impair male fertility26,27.
Therefore, this study investigates age-related differences in semen quality (viability, concentration, acrosome integrity, morphology) and oxidative stress markers in the semen of mature and senile dogs, as well as the ability of frozen-thawed spermatozoa from these groups to bind to the zona pellucida of oocytes. The analysis includes assessments of membrane LPO, sperm DNA fragmentation, and the activities of key antioxidant enzymes, such as SOD, CAT, and GPx, in both the cellular and plasma fractions of semen. Furthermore, a sperm-zona pellucida binding assay was performed to evaluate the impact of aging on fertilization potential in dogs.
Results
Experiment 1
Figure 1 summarizes the sperm characteristics for the mature and senile age groups. Spermatozoa from mature dogs exhibited a higher percentage of total motility than those from senile dogs (P < 0.001). As reported in our previous study, mature dogs also showed superior progressive motility and kinematic parameters28. Sperm concentration and TSC were lower in senile dogs than in mature dogs (P < 0.001) (Table 1). Mature dogs had a lower proportion of proximal cytoplasmic droplets and tail defects (P < 0.05), while no differences were observed in head and midpiece defects.
(a) Total motility, (b) sperm concentration, (c) total sperm count, (d) proximal cytoplasmic droplets, (e) tail defects, (f) head defects, (g) midpiece defects in the fresh semen of mature (n = 20; 2–4 years) and senile (n = 20; ≥ 9 years) dogs. Dots represent individual ejaculates and the circle indicates mean values. M = 106.
Mature dogs exhibited a significantly higher percentage of sperm with intact acrosome and intact membrane (IAIM) compared to senile dogs (P < 0.05; Table 2). Conversely, the proportion of sperm with intact acrosome and damaged membrane (IADM) and damaged plasma membrane (DM) was higher in senile dogs compared to mature dogs (P < 0.05). However, no significant difference was observed in the percentages of sperm with damaged acrosome and intact membrane (DAIM), damaged acrosome and damaged membrane (DADM), and damaged acrosome (DA) between the two groups (Table 2).
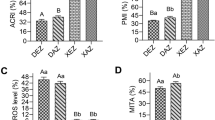
As illustrated in Fig. 2, no significant differences were detected between the mature and senile groups in the following parameters: the proportion of live spermatozoa with lipid peroxidation (P = 0.838), dead spermatozoa with lipid peroxidation (P = 0.238), DNA fragmentation index (DFI) (P = 0.148), and high DNA stainability (HDS) (P = 0.309) (Table 1).
The activities of SOD, CAT, and GPx in seminal plasma and spermatozoa are summarized in Table 3. No significant differences were observed in the activities of these enzymes between the mature and senile age groups.
Among the covariates included in the statistical model, individual animal affected midpiece defects, TSO, and proximal droplets (Table 1).
Experiment 2
The results of the zona binding assay and post-thaw motilities and kinetic parameters are presented in Table 4. The mean number of spermatozoa bound to the zona pellucida was significantly lower in senile dogs than in mature dogs (P < 0.05). Within both groups, variability was observed, with some oocytes not bound by any spermatozoa, while others were bound by multiple spermatozoa (Fig. 3a and b). The post-thaw motility and kinematic parameters of mature dogs were significantly higher than in senile dogs (P < 0.05) (Table 4).
Discussion
To our knowledge, the present study is the first to examine the impact of aging on enzymatic antioxidant levels in dog spermatozoa and seminal plasma, as well as the ability of frozen-thawed spermatozoa to bind to the zona pellucida of oocytes. Additionally, parameters such as TSC, LPO, acrosome integrity, DNA fragmentation, and proximal cytoplasmic droplets were assessed in dogs of optimal reproductive age and those of advanced age, typically associated with a decline in semen quality. The increasing use of AI in dogs, along with the freezing, storage, and shipment of semen from valuable stud dogs, including those of advanced age, highlights the importance of this study. TSC has been well-documented to decline with age in humans, primarily due to alterations in testicular, epididymal, and reproductive accessory gland function29,30,31,32. In contrast, limited research has explored this parameter in dogs33,34. Previous studies combining data from fertile and subfertile dogs have indicated that age significantly influences TSC33,34. The findings of the present study align with those of Rijsselaere et al.34suggesting that aging in dogs may lead to reduced epididymal and accessory sex gland secretions (manifesting as decreased semen volume) and impaired spermatogenesis in the seminiferous tubules. These age-related changes, including diminished germ cell populations, likely contribute to the reduced TSC observed in senile dogs3,29.
The increased presence of proximal cytoplasmic droplets in sperm from senile dogs, along with the higher motility observed in mature dogs in this study, reinforces the hypothesis that impaired cytoplasmic droplet migration negatively affects sperm motility35,36,37. Cytoplasmic droplets are believed to harbor enzymes and substrates incorporated into sperm cells during droplet migration and sperm maturation, facilitating plasma membrane remodeling and the acquisition of motility38,39,40. Furthermore, cytoplasmic droplets are reported to modulate sperm motility by activating mitochondrial function during epididymal maturation35,41. Consistent with this, our previous report demonstrated that mature dogs exhibit significantly higher mitochondrial potential than senile dogs, further underscoring the critical role of cytoplasmic droplets in regulating sperm motility28.
ROS production and LPO are widely believed to increase with age in mammals42,43. LPO can alter the physiological functions of sperm cell membranes and contribute to cellular damage, including DNA fragmentation9,44,45. Elevated LPO levels have been observed in the spermatozoa of aged Brown Norway rats and was associated with reduced sperm quality15. Similarly, increased LPO and oxidative DNA damage have been reported in the frozen-thawed semen of senior bulls9. In contrast, our study found no significant difference in sperm LPO levels between aged and mature dogs. These findings support the assertion by Rikans and Hornbrook46 that increased LPO is not a universal feature of aging and varies depending on the species and tissue type. Our findings are consistent with two prior studies in dogs, which also reported no significant differences in LPO between age groups in fresh and cryopreserved semen22,47. Similarly, a study on Nili-Ravi buffalo bulls found no differences in LPO between aged and young individuals48. Furthermore, Neagu et al.49 suggested that LPO appears to have a minimal role in sperm cryodamage in dogs and is unrelated to the antioxidant activity of seminal plasma. Based on our findings and these previous studies, we propose that LPO in dog spermatozoa is independent of age. The age-associated decline in semen quality in dogs may instead result from molecular changes and disruptions in apoptosis during spermatogenesis and sperm maturation, without significant involvement of the oxidative stress pathway.
The observations in this study regarding spermatozoa and seminal plasma enzymatic antioxidants differ from the findings of Kelso et al.6 in bulls, where a significant reduction in seminal plasma and spermatozoa SOD and GPx levels was observed in older bulls (> 9 years of age). This discrepancy may be explained by the identical and relatively low levels of lipid peroxidation observed in both the senile and mature groups of dogs in the present study, suggesting comparable levels of antioxidant protection. In bulls, a reduction in antioxidant activity was linked to decreased concentrations of polyunsaturated and unsaturated fatty acids, such as arachidonic and docosahexaenoic acid, making spermatozoa more susceptible to peroxidative damage in older individuals6. Similarly, in senior mice, reduced antioxidant activity in sperm was associated with elevated ROS production and increased LPO13. Our findings highlight the species-specific differences in sperm metabolism and oxidative stress responses. Unlike bulls and mice, dogs may maintain consistent antioxidant levels and LPO across age groups, reflecting potential differences in sperm physiology and protection mechanisms.
The current study demonstrated that the semen of senile dogs is more susceptible to the freezing-thawing process than that of mature dogs. The immediate post-thaw sperm motility and kinematic parameters were significantly lower in senile dogs compared to mature dogs. This finding agrees with the report by Lechner et al.50who retrospectively analyzed data from frozen-thawed canine semen and found that the effects of cryopreservation on progressively motile sperm are more pronounced in dogs aged 10 and 11 years. Previous studies investigating the influence of aging on cryopreserved sperm motility have reported variable results, likely due to the inclusion of dogs ≥ 7 years in the senior group22,47,51. Consistent with Lechner et al.50our findings suggest that sperm quality and cryotolerance in dogs remain relatively stable throughout much of their lifespan but decline more noticeably in individuals aged nine years and older.
The ZBA provides insights into the fertilizing potential of spermatozoa26. Sperm–zona pellucida interaction is a critical step in fertilization, reflecting sperm competence in recognizing the oocyte52,53. This process begins with the interaction between receptors on the sperm membrane and glycoproteins of the zona pellucida27. Therefore, the ZBA can assess damage to molecular binding structures that routine sperm functional analyses may not detect. Additionally, the assay is useful in evaluating not only sperm damage caused by storage but also inherent dysfunctions in untreated spermatozoa that may contribute to reduced fertility26,54.
In the present study, significantly more spermatozoa from mature dogs bound to the zona pellucida compared to those from senile dogs. While this finding may suggest reduced fertility in senile dogs in vivo, caution is warranted in its interpretation. The capacity of spermatozoa to bind to the oocyte varies between replicates due to multiple factors, including oocyte quality, the duration of ovarian cryostorage, sperm population characteristics, and individual male variation27,55. In domestic animals, attempts to establish a correlation between sperm–zona binding ability and field fertility have often yielded inconsistent results26,56,57,58,59. The discrepancy between in vivo and in vitro fertility assessments is unsurprising, as the ZBA does not account for sperm transport within the female reproductive tract or post–zona binding events53,58.
Several factors influence sperm binding capacity to the zona pellucida, including in vitro capacitation, acrosomal status, and specific motile subpopulations26,53,60,61. Previous studies suggest that both acrosome-intact and acrosome-reacted dog spermatozoa can bind to the zona pellucida62and no correlation has been found between acrosomal status and sperm penetration of the oocyte63. Given the significant differences in the rapid and progressively motile sperm subpopulations between mature and senile dogs in this study, we propose that this factor may explain the observed differences in zona pellucida binding, as this subpopulation has been shown to exhibit superior responses to in vitro capacitation53.
Conclusions
In conclusion, no significant age-related changes in lipid peroxidation or enzymatic antioxidant activities were observed, suggesting that the observed decline in semen quality is not driven by increased susceptibility to oxidative stress. Furthermore, frozen-thawed spermatozoa from mature dogs exhibit a significantly higher zona pellucida-binding capacity than those from senile dogs, likely due to a greater proportion of progressively motile sperm subpopulations.
Materials and methods
Animals and experimental design
This study included 40 domestic dogs of various breeds presented for semen quality assessment at the Ambulatory of the Department of Reproduction and Clinic of Farm Animals, Faculty of Veterinary Medicine, Wroclaw University of Environmental and Life Sciences. Two groups of stud dogs were evaluated: the mature group (aged 2–4 years; 3 ± 0.2, n = 20) and the senile group (aged 9–11 years; 10 ± 0.2, n = 20), with a total of 107 ejaculates (2–3 per dog) collected for analysis. The dogs were similarly distributed among small, medium, and large breeds within each group: 8 small breeds (< 20 kg), 8 medium breeds (20–40 kg), and 4 large breeds (> 40 kg) in each group. All dogs were fed commercial diets without supplementation aimed at enhancing semen quality. Before semen collection, all dogs underwent a comprehensive examination, including ultrasonography to assess testicular and prostate health, confirming their reproductive health status. Semen was collected via preputial massage using a gloved hand in the absence of an estrous bitch. Ejaculates were obtained once per week from each dog. Semen collection during routine veterinary procedures was approved by the Animal Welfare Committee of Wroclaw University of Environmental and Life Sciences (Statement n. 15/2024). All experiments were conducted in compliance with the national Act on the Protection of Animals Used for Scientific or Educational Purposes (Dz. U. poz. 266) and are reported in accordance with the ARRIVE guidelines.
Additionally, semen from 10 mature (2–4 years; 3.3 ± 0.3 years) and 10 senile (9–11 years; 10.1 ± 0.2) dogs was cryopreserved and used in the zona pellucida binding assay (ZBA). Ovaries were collected from 20 bitches following routine ovariohysterectomy at the clinic and stored at -20 °C until use in the ZBA.
Experiment 1: assessment of sperm cell characteristics in fresh semen
Evaluation of spermatozoa
Immediately after collection, 10 µL of semen was diluted in Tris-citric acid-fructose extender, which consisted of Tris (hydroxymethyl)- aminomethane (0.2 M), citric acid monohydrate (0.06 M), fructose (0.05 M), and distilled water, and sperm concentration, TSC, and total motility were assessed using the Ceros II Computer-Assisted Sperm Analysis (CASA) system (Hamilton-Thorne Biosciences, Beverly, MA, USA). The software settings for the CASA system used in this study were presented in our previous study28. To evaluate sperm abnormalities, 200 spermatozoa were examined under a phase-contrast microscope (Nikon Eclipse E200). Morphological abnormalities were categorized as head defects, midpiece defects, tail defects, or the presence of cytoplasmic droplets64.
Evaluation of acrosome integrity
For the simultaneous evaluation of acrosome and plasma membrane integrity, 10 µL of peanut agglutinin (PNA; 1 µg/mL) working solution (Life Technologies Ltd., Grand Island, NY, USA) was added to 500 µL of diluted semen containing 8 × 106 spermatozoa. The mixture was incubated for 5 min at room temperature in the dark. Subsequently, 3 µL of propidium iodide (PI) (1 mg/mL solution in water; P3566, Thermo Fisher Scientific Inc.) was added before analysis to differentiate between live and dead spermatozoa65. All samples were analyzed using a Guava EasyCyte 5® flow cytometer (Merck KGaA, Darmstadt, Germany), with 10,000 events recorded per sample. Fluorescence detection was performed using an argon-ion laser (488 nm). Based on a simultaneous assessment of acrosome and membrane integrity, spermatozoa were classified into four categories: (i) intact acrosome and intact membrane (IAIM), (ii) damaged acrosome and intact membrane (DAIM), (iii) intact acrosome and damaged membrane (IADM), and (iv) damaged acrosome and damaged membrane (DADM). Furthermore, parameters for overall acrosome damage (DA) and membrane damage (DM) were analyzed by grouping spermatozoa as follows: DA = (DAIM + DADM) and DM = (IADM + DADM).
Lipid peroxidation
LPO was assessed using the fluorescent lipid probe C11-BODIPY581/591 (Life Technologies Ltd., Grand Island, NY, USA). This probe mimics fatty acids and integrates into the plasma membrane, emitting fluorescence upon lipid peroxidation. Under non-oxidized conditions, the probe emits red fluorescence; however, when subjected to oxidation, such as by peroxyl and alkoxyl radicals, the emission shifts to green fluorescence66. To conduct the assay, 1 µL of 2 mM C11-BODIPY581/591 in ethanol was added to the diluted sperm samples, resulting in a final probe concentration of 4 µM. The samples were incubated for 30 min at 37 °C in the dark. Following incubation, the samples were centrifuged at 500×g for 3 min, and the pellet was resuspended in 500 µL of Tris buffer to remove unbound or excess dye. To assess cell viability, samples were stained with PI and further incubated for 5 min at room temperature before cytometric analysis. Dot plots of C11-BODIPY 581/591/PI -stained spermatozoa revealed four distinct cell populations: (1) live spermatozoa without LPO (PI-BODIPYred), (2) live spermatozoa with LPO (PI-BODIPYgreen), (3) dead spermatozoa without LPO (PI + BODIPYred), and (4) dead spermatozoa with LPO (PI + BODIPYgreen). Green fluorescence was detected using an FL-1 detector, while red fluorescence was measured using an FL-3 detector67.
Sperm chromatin status
Chromatin status was evaluated using the sperm chromatin structure assay (SCSA) as described by Evenson68which exploits the metachromatic properties of Acridine Orange (AO) dye (Life Technologies Ltd., Grand Island, NY, USA). Initially, 200 µL of denaturation fluid (0.1% (v/v) Triton X-100, 0.15 M NaCl, and 0.08 M HCl, pH 1.4) was added to 100 µL of diluted sperm. After a 30-second incubation, 600 µL of AO solution (6 µg AO/mL in buffer containing 0.1 M citric acid, 0.2 M Na2HPO4, 1 mM EDTA, and 0.15 M NaCl, pH 6) was added. Spermatozoa with intact DNA exhibited green fluorescence, whereas those with fragmented DNA displayed red fluorescence, classified as DFI68. A distinct subgroup of spermatozoa exhibiting high DNA stainability (HDS) was also identified. The high DNA-stainable sperm are believed to have abnormally high DNA staining due to defective protamination, resulting in an increased amount of retained histones. These cells appeared as a separate population positioned to the right of the normal sperm population (FL-3) and above the population characterized by fragmented DNA.
Enzyme quantification
Seminal plasma was obtained by centrifuging each semen ejaculate at 1,500 × g for 15 min. Enzymatic antioxidant activity was analyzed from a single ejaculate per dog. The activities of SOD, CAT, and GPx in semen samples (both seminal plasma and spermatozoa) of dogs were assessed spectrophotometrically using a BioTek ELx800 Absorbance Microplate Reader (Biotek, Winooski, VT, USA). SOD (19160-1KT-F) and GPx (MAK437) activities were measured using commercial Sigma-Aldrich kits (Darmstadt, Germany) at wavelengths of 450 nm and 340 nm, respectively. CAT activity was quantified using the catalase colorimetric activity kit (EIACATC, Invitrogen, USA) at a wavelength of 540 nm. CAT activity was assessed based on the enzyme’s ability to catalyze the breakdown of hydrogen peroxide (H2O2) into water and oxygen. SOD activity was determined using the water-soluble tetrazolium salt WST-1[2-(4-iodophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2 H-tetrazolium, monosodium salt], which produces a water-soluble formazan dye upon reduction by superoxide anions. The rate of this reduction, which is proportional to xanthine oxidase (XO) activity, is inhibited by SOD. The IC50 (50% inhibition activity of SOD) was determined colorimetrically under the conditions specified by the manufacturer. GPx activity was measured by monitoring the oxidation of reduced glutathione (GSH) to oxidized glutathione (GSSG) by GPx. The resulting GSSG was subsequently reduced back to GSH through the action of glutathione reductase and nicotinamide adenine dinucleotide phosphate (NADPH), with NADPH consumption measured spectrophotometrically. The enzyme activities were expressed as follows: SOD activity as % inhibition rate, CAT activity as U/mL, and GPx activity as U/L, in accordance with the manufacturer’s guidelines.
Experiment 2: Zona pellucida binding assay (ZBA)
The ZBA was employed in this experiment to evaluate the ability of frozen-thawed spermatozoa from mature and senile dogs to bind to the zona pellucida. Frozen-thawed spermatozoa were utilized in this study because freshly collected semen underwent multiple quality assessments immediately after collection. Performing the ZBA several hours post-collection would have reduced semen viability. Additionally, the ZBA is a time-consuming procedure, making it impractical to conduct alongside other sperm functional tests on the same day. Furthermore, quality parameters of fresh semen could not be directly compared with frozen-thawed semen, as only approximately half of the senile dogs produced sufficient semen for both comprehensive quality assessments and cryopreservation.
Semen freezing
After semen collection and CASA evaluation, an aliquot containing approximately 200 × 10⁶ spermatozoa was aspirated and placed in a tube, then centrifuged at 500 × g for 5 min to remove seminal plasma. The resulting pellet was diluted in a Tris-Fructose-Equex extender with egg yolk and frozen according to the method described by Nizanski69, . Briefly, each extended semen sample was gradually cooled to 5 °C over 1 h, followed by the addition of 6% glycerol. After a 90-minute equilibration period at 5 °C, the samples were frozen in 0.5 mL straws at -140 °C using nitrogen vapor (5 cm above the liquid nitrogen surface) and subsequently stored in liquid nitrogen. Several months later, the samples were thawed at 37 °C in a water bath for 60 s for post-thaw motility and kinematic assessment, as well as for zona pellucida binding assay (ZBA).
Oocyte recovery
Ovaries from 20 bitches, aged 2 to 4 years and of various breeds, were collected following routine ovariohysterectomy procedures at the clinic. Among these, seven ovaries contained corpora lutea, three had follicles, and ten displayed no functional structures on the ovarian surface. The ovaries were rinsed with phosphate-buffered saline (PBS), immersed in 20 mL of NaCl solution, and stored at -20 °C. On the day of the experiment, ovaries were randomly selected and thawed at room temperature. For each trial, one pair of ovaries was designated for spermatozoa from mature and senile dogs. Cumulus-oocyte complexes (COCs) were retrieved by repeatedly slicing the ovaries in a Petri dish containing PBS with 0.5% (w/v) bovine serum albumin (BSA) under a stereomicroscope. Cumulus cells were removed by incubating the oocytes in 75 mmol/L sodium citrate for 15 min, followed by vortexing for an additional 15 min, as described by Strom Holst et al.26. The denuded oocytes were stored overnight at 4 °C in PBS containing 0.5% (w/v) BSA and used the following day for the ZBA.
Semen processing and ZBA
On the assay day, after thawing, semen was centrifuged at 300 × g for 5 min, and the supernatant was discarded. The sperm pellet was washed in 2 mL of Canine Capacitation Medium (CCM)70 by centrifugation at 300 × g for 5 min, followed by the removal of the supernatant. The pellet was then resuspended in 2.5 mL of CCM to achieve a final concentration of 40 × 10⁶ sperm/mL and incubated at 37 °C in 5% CO₂ for 2 h.
Forty-microliter droplets of CCM were prepared in four-well Petri dishes, with each well overlaid with 1.2 mL of mineral oil. Ten oocytes were added to each droplet and equilibrated at 37 °C in 5% CO₂ for at least 1 h, following the protocol described by Strom Holst et al.26 Subsequently, a 25 µL aliquot of the sperm suspension was diluted with 75 µL of CCM, and a 10 µL aliquot of this dilution (containing approximately 50 × 10³ spermatozoa) was added to each droplet27. The sperm-oocyte droplets were co-incubated at 37 °C in 5% CO₂ for 6 h.
Following incubation, sperm-oocyte complexes were transferred to 100 µL droplets of 1.5% (v/v) glutaraldehyde in 0.1 M sodium cacodylate buffer, fixed for 15 min, and stored in PBS containing 0.5% (w/v) BSA until evaluation. To remove loosely bound spermatozoa, sperm-oocyte complexes were washed by repeated pipetting in 100 µL droplets of PBS with 0.5% BSA. The oocytes were then stained with 10 µL of PI in 1 mL of PBS with 0.5% BSA for 10 min.
Stained sperm-oocyte complexes were placed on microscope slides and overlaid with a coverslip supported by four droplets of a Vaseline–paraffin mixture. Gentle pressure was applied to immobilize the oocytes in the medium. The number of spermatozoa strongly bound to the zona pellucida was determined using a fluorescence microscope equipped with a 488 nm excitation laser (100 mW output). Observations were performed at 400× magnification. Multiple optical sections were captured at 10 μm intervals for each oocyte. Images were compiled into a single visual layer using OLYMPUS cellSens Entry 1.18 software, and strongly bound spermatozoa were counted.
Statistical analysis
Data analysis was conducted using Jamovi version 2.3.21 (The Jamovi project, 2022). Statistical evaluation of all semen parameters was performed using repeated measures ANOVA, with age group specified as the between-subject factor, while individual dog and age were included as covariates. Ejaculate number was modeled as the within-subject factor. Data normality was assessed using the Shapiro-Wilk test, and non-normally distributed variables were log-transformed to meet parametric assumptions. Group comparisons of mean SOD, CAT, and GPx levels were performed using independent samples t-tests. Statistical significance was defined at P ≤ 0.05, with exact P-values reported unless P < 0.001. Figures present scatter plots of individual ejaculates alongside corresponding mean values.
Data availability
The data that support the findings in this study are available upon reasonable request from the corresponding author [Kenneth Owoicho Abah].
Change history
28 October 2025
The original online version of this Article was revised: The Funding section in the original version of this Article was incomplete. The Funding section now reads: "This work was supported by Wroclaw University of Environmental and Life Sciences (Poland) under the Ph.D. research programme “Doctoral student grant at the Doctoral School of Wroclaw University of Environmental and Life Sciences” no. N020/0014/2022, and the Polish National Agency for Academic Exchange under grant number PPI/APM/2019/1/00044/U/ 00001. The APC is financed by Wrocław University of Environmental and Life Sciences". The original Article has been corrected.
References
Aitken, R. J. Impact of oxidative stress on male and female germ cells: implications for fertility. Reproduction 159, R189–R201 (2020).
Balić, I. M., Milinković-Tur, S., Samardžija, M. & Vince, S. Effect of age and environmental factors on semen quality, glutathione peroxidase activity and oxidative parameters in Simmental bulls. Theriogenology 78, 423–431 (2012).
Halvaei, I., Litzky, J. & Esfandiari, N. Advanced paternal age: effects on sperm parameters, assisted reproduction outcomes and offspring health. Reprod. Biol. Endocrinol. 18, 110 (2020).
Vince, S. et al. Age-related differences of semen quality, seminal plasma, and spermatozoa antioxidative and oxidative stress variables in bulls during cold and warm periods of the year. Animal 12, 559–568 (2018).
Zmudzinska, A., Wisniewski, J., Mlynarz, P., Olejnik, B. & Mogielnicka-Brzozowska, M. Age-dependent variations in functional quality and proteomic characteristics of canine (Canis lupus familiaris) epididymal spermatozoa. Int. J. Mol. Sci. 23, 9143 (2022).
Kelso, K. A., Redpath, A., Noble, R. C. & Speake, B. K. Lipid and antioxidant changes in spermatozoa and seminal plasma throughout the reproductive period of bulls. J. Reprod. Fertil. 109, 1–6 (1997).
Koh, S.-A., Sanders, K. & Burton, P. Effect of male age on oxidative stress markers in human semen. J. Reprod. Biotechnol. Fertil. 5, 2058915816673242 (2016).
Nguyen-Powanda, P. & Robaire, B. Oxidative stress and reproductive function in the aging male. Biology (Basel) 9, 282 (2020).
Trevizan, J. T. et al. Does lipid peroxidation and oxidative DNA damage differ in cryopreserved semen samples from young, adult and aged Nellore bulls?. Anim. Reprod. Sci. 195, 8–15 (2018).
Zubkova, E. V., Wade, M. & Robaire, B. Changes in spermatozoal chromatin packaging and susceptibility to oxidative challenge during aging. Fertil. Steril. 84(Suppl 2), 1191–1198 (2005).
Zubkova, E. V. & Robaire, B. Effects of ageing on spermatozoal chromatin and its sensitivity to in vivo and in vitro oxidative challenge in the Brown Norway rat. Hum. Reprod. 21, 2901–2910 (2006).
Ozkosem, B., Feinstein, S. I., Fisher, A. B. & O’Flaherty, C. Advancing age increases sperm chromatin damage and impairs fertility in peroxiredoxin 6 null mice. Redox. Biol. 5, 15–23 (2015).
Weir, C. P. & Robaire, B. Spermatozoa have decreased antioxidant enzymatic capacity and increased reactive oxygen species production during aging in the Brown Norway rat. J. Androl. 28, 229–240 (2007).
Paul, C., Nagano, M. & Robaire, B. Aging results in differential regulation of DNA repair pathways in pachytene spermatocytes in the Brown Norway rat. Biol. Reprod. 85, 1269–1278 (2011).
Selvaratnam, J., Paul, C. & Robaire, B. Male rat germ cells display age-dependent and cell-specific susceptibility in response to oxidative stress challenges. Biol. Reprod. 93, 72 (2015).
Januskauskas, A., Johannisson, A. & Rodriguez-Martinez, H. Subtle membrane changes in cryopreserved bull semen in relation with sperm viability, chromatin structure, and field fertility. Theriogenology 60, 743–758 (2003).
Roldan, E. R. & Harrison, R. A. Diacylglycerol in the exocytosis of the mammalian sperm acrosome. Biochem. Soc. Trans. 21, 284–289 (1993).
Fujii, J., Iuchi, Y., Matsuki, S. & Ishii, T. Cooperative function of antioxidant and redox systems against oxidative stress in male reproductive tissues. Asian J. Androl. 5, 231–242 (2003).
de Lamirande, E., Jiang, H., Zini, A., Kodama, H. & Gagnon, C. Reactive oxygen species and sperm physiology. Rev. Reprod. 2, 48–54 (1997).
Makker, K., Agarwal, A. & Sharma, R. Oxidative stress & male infertility. Indian J. Med. Res. 129, 357–367 (2009).
Aitken, R. J. A free radical theory of male infertility. Reprod. Fertil. Dev. 6, 19–23 (1994).
Hesser, A. et al. Semen evaluation and fertility assessment in a purebred dog breeding facility. Theriogenology 87, 115–123 (2017).
Ligocka, Z., Partyka, A., Bonarska-Kujawa, D., Mucha, A. & Niżański, W. Addition of low concentration of cholesterol-loaded cyclodextrin (CLC) has a positive effect on cryopreserved canine spermatozoa evaluated by andrological and biophysical methods. BMC Vet. Res. 20, 7 (2024).
Kumaresan, A., Johannisson, A., Al-Essawe, E. M. & Morrell, J. M. Sperm viability, reactive oxygen species, and DNA fragmentation index combined can discriminate between above- and below-average fertility bulls. J. Dairy Sci. 100, 5824–5836 (2017).
Narud, B. et al. Differences in sperm functionality and intracellular metabolites in Norwegian Red bulls of contrasting fertility. Theriogenology 157, 24–32 (2020).
Ström Holst, B., Larsson, B., Linde-Forsberg, C. & Rodriguez-Martinez, H. Evaluation of chilled and frozen-thawed canine spermatozoa using a zona pellucida binding assay. J. Reprod. Fertil. 119, 201–206 (2000).
Ligocka, Z., Partyka, A., Schäfer-Somi, S., Mucha, A. & Niżański, W. Does better post-thaw motility of dog sperm frozen with CLC mean better zona pellucida binding ability?. Animals (Basel) 13, 1580 (2023).
Abah, K. O. et al. Deciphering the mechanism of sperm cell aging – Functional, structural, and kinematic characteristics in fresh semen of mature and aged dogs. Anim. Reprod. Sci. 274, 107798 (2025).
Johnson, S. L., Dunleavy, J., Gemmell, N. J. & Nakagawa, S. Consistent age-dependent declines in human semen quality: a systematic review and meta-analysis. Ageing Res. Rev. 19, 22–33 (2015).
Molina, R. I. et al. Semen quality and aging: analysis of 9.168 samples in Cordoba. Argentina. Arch Esp Urol 63, 214–222 (2010).
Pino, V., Sanz, A., Valdés, N., Crosby, J. & Mackenna, A. The effects of aging on semen parameters and sperm DNA fragmentation. JBRA Assist. Reprod. 24, 82–86 (2020).
Verón, G. et al. Impact of age, clinical conditions, and lifestyle on routine semen parameters and sperm kinematics. Fertil. Steril. 110, 68-75.e4 (2018).
Hendrikse, J. & Antonisse, H. W. Evaluation of dog sperm. Tijdschr Diergeneeskd 109, 171–174 (1984).
Rijsselaere, T., Maes, D., Hoflack, G., de Kruif, A. & Van Soom, A. Effect of body weight, age and breeding history on canine sperm quality parameters measured by the Hamilton-Thorne analyser. Reprod. Domest. Anim. 42, 143–148 (2007).
Angrimani, D. S. R. et al. Cytoplasmic droplet acting as a mitochondrial modulator during sperm maturation in dogs. Anim. Reprod. Sci. 181, 50–56 (2017).
Cooper, T. G. Cytoplasmic droplets: the good, the bad or just confusing?. Hum. Reprod. 20, 9–11 (2005).
Xu, H., Yuan, S.-Q., Zheng, Z.-H. & Yan, W. The cytoplasmic droplet may be indicative of sperm motility and normal spermiogenesis. Asian J. Androl. 15, 799–805 (2013).
Cooper, T. G. & Yeung, C.-H. Acquisition of volume regulatory response of sperm upon maturation in the epididymis and the role of the cytoplasmic droplet. Microsc. Res. Tech. 61, 28–38 (2003).
Cortadellas, N. & Durfort, M. Fate and composition of cytoplasmic droplet of hamster epididymal spermatozoa. J. Morphol. 221, 199–210 (1994).
Oko, R., Hermo, L., Chan, P. T. K., Fazel, A. & Bergeron, J. The cytoplasmic droplet of rat epididymal spermatozoa contains saccular elements with Golgi characteristics. J. Cell. Biol. 123, 809–821 (1993).
Yuan, S., Zheng, H., Zheng, Z. & Yan, W. Proteomic analyses reveal a role of cytoplasmic droplets as an energy source during epididymal sperm maturation. PLoS ONE 8, e77466 (2013).
Beckman, K. B. & Ames, B. N. The free radical theory of aging matures. Physiol. Rev. 78, 547–581 (1998).
Cocuzza, M. et al. Age-related increase of reactive oxygen species in neat semen in healthy fertile men. Urology 71, 490–494 (2008).
da Silva, C. M. B. et al. Melatonin reduces lipid peroxidation and apoptotic-like changes in stallion spermatozoa. J. Pineal. Res. 51, 172–179 (2011).
Partyka, A., Rodak, O., Bajzert, J., Kochan, J. & Niżański, W. The effect of L-carnitine, hypotaurine, and taurine supplementation on the quality of cryopreserved chicken semen. Biomed. Res. Int. 2017, 7279341 (2017).
Rikans, L. E. & Hornbrook, K. R. Lipid peroxidation, antioxidant protection and aging. Biochim. Biophys. Acta 1362, 116–127 (1997).
Brito, M. M. et al. Effect of senescence on morphological, functional and oxidative features of fresh and cryopreserved canine sperm. Aging Male 23, 279–286 (2020).
Ahmed, S., Khan, M.I.-R., Ahmad, M. & Iqbal, S. Effect of age on lipid peroxidation of fresh and frozen-thawed semen of Nili-Ravi buffalo bulls. Ital. J. Anim. Sci. 17, 730–735 (2018).
Neagu, V. R. et al. Determination of glutation peroxidase and superoxide dismutase activities in canine seminal plasma and its relation with sperm quality and lipid peroxidation post thaw. Theriogenology 75, 10–16 (2011).
Lechner, D., Aurich, J., Schäfer-Somi, S. & Aurich, C. Effects of age, size and season on cryotolerance of dog semen – A retrospective analysis. Anim. Reprod. Sci. 236, 106912 (2022).
de la Fuente-Lara, A., Hesser, A., Christensen, B., Gonzales, K. & Meyers, S. Effects from aging on semen quality of fresh and cryopreserved semen in Labrador retrievers. Theriogenology 132, 164–171 (2019).
Oehninger, S., Franken, D. R. & Ombelet, W. Sperm functional tests. Fertil. Steril. 102, 1528–1533 (2014).
Peña, A. I., Adán, S., Quintela, L. A., Becerra, J. J. & Herradón, P. G. Relationship between motile sperm subpopulations identified in frozen-thawed dog semen samples and their ability to bind to the zona pellucida of canine oocytes. Reprod. Domest. Anim. 53, 14–22 (2018).
Mayenco-Aguirre, A. M. & Pérez Cortés, A. B. Preliminary results of hemizona assay (HZA) as a fertility test for canine spermatozoa. Theriogenology 50, 195–204 (1998).
Peña, A. I., Barrio, M., Becerra, J. J., Quintela, L. A. & Herradón, P. G. Infertility in a dog due to proximal cytoplasmic droplets in the ejaculate: investigation of the significance for sperm functionality in vitro. Reprod. Domest. Anim. 42, 471–478 (2007).
Braundmeier, A. G., Demers, J. M., Shanks, R. D., Saacke, R. G. & Miller, D. J. Examination of the binding ability of bovine spermatozoa to the zona pellucida as an indicator of fertility. J. Androl. 23, 645–651 (2002).
Codde, J. M. & Berger, T. In vivo fertility of rams in relation to sperm-zona pellucida binding and sperm-zona pellucida penetration of ovine oocytes. Theriogenology 44, 901–906 (1995).
Collins, E. D., Flowers, W. L., Shanks, R. D. & Miller, D. J. Porcine sperm-zona binding ability as an indicator of fertility. Anim. Reprod. Sci. 104, 69–82 (2008).
Waberski, D. et al. Importance of sperm-binding assays for fertility prognosis of porcine spermatozoa. Theriogenology 63, 470–484 (2005).
Franken, D. R. New aspects of sperm-zona pellucida binding. Andrologia 30, 263–268 (1998).
Topper, E. K., Killian, G. J., Way, A., Engel, B. & Woelders, H. Influence of capacitation and fluids from the male and female genital tract on the zona binding ability of bull spermatozoa. J. Reprod. Fertil. 115, 175–183 (1999).
Kawakami, E., Vandevoort, C. A., Mahi-Brown, C. A. & Overstreet, J. W. Induction of acrosome reactions of canine sperm by homologous zona pellucida. Biol. Reprod. 48, 841–845 (1993).
Hewitt, D. A. & England, G. C. The canine oocyte penetration assay; its use as an indicator of dog spermatozoal performance in vitro. Anim. Reprod. Sci. 50, 123–139 (1998).
Blom, E. The ultrastructure of some characteristic sperm defects and a proposal for a new classification of the bull spermiogram (author’s transl). Nord. Vet. Med. 25, 383–391 (1973).
Graham, J. K. Assessment of sperm quality: a flow cytometric approach. Anim. Reprod. Sci. 68, 239–247 (2001).
Drummen, G. P. C., van Liebergen, L. C. M., Op den Kamp, J. A. F. & Post, J. A. C11-BODIPY(581/591), an oxidation-sensitive fluorescent lipid peroxidation probe: (micro)spectroscopic characterization and validation of methodology. Free Radic. Biol. Med. 33, 473–490 (2002).
Partyka, A. et al. Flavone and 3-hydroxyflavone supplementation in cryopreservation medium protects canine sperm against apoptosis and lipid peroxidation. Theriogenology 226, 319–327 (2024).
Evenson, D. P. The sperm chromatin structure assay (SCSA(®)) and other sperm DNA fragmentation tests for evaluation of sperm nuclear DNA integrity as related to fertility. Anim. Reprod. Sci. 169, 56–75 (2016).
Nizański, W. Intravaginal insemination of bitches with fresh and frozen-thawed semen with addition of prostatic fluid: use of an infusion pipette and the Osiris catheter. Theriogenology 66, 470–483 (2006).
Mahi, C. A. & Yanagimachi, R. Capacitation, acrosome reaction, and egg penetration by canine spermatozoa in a simple defined medium. Gamete. Res. 1, 101–109 (1978).
Acknowledgements
The authors would like to thank Barbara Smalec of the Andrology Laboratory, Department of Reproduction and Clinic of Farm Animals, Faculty of Veterinary Medicine, Wroclaw University of Environmental and Life Sciences, for her invaluable assistance during the experiment and sample processing. Appreciation also goes to Patricia Kalak for her support during semen collection.
Funding
This work was supported by Wroclaw University of Environmental and Life Sciences (Poland) under the Ph.D. research programme “Doctoral student grant at the Doctoral School of Wroclaw University of Environmental and Life Sciences” no. N020/0014/2022, and the Polish National Agency for Academic Exchange under grant number PPI/APM/2019/1/00044/U/ 00001. The APC is financed by Wrocław University of Environmental and Life Sciences.
Author information
Authors and Affiliations
Contributions
KOA: Writing—original draft, Resources, Methodology, Formal analysis, Funding acquisition, Data curation. ZLK: Resources, Investigation, Writing—review & editing. AP: Writing—review & editing, Resources. SP: Resources, Investigation, Writing—review & editing. AF: Supervision, review. WN: Writing—review & editing, Supervision, Methodology, Funding acquisition, Conceptualization.All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Abah, K.O., Ligocka-Kowalczyk, Z., Partyka, A. et al. Exploring the characters of sperm lipid peroxidation, enzymatic antioxidant activity, and spermatozoa zona binding capacity with aging in dogs. Sci Rep 15, 30976 (2025). https://doi.org/10.1038/s41598-025-16410-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-16410-w