Abstract
In bean beetles (Callosobruchus maculatus), population density affects larval development. Under high density, individuals develop into a dispersal morph, with larger wings and smaller gonads, while under low density we observe the flightless morph with smaller wings and larger gonads. Past research has shown that the effect of density on wing size is stronger in males than in females, and we hypothesized that this difference is caused by a sex-specific trade-off between immunocompetence and reproduction. We predicted that females reared under high larval density would have an overall stronger immune response compared to those reared in low densities, and to males in general. We tested this hypothesis by rearing males and females under different density treatments and measuring wing size, gonad size, and immune response through phenoloxidase and lytic activity. Females reared under the high-density treatment displayed stronger immune responses, and a trade-off between immunity and gonad size. This provides evidence for a sex-specific, density-specific trade-off between immunocompetence and reproduction.
Similar content being viewed by others
Introduction
Trade-offs are a central concept to life history theory, arising because condition-dependent plastic mechanisms and developmental constraints allow for differential trait investment that can produce negative covariances between traits that compete for resources1,2,3. Since ecological perturbations affect differential trait investment4,5,6,7,8,9previous work implicates trade-offs, and their underlying plastic mechanisms, as adaptations optimizing the fitness of individuals across environments7,10,11,12. Immunity is an important life history trait as it affects survivorship, lifespan and ultimately fitness13. On the other hand, maintaining an immune system and mounting an immune response is energetically expensive, which may lead to trade-offs between immunity and other life history traits13. Trade-offs involving immunocompetence and other life history traits such as growth and reproduction have been substantially documented in the literature in a variety of taxa14,15,16,17,18.
Differential allocation to immunity traits may result from environmental factors that impact patterns of infection. For example, the density-dependent prophylaxis hypothesis argues that optimal expression of immunity-related traits is expected to vary with population density: Higher densities may favor increased investment in immune response as crowded environments favor increased rates of disease transmission19,20. Sex differences in life history can also cause differences in immunocompetence, leading to variation in resource allocation and resulting trade-offs between immunity and other costly traits such as reproduction. Specifically, as in most species, sexual selection is stronger in males21,22,23selection in males is expected to favor traits that will give them an advantage in direct or indirect mate competition at the cost of longevity. On the other hand, selection in females should favor increased longevity and fecundity3. This pattern has indeed been demonstrated in many taxa, including insects24,25,26.
This study addresses the effects of sex and the social environment on life-history trade-offs between immunocompetence, reproduction and dispersal traits in the bean beetle (Callosobruchus maculatus). Immunocompetence can be defined as an individual’s ability to resist infection by a pathogen. In insects, different types of pathogens trigger different types of immune responses, and immunocompetence can be measured through different components of the immune system. Multicellular pathogens typically trigger the encapsulation response, where the pathogen is entrapped by haemocytes and subsequently cleared27,28,29. After capsule formation, melanin is deposited within and around the capsule, a result of a biochemical cascade trigged by the enzyme phenoloxidase30. Bacterial infection triggers the lytic response, where a pathogen is killed through lysis. The disruption of the pathogen’s cell membrane is accomplished by antimicrobial peptides (AMPs), which are activated by immune signaling pathways31. Both encapsulation and lytic response abilities have important fitness consequences in a number of species27,29,32,33.
A previous study in C. maculatus demonstrated that variation in larval density during development affects differential allocation to both reproductive and dispersal traits, leading to differences in trade-offs: individuals reared under high density invested more in dispersal traits (wings) and less in reproduction (gonad size), and vice-versa34. Interestingly, there were sex differences in trait covariances and individual response to larval density: density affected gonad size similarly in both sexes, but the effect size of density on wing length was substantially larger in males than in females34. In other words, while we observe a clear trade-off between gonad and wing size in males, variation in wing size between density treatments does not fully explain variation in ovariole size in females. It should be noted that these differences in ovariole size do not result from differences in resource acquisition, since adult body size did not vary between larval density treatments. Gonad size in females may therefore be limited by allocation to another competing life-history trait, such as immunocompetence. Here we tested the hypothesis that there is a sex-specific trade-off between immunocompetence and fecundity in C.maculatus females. This hypothesis generated the following predictions: (1) immunocompetence will be overall lower in males than in females regardless of density; (2) females reared under high larval densities will have stronger immunocompetence and smaller gonads than those reared under lower densities and (3) we will find a trade-off between immunocompetence and gonad size in females, but not in males. For a comprehensive quantification of immunocompetence, we measured two different components of the immune system: encapsulation, through a phenoloxydase (PO) activity assay, and lytic response, through a lytic activity assay. Therefore, we also investigated whether there are trade-offs between these two immune responses, or whether they would be positively correlated. As males are expected to allocate overall fewer resources to immunocompetence, we predicted a negative covariance between PO and lytic activity in males and a positive covariance in females.
Results
Effects of sex and larval density in immune response
When we examined the effects of sex and larval density in immune response, we found significant effects of sex (Two-Way ANOVA, F1,127= 64.72, p < 0.0001) and larval density (F1,127= 22.98, p < 0.0001) on PO activity, where it was highest in females reared under higher larval density (Fig. 1a). We also found a significant effect of sex (F1,78=13.06, p = 0.0002) but not larval density (F1, 78=1.27, p = 0.26) on lytic activity, which was highest on females (Fig. 1b). We did not find significant sex*larval density interactions on either PO activity (F1, 127=1.86, p = 0.17) or lytic activity (F1,78=0.21, p = 0.65, Fig. 1).
Effects of larval density level and sex on two components of immune response: PO activity (A) and lytic activity (B). Females are represented in red and males in gray. Hatched colors represent low density and solid colors represent high density. There is a significant sex*larval density interaction effect on PO activity, and significant effects of sex and larval density on lytic activity.
Covariance between gonad size and immune response
Larval density had a significant effect on gonad size in both females (Fig. 2a) and males (Fig. 2b), where individuals reared under high larval density had smaller gonads (t-test, females: t59 = 17.44, p < 0.0001; males: t61 = 54.36, p < 0.0001). When we examined the covariance between gonad size and immune response, we found a significant interaction between gonad size and larval density on female PO activity (F1,55= 7.75, p = 0.007): there was a negative covariance between gonad size and PO activity in females reared under the high-density treatment, but no covariance in low-density females (Fig. 3a). We did not find significant covariances between gonad size and lytic activity in females (density: F1,28= 0.0007, p = 0.98, relative ovariole size: 1,28= 0.28, p = 0.59, density*relative ovariole size: F1,28= 0.01, p = 0.91, Fig. 3b). In males, we did not find significant effects of gonad size in PO activity (density: F1,59=2,44, p = 0.12, testes size: F1,59=0.008, p = 0.93, density*testes size: F1,59=1.1, p = 0.29, Fig. 3c). or lytic activity (density: F1,39=0.08, p = 0.77, relative testes size: F1,39=0.03, p = 0.85, density*relative testes size: F1,39=0.14, p = 0.71, Fig. 3d).
Effects of density on relative gonad size of females (A) and males (B). Females are represented in red and males in gray. Hatched colors represent low density and solid colors represent high density. In both sexes, individuals raised under high larval density developed smaller gonads. Asterisks show significant effects (****p < 0.0001).
Effect of density on the covariance between immune response and gonad size in females (A, B) and males C, D). Females are represented in red and males in gray. Hatched lines and triangles represent low density and solid lines and circles represent high density. Females reared under high larval density showed a negative covariance between PO activity and ovariole size.
Covariance between different components of immunity
We did not find a significant covariance between the two components of immunity we measured, PO and lytic activity, in females (density: F1,30= 19.76, p=0.0001, lytic activity: F1,30= 0.49, p=0.49, density* lytic activity: F1,30= 2.75, p=0.11, Fig. 4a) or males (density: F1,43=4.17, p=0.04, lytic activity: F1,43=0.65, p=0.42, density* lytic activity: F1,43=0.46, p=0.5, Fig. 4b).
Effect of density on the covariance between two components of immune response, PO activity and lytic activity, in females (A) and males (B). Hatched lines and triangles represent low density and solid lines and circles represent high density. There is no significant covariance between the two components of immunity in either sex.
Discussion
The results presented here support the hypothesis of a sex-specific trade-off between immunity and reproduction in C. maculatus. We found that females had a higher overall immune response compared to males. Additionally, females reared under a high larval density had higher immunocompetence measured as PO activity than low-density females and than males reared under either larval density. These high-density females also developed smaller gonads, and gonad size negatively covaried with PO activity. These results support the density-dependent prophylaxis hypothesis, as we observed an increased immune response of females reared under high larval density, a social context where there is higher risk of disease transmission20. These findings are also in agreement with several previous studies that found support for the density-dependent prophylaxis hypothesis, particularly in insects where density-dependent polyphenism is present35,36,37,38. Interestingly, we only observed an effect of density in immunity on females, suggesting sex differences in trade-offs involving immune responses, and mirroring results from previous studies in sand crickets39,40.
In studies of life history trade-offs, immunity is a trait of particular interest due to being energetically costly24 and several previous studies have reported sex differences in immunity where females typically have higher immune responses. In insects, stronger immunity in females may be adaptive because reproduction suppresses immune response. For example, female immune function has been found to be lower during mating in the mealworm beetle Tenebrio molitor, where copulation triggers the release of the hormone ecdysteroid, which suppresses the immune system41. Male seminal fluid has also been found to contain proteins that suppress female immune responses42. In Drosophila melanogaster, seminal fluid proteins bind to the surface of immune cells and inhibit their lytic activity; the Acp36DE seminal protein has been found to prevent Toll pathway activation42. This suppression of immunity is adaptive to males as sperm might otherwise be attacked by the female immune system, but may otherwise negatively impact female fitness by increasing susceptibility to pathogens. This widespread pattern may explain why there is a higher baseline immunity in females, especially females in dense populations where disease transmission rates are higher.
Additionally, hormones play an important role in sex-specific life history strategies. In C. maculatus, males and females experience different gene expression for hormonal pathways, including ecdysone and juvenile hormone; both of these hormones are involved in reproduction and immunity43. Juvenile hormones in particular are likely to play a role in the trade-offs described here, as they have been implicated in the generation of dispersal and non-dispersal morphs, and in trade-offs between wing and gonad size, in wing-polymorphic insects44. Juvenile hormones have also been shown to downregulate immune function in both males and females41 and these roles of juvenile hormones are highly conserved in insects.
The density effects in immune response revealed here are consistent with predictions in light of disease transmission models. Under high densities, an increased baseline immunocompetence is expected to be found due to the risk of disease transmission. Notably, we only found a trade-off between PO activity and gonad size in females reared under high densities: there is no covariance between these traits in individuals reared under low density. The absence on this trade-off under low density may be due to overall lower allocation to immunity under this condition, because it may not necessarily compete in energy allocation with other life-history traits. Additionally, under lower competition levels, individuals may have higher resource acquisition under low densities, which would explain the absence of trade-offs. Ultimately, these results illustrate sex differences in immunity, trade-offs and life history strategies.
Methods
Study species
The bean beetle Callosobruchus maculatus feeds and develops exclusively on dry beans. Mated females adhere eggs to the surface of dry beans, and larvae burrow into the bean after eclosion45,46 where they undergo a series of molts and pupation, and later on, winged adults emerge. They are capital breeders; larvae are restricted to the bean where their egg was oviposited and feeding only occurs during the larval stage47. Therefore, all of the resources to be allocated to different traits are fixed at adulthood, when they have a limited lifespan of 10–14 days46. Larval density is known to induce differential resource allocation in this species34,48. The individuals used in this experiment come from a laboratory population of Callosobruchus maculatus adapted to cowpeas (Vigna unguiculata) started in 2018 at Lake Forest College in Lake Forest, Illinois, USA. This population is housed in 4-liter plastic containers with vented lids in a growth chamber at 27 C and 12:12 h dark: light photoperiod. Lake Forest College does not require research performed in invertebrates to be reviewed by a regulatory body, so no permits were required.
Experimental setup
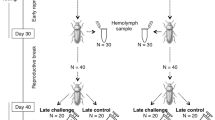
To test the hypothesis of a sex-specific trade-off between immune function and gonad size, we reared beetles under two larval density treatments: low and high. These treatments correspond to up to 3 eggs per bean and over 3 eggs per bean respectively, and are known to induce differential allocation to wing and gonad size in adults34. We set up these treatments by placing a single female and a cowpea from the laboratory population in each well of a clear 48-well cell culture plates, for a total of 12 plates. These females were allowed to lay eggs in isolation for 72 h, at which point they had produced more than 13 eggs per bean. We then removed the females and haphazardly assigned the beans to high- and low-density treatments. We then removed some eggs from the beans to match the two density treatments. We allowed these eggs to develop to adulthood in a climate-controlled growth chamber (Power Scientific environmental chamber DS27SD) at 27 °C and 12:12 dark: light photoperiod. We checked the plates daily for adult emergences to ensure that virgin individuals were collected the day they emerged, allowing us to standardize the age and mating status of experimental individuals.
Measuring allocation to immunity
Haemolymph collection and immunoassay protocols were adapted from methods described in Dougherty et al.49. We collected the first adult to emerge from each well, weighed them (Mettler-Toledo XP26 microbalance), and immediately euthanized them by freezing at -20 C. At this point, we separated the thorax for haemolymph collection and stored the abdomen, head, legs and wings in ethanol for later dissection. We crushed the thorax in a microcentrifuge tube with 20 uL of PBS buffer and centrifugated for 10 min at 13.1 RPM. We stored the supernatant at – 80 °C to be used for subsequent immune assays. We measured two components of inmmune response: phenoloxydase activity and lytic activity.
For the phenoloxydase assay, we added 10 uL of thawed haemolymph sample, 100 uL of PBS buffer and 90 uL of 8 mm dopamine hydrochloride (Sigma-Aldrich H8502) into each well of a 96-well microtiter plate. We loaded the plate into a plate reader (BioTek Microplate Reader) and measured the absorbance of the samples at 494 nm every 5 min for 30 min to estimate PO Vmax as the maximum linear rate of substrate conversion during this period.
For the lytic activity assay, we made a 5 mg/mL Micrococcus luteus (Sigma Aldrich M3770) suspension in tryptic soy broth with 15 ug/mL streptomycin sulfate and 1% agar. We poured this sterile solution into petri plates, and once the agar solidified, we punched two wells with a Pasteur pipette and added 2 uL of thawed haemolyph sample into each well. We incubated the plates at 33 C for 36 h, when we then imaged the areas around the wells under 10x magnification. We measured the zone of growth inhibition around each well using the software ImageJ version 1.53 (available at www.imagej.net)50.
Measuring allocation to morphological traits
We dissected experimental individuals with FST fine forceps under Leica MZ60-series stereomicroscopes and mounted specimens in DMHF (2,5-dimethyl-4-hydroxy-3(2 H)-furanone) resin. We mounted and measured the following appendages to test for overall differences in body size: antennae, elytra and hind tibia. To measure allocation to dispersal and reproduction, we dissected and measured hind wings and gonads (ovarioles and testes) respectively. We imaged the mounted specimes using a Leica MZ16A stereoscope with a Leica DFC429 digital camera and Leica Application Suite (LAS) imaging software, version 2.0 (available at https://www.leica-microsystems.com)51. We then measured these body parts with ImageJ version 1.53 (available at www.imagej.net)50 following the protocol in34.
Statistical analysis
We performed data transformations on all immunity data to normalize variance using the bestNormalize R package52and used an orderNorm transformation for PO activity and a standardized double-reversed Log_b(x + a) transformation for lytic activity52. We then performed Two-Way ANOVAs to test for the effects of sex, larval density and sex*larval density interaction on immune response.
To correct gonad size for body size, we used the residuals from a linear regression of body mass and gonad size. Then, we conducted two t-tests to test for the effect of density on gonad size of males and females. Finally, we performed linear models to test (1) how gonad size covaries with immune response under different larval density treatments and (2) how the different components of immune response (PO activity and lytic activity) covary under different larval density treatments. All statistical analyses were performed using JMP statistical software version 13 (available at https://www.jmp.com)53.
Data availability
The data supporting this article have been uploaded as supplementary material.
References
Noordwijk, A. J. & van de Jong, G. Acquisition and allocation of resources : their influence on variation in life history tactics. Am. Nat. 128, 137–142 (1986).
Stearns, S. C. Trade-offs in life-history evolution. Funct. Ecol. 3, 259–268 (1989).
Roff, D. A. Life History Evolution. (2002).
J Emlen, D. Environmental control of Horn length dimorphism in the beetle onthophagus acuminatus (Coleoptera: Scarabaeida). Proc. R Soc. Lond. B Biol. Sci. 256, 131–136 (1994).
Moczek, A. P. & Emlen, D. J. Male Horn dimorphism in the scarab beetle, Onthophagus taurus: do alternative reproductive tactics favour alternative phenotypes? Anim. Behav. 59, 459–466 (2000).
Braendle, C., Friebe, I., Caillaud, M. C. & Stern, D. L. Genetic variation for an aphid wing polyphenism is genetically linked to a naturally occurring wing polymorphism. Proc. R Soc. B Biol. Sci. 272, 657–664 (2005).
Yamane, T., Okada, K., Nakayama, S. & Miyatake, T. Dispersal and ejaculatory strategies associated with exaggeration of weapon in an armed beetle. Proc. R Soc. B Biol. Sci. 277, 1705–1710 (2010).
Smallegange, I. M., Deere, J. A. & Coulson, T. Correlative changes in life-history variables in response to environmental change in a model organism. Am. Nat. 183, 784–797 (2014).
Katsuki, M. & Lewis, Z. A trade-off between pre- and post-copulatory sexual selection in a bean beetle. Behav. Ecol. Sociobiol. 69, 1597–1602 (2015).
Johnson, T. L., Symonds, M. R. E. & Elgar, M. A. Anticipatory flexibility: larval population density in moths determines male investment in antennae, wings and testes. Proc. R. Soc. B Biol. Sci. 284, (2017).
Peterson, M. L., Doak, D. F. & Morris, W. F. Both life-history plasticity and local adaptation will shape range-wide responses to climate warming in the tundra plant Silene acaulis. Glob Change Biol. 24, 1614–1625 (2018).
Snell-Rood, E. C. & Moczek, A. P. Insulin signaling as a mechanism underlying developmental plasticity: the role of FOXO in a nutritional polyphenism. PLoS One 7.4, e34857 (2012).
Brommer, J. E. The evolution of fitness in life-history theory. Biol. Rev. 75, 377–404 (2000).
Freitak, D., Wheat, C. W., Heckel, D. G. & Vogel, H. Immune system responses and fitness costs associated with consumption of bacteria in larvae of trichoplusia Ni. BMC Biol. 5, 56 (2007).
Hanson, M. A., Lemaitre, B. & Unckless, R. L. Dynamic evolution of antimicrobial peptides underscores Trade-Offs between immunity and ecological fitness. Front Immunol 10, 2620 (2019).
Hosken, D. J. Sex and death: microevolutionary trade-offs between reproductive and immune investment in Dung flies. Curr. Biol. 11, 379–380 (2001).
Iglesias-Carrasco, M., Head, M. L., Jennions, M. D. & Cabido, C. Condition-dependent trade-offs between sexual traits, body condition and immunity: the effect of novel habitats. BMC Evol. Biol. 16, 1–10 (2016).
Leman, J. C. et al. Lovesick: immunological costs of mating to male sagebrush crickets. J. Evol. Biol. 22, 163–171 (2009).
Fuxa, J. R. & Tanada, Y. Epizootiology of Insect Diseases (Wiley, 1991).
Wilson, K. & Cotter, S. Density-Dependent Prophylaxis in Insects. in Phenotypic Plasticity of Insects (eds. Whitman, D. & Ananthakrishnan, T.)Science Publishers, (2009). https://doi.org/10.1201/b10201-7
Møller, A. P. Parasites and sexual selection: current status of the Hamilton and Zuk hypothesis. J. Evol. Biol. 3, 319–328 (1990).
Dewsbury, D. A. The Darwin-Bateman paradigm in historical Context1. Integr. Comp. Biol. 45, 831–837 (2005).
Janicke, T., Häderer, I. K., Lajeunesse, M. J. & Anthes, N. Darwinian sex roles confirmed across the animal Kingdom. Sci. Adv. 2, e1500983 (2016).
Sheldon, B. C. & Verhulst, S. Ecological immunology: costly parasite defences and trade-offs in evolutionary ecology. Trends Ecol. Evol. 11, 317–321 (1996).
Zuk, M. & McKean, K. A. Sex differences in parasite infections: patterns and processes. Int. J. Parasitol. 26, 1009–1024 (1996).
Rolff, J. Bateman’s principle and immunity. Proc. R Soc. B Biol. Sci. 269, 867–872 (2002).
Marmaras, V. J., Charalambidis, N. D. & Zervas, C. G. Immune response in insects: the role of phenoloxidase in defense reactions in relation to melanization and sclerotization. Arch. Insect Biochem. Physiol. 31, 119–133 (1996).
Gillespie, J. P., Kanost, M. R. & Trenczek, T. Biological mediators of insect immunity. Annu. Rev. Entomol. 42, 611–643 (1997).
Kanost, M. R. & Gorman, M. J. Phenoloxidases in insect immunity. Insect Immunol. 1, 69–96 (2008).
Schmid-Hempel, P. Evolutionary ecology of insect immune defenses. Annu. Rev. Entomol. 50, 529–551 (2005).
Roers, A., Hiller, B. & Hornung, V. Recognition of endogenous nucleic acids by the innate immune system. Immunity 44, 739–754 (2016).
Kelly, C. D., Stoehr, A. M., Nunn, C., Smyth, K. N. & Prokop, Z. M. Sexual dimorphism in immunity across animals: a meta-analysis. Ecol. Lett. 21, 1885–1894 (2018).
McAfee, A., Chapman, A., Pettis, J. S., Foster, L. J. & Tarpy, D. R. Trade-offs between sperm viability and immune protein expression in honey bee queens (Apis mellifera). Commun. Biol. 4, 1–11 (2021).
Gascoigne, S. J. L., Nalukwago, U., Barbosa, F. & D. I. & Larval density, sex, and allocation hierarchy affect life history trait covariances in a bean beetle. Am. Nat. 199, 291–301 (2022).
Nokelainen, O., Lindstedt, C. & Mappes, J. Environment-mediated morph‐linked immune and life‐history responses in the aposematic wood tiger moth. J. Anim. Ecol. 82, 653–662 (2013).
Silva, F. W. et al. Two’s a crowd: phenotypic adjustments and prophylaxis in Anticarsia gemmatalis larvae are triggered by the presence of conspecifics. PloS One. 8, e61582 (2013).
Bailey, N. W., Gray, B. & Zuk, M. Does immunity vary with population density in wild populations of Mormon crickets? Evol. Ecol. Res. 10, 599–610 (2008).
Wilson, K. et al. Coping with crowds: density-dependent disease resistance in desert locusts. Proc. Natl. Acad. Sci. 99, 5471–5475 (2002).
Kelly, C. D. & L’Heureux, V. Effect of rearing density on female investment in reproduction and melanotic encapsulation response in the sand cricket (Gryllus firmus) (Orthoptera: Gryllidae). Biol. J. Linn. Soc. 144, blae023 (2024).
Kelly, C. D., L’Heureux, V., Wey, T. W. & Réale, D. Effect of rearing density on the expression of fitness-related traits in male sand field crickets (Gryllus firmus). Evol. Ecol. 37, 835–846 (2023).
Rolff, J. & Siva-Jothy, M. T. Copulation corrupts immunity: a mechanism for a cost of mating in insects. Proc. Natl. Acad. Sci. 99, 9916–9918 (2002).
Avila, F. W., Sirot, L. K., LaFlamme, B. A., Rubinstein, C. D. & Wolfner, M. F. Insect seminal fluid proteins: identification and function. Annu. Rev. Entomol. 56, 21–40 (2011).
Immonen, E., Sayadi, A., Bayram, H. & Arnqvist, G. Mating changes sexually dimorphic gene expression in the seed beetle Callosobruchus maculatus. Genome Biol. Evol. 9, 677–699 (2017).
Zera, A. J. The endocrine regulation of wing polymorphism in insects: state of the art, recent surprises, and future directions1. Integr. Comp. Biol. 43, 607–616 (2003).
Wilson, K. Evolution of clutch size in insects. II. A test of static optimality models using the beetle Callosobruchus maculatus (Coleoptera: Bruchidae). J. Evol. Biol. 7, 365–386 (1994).
Vamosi, S. M. Interactive effects of larval host and competition on adult fitness: an experimental test with seed beetles (Coleoptera: Bruchidae). Funct. Ecol. 19, 859–864 (2005).
Beck, C. W. & Blumer, L. S. A handbook on bean beetles, Callosobruchus maculatus. Caryologia 24, 157–166 (2011).
Utida, S. Density dependent polymorphism in the adult of Callosobruchus maculatus (Coleoptera, Bruchidae). J. Stored Prod. Res. 8, 111–125 (1972).
Dougherty, L. R. et al. Sexual conflict and correlated evolution between male persistence and female resistance traits in the seed beetle Callosobruchus maculatus. Proc. R. Soc. B Biol. Sci. 284, (2017).
Rasband, W. S. ImageJ. (1997).
Microsystems, L. Leica application Suite version 2.0. (2010).
Peterson, R. A. Finding optimal normalizing transformations via bestNormalize. (2021).
SAS Institute Inc. JMP®. (1989).
Acknowledgements
The authors are grateful to Frances Rice, Sarah McField, Beth DeFoe and Raneem Samman for support in data collection, animal husbandry, and project set up. This material is based upon work supported by the Department of Biology at Lake Forest College and by the National Science Foundation under Grant IOS-2113134. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the NSF.
Author information
Authors and Affiliations
Contributions
IZS helped design the study, contributed to carrying out the outlined methods, contributed to data analysis and helped draft the manuscript. FB contributed to the inception of the study, designing study methods, data analysis, manuscript drafting and revision and supervision of the study. KL contributed to carrying out the outlined methods, contributed to data analysis and helped revise the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shepard, I.Z., Lucas, K. & Barbosa, F. Effects of population density on immunity and reproduction in bean beetles Callosobruchus maculatus. Sci Rep 15, 32240 (2025). https://doi.org/10.1038/s41598-025-17811-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17811-7