Abstract
Acellular dermal matrix (ADM) has been widely used in skin grafting to provide a dermal layer for facilitating graft take and mature scarring. This randomized controlled, double-blinded superiority study aimed to quantitatively assess the effects of this technique on the resulting graft sites via functional measurements including elasticity, humidification, transepidermal water loss (TEWL), pigmentation, and erythema at 6 months after surgery. A survey was performed to evaluate the satisfaction levels of both patients and surgeons. We considered patients with full-thickness skin defects for whom split-thickness skin grafts (STSGs) were used. By random assignment, they were allocated to the ADM group or the STSG-only group. The ADM group received STSG with a ADM layer on the defect, whereas the STSG-only group was reconstructed with only STSG. At 6 months after surgery, patients were evaluated for elasticity, humidification, TEWL, erythema, and pigmentation. Defect areas before and 2 weeks after surgery were traced and measured. Patients and surgeons rated the graft sites as excellent, good, fair or poor at 6 months after the operation. Among the 89 patients who were screened, 54 patients were enrolled in this study. Compared with those in the STSG-only group, defects that were reconstructed with STSG and ADM resulted in significantly greater elasticity, lower TEWL, and less erythema. There were no significant differences in humidification or pigmentation. The ADM co-grafted with STSGs was associated with greater satisfaction among surgeons, whereas patient satisfaction was not significantly different between the two groups. Co-grafting ADM with STSG in full-thickness wounds can lead to favorable wound healing in terms of elasticity, TEWL, and erythema. This approach also has a higher satisfaction rate for surgeons.
Registry: Clinical Research Information Service (https://cris.nih.go.kr), Registration number: KCT0007040, Registration date: 24/02/2022.
Similar content being viewed by others
Introduction
The acellular dermal matrix (ADM) is a versatile tool in the fields of reconstructive surgery and wound healing. It is an allogenic or xenogenic immunologically inert processed layer of dermis that can be utilized as an extracellular matrix that provides scaffolding and facilitating wound healing1,2,3. It is widely used in various areas, including breast reconstructions4, head and neck surgeries5, lower extremity reconstructions6,7, burn wounds, and skin grafts8. While synthetic skin replacements are under exploration, ADM is a convenient and relatively affordable source of soft tissue that could be otherwise difficult to procure9.
One of the main functions of ADM is providing a protective and incorporable layer for wound coverage. For shallow and expansive wounds, finding a source of dermal replacement with limited donor sites is challenging. For deeper wounds, protecting the underlying structures, such as tendons, nerves, muscles, and bones, and facilitating skin graft integration is important8,10. In both cases, split-thickness skin grafts (STSGs) constitute a central method of treatment for large, shallow defects and are often cografted with ADMs11.
While ADM is a widely used tool, its effectiveness on STSG outcomes has not been quantitatively analyzed. Many studies have affirmed that ADM is beneficial to reconstruction with skin grafts, but few have objectively inspected the healed tissues in terms of measured parameters. Among many parameters, we have chosen to inspect elasticity, skin humidification, transepidermal water loss (TEWL), erythema, and pigmentation in order to assess the biomechanical properties of the skin barrier12,13,14,15.
This randomized, double-blinded, superiority trial investigates the effect of ADM on STSG healing by comparing tissue characteristics, including elasticity, erythema, scar humidification, transepidermal water loss, and the melanin index. In addition, patient and surgeon satisfaction was assessed using subjective surveys.
Method
Study design
This study was a randomized, double-blinded, superiority trial that was conducted at Seoul St. Mary’s Hospital, Yonsei Severance Hospital, and Seoul Asan Hospital to evaluate the efficacy of combining STSG with human ADM (L&C Bio, Seongnam-si, Gyeonggi-do, South Korea) in the reconstruction of full-thickness skin defects between 2022 and 2024.
Trial protocol and data are available in supplementary files.
Ethical review
This study was reviewed and approved by the Institutional Review Boards (IRBs) of Seoul St. Mary’s Hospital, Asan Medical Center, and Severance Hospital before and throughout the experiment. Any significant modifications related to the study were subject to IRB approval, and all results have been registered in the Clinical Research Information Service on 24/02/22 (https://cris.nih.go.kr, Registration number KCT0007040). It was conducted in compliance with the principles of the International Conference on Harmonization (ICH) Guidelines and the Declaration of Helsinki. This study was carried out in accordance with the Korean Good Clinical Practice (KGCP) guidelines and relevant regulations, with due consideration for the rights and safety of the study participants.
As a postmarketing clinical study of Megaderm Plus (L&C Bio, Seongnam-si, Gyeonggi-do, South Korea) performed within the approved indications, it is not subject to approval by the Ministry of Food and Drug Safety (MFDS).
Study population
Patients between 19 and 70 years of age with full-thickness skin defects requiring reconstruction were provided with informed consent to participate in the clinical trial. Defects were acute, uninfected wounds on flat surfaces that were not affected by joint movements. Target enrollment was 84 subjects, with 42 per group, accounting for 20% dropout. Patients were excluded if they were pregnant, nursing, or had a history of potentially relevant or unsuitable conditions that could interfere with wound healing.
Randomization and maintaining blindness
Each enrolled patient was assigned a screening number and screened per the eligibility criteria. After completing the screening process, each eligible patient was randomly assigned a 3-digit number and assigned to either the experimental (ADM and STSG) or control group (STSG only) according to a computer-generated randomization table. This allotment was performed via the block randomization method, in which each patient was stratified to each institution so that the control group and experimental groups could be balanced. The three-digit randomization number was used as the patient identification code during the experiment.
This study maintained double-blindness by establishing an independent investigator to measure and analyze the functional assessments. Because the investigator was not involved in the surgical process, he or she was able to make objective measurements without bias. The operator was aware of the patient allocation during the surgery but was blinded during the satisfaction survey.
Safety analysis and discontinuation of trial
Adverse events were assessed at every visit after the operation and were classified according to severity; mild, if it does not interfere with normal daily activities, moderate if it causes noticeable discomfort affecting normal daily activities, or severe if it prevents normal daily activities. If unexpected surgical complications occur in more than 30% of participants, the trial was to be discontinued.
Surgical procedure and postoperative care
Before skin grafting, all wounds were debrided, thoroughly irrigated, and cauterized for bleeding. When the full-thickness skin defect was prepared for skin grafting, it was traced for area measurements before grafting. Split-thickness skin grafts were harvested from either thigh with a pneumatic dermatome at 0.002 mm thickness. The ADM group received a combination of cadaveric ADM (MegadermPlus, L&C Bio, Seongnam-si, Gyeonggi-do, South Korea) and STSG, whereas the control group received STSG alone. For the ADM group, human ADM was placed on the defect and was trimmed to fit before applying the STSG (Fig. 1). For the control group, skin grafts was placed directly on the defects. STSGs were meshed, trimmed to the defect size, and fixed on the wounds with skin stapler (Fig. 2). The wounds were dressed under negative pressure wound therapy (NPWT). When applying NPWT (Curasys, CG Bio, South Korea), a layer of gel matrix dressing (UrgoTul, Urgo medical, France) was placed on the skin graft before applying the sponge and were then vacuum sealed with film at continuous negative pressure of 120 mmHg. The dressing was removed after 3 to 5 days, followed by gauze or foam dressing, which was changed every two days. Dressings were discontinued once the wounds healed and no longer showed exudates. A skin moisturizer provided by the investigator was applied for at least 3 months, except for the 7 days preceding the follow-up visits. The moisturizer (Bythedoctor Ato lotion, L&C Bio, South Korea) contained hyaluonic acid, ceramide, and lecithin and was used as an emollient barrier to promote skin remodeling and healthy scar formation to the grafted sites. It was discontinued before data collection to prevent its impact on data measurements.
Postoperative follow-ups and evaluations
Follow-up observations were performed at 2 weeks, 3 months, 6 months, and 12 months after surgery. All assessments and measurements were done by a blinded investigator who had not participated in the randomization process or the surgical procedures. At each visit, the patients’ vital signs were measured, photographs of their surgical sites were taken, and any adverse effects were observed. At the 2 week visit, pictures of graft sites were taken and areas of successfully healed grafts were measured by ImageJ program. At the 6-month visit, the patients were analyzed for their functional skin assessments and satisfaction.
Primary outcome was skin elasticity at 6 months after surgery. Secondary outcomes included other functional skin assessments at 6 months after surgery, such as scar humidification, TEWL, melanin index, and erythema index. Elasticity was measured with a Cutometer MPA-580 (Courage & Khazaka Electronic GmbH, Germany) under 500 mbar vacuum. Scar humidification was measured with a Corneometer (Courage & Khazaka Electronic GmbH, Germany) and a CM825 probe. TEWL was measured with a Tewameter (Courage & Khazaka Electronic GmbH, Germany) and TM300 probe. The melanin index and erythema index were measured with a Mexameter (Courage & Khazaka Electronic GmbH, Germany) with an MX18 probe.
The graft areas were traced during surgery before grafting and 2 weeks after surgery. Tracings were then scanned, and the area was measured via the ImageJ program. A blinded satisfaction survey was performed at 6 months after surgery by both patients and surgeons, who rated their resulting skin graft sites as excellent, good, fair, or poor.
Statistical analysis
The functional assessment of skin elasticity, humidification, transepidermal water loss (TEWL), the melanin index, and erythema at 6 months postsurgery was performed. Group comparisons were performed via either the independent two-sample t test or Wilcoxon rank-sum test, which is based on normality assumptions per the Shapiro‒Wilk test. If the patient was missing any data, the data was excluded from analysis.
The satisfaction scores from both the investigators and the participants are presented as frequencies. Comparisons between groups were made via Fisher’s exact test if there were more than 5 cells with expected counts of less than 5.
The graft rate at 2 weeks postsurgery was calculated by comparing the engrafted area to the initial treated area for each treatment group. Group comparisons were performed via the Mann‒Whitney U test after testing for a normal distribution via the Shapiro‒Wilk test.
Results
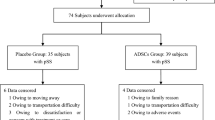
Eighty-nine patients were screened for eligibility, and 85 patients who met the eligibility criteria were randomized into ADM-only and STSG-only groups. Forty-one and 44 patients were allocated to the ADM-only and STSG-only groups, respectively. During the surgical procedures, a different surgical method was selected for 3 patients in the ADM group and 1 patient in the STSG-only group. During postoperative follow-up, 10 and 17 patients, respectively, were withdrawn from the study because of noncompliance (4 and 7 patients, respectively), withdrawal of consent (1 and 2 patients, respectively), and otherwise, miscellaneous reasons (5 and 8 patients, respectively). As a result, a total of 54 patients were enrolled per protocol this study, with 28 in the ADM group and 26 in the STSG only group (Fig. 3).
Both groups did not differ significantly in terms of demographic factors, such as age, sex, previous medical history, defect location, or cause of defects. The mean ages of the ADM and STSG-only groups were 56.6 and 57.8 years, respectively. Nearly all patients had previous illnesses, including soft tissue malignancies. Most of the defects were free flap donor sites (92.9% and 88.5%, respectively) located at thigh and forearm areas. Other defects were caused by other means such as trauma, burn, fasciotomy, and tumor excision. All defects were clean, acute wounds located on flat surfaces not associated with joint movements (Table 1).
Skin assessment at 6 months after surgery revealed elasticity, humidification, transepidermal water loss (TEWL, g/hr/m2), pigmentation, and erythema. Elasticity index, the primary outcome, was significantly greater in the ADM group (0.82 ± 0.13) than in the STSG-only group (0.74 ± 0.12), with a p-value of 0.032. Secondary outcomes differed by category. Humidification was slightly greater in the STSG group than in the ADM group, at 32.5 ± 12.6 and 28.2 ± 14.1, respectively, but the difference was not statistically significant. TEWL was greater in the STSG-only group (12.29 ± 8.24) than in the ADM group (8.99 ± 5.57) with a p-value of 0.038. Pigmentation, as measured by the melanin index, was 269.2 ± 130.2 and 270.2 ± 144.8 for the ADM group and the STSG group, respectively, and the difference was not statistically significant. Erythema index was significantly greater in the STSG group (399.8 ± 90.9) than in the ADM group (350.4 ± 64.02), with a p value of 0.024. (Fig. 4; Table 2)
The graft area was measured before grafting and 2 weeks after surgery. The mean defect areas were 34.51 ± 29.81 cm2 and 46.76 ± 46.59 cm2 in the ADM and STSG-only groups respectively, with no significant differences. The area of healed grafts at 2 weeks was 33.57 ± 29.84 cm2 (95.4 ± 9.2%) for the ADM group and 46.37 ± 46.14 cm2 (98.8 ± 2.15%) for the STSG-only group. More than 95% of the grafts in both groups were successfully engrafted by 2 weeks after surgery, and there were no significant differences between the groups.
Patient and surgeon satisfaction analysis was performed at 6 months after surgery. (Table 3) Surveys were double-blinded and categorized into 4 categories: excellent, good, fair, and poor. The surgeons’ responses revealed that the ADM group was significantly more favorable with a p-value of 0.046. A total of 96.8% of the surgeons reported that the ADM co-grafted STSG sites were excellent or good, whereas only 2 (7.14%) thought that they were fair. No surgeon reported that the STSGs co-grafted with ADMs was poor. The majority, 89.5% and 73.1%, of the patients in the ADM group and the STSG-only group, respectively, reported favorable answers of excellent or good, and 3.6% and 11.5%, respectively, reported that the graft site quality was poor. There was no significant difference between the ADM group and the STSG-only group (Fig. 5).
Nine adverse events were reported among 6 subjects (21.4%) in the test group and 37 among 10 subjects (38.5%) in the control group, but none were relevant to the procedure. In the test group, the adverse events were operation site pain, hypertension, vertigo, hemoptysis, arm swelling, arm pain, soft tissue abscess, and cancer metastasis. The control group reported symptoms such as vomiting, diarrhea, pruritus, and facial edema, but none were relevant to the procedure. There was no statistically significant difference between the two groups (p = 0.35). There was no graft failure or complications related to wound healing.
Discussion
Skin grafting is a useful reconstructive method that is versatile for most defects, but it is limited by donor site availability16 and its thinness17,18. To compensate for these disadvantages, various combinations of allogenous and autogenous grafts have been explored19,20, finally giving way to ADMs in the 1990s. ADMs have been introduced as an immunologically inert form of allogenous or xenogenous skin layer21,22 for coverage of expansive full-thickness skin injuries.
Various forms of ADMs have been explored, ranging from allografts and xenografts to artificial matrices, that can facilitate tissue growth with minimal immunologic reactions or scarring23,24. Allogenic ADMs are obtained from human cadaveric skin, treated to minimize immunologic triggers. Xenogenic ADMs are most frequently prepared from bovine or porcine skin and are economically favorable. Some animal model studies have shown, however, that xenografts are associated with increased cell-mediated immune reactions25,26 and poor wound healing26,27.
In the clinical setting, both allogenic and xenogenic ADMs are actively used in reconstructive surgery. They are mainstay in breast reconstruction for natural aesthetic outcomes and to prevent capsular contracture in breast28,29. Cosmetic surgery also uses ADMs as a source of augmentation that can minimize foreign body reactions30, since inflammation can cause significant complications. All areas of reconstructions, including head and neck, genitourinary, and lower extremities, are actively utilizing ADM as an additional source of soft tissue. However, ADMs are costly1,31,32, requiring both patient and surgeon to weigh the pros and cons of this reconstructive tool. While there are several studies that warrant the use of ADM resulting in an overall reduction in time and cost31,33, some assert that it does pose a financial burden on patients31,34 and to be wary of industrial fundings that may be a cause of this surge of ADM in the clinical and academic settings32.
Especially in split-thickness skin grafts, supplementation with ADM can provide a layer of acellular extracellular matrix (ECM), a kind of biocompatible scaffold for tissue ingrowth, and mimic the physiologic dermis layer that STSGs lack. Full thickness skin grafts (FTSGs), skin grafts that include the dermis layer, are often considered as an alternative, but they are limited by donor site morbidity in expansive defect coverage. ADM acts as a supplementary dermis in STSGs, and is known to facilitate wound healing by providing a template3,35 for fibroblasts to infiltrate and provide collagen for wound coverage36. Owing to its compositional similarity to the dermis36,37, it can assist in the functions of the skin barrier, such as durability37 and protection against loss of humidification, as well as deterrence of hyperpigmented and immature scarring. Some studies6,9 have used ADM as a single treatment option to promote wound healing without autologous tissue transfer.
This study aimed to assess the function and aesthetics of healed defects to objectively analyze the effect of ADM on STSG by quantitative parameters and surgeon-/patient-reported quality outcomes. Functional measures were reported by elasticity, a parameter reflecting skin plasticity and durability37. TEWL and skin humification measured skin barrier integrity38, reflecting the ability of stratum corneum’s retention of the body’s moisture held from the external environment13,14. For aesthetic considerations, we measured erythema and pigmentation, with erythema an important predictor of clinical symptoms of scar formation39, such as pruritus and pain.
At 6 months after surgery, the primary outcome showed that skin elasticity was higher in the STSG and ADM group. Lee et al.40 reported that the elasticity of the human ADM co-grafted to STSG was similar to that of adjacent normal skin. Because ADM is composed of type I collagen35, it can be expected that applying ADM with STSG can increase skin elasticity. Other studies have recently explored the modification of this scaffold to augment its elasticity37,41.
For secondary outcomes, we measured other variables to attest for the biomechanical properties of grafted skin. Our experiment revealed that skin grafts with ADM presented a lower TEWL, confirming that ADM can act as a functional assistance against moisture loss. Other studies40 also reported similar results, where human or bovine ADM co-grafted STSGs presented lower TEWL than did the STSG-only group. Although there was no significant difference in scar humidification between the two groups, the resistance to water loss was greater in the ADM group than in the control group, indicating that the skin barrier function improved.
ADM co-grafted STSGs were significantly less erythematous than STSG-only group, indicating that ADM co-grafted with STSGs may reduce the possibility of future scar symptoms. Pigmentation was not significantly different between the groups while other studies40 explored the differences of pigmentations in allogenic and xenogenic ADMs.
According to our satisfaction survey, ADM was significantly favored by surgeons, while there was no significant difference among patients. Given that this is a double-blinded study, STSG sites co-grafted with ADM were rated more favorable to the professional eye. It also shows that there was no outstanding downside in the use of ADM compared to STSG only reconstructions.
Limitations of this study are underscored by the shortness of the follow-up period. Evaluations of functional assessments would benefit from a longer observation to see the changes made with skin remodeling. Satisfaction survey could be done in more detail to analyze the reasons behind the responses.
Conclusion
This double-blinded controlled study objectively shows that human ADM can enhance the skin grafts in elasticity, TEWL, and erythema. Subjective survey showed that surgeons viewed ADM co-grafted skin as more favorable to the surgeons and that patients did not complain of significant differences. We corroborate that ADM is a beneficial tool in STSG and suggest a long-term study for its impact on skin remodeling.
Data availability
The datasets generated and/or analysed during the current study are available in the CRIS repository (cris.nih.go.kr) under Registration number KCT0007040, registraction date of 24/02/2022. The datasets used and/or analysed during the current study available from the corresponding author on reasonable request. All data generated or analysed during this study along with the trial protocol are included in this published article and its supplementary information files.
References
Mohammadyari, F. et al. Acellular dermal matrix in reconstructive surgery: Applications, benefits, and cost. Front Transplant. 2, 1133806. https://doi.org/10.3389/frtra.2023.1133806 (2023) (Epub 20230310).
Gierek, M. et al. Human acellular dermal matrix in reconstructive surgery-a review. Biomedicines. https://doi.org/10.3390/biomedicines10112870 (2022) (Epub 20221109).
Brown, B. N. et al. Comparison of three methods for the derivation of a biologic scaffold composed of adipose tissue extracellular matrix. Tissue Eng. Part C Methods 17(4), 411–421. https://doi.org/10.1089/ten.TEC.2010.0342 (2011) (Epub 20110205).
Gravina, P. R., Pettit, R. W., Davis, M. J., Winocour, S. J. & Selber, J. C. Evidence for the use of acellular dermal matrix in implant-based breast reconstruction. Semin. Plast. Surg. 33(4), 229–235. https://doi.org/10.1055/s-0039-1696986 (2019) (Epub 20191017).
Patel, S., Ziai, K., Lighthall, J. G. & Walen, S. G. Biologics and acellular dermal matrices in head and neck reconstruction: A comprehensive review. Am. J. Otolaryngol. 43(1), 103233. https://doi.org/10.1016/j.amjoto.2021.103233 (2022) (Epub 20210915).
Li, G., Shen, Q., Zhou, P., Liu, H. & Chen, J. Acellular dermal matrix for one-stage treatment of lower extremity full-thickness skin defect: a case series. BMC Surg. 23(1), 17. https://doi.org/10.1186/s12893-022-01871-x (2023) (Epub 20230124).
Fuller, D. A. Split thickness skin graft to lower leg. J. Orthop. Trauma. 30(Suppl 2), S34. https://doi.org/10.1097/BOT.0000000000000605 (2016).
Tognetti, L. et al. The use of human acellular dermal matrices in advanced wound healing and surgical procedures: State of the art. Dermatol. Ther. 34(4), e14987. https://doi.org/10.1111/dth.14987 (2021) (Epub 20210524).
Lewis, R. E. et al. Human acellular dermal matrix is a viable alternative to autologous skin graft in patients with cutaneous malignancy. Am. Surg. 85(9), 1056–1060 (2019).
Woo, S. J., Kwon, S. T. & Kim, B. J. Serratus anterior fascia free flap for functional surgery of subungual melanoma: case series and literature review. Arch. Plast. Surg. 52(2), 90–95. https://doi.org/10.1055/s-0044-1792109 (2025) (Epub 20250311).
Hahn, H. M., Jeong, Y. S., Lee, I. J., Kim, M. J. & Lim, H. Efficacy of split-thickness skin graft combined with novel sheet-type reprocessed micronized acellular dermal matrix. BMC Surg. 22(1), 358. https://doi.org/10.1186/s12893-022-01801-x (2022) (Epub 20221011).
Jaspers, M. E. H. et al. Assessing blood flow, microvasculature, erythema and redness in hypertrophic scars: A cross sectional study showing different features that require precise definitions. Burns 43(5), 1044–1050. https://doi.org/10.1016/j.burns.2017.01.017 (2017) (Epub 20170130).
Lamke, L. O. The influence of different “skin grafts” on the evaporative water loss from burns. Scand. J. Plast. Reconstr. Surg. 5(2), 82–86. https://doi.org/10.3109/02844317109042943 (1971).
Szymoniak-Lipska, M. et al. Transepidermal water loss (TEWL) and transonychial water loss (TOWL) measurements in healthy nail apparatus. Skin. Res. Technol. 30(7), e13851. https://doi.org/10.1111/srt.13851 (2024).
Yang, Y., Wu, X. & Liu, W. Scar symptom: erythema and thickness. In Textbook on Scar Management: State of the Art Management and Emerging Technologies (eds Teot, L. et al.) 103–108 (2020).
Ng, J. C., Ahmad Zaidi, A. I., Lee, J. & Jabar, M. F. Meek micrografting technique for reconstruction of extensive necrotizing fasciitis of the anterior abdomen and bilateral femoral region: a case report. Arch. Plast. Surg. 50(6), 610–614. https://doi.org/10.1055/a-2077-5745 (2023) (Epub 20230824).
Corps, B. V. The effect of graft thickness, donor site and graft bed on graft shrinkage in the hooded rat. Br. J. Plast. Surg. 22(2), 125–133. https://doi.org/10.1016/s0007-1226(69)80053-6 (1969).
Wainwright, D. J. & Bury, S. B. Acellular dermal matrix in the management of the burn patient. Aesthet. Surg. J. 31(7 Suppl), 13S-23S. https://doi.org/10.1177/1090820X11418202 (2011).
Cuono, C. B., Langdon, R., Birchall, N., Barttelbort, S. & McGuire, J. Composite autologous-allogeneic skin replacement: development and clinical application. Plast. Reconstr. Surg. 80(4), 626–637. https://doi.org/10.1097/00006534-198710000-00029 (1987).
Yukna, R. A., Turner, D. W. & Robinson, L. J. Variable antigenicity of lyophilized allogeneic and lyophilized xenogeneic skin in guinea pigs. J. Periodontal. Res. 12(3), 197–203. https://doi.org/10.1111/j.1600-0765.1977.tb00122.x (1977).
Sedmak, D. D. & Orosz, C. G. The role of vascular endothelial cells in transplantation. Arch. Pathol. Lab. Med. 115(3), 260–265 (1991).
Livesey, S. A., Herndon, D. N., Hollyoak, M. A., Atkinson, Y. H. & Nag, A. Transplanted acellular allograft dermal matrix. Potential as a template for the reconstruction of viable dermis. Transplantation 60(1), 1–9 (1995).
Castagnoli, C., Stella, M., Magliacani, G., Alasia, S. T. & Richiardi, P. Anomalous expression of HLA class II molecules on keratinocytes and fibroblasts in hypertrophic scars consequent to thermal injury. Clin. Exp. Immunol. 82(2), 350–354. https://doi.org/10.1111/j.1365-2249.1990.tb05451.x (1990).
Carvalho-Junior, J. D. C., Zanata, F., Aloise, A. C. & Ferreira, L. M. Acellular dermal matrix in skin wound healing in rabbits - histological and histomorphometric analyses. Clinics 76, e2066. https://doi.org/10.6061/clinics/2021/e2066 (2021) (Epub 20210308).
Wang, R. M. et al. Humanized mouse model for assessing the human immune response to xenogeneic and allogeneic decellularized biomaterials. Biomaterials 129, 98–110. https://doi.org/10.1016/j.biomaterials.2017.03.016 (2017) (Epub 20170311).
DeSagun, E. Z., Botts, J. L., Srivastava, A., Hanumadass, M. & Walter, R. J. Long-term outcome of xenogenic dermal matrix implantation in immunocompetent rats. J. Surg. Res. 96(1), 96–106. https://doi.org/10.1006/jsre.2000.6060 (2001).
Srivastava, A. et al. Xenogeneic acellular dermal matrix as a dermal substitute in rats. J. Burn Care Rehabil. 20(5), 382–390. https://doi.org/10.1097/00004630-199909000-00010 (1999).
Marra, C., Cuomo, R., Ceccaroni, A., Pentangelo, P. & Alfano, C. Acellular dermal matrix in breast augmentation surgery: A systematic review. JPRAS Open. 40, 111–117. https://doi.org/10.1016/j.jpra.2024.02.004 (2024) (Epub 20240215).
du Plessis, M. I., Cottler, P. S. & Campbell, C. A. Acellular dermal matrix favorably modulates the healing response after surgery. Plast. Reconstr. Surg. 150(2), 290e-e299. https://doi.org/10.1097/PRS.0000000000009338 (2022) (Epub 20220603).
Mihalecko, J. et al. Acellular dermal matrix in plastic and reconstructive surgery. Physiol. Res. 71(Suppl 1), S51–S57. https://doi.org/10.33549/physiolres.935045 (2022).
Krishnan, N. M. et al. The cost effectiveness of acellular dermal matrix in expander-implant immediate breast reconstruction. J. Plast. Reconstr. Aesthet. Surg. 67(4), 468–476. https://doi.org/10.1016/j.bjps.2013.12.035 (2014) (Epub 20140123).
Hirpara, M. M., Clark, R. C., Hogan, E., Dean, R. & Reid, C. M. Rise of acellular dermal matrix: cost consciousness, industry payment, and publication productivity. J. Am. Coll. Surg. 236(6), 1189–1197. https://doi.org/10.1097/XCS.0000000000000648 (2023) (Epub 20230209).
Aliotta, R. E. et al. A controlled cost and outcomes analysis of acellular dermal matrix and implant-based reconstruction. J. Plast. Reconstr. Aesthet. Surg. 74(6), 1229–1238. https://doi.org/10.1016/j.bjps.2020.10.079 (2021) (Epub 20201108).
Viezel-Mathieu, A. et al. Acellular dermal matrix-sparing direct-to-implant prepectoral breast reconstruction: A comparative study including cost analysis. Ann. Plast. Surg. 84(2), 139–143. https://doi.org/10.1097/SAP.0000000000001997 (2020).
Badylak, S. F., Freytes, D. O. & Gilbert, T. W. Extracellular matrix as a biological scaffold material: Structure and function. Acta Biomater. 5(1), 1–13. https://doi.org/10.1016/j.actbio.2008.09.013 (2009) (Epub 20081002).
Chen, R. N., Ho, H. O., Tsai, Y. T. & Sheu, M. T. Process development of an acellular dermal matrix (ADM) for biomedical applications. Biomaterials 25(13), 2679–2686. https://doi.org/10.1016/j.biomaterials.2003.09.070 (2004).
Versteegden, L. R. et al. Design of an elasticized collagen scaffold: A method to induce elasticity in a rigid protein. Acta Biomater. 44, 277–285. https://doi.org/10.1016/j.actbio.2016.08.038 (2016) (Epub 20160821).
Green, M., Feschuk, A. M., Kashetsky, N. & Maibach, H. I. “Normal” TEWL-how can it be defined? A systematic review. Exp. Dermatol. 31(10), 1618–1631. https://doi.org/10.1111/exd.14635 (2022) (Epub 20220731).
Lee, Y. J. et al. Effectiveness of acellular dermal matrix on autologous split-thickness skin graft in treatment of deep tissue defect: esthetic subjective and objective evaluation. Aesthetic Plast. Surg. 41(5), 1049–1057. https://doi.org/10.1007/s00266-017-0891-2 (2017) (Epub 20170519).
Lee, M. C., Jang, Y. J., Yun, I. S., Lew, D. H. & Lee, W. J. Comparative skin evaluation after split-thickness skin grafts using 2 different acellular dermal matrices to cover composite forearm defects. J. Hand Surg. Am. 42(4), 297.e1-e10. https://doi.org/10.1016/j.jhsa.2017.01.026 (2017) (Epub 20170301).
Ryan, A. J. & O’Brien, F. J. Insoluble elastin reduces collagen scaffold stiffness, improves viscoelastic properties, and induces a contractile phenotype in smooth muscle cells. Biomaterials 73, 296–307. https://doi.org/10.1016/j.biomaterials.2015.09.003 (2015) (Epub 20150925).
Funding
ADM was provided by L&C Bio. No other source of funding was used.
Author information
Authors and Affiliations
Contributions
C.L. participated in statistical analysis and prepared manuscript and figures.W.J. was in charge of data collection in Asan medical center and overall experimental design.Y.S. was in charge of data collection in Severance hospital and overall experimental design.S.M. in charge of data collection in Seoul St. Mary’s hospital and overall experimental design.All authors reviewed the manuscript and statistical analyses.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, C.R., Jeong, W.S., Suh, Y.C. et al. A randomized, controlled, double-blinded trial on the effects of acellular dermal matrices on the functional assessment and qualitative satisfaction of split-thickness skin grafts. Sci Rep 15, 33579 (2025). https://doi.org/10.1038/s41598-025-18705-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-18705-4