Abstract
Lead (Pb) induces physiological, morphological, and metabolic effects in plants. The exogenous application of ethylenediaminetetraacetic acid (EDTA) and citric acid (CA) is a common method to enhance the phytoextraction of Pb from plants. However, the impact of these treatments on harmel’s ability to tolerate and accumulate Pb remains unclear. This study aimed to investigate the effects of CA (0, 2.5, and 5 mM) and EDTA (0, 2.5, and 5 mM) on physiological growth responses, antioxidant enzymes, as well as lead translocation factor (TF) and uptake of harmel under Pb stress (0, 5, and 25 mg L−1). The results indicated that Pb treatment significantly reduced growth parameters in harmel plants. The application of 5 mM EDTA adversely affected plant growth, while the 2.5 and 5 mM CA treatments enhanced it. The application of 2.5 mM CA and EDTA significantly decreased lipid peroxidation and improved biomass production in Pb-stressed plants. The diversity of enzymatic activity in the 2.5 mM EDTA treatments and both CA concentrations was greater than in the control and the 5 mM EDTA treatment. CA and EDTA primarily increased the levels of ascorbate, dehydroascorbate, glutathione, and protein in the leaves, thereby enhancing the Pb tolerance of harmel plants. Furthermore, CA and EDTA significantly elevated TF values, with the highest amount observed in Pb 5 + EDTA 5, compared to plants treated with Pb alone. Correlation analysis, visualized through a heat map, revealed strong positive relationships among all growth indicators and a consistently strong negative correlation between root Pb concentration and plant growth, underscoring root Pb accumulation as the primary constraint on development. Conversely, shoot Pb concentration showed weaker associations, and TF exhibited no consistent correlation with growth suppression. Principal Component Analysis (PCA) effectively distinguished Pb-stressed from treated plants and confirmed the ameliorative effects of CA and EDTA. The PCA further highlighted distinct physiological response patterns induced by the two chelating agents, suggesting different underlying mechanisms of action. Overall, CA and EDTA improved Pb tolerance in harmel by modulating antioxidant defense systems and influencing Pb distribution, offering promising tools for enhancing phytoremediation potential under Pb-contaminated conditions.
Similar content being viewed by others
Introduction
Soil contamination with heavy metals, particularly Pb, poses a significant global environmental challenge due to its persistence, toxicity, and adverse effects on soil fertility and human health1,2,3,4. Originating from mining, smelting, and agrochemical overuse, Pb accumulates in plants, disrupting photosynthesis, nutrient uptake, and antioxidant defenses, which leads to reduced growth and productivity5,6,7,8.
Phytoremediation, a plant-based approach to detoxify polluted environments, offers a sustainable alternative to conventional methods9,10. However, its efficiency is often constrained by low metal bioavailability in soils and the limited accumulation capacity of most plant species. To overcome these challenges, chelating agents such as EDTA and CA have been applied to improve Pb solubility and uptake 11,12,13,14,15.
In addition, chelating agents such as EDTA and CA address these limitations by increasing Pb solubility and uptake. EDTA forms stable complexes that enhance Pb translocation to shoots12, while CA, a biodegradable acid, acidifies the rhizosphere and boosts antioxidant responses12,13,14,15. Despite their efficacy, the mechanisms by which these agents mediate Pb tolerance in specific plants remain poorly understood8,12.
Furthermore, P. harmala is a perennial herb native to arid and semi-arid regions. Although not classified as a hyperaccumulator, its tolerance to harsh conditions and moderate metal accumulation capacity make it a promising candidate for phytoremediation in marginal soils5,7,16,17. Yet, its response to Pb stress and the role of chelators like EDTA and CA are underexplored18,19,20,21.
Consequently, this study addresses these gaps by investigating harmel’s physiological responses under Pb stress using a hydroponic system, which provides a controlled environment to assess chelator effects without the complexities of soil matrices. While this approach minimizes variability, its findings require validation for extrapolation to soil conditions. Therefore, the objectives are to (1) assess the physiological effects of Pb on harmel seedlings after a 14-day exposure period, (2) compare the effects of different concentrations of CA and EDTA on seedling growth, Pb accumulation, and physiological attributes under Pb stress, and (3) evaluate the relationship between antioxidant enzyme activities and Pb tolerance in harmel seedlings.
Materials and methods
Plant materials and Pb treatments
P. harmala is a wild and widely distributed plant species that is not currently considered at risk of extinction. However, to ensure the sustainability of its populations and in accordance with scientific and ethical standards, seed collection was conducted carefully and in compliance with local regulations. The identification of P. harmala was confirmed by S. M. Mir Tajadiny. Voucher specimens of both the plant and its seeds have been deposited in the Herbarium of Payame Noor University of Kerman. We confirm that the necessary permits were obtained for collecting seeds from the metallicolous population located in the Koshk area. Bulk seed collections (> 25 plants) of P. harmala L. were made from the 60-year-old Koshk Pb/Zn mining site (metallicolous population). The collected seeds were surface-sterilized in 2% sodium hypochlorite solution for 10 min and subsequently washed three times with double-distilled water to remove any residual sterilizing agent. Following sterilization, the seeds were evenly planted in plastic pots filled with sterile perlite as the growing medium. Six seeds were placed in each pot to ensure adequate germination rates and uniform growth. The seedlings were grown in a controlled greenhouse environment maintained at day/night temperatures of 25/20 °C with a light intensity of 200 μE m-2 s−1. Plants were watered with Hoagland nutrient solution throughout the initial 40-day growth period to ensure optimal conditions.
Experimental design and lead treatments
The concentrations of Pb (0, 5 and 25 mg L−1), CA (0, 2.5 and 5 mM), and EDTA (0, 2.5 and 5 mM) were selected based on previous studies on heavy metal stress and chelator-assisted phytoextraction, as well as preliminary optimization experiments conducted specifically for P. harmala under hydroponic conditions. These concentrations were found to be physiologically relevant for inducing measurable responses in growth, antioxidant activity, and Pb uptake, without causing acute toxicity. The experiment was conducted following a Completely Randomized Design (CRD). After an initial 40-day growth period, uniform P. harmala seedlings were subjected to treatments with Pb, CA, and EDTA treatments for a duration of 14 days. The study comprised 15 treatment combinations: T1: 0 mg L−1 Pb (control); T2: 5 mg L−1 Pb; T3: 25 mg L−1 Pb; T4: 0 mg L−1 Pb + 2.5 mM CA; T5: 0 mg L−1 Pb + 5 mM CA; T6: 5 mg L−1 Pb + 2.5 mM CA; T7: 5 mg L−1 Pb + 5 mM CA; T8: 25 mg L−1 Pb + 2.5 mM CA; T9: 25 mg L−1 Pb + 5 mM CA; T10: 0 mg L−1 Pb + 2.5 mM EDTA; T11: 0 mg L−1 Pb + 5 mM EDTA; T12: 5 mg L−1 Pb + 2.5 mM EDTA; T13: 5 mg L−1 Pb + 5 mM EDTA; T14: 25 mg L−1 Pb + 2.5 mM EDTA; T15: 25 mg L−1 Pb + 5 mM EDTA, treatments7. Each treatment was replicated three times (n = 3), with each replicate consisting of one pot containing six uniform seedlings. Treatments were administered hydroponically by simultaneously adding Pb (as Pb(NO3)2), CA, and EDTA at the designated concentrations to the Hoagland nutrient solution. Plants were harvested seven days after the 14-day treatment period, at 61 days of age.
Growth criteria
Root and shoot lengths were measured immediately after harvesting using a standard ruler. Following thorough rinsing and drying, the dry biomass of root and shoot tissues was determined by weighing the oven-dried samples with an analytical balance.
Lead determination and translocation factor estimation
After completion of the treatments, root and shoot samples were thoroughly rinsed three times with demineralized water to remove surface contaminants. The samples were then oven-dried at 70 °C for 48 h, and their dry biomass was recorded. Dried tissues were ground into a fine powder and digested using a mixture of concentrated nitric acid and hydrogen peroxide (HNO3:H2O2, 3:1 v/v) in a microwave digestion system. The concentration of lead in the digested samples was determined using flame atomic absorption spectrometry (FAAS), following the methodology described by Mahdavian et al.7.
Lead uptake and its internal movement within P. harmala were assessed by calculating the TF, which indicates the plant’s efficiency in transporting heavy metals from roots to shoots. TF was determined as the ratio of Pb concentration in the shoot to that in the root, using the following formula:
Lipid peroxidation
Lipid peroxidation was assessed by quantifying malondialdehyde (MDA) content, a widely used indicator of oxidative damage. Briefly, plant tissue homogenates were mixed with thiobarbituric acid (TBA) in trichloroacetic acid (TCA), heated at 95 °C for 30 min, cooled on ice, and subsequently centrifuged. The absorbance of the resulting supernatant was measured at 532 nm, with non-specific absorbance at 600 nm subtracted to correct for turbidity and other interferences. MDA concentration was calculated using an extinction coefficient of 155 mM−1 cm−1 and expressed as nmol g−1 fresh weight22. Additionally, other aldehydes were quantified by measuring absorbance at 455 nm23.
Measurement of glutathione (GSH), dehydroascorbate (DHA) and ascorbate (ASC)
The levels of ascorbate (ASC) and dehydroascorbate (DHA) were determined following the method of De Pinto et al.24. Total ascorbate was quantified by reducing DHA to ASC using dithiothreitol (DTT). The ASC content was measured spectrophotometrically based on its reaction with 2,2’-dipyridyl in the presence of Fe3+, with absorbance recorded at 525 nm. DHA concentration was calculated as the difference between total ascorbate and reduced ascorbate.
Glutathione (GSH) content was measured using 5,5’-dithiobis-(2-nitrobenzoic acid) (DTNB) according to Ellman’s method25. The reaction mixture contained plant extract, phosphate buffer (pH 8.0), and DTNB. The formation a yellow-colored complex was monitored at 412 nm, and GSH concentration was determined using a standard curve and expressed as µmol g−1 fresh weight.
Enzyme extraction and assays
Protein concentration in enzyme extracts was determined using the Bradford method26, with bovine serum albumin (BSA) as the standard.
Fresh leaf samples (1.0 g) were homogenized in 6 mL of cold 50 mM potassium phosphate buffer (pH 7.8) containing 0.2 mM EDTA and 2% (w/v) polyvinylpyrrolidone (PVP) using a mortar and pestle on ice. The homogenate was centrifuged at 12,000 rpm for 20 min at 4 °C, and the resulting supernatant was used for the determination of enzyme activities, including catalase (CAT), ascorbate peroxidase (APX), guaiacol peroxidase (GPX), superoxide dismutase (SOD) and lipoxygenase (LOX).
-
Catalase (CAT) activity was measured spectrophotometrically according to Aebi27. The reaction mixture contained 5 mM H2O2 in 50 mM potassium phosphate buffer (pH 7.0). The reaction was initiated by adding 100 μL of enzyme extract to 900 μL of the reaction solution. The decrease in absorbance at 240 nm was monitored, and CAT activity was calculated using an extinction coefficient of 0.039 mM−1 cm−1. One unit of CAT was defined as the amount of enzyme that decomposes 1 μmol of H2O2 per minute at 25 °C.
-
Ascorbate peroxidase (APX) activity was determined using the method described by Boominathan and Doran28 and Nakano and Asada29. The reaction mixture contained of 725 μL of 50 mM potassium phosphate buffer (pH 7.0) with 0.2 mM EDTA, 175 μL of 0.5 mM ascorbic acid, and 50 μL of enzyme extract. The reaction was initiated by the addition of 50 μL of 200 mM H2O2. The decrease in absorbance at 290 nm was monitored, and APX activity was calculated using an extinction coefficient of 2.6 mM−1 cm−1. One unit of APX was defined as the oxidation of 1 μmol of ascorbate per minute at 25 °C.
-
Guaiacol peroxidase (GPX) activity was assayed following the method of Plewa et al.30 by measuring the increase in absorbance at 470 nm due to tetraguaiacol formation. The reaction mixture included 50 mM potassium phosphate buffer (pH 7.0), 0.3% H2O2, 1% guaiacol, and 20 μL of enzyme extract. The extinction coefficient of 25.5 mM−1 cm−1 was used to calculate GPX activity. One unit of GPX was defined as the amount of enzyme producing 1 μmol of tetraguaiacol per minute.
-
Superoxide dismutase (SOD) activity was determined based on the method of Giannopolitis and Ries31. The 3 mL reaction mixture included 50 μL enzyme extract, 2 mM riboflavin, 75 mM nitroblue tetrazolium (NBT), 13 mM methionine, 0.1 mM EDTA, and 50 mM phosphate buffer (pH 7.8). One unit of SOD was defined as the amount of enzyme required to inhibit 50% of NBT reduction under light. Absorbance was measured at 560 nm. Although no extinction coefficient is strictly required, an approximate ε value of 12.8 mM−1 cm−1 at 560 nm may be used for quantification if necessary.
-
Lipoxygenase (LOX) activity was measured according to Doderer et al.32. The reaction mixture contained 100 mM linoleic acid and 100 mM acetic acid buffer (pH 5.5). The reaction was initiated by adding 50 μL of enzyme extract, and the increase in absorbance at 234 nm was recorded. LOX activity was expressed as units per mg protein using an extinction coefficient of 25,000 M−1 cm−1.
Statistical analysis
All results are presented as mean values ± standard error (SE) based on three biological replicates. The experiment was conducted using a completely randomized design (CRD) with a factorial arrangement. Data were analyzed using two-way analysis of variance (ANOVA) to assess the main effects of treatments and their interactions. Statistical analyses were performed using IBM SPSS Statistics version 26. Post hoc comparisons were conducted using Tukey’s Honestly Significant Difference (HSD) test at a 5% significance level (p < 0.05). Graphs were generated using Microsoft Excel 2016. The PCA was carried out using Python version 3.x, with the ‘scikit-learn’ library employed for dimensionality reduction and the ‘seaborn’ and ‘matplotlib’ libraries for visualization. Pearson correlation coefficients were calculated to assess the relationships between growth parameters and Pb accumulation. Correlation matrices were visualized as heatmaps using the Seaborn library in Python.
Results
Plant growth
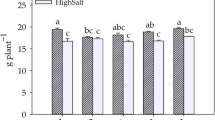
Exposure to Pb at concentrations of 5 and 25 mg L−1 resulted in a significant reduction in the growth of harmel plants compared to the control. Specifically, shoot lengths decreased by 16% and 38% at 5 mg L−1 and 25 mg L−1 Pb, respectively, relative to untreated plants (Fig. 1). Similar declines were observed in root length. Additionally, dry weights of both shoots and roots showed significant reductions in the presence of Pb, as illustrated in Fig. 2. Interestingly, the co-application of Pb (5 and 25 mg L−1) with CA (2.5 and 5 mM) notably improved growth metrics. The highest increases in shoot and root lengths—69% and 35%, respectively—were recorded under a combined treatment of 2.5 mM CA and 5 mg L−1 Pb, compared to their control. Similarly, dry weights of shoots and roots improved by 72% and 6%, respectively, under CA (2.5 mM) and Pb (5 mg L−1) compared to Pb-treated plants (5 mg L−1) alone (Fig. 1, 2). EDTA (2.5 mM) alone, and with Pb (5 mg L−1) significantly increased all growth parameters. The lowest effect was observed in all growth parameters measured at Pb 25 mg L−1 + EDTA 5 mM concentration, and the highest was obtained with 2.5 mM CA treatment (Figs. 1, 2).
Malondialdehyde (MDA) and other aldehydes
Lead exposure significantly elevated malondialdehyde (MDA) and other aldehydes in harmel plants. At a concentration of 25 mg L−1 Pb, MDA and aldehyde levels increased by 36% and 76%, respectively, compared to the control (Fig. 3). However, the addition of CA and EDTA mitigated these effects, reducing MDA levels by 33% and aldehydes by 15% in plants treated with 25 mg L−1 Pb and 2.5 mM CA, respectively, compared to Pb alone. Also, Pb (25 mg L−1) and EDTA (2.5 mM) significantly decreased MDA and other aldehyde by 38% and 12%, respectively, in comparison with treated plants under Pb alone (Fig. 3A, B).
Ascorbate, dehydroascorbate and glutathione concentrations
In this study, harmel plants showed a significant increase in ascorbate and dehydroascorbate content in Pb conditions compared to the control. The ascorbate and dehydroascorbate in the leaves at a concentration of 5 mg L−1 Pb increased by 83% and 50%, respectively, compared to the control. However, CA and EDTA increased the content of ascorbate and dehydroascorbate. The highest amount of dehydroascorbate and ascorbate was obtained in plants that used Pb (5 mg L−1) and EDTA (2.5 and 5 mM), respectively, compared with Pb alone, and the lowest contents were achieved with a 25 mg L⁻1 Pb treatment. Pb (5 mg L−1) and EDTA (2.5 and 5 mM) significantly increased ascorbate by 76% and 99%, while dehydroascorbate by 160% and 34%, respectively, compared with Pb alone. Also, Pb (5 mg L−1) and CA (2.5 and 5 mM) significantly increased ascorbate by 34% and 27%, while dehydroascorbate by 26% and 147%, respectively, in comparison with Pb alone (Fig. 4). In this study, the concentration of glutathione in harmel plants was significantly elevated when exposed to Pb. Specifically, treatment with 5 mM and 25 mM Pb resulted in a 36% and 63% increase in glutathione levels, respectively, compared to the control group (Fig. 5). Additionally, the application of CA and EDTA at 2.5 mM concentrations further enhanced the glutathione content by 94% and 83%, respectively, 25 mM Pb compared to plants treated with Pb alone. The most pronounced increase in glutathione, 272%, was observed in plants treated with 2.5 mM CA, in comparison to the control group (Fig. 5).
Protein content
The application of 5 and 25 mg L−1 Pb resulted in a decrease in leaf protein concentration by 19% and 46%, respectively, compared to the control group. Adding of CA and EDTA significantly reduced the harmful effect of Pb on protein content. The highest amount of protein content (31%) was obtained in plants that used Pb (5 mg L−1) and CA (5 mM), In contrast to the plants under Pb (5 mg L−1) alone. Also, Pb (5 mg L−1) and EDTA (2.5 mM) significantly enhanced the protein content by 28% in comparison with treated plants under Pb alone (Fig. 6).
Antioxidant enzyme activity
In the 5 and 25 mg L−1 Pb treatments, the antioxidant enzyme activities in the leaves were higher than in the control. At 25 mg L⁻1 Pb, CAT, SOD, and GPX activities increased by 35.3%, 156.5%, and 17.8%, respectively, compared to the control. However, APX and LOX showed minimal change. Application of CA and EDTA in combination with Pb further altered enzyme activities. The highest CAT and SOD activities were observed in Pb 25 + EDTA 5, with increases of 355.7% and 174.9%, respectively, over the control. APX activity peaked in Pb5 + CA 5, representing a 272.4% increase over the control. Similarly, LOX activity was highest in Pb 5 + CA 5, a 399.0% increase compared to the control. Conversely, GPX activity generally decreased under CA and EDTA treatments. For instance, in Pb 0 + EDTA 5, GPX activity dropped, a 81.9% reduction relative to Pb 25 alone, suggesting an inhibitory effect of EDTA on this enzyme. These results suggest that Pb stress induces oxidative responses in harmel, and both CA and EDTA modulate antioxidant activity differently, with CA being more effective in enhancing APX and LOX, while EDTA significantly boosts CAT and SOD activities. (Fig. 7).
Lead uptake and translocation factor
As illustrated in Fig. 8, the concentration of Pb varied across different treatments, with the highest levels observed in the roots, followed by the shoots. This suggests that the roots serve as the primary site for Pb storage. (Fig. 8). Lead concentrations increased in plants treated with 25 mg L−1 lead by 39% and 226% in shoots and roots, respectively, compared with plants treated with 5 mg L−1 Pb soil. The addition of CA and EDTA affected Pb uptake and harmel transport. Plants exposed to 2.5 mM CA exhibited a reduced accumulation of Pb in various plant tissues when compared to those grown in Pb -contaminated soil (Fig. 8). The application of CA further reduced Pb concentrations by 79% in shoots, while 14% in roots, when applying a combination of 2.5 mM CA and 5 mg L−1 Pb compared with the control. The concentration in both the shoot and root increased by 317% and 108%, respectively, when exposed to 2.5 mM EDTA and 5 mg L−1 Pb, in comparison to their corresponding control treatments. The most notable increase was observed in the case of Pb. 5 + EDTA 2.5 treatment in shoots and Pb 25 + EDTA 5 treatment in roots (Fig. 8).
The analysis of variance revealed a significant influence of Pb concentration on the transfer factor (TF) in harmel plants. As Pb concentration in the soil increased, the displacement coefficient of harmel plants decreased. Furthermore, the application of CA and EDTA notably enhanced the TF values, with the highest values recorded in the treatment combining Pb at 5 mg L−1 and EDTA at 5. In comparison to plants exposed solely to Pb, the transfer factor exceeded one in this treatment. (Fig. 8).
Correlation analysis between growth indicators and Pb uptake
To comprehensively evaluate the interrelationships between various plant growth indicators and Pb uptake and accumulation, a Pearson correlation analysis was performed. The results are presented as a heatmap in Fig. 9. Correlation coefficients (r) range from −1 to + 1, where values close to + 1 indicate a strong positive correlation, values near -1 represent a strong negative correlation, and values close to zero suggest the absence of a linear relationship.
Heatmap of Pearson correlation coefficients between plant growth indicators and Pb uptake parameters. The heatmap was generated using Python (version 3.x) in a Google Colab environment with the libraries pandas (v2.2.2), Seaborn (v0.13.2, https://seaborn.pydata.org/), and matplotlib (v3.10.0).
Strong positive correlations among growth indicators
All measured growth parameters—shoot length (cm), root length (cm), shoot dry weight (DW, g), and root dry weight (DW, g)—exhibited strong positive correlations with one another (ranging from 0.73 to 1.00). This finding implies that any factor enhancing one growth parameter tends to positively influence the others. For instance, shoot and root dry weights showed a particularly strong correlation (r = 0.93), indicating coordinated biomass accumulation.
Strong negative correlation between growth indicators and root Pb accumulation
A strong negative correlation was observed between all growth indicators and the concentration of Pb in the root (Pb Root). These correlation coefficients ranged from r = -0.53 (between Shoot Length and Pb Root) to r = -0.77 (between Root DW and Pb Root). This robust negative relationship signifies that an increase in Pb accumulation within the roots is significantly associated with a decrease in overall plant growth across all dimensions (shoot and root length, and dry weight). In essence, high Pb accumulation in roots acts as a limiting factor for the plant’s overall development.
Weaker negative correlation between growth indicators and shoot Pb accumulation
The concentration of Pb in the shoot (Pb Shoot) exhibited weaker negative correlations with growth indicators (correlation coefficients ranging from r = −0.22 to r = −0.46). This observation might suggest that the primary damage inflicted by Pb on the plant predominantly originates from its accumulation in the roots, or that the plant possesses mechanisms to either reduce Pb translocation from roots to shoots or tolerate Pb better within shoot tissues.
Correlations among Pb uptake indicators
Shoot and root Pb concentrations showed a moderate positive correlation with each other (r = 0.41). This indicates that increased Pb in the roots can Pb to greater translocation to the shoots, although this relationship is not as strong as the one between root Pb and growth parameters. The Translocation Factor, which represents the ratio of Pb transferred from roots to shoots, had a strong positive correlation (r = 0.58) with the Pb concentration in the shoot, which is logically expected as higher shoot Pb contributes to a higher TF. However, TF exhibited weak and mostly negative correlations with both growth indicators and root Pb levels (ranging from −0.14 to −0.28). This implies that the proportion of Pb translocated to shoots does not directly influence growth and that other factors may play a more dominant role in determining plant performance under Pb stress.
In conclusion, the results of the correlation analysis confirm that Pb accumulation—particularly in the roots—has a strong inhibitory effect on plant growth. These findings highlight the central role of root-level Pb toxicity and reveal the intricate balance between heavy metal uptake, translocation, and plant physiological responses. Understanding these relationships is crucial for developing effective phytoremediation strategies and improving plant resilience under metal stress.
The PCA of treatments effects
To comprehensively evaluate the complex interactions and overall plant responses to Pb toxicity, CA, and EDTA, PCA was performed on the treatment data, with 95% confidence ellipses added to assess the statistical reliability of the observed clusters (Fig. 10). The PCA score plot (Fig. 10) illustrates the distribution and relationships among the different treatments, with PC1 and PC2 accounting for 36.41% and 19.23% of the total variance, respectively. The PCA revealed distinct clusters, reflecting the varying effectiveness of CA and EDTA in mitigating Pb-induced stress.
Lead-only treatments (T1, T2, T3)
The control (T1: 0 mg L−1 Pb) is positioned distinctly on the plot, with its 95% confidence ellipse showing minimal overlap with T2 (5 mg L−1 Pb) and T3 (25 mg L-1 Pb), which shift progressively along PC1. The distinct separation of T3’s ellipse signifies severe Pb-induced stress.
Impact of CA on Pb-toxicity (T6, T7, T8, T9)
Treatments combining Pb and CA (T6: 5 mg L−1 Pb + 2.5 mM CA; T7: 5 mg L−1 Pb + 5 mM CA; T8: 25 mg L−1 Pb + 2.5 mM CA; T9: 25 mg L−1 Pb + 5 mM CA) exhibit overlapping 95% confidence ellipses, suggesting similarity in response, though T9 shows a distinct ellipse in the upper-left quadrant, indicating effective mitigation of high Pb stress. T7 is positioned toward the upper region, reflecting a beneficial impact of 5 mM CA under moderate Pb exposure, while T6, closer to T2, indicates a less pronounced effect with 2.5 mM CA.
Effect of EDTA on Pb-toxicity (T12, T13, T14, T15)
Treatments combining EDTA with Pb (T12: 5 mg L−1 Pb + 2.5 mM EDTA; T13: 5 mg L−1 Pb + 5 mM EDTA; T14: 25 mg L−1 Pb + 2.5 mM EDTA; T15: 25 mg L−1 Pb + 5 mM EDTA) display distinct patterns. T15 is located in the bottom-right quadrant with a non-overlapping 95% confidence ellipse, suggesting a unique physiological response compared to T3 and T9. T13 and T12 are closely grouped with overlapping ellipses, indicating that even low EDTA concentrations significantly affect plant responses under moderate Pb stress.
Comparison of CA and EDTA responses
The PCA plot illustrates a notable divergence between CA- (T6–T9) and EDTA-treated groups (T12–T15), with non-overlapping 95% confidence ellipses highlighting distinct physiological responses. CA-treated plants occupy the upper regions, while EDTA-treated plants disperse toward the lower quadrants, reflecting differences in chelation efficiency. The control treatments involving only CA (T4, T5) or EDTA (T10, T11) cluster near T1 with overlapping ellipses, indicating minimal impact in the absence of Pb. Overall, the PCA effectively distinguishes between the various treatment groups and provides visual confirmation of the ameliorative roles of CA and EDTA in mitigating Pb toxicity.
Discussion
This study aimed to evaluate the effects of CA and EDTA on growth, physiological responses, antioxidant defense mechanisms, and Pb accumulation in P. harmala under Pb stress. The findings elucidate the mechanistic pathways by which these chelators regulate Pb uptake, detoxification, and stress tolerance in this species.
Pb exposure significantly inhibited plant growth and biomass accumulation, consistent with previous evidence that Pb binds to sulfhydryl groups in enzymes, disrupting cellular metabolism, impairing photosynthesis, and inducing oxidative stress via reactive oxygen species (ROS)33,34,35,36. However, CA and EDTA mitigated these effects through distinct mechanisms. Both chelators enhanced Pb bioavailability by forming soluble Pb–chelate complexes, facilitating root uptake via transporters like ZIP family proteins1,37. EDTA forms stable, high-affinity Pb–EDTA complexes, promoting apoplastic transport and xylem loading through upregulation of HMA4 transporters, thus enhancing long-distance translocation12. In contrast, CA mobilizes Pb via weaker Pb–CA complexes and rhizosphere acidification, potentially activating ALMT-family organic acid efflux pumps, which increases root uptake while favoring Pb retention to protect photosynthetic tissues14,15.
The concentration-dependent growth response was notable: 2.5 mM EDTA improved plant height and biomass by stabilizing Pb and reducing free ion toxicity, whereas 5 mM EDTA caused inhibition, likely due to excessive chelation overwhelming membrane transporters (e.g., IRT1) and disrupting nutrient uptake38,39,40. CA-treated plants exhibited greater biomass and reduced toxicity symptoms, attributable to CA’s lower phytotoxicity and biodegradability, which minimizes cellular stress compared to EDTA’s synthetic complexes41,42,43,44. These trends align with findings in Brassica napus41, Typha latifolia42, centipedegrass43 and Iris halophila 44.
Supporting evidence from Zhang et al.45 showed that low EDTA concentrations preserved Festuca arundinacea biomass, while high doses impaired growth, consistent with our results. Similarly, low EDTA levels in Sedum hybridum had minimal impact, likely due to reduced free ionic metal stress via chelation46, highlighting the threshold effect of chelator concentration.
Both CA and EDTA activated specific antioxidant pathways to counter Pb-induced ROS. Elevated MDA levels indicated lipid peroxidation47, but chelator treatments enhanced the AsA–GSH cycle. Increased ASC, DHA, and GSH levels in CA-treated plants (Figs. 4, 5) suggest robust H2O2 detoxification via APX and GR, potentially driven by CA-induced upregulation of GST and APX genes48,49. EDTA also boosted antioxidant activity, but its higher translocation of Pb–EDTA complexes may exceed redox capacity at 5 mM, contributing to toxicity50. Enzymatic antioxidants—SOD, CAT, POX, and APX—showed increased activity in Pb-stressed plants (Fig. 7), with CA preferentially enhancing APX/CAT pathways via SOD1 and CAT1 gene expression, while EDTA supported cofactor availability (e.g., Fe for CAT)50,51,52. These findings align with studies on B. napus41, centipedegrass43, and rice50.
Pb stress reduced protein content (Fig. 6), likely due to oxidative degradation, but CA and EDTA restored levels by mitigating ROS and improving nutrient uptake, possibly via enhanced NRT1 gene expression for nitrogen assimilation43. Regarding Pb distribution, greater root accumulation than shoots (Fig. 8) reflects a defense strategy. EDTA significantly increased shoot Pb and TF at 5 mM, confirming its role in xylem loading, while CA promoted moderate uptake with minimal toxicity, supporting its safer profile 1,53,54,55,56,57.
Correlation analysis (Fig. 9) showed strong negative correlations between root Pb and growth traits (e.g., r = − 0.77 with root DW), indicating root-level toxicity as a key constraint37,58,59. PCA (Fig. 10) substantiated these patterns, with distinct clusters for CA (T6–T9) and EDTA (T12–T15) treatments, reflecting differential mechanistic pathways, consistent with wheat and maize studies60.
Overall, EDTA enhances Pb translocation via stable complexes but poses phytotoxicity risks at high doses, while CA improves uptake and tolerance through acidification and antioxidant activation. These mechanistic insights, derived from hydroponic conditions, provide a foundation for soil-based applications, requiring further optimization of chelator doses.
Conclusion
This study confirms Peganum harmala’s tolerance to Pb stress and its phytoremediation potential. Pb accumulates predominantly in roots, limiting shoot translocation and protecting photosynthetic tissues. At 2.5 mM, CA and EDTA enhanced growth, reduced oxidative stress (MDA), and boosted antioxidants (AsA, GSH, proteins), with CA showing greater efficacy. However, 5 mM EDTA increased Pb translocation but impaired growth due to toxicity. PCA highlighted distinct mitigation mechanisms for CA and EDTA, supporting CA-assisted phytoextraction as a safer, sustainable option. These hydroponic findings warrant further soil-based validation.
Data availability
The raw data of this article will be made available by corresponding author (Dr. Kobra Mahdavian; k.mahdavian@pnu.ac.ir), according to the personal requests.
References
Chen, Y. et al. A critical review on EDTA washing in soil remediation for potentially toxic elements (PTEs) pollutants. Rev. Environ. Sci. Biotechnol. 15, 1–25 (2022).
Plunkett, S. A., Eckley, C. S., Luxton, T. P. & Johnso, M. G. The effects of biochar and redox conditions on soil Pb bioaccessibility to people and waterfowl. Chemosphere 294, 133675 (2022).
Oladoye, P. O., Olowe, O. M. & Asemoloye, M. D. Phytoremediation technology and food security impacts of heavy metal contaminated soils: A review of literature. Chemosphere 288, 132555 (2022).
Sidhu, G. P. S., Bali, A. S., Singh, H. P., Batish, D. R. & Kohli, R. K. Insights into the tolerance and phytoremediation potential of Coronopus didymus L. (Sm) grown under zinc stress. Chemosphere 244, 125350 (2020).
Mahdavian, K. The role of organic acids and thiol compounds in detoxification and tolerance of Zn stress in two populations of harmel. Russ. J. Plant Physiol. 69, 58 (2022).
Mahdavian, K. Detoxification role of amino acids and phytochelatins on two populations of harmel plant under silver stress. Environ. Sci. Pollut. Res. 30, 110970–110980 (2023).
Mahdavian, K., Ghaderian, S. M. & Schat, H. Pb accumulation, Pb tolerance, antioxidants, thiols, and organic acids in metallicolous and non-metallicolous Peganum harmala L. under Pb exposure. Environ. Exp. Bot. 126, 21–31 (2016).
Fu, X. P. et al. A review of progress in roles of organic acids on heavy metal resistance and detoxification in plants. Chin. J. Plant Ecol. 534, 1354–1358 (2010).
Li, Z. M., Song, Z. L., Parr, J. F. & Wang, H. L. Occluded C in rice phytoliths: Implications to biogeochemical carbon sequestration. Plant Soil 370, 615–623 (2013).
Li, Y. et al. Effect of plant extracts and citric acid on phytoremediation of metal-contaminated soil. Ecotoxicol. Environ. Saf. 211, 111902 (2021).
Shukla, M. et al. Remediation of chromium contaminated soil by soil washing using EDTA and N-acetyl-L-cysteine as the chelating agents. Prog. Org. Coat. 165, 106704 (2022).
Madhupriyaa, D. et al. Efficacy of Chelated micronutrients in plant nutrition. Commun. Soil Sci. Plant Anal. 55(22), 3609–3637 (2024).
Nas, F. S. & Ali, M. The effect of lead on plants in terms of growing and biochemical parameters: A review. MOJ Ecol. Environ. Sci. 3(4), 265–268 (2018).
Jones, D. L. Organic acids in the rhizosphere–a critical review. Plant Soil 205, 25–44 (1998).
Dakora, F. D. & Phillips, D. A. Root exudates as mediators of mineral acquisition in low-nutrient environments. Plant Soil 245(1), 35–47 (2002).
Mahdavian, K., Ghaderian, S. M. & Torkzadeh-Mahani, M. Accumulation and phytoremediation of Pb, Zn, and Ag by plants growing on Koshk lead-zinc mining area, Iran. J. Soil Sediment 17, 1310–1320 (2017).
Cao, X. R. et al. Responses of soil bacterial community and Cd phytoextraction to a Sedum alfredii-oilseed rape (Brassica napus L. and Brassica juncea L.) intercropping system. Sci. Total Environ. 723, 138152 (2020).
Mahdavian, K. Effect of citric acid on antioxidant activity of red bean (Phaseolus calcaratus L.) under Cr+6 stress. S. Afr. J. Bot. 139, 83–91 (2021).
Rostami, S. & Azhdarpoor, A. The application of plant growth regulators to improve phytoremediation of contaminated soils: A review. Chemosphere 220, 818–827 (2019).
Suthar, V., Memon, K. S. & Mahmood-Ul-Hassan, M. EDTA-enhanced phytoremediation of contaminated calcareous soils: Heavy metal bioavailability, extractability, and uptake by maize and sesbania. Environ. Monit. Assess. 186, 3957–3968 (2014).
Guo, J. et al. Effects of EDTA and plant growth-promoting rhizobacteria on plant growth and heavy metal uptake of hyperaccumulator Sedum alfredii Hance. J. Environ. Sci. 88, 361–369 (2020).
Heath, R. L. & Packer, L. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 125, 189–198 (1968).
Meirs, S., Philosoph-Hadas, S. & Aharoni, N. Ethylene increased accumulation of fluorescent lipid peroxidation products detected during senescence of parsley by a newly developed method. J. Am. Soc. Hortic. Sci. 117, 128–132 (1992).
De Pinto, M. C., Francis, D. & De Gara, L. The redox state of the ascorbate-dehydroascorbate pair as a specific sensor of cell division in tobacco BY-2 cells. Protoplasma 209, 90–97 (1999).
Ellman, G. L. Tissue sulfydryl groups. Arch. Biochem. Biophys. 82, 70–77 (1959).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Aebi, H. E. & Bergmeyer, H. U. (eds) Methods of enzymatic analysis 3rd edn, 273–286 (Elsevier, 1983).
Boominathan, R. & Doran, P. M. Ni-induced oxidative stress in roots of the Ni hyperaccumulator, Alyssum bertolonii. New Phytol. 156, 205–215 (2002).
Nakano, Y. & Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 22, 867–880 (1981).
Plewa, M. J., Smith, S. R. & Wanger, E. D. Diethyldithiocarbamate suppresses the plant activation of aromatic amines into mutagens by inhibiting tobacco cell peroxidase. Mutat. Res. 247, 57–64 (1991).
Giannopolitis, C. N. & Ries, S. K. Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol. 59, 309–314 (1977).
Doderer, A. et al. Purification and characterization of two lipoxygenase isoenzymes from germinating barley. Biochem. Biophys. Acta 1120, 97–104 (1992).
Salt, D. E. et al. Phytoremediation: A novel strategy for the removal of toxic metals from the environment using plants. Bio/Technology 13(5), 468–474 (1995).
Tahjib-Ul-Arif, M. et al. Citric acid-mediated abiotic stress tolerance in plants. Int. J. Mol. Sci 22(13), 7235 (2021).
Evangelou, M. W., Ebel, M. & Schaeffer, A. Chelate assisted phytoextraction of heavy metals from soil. Effect, mechanism, toxicity, and fate of chelating agents. Chemosphere 68(6), 989–1003 (2007).
Rahman, S. U. et al. Pb uptake, accumulation, and translocation in plants: Plant physiological, biochemical, and molecular response: A review. Heliyon 10(6), e27724 (2024).
Zhou, J., Zhang, Z., Zhang, Y., Wei, Y. & Jiang, Z. Effects of lead stress on the growth, physiology, and cellular structure of privet seedlings. PLoS ONE 13(3), e0191139 (2018).
Hosseini, S. S., Lakzian, A., Halajnia, A. & Razavi, B. S. Optimization of EDTA and citric acid for risk assessment in the remediation of lead contaminated soil. Rhizosphere 17, 100277–100287 (2021).
Vigliotta, G., Matrella, S., Cicatelli, A., Guarino, F. & Castiglione, S. Effects of heavy metals and chelants on phytoremediation capacity and on rhizobacterial communities of maize. J. Environ. Manage 179, 93–102 (2016).
Cay, S., Uyanik, A., Engin, M. S. & Kutbay, H. G. Effect of EDTA and Tannic Acid on the Removal of Cd, Ni, Pb and Cu from Artificially Contaminated Soil by Althaea rosea Cavan. Int. J. Phytoremediat. 17(6), 568–574 (2015).
Shakoor, M. B. et al. Citric acid improves lead (Pb) phytoextraction in Brassica Napus L. by mitigating Pb-induced morphological and biochemical damages. Ecotoxicol. Environ. Saf. 109, 38–47 (2014).
Amir, W. et al. Accumulation potential and tolerance response of Typha Latifolia L. under citric acid assisted phytoextraction of lead and mercury. Chemosphere 257, 127247 (2020).
Cai, X. et al. Low-molecular-weight organic acid-mediated tolerance and Pb accumulation in centipedegrass under Pb stress. Ecotoxicol. Environ. Saf. 241, 113755 (2022).
Han, Y. L., Zhang, L. L., Gu, J. G., Zhao, J. Z. & Fu, J. J. Citric acid and EDTA on the growth, photosynthetic properties and heavy metal accumulation of Iris halophila Pall. cultivated in Pb mine tailings. Int. Biodeterior. Biodegrad. 128, 15–21 (2018).
Zhang, T. et al. Chelant extraction of heavy metals from contaminated soils using new selective EDTA derivatives. J. Hazard. Mater. 262, 464–471 (2013).
Shahid, M. et al. EDTA-enhanced phytoremediation of heavy metals: A review. Soil Sediment Contam. 23(4), 389–416 (2014).
Khan, M. et al. Reduced glutathione protects subcellular compartments from Pb-induced ROS injury in leaves and roots of upland cotton (Gossypium hirsutum L.). Front. Plant. Sci 11, 412 (2020).
Peco, J. D. et al. Deciphering lead tolerance mechanisms in a population of the plant species Biscutella auriculata L. from a mining area: Accumulation strategies and antioxidant defenses. Chemosphere 261, 127721 (2020).
Xie, C. C. et al. Melatonin-assisted phytoremediation of Pb-contaminated soil using bermudagrass. Environ. Sci. Pollut. Res. Int 28, 44374–44388 (2021).
Khatun, M. R., Mukta, R. H., Islam, M. A. & Huda, A. N. Insight into citric acid-induced chromium detoxification in rice (Oryza Sativa L.). Int. J. Phytoremediat. 21, 1234–1240 (2019).
Zhang, H., Zhang, L. L., Li, J., Chen, M. & An, R. D. Comparative study on the bioaccumulation of lead, cadmium and nickel and their toxic effects on the growth and enzyme defence strategies of a heavy metal accumulator, Hydrilla verticillata (L. f.) Royle. Environ. Sci. Pollut. Res. Int. 27, 9853–9865 (2020).
Song, J. et al. Exogenous oxalic acid and citric acid improve lead (Pb) tolerance of Larix olgensis A. Henry Seedlings. Forests 9, 510 (2018).
Wu, L. H., Luo, Y. M., Xing, X. R. & Christie, P. EDTA-enhanced phytoremediation of heavy metal contaminated soil with Indian mustard and associated potential leaching risk. Agric. Ecosyst. Environ. 102, 307–318 (2004).
Jiang, M. et al. EDTA -facilitated toxic tolerance, absorption and translocation and phytoremediation of lead by dwarf bamboos. Ecotoxicol. Environ. Saf. 170, 502–512 (2019).
Masoudi, F., Shirvani, M., Shariatmadari, H. & Sabzalian, M. R. Performance of new biodegradable chelants in enhancing phytoextraction of heavy metals from a contaminated calcareous soil. J. Environ. Health Sci. Eng. 18(2), 655–664 (2020).
Aran, D. S., Harguinteguy, C. A., Fernandez-Cirelli, A. & Pignata, M. L. Phytoextraction of Pb, Cr, Ni, and Zn using the aquatic plant Limnobium laevigatum and its potential use in the treatment of wastewater. Environ. Sci. Pollut. Res 24, 18295–18308 (2017).
Kafle, A. et al. Phytoremediation: Mechanisms, plant selection and enhancement by natural and synthetic agents. Environ. Adv. 8, 100203 (2022).
El-Mahrouk, E. S. M. et al. Evaluation of the growth, enzymatic activity, electrolyte leakage, and phytoremediation efficiency of Conocarpus erectus under cadmium and lead stress. Front. Plant Sci. 15, 1466697 (2024).
Gupta, M. et al. Lead toxicity in plants: mechanistic insights into toxicity, physiological responses of plants and mitigation strategies. Plant Signal. Behav. 19(1), 2365576 (2024).
Natasha, K., Ahmad, I. & Nafees, M. Comparative analysis of Ethylenediaminetetraacetic acid and Citric Acid for enhanced Lead Phytoremediation in Wheat and Maize Plants. Biocatal. Agric. Biotechnol. 101, 103612 (2025).
Acknowledgements
I wish to thank Payame Noor University Research Council for approval and providing financial support.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, ornot-for-profit sectors.
Author information
Authors and Affiliations
Contributions
K. M was instrumental in the research project, contributing to its design by establishing objectives and methodologies. During implementation, they ensured adherence to protocols, and in the analysis phase, K. M applied statistical methods to interpret the findings. Their involvement extended to crafting the manuscript, making the research accessible and impactful.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethical approval
Not applicable.
Consent to participate
We consent to participate in this manuscript.
Consent for publication
We consent to publish this manuscript.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mahdavian, K. Synergistic modulation of antioxidant enzymes by citric acid and EDTA to enhance lead tolerance and phytoextraction efficiency in harmel. Sci Rep 15, 37890 (2025). https://doi.org/10.1038/s41598-025-21662-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-21662-7