Abstract
Major depressive disorder (MDD) is a prevalent mental health condition associated with substantial emotional and functional impairment. While interest in neurophysiological markers is growing, gender-specific neural oscillatory patterns measured by portable electroencephalogram (EEG) devices remain underexplored. This study examined whether relative spectral power in the high alpha band (11–13 Hz), recorded by a three-channel prefrontal EEG device during eyes-closed resting state, is associated with emotional states and clinical diagnosis, with attention to gender differences. Forty participants from the MODMA dataset were included, comprising 18 patients with MDD (8 females, 10 males; mean age = 30.39 ± 10.91 years) and 22 healthy controls (8 females, 14 males; mean age = 30.86 ± 9.40 years). All participants completed EEG recordings using both a 128-channel laboratory system and a portable three-channel device (Fp1, Fpz, Fp2). Emotional status was assessed using the Patient Health Questionnaire-9 (PHQ-9), Generalized Anxiety Disorder-7 (GAD-7), and Childhood Trauma Questionnaire-Short Form (CTQ-SF). Pearson correlations between relative spectral power and emotional scale scores were computed separately for males and females, with false discovery rate (FDR) correction applied to multiple comparisons. Spectral features from the portable EEG closely matched those of the lab system (r = 0.975). High alpha power was positively correlated with PHQ-9 and GAD-7 scores, with stronger and more extended correlations observed in females, sometimes reaching into the low beta range. After FDR correction, significant correlations were observed primarily at 11–14 Hz. At 11.9 Hz, PHQ-9 correlations were significant for both males (r = 0.40, p = 0.011) and females (r = 0.33, p = 0.037). High alpha power also distinguished MDD from controls (r = 0.32, p = 0.042), independent of age and education. Frontal high alpha activity measured by a portable EEG device is a promising neural marker of emotional dysregulation, with stronger and broader associations observed in females. These findings support gender-sensitive EEG-based screening approaches for mood disorders in community and clinical settings.
Similar content being viewed by others
Introduction
Major depressive disorder (MDD) is a global mental health condition that significantly affects individual well-being and socio-economic stability. Clinically, MDD is characterized by persistent sadness, hopelessness, anhedonia, and often co-occurring with anxiety, guilt, psychomotor changes, and sleep disturbances1. Alarmingly, MDD is one of the leading causes of suicide worldwide2, emphasizing the urgent need for effective and early screening approaches. Notably, the clinical presentation and prevalence of MDD differ by gender, with females exhibiting two to three times higher rates than males3,4. This disparity may relate to factors such as estrogen’s modulation of the limbic system5, increased sensitivity to social stressors, and internalizing coping styles. However, many prior studies have underrepresented females or employed male-centric animal models6,7,8, limiting the understanding of sex-specific neurobiological mechanisms9.Currently, the diagnosis of MDD relies on subjective self-reports and clinical observations, such as those outlined in the DSM-IV10. While widely used, these methods are prone to bias and inconsistencies, emphasizing the need for objective diagnostic tools11. the use of neural oscillations has emerged as a promising modality for uncovering neural biomarkers of psychiatric disorders, including MDD12,13,14,15,16,17.Among them, prefrontal alpha rhythms particularly frontal alpha asymmetry (FAA) have shown consistent associations with depression severity18,19,20, with some evidence supporting its relevance in female patients21. Though findings on prefrontal beta activity are more mixed, they also point to potential affective relevance22,23,24,25. Importantly, females often exhibit stronger prefrontal responses to negative emotional stimuli, suggesting underlying neural differences relevant to MDD pathophysiology26,27,28.
While EEG-based biomarkers offer great promise, traditional multi-channel EEG systems remain impractical for large-scale deployment due to their high cost, operational complexity, and lengthy setup times. Recent studies have shown that negative emotion regulation strategies, such as rumination, are associated with significant changes in the electroencephalogram (EEG) of the prefrontal region29,30 For patients with severe major depressive disorder (MDD), the electroencephalogram (EEG) was analyzed using power spectral density (PSD) to detect an increase in power in the theta and beta frequency bands in the frontal lobe region, while the power in the alpha frequency band decreased31. These findings indicate that the power of EEG from theta, alpha to beta is a sensitive biomarker for depression. Power spectral density (PSD) analysis is one of the most widely used methods in neuropsychiatry, used to describe the characteristics of specific frequency neural oscillations. Based on the above evidence, if we need to explore how portable electroencephalogram devices can effectively capture brain signals related to depression, then detecting in the prefrontal cortex (Fp1, Fpz, Fp2) and focusing on the power spectral density analysis of 4–20 Hz is a simple and effective solution. In recent years, Portable EEG systems offer a scalable, cost-effective, and user-friendly alternative32,33,34,35, and recent research has further validated their ability to detect emotional states, even with single-channel recordings36,37. However, their ability to capture gender-sensitive neural markers has not been fully explored, which is a key gap our research aims to address.
The present study sought to bridge this gap by using a portable prefrontal EEG to investigate gender-sensitive neural signals associated with depression and anxiety. We focus on the spectral activities in specific frequency bands, particularly the 4–20 Hz band, which encompasses theta, alpha and beta oscillations and is associated with emotional and cognitive processes. We aimed to investigate whether portable EEG could reveal different patterns across gender and psychological symptom dimensions. Our results contribute to the development of objective, accessible, and gender-related biomarkers of depression, particularly by utilizing a portable system optimized for prefrontal regions, enabling scalable and efficient screening.
Materials and methods
Dataset and participants
The dataset used in this study was sourced from the MODMA dataset 38 (https://modma.lzu.edu.cn/data/index/), established by the Key Laboratory of Wearable Computing at Lanzhou University. All participants provided written informed consent prior to participation. The study design and consent procedures were approved by the local Ethics Committee for Biomedical Research at Lanzhou University Second Hospital, in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki).
From this dataset, we selected 40 participants, including 18 patients with major depressive disorder (MDD; 8 females, 10 males; mean age = 30.39 ± 10.91 years) and 22 healthy controls (HC; 8 females, 14 males; mean age = 30.86 ± 9.40 years). All participants were aged between 18 and 55 years and had at least a primary school education level. Only individuals with complete EEG recordings acquired using both a 128-channel high-density system and a 3-channel wearable system during resting-state, eyes-closed conditions were included.
Data collection
Each participant completed two EEG recording sessions under eyes-closed resting-state conditions: one using a 128-channel EEG cap and the other using a 3-channel wearable EEG device. The recordings were conducted at different times, and participants were instructed to remain still and relaxed during each 5-min session to minimize motion artifacts and external interference.
Inclusion and exclusion criteria
-
MDD Group: Patients were diagnosed with MDD based on the Mini-International Neuropsychiatric Interview (MINI) and had a PHQ-9 score ≥ 5.
-
Healthy Controls: Healthy participants had no personal or family history of mental disorders.
Exclusion criteria and experimental procedure
Participants were excluded if they had severe physical or psychiatric conditions, a history of substance abuse, or if they were pregnant or breastfeeding.
All EEG recordings were conducted in a quiet, dimly lit room free from electromagnetic interference. During the sessions, participants were seated comfortably and instructed to remain still with their eyes closed for approximately five minutes. They were asked to stay awake, avoid deliberate thinking, and minimize physical movements to reduce potential noise from electromyographic (EMG) and electrooculographic (EOG) artifacts.
Group characteristics
Statistical analyses were conducted to examine demographic and clinical differences between the MDD and healthy control groups. As shown in Table 1, no significant differences were found in age or sex between the two groups. However, participants with MDD exhibited significantly higher scores on both the PHQ-9 and GAD-7 compared to controls.
Ethics statement
The study was approved by the Ethics Committee of the Second Affiliated Hospital of Lanzhou University. Written informed consent was obtained from all participants.
Data acquisition
Resting-state EEG data were obtained using the HydroCel Geodesic Sensor Net (HCGSN) system (Electrical Geodesics Inc., Oregon, USA), with recordings conducted using a 128-channel EEG cap equipped with Ag/AgCl electrodes. The EEG signals were recorded at a sampling rate of 250 Hz, and the contact impedance between the electrodes and the skin was kept below 50 kΩ. For the three-electrode EEG experiment, electrodes were placed on the prefrontal lobe (Fp1, Fpz, and Fp2). The data were stored in a referential montage in TXT format, with each file representing an M × N array, where M is the number of electrodes and N is the number of sample points.
Preprocessing of EEG
Both the standard 128-channel system and the 3-channel portable device recorded EEG signals were analyzed offline using Matlab and EEGLAB. The sampling frequency was 250 Hz. Subsequent analyses focused on the frontal electrodes (Fp1, Fpz, and Fp2). A 1–50 Hz bandpass filter was used to eliminate slow drift, high frequency noise, and 50 Hz line interference. To reduce artifacts, we asked participants to remain still with their eyes closed, and all data were checked visually.
Subsequent analyses were limited to the 4–20 Hz range, including the theta (4–8 Hz), alpha (8–13 Hz), and low-beta (13–20 Hz) frequency bands, which are strongly associated with affective and cognitive processes.
We estimated the power spectrum of EEG signals using Welch’s method, which is based on fast Fourier transform (FFT). By increasing the window and overlap, the variance is reduced and the stability of the spectrum estimation is improved39.
For each electrode (Fp1, Fpz, Fp2), relative power was computed for each frequency bin (0.1 Hz resolution), defined as:
where Pf is the power at a specific frequency and Ptotal is the total power in the 4–20 Hz range. This relative normalization reduces interindividual variation caused by absolute amplitude differences and enhances the robustness of cross-subject comparisons consistent with methods established in previous biomedical and neuroscience studies37,40,41,42,43.
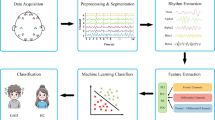
Finally, the average relative power across frontal electrodes (Fp1, Fpz, and Fp2) was extracted as a feature representing frontal EEG activity. This feature extraction approach has been widely applied in affective neuroscience and depression research to obtain reliable indicators of frontal activity44,45. The overall workflow of EEG preprocessing is illustrated in Fig. 1.
Psychological assessments
To evaluate participants’ emotional states and clinical characteristics, three standardized self-report scales were used:
PHQ-9 (Patient Health Questionnaire-9): A 9-item instrument for assessing depressive symptoms over the past two weeks. Scores range from 0 to 27. A score ≥ 5 is commonly used as a threshold for identifying possible clinical depression and was one of the criteria for MDD group inclusion.
GAD-7 (Generalized Anxiety Disorder-7): A 7-item scale for evaluating anxiety symptoms. Although not specific to depression, GAD-7 is often comorbid with depressive disorders and helps capture broader emotional dysregulation.
CTQ-SF (Childhood Trauma Questionnaire—Short Form): A 28-item retrospective measure evaluating five types of childhood adversity. Higher scores indicate more severe early-life trauma, a known risk factor for depression.
These scales complement each other by capturing both current mood symptoms (PHQ-9, GAD-7) and early-life risk factors (CTQ-SF), providing a multidimensional profile of participants’ psychological vulnerability and aiding in the understanding of EEG–emotion associations in depression.
Statistical analysis
The overall workflow is summarized in Fig. 1. For each participant, the average relative power at each 0.1 Hz frequency bin within 4–20 Hz from frontal electrodes (Fp1, Fpz, Fp2) was computed.
The reliability between the portable 3-channel and the standard 128-channel EEG systems was assessed using intraclass correlation coefficients (ICC(3,k)) and Pearson correlation analysis.
Gender effects were evaluated per frequency bin using the nonparametric Mann–Whitney U test, due to the relatively small sample size and the potential non-normal distribution of EEG relative power values. False discovery rate (FDR) correction was applied to account for multiple comparisons.
Relationships between frontal EEG relative power and psychological scale scores (PHQ-9, GAD-7, CTQ-SF) were examined using Pearson correlations, separately for male and female participants, with FDR correction applied in the 11–14 Hz high-alpha range.
To evaluate the clinical utility of high-alpha power, associations with diagnostic category (0 = HC, 1 = MDD) were tested using point-biserial correlation and receiver operating characteristic (ROC–AUC) analysis. Additional correlations with demographic variables (age and years of education) were computed to examine potential confounding effects.
Statistical significance was defined as FDR-corrected p < 0.05 where applicable.
Results
Validation of portable EEG device
To assess the consistency between the portable three-channel EEG device and the standard 128-channel laboratory system in frequency-domain measurements, we extracted the relative power from corresponding prefrontal channels (Fp1, Fp2, Fpz), averaged them, and compared the resulting frequency profiles across the 4–20 Hz range for each participant.
As shown in Fig. 2A, most participants exhibited highly similar frequency profiles across the two devices, with clearly identifiable alpha peaks (8–13 Hz) during the eyes-closed resting state.
Spectral comparison and gender-based spectral analysis between a portable three-channel EEG device and a 128-channel laboratory EEG system. (A) Frequency power distribution for each participant under both devices. The x-axis represents frequency (4–20 Hz), and the y-axis indicates relative spectral power. The red line depicts the power spectrum from the portable three-channel EEG device, while the blue line shows the spectrum from the 128-channel EEG system. (B) Group-averaged EEG spectral power for both devices. The red line represents the average spectrum of the portable device; the blue line, the average spectrum from the 128-channel device. Shaded areas denote ± 1 standard deviation (SD). High agreement was observed between devices across the 4–20 Hz band (Pearson r = 0.975, p < 0.0001). (C) Gender-based EEG spectral power recorded using the three-channel device. Red and blue lines represent mean spectra for male and female participants, respectively. Shaded areas show ± 1 SD for each group. Spectral distributions were highly consistent between genders (r = 0.933, p < 0.0001). Note for gender-based analysis, participants from both diagnostic groups (MDD and HC) were combined, as the primary aim of this figure was to illustrate device comparability and general sex-related effects rather than diagnostic subgroup differences.
Figure 2B presents the mean spectra and standard deviation intervals for all participants, and the two devices were highly similar in the overall shape and amplitude of frontal region frequencies. Quantitative analysis confirmed this high agreement: The Pearson correlation between the two instruments was r = 0.975, p < 0.0001, ICC(3,k) = 0.980. These results demonstrate that the portable three-channel EEG system reliably reproduces the frontal spectral profile measured by the standard 128-channel system.
To further explore the potential influence of gender on frontal EEG spectral profiles, participants were divided into male and female groups. The mean relative power spectra (± SD) were computed separately for the 3-channel device (Fig. 2C). Per-frequency comparisons (Mann–Whitney U, FDR-corrected) revealed no significant differences at any frequency bin, indicating minimal gender-related effects across 4–20 Hz, consistent with the high overall correlation (r = 0.933, p < 0.0001). These results indicate minimal gender-related effects across the 4–20 Hz range, although males tended to exhibit slightly higher alpha power, consistent with previous reports in young adults46.
Correlations between EEG activity and psychological scales
We next examined the relationship between frontal EEG activity during eyes-closed resting state and emotional states, analyzing both male and female groups separately.
Pearson correlations between frontal EEG relative power and psychological scale scores were computed per 0.1 Hz frequency bin. Multiple comparisons were corrected using the FDR method. After FDR correction, significant correlations were observed primarily in the 11–14 Hz range for PHQ-9 and GAD-7, while CTQ-SF showed no FDR-significant correlations. Gender differences were evident: female participants showed stronger and more sustained correlations across the full 11–14 Hz range, whereas male participants exhibited more localized associations, primarily centered around 12 Hz. These differences remained after FDR correction (Figs. 3A, 4A, 5A).
Correlation analysis between PHQ-9 scores and relative spectral power at the forehead. (A) Pearson correlation coefficients between PHQ-9 scores and relative spectral power across the 4–20 Hz frequency range. The x-axis represents frequency (Hz), and the y-axis represents the Pearson correlation coefficient (r). The red line denotes male participants, and the blue line denotes female participants. (B) Scatter plots of PHQ-9 scores versus relative spectral power at 11.9 Hz. Red dots represent male participants (r = 0.40, p = 0.011), and purple dots represent female participants (r = 0.33, p = 0.037).
Correlation analysis between GAD-7 scores and relative spectral power at the forehead. (A) Pearson correlation coefficients between GAD-7 scores and relative spectral power across the 4–20 Hz frequency band. The x-axis represents frequency (Hz), and the y-axis indicates the correlation coefficient (r). The red line represents male participants, and the blue line represents female participants. (B) Scatter plots of GAD-7 scores versus relative spectral power at 11.9 Hz. Red dots represent male participants (r = 0.37, p = 0.018), and purple dots represent female participants (r = 0.40, p = 0.011).
Correlation analysis between CTQ-SF scores and relative spectral power at the forehead. (A) Pearson correlation coefficients between CTQ-SF scores and relative spectral power across the 4–20 Hz frequency band. The x-axis represents frequency (Hz), and the y-axis indicates the correlation coefficient (r). The red line represents male participants, and the blue line represents female participants. (B) Scatter plots of CTQ-SF scores versus relative spectral power at 11.9 Hz. Red dots represent male participants (r = 0.32, p = 0.043), and purple dots represent female participants (r = 0.34, p = 0.030).
To illustrate this pattern, 11.9 Hz was selected as a representative (not peak) frequency based on two criteria: (1) both genders showed relatively strong and similar correlations at this point; (2) the selection avoids presenting only peak values and instead reflects a broader, shared trend.
Scatter plots at 11.9 Hz are presented in Figs. 3B, 4B, and 5B. At this representative frequency, the correlation between PHQ-9 scores and relative EEG power was r = 0.40 (p = 0.011) in males and r = 0.37 (p = 0.018) in females. Comparable gender differences were observed for GAD-7 and CTQ-SF scores.
Overall, the results suggest that prefrontal high-alpha activity during resting state is positively associated with emotional symptom severity, particularly among female participants, as confirmed by FDR-corrected significance in the 11–14 Hz range. While males also showed significant associations, these were more narrowly distributed in frequency.
Association with clinical diagnosis
To explore the potential of relative EEG power as a clinical biomarker, we examined the association between high alpha band activity during the eyes-closed resting state and diagnostic category (0 = healthy controls [HC], 1 = major depressive disorder [MDD]). As shown in Fig. 6, individuals diagnosed with MDD exhibited significantly higher prefrontal relative power in the high alpha range, with a representative correlation observed at 11.9 Hz (r = 0.32, p = 0.042). Consistently, ROC analysis at this frequency yielded an AUC of 0.652, suggesting that while high alpha activity carries some discriminatory value between HC and MDD, its standalone classification performance remains modest. These findings suggest that elevated high alpha activity during resting state may be indicative of greater vulnerability to depression, though additional markers would be required for robust clinical prediction.
Control for demographic variables
To rule out potential confounding effects of demographic variables, we examined correlations between 11.9 Hz relative power and age and years of education in both male and female participants. As shown in Fig. 7, no significant correlations were found in either group, suggesting that the EEG markers observed were more specifically related to emotional states rather than general demographic characteristics.
Relative prefrontal EEG Power as a function of Age and Years of Education. (A) Scatter plot of the correlation between 11.9 Hz relative energy and age. Red dots indicate males and blue dots indicate females. No significant associations were found for either sex. (B) Scatter plot of the correlation between 11.9 Hz relative energy and years of education. Red dots indicate males and blue dots indicate females. No significant associations were found for either sex.
Discussion
This study focused on exploring neurophysiological biomarkers of depressive states using a portable EEG device. As emotional health issues become increasingly prevalent due to fast-paced lifestyles and heightened societal pressure, depression has emerged as one of the most common mental disorders. Current diagnoses of depression largely rely on subjective assessments, underscoring the urgent need for objective and reliable biomarkers. EEG technology offers unique value in this context by capturing real-time brain activity. Portable EEG systems, in particular, hold great promise for scalable early screening and proactive detection of emotional disturbances in community or school settings.
Data quality and advantages of the portable EEG device
The results demonstrate that the three-channel portable EEG device closely replicated the spectral patterns obtained from the standard 128-channel laboratory system. In particular, the presence of clear alpha peaks (8–13 Hz) during the eyes-closed resting state is consistent with typical electrophysiological characteristics, indicating high signal quality and a favorable signal-to-noise ratio47,48. Previous studies have shown that the alpha band exhibits higher test–retest reliability compared to other frequency bands, regardless of whether eyes are open or closed49. Moreover, the closed-eyes state is less susceptible to oculomotor artifacts, resulting in lower noise levels50. These findings validate the measurement reliability of the portable device and support its use for emotional assessment in non-laboratory environments.
Compared to high-density lab-based systems, portable EEG devices offer advantages such as lower cost, ease of use, and scalability, making them particularly suitable for large-scale screening51. With continued advancements, these systems could be implemented in schools and community settings to support early identification and prevention of emotional disorders, contributing to broader public mental health initiatives. Portable EEG devices are being increasingly applied in mobile mental health contexts52, such as home use, community screening, and early intervention. Our findings offer initial support for their utility in identifying affective disorders.
High alpha activity as a biomarker for emotional disturbance
Our findings revealed significant positive correlations between relative power in the high alpha band (11–13 Hz) recorded over the prefrontal region during the eyes-closed resting state and scores on all three psychological scales (PHQ-9, GAD-7, CTQ-SF) in both male and female participants.
These results are consistent with previous studies that have reported similar associations between increased high-alpha activity over prefrontal areas and higher levels of depressive and anxiety symptoms53,54. Moreover, recent evidence has shown systematic differences in resting-state EEG measures between patients with major depressive disorder and healthy controls, further supporting the utility of spectral features as potential biomarkers55.
The high alpha band has been consistently associated with key cognitive and affective processes, including attentional selection56,57, self-referential thinking58,59,60, cognitive inhibition61, and emotional dysregulation62. Importantly, abnormalities in these functions are frequently reported in individuals with mood and anxiety disorders. For instance, patients with depression and anxiety often exhibit disrupted attentional control63, self-referential thinking64, impaired cognitive function65,66 and emotional dysregulation67,68. Consequently, high alpha bands are highly likely to be biomarkers of emotional dysregulation.
It should be noted, however, that EEG power in the high alpha range may be influenced not only by true oscillatory activity (periodic components) but also by the underlying aperiodic background (1/f) power. Recent studies have shown that separating periodic and aperiodic components can provide more precise insights into the neural mechanisms of depression, avoiding potential confounds from global spectral shifts69,70. Thus, the observed associations between prefrontal high alpha power and emotional symptoms may reflect a combination of genuine oscillatory changes and contributions from the non-oscillatory background. Future studies explicitly disentangling these components would help clarify the specific neural dynamics underlying high alpha as a biomarker.
Moreover, we explored the potential of high alpha activity during eyes-closed resting state to distinguish between individuals with major depressive disorder (MDD) and healthy controls (HC). The analysis revealed a moderate association (r = 0.32, p = 0.042), which was consistent with previous analyses71,72, indicating that prefrontal high alpha power during resting state may serve as an indicator of clinical status. While not sufficient as a standalone diagnostic tool, this EEG feature holds promise for early screening and risk identification. Importantly, we found no significant correlations between high alpha activity and demographic variables such as age or years of education, enhancing its specificity as an emotional biomarker.
In resource-limited settings, prefrontal high alpha activity captured by portable EEG devices may offer a practical and effective neural marker for assessing emotional risk.
Gender differences and frequency range extension
The observed gender differences in EEG–emotion associations are particularly noteworthy and warrant deeper attention. Our results showed that while both male and female participants exhibited significant correlations between frontal high alpha power and emotional scale scores, females demonstrated stronger and more broadly distributed associations. Specifically, in females, the correlations extended beyond the high alpha range (11–13 Hz) into the low beta range (13–14 Hz), suggesting that the neural coupling between emotional states and oscillatory activity may be more extensive in women.
This phenomenon may be attributed to the interaction of multiple factors. First, in emotion-related cognitive tasks, females demonstrate greater activation in the prefrontal cortex (PFC) and limbic regions than males, indicating higher neural engagement during emotional processing27,73,74,75. Second, fluctuations in female sex hormones—particularly estradiol and progesterone—have been shown to modulate the excitability of key emotional brain regions such as the amygdala and medial prefrontal cortex, thereby influencing the intensity and duration of emotional responses76,77,78,79. Third, social expectations regarding emotional expressivity tend to be more strongly imposed on women, and this long-term social conditioning may heighten their sensitivity and responsiveness to emotional cues80,81,82. Lastly, in terms of regulatory strategies, women are more likely to employ cognitively demanding approaches such as reappraisal and expressive suppression, whereas men often rely on more automatic, resource-efficient mechanisms74,83. Together, these physiological, cognitive, and sociocultural factors may help explain the heightened emotional reactivity and broader oscillatory-emotion coupling observed in women.
From a methodological perspective, these findings underscore the importance of considering gender as a critical moderating factor in EEG-based emotional assessment. Models trained on mixed-gender data may underperform or produce biased predictions if gender-specific patterns are not accounted for. Future research should prioritize gender-stratified analyses and develop sex-specific EEG emotion markers or adaptive algorithms that account for individual differences.
In applied settings, such as school or clinical screenings, understanding these gender-based patterns may enable tailored detection strategies, improving the sensitivity and specificity of early warning systems for emotional distress.
Practical implications
In the context of education, students are highly sensitive to mood disorders. However, their mental state is often overlooked due to limited resources and expressive challenges. Integrating portable EEG assessments into routine school activities (e.g., mass screenings per school year, pre/post-exam assessments) and using spectral relative power markers in the high alpha range could identify people at high risk for anxiety or depression for further action. This will provide invaluable quantitative support to school counsellor and psychology teachers to facilitate the implementation of preventive mental health services.
In clinical practice, depression and related mood disorders have the characteristics of high prevalence, high recurrence rate, high disability rate, and long asymptomatic period. Because of the limitations of self-report questionnaires in primary care Settings, a portable EEG device with high alpha biomarkers at the forehead location could provide a cost-effective, noninvasive, and objective pre-screening tool. These tools can be used in community health centers, school medical offices, and workplace health programs to rapidly identify at-risk individuals and initiate timely interventions to advance primary prevention systems for mental health on a larger scale.
In the commercial sector, EEG-based emotion recognition models can be embedded in wearable devices such as headbands, smart headsets, or VR systems. These apps can support daily mood self-monitoring, mindfulness training, and real-time emotional feedback, especially for high-priority populations such as adolescents, older adults, and people with chronic diseases.
In summary, the spectral characteristics of the high-alpha band captured by portable EEG devices show potential for early psychological risk detection, population-level mood assessment, and personalized interventions. These findings open new avenues for scalable, accessible, and data-supported mental health monitoring approaches84.
Limitations and considerations
High-alpha activity is not specific to major depressive disorder (MDD); it can also reflect non-pathological states such as fatigue, relaxation, or temporary attentional disengagement85. Therefore, we emphasize that elevated high-alpha power may indicate potential emotional risk and can serve as a preliminary warning signal for further assessment, but it should not be interpreted as definitive evidence of depression, particularly in non-clinical populations.
It should also be noted that high-alpha power may be influenced by both periodic (true oscillatory) and aperiodic (1/f) components, which could confound interpretation at the individual level. Furthermore, averaging across Fp1, Fpz, and Fp2 may mask potential lateralization effects, such as frontal alpha asymmetry, thereby limiting the resolution of spatial or hemisphere-specific insights.
Future studies could separate periodic and aperiodic components, examine lateralization metrics, and consider context-specific factors (e.g., fatigue, relaxation, attention) to refine individual-level predictions and enhance interpretive depth. This approach will improve the precision of individual-level assessments and increase the clinical and educational utility of portable EEG evaluations.
Data availability
The MODMA dataset used in this study is publicly available at the following URL: https://modma.lzu.edu.cn/data/index/ This dataset is described in the publication: Cai, H. et al. A multi-modal open dataset for mental-disorder analysis. Sci Data 9, 1–10 (2022). https://doi.org/10.1038/s41597-022-01211-x. Researchers interested in additional details or data access should contact the corresponding author.
References
Malhi, G. S. & Mann, J. J. Depression. The Lancet 392, 2299–2312 (2018).
Schuch, J. J. J., Roest, A. M., Nolen, W. A., Penninx, B. W. J. H. & de Jonge, P. Gender differences in major depressive disorder: Results from the Netherlands study of depression and anxiety. J. Affect Disord. 156, 156–163 (2014).
Kuehner, C. Gender differences in unipolar depression: an update of epidemiological findings and possible explanations. Acta Psychiatr. Scand. 108, 163–174 (2003).
Breslau, J. et al. Sex differences in recent first-onset depression in an epidemiological sample of adolescents. Transl. Psychiatry 7, e1139–e1139 (2017).
Sun, Q. et al. Role of estrogen in treatment of female depression. Aging 16, (2024).
Zucker, I. & Beery, A. K. Males still dominate animal studies. Nature 465, 690–690 (2010).
Fuchikami, M. et al. Optogenetic stimulation of infralimbic PFC reproduces ketamine’s rapid and sustained antidepressant actions. Proc. Natl. Acad. Sci. 112, 8106–8111 (2015).
Mir, F. R. & Rivarola, M. A. Sex Differences in Anxiety and Depression: What Can (and Cannot) Preclinical Studies Tell Us?. Sexes 3, 141–163 (2022).
Weinberger, A. H., McKee, S. A. & Mazure, C. M. Inclusion of women and gender-specific analyses in randomized clinical trials of treatments for depression. J. Womens Health 19, 1727–1732 (2010).
Zimmerman, M., McGlinchey, J. B., Chelminski, I. & Young, D. P. Diagnosing major depressive disorder V applying the DSM-IV exclusion criteria in clinical practice. J. Nerv. Ment. Dis. 194, 530–533 (2006).
Pan, J.-X. et al. Diagnosis of major depressive disorder based on changes in multiple plasma neurotransmitters: a targeted metabolomics study. Transl. Psychiatr. 8, 130 (2018).
Liu, X. et al. EEG-based major depressive disorder recognition by neural oscillation and asymmetry. Front Neurosci 18, (2024).
Stolz, L. A., Kohn, J. N., Smith, S. E., Benster, L. L. & Appelbaum, L. G. Predictive Biomarkers of Treatment Response in Major Depressive Disorder. Brain Sci 13, (2023).
Watts, D. et al. Predicting treatment response using EEG in major depressive disorder: A machine-learning meta-analysis. Transl Psychiatry 12, (2022).
Yun, S. Advances, challenges, and prospects of electroencephalography-based biomarkers for psychiatric disorders: a narrative review. J. Yeungnam Med. Sci. 41, 261–268 (2024).
Key, A. P., Thornton-Wells, T. A. & Smith, D. G. Electrophysiological biomarkers and age characterize phenotypic heterogeneity among individuals with major depressive disorder. Front Hum Neurosci 16, (2023).
Alagapan, S. et al. Cingulate dynamics track depression recovery with deep brain stimulation. Nature 622, 130–138 (2023).
van der Vinne, N., Vollebregt, M. A., van Putten, M. J. A. M. & Arns, M. Frontal alpha asymmetry as a diagnostic marker in depression: Fact or fiction? A Meta-Analysis. Neuroimage Clin. 16, 79–87 (2017).
Tsai, Y. C., Li, C. T. & Juan, C. H. A review of critical brain oscillations in depression and the efficacy of transcranial magnetic stimulation treatment. Front Psychiatry 14, (2023).
Arns, M. et al. EEG alpha asymmetry as a gender-specific predictor of outcome to acute treatment with different antidepressant medications in the randomized iSPOT-D study. Clin. Neurophysiol. 127, 509–519 (2016).
Allen, J. J. B., Urry, H. L. & Hitt, S. K. The stability of resting frontal electroencephalographic asymmetry in depression. Psychophysiology 41, 269–280 (2004).
Nofzinger, E. Towards a neurobiology of dysfunctional arousal in depression: the relationship between beta EEG power and regional cerebral glucose metabolism during NREM sleep. Psychiatry Res. Neuroimaging 98, 71–91 (2000).
Li, P., Yokoyama, M., Okamoto, D., Nakatani, H. & Yagi, T. Resting-state EEG features modulated by depressive state in healthy individuals: insights from theta PSD, theta-beta ratio, frontal-parietal PLV, and sLORETA. Front. Hum. Neurosci. 18, 1–12 (2024).
Spironelli, C. et al. Evidence of language-related left hypofrontality in major depression: An EEG beta band study. Sci. Rep. 10, 1–12 (2020).
Jang, K. I. et al. Increased beta power in the bereaved families of the Sewol ferry disaster: A paradoxical compensatory phenomenon? A two-channel electroencephalography study. Psychiatry Clin. Neurosci. 71, 759–768 (2017).
Lee, S. A., Kim, C. Y., Shim, M. & Lee, S. H. Gender differences in neural responses to perceptually invisible fearful face—an ERP study. Front Behav Neurosci 11, (2017).
Stevens, J. S. & Hamann, S. Sex differences in brain activation to emotional stimuli: A meta-analysis of neuroimaging studies. Neuropsychologia 50, 1578–1593 (2012).
Lungu, O., Potvin, S., Tikàsz, A. & Mendrek, A. Sex differences in effective fronto-limbic connectivity during negative emotion processing. Psychoneuroendocrinology 62, 180–188 (2015).
Aydın, S. & Akın, B. Machine learning classification of maladaptive rumination and cognitive distraction in terms of frequency specific complexity. Biomed Signal Process Control 77, (2022).
Aydın, S. Investigation of global brain dynamics depending on emotion regulation strategies indicated by graph theoretical brain network measures at system level. Cogn. Neurodyn. 17, 331–344 (2023).
Özçoban, M. A. & Tan, O. Electroencephalographic markers in Major Depressive Disorder: insights from absolute, relative power, and asymmetry analyses. Front Psychiatry 15, Preprint at https://doi.org/10.3389/fpsyt.2024.1480228 (2024)
Wei, L., Li, Y. & Yao, X. The different EEG activity during cognitive reappraisal task for fearful and sad stimuli. Chin. J. Biomed. Eng. 40, 44–52 (2021).
Ratti, E., Waninger, S., Berka, C., Ruffini, G. & Verma, A. Comparison of medical and consumer wireless EEG systems for use in clinical trials. Front. Hum. Neurosci. 11, 1–7 (2017).
Blanco-Ríos, M. A. et al. Real-time EEG-based emotion recognition for neurohumanities: perspectives from principal component analysis and tree-based algorithms. Front Hum Neurosci 18, (2024).
Yu, X., Li, Z., Zang, Z. & Liu, Y. Real-Time EEG-Based Emotion Recognition. Sensors 23, 1–12 (2023).
Lin, Y. et al. The neural oscillatory mechanism underlying human brain fingerprint recognition using a portable EEG acquisition device. Neuroimage 294, 120637 (2024).
Han, C. et al. Monitoring sleep quality through low α-band activity in the prefrontal cortex using a portable electroencephalogram device: Longitudinal study. J. Med. Internet Res. 27, e67188 (2025).
Cai, H. et al. A multi-modal open dataset for mental-disorder analysis. Sci. Data 9, 1–10 (2022).
Aydın, S. Comparison of power spectrum predictors in computing coherence functions for intracortical EEG signals. Ann. Biomed. Eng. 37, 192–200 (2009).
Wang, B. et al. Superimposed gratings induce diverse response patterns of gamma oscillations in primary visual cortex. Sci. Rep. 11, 1–20 (2021).
Han, C. et al. Enhancement of the neural response during 40 Hz auditory entrainment in closed-eye state in human prefrontal region. Cogn. Neurodyn. 17, 399–410 (2023).
Han, C., Shapley, R. & Xing, D. Gamma rhythms in the visual cortex: functions and mechanisms. Cogn. Neurodyn. 16, 745–756 (2022).
Han, C. et al. Neural mechanism of orientation selectivity for distinct gamma oscillations in cat V1. J. Vis. 20, 1116 (2020).
Davidson, R. J. Anterior electrophysiological asymmetries, emotion, and depression: Conceptual and methodological conundrums. Psychophysiology 35, 607–614 (1998).
Jernajczyk, W. et al. Alpha Wavelet Power as a Biomarker of Antidepressant Treatment Response in Bipolar Depression. Adv Exp Med Biol 968, (2017).
Han, C., Cheung, V. C. K. & Chan, R. H. M. Aging amplifies sex differences in low alpha and low beta EEG oscillations. Neuroimage 312, 121231 (2025).
Barry, R. J., Clarke, A. R., Johnstone, S. J., Magee, C. A. & Rushby, J. A. EEG differences between eyes-closed and eyes-open resting conditions. Clin. Neurophysiol. 118, 2765–2773 (2007).
Niedermeyer, E. Alpha rhythms as physiological and abnormal phenomena. Int. J. Psychophysiol. 26, 31–49 (1997).
Qin, H., Ding, L., Duan, W. & Lei, X. Test-retest reliability of EEG: A comparison across multiple resting-state and task-state experiments. Acta Psychol. Sin. 55, 1587–1596 (2023).
Ding, L., Duan, W., Wang, Y. & Lei, X. Test-retest reproducibility comparison in resting and the mental task states: A sensor and source-level EEG spectral analysis. Int. J. Psychophysiol. 173, 20–28 (2022).
Bhavnani, S. et al. The acceptability, feasibility, and utility of portable electroencephalography to study resting-state neurophysiology in rural communities. Front. Hum. Neurosci. 16, 1–13 (2022).
LaRocco, J., Le, M. D. & Paeng, D. G. A Systemic Review of Available Low-Cost EEG Headsets Used for Drowsiness Detection. Front Neuroinform 14, Preprint at https://doi.org/10.3389/fninf.2020.553352 (2020)
Zhao, X. et al. Distinguishing major depressive disorder from bipolar disorder using alpha-band activity in resting-state electroencephalogram. J. Affect Disord. 376, 333–340 (2025).
Wang, B. et al. Characterizing major depressive disorder (MDD) using alpha-band activity in resting-state electroencephalogram (EEG) combined with MATRICS consensus cognitive battery (MCCB). J. Affect Disord. 355, 254–264 (2024).
Kovacevic, N. et al. Differences in resting state and task-based EEG measures between patients with major depressive disorder and healthy controls. Clin. Neurophysiol. 173, 190–198 (2025).
Ji, J. L., Grafton, B. & MacLeod, C. Referential focus moderates depression-linked attentional avoidance of positive information. Behav. Res. Ther. 93, 47–54 (2017).
Klimesch, W. Alpha-band oscillations, attention, and controlled access to stored information. Trends Cogn. Sci. 16, 606–617 (2012).
Mu, Y. & Han, S. Neural oscillations involved in self-referential processing. Neuroimage 53, 757–768 (2010).
Knyazev, G. G. EEG correlates of self-referential processing. Front. Hum. Neurosci. 7, 1–14 (2013).
Wagner, G., Schachtzabel, C., Peikert, G. & Bär, K. J. The neural basis of the abnormal self-referential processing and its impact on cognitive control in depressed patients. Hum. Brain Mapp. 36, 2781–2794 (2015).
Finlayson-Short, L., Harrison, B. J. & Davey, C. Self-other referential neural processing in social anxiety disorder and major depressive disorder. Neuroimage Clin. 30, 102669 (2021).
Liu, P. & Tan, J. X. Y. Late positive potentials elicited by negative self-referential processing predict increases in social anxiety, but not depressive, symptoms from age 11 to age 12. Dev Psychopathol 1–11 (2025). https://doi.org/10.1017/S0954579424001548
Hsu, K. J. et al. Transdiagnostic mechanisms in depression and anxiety: The role of rumination and attentional control. J. Affect Disord. 188, 22–27 (2015).
Northoff, G. et al. Self-referential processing in our brain—A meta-analysis of imaging studies on the self. Neuroimage 31, 440–457 (2006).
Lyche, P., Jonassen, R., Stiles, T. C., Ulleberg, P. & Landrø, N. I. Cognitive control functions in unipolar major depression with and without co-morbid anxiety disorder. Front. Psychiatry 1, 1–9 (2010).
Kircanski, K. & Gotlib, I. H. Cognitive Aspects of Depression. 3, 301–313 (2012).
Aldao, A., Nolen-Hoeksema, S. & Schweizer, S. Emotion-regulation strategies across psychopathology: A meta-analytic review. Clin. Psychol. Rev. 30, 217–237 (2010).
Schäfer, J. Ö., Naumann, E., Holmes, E. A., Tuschen-Caffier, B. & Samson, A. C. Emotion regulation strategies in depressive and anxiety symptoms in youth: A meta-analytic review. J. Youth Adolesc 46, 261–276 (2017).
Li, J. et al. Individualized spectral features in first-episode and drug-naïve major depressive disorder: Insights from periodic and aperiodic electroencephalography analysis. Biol. Psychiat. Cogn. Neurosci. Neuroimaging 10, 574–586 (2025).
Zandbagleh, A., Sanei, S. & Azami, H. Implications of aperiodic and periodic EEG components in classification of major depressive disorder from source and electrode perspectives. Sensors 24, Preprint at https://doi.org/10.3390/s24186103 (2024).
Knott, V., Mahoney, C., Kennedy, S. & Evans, K. EEG power, frequency, asymmetry and coherence in male depression. Psychiatry Res. Neuroimaging 106, 123–140 (2001).
Jaworska, N., Blier, P., Fusee, W. & Knott, V. Alpha power, alpha asymmetry and anterior cingulate cortex activity in depressed males and females. J. Psychiatr. Res. 46, 1483–1491 (2012).
Whittle, S., Yücel, M., Yap, M. B. H. & Allen, N. B. Sex differences in the neural correlates of emotion: Evidence from neuroimaging. Biol. Psychol. 87, 319–333 (2011).
McRae, K., Ochsner, K. N., Mauss, I. B., Gabrieli, J. J. D. & Gross, J. J. Gender differences in emotion regulation: An fMRI study of cognitive reappraisal. Group Process. Intergroup Relat. 11, 143–162 (2008).
Domes, G. et al. The neural correlates of sex differences in emotional reactivity and emotion regulation. Hum. Brain. Mapp 31, 758–769 (2010).
Andreano, J. M. & Cahill, L. Menstrual cycle modulation of medial temporal activity evoked by negative emotion. Neuroimage 53, 1286–1293 (2010).
Pompili, A., Arnone, B., D’Amico, M., Federico, P. & Gasbarri, A. Evidence of estrogen modulation on memory processes for emotional content in healthy young women. Psychoneuroendocrinology 65, 94–101 (2016).
Toffoletto, S., Lanzenberger, R., Gingnell, M., Sundström-Poromaa, I. & Comasco, E. Emotional and cognitive functional imaging of estrogen and progesterone effects in the female human brain: A systematic review. Psychoneuroendocrinology 50, 28–52 (2014).
Dreher, J. C. et al. Menstrual cycle phase modulates reward-related neural function in women. Proc. Natl. Acad. Sci. U S A 104, 2465–2470 (2007).
Chaplin, T. M., Hong, K., Bergquist, K. & Sinha, R. Gender differences in response to emotional stress: An assessment across subjective, behavioral, and physiological domains and relations to alcohol craving. Alcohol Clin. Exp. Res. 32, 1242–1250 (2008).
Wiggert, N., Wilhelm, F. H., Derntl, B. & Blechert, J. Gender differences in experiential and facial reactivity to approval and disapproval during emotional social interactions. Front. Psychol. 6, 1–10 (2015).
Else-Quest, N. M., Higgins, A., Allison, C. & Morton, L. C. Gender differences in self-conscious emotional experience: A meta-analysis. Psychol. Bull. 138, 947–981 (2012).
Nolen-Hoeksema, S. & Aldao, A. Gender and age differences in emotion regulation strategies and their relationship to depressive symptoms. Pers. Individ. Dif. 51, 704–708 (2011).
Liu, Y., Sourina, O. & Nguyen, M. K. Real-time EEG-based human emotion recognition and visualization. In: Proceedings - 2010 International Conference on Cyberworlds, CW 262–269 (2010). https://doi.org/10.1109/CW.2010.37
Li, G. et al. The impact of mental fatigue on brain activity: A comparative study both in resting state and task state using EEG. BMC Neurosci. 21, 1–9 (2020).
Acknowledgements
This research was supported by data from the MODMA Dataset (Multi-modal Open Dataset for Mental-disorder Analysis), provided by the Key Laboratory of Wearable Computing at Lanzhou University. We thank the dataset creators for enabling this work.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
CH and XL conceived and designed the study. XL provided supervision and overall guidance throughout the research process. CH and ST performed the literature review and data analysis. ST was responsible for data preprocessing, figure generation, and manuscript writing. CH led the interpretation of results. CH and XL were responsible for manuscript review and editing. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tang, S., Han, C. & Li, X. High alpha oscillations in portable prefrontal EEG indicate gender-sensitive biomarkers for emotional disorders. Sci Rep 15, 39289 (2025). https://doi.org/10.1038/s41598-025-23112-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-23112-w