Abstract
Pharmacological interventions that treat core and co-occurring features of autism spectrum disorder (ASD) are a persistent unmet need. As such, use of cannabis to manage ASD features is common in the autistic community. Yet, few studies have examined the acute effects of cannabis on symptoms associated with ASD. Therefore, we measured changes in symptom ratings from before to after cannabis use in a sample of 111 self-identified autistic adults. Anonymized archival data sourced from the Strainprint® app were analyzed. A subset of tracked information that reflected changes in core and co-occurring symptoms associated with ASD (i.e., Sensory Sensitivity, Repetitive Behaviors, Mental Control, and Negative Affect) were used to assess the impacts of cannabis on symptom severity. Overall, symptom severity ratings were reduced by 73.09% from before to after cannabis use. More severe symptoms were associated with greater reductions in severity ratings after use. Higher doses predicted greater reductions in severity of Repetitive Behaviors, Mental Control, and Negative Affect but dose of cannabis used to manage all symptoms remained static across time. Results from this first empirical examination of the perceived acute effects of cannabis in autistic adults suggest that cannabis provides temporary relief from symptoms associated with ASD.
Similar content being viewed by others
Introduction
Autism Spectrum Disorder (ASD) is the ninth leading cause of neurological disability in worldwide1. Yet, targeted pharmacological interventions for ASD is a chronic and persistent unmet need2. The lack of targeted pharmacological interventions has been attributed to the difficulty in developing interventions for such a widely heterogenous patient population3. While ASD is a developmental brain disorder, it is clinically diagnosed through the observation of atypical behaviors in two broad domains: social functioning and restricted, repetitive behaviors. Each domain encompasses a wide range of behaviors resulting in a highly complex, heterogenous symptom profile that varies widely across autistic individuals. The social functioning domain includes atypical social interactions, challenges in social communication, and issues with the fostering of social relationships into social connections4. The restricted, repetitive behaviors domain is highly varied and includes actions beyond the expected repetitive movement patterns like limb flapping and stimming. Specifically, this domain also comprises insistence on sameness (i.e., need for routines), ritualistic behaviors (i.e., strong attachments to specific objects), atypical sensory reactivity (e.g., skin/touch sensitivity, sound sensitivity, sensory overload), and circumscribed interests4,5,6,7. Repetitive thoughts can also be categorized in the restricted, repetitive behaviors domain8,9 when associated with circumscribed interests10,11.
The diagnostic features of ASD are further complicated by the prevalence of co-occurring psychological disorders in this patient population. Approximately 40–50% of autistic individuals are diagnosed with four or more co-occurring conditions, all of which can be difficult to recognize due to atypical manifestations and symptomatology12. The most common co-occurring conditions are anxiety, attention-deficit/hyperactivity disorder (ADHD), obsessive-compulsive disorder (OCD), intellectual developmental disorders, and/or other developmental delays13,14. As such, most autistic individuals are prescribed more than three different medications to address features of their ASD diagnosis, co-occurring conditions, and/or the overlap between core and co-occurring features15. Importantly, among the medications that contribute to the high rates of polypharmacy (i.e., ≥ 3 medications given at one time to treat a single condition or disorder) in ASD, only two medications, aripiprazole and risperidone are approved by the FDA to “treat” ASD16,17. Both medications have known undesirable side effects including increased risk of gynecomastia (i.e., development of breast tissue in males), metabolic changes, and weight gain. Further, data suggest these pharmacological interventions have poor long-term effectiveness and negatively impact hormone regulation (i.e., changes to prolactin)18,19. Given the limitations of the FDA approved interventions for ASD, there has been increasing attention to alternative pharmacological interventions20.
Recent reports from caregivers indicate growing interest in cannabis as an unconventional therapeutic for their autistic children, provided there is a solid evidence base21. Additionally, reports that perturbations in endogenous cannabinoid signaling may contribute to ASD pathophysiology further highlight the need to assess the impact of cannabis use in this population22. However, there is a scarcity of observational, open-label, and randomized clinical trials on the safety, tolerability, and efficacy of cannabinoids, such as delta-9 tetrahydrocannabinol (THC) and cannabidiol (CBD), for treating symptoms associated with ASD. While the autism community awaits publication of results from ongoing clinical trials, data from autistic adults currently using cannabis are needed to provide critical information to enhance harm reduction strategies for individuals that have access to cannabis but lack access to research on potential effects of cannabis use on symptoms associated with ASD. Moreover, data from current cannabis users can uniquely inform future clinical trials evaluating the impact of cannabinoids on symptoms associated with ASD features.
Therefore, the primary objective of the present study was to examine changes in self-reported core and co-occurring features associated with ASD from before to after acute cannabis use in a large sample of autistic cannabis users. The second objective was to explore potential predictors of changes in symptom ratings, including gender, age, dose, cannabinoid content, and/or cannabis use sessions across time. The third objective was to examine potential changes in doses and baseline (pre-cannabis use) symptoms over time as a function of the repeated use of cannabis to manage these symptoms. The study also provides general knowledge on the use patterns (i.e., dosages, cannabinoid concentrations) and demographics of autistic adults using cannabis to expand the currently available data.
Methods
Procedure
Anonymous archival data were obtained from Strainprint® Technologies (Strainprint®), a medical cannabis real-world evidence technology platform with a journaling app that allows users to track changes in symptom severity before and after using different strains and doses of cannabis. In the initial set-up period, app users enter basic demographic information as well as their medical conditions and symptoms they use cannabis to manage. Prior to engaging in medical cannabis use, Strainprint® users can select the specific symptoms they are using cannabis to manage and rate their baseline severity from 1 (least severe) to 10 (most severe). Users are further prompted to indicate their method of administration (e.g., smoke, vape, edible), the strain of cannabis they are about to use as well as the producer/distributor of that strain by choosing from a selection of cannabis products sold in Canada. Lab verified cannabinoid content (% THC and CBD) is pre-populated within the app using data from cannabis distributors in Canada. If the users’ product is not prepopulated within the app, they can manually enter the strain name and cannabinoid content of the product they are about to use. After rating their initial symptom severity and identifying their cannabis product and method of administration, app users then indicate the dose (total number of puffs) used. After an onset period determined by the method of administration chosen (e.g., 20 min after smoking/vaping), the app sends a push notification to prompt users to re-rate the severity of their symptoms. However, individuals can re-rate their symptoms beyond the 20 min they receive the prompt.
For the present study, we obtained anonymous archival data from Strainprint® for users who self-identified as having ASD as a condition and who used the app to track one or more of the following symptoms: skin sensitivity, touch sensitivity, sound sensitivity, sensory overload, limb flapping, repetitive behavior, compulsive behavior, stimming, challenge concentrating, intrusive thoughts, anxiety, irritability. These specific symptoms were selected a priori by reviewing the complete list of symptoms in the Strainprint app and identifying those most closely related to the core and co-occurring features of ASD. The following questions were used in the Strainprint app to prompt users to rate the severity of each of these symptoms: “How severe is your skin sensitivity?”, “How bad is your touch sensitivity?”, “How sensitive are you to sound?”, “How bad is your sensory overload?”, “How bad is your limb flapping?”, “How bad is your repetitive behavior?”, “How bad is your compulsive behavior?”, “How bad is your stimming?”, “How bad is your concentration?”, “How intrusive are your thoughts?”, “How bad is your anxiety?”, “How bad is your irritability?” For each, the response scale ranges from 1 to 10 with 1 indicating low severity and 10 indicating the highest severity.
The data included anonymous ID codes, gender, age, symptoms, self-reported symptom severity before and after each tracked medical cannabis use session, cannabinoid content (% THC, % CBD), the method of obtaining the cannabinoid content data (i.e., lab-verified cannabis producer vs. user-generated), the method of administration, the dose for each cannabis use session, and time/cannabis use sessions (a count variable ranging from 1 to the total number of cannabis use sessions they tracked with the app, which reflects the number of times they have used cannabis to manage their symptoms over time).
Inclusion/exclusion criteria
Due to differences in the onset of different routes of administration of cannabis (e.g., oral vs. inhaled), we included only tracked sessions in which individuals indicated that they used an inhaled method of administration (e.g., smoking, vaping) which is the most common route of administration23. Further, only tracked sessions in which individuals re-rated their symptoms within 4 h of cannabis use were included, given that the acute effects of inhaled cannabis peak within 10–30 min of ingestion, and dissipate after 3–4 h24,25. Finally, due to concerns with the reliability and validity of self-reported cannabinoid content information, we only included sessions with lab-verified cannabinoid content data.
Sample
The sample comprised 111 adults (age range 19–70) who reported using cannabis for symptoms associated with ASD (47 women, 49 men, 14 other, 1 undisclosed). This sample collectively used the Strainprint® app 5932 times over a span of 74 months (July 2017 to August 2023) to track symptoms of Sensitivity, Repetitive Behaviors, Mental Control, and Negative Affect. Table 1 displays demographic characteristics (age, gender) and information on the total number of tracked cannabis use sessions for the total sample, as well as for sub-samples broken down by symptom cluster. The Washington State University Office of Research Assurances determined that the use of anonymous archival study was exempt from the need for review by the Institutional Review Board (IRB). All research was performed in accordance with the Declaration of Helsinki. Additionally, the end user license agreement of the Strainprint® app includes informed consent for the collection and use of anonymous user data.
Data analysis
To streamline the analyses, the 12 symptoms were clustered into the following four domains: Sensitivity (skin sensitivity, touch sensitivity, sound sensitivity, and sensory overload), Repetitive Behaviors (limb flapping, stimming, repetitive behavior, compulsive behavior), Mental Control (challenge concentrating, intrusive thoughts), and Negative Affect (anxiety and irritability).
The percentage of cannabis use sessions involving a reduction in ASD symptom severity, an exacerbation in severity, and no change in severity ratings following cannabis use were computed for each symptom cluster.
To examine change in symptom severity from before to after cannabis use, two-time points latent change score (LCS) models were used. This approach allowed us to examine changes in ASD symptoms within subjects across time26 and as a function of specific predictors of interest. One series of LCS models were tested using time/cannabis use session and age as predictors and a separate series of LCS models were conducted using time/cannabis use session, dose, and cannabinoid content (% THC and CBD) as predictors. A detailed description of this approach is provided in Cuttler et al.27. All LCS models were fit using Mplus version 8.328.
Separate repeated measures multilevel models were used to examine changes in baseline symptom severity ratings and dose of cannabis used as a function of time/cannabis use sessions. Cannabis use session was centered at Time 1 so that the intercept (Time 0) represented the first session of cannabis use in each model. The fixed and random linear effects of time/cannabis use session on baseline severity and dose were estimated using SAS Proc Mixed, with maximum likelihood estimation and incomplete data were treated using missing at random assumptions. Unspecified covariance matrices were used in all multilevel models. In instances where models failed to converge using an unspecified covariance matrix, an autoregressive covariance matrix was used. This occurred for unconditional growth models predicting change in starting symptom severity for the Repetitive Behaviors and Sensitivity symptom clusters and change in dose over time for the Repetitive Behaviors symptom cluster. All other models met convergence criteria using the standard unspecified covariance matrix.
Given the size of the dataset and number of analyses a Bonferroni corrected alpha of 0.013 (calculation: 0.05/4 symptom clusters) was used to determine statistical significance and reduce family-wise Type I error rate.
Results
Cannabis use characteristics
Table 2 displays cannabis use characteristics for the entire sample, and for each symptom cluster. Specifically, the table displays descriptive statistics pertaining to dose of cannabis used (# of puffs), as well as THC and CBD concentrations (measured in %) in the cannabis used to manage each symptom cluster. A small number (n = 88, 1.48% of total sessions) of lab-verified tracked sessions measured THC and CBD concentrations in mg/dose, mg/inhalation, or mg/ml, or did not list the measurement scale. These sessions were excluded from analysis of mean THC/CBD content listed in Table 2, as well as latent change score analyses where the influence of dose, %THC, and %CBD were included as predictors of change in ASD symptom severity.
Overall change in symptom severity ratings
ASD symptoms were reported as being reduced (as opposed to exacerbated or unchanged) in the vast majority of tracked sessions (98.33%). With respect to each symptom cluster, Sensitivity symptom ratings were reduced in 97.26% of tracked sessions, ratings of Repetitive Behaviors were reduced in 99.71% of tracked sessions, Mental Control symptom ratings were reduced in 97.68% of sessions, and Negative Affect symptom ratings were reduced in 98.40% of sessions.
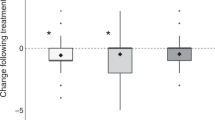
As further depicted in Fig. 1, mean Sensitivity severity ratings were significantly reduced from 7.27 (SE = 0.12) to 2.32 (SE = 0.11) (p < .001) which represents a decrease of 68.09% from before to after cannabis use. Similarly, ratings of Repetitive Behavior severity were significantly reduced from 6.86 (SE = 0.03) to 2.03 (SE = 0.04) from before to after use (p < .001) (representing a 70.41% reduction). Ratings of the severity of problems with Mental Control were significantly reduced from 6.75 (SE = 0.03) to 2.12 (SE = 0.04) (p < .001) which indicates a 68.59% reduction, and ratings of the severity of symptoms of Negative Affect were significantly reduced from 7.02 (SE = 0.03) before cannabis use to 1.70 (SE = 0.03) after cannabis use (p < .001) representing a 75.78% reduction in severity of this symptom cluster. Collapsed across all three symptom domains, symptom severity ratings were reduced from a mean of 6.95 (SE = 0.02) before cannabis use to 1.87 (SE = 0.02) after cannabis use which represents an overall reduction in all symptom ratings combined of 73.09%.
Baseline unconditional LCS models predicting change in symptom severity
The fixed effects of baseline LCS models indicated that ratings of all symptom clusters were significantly reduced from before to after cannabis use (Sensitivity: µΔ = -4.96, SE = 0.53, p < .001; Repetitive Behaviors: µΔ = -4.82, SE = 0.44, p < .001; Mental Control: µΔ= -4.63, SE = 0.37 p < .001; Negative Affect: µΔ = -5.32, SE = 0.32, p < .001).
The covariance between symptom severity ratings before cannabis use and the latent change factor was statistically significant and negative for all four of the symptom clusters (Sensitivity: covariance estimate = -3.17, SE = 0.39, p < .001; Repetitive Behaviors: covariance estimate = -0.93, SE = 0.32, p = .003; Mental Control: covariance estimate = -1.35, SE = 0.43, p = .002; Negative Affect: covariance estimate = -2.82, SE = 0.62, p < .001). These results indicate that more severe symptoms prior to cannabis use were associated with greater reductions in symptom severity ratings following use.
The variance (i.e., random effects tested) of the latent change factors were also statistically significant for all four symptom clusters (Sensitivity: variance estimate = 5.55, SE = 0.60, p < .001; Repetitive Behaviors: variance estimate = 2.41, SE = 0.59, p < .001; Mental Control: variance estimate = 3.37, SE = 1.06, p = .001; Negative Affect: variance estimate = 5.04, SE = 1.11, p < .001). This suggests that there were significant differences across individuals in the magnitude of change in ratings of each symptom cluster from before to after cannabis use.
Conditional LCS models with predictors of change
Two sets of conditional LCS Models were run to assess the influence of predictors of interest on change in ASD symptom severity. Models including time/cannabis use session, gender, and age were run with the total sample for each symptom cluster. Separate models including time/cannabis use session, dose, %THC, and %CBD were run using sessions where THC and CBD were measured in percent (%). Once again, sessions with cannabinoid content measured in mg/ml, mg/dose, or mg/inhalation constituted less than 1.5% of the total number of observations and were excluded from analyses where %THC, %CBD and dose were included as predictors. As shown in Table 3, conditional LCS models revealed that time/cannabis use session, and age were significant predictors of symptom change for the Sensitivity symptom cluster. More specifically, Sensitivity symptom relief lessened significantly over time as a function of using cannabis to self-medicate for this symptom cluster, and older individuals reported greater relief that younger individuals. In contrast as shown in Table 4, higher doses of cannabis predicted greater reductions in severity of Repetitive Behaviors, Mental Control, and Negative Affect. Time/cannabis use session, age, gender, %THC, and %CBD were not significant predictors of symptom change for symptoms of Negative Affect, Repetitive Behaviors, or Mental Control.
MLM predicting changes across cannabis use sessions
Changes in dose across time. Four separate multilevel models revealed that the dose of cannabis used to manage all four symptom clusters did not increase significantly across cannabis use sessions over time (Sensitivity: β = 0.005, SE = 0.07, p = .94; Repetitive Behavior: β = 0.15, SE = 2.00, p = .94; Mental Control: β = 0.01, SE = 0.01, p = .15; Negative Affect: β = 0.03, SE = 0.02, p = .21).
Change in baseline symptoms. An additional set of four multilevel models predicting changes in baseline symptoms as a function of cannabis use sessions across time revealed that severity of baseline ratings did not increase significantly across time for Sensitivity (β = − 0.18, SE = 0.21, p = .41) or Mental Control (β = 0.003, SE = 0.002, p = .10). In contrast, baseline severity ratings of Repetitive Behaviors (β = 0.004, SE = 0.0003, p < .001) and Negative Affect (β = 0.006, SE = 0.001, p < .001) increased significantly across cannabis use sessions over time.
Discussion
Globally, cannabis is one of the most widely used psychoactive substances, which parallels recent data that ASD is the ninth leading cause of neurological disability worldwide1,29,30. Nevertheless, there is a conspicuous lack of data on the acute effects of cannabis on core and co-occurring features ASD. This issue is complicated by a historical abundance of data on the potential negative effects of cannabis use in allistic people and recent reports of increased rates of substance use disorder in autistic people (i.e., up to 36% of cases)31,32,33. As such, a fundamental understanding of the outcomes of cannabis use in autistic people has been persistently absent from the research landscape. Therefore, this study represents the first to investigate the perceived acute effects of cannabis on symptoms associated with ASD using an innovative methodology that allowed us to bypass federal restrictions governing the acute administration of cannabis to humans. We evaluated whether age, dose, cannabinoid content of cannabis used, and/or number of cannabis use sessions across time predict changes in self-identified autistic adult’s symptom severity ratings from before to after cannabis use.
Using a large dataset from self-identified autistic adults self-medicating with cannabis, we found that ratings of the severity of Negative Affect were reduced by 75.78%, ratings of the severity of Repetitive Behaviors were reduced by 70.41%, ratings of the severity of problems with Mental Control were reduced by 68.59%, and ratings of Sensitivity severity were reduced by 68.09%, from immediately before to shortly after inhaling cannabis. Moreover, these symptoms were reduced (rather than exacerbated or unchanged) in 98.33% of the cannabis use sessions. These findings are among the first to indicate that cannabis is perceived to improve highly prevalent symptoms associated with ASD in adults using cannabis for symptom management.
While inhaled cannabis resulted in significant and substantial reductions in ratings of all four symptom clusters, acute cannabis use was perceived to be most effective for individuals with the highest baseline severity ratings and there was significant heterogeneity in the magnitude of symptom reductions across individuals. This indicates that not all people reported symptom reductions and different people reported different magnitudes of reductions in the severity of symptoms from before to after use. Future research is needed to examine individual difference variables that contribute to the perceived impacts of cannabis on ASD symptoms (e.g., prior experience using cannabis).
The first series of LCS models revealed that age was a significant predictor of changes in self-reported symptoms of Sensitivity, with increased age associated with larger perceived reductions in Sensitivity ratings after cannabis use. These findings are somewhat consistent with reports that older adults experience fewer detrimental effects from acute cannabis use34 and that sensory symptoms are most severe in middle adulthood. The LCS models also indicated that time/cannabis use session significantly predicted the magnitude of symptom change for the Sensitivity cluster. The negative coefficient for this variable indicates that cannabis may become less effective for reducing Sensitivity symptoms as a function of the repeated use of cannabis to self-medicate for this symptom cluster over time. In contrast, the efficacy of cannabis in reducing the severity of the remaining three symptom clusters remained static across cannabis use sessions over time.
The second series of LCS models indicated that dose of cannabis used (# puffs) was a robust predictor of changes in Repetitive Behaviors, Mental Control, and Negative Affect, with higher doses predicting greater reductions in all three symptom clusters. Despite experiencing more relief from larger doses, the MLM models indicate that individuals are reporting using consistent doses across time. Collectively, these results demonstrating consistency in perceived efficacy and dose over time suggest that tolerance to the effects of inhaled cannabis on Repetitive Behaviors, problems with Mental Control, and Negative Affect may not develop in autistic adults using cannabis to manage their symptoms.
Finally, cannabinoid concentrations (% THC and CBD) did not have a significant influence on changes in any of the four symptom clusters assessed. These results contrast with the design and preliminary data of most ongoing clinical trials which are geared towards assessing the therapeutic tolerability, safety, and impact of mainly CBD products35. In human studies, the underlying rationale to investigate products that have a high ratio of CBD to THC is to leverage the neuroprotective properties of CBD36,37,38,39,40 while decreasing the potential negative effects of THC (e.g., detrimental effects on cognition, psychosis, and brain development; for a detailed review see 40).
Nevertheless, our findings of perceived reductions in symptoms associated with ASD in autistic adults using cannabis align with several recent clinical trials including an open-label study with 60 autistic children orally administered a whole plant extract with a 1:20 THC: CBD ratio (resulting in a mean dose of 3.8 ± 2.6 mg/kg/day CBD and 0.29 ± 0.22 mg/kg/day THC) that revealed improvements in aggressive behaviors, anxiety, and communication41. Another study evaluated 188 pediatric autistic participants (mean age of 12.9) administered a 1:30 THC: CBD cannabis product (for a mean dose of 79.5 ± 61.5 mg CBD and 4.0 ± 3.0 mg THC) sublingually three times a day42. Parents reported that their children showed improvements in positive mood, independent hygienic care (i.e., ability to dress and shower), sleep, and concentration that persisted for up to six months after treatment. In a third study, 53 autistic children (mean age of 11) administered a cannabinoid oil solution with a 1:20 THC: CBD ratio (resulting in a maximal dose of 600 mg CBD and 40 mg of THC) showed improvements in sleep, self-injurious behaviors, hyperactivity, disturbances, and anxiety43. Further, in a nine-month uncontrolled observational study of 18 pediatric autistic participants using a 1:75 THC: CBD cannabis extract taken orally in capsules (resulting in an average dose of CBD was 4.6 mg/kg/day and 0.06 mg/kg/day of THC), parents reported improvements in sleep, seizures, and behavioral crises (i.e., binge eating, excessive fear reactions, pseudobulbar affect responses like uncontrollable crying or inappropriate laughter)44. Finally, international researchers evaluated the safety, tolerability, and efficacy of cannabinoid interventions in children and adolescents using a double blind, randomized, placebo-controlled crossover trial using a 20:1 CBD to THC whole plant extract as well as a purified CBD and THC intervention at the same ratio. Cannabinoid interventions were taken sublingually, three times a day with an average dose of whole plant extracts of 5.7 ± 2.6 mg/kg/day CBD and a 5.9 ± 2.7 mg/kg/day CBD dose for the pure cannabinoids. Total scores were unchanged on the Home Situations Questionnaire-Autism Spectrum Disorder between treatments, but the whole plant extract intervention demonstrated a significant improvement compared placebo on the Clinical Global Improvement scale45. Additionally, whole plant extracts improved scores on the social responsiveness scale, a gold-standard assessment for evaluating autistic social behaviors45. In sum, data from clinical trials of children and adolescents presents an evidence base that supports a continued focus on the impact of CBD on ASD features, while the present data from cannabis-using autistic adults indicates that a sole focus on CBD may not fully capture the potential impact of cannabinoids as a pharmacological intervention for adults with ASD. Thus, additional placebo-controlled clinical trials are needed where THC, CBD, and other non-intoxicating cannabinoids (e.g., cannabigerol), terpenes, and/or medications that modulate the functioning of the endocannabinoid system are administered to autistic adults to determine their relative effects on symptoms associated with ASD.
These generally promising results are tempered by our findings that baseline severity ratings of Repetitive Behaviors and Negative Affect significantly increased as a function of the repeated use of cannabis over time. This suggests that regular use of cannabis to self-medicate for Repetitive Behaviors and Negative Affect may intensify these symptoms in the long-term and is consistent with previous findings that depression scores are exacerbated by the repeated use of cannabis to self-medicate for depression46. Thus, while acute use of cannabis may provide a temporary reprieve from Repetitive Behaviors and Negative affect without the development of tolerance to these effects; regular cannabis use may intensify these symptom clusters in the long-term. Alternatively, given the non-experimental nature of the study it is possible that our findings that baseline symptoms of Repetitive Behaviors and Negative Affect increased as a function of cannabis use over time reflects changes in people’s threshold for tolerating these symptoms over time and tendencies to begin to self-medicate with cannabis at higher levels of these symptom clusters. Future controlled longitudinal research is needed to tease apart these alternative explanations.
Limitations include the sample size, sampling bias, lack of assessment of adverse events, lack of clinician-verified diagnoses of ASD, a lack of representation or underrepresentation of some core features of ASD (e.g., deficits in social communication and interaction), and the non-experimental nature of the study.
The sample was large enough to afford ample power to detect effects but was somewhat small relative to samples with other conditions we have examined using Strainprint® app data over shorter timeframes (e.g., Anxiety, n = 770 46; Depression, n = 561 46; PTSD, n = 404 46; pain, n = 3548 46), suggesting adults with autism may be less likely to self-medicate with cannabis relative to adults with other conditions. Further, most people in the sample (> 50%) only used the app 1–2 times suggesting the majority may not be heavy cannabis users and/or may not rely heavily on cannabis to manage their symptoms. Nevertheless, it is still important to acknowledge that the sample may overrepresent individuals who tolerate cannabis well and find it effective in managing their symptoms, as those who don’t find it effective would be unlikely to continue using cannabis and the app to track such use. As such, our sample likely underrepresents individuals who find cannabis to be aversive or ineffective at reducing their symptoms. Moreover, a minority of people reported symptom exacerbation and individual differences in the efficacy of cannabis in reducing symptoms were detected. As such, it is likely that a sub-set of individuals with ASD would not find cannabis beneficial for reducing their symptoms. Future more controlled research using cannabis-naïve autistic adults is needed to better understand the relative percentages of autistic adults who find cannabis beneficial versus detrimental for their symptoms.
We also did not obtain data on adverse events and as such it is unclear how many people experienced adverse events, what those adverse events may have been, and which factors (e.g., dose, gender, age) best predicted adverse events. Future clinical trials are needed to better understand the potential for adverse events in autistic adults using cannabis for symptom management and how best to mitigate them.
Unfortunately, we could not verify ASD diagnoses, not all clinically recognized symptoms associated with ASD were assessed by the app, and some of the outcomes were rated by a small number of people who tracked a low number of sessions. More specifically, people in this study were self-diagnosed as autistic and as such some may not have a formal, clinical diagnosis of ASD. However, ASD self-diagnosis has been shown to accurately predict a formal, clinical diagnosis in approximately 80% of cases47, providing reasonable support that our sample of self-diagnosed autistic adults likely predominantly represents those with a clinical diagnosis. Our use of a self-identified autistic sample may explain the fairly equal balance of autistic men and women in our sample despite the 3:1 male to female gender ratio in clinical ASD diagnoses. Due to inadequate diagnostic tools and increased camouflaging/masking, autistic women are diagnosed less frequently than men (with up to 80% of autistic adult women remaining undiagnosed by age 18)48. As a result of these increased barriers, many autistic women resort to self-diagnosis making it highly probable that a study utilizing self-diagnosed adults would result in a great inclusion and proportion of autistic women48,49,50.
Some outcomes, particularly self-regulatory behaviors (i.e., stimming), were rated by a small number of people (n = 4) over a small number of sessions (n = 7), perhaps because they were unfamiliar with the term or because they don’t view stimming as a negative symptom and were therefore disinclined to use cannabis to reduce the behavior or provide ratings for the statement “How bad is your stimming?” Indeed, reports from the autistic community suggest that the use of self-stimulatory behaviors is an adaptive and compensatory mechanism to assists in increasing calm comfort, while reducing the impact of internal or environmental factors like overpowering emotion or intense sensory environments51,52. Moreover, we were unable to assess the impact of cannabis use on social behavior and communication as the Strainprint® app does not include symptoms pertaining to this hallmark feature of ASD. More generally, use of archival data limited our ability to directly assess core diagnostic symptom features in ASD. Although many of the tracked symptoms do not represent pure diagnostic features in ASD, they are inclusive of many of the features found in diagnostic inventories as well as symptoms that are observed at a high prevalence within the autistic community. For example, although irritability is not a core diagnostic symptom of ASD, its prevalence in ASD is between 19 and 80% 53. Moreover, the only FDA-approved pharmacological interventions for ASD specifically target irritability rather than the core diagnostic symptoms. Similarly, in autistic adults, anxiety is one of the most prevalent co-occurring features in ASD, with 20–50% of autistic adults experiencing anxiety54.
It was also not possible to experimentally manipulate cannabis use or obtain a placebo control group. As such, we cannot make causal conclusions, and expectancy effects are likely contributing to some of the reductions in symptom ratings. The lack of experimental manipulation and the presence of individual differences in the number of tracked sessions also complicate interpretations of changes in baseline symptoms across time.
These limitations are offset by very high ecologically validity as the data were obtained from over 100 individuals spanning a wide range of ages (19–70) who were legally using a broad array of market cannabis products in their natural environments. The ability to track changes in symptoms across a large number of cannabis sessions (> 5,900) over a 74-month time-period are additional strengths of the study.
The present study represents the first to demonstrate acute perceived beneficial effects of inhaled cannabis on core and co-occurring symptoms associated with ASD in a large adult sample. While all symptom clusters were reduced by a 68–75% in the short-term, there was evidence that the repeated use of cannabis to self-medicate may exacerbate Repetitive Behaviors and Negative Affect over time. Collectively these results indicate that people using cannabis to manage symptoms associated with ASD may experience short-term, but not long-term, reductions in their symptoms. These findings indicate that well-powered placebo-controlled trials are warranted to examine the acute effects of various cannabinoids and manipulations of the endocannabinoid system on ASD symptoms. Moreover, they can be used to guide clinical practice and contextualization of results from ongoing clinical trials.
Patient and public involvement
Community members were not involved in the development or design of the research question, measures, implementation, or interpretation and dissemination of the findings. The research study was led by a neurodiverse team and used inclusive data collection practices, such as the self-diagnosis inclusion criteria.
Data availability
Data are available upon request to [carrie.cuttler@wsu.edu] (mailto: carrie.cuttler@wsu.edu).
References
Steinmetz, J. D. et al. Global, regional, and national burden of disorders affecting the nervous system, 1990–2021: A systematic analysis for the global burden of disease study 2021. Lancet Neurol. 23, 344–381 (2024).
Maniram, J., Karrim, S. B. S., Oosthuizen, F. & Wiafe, E. Pharmacological management of core symptoms and comorbidities of autism spectrum disorder in children and adolescents: A systematic review. Neuropsychiatr Dis. Treat. 18, 1629–1644 (2022).
Pérez-Cano, L. et al. Translating precision medicine for autism spectrum disorder: A pressing need. Drug Discov Today. 28, 103486 (2023).
CDC. Diagnostic criteria | autism spectrum disorder (ASD) | NCBDDD | CDC. Centers Disease Control Prevention (2020). https://www.cdc.gov/ncbddd/autism/hcp-dsm.html
Chen, Y. H., Rodgers, J. & McConachie, H. Restricted and repetitive behaviours, sensory processing and cognitive style in children with autism spectrum disorders. J. Autism Dev. Disord. 39, 635–642 (2009).
Leekam, S. R., Prior, M. R. & Uljarevic, M. Restricted and repetitive behaviors in autism spectrum disorders: A review of research in the last decade. Psychol. Bull. 137, 562–593 (2011).
Tian, J., Gao, X. & Yang, L. Repetitive restricted behaviors in autism spectrum disorder: from mechanism to development of therapeutics. Front Neurosci 16, 780407 (2022).
Thom, R. P. & McDougle, C. J. Repetitive thoughts and behaviors in autism spectrum disorder: A symptom-based framework for novel therapeutics. ACS Chem. Neurosci. 14, 1007–1016 (2023).
McDougle, C. J. et al. A case-controlled study of repetitive thoughts and behavior in adults with autistic disorder and obsessive-compulsive disorder. Am. J. Psychiatry. 152, 772–777 (1995).
Hemmers, J., Baethge, C., Vogeley, K. & Falter-Wagner, C. M. Are executive dysfunctions relevant for the autism-specific cognitive profile? Front. Psychiatry. 13, 886588 (2022).
Ozonoff, S., Pennington, B. F. & Rogers, S. J. Executive function deficits in high-functioning autistic individuals: Relationship to theory of Mind. J. Child. Psychol. Psychiatry. 32, 1081–1105 (1991).
Khachadourian, V. et al. Comorbidities in autism spectrum disorder and their etiologies. Transl. Psychiatry. 13, 1–7 (2023).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5 (5th Ed.). xliv, 947. (American Psychiatric Publishing Inc., Arlington, 2013). https://doi.org/10.1176/appi.books.9780890425596
Frazier, T. W. et al. Validation of proposed DSM-5 criteria for autism spectrum disorder. J. Am. Acad. Child. Adolesc. Psychiatry. 51, 28–40e3 (2012).
Feroe, A. G. et al. Medication use in the management of comorbidities among individuals with autism spectrum disorder from a large nationwide insurance database. JAMA Pediatr. 175, 957–965 (2021).
Aishworiya, R., Valica, T., Hagerman, R. & Restrepo, B. An update on psychopharmacological treatment of autism spectrum disorder. Neurotherapeutics 19, 248–262 (2022).
Ritter, C., Hewitt, K. & McMorris, C. A. Psychotropic polypharmacy among children and youth with autism: A systematic review. J. Child. Adolesc. Psychopharmacol. 31, 244–258 (2021).
Findling, R. L. et al. Prolactin levels during long-term risperidone treatment in children and adolescents. J. Clin. Psychiatry. 64, 1362–1369 (2003).
Roke, Y., Buitelaar, J. K., Boot, A. M., Tenback, D. & van Harten, P. N. Risk of hyperprolactinemia and sexual side effects in males 10–20 years old diagnosed with autism spectrum disorders or disruptive behavior disorder and treated with risperidone. J. Child. Adolesc. Psychopharmacol. 22, 432–439 (2012).
Fletcher, S., Pawliuk, C., Ip, A., Huh, L., Rassekh, S. R., Oberlander, T. F., & Siden, H. Medicinal Cannabis in Children and adolescents with autism spectrum disorder: A scoping review. Child Care Health Dev. 48, 33–44 (2022). https://doi.org/10.1111/cch.12909
Sivori, T. T., Capron, M., Graves, D., Harris, J. & Schaaf, R. C. Caregivers’ views on cannabis use for their children with autism. Res. Autism Spectr. Disord. 102, 102130 (2023).
Karhson, D. S. et al. Plasma anandamide concentrations are lower in children with autism spectrum disorder. Mol. Autism. 9, 18 (2018).
Sexton, M., Cuttler, C., Finnell, J. S. & Mischley, L. K. A cross-sectional survey of medical cannabis users: Patterns of use and perceived efficacy. Cannabis Cannabinoid Res. 1, 131–138 (2016).
Grotenhermen, F. Pharmacokinetics and pharmacodynamics of cannabinoids. Clin. Pharmacokinet. 42, 327–360 (2003).
Menkes, D. B., Howard, R. C., Spears, G. F. & Cairns, E. R. Salivary THC following cannabis smoking correlates with subjective intoxication and heart rate. Psychopharmacol. (Berl.). 103, 277–279 (1991).
McArdle, J. J. Latent variable modeling of differences and changes with longitudinal data. Annu. Rev. Psychol. 60, 577–605 (2009).
Cuttler, C., Spradlin, A., Cleveland, M. J. & Craft, R. M. Short- and Long-Term effects of cannabis on headache and migraine. J. Pain. 21, 722–730 (2020).
Muthén, B. O., Muthén, L. K. & Asparouhov, T. Regression and Mediation Analysis Using Mplus (Muthén & Muthén, 2017).
Talantseva, O. I. et al. The global prevalence of autism spectrum disorder: A three-level meta-analysis. Front Psychiatry 14, 1071181 (2023).
Zeidan, J. et al. Global prevalence of autism: A systematic review update. Autism Res. 15, 778–790 (2022).
Arnevik, E. A. & Helverschou, S. B. Autism spectrum disorder and Co-occurring substance use disorder: A systematic review. Subst. Abuse Res. Treat. 10, SART-S39921. https://doi.org/10.4137/SART.S39921 (2016).
Weir, E., Allison, C. & Baron-Cohen, S. Understanding the substance use of autistic adolescents and adults: A mixed-methods approach. Lancet Psychiatry. 8, 673–685 (2021).
Weir, E., Allison, C., Warrier, V. & Baron-Cohen, S. Increased prevalence of non-communicable physical health conditions among autistic adults. Autism Int. J. Res. Pract. 25, 681–694 (2021).
Sexton, M., Cuttler, C. & Mischley, L. K. A survey of cannabis acute effects and withdrawal symptoms: Differential responses across user types and age. J. Altern. Complement. Med.. 25, 326–335 (2019).
Aran, A. & Cayam Rand, D. Cannabinoid treatment for the symptoms of autism spectrum disorder. Expert Opin. Emerg. Drugs 29, 65–79 (2024).
Skaper, S. D. & Di Marzo, V. Endocannabinoids in nervous system health and disease: The big picture in a nutshell. Philos. Trans. R Soc. Lond. B Biol. Sci. 367, 3193–3200 (2012).
Devinsky, O. et al. Cannabidiol: Pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia 55, 791–802 (2014).
Fernández-Ruiz, J. et al. Cannabidiol for neurodegenerative disorders: important new clinical applications for this phytocannabinoid? Br. J. Clin. Pharmacol. 75, 323–333 (2013).
Aychman, M. M., Goldman, D. L. & Kaplan, J. S. Cannabidiol’s neuroprotective properties and potential treatment of traumatic brain injuries. Front Neurol 14, 1087011 (2023).
Hill, M. N., Haney, M., Hillard, C. J., Karhson, D. S. & Vecchiarelli, H. A. The endocannabinoid system as a putative target for the development of novel drugs for the treatment of psychiatric illnesses. Psychol. Med. 53, 7006–7024. https://doi.org/10.1017/S0033291723002465 (2023).
Aran, A., Cassuto, H., Lubotzky, A., Wattad, N. & Hazan, E. Brief report: Cannabidiol-rich cannabis in children with autism spectrum disorder and severe behavioral problems—A retrospective feasibility study. J. Autism Dev. Disord. 49, 1284–1288. https://doi.org/10.1007/s10803-018-3808-2 (2018).
Barchel, D. et al. Oral cannabidiol use in children with autism spectrum disorder to treat related symptoms and co-morbidities. Front. Pharmacol. 9, 1521 (2018).
Bar-Lev Schleider, L., Mechoulam, R., Saban, N., Meiri, G. & Novack, V. Real life experience of medical cannabis treatment in autism: Analysis of safety and efficacy. Sci Rep 9, 200 (2019).
Fleury-Teixeira, P., Caixeta, F. V., Ramires da Silva, L. C., Brasil-Neto, J. P. & Malcher-Lopes, R. Effects of CBD-enriched cannabis sativa extract on autism spectrum disorder symptoms: An observational study of 18 participants undergoing compassionate use. Front. Neurol. 10, 1145 (2019).
Aran, A. et al. Cannabinoid treatment for autism: A proof-of-concept randomized trial. Mol. Autism. 12, 6 (2021).
Cuttler, C., Spradlin, A. & McLaughlin, R. J. A naturalistic examination of the perceived effects of cannabis on negative affect. J. Affect. Disord. 235, 198–205 (2018).
Sizoo, B. B. et al. Predictive validity of self-report questionnaires in the assessment of autism spectrum disorders in adults. Autism Int. J. Res. Pract. 19, 842–849 (2015).
Friedman, A., Paltoglou, A. & Sorte, R. A qualitative exploration of the experiences of self-diagnosed autistic women and gender-diverse individuals who are not pursuing an autism diagnosis. Neurodiversity 2, 27546330241307828 (2024).
Chester, V. Autistic women and girls: under-recognised, under-researched and under-served. Adv. Autism. 5, 1–1 (2019).
Lockwood Estrin, G., Milner, V., Spain, D., Happé, F. & Colvert, E. Barriers to autism spectrum disorder diagnosis for young women and girls: A systematic review. Rev. J. Autism Dev. Disord. 8, 454–470 (2021).
Kapp, S. K. et al. People should be allowed to do what they like’: Autistic adults’ views and experiences of stimming. Autism 23, 1782–1792 (2019).
Charlton, R. A., Entecott, T., Belova, E. & Nwaordu, G. It feels like holding back something you need to say: Autistic and non-autistic adults accounts of sensory experiences and stimming. Res. Autism Spectr. Disord. 89, 101864 (2021).
Alatrash, S. et al. Predictors of irritability in pediatric autistic populations: A scoping review. Front. Child. Adolesc. Psychiatry. 3, 1393231 (2024).
Nimmo-Smith, V. et al. Anxiety disorders in adults with autism spectrum disorder: A population-based study. J. Autism Dev. Disord. 50, 308–318 (2020).
Acknowledgements
The authors extend their sincere gratitude and acknowledgement of the autistic participants who generously contributed their time and insights to this study. We also thank the staff and colleagues at the University of New Orleans and Washington State University for their support and assistance throughout this research.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
D.S.K. and C.C. equally contributed to the conceptualization, interpretation, and writing—original draft, review and editing of the manuscript. C.C. and E.M.L. equally contributed to data curation and formal analysis. E.M.L. also participated in writing, reviewing, and editing. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Karhson, D.S., LaFrance, E.M. & Cuttler, C. Acute effects of cannabis on core and co-occurring features associated with autism spectrum disorder in adults. Sci Rep 15, 39849 (2025). https://doi.org/10.1038/s41598-025-23472-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-23472-3