Abstract
Myocardial infarction (MI) remains a leading cause of heart failure, yet the relationship between functional decline and structural remodeling is not fully defined. This study evaluated how echocardiographic parameters correspond with stereological measures of myocardial tissue in a rabbit MI model. Sixteen male New Zealand White rabbits were initially enrolled (MI, n = 12; sham, n = 4). Following two perioperative deaths, fourteen animals completed the study (MI, n = 10; sham, n = 4). MI was induced by permanent ligation of the left circumflex artery. Echocardiography (B-mode, M-mode, Doppler) was performed at baseline, immediately after surgery, and at 3 and 6 weeks (sham animals at baseline and post-surgery only). After 6 weeks, stereological analysis quantified cardiomyocyte, connective tissue, and vascular volumes globally and within infarct regions. MI rabbits showed directional trends toward ventricular dilation, wall thinning, and reductions in ejection fraction and fractional shortening, with a significant decline in early transmitral filling velocity (E-wave, p < 0.05). Global stereological analysis revealed no significant differences between MI and sham groups, but regional assessment suggested trends toward localized fibrosis, cardiomyocyte loss, and microvascular rarefaction in infarcted areas. Echocardiography sensitively detects early post-infarction functional impairment, whereas global stereology may underestimate structural remodeling. Regional stereological evaluation provides additional insight, highlighting the value of combining functional imaging with quantitative tissue analysis in experimental MI.
Similar content being viewed by others
Introduction
Cardiovascular diseases (CVD) remain the leading cause of mortality worldwide, with ischemic heart disease representing a major contributor to this burden1,2,3. Among ischemic conditions, myocardial infarction (MI), characterized by irreversible cardiomyocyte death due to prolonged ischemia, is a key driver of heart failure and long-term cardiac dysfunction. Despite improvements in acute management, post-infarction ventricular remodeling—encompassing structural, geometric, and functional changes—remains a critical determinant of morbidity and mortality4,5. A deeper understanding of the mechanisms underlying cardiac remodeling, and the development of reliable, translational assessment tools, are essential to improving clinical outcomes after MI.
Ventricular remodeling involves complex, dynamic active and passive tissue-level processes, including cardiomyocyte loss, fibrotic matrix expansion, and alterations in myocardial stiffness and mechanics. Recent studies in small animal models have demonstrated that infarction initiates both passive remodeling, characterized by extracellular matrix (ECM) expansion, collagen deposition, and increased myocardial stiffness, as well as active remodeling processes, including cardiomyocyte hypertrophy, sarcomere disarray, and reorientation of myocardial fiber architecture6,7.These microstructural adaptations disrupt myocardial mechanical efficiency and precede the macroscopic geometric alterations detectable by imaging modalities8,9,10.
Echocardiography is a widely accessible, non-invasive imaging technique that provides real-time evaluation of cardiac geometry, contractility, and diastolic function. It has become a cornerstone for monitoring left ventricular remodeling after MI11,12. However, echocardiography predominantly assesses macroscopic functional parameters and may fail to capture microscopic tissue-level changes, including cardiomyocyte loss, extracellular matrix remodeling, or alterations in microvascular architecture. Conversely, histological techniques—particularly stereological analysis—enable quantitative assessment of myocardial microstructure, providing insight into cellular and extracellular matrix composition13. Despite the complementary value of these approaches, few studies have systematically examined the relationship between echocardiographic indices and quantitative, tissue-level structural remodeling, especially in translational animal models of MI.
Animal models remain indispensable for investigating the pathophysiological processes of MI and evaluating diagnostic tools under controlled conditions14. Although small rodents such as mice and rats are frequently used for preclinical cardiac research, their small heart size, rapid heart rate, and technical imaging challenges limit their translational relevance for certain applications. In contrast, rabbits offer a favorable intermediate model, with a manageable body size, slower heart rate, and cardiac anatomy that enhances echocardiographic resolution of structural and functional parameters14. Previous studies across species—including rats, dogs, pigs, and even earlier rabbit work—have described structural remodeling after MI, such as collagen deposition, infarct expansion, or ventricular dilation. However, these investigations were generally limited to descriptive histology or global functional measures, without systematic correlation between imaging parameters and unbiased quantitative stereology, and rarely distinguished infarct, remote myocardial zones. Despite these advances, the extent to which echocardiographic indicators of ventricular dysfunction reflect underlying microstructural remodeling—including cardiomyocyte loss, fibrosis, and vascular alterations—remains poorly defined.
The present study provides the first integrated, longitudinal correlation of echocardiographic function with stereological quantification in a rabbit MI model, incorporating regional tissue analysis (infarct and remote) to capture spatial heterogeneity of remodeling. By directly linking functional deterioration to microstructural remodeling, this work addresses critical gaps left by prior species studies that focused on either imaging or histology in isolation. This combined approach highlights the potential translational value of regional stereological analysis as a complement to echocardiography, offering novel insights into the structure–function relationship during post-infarction remodeling.
Materials and methods
Ethical approval and animal welfare
All experimental procedures were conducted in strict accordance with institutional and national guidelines for the care and use of laboratory animals, as established by the University of Tehran. The study protocol adhered to the ethical principles outlined in the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines to ensure the humane treatment of research animals. Ethical approval for this study was granted by the Iran National Committee for Ethics in Biomedical Research under approval code IR.UT.VETMED.REC.1402.041. All efforts were made to minimize animal suffering, including the use of appropriate anesthesia, analgesia, and humane endpoints throughout the experimental period.
Animal model description
Sixteen healthy male New Zealand White rabbits (Oryctolagus cuniculus), aged 12–14 weeks and weighing 2.5–3.0 kg, were used in this study. All animals were obtained from the Pasteur Institute of Karaj (Alborz Province, Iran), a certified national supplier of laboratory animals. Rabbits were housed individually in temperature-controlled cages under standard environmental conditions (22 ± 2 °C) with a 12-h light/dark cycle and free access to food and water. A minimum acclimatization period of 1 week was provided before the start of experiments to ensure physiological stabilization. Only male rabbits were included to reduce potential variability in cardiac physiology associated with hormonal fluctuations in females. This approach is consistent with previous studies on myocardial infarction models and ventricular remodeling, which have demonstrated sex-dependent differences in cardiac adaptation. While restricting the study to male rabbits reduced biological variability, it may also limit the generalizability of findings across sexes. Furthermore, the relatively small cohort size (n = 16) reflects ethical considerations in animal use.
Experimental design and group allocation
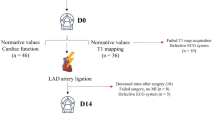
This study was designed to evaluate the relationship between functional cardiac changes assessed by echocardiography and structural remodeling quantified by stereological analysis following myocardial infarction (MI) in a rabbit model. A total of sixteen male New Zealand White rabbits were randomly assigned to either the MI group or the sham-operated control group using a randomization process to minimize selection bias and ensure comparable baseline characteristics between groups. Of the sixteen animals initially enrolled, eleven rabbits were allocated to the MI group, where myocardial infarction was induced by permanent ligation of the left circumflex coronary artery (LCx). The remaining five rabbits were assigned to the sham group, undergoing identical surgical preparation without coronary ligation to serve as procedural controls.
During the experimental period, one rabbit in the MI group died during anesthesia induction, and another MI animal died within a few hours postoperatively due to complications unrelated to the experimental intervention. Accordingly, all sixteen rabbits were included in baseline echocardiographic assessments, but only fourteen animals (10 MI and 4 sham) survived to complete the study and were available for longitudinal and stereological analysis.
Echocardiographic assessments were performed at multiple time points to monitor cardiac structure and function. All animals underwent baseline echocardiography prior to surgery, followed by immediate postoperative imaging to evaluate acute changes. Sham-operated animals were only re-imaged immediately after surgery (AS) and did not undergo scheduled follow-up at 3 or 6 weeks, in order to minimize animal use while still controlling for surgical effects. In contrast, animals in the MI group received additional follow-up echocardiographic assessments at both 3 and 6 weeks after surgery to evaluate progressive ventricular remodeling. This experimental design therefore provided time-matched controls at baseline and immediately post-surgery, while allowing longitudinal evaluation of infarct-induced remodeling in the MI group.
Myocardial infarction induction
Myocardial infarction (MI) was induced using a well-established surgical ligation model targeting the left circumflex coronary artery (LCx), a method widely applied in rabbit models of ischemic heart disease. Prior to surgery, each rabbit was sedated with an intramuscular injection of ketamine (10% solution, 30 mg/kg) combined with xylazine (2%, 5 mg/kg). After confirming adequate sedation, baseline transthoracic echocardiography was performed, as detailed in the following section. Subsequently, animals were intubated and mechanically ventilated using a Dräger Sulla 808 V anesthesia machine. General anesthesia was maintained with 2.0–2.5% isoflurane delivered in oxygen at a flow rate of 1 L per minute to ensure a stable surgical plane.
A left-sided thoracotomy was performed via the fourth or fifth intercostal space to access the thoracic cavity15. Muscle and soft tissue layers were carefully dissected to minimize trauma, and a surgical retractor was used to facilitate exposure16. The pericardium was gently incised to avoid injury to the lungs. The left circumflex coronary artery was visually identified on the lateral/posterior surface of the left ventricle, consistent with its anatomical course in rabbits. In animals assigned to the MI group, permanent ligation of the proximal LCx was performed using a 5-0 nylon suture (Ethicon, USA), inducing localized myocardial ischemia (Supplementary Fig. 1). This procedure typically results in infarction of the posterior-lateral free wall of the left ventricle, as described in previous rabbit MI models, corresponding to ~ 20–30% of the left ventricular circumference. The ligation site was consistent with protocols established in previous rabbit infarction studies to ensure reproducibility of the ischemic insult.
In the sham-operated control group, identical surgical exposure of the thoracic cavity and pericardium was conducted, but no coronary ligation was performed. This allowed assessment of the effects of surgery and anesthesia independent of ischemic injury.
Prior to thoracic closure, residual air was evacuated from the pleural space using a silicone catheter connected to a 10-ml syringe to prevent postoperative pneumothorax. Local infiltration of lidocaine (1 mg/kg) was administered at the intercostal incision site for perioperative analgesia. Postoperatively, all animals received a single intramuscular dose of enrofloxacin (10 mg/kg, 5% solution, Hydra) for infection prophylaxis and subcutaneous tramadol (5 mg/kg) to manage pain. Animals were continuously monitored throughout recovery to ensure hemodynamic stability, adequate analgesia, and humane care. As expected in surgical MI models, perioperative mortality occurred in two rabbits, consistent with reports of procedure-related risk in this species.
Echocardiography protocol
Transthoracic echocardiography was performed to assess cardiac geometry, systolic performance, and diastolic filling characteristics at multiple time points throughout the study period. All examinations were conducted using a high-resolution ultrasound system (Mylab 70 XVision, Esaote, Genoa, Italy) equipped with a 10 MHz micro-convex transducer optimized for small animal imaging. System parameters were adjusted to maximize frame rates, ensuring accurate capture of the rapid cardiac cycles characteristic of rabbits.
Prior to each imaging session, animals were sedated with an intramuscular injection of ketamine (15 mg/kg) combined with xylazine (2.5 mg/kg), representing half the dose used for surgical anesthesia. This protocol provided sufficient immobilization while preserving spontaneous respiration and minimizing cardiovascular depression. Rabbits were positioned in right lateral recumbency to obtain standard parasternal images, followed by left lateral decubitus positioning to acquire apical views.
Echocardiographic assessments included B-mode, M-mode, and Doppler imaging. B-mode images were obtained from right parasternal long-axis and short-axis views to visualize global cardiac structures and chamber morphology. The apical four-chamber view was acquired from the left lateral decubitus position to evaluate ventricular chamber size and atrial dimensions. For systolic function, end-diastolic and end-systolic left ventricular areas were manually traced on B-mode images in the apical four-chamber view, and ejection fraction (EF) was calculated using Simpson’s method (Supplementary Fig. 2A). M-mode images were recorded from the right parasternal short-axis view at the level of the mid-papillary muscles to measure interventricular septal thickness, left ventricular internal dimensions, and posterior wall thickness in both systole and diastole. This imaging plane consistently included the lateral/posterior left ventricular free wall (LCx territory); thus, LVIDd and LVIDs measurements intersected the infarct region in all MI animals. All M-mode measurements followed the leading-edge methodology recommended by the American Society of Echocardiography17 (Supplementary Fig. 2B).
Diastolic function was assessed using pulsed-wave Doppler imaging of transmitral inflow velocities. Recordings were obtained from the apical four-chamber view, with the Doppler sample volume positioned at the tips of the mitral valve leaflets. Peak early diastolic filling velocity (E wave) and late atrial contraction velocity (A wave) were measured, and the E/A ratio was calculated to characterize left ventricular filling patterns (Supplementary Fig. 3).
To minimize variability, three representative cardiac cycles were analyzed for each parameter, and results were averaged. All echocardiographic data were digitally stored and analyzed offline by a single experienced operator blinded to group allocation. Baseline Doppler values were recorded separately for sham and MI animals and are therefore presented independently in Table 2 as Pre-sham (BS) and Pre-MI (BS) to allow within-group longitudinal comparisons. Parameters derived from these measurements are summarized in Table 1 (structural and systolic indices) and Table 2 (Doppler indices).
Histology and stereological analysis
After the final echocardiographic assessment, deep anesthesia was induced with an intraperitoneal injection of ketamine (60 mg/kg body weight) and xylazine (8 mg/kg body weight)17. Following confirmation of surgical anesthesia, euthanasia was performed via intravenous administration of pentobarbital sodium (150 mg/kg). The thorax was then opened, and the hearts were excised and immediately immersed in neutral-buffered formaldehyde for fixation.
The left ventricle, including the interventricular septum, was carefully separated, while the chordae tendineae and trabecular muscles were removed. Approximately 8–10 transverse slices (depending on cardiac size) were prepared from each heart using a tissue slicer according to systematic uniform random sampling principles.
Standard histological processing was conducted, including dehydration through a graded ethanol series, clearing in xylene, and paraffin embedding. Section (5 μm thick) were cut using a microtome and stained with Masson’s Trichrome to differentiate connective tissue, cardiomyocytes, and vascular structures.
Stained sections were photographed using an Olympus light microscope (CX40, Olympus) equipped with a digital camera (MB-225). Microscopic fields were selected randomly and systematically for stereological evaluation. A test grid of equally spaced points was superimposed on the digital images, and the number of points hitting each tissue component (cardiomyocytes, blood vessels, or connective tissue) was counted using ImageJ software (National Institutes of Health, Bethesda, MD, USA; Version 1.54p, https://imagej.nih.gov/ij/). The relative volume fraction (Vv) of each component was calculated according to the standard point-counting formula:
where ΣP(structure) is the number of points on the tissue of interest and ΣP(total) is the total number of test points over the ventricular section (Fig. 1). In cases of overlap, classification was based on the predominant tissue within the respective grid cell.
Representative stereological analysis of myocardial tissue before and after infarction. Masson’s Trichrome–stained ventricular sections demonstrate pre-infarction myocardium with organized cardiomyocyte alignment and minimal connective tissue (A) compared to post-infarction myocardium showing disrupted fiber architecture, increased interstitial fibrosis (blue), and reduced viable cardiomyocyte density (B). Scale bar = 200 μm.
For interpretative purposes, analyses were performed both globally for the entire left ventricle and separately for infarcted and remote myocardium. Infarct size was expressed as the proportion of scar tissue relative to the total left ventricular myocardium on Masson’s Trichrome sections. All stereological analyses were conducted by a single investigator blinded to group allocation to minimize observational bias.
Statistical analysis
Data analysis was performed using Python (v3.11) with Pandas (v1.5.3), SciPy (v1.11.1), Matplotlib (v3.7.1), and Seaborn (v0.12.2). Continuous variables were tested for normality with the Shapiro–Wilk test and for homogeneity of variance using Levene’s test. Parametric data were analyzed using paired or independent two-tailed Student’s t tests, as appropriate. For non-parametric data, the Wilcoxon signed-rank test or Mann–Whitney U test was applied. Time-dependent changes in echocardiographic parameters within the MI group were assessed using repeated-measures ANOVA with Bonferroni correction for multiple comparisons. A p value < 0.05 was considered statistically significant. Exact p values are reported for key comparisons, as recommended by journal guidelines. Results are expressed as mean ± standard error of the mean (SEM). Graphs include error bars representing SEM, with statistical significance indicated using standard symbols (p < 0.05).
Results
Animal survival and experimental completion
A total of sixteen male New Zealand White rabbits were initially enrolled. One animal in the myocardial infarction (MI) group died during anesthesia induction and was excluded from further analysis. Another MI rabbit died within several hours postoperatively from complications unrelated to the experimental procedure. No mortality occurred in the sham-operated group. Thus, fourteen rabbits successfully completed the study, comprising ten animals in the MI group and four in the sham group. Data from all surviving animals were included in the final echocardiographic and stereological analyses.
Echocardiographic assessment of left ventricular geometry
Serial echocardiographic measurements demonstrated progressive alterations in left ventricular geometry following myocardial infarction (MI). In the MI group, the left ventricular internal dimension during diastole (LVIDd) increased from 14.63 ± 0.78 mm at baseline to 16.83 ± 1.45 mm at 6 weeks, although this change was not statistically significant (p = 0.2031). Similarly, the systolic internal dimension (LVIDs) rose from 9.28 ± 1.05 mm to 12.10 ± 1.74 mm (p = 0.1858) (Fig. 2). These changes were accompanied by a reduction in left ventricular wall thickness: parietal wall thickness decreased from 3.66 ± 0.38 mm to 2.57 ± 0.53 mm (p = 0.1137), and interventricular septal thickness declined from 2.97 ± 0.41 mm to 2.16 ± 0.45 mm (p = 0.2001) (Supplementary Fig. 3). Left ventricular mass increased slightly from 20.86 ± 1.17 mm² at baseline to 21.79 ± 0.61 mm² at 6 weeks (p = 0.4928). Sham-operated animals (baseline vs. immediate post-surgery) exhibited no significant changes in chamber dimensions, wall thickness, or ventricular mass, confirming that these trends were specific to the infarction model.
Progressive changes in left ventricular internal dimensions following myocardial infarction. Time-dependent alterations in left ventricular internal diameter at diastole (LVIDd) and systole (LVIDs) are shown for MI and sham-operated rabbits across four timepoints: BS = baseline (before surgery), AS = after surgery, 3 W = 3 weeks, and 6 W = 6 weeks. Sham animals were imaged only at BS and AS; therefore, no sham data are available at 3–6 W. Both LVIDd and LVIDs showed progressive increases in the MI group over 6 weeks, consistent with post-infarction ventricular remodeling, while sham animals remained stable between BS and AS. Statistical comparisons (MI group, BS vs. 6 W) yielded p = 0.2031 (LVIDd) and p = 0.1858 (LVIDs). Values are expressed as mean ± SEM.
Systolic function: ejection fraction and fractional shortening
Progressive deterioration in systolic function was observed in the MI group over 6 weeks. Ejection fraction (EF), measured from the apical four-chamber view using Simpson’s method, decreased from 62.15 ± 4.47% at baseline to 52.19 ± 5.61% at 3 weeks (p = 0.1828) and remained reduced at 52.14 ± 4.18% at 6 weeks (p = 0.1194) (Fig. 3). M-mode–derived EF showed a similar decline, from 71.20 ± 10.07% at baseline to 63.80 ± 5.24% at 3 weeks (p = 0.5254) and 56.29 ± 9.96% at 6 weeks (p = 0.3064), though these changes were not statistically significant. Fractional shortening (FS) also decreased, from 36.50 ± 6.40% at baseline to 31.20 ± 6.46% at 3 weeks (p = 0.5672) and 28.29 ± 5.59% at 6 weeks (p = 0.3470). Sham-operated animals (baseline vs. immediate post-surgery) showed no significant changes in EF or FS, with values remaining within physiological ranges. Notably, differences in EF values reflect the use of distinct echocardiographic methods: Simpson’s B-mode tracing of the apical four-chamber view yielded lower EF values (≈ 52–62%), whereas M-mode measurements from the short-axis consistently gave higher EF values (≈ 56–71%). This methodological variability is consistent with previous rabbit echocardiography studies and does not affect the within-group trends reported.
Comparison of ejection fraction (EF) measurements obtained using M-mode and Simpson’s area-tracing methods across timepoints. Data are shown for sham and myocardial infarction (MI) groups at baseline (BS = before surgery), after surgery (AS), and follow-up (3 W = 3 weeks, 6 W = 6 weeks). Sham animals underwent echocardiography only at BS and AS; therefore, no sham data are available at 3–6 W. Both methods demonstrate a progressive decline in EF in MI animals, confirming consistent detection of systolic dysfunction. Statistical comparisons (MI group, BS vs. 6 W) yielded p = 0.3064 (M-mode) and p = 0.1194 (Simpson’s). Values are presented as mean ± SEM.
Left atrial size and aortic root ratio (LA/AO)
The left atrial to aortic root diameter ratio (LA/AO) showed a modest, time-dependent increase in the MI group, rising from 1.54 ± 0.06 at baseline to 1.61 ± 0.05 at 3 weeks (p = 0.4435) and 1.66 ± 0.04 at 6 weeks (p = 0.1852) (Table 1). Although the trend suggested progressive left atrial enlargement, the changes did not reach statistical significance. Sham-operated animals (baseline vs. immediate post-surgery) maintained stable LA/AO ratios (p = 0.8913), confirming that the observed increase was specific to infarcted animals.
Diastolic function: transmitral doppler flow analysis
Pulsed-wave Doppler assessment of transmitral inflow revealed progressive diastolic dysfunction in the MI group over 6 weeks. The peak early diastolic filling velocity (E wave) was 0.64 ± 0.11 cm/s at baseline and declined significantly to 0.47 ± 0.05 cm/s at 6 weeks (p = 0.0451), indicating impaired early filling. The peak atrial contraction velocity (A wave) decreased from 0.41 ± 0.07 cm/s to 0.35 ± 0.04 cm/s, though this change was not statistically significant (p = 0.1663). The E/A ratio declined from 1.54 ± 0.16 to 1.37 ± 0.15 after MI, but this reduction was also non-significant (p = 0.2348) (Fig. 4). Immediately after surgery, no significant changes were observed in E wave, A wave, or E/A ratio in either group (all p > 0.05), suggesting that diastolic impairment developed progressively rather than acutely. Sham-operated animals (baseline vs. immediate post-surgery) maintained stable transmitral flow parameters (all p > 0.05).
Assessment of diastolic function using transmitral Doppler flow analysis. The E/A ratio (mean ± SEM) is shown for myocardial infarction (MI) and sham-operated rabbits at baseline (BS), after surgery (AS), and during follow-up (3 W, 6 W). In the MI group, the early diastolic filling velocity (E wave) declined significantly (p = 0.0451), while the atrial contraction velocity (A wave) showed a non-significant reduction (p = 0.1663). The overall E/A ratio also decreased (p = 0.2348), reflecting progressive impairment of left ventricular relaxation after infarction.
Stereological analysis of myocardial structure
Quantitative stereological evaluation showed no statistically significant differences in the global volume densities of cardiomyocytes, connective tissue, or vasculature between MI and sham groups (Table 2; Fig. 3). The cardiomyocyte volume fraction was 74.8 ± 1.3% in the MI group versus 74.6 ± 1.2% in shams (p = 0.82). Connective tissue volume increased slightly from 18.9 ± 1.1% in shams to 19.9 ± 1.2% after MI (p = 0.44), while vascular volume density decreased from 6.0 ± 0.5% to 5.0 ± 0.6% (p = 0.37). Variability in stereological parameters was greater in the MI group, particularly for myocardial and capillary volumes, reflecting heterogeneous remodeling responses across animals (Supplementary Fig. 4).
The MI group exhibited a mean infarct size of 15.2 ± 1.5% LV (range 8.5–21.7%), whereas sham hearts showed 0% (no scar). Individual animal values are presented in Supplementary Fig. 5, which illustrates the distribution and heterogeneity of infarct size across animals.
Regional analysis revealed early remodeling trends in the infarct zone compared with remote myocardium. Connective tissue volume was higher (22.5 ± 1.3% vs. 18.7 ± 1.2%, p = 0.07), while cardiomyocyte density was lower (71.2 ± 1.5% vs. 75.3 ± 1.4%, p = 0.06). Vascular density also declined (4.2 ± 0.4% vs. 5.4 ± 0.5%), approaching statistical significance (p = 0.05) (Table 3). These findings suggest localized fibrotic remodeling and microvascular rarefaction in the infarct region, despite no significant global changes.
To assess the relationship between myocardial structure and systolic performance, left ventricular ejection fraction (EF) was correlated with cardiomyocyte volume across all animals (Fig. 5). A strong positive relationship was observed (R² = 0.80), indicating that reduced cardiomyocyte volume was associated with lower EF in MI rabbits, consistent with impaired contractile performance secondary to structural remodeling.
Relationship between cardiomyocyte volume and left ventricular ejection fraction (EF) in myocardial infarction (MI) and sham rabbits. Each point represents one animal (MI: n = 10; Sham: n = 4). A strong positive correlation was observed between cardiomyocyte volume (Vc) and EF (R² = 0.80), suggesting that smaller cardiomyocyte volumes are associated with reduced systolic function after MI. Sham animals clustered at higher EF and larger Vc values, reflecting preserved myocardial structure and function.
Discussion
In this study, myocardial infarction (MI) induced by permanent ligation of the left circumflex coronary artery (LCx) in rabbits resulted in progressive structural and functional alterations, as demonstrated by longitudinal echocardiographic assessments. The left ventricular internal diameter during diastole (LVIDd) increased from 14.63 ± 0.78 mm at baseline to 16.83 ± 1.45 mm by 6 weeks post-MI; however, this change did not reach statistical significance (p = 0.2031). Similarly, the left ventricular internal diameter during systole (LVIDs) increased from 9.28 ± 1.05 mm to 12.10 ± 1.74 mm, though this difference was not statistically significant (p = 0.1858). These geometric alterations were accompanied by reductions in parietal wall thickness (3.66 ± 0.38 mm to 2.57 ± 0.53 mm, p = 0.1137) and septal wall thickness (2.97 ± 0.41 mm to 2.16 ± 0.45 mm, p = 0.2001), but neither of these changes reached statistical significance. Despite the lack of statistically significant remodeling, the observed directional trends toward ventricular dilation and wall thinning are consistent with the early stages of adverse ventricular remodeling frequently reported following MI18. In addition, stereological quantification confirmed that LCx ligation produced infarcts involving on average ~ 22% of the LV (range 11–32%), consistent with expectations for this model and providing important context for the heterogeneous functional outcomes observed across animals.
While numerous animal models dating back to the 1970s have described structural and functional remodeling after MI in mice, dogs, pigs, and other species, the present study offers two key contributions. First, it uniquely combines longitudinal echocardiography with unbiased stereological quantification within the same rabbit cohort, enabling direct comparison of functional decline with tissue-level remodeling. Second, by applying regional stereological analysis, we show that early fibrosis and cardiomyocyte loss can be detected locally in infarct zones even when global volume fractions remain unchanged. This dissociation between global function and structure, rarely emphasized in prior rabbit studies, highlights the importance of region-specific evaluation in early post-MI remodeling.
Functionally, left ventricular systolic performance exhibited directional decline over the 6-week follow-up period. Ejection fraction (EF) derived from M-mode echocardiography decreased from 71.20 ± 10.07% at baseline to 56.29 ± 9.96% at 6 weeks, though this reduction was not statistically significant (p = 0.3064). Simpson’s B-mode tracing of the apical four-chamber view yielded lower EF values overall, declining from 62.15 ± 4.47 to 52.14 ± 4.18% (p = 0.1194). Fractional shortening (FS) also declined from 36.50 ± 6.40 to 28.29 ± 5.59%, without reaching statistical significance (p = 0.3470). Although these systolic parameters did not show statistically significant deterioration, the downward trends align with the progression of systolic dysfunction described in previous rabbit MI models19, including the study by Zhang et al. (2023), which reported a reduction in EF from approximately 65% to below 50% within 4 weeks post-MI15.
In terms of diastolic function, transmitral Doppler assessments revealed a significant decrease in early diastolic filling velocity (E wave) from 0.64 ± 0.11 cm/s to 0.47 ± 0.05 cm/s at 6 weeks post-MI (p = 0.0451), indicating impaired ventricular relaxation. In contrast, the atrial contraction velocity (A wave) decreased from 0.41 ± 0.07 to 0.35 ± 0.04 cm/s, though this change was not statistically significant (p = 0.1663). Consequently, the E/A ratio decreased from 1.54 ± 0.16 to 1.37 ± 0.15, but this difference also did not reach statistical significance (p = 0.2348). These trends are consistent with early diastolic dysfunction observed in both preclinical and clinical settings20. For example, Prasad et al. (2018) demonstrated that reduced E/A ratios following MI are associated with increased risk of adverse outcomes in human patients21.
Although echocardiographic assessments revealed evidence of functional decline, stereological analysis detected only modest structural remodeling at the global myocardial level22. The cardiomyocyte volume fraction remained unchanged between MI and sham animals (74.8% vs. 74.6%, p = 0.82), connective tissue volume demonstrated a slight but non-significant increase (19.9% vs. 18.9%, p = 0.44), and vascular density decreased marginally (5.0% vs. 6.0%, p = 0.37). These findings are consistent with prior studies such as Guevara et al. (2024), where sympathetic denervation following MI occurred predominantly in infarct zones, while global myocardial structure remained stable23.
Regional stereological analysis revealed early, localized trends toward remodeling in infarcted regions. Specifically, connective tissue volume increased to 22.5 ± 1.3% in infarct zones compared to 18.7 ± 1.2% in remote myocardium (p = 0.07), while cardiomyocyte volume decreased to 71.2 ± 1.5% versus 75.3 ± 1.4% (p = 0.06). Although these differences did not achieve statistical significance, the observed patterns suggest localized fibrosis and cardiomyocyte loss consistent with early post-infarction remodeling processes24,25. Similar region-specific remodeling has been reported by Zhang et al. (2023) and Guevara et al. (2024), emphasizing the limitations of relying solely on global tissue quantification15,23.
Importantly, our correlation analysis revealed no significant relationship between global left ventricular EF and global cardiomyocyte volume density (r = 0.21, p = 0.46), underscoring the dissociation between functional decline and detectable whole-heart structural remodeling during the early post-MI period26,27. The relatively large standard deviations observed in post-MI EF and stereological values indicate biological heterogeneity across animals. Quantitative infarct measurements confirmed that LCx ligation produced infarcts involving on average ~ 22% of the LV (range 11–32%). Some rabbits with larger infarcts exhibited pronounced functional decline and histological remodeling, whereas others with smaller infarcts maintained near-baseline structure and function. This variability likely reflects differences in infarct size and collateral circulation following LCx ligation and highlights the value of evaluating individual-animal and regional outcomes rather than relying solely on global group means. Inspection of individual trajectories further confirmed this variability, with certain animals showing early structural and functional deterioration while others remained largely preserved, reinforcing the importance of case-level interpretation alongside group averages. This finding is supported by work from Botting et al. (2014), who demonstrated that cardiomyocyte loss in sheep subjected to chronic hypoxia was only detectable after extended observation periods, suggesting that early structural remodeling may initially be confined to localized regions and insufficient to alter global tissue metrics28.
Compared to existing rabbit MI studies, our 6-week follow-up provides a longer temporal window for evaluating progressive remodeling. For instance, Chang et al. (2024) assessed functional outcomes at 3 weeks post-MI, identifying increased arrhythmia susceptibility but without accompanying structural data29. In contrast, our extended timeline allowed for the detection of evolving functional decline alongside localized histological remodeling.
To further illustrate the heterogeneity observed across animals, we plotted individual EF trajectories for all MI rabbits (Supplementary Fig. 6). This visualization highlights that, while several animals experienced substantial declines in systolic function over 6 weeks, others maintained near-baseline EF despite undergoing the same infarction protocol. These findings emphasize the biological variability of remodeling outcomes, likely reflecting differences in infarct size and collateral circulation, and reinforce the importance of evaluating individual-animal data alongside group averages.
This study has several limitations. First, although stereology provides robust quantitative assessment, global volume density measurements may fail to capture subtle, localized remodeling. Our regional analysis partly addressed this; however, the relatively small sample size—particularly in the sham group—likely reduced the statistical power needed to detect modest but biologically meaningful differences. The observed trends toward increased fibrosis and reduced cardiomyocyte volume in infarct zones approached, but did not reach, statistical significance, suggesting that larger studies are necessary to validate these findings.
Second, the 6-week follow-up may not fully reflect later stages of remodeling. Prior work, such as Botting et al. (2014), demonstrated that structural changes including measurable cardiomyocyte deficits can emerge only after longer observation periods, indicating that extended timelines are warranted in future investigations28. Third, only male rabbits were included to minimize variability, which limits the generalizability of our results across sexes. In addition, echocardiography was performed under anesthesia, which was necessary for image acquisition but may have influenced cardiac function measurements. Finally, although all analyses were performed by a single experienced investigator to reduce variability, incorporating blinded multi-observer assessments would provide greater robustness30.
Taken together, these limitations highlight the importance of larger, longer-term, and sex-inclusive studies that integrate functional imaging with region-specific histological and molecular approaches. Such efforts will better clarify the dissociation we observed between functional impairment and global stereological stability, while confirming the early, localized remodeling trends within infarct zones. Ultimately, advancing toward region-specific structure–function analyses will strengthen the translational value of the rabbit MI model for studies of myocardial repair and disease progression31.
Conclusion
This study demonstrates that myocardial infarction induced by LCx ligation in rabbits leads to early functional deterioration, particularly impaired diastolic filling, alongside directional but not statistically significant systolic and geometric changes. Despite these alterations, global stereological analysis did not reveal significant structural remodeling, while regional analysis suggested localized trends toward fibrosis and cardiomyocyte loss. Variability in outcomes across animals was partly explained by differences in infarct size, emphasizing the importance of considering individual-animal and region-specific data when interpreting remodeling. The rabbit MI model therefore provides a valuable platform for investigating early myocardial remodeling and supports future studies incorporating larger cohorts, longer follow-up, and sex-inclusive designs to enhance translational relevance.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Savarese, G. et al. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovasc. Res. 118, 3272–3287 (2022).
Münzel, T. et al. Environmental risk factors and cardiovascular diseases: a comprehensive expert review. Cardiovasc. Res. 118, 2880–2902 (2022).
Townsend, N. et al. Epidemiology of cardiovascular disease in Europe. Nat. Rev. Cardiol. 19, 133–143 (2022).
Jenča, D. et al. Heart failure after myocardial infarction: incidence and predictors. ESC Heart Fail. 8, 222–237 (2021).
Triposkiadis, F., Xanthopoulos, A., Parissis, J., Butler, J. & Farmakis, D. Pathogenesis of chronic heart failure: cardiovascular aging, risk factors, comorbidities, and disease modifiers. Heart Fail. Rev. 27, 337–344 (2022).
Mendiola, E. A. et al. An image-driven micromechanical approach to characterize multiscale remodeling in infarcted myocardium. Acta Biomater. 173, 109–122 (2024).
Neelakantan, S. et al. Multiscale characterization of left ventricle active behavior in the mouse. Acta Biomater. 162, 240–253 (2023).
Mandoli, G. E. et al. Novel approaches in cardiac imaging for non-invasive assessment of left heart myocardial fibrosis. Front. Cardiovasc. Med. 8, 614235 (2021).
Lindsey, M. L. et al. Guidelines for in Vivo Mouse Models of Myocardial Infarction (American Journal of Physiology-Heart and Circulatory Physiology, 2021).
Zhang, J. et al. Early and dynamic detection of doxorubicin induced cardiotoxicity by myocardial contrast echocardiography combined with two-dimensional speckle tracking echocardiography in rats. Front. Cardiovasc. Med. 9, 1063499 (2023).
Lindner, J. R. Contrast echocardiography: current status and future directions. Heart 107, 18–24 (2021).
Barry, T. et al. The role of artificial intelligence in echocardiography. J. Imaging. 9, 50 (2023).
Mühlfeld, C. & Schipke, J. Methodological progress of stereology in cardiac research and its application to normal and pathological heart development. Cells 11, 2032 (2022).
Pilz, P. M. et al. Large and small animal models of heart failure with reduced ejection fraction. Circ. Res. 130, 1888–1905 (2022).
Zhang, J. et al. Adult bone marrow-derived mesenchymal stem cells seeded on tissue-engineered cardiac patch contribute to myocardial scar remodeling and enhance revascularization in a rabbit model of chronic myocardial infarction. In The Heart Surgery Forum E148–55 (2023).
Monnet, E. Small Animal Soft Tissue Surgery (Wiley, 2023).
Hahn, R. T. et al. Recommended standards for the performance of transesophageal echocardiographic screening for structural heart intervention: from the American society of echocardiography. J. Am. Soc. Echocardiogr. 35, 1–76 (2022).
Leancă, S. A. et al. Left ventricular remodeling after myocardial infarction: from physiopathology to treatment. Life 12, 1111 (2022).
Wohlfahrt, P. et al. Trajectories and determinants of left ventricular ejection fraction after the first myocardial infarction in the current era of primary coronary interventions. Front. Cardiovasc. Med. 9, 1051995 (2022).
Giga, V. et al. Predictors of diastolic deceleration time of coronary flow velocity of infarct related and reference coronary artery assessed by transthoracic doppler echocardiography in the chronic phase of successfully reperfused anterior myocardial infarction: relation to infarct size. Front. Cardiovasc. Med. 10, 1196206 (2023).
Prasad, S. B. et al. Diastolic dysfunction assessed using contemporary guidelines and prognosis following myocardial infarction. J. Am. Soc. Echocardiogr. 31, 1127–1136 (2018).
Campbell, D. J., Francis, V. C., Young, G. R. & Woodford, N. W. Association of coronary microvascular rarefaction and myocardial fibrosis with coronary artery disease. J. Am. Heart Assoc. 13, e037332 (2024).
Guevara, A. et al. Sympathetic structural and electrophysiological remodeling in a rabbit model of reperfused myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 327, H631–H638 (2024).
Baudouy, D., Michiels, J-F., Vukolic, A., Wagner, K-D. & Wagner, N. Echocardiographic and histological examination of cardiac morphology in the mouse. J. Vis. Exp. 126, e55843 (2017).
Schipke, J. et al. Experimentally induced intrauterine growth restriction in rabbits leads to differential remodelling of left versus right ventricular myocardial microstructure. Histochem. Cell Biol. 148, 557–567 (2017).
Zhang, H. et al. Detection of diffuse to focal myocardial fibrosis by cardiovascular magnetic resonance against histology in Mini-Swine. BioRxiv 632871 (2025).
Yang, Z. et al. Contrast-enhanced echocardiographic diagnosis of benign and malignant cardiac tumors and its correlation with pathology. Front. Cardiovasc. Med. 10, 1182334 (2023).
Botting, K. J., McMillen, I. C., Forbes, H., Nyengaard, J. R. & Morrison, J. L. Chronic hypoxemia in late gestation decreases cardiomyocyte number but does not change expression of hypoxia-responsive genes. J. Am. Heart Assoc. 3, e000531 (2014).
Chang, P-C. et al. Vericiguat suppresses ventricular tachyarrhythmias inducibility in a rabbit myocardial infarction model. PLoS One. 19, e0301970 (2024).
Pergola, V. et al. Unveiling the hidden chamber: exploring the importance of left atrial function and filling pressure in cardiovascular health. J. Cardiovasc. Echogr. 33, 117–124 (2023).
Tani, H. et al. Direct reprogramming improves cardiac function and reverses fibrosis in chronic myocardial infarction. Circulation 147, 223–238 (2023).
Acknowledgements
The authors thank the staff of the Faculty of Veterinary Medicine, University of Tehran, for their technical support.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
F.A. and M.M. conceptualized the study. F.A. performed the surgeries, echocardiography, and data collection. M.P. assisted with echocardiographic analysis. J.S. conducted the stereological evaluations. R.I. contributed to animal care and postoperative monitoring. M.M. supervised the project. All authors contributed to data interpretation, manuscript writing, and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All experimental procedures were conducted in accordance with institutional guidelines and the ARRIVE (Animal Research: Reporting of In Vivo Experiments) recommendations. The study protocol was reviewed and approved by the Iran National Committee for Ethics in Biomedical Research under ethical approval code IR.UT.VETMED.REC.1402.041.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.






Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Aramesh, F., Masoudifard, M., Pedram, M. et al. Correlation between echocardiographic and stereological parameters in rabbit models of myocardial infarction. Sci Rep 15, 40753 (2025). https://doi.org/10.1038/s41598-025-24457-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24457-y