Abstract
Acute lymphoblastic leukemia (ALL) is the most common childhood cancer, with central nervous system (CNS) complications occurring in approximately 10% of cases. This study aimed to analyze cerebrovascular complications in pediatric ALL patients. We conducted a retrospective analysis of 144 pediatric ALL patients treated at the University Children’s Hospital in Lublin from 2014 to 2023. Statistical analyses were performed using PQStat software. Among the patients, 12.5% (n = 18) developed cerebrovascular complications, with equal distribution between genders. The median age was 6.3 years. Most complications (83.3%) occurred during induction therapy. The most frequent symptom was seizures (50%), followed by impaired consciousness (39%). Magnetic resonance imaging (MRI) detected ischemic complications in 44.4% of affected patients. No significant correlation was found between risk group, diagnostic parameters, or gender and CNS complications. CNS complications, particularly seizures and ischemic events, are common in pediatric ALL, primarily during induction therapy. MRI plays a crucial role in early detection. Further multicenter studies are necessary to improve management and reduce treatment-related neurotoxicity.
Similar content being viewed by others
Introduction
Leukemia is the most common childhood cancer, representing 28.8% of cancer cases worldwide1. CNS complications that appear in the course of ALL treatment occur in appro−ximately 10% of patients in retrospective cohort groups2,3,4. The majority of these complications appear during the first 6 months, with 37% to 56.5% occurring in the induction phase2,3,4. and may be caused by leukemia itself and/or by cancer therapies, including chemotherapy6,7,8,9,10,11. Recent studies report that age 10 years and older, a high-risk phenotype, T-cell ALL, and CNS leukemia are associated with a higher risk of CNS toxicity3,5. Based on the current literature, the most common CNS symptoms that appear in children during or shortly after ALL treatment include seizures, disturbances of consciousness, constant headaches, hemiplegia, hallucinations, and visual impairment12,13. The most common causes of those disorders include posterior reversible encephalopathy syndrome (PRES)14,15, methotrexate-related stroke-like syndrome (SLS), cerebral venous sinus thrombosis16,17,18,19, other toxic leukoencephalopathies (LE)20, CNS bleeding, including stroke21,22., and neuroinfections2,3,23. Chemotherapy drugs known to cause CNS complications and toxicity in ALL are MTX, glucocorticosteroids, vincristine, L-asparaginase (L-ASP), and cytarabine24,25. Methotrexate-derived neurotoxicity occurs in 3–7% of ALL pediatric patients26. Methotrexate metabolites are known to inhibit ribonucleotide 5-aminoimidazole-4-carboxamide formyltransferase (ATIC), thereby promoting the release of adenosine, which, by binding to adenosine receptors, modifies neuronal responses in the CNS27. The most frequently reported CNS complications associated with the use of glucocorticoids include mental and cognitive disorders.
Approximately 20% of patients develop depression, mania, and psychosis28., which require pharmacological treatment, and 75% report psychiatric symptoms that resolve after therapy discontinuation29. The incidence of neurotoxic consequences of vincristine use is not precisely known because of variability among study groups. The most significant side effect of vincristine therapy is neurotoxicity, primarily peripheral neuropathy30. According to several studies, up to 33% of children suffer from CNS disorders during L-ASP treatment31. The mechanism of L-ASP neurotoxicity derives from the breakdown of aspartate and glutamine into aspartic and glutamic acids and increased ammonia levels32. Excessive stimulation of the N-methyl-D-aspartate (NMDA) receptor can lead to cell death in CNS neurons33. Notably, the rate of severe CNS adverse events caused by cytarabine, such as somnolence, confusion, and, more infrequently, seizures, cerebral dysfunction, and acute cerebellar syndrome, is around 12% of all patients34, possibly due to wide-ranging loss of Purkinje cells in the cerebellum35.
The study’s objective was to analyze the cause, number, and type of cerebrovascular complications developed by children with ALL hospitalized for anti-cancer treatment in the Department of Pediatric Hematology, Oncology, and Transplantology of the University Children’s Hospital in Lublin from 2014 to 2023.
Index case
A 4-year-old girl assigned to a standard risk group of pre-B-ALL common ( +) developed PRES syndrome during chemotherapy induction phase. In the treatment according to the current protocol, the patient received vincristine, daunorubicin, PEG-Lasparaginase (PEG-ASP), methotrexate, and prednisone. On the 28th day of treatment, the girl developed a seizure, facial muscle spasm near the corner of the mouth on the left side, eye turn and dilated pupils without reaction to light. Laboratory test results revealed anemia (HGB 9 g/dl vs reference range 11.5–15.5 g/dl) and thrombocytopenia (26 × 103/µL vs normal: 140–410 × 103/µL). Moreover, an elevated procalcitonin level reaching 1.11 ng/mL (normal: < 0.5 ng/mL) and a C-reactive protein (CRP) level of 6.71 mg/dL (reference range 0–0.5 mg/dL) were detected. The following abnormalities were found in the coagulogram:
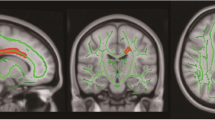
PT 26 s (normal: 12–16 s), ATIII 68% (reference range 75–150%), D-dimer 959 ng/ml (normal < 500 ng/ml). The radiological report from imaging studies showed features of PRES syndrome (Figs. 1 and 2). Cerebralfluid examination revealed pleocytosis (25 cells/ul). The neurological examination showed neck stiffness, weakened tendonreflexes, and decreased muscle tone in the upper and lower limbs. On the EEG examination, abnormal recording with periods of generalized paroxysmal, sometimes exacerbated, slow delta-theta waves (2–4 Hz to 200–250 uV) against a background of significantly slowed, with features of disorganization, baseline delta-theta waves (2–4-7 Hz to 80–150 uV). The patient communicated with gestures, reported headache, had vomiting reflexes, nausea. Treatment included diazepam, mannitol, and amlodipinum. Additionally, the girl developed profound bone marrow aplasia, leukopenia, neutropenia, thrombocytopenia, toxic liver damage, polyneuropathy, and oral mucositis, so she required broad-spectrum antibiotic therapy, blood transfusion, parenteral nutrition and rehabilitation. Interruption of anticancer treatment lasted 24 days. Currently, the patient is under the constant care of a hematologist, cardiologist, nephrologist, neurologist and speech therapist, due to heart rhythm disturbances, hypertension, epilepsy and speech disorders. The patient does not show any hematooncological changes and remains in oncological remission.
LA. Bilateral cortical/subcortical areas, mainly in the parietal/occipital (predominantly left side) and temporal (predominantly right side) regions, and partly in the thalamus, with increased IS in DF and T2-weighted images. Imaging of the lesions in correlation with the clinical picture suggests changes in the course of PRES. B. On follow-up examination: partial regression of the described lesions. In the right parietal lobe, a small, approximately 4 mm in diameter, signal area not seen in the previous examination.
Results of the study
The clinical information about children with ALL and cerebrovascular complications was summarized in Table 1. Patients’ laboratory tests were summarised in Table 2. From 2014 to 2023 (10 years) 144 young patients with ALL were hospitalized in the Department of Pediatric Hematology, Oncology, and Transplantology of the University Children’s Hospital in Lublin, Poland, among whom 12,5% (n = 18 patients) developed cerebrovascular toxicities during treatment. 58.8% of patients were treated with AIEOP 2017 and 41.2% with ALLIC 2009. All patients were hospitalized at the time when their symptoms first appeared. Cerebrovascular complications were equally distributed in girls and in boys. In our analysis, there were no cases of individuals having more than one neurological incident throughout the course of therapy. The patients’ ages averaged 7.1 years and the median distribution was 6.3 years, with a range of results from 0.6 to 15.3 years. Half of the group were patients from 2.9 to 10.8 years old. Five cases, representing 29% of the group, were found to have genetic aberrations, of which four patients had ETV6::RUNX1, and one patient KMT2A::AFF1. One case, representing 5.88% of the study group, had a family history of cystic kidney disease, which was found in the patient’s sister. Chronic diseases were present in 44% of the subjects, including SIADH and mild intellectual disability (n = 2 each ), as well as epilepsy, visual impairment, and ataxia-telangiectasia syndrome (n = 1 each). Among the chronic illnesses present in subjects, no central nervous system (CNS) related diseases or hypercoagulable states were identified.B-ALL cases represented 61.11% (n = 11) of the study group, and T-ALL patients represented 38.59% (n = 7). According to the AIEOP-BFM ALL 2017 POLAND protocol, patients are classified into standard risk (SR), intermediate risk (IR), or high risk (HR) groups based on their early response to induction therapy and the presence of high-risk genetic abnormalities. In our cohort, the SR group accounted for 27.78% of the patients, the IR group included 22.22%, and the HR group comprised 38.89%. The remaining 11.11% of patients were not assigned to a risk group at the time of CNS involvement occurrence. Most of the complications (83.33%) occured during treatment induction. The most commonly indicated symptom was seizures, occurring in half the patients. Other symptoms included impaired consciousness (39% of the subjects), petechiae (22%), limb tremors (11%), headache (11%), vomiting (6%), and nausea (6%). In a group of six (33.33%) patients, an increase in inflammatory markers, i.e.C-reactive protein and procalcitonin, was observed. Three patients had elevated protein C levels, with concentrations of 77 mg/L, 82 mg/L and 90 mg/L. Ischaemic lesions (44.44%) were most frequently observed on MRI; in a group representing 40% of the patients, lesions were found on head CT, and 20% had no lesions. The results of the imaging studies are shown in Table 1. The results of laboratory tests are shown in Table 2.
In a group representing 11.11% (n = 2) of the patients, changes were found on ultrasound of the carotid vessels and in 55.56% (n = 10) no changes were found. Treatment interruption time averaged 14.0 days and the median was 18.5 days, with a range of 0 to 30 days. No significant (p < 0.05) associations were found between gender and the presence of individual symptoms. Neither any significant (p < 0.05) associations were observed between risk groups and the incidence of individual symptoms.
There were no significant (p < 0.05) associations between risk groups and radiological changes. Nor any significant (p < 0.05) associations were found between the diagnosis and the occurrence of individual symptoms.
Discussion
Cerebrovascular complications during treatment of acute lymphoblastic leukemia (ALL) pose significant clinical challenges, often requiring modifications to chemotherapy protocols that may directly affect patient outcomes. In our 10-year retrospective analysis, 12.5% of pediatric ALL patients exhibited clinical signs of cerebrovascular involvement, a figure consistent with the reported incidence range of 3–18.4%. These complications frequently necessitate interruption or alteration of therapy, highlighting the need for adaptive strategies that balance toxicity management with the preservation of treatment efficacy The clinical relevance of these complications stems from their potential to disrupt curative therapy. Despite the protective role of the blood–brain barrier, leukemic cells can infiltrate the central nervous system (CNS) by accumulating at microvascular branching points, leading to cerebrovascular lesions. This pathophysiological mechanism underlies many of the neurological sequelae observed during ALL therapy36,37,38,39,40,41,42. Methotrexate-induced leukoencephalopathy—a well-documented neurotoxic effect—may require replacement with intrathecal cytarabine to limit CNS damage while maintaining antileukemic efficacy43,44,45,46,47. Posterior reversible encephalopathy syndrome (PRES), often associated with agents such as methotrexate, L-asparaginase,
vincristine, and corticosteroids48,49,50,51,52, may paradoxically require reintroduction of corticosteroids to control vasogenic edema and associated neurological symptoms52,53,54,55,56.
Cerebral thrombosis linked to L-asparaginase—reported in 2.1–15% of cases, depending on the study8,57,58,59,60—presents a particular therapeutic dilemma. Although thrombosis frequently necessitates temporary cessation of asparaginase, withholding this agent has been correlated with worse outcomes, particularly in adolescents and young adults (AYA)61. In such cases, substitution with alternative formulations, such as Erwinia chrysanthemi–derived asparaginase, offers a viable approach that allows therapy to continue without compromising efficacy. These examples underscore the importance of maintaining dose intensity while managing toxicity to optimise survival. In our cohort, glucocorticosteroids, vincristine, and methotrexate were the chemotherapeutics most commonly associated with CNS toxicity, consistent with previous literature24,62,63,64,65,66. Methotrexate toxicity often presents with stroke-like symptoms such as hemiparesis or altered consciousness, typically within 13 days post-administration67,68,69. Vincristine, known for inducing neuropathy involving peripheral, autonomic, and occasionally central nervous systems, can necessitate dose reductions that may impair treatment continuity and quality of life. Unfortunately, no reliable clinical predictors of vincristine-induced neuropathy have been identified thus far. L-asparaginase–related CNS complications manifest in various forms, including cerebral hemorrhage (2.1%), thrombosis (15%), and thromboembolic events (4.2%). Sinus thrombosis is most frequently observed during remission induction, although it can occur at later treatment stages. Prolonged seizures have also been documented as a presenting symptom of asparaginase-associated cerebral complications. PRES occurs more commonly in ALL patients (55%) than in those with AML (9%) and is typically characterised by seizures and altered mental status64,65,66. These manifestations highlight the importance of vigilant neurological monitoring during high-risk phases of treatment.
Cerebrovascular complications in patients treated for acute lymphoblastic leukemia (ALL) result from a complex interplay of cancer-associated hypercoagulability, treatment-induced vascular injury, and hematological abnormalities such as hyperleukocytosis and thrombocytopenia. Acute management involves rapid correction of hematological parameters, including platelet transfusions and coagulation factor replacement, to reduce the risk of hemorrhagic and ischemic events. However, anticoagulant or antiplatelet therapies must be used cautiously due to bleeding risks inherent in thrombocytopenic patients. Monitoring minimal residual disease (MRD) is essential not only for relapse risk assessment but also for guiding therapy intensity. Persistent MRD often necessitates intensified chemotherapy, which may prolong exposure to neurotoxic and vasculotoxic agents, thereby indirectly increasing cerebrovascular risk. Therefore, strategies aimed at rapid MRD eradication could reduce cerebrovascular complications by shortening intensive treatment duration. Advances in radiotherapy, such as intensity-modulated radiotherapy (IMRT), allow precise targeting of leukemic sites while sparing healthy brain tissue and vasculature, thus mitigating late radiation-induced vasculopathy characterized by endothelial damage and accelerated atherosclerosis Additionally, hematopoietic stem cell transplantation (HSCT) and hematopoietic growth factors (HGFs) influence endothelial function and blood counts, potentially increasing cerebrovascular risks, which necessitates vigilant neurological monitoring during and after transplantation. There is potential to explore endothelial-protective agents and optimised management of comorbidities to preserve vascular health in ALL patients. Pharmacogenetic profiling may further enable personalised treatment adjustments to minimise neurotoxicity. An integrated, multidisciplinary approach combining hematological monitoring, MRD-guided therapy, advanced radiotherapy techniques, and neuroprotective strategies is critical to improving outcomes and quality of life in ALL patients at risk of cerebrovascular complications5,70,71,72,73. Although computed tomography (CT) remains the first-line imaging modality due to its accessibility, its limited sensitivity, missing up to 75% of hemorrhages—demonstrates the superiority of magnetic resonance imaging (MRI) in detecting ischemic changes and subtle lesions74,75,76,77,78. Age is a known modifier of risk, with children over 10 years more susceptible to CNS toxicity. Notably, however, 72.2% of affected patients in our study were aged 10 years. B-cell ALL (B-ALL) predominated (61.11%), in contrast to findings from some previous reports. While genetic alterations such as the ETV6::RUNX1 fusion were identified, no specific mutations definitively linked to increased CNS toxicity were observed79,80,81. Approximately 80% of acute CNS complications resolve with appropriate intervention, yet mortality remains around 15%. Survivors may experience long-term neurocognitive deficits, including reduced processing speed and impaired attention, which are thought to correlate with caudate nucleus volume depletion82,83. These findings support the need for early and continuous neuropsychological assessment throughout treatment.
This study has several limitations, including its retrospective, single-centre design and relatively small sample size, which may limit the generalizability of the findings. Additionally, the often nonspecific clinical presentation of CNS complications necessitated a strong reliance on neuroimaging for diagnosis. Future multicenter, prospective studies incorporating standardised imaging protocols and biomarker evaluation are needed to better stratify risk and refine therapeutic approaches.
In conclusion, cerebrovascular complications during ALL treatment significantly affect therapy continuation and overall prognosis. Timely recognition and multidisciplinary management are essential. Therapeutic adaptations—such as replacing intrathecal methotrexate with cytarabine in cases of leukoencephalopathy, reintroducing corticosteroids for PRES management, and utilising alternative asparaginase formulations in thrombotic events—can effectively mitigate toxicity while preserving therapeutic efficacy. Close collaboration between hematologists, neurologists, and radiologists is critical to optimising care and improving patient outcomes.
Methods
The study was conducted as a retrospective single-center study. The study group included patients from 0 to 18 years old with acute lymphoblastic leukemia hospitalized in the Department of Pediatric Hematology, Oncology, and Transplantology of the University Children’s Hospital in Lublin. Due to retrospective nature of the study, informed consent was waived by the Bioethics Committee of the Medical University of Lublin (KE-0254/208/10/2022). Statistical analyses were carried out using the statistical 1.8.4.152 PQStat package version. Correlation coefficients of biochemical parameters between the sexes were estimated by means of Pearson’s linear correlation coefficients and Spearman’s monotonic correlation coefficients. The relationship between the sex and symptoms was analyzed using Fisher’s exact test. The association of risk groups with symptoms and radiological changes was analyzed with Fisher’s exact test. Also the association of diagnosis with symptoms and treatment protocol was analyzed with the use of Fisher’s exact test. Similarly, Fisher’s exact test was used to analyze the association of the treatment protocol with symptoms and radiological changes . A test probability p < 0.05 was considered significant and it was judged as highly significant at p < 0.01. This study obtained approval of the Bioethics Committee of the Medical University of Lublin, KE-0254/208/10/2022. In this study, diagnostic methods were applied in accordance with the current Polish standards for the diagnostic management of acute lymphoblastic leukemia in children, as recommended by the Polish Society of Pediatric Oncology and Hematology. Genetic diagnostics primarily relied on conventional cytogenetic analysis, which allowed for the detection of chromosomal abnormalities. Additionally, molecular techniques such as polymerase chain reaction (PCR) and fluorescence in situ hybridization (FISH) were employed to identify characteristic translocations and genetic mutations relevant for prognosis and treatment decisions. All testing was performed following national guidelines to ensure consistency, reliability, and appropriate clinical interpretation of the results.
Data availability
All data generated or analyzed during this study are included in this published article. The datasets supporting the conclusions of this article are available within the article. No additional data are available. Should any further clarification or raw data be required, they can be made available by the corresponding author upon reasonable request.
References
Childhood cancer: Estimating regional and global incidence. Cancer Epidemiology [Internet]. 71:101662. Available from: https://www.sciencedirect.com/science/article/pii/S1877782119301729?via%3Dihub (2021).
Baytan, B., Evim, M. S., Güler, S., Güneş, A. M. & Okan, M. Acute central nervous system complications in pediatric acute lymphoblastic leukemia. Pediatr. Neurol. 53(4), 312–318 (2015).
Hamoda A, Bedair M, Semary SF, Youssef A, Hafez H. Patterns and outcomes of acute central nervous system complications during treatment of childhood acute lymphoblastic leukemia: a single-center experience. J. Pediatr. Hematol./Oncol. [Internet]. 2024 Jan;46(2):72–9. Available from: https://pubmed.ncbi.nlm.nih.gov/38132573/
Parasole, R. et al. Central nervous system complicationsduring treatment of acute lymphoblastic leukemia in a single pediatric institution. Leuk Lymphoma 51(6), 1063–1071 (2010).
Anastasopoulou, S. et al. Acute central nervous system toxicity during treatment of pediatric acute lymphoblastic leukemia: Phenotypes, risk factors and genotypes. Haematologica 107(10), 2318 (2022).
Chen CY, Zimmerman RA, Faro S, Bilaniuk LT, Chou TY, Molloy PT. Childhood leukemia: Central nervous system abnormalities during and after treatment. AJNR American journal of neuroradiology [Internet]. 1996 Feb;17(2):295–310. https://pubmed.ncbi.nlm.nih.gov/8938302/
Walker, R.W. Neurologic complications of leukemia. Neurologic clinics [Internet]. 1991 Nov;9(4):989–99. https://pubmed.ncbi.nlm.nih.gov/1758436/
Feinberg, W.M. & Swenson, M.R. Cerebrovascular complications of L-asparaginase therapy. Neurology [Internet]. 1988 Jan;38(1):127–33. https://pubmed.ncbi.nlm.nih.gov/3275903/
Biti, G.P., Magrini, S.M., Villari, N., Caramella, D., Guazzelli, G., Rosi, A., et al. Brain damage after treatment for acute lymphoblastic leukemia. A report on 34 patients with special regard to MRI findings. Acta oncologica (Stockholm, Sweden) [Internet]. 1989;28(2):253–6. https://pubmed.ncbi.nlm.nih.gov/2736115/
Flament-Durand, J., Ketelbant-Balasse, P., Maurus, R., Regnier, R. & Spehl, M. Intracerebral calcifications that appear during the course of acute lymphocytic leukemia treated with methotrexate and X-rays. Cancer 35(2), 319–325 (1975).
Snyder, E. J., Sarma, A., Poussaint, T. Y., Krishnasarma, R. & Pruthi, S. Complications of cancer therapy in children: A comprehensive review of neuroimaging findings. J. Comput. Assist. Tomogr. 47(5), 820–832 (2023).
AlGhamdi, A. S. Novel ensemble model recommendation approach for the detection of dyslexia. Children 9(9), 1337 (2022).
Ochs JJ, Bowman WP, Pui CH, Abromowitch M, Mason C, Simone JV. Seizures in childhood lymphoblastic leukaemia patients. Lancet (London, England) [Internet]. 1984 Summer;2(8417–8418):1422–4. https://pubmed.ncbi.nlm.nih.gov/6151043
Ghali, M. G. Z., Davanzo, J., Leo, M. & Rizk, E. Posterior reversible encephalopathy syndrome in pediatric patients: Pathophysiology, diagnosis, and management. Leuk. Lymphoma 60(10), 2365–2372 (2019).
Zama, D. et al. A survey on hematology-oncology pediatric AIEOP centres: The challenge of posterior reversible encephalopathy syndrome. Eur. J. Haematol. 100(1), 75–82 (2017).
Vora, A. Childhood acute lymphoblastic leukemia (Springer, 2017).
Bond, J. et al. Stroke-like syndrome caused by intrathecal methotrexate in patients treated during the UKALL 2003 trial. Leukemia 27(4), 954–956 (2012).
Bhojwani, D. et al. Methotrexate-induced neurotoxicity and leukoencephalopathy in childhood acute lymphoblastic leukemia. J. Clin. Oncol. 32(9), 949–959 (2014).
Ranta, S. et al. Cerebral sinus venous thromboses in children with acute lymphoblastic leukaemia - a multicentre study from the Nordic Society of Paediatric Haematology and Oncology. Br. J. Haematol. 168(4), 547–552 (2014).
Ozturk, K., Rykken, J. & McKinney, A. M. Pediatric acute toxic leukoencephalopathy: Prediction of the clinical outcome by FLAIR and DWI for various etiologies. Am. J. Neuroradiol. 41(8), 1517–1524 (2020).
Zadeh, C., AlArab, N., Muwakkit, S., Atweh, L.A., Tamim, H., Makki, M., et al. Stroke in Middle Eastern children with cancer: Prevalence and risk factors. BMC neurology [Internet].22(1):31.: https://pubmed.ncbi.nlm.nih.gov/35042459/ (2022)
Intusoma, U., Nakorn, C. N. & Chotsampancharoen, T. Intracranial hemorrhage in childhood acute Leukemia: Incidence, characteristics, and contributing factors. Pediatr. Neurol. 99, 23–30 (2019).
Rahiman, E.A., Rajendran, A., Sankhyan, N., Singh, P., Muralidharan, J., Bansal, D., et al. Acute neurological complications during acute lymphoblastic leukemia therapy: A single-center experience over 10 years. Indian journal of cancer [Internet]. 58(4):545–52. https://pubmed.ncbi.nlm.nih.gov/34380827/ (2021).
Peddi, P. F., Peddi, S., Santos, E. S. & Morgensztern, D. Central nervous system toxicities of chemotherapeutic agents. Expert Rev. Anticancer Ther. 14(7), 857–863 (2014).
How, J., Blattner, M., Fowler, S., Wang-Gillam, A. & Schindler, S. E. Chemotherapy-associated posterior reversible encephalopathy syndrome. Neurologist 21(6), 112–117 (2016).
Millan, N. C. et al. Acute and sub-acute neurological toxicity in children treated for acute lymphoblastic leukemia. Leuk. Res. 65, 86–93 (2018).
Vagace, J. M. et al. Methotrexateinduced subacute neurotoxicity in a child with acute lymphoblastic leukemia carrying genetic polymorphisms related to folate homeostasis. Am. J. Hematol. 86(1), 98–101 (2010).
Boston Collaborative Drug Surveillance Program. Acute adverse reactions to prednisone in relation to dosage. Clin. Pharmacol. Therapeut. 13(51), 694–698 (1972).
Wolkowitz, O. Prospective controlled studies of the behavioral and biological effects of exogenous corticosteroids. Psychoneuroendocrinology 19(3), 233–255 (1994).
Lavoie, E. M. et al. Patterns and severity of vincristine-induced peripheral neuropathy in children with acute lymphoblastic leukemia. J. Peripher. Nerv. Syst. 20(1), 37–46 (2015).
Moghrabi, A., Levy, D.E., Asselin, B., Barr, R., Clavell, L., Hurwitz, C., et al. Results of the Dana-Farber Cancer Institute ALL Consortium Protocol 95-01 for children with acute lymphoblastic leukemia. Blood [Internet]. [cited 2022 Sep 28];109(3):896–904. https://ashpublications.org/blood/article/109/3/896/23859/Results-of-the-Dana-FarberCancer-Institute-ALL (2006)
Peng, C. T. et al. Amino acid concentrations in cerebrospinal fluid in children withacute lymphoblastic leukemia undergoing chemotherapy. Eur. J. Cancer 41(8), 1158–1163 (2005).
Gruenbaum, S. E. et al. Branched-chain amino acids and seizures: A systematic review of the literature. CNS Drugs 33(8), 755–770 (2019).
Herzig, R. H. et al. Cerebellar toxicity with high-dose cytosine arabinoside. J. Clin. Oncol. 5(6), 927–932 (1987).
Salinsky, M. C., Levine, R. L., Aubuchon, J. P. & Schutta, H. S. Acute cerebellar dysfunction with high-dose ARA-C therapy. Cancer 51(3), 426–429 (1983).
Yao, H. et al. Leukaemia hijacks a neural mechanism to invade the central nervous system. Nature 560(7716), 55–60 (2018).
Ray, M., Marwaha, R. K. & Trehan, A. Chemotherapy related fatal neurotoxicity during induction in acute lymphoblastic leukemia. Ind. J. Pediatr. 69(2), 185–187 (2002).
Mahoney, D. H. et al. Acute neurotoxicity in children with B-precursor acute lymphoid leukemia: An association with intermediate-dose intravenous methotrexate and intrathecal triple therapy–a Pediatric Oncology Group study. J. Clin. Oncol. 16(5), 1712–1722 (1998).
Kuskonmaz, B. et al. The neurologic complications in pediatric acute lymphoblastic leukemia patients excluding leukemic infiltration. Leuk. Res. 30(5), 537–541 (2006).
Aytaç, S., Yetgin, S. & Tavil, B. Acute and long-term neurologic complications in children with acute lymphoblastic leukemia. The Turkish J. Pediatr. 48(1):1–7. https://pubmed.ncbi.nlm.nih.gov/16562778/ (2006).
Nigro, L. L., Cataldo, A. D. & Schiliro, G. Acute neurotoxicity in children with B-lineage acute lymphoblastic leukemia (B-ALL) treated with intermediate risk protocols. Med. Pediatr. Oncol. 35(5), 449–455 (2000).
Kamath, N., Sankhe, S. & Sahu, A. A rare case of chemotherapy induced reversible cerebral vasoconstriction syndrome in a patient of acute lymphocytic leukemia. J Canc Res Therapeut 11(4), 1012–1012e (2015).
Avila, J. D. Methotrexate-induced neurotoxicity: An uncommon stroke mimic. The Neurohospitalist 8(1), 46–47 (2017).
Hafez, H. A., Soliaman, R. M., Bilal, D., Hashem, M. & Shalaby, L. M. Early deaths in pediatric acute leukemia: A major challenge in developing countries. J. Pediatr. Hematol. Oncol. 41(4), 261–266 (2019).
Maytal, J. et al. Prognosis and treatment of eizures in children with acute lymphoblastic leukemia. Epilepsia 36(8), 831–836 (1995).
Shin, R. K., Stern, J. W., Janss, A. J., Hunter, J. A. & Liu, G. R. Reversible posterior leukoencephalopathy during the treatment of acute lymphoblastic leukemia. Neurology 56(3), 388–391 (2001).
Pratt, G. et al. Cyclosporin neurotoxicity after chemotherapy. BMJ 319(7201), 54–64 (1999).
Wong, R. et al. Tacrolimus-associated posterior reversible encephalopathy syndrome after allogeneic haematopoietic stem cell transplantation. Br. J. Haematol. 122(1), 128–134 (2003).
Rogers, P., Pan, W. J., Drachtman, R. A. & Haines, C. A stroke Mimic: Methotrexate-induced neurotoxicity in the emergency department. J. Emerg. Med. 52(4), 559–561 (2017).
Hualde Olascoaga, J. et al. Leucoencefalopatía posterior reversible: Descripción de dos casos asociados a vincristina. An Pediatr (Barc) 68(3), 282–285 (2008).
Banerjee, J. et al. The spectrum of acute central nervous system symptoms during the treatment of childhood acute lymphoblastic leukaemia. Pediatr. Blood Canc. 67(2), e2799 (2019).
Vora, A. et al. Treatment reduction for children and young adults with low-risk acute lymphoblastic leukaemia defined by minimal residual disease (UKALL 2003): A randomised controlled trial. Lancet Oncol. 14(3), 199–209 (2013).
Schmiegelow, K. et al. Consensus definitions of 14 severe acute toxic effects for childhood lymphoblastic leukaemia treatment: A Delphi consensus. Lancet Oncol. 17(6), e231–e239 (2016).
Ando, Y., Ono, Y., Sano, A., Fujita, N. & Ono, S. Posterior reversible encephalopathy syndrome: A review of the literature. Intern. Med. 61(2), 135–141 (2022).
Li, R. et al. Clinical analysis of childhood acute lymphoblastic leukemia with epilepsy seizures. Front. Neurol. 13, 824268 (2022).
Fleischhack, G. et al. Bildgebende Verfahren in der Diagnostik zerebrovaskulärer Komplikationen unter L-Asparaginase-Therapie. Klin. Padiatr. 206(4), 334–341 (1994).
Nowak-Göttl, U. et al. Enhanced thrombin generation, P-von willebrand factor, P-fibrin D-dimer and P-plasminogen activator inhibitor 1: Predictive for venous thrombosis in asparaginasetreated children. Fibrinol. Proteol. 8, 63–65 (1994).
Corso, A., Castagnola, C. & Bernasconi, C. Thrombotic events are not exclusive to the remission induction period in patients with acute lymphoblastic leukemia: A report of two cases of cerebral sinus thrombosis. Ann. Hematol. 75(3), 117–119 (1997).
Wong, R. et al. Tacrolimus-associated posterior reversible encephalopathy syndrome after allogeneic haematopoietic stem cell transplantation. Br. J. Haemat. 122(1), 128–134 (2003).
Sánchez-Carpintero, R., Narbona, J., López de Mesa, R., Arbizu, J. & Sierrasesúmaga, L. Transient posterior encephalopathy induced by chemotherapy in children. Pediatr. Neurol. 24(2), 145–148 (2001).
Gupta, S. et al. Impact of asparaginase discontinuation on outcome in childhood acute lymphoblastic leukemia: A report from the children’s oncology group. J. Clin. Oncol. 38(17), 1897–1905 (2020).
Ocaña Rico, M., Javier Manchón, G., Coroleu Lletget, W. & Capellades, F. J. Encefalopatía posterior reversible durante el tratamiento de inducción de la leucemia linfoblástica aguda. Med. Clin. 119(20), 795–796 (2002).
Bansal, R. et al. Progression of brain injuries associated with methotrexate chemotherapy in childhood acute lymphoblastic leukemia. Pediatr. Res. 97(1), 348–359 (2025).
Appachu, M. A., Purohit, S., Lakshmaiah, K. C., Aruna, B. K. & Appaji, L. Posterior reversible encephalopathy syndrome in pediatric acute leukemia: Case series and literature review. Indian J. Med. Paediatr. Oncol. 35(1), 79–82 (2014).
Musioł, K. et al. PRES in the course of hemato-oncological treatment in children. Child’s Nerv. Syst. 34(4), 691 (2017).
Rogers, P., Pan, W. J., Drachtman, R. A. & Haines, C. A stroke mimic: Methotrexate-induced neurotoxicity in t6he emergency department. J. Emerg. Med. 52(4), 559–61.43 (2017).
Watanabe, K. et al. Characteristics of methotrexate-induced stroke-like neurotoxicity. Int. J. Hematol. 108(6), 630–636 (2018).
Antunes, N. L. Seizures in children with systemic cancer. Pediatr. Neurol. 28(3), 190–193 (2003).
Madsen, M. L. et al. Aspects of vincristine-induced neuropathy in hematologic malignancies: A systematic review. Canc. Chemother. Pharmacol. 84(3), 471–485 (2019).
Faber, K. et al. Neurological toxicities among children patients during treatment for acute lymphoblastic leukemia and occurrence of neuropsychological late effects after the treatment. Psychoonkologia 18(4), 144–152 (2014).
Krasinka, J., Trelinska, J., Walenciak, J., Dachowska-Kałwak, I. & Młynarski, W. Central neurotoxicity ´ as a complication in course of treatment of acute lymphoblastic leukemia in children: A single center experience. Acta Haematol. Pol. 54(3), 145–153 (2023).
Mizutani, T. et al. Radiation-induced vascular injury and its management in cancer patients. J. Neurooncol. 108(3), 423–432 (2012).
Rahiman, E. A. et al. Acute neurological complications during acute lymphoblastic leukemia therapy: A single-center experience over 10 years. Ind. J. Canc. 58(4), 545–552 (2021).
Kieslich, M. et al. Cerebrovascular complications of l-Asparaginase in the therapy of acute lymphoblastic leukemia. J. Pediatr. Hematol. Oncol. 25(6), 484–487 (2003).
Zajac-Spychała, O. et al. Long-term brain structural magnetic resonance imaging and cognitive functioning in children treated for acute lymphoblastic leukemia with high-dose methotrexate chemotherapy alone or combined with CNS radiotherapy at reduced total dose to 12 Gy. Neuroradiology 59(2), 147–156 (2017).
Tsai, P. F. et al. Hippocampal dosimetry correlates with the change in neurocognitive function after hippocampal sparing during whole brain radiotherapy: A prospective study. Radiat. Oncol. 10(1), 253 (2015).
Monje, M. et al. Functional and structural differences in the hippocampus associated with memory deficits in adult survivors of acute lymphoblastic leukemia. Pediatr. Blood Canc. 60(2), 293–300 (2012).
Seigers, R. et al. Long-lasting suppression of hippocampal cell proliferation and impaired cognitive performance by methotrexate in the rat. Behav. Brain Res. 186(2), 168–175 (2008).
Oskarsson, T. et al. Relapsed childhood acute lymphoblastic leukemia in the Nordic countries: Prognostic factors, treatment and outcome. Haematologica 101(1), 68–76 (2015).
Sliwa-Tytko, P., Kaczmarska, A., Lejman, M. & Zawitkowska, J. Neurotoxicity associated with treatment of Acute ´ lymphoblastic leukemia chemotherapy and immunotherapy. Int. J. Mol. Sci. 23(10), 5515 (2022).
Mateos, M. K. et al. Methotrexate-related central neurotoxicity: Clinical characteristics, risk factors and genome-wide association study in children treated for acute lymphoblastic leukemia. Haematologica [Internet]. 107(3), 635–643 (2022).
Wardlaw, J. M., Keir, S.L., & Dennis, M.S. The impact of delays in computed tomography of the brain on the accuracy of diagnosis and subsequent management in patients with minor stroke. J. Neurol., Neurosurg. Psychiatry [Internet]. 74(1):77–81. https://jnnp.bmj.com/content/74/1/77 (2003).
Siddiqui, F.M., Bekker, S.V. & Qureshi, A.I. Neuroimaging of hemorrhage and vascular defects. Neurotherapeut: J. Am. Soci. Exper. NeuroTherapeut [Internet]. 8(1):28–38. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3075731/ (2011).
Funding
This study received no external funding, they can be made available by the corresponding author upon reasonable request.
Author information
Authors and Affiliations
Contributions
A.R., M.L. (Maria Lesniak), P.M., J.L. and M.S. contributed to the study concept and design, analysis and interpretation of data, and drafting of manuscript; J.Z., M.L. (Monika Lejman) and M.W. contributed to the study concept and design, interpretation of data, and critical revision of manuscript for important intellectual content. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration 105 of Helsinki, and approved by the Bioethics Committee of the Medical University of Lublin KE-0254/208/10/2022.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rocka, A., Lesniak, M., Majka, P. et al. Cerebrovascular complications in pediatric patients with acute lymphoblastic leukemia. Sci Rep 15, 40825 (2025). https://doi.org/10.1038/s41598-025-24634-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-24634-z