Abstract
Tick-borne spotted fever group (SFG) rickettsioses are considered emerging vector-borne infectious diseases with potentially severe consequences for public health, particularly in the temperate regions of Europe. The aim of the study was to determine the prevalence and the diversity of Rickettsia spp. in questing Ixodes ricinus ticks in north-eastern Poland, evaluate the serological evidence of exposure to SFG rickettsiae among adult residents of this region, as well as to determine the level of SFG rickettsiae co-infection with Borrelia spirochaetes in ticks and humans. In a total of 4090 ticks, the presence of Rickettsia spp. was detected by PCR and confirmed by sequencing. IgG antibodies against SFG Rickettsia and Borrelia were detected using ELISA. Rickettsial DNA was revealed in 11.3% of females, 11.8% of males and nymphs of 6.5% (MIR, minimum infection rate). Based on DNA sequencing, R. helvetica and R. monacensis were identified. IgG antibodies against SFG rickettsiae were confirmed in 38.7% of adult residents of north-eastern Poland. The co-infection rate of SFG rickettsiae with Borrelia spirochaetes in questing adult I. ricinus ticks was low (2.6%), compared to the co-infection rate (16.1%) observed based on the presence of immunological responses in the study group of residents from northeastern Poland. These findings provide important evidence filling the gap between the environmental detection of SFG rickettsiae in vector populations and the limited number of diagnosed clinical cases in humans.
Similar content being viewed by others
Introduction
The rickettsiae are obligate, intracellular Gram-negative bacteria, transmitted via hematophagous ectoparasitic arthropod vectors such as ticks, fleas, lice or mites from wild or domestic vertebrate hosts1. Within the genus Rickettsia, one of the most numerous groups classified based on phenotypic characteristics, vector hosts and phylogenetic organization is the spotted fever group (SFG), which is almost exclusively tick-borne2. In recent decades, the medical importance of the Rickettsia species transmitted by ticks has increased considerably3,4. Among those, species such as R. rickettsii and R. conorii are causative agents of the oldest known tick-borne diseases, Rocky Mountain Spotted Fever and Mediterranean Spotted Fever, respectively5. Many other Rickettsia spp. from SFG, considered to be nonpathogenic for decades, such as R. helvetica5, R. monacensis6 or R. slovaca7, are now associated with human infections. Moreover, novel species (i.e. Candidatus R. mendelii8, Candidatus R. thierseensis9) of undetermined pathogenicity continue to be detected in or isolated from ticks around the world10,11,12.
SFG rickettsioses, like other tick-borne diseases, belong to the emerging and re-emerging infectious diseases10,13,14. They are considered some of the most rapidly expanding vector-borne diseases, with potentially severe consequences for public health, particularly in the temperate regions of Europe15,16. In humans, spotted fever rickettsioses do not have pathognomonic features; however, the typical clinical symptoms include flu-like signs (fever, headache, muscle pain), skin manifestations (rash or a characteristic inoculation eschar ‘tâche noire’ at the site of the tick bite), as well as in some cases local lymphadenopathy. These major clinical signs vary depending on the rickettsial species involved and may be absent or unnoticed during an undirected clinical examination10,17.
The risk of rickettsioses in a given region may depend on the presence of specific tick species and tick population density, the geographical and climatic conditions, as well as the duration of tick attachment to the host and human activity5. The main vectors and reservoirs of SFG rickettsiae are hard ticks from the Ixodidae family12. In Europe, a significant role in the circulation cycle of SFG rickettsiae is attributed to the most abundant tick species, Ixodes ricinus, Dermacentor reticulatus and Rhipicephalus sanguineus (in Mediterranean countries)13,18,19. Monitoring the level of tick infection with pathogens as Borrelia spirochetes or microorganisms potentially pathogenic to humans, as well as conducting serological investigations of the general population or selected healthy groups, is one of the key elements in assessing the circulation of tick-borne pathogens and the risk of tick-borne diseases in a given area20. Such monitoring is particularly important for SFG Rickettsia. It is assumed that this group of microorganisms will continue to evolve and should be closely monitored by data collected from properly designed field surveys of vectors and animal hosts, periodic serosurveys in healthy populations, and improved surveillance systems for human cases4.
The aims of the study were: (1) to assess the prevalence of Rickettsia spp. in questing I. ricinus ticks in north-eastern Poland; (2) to determine the species diversity of the Rickettsia genus in the study area, examining the impact of subregional conditions, biotope, and year of study on the frequency of Rickettsia spp.; (3) to assess contact with SFG rickettsiae among adult residents of the north-eastern region of Poland based on serological investigations; and (4) to determine the level of SFG Rickettsia co-infection with Borrelia spirochetes in ticks and humans.
Materials and methods
Molecular detection of the spotted fever group rickettsiae in ticks
Study area and tick collection
Rickettsia spp. were examined in a total of 4,090 I. ricinus ticks, including 381 females, 450 males, and 3,259 nymphs (in 651 pools of five and one pool of four specimens). Questing ticks were collected between April and June of 2016 and 2017 at 14 sites located in the western, central, and eastern subregion of the Warmia-Mazury province in north-eastern Poland (Fig. 1). The tick collection sites represented forest landscapes and ecotone habitats (zones between grassy and forested areas). At each site, ticks were collected twice per month in each year of the study during the daytime between 9 a.m. and 4 p.m. by two persons for at least 30 min using the standard flagging method21. Ticks were preserved in 70% ethanol. In the laboratory, species, sex (female, male) and developmental stage (nymph, adult) of collected ticks were morphologically identified based on a taxonomic key22. Subsequently, adult ticks were stored individually, and nymphs in pools at − 80°C for further molecular analyses.
Ixodes ricinus tick collection sites located in the Warmia-Mazury province (north-eastern Poland). The colors indicate the western, central, and eastern subregions of the Warmia-Mazury province, from left to right, respectively. Geographical coordinates and detailed descriptions of the habitats of the tick collection sites were published in Kubiak et al.27[. The map was created based on Google Maps (https://www.google.pl/maps) in CorelDRAWX5 software.
DNA extraction
Genomic DNA from I. ricinus ticks was isolated using the universal Sherlock AX kit (A&A Biotechnology, Gdynia, Poland) according to the manufacturer’s protocol. DNA was extracted from individual adult ticks and from pools of nymphs and then frozen at − 80°C for further analysis. The procedure was described in detail in Kubiak et al.27.
The presence of Rickettsia spp. DNA in tick genomic DNA samples was confirmed by the conventional PCR method using two sets of primers: (a) CS409/Rp125823 - specific to the citrate synthase (gltA) gene and (b) ITS-F/ITS-R24 - specific to the intergenic spacer region (ITS) between the 23 S rRNA and 5 S rRNA genes. Target regions, expected length of the PCR products and the oligonucleotide sequences of primers are detailed in Table 1.
The total volume of the PCR mixture (25 µL) contained: 12.5 µL of 2 × PCR Master Mix Plus (0.1 U/µL of Taq polymerase supplied in a PCR buffer, 4 mM of MgCl2 and 0.5 mM of each dNTP) (A&A Biotechnology, Gdynia, Poland), 0.5 µL of each primer (10 µM), 1.5/2 µL of template DNA and an appropriate volume of sterile nuclease-free water. Sequence-verified DNA of R. helvetica, obtained from an infected I. ricinus tick, as a positive control was used. PCR was performed on a Mastercycler®nexus GSX1 thermocycler (Eppendorf AG, Hamburg, Germany). PCR amplicons were analysed by electrophoresis in 1.5% agarose gels stained with Midori Green Stain (Nippon Genetics Europe, Düren, Germany). The PCR products of approximately 769 bp of the gltA gene23 and between 300 and 533 bp of the rrl-rrf ITS region24 were considered Rickettsia-positive.
Rickettsia species identification
The CleanUp purification kit (A&A Biotechnology, Gdynia, Poland) was used to purify randomly selected PCR products (46 for the gltA gene and 4 for the rrl-rrf ITS). The amplicons were then bidirectionally sequenced by Macrogen Europe (Amsterdam, The Netherlands). The nucleotide sequences obtained in this study were manually edited using BioEdit, v. 7.2 software25 (https://bioedit.software.informer.com, accessed July 9, 2024), and then compared using the Basic Local Alignment Search Tool (http://www.ncbi.nlm.nih.gov/BLAST/, accessed on 20 July 2024) to nucleotide sequences available in the GenBank database (http://www.ncbi.nih.gov/Genbank/index.html, accessed July 20, 2024). Representative sequences of the Rickettsia gltA gene and rrl-rrf ITS were registered in the GenBank database.
Phylogenetic analysis
Nucleotide sequences of the gltA gene (GenBank: PQ119728-729) obtained in this study were used to demonstrate the molecular relationship between the identified Rickettsia species and reference sequences in the GenBank database. The phylogram was constructed using the Maximum Likelihood method based on the Kimura 2-parameter model. The topology of the phylogenetic tree was evaluated using the bootstrap method with 1,000 replicates. Phylogenetic analysis was conducted using MEGA 11 software26 (https://www.megasoftware.net, accessed on 1 August 2024).
Coinfections of Rickettsia spp. with Borrelia burgdorferi s.l. in ticks
Additionally, co-infections of Rickettsia spp. with Borrelia spirochaetes were also analysed in the same tick DNA samples. Data on the prevalence of spirochaetes (B. burgdorferi s.l. and B. miyamotoi) in I. ricinus ticks from the same study sites and years were published by Kubiak et al.27. Due to the fact that the level of nymph infection was studied in pools of specimens, the analysis of co-infections was conducted only among adults of I. ricinus.
IgG seroreactivity to spotted fever group rickettsiae and Borrelia spirochaetes in humans
Selection and recruitment of participants
The material for the serological studies consisted of 155 serum samples collected in 2023–2024 from two groups of adult residents (≥ 18 years) of the north-eastern region of Poland, with varying degrees of exposure to tick bites. The high-risk group (HR) consisted of 102 forest workers (22 females, 80 males) from two forest inspectorates: Jedwabno (n = 61) and Olsztynek (n = 41) in the Warmia-Mazury province. The low-risk group (LR) consisted of 53 volunteers (39 females, 14 males), residents of the Olsztyn city area, who occasionally came into contact with tick-infested environments as part of their recreational activities. The qualification for the LR group was carried out based on an original survey questionnaire, which provided information on socio-demographic data, history of tick bites, as well as previously diagnosed borreliosis or other tick-borne diseases. All study participants had no history of Lyme disease.
Collected serum samples were used to test for the presence of specific IgG antibodies for SFG rickettsiae and Borrelia spirochaetes in each sample.
Blood sampling and serological tests
Blood sera (~ 2 ml) were obtained by centrifugation of venous blood collected in a closed system under the applicable procedures by qualified medical personnel of the Epidemiological and Clinical Research Laboratory, the Voivodeship Sanitary and Epidemiological Station in Olsztyn, the Medical Diagnostic Laboratory of the Clinical Hospital of the Ministry of Internal Affairs and the Administration of the Warmia-Mazury Oncology Centre in Olsztyn, for both the HR and LR group. All serum samples were stored at −20°C until further analysis.
To detect IgG antibodies to antigens of SFG rickettsiae, the Rickettsia IgG (Spotted Fever Group) ELISA kit (DRG International Inc., Springfield, New Jersey, USA) was used. The assays were performed according to the manufacturer’s instructions. The cut-off calibrator was set at an index of 1.0 to provide for discrimination between reactive and non-reactive sera. The index from above 1.1 was considered to be positive and below 0.9 was considered to be negative for Rickettsia spp. An index between 0.9 and 1.1 was considered equivocal.
The presence of anti-B. burgdorferi s.l. IgG antibodies were examined with the use of the Borrelia IgG ELISA Recombinant Antigen kit (Biomedica Laboratories, Vienna, Austria). The test was conducted in accordance with the manufacturer’s instructions. Positive or negative results were determined by the manufacturer’s recommendation.
Serological tests were conducted and interpreted by qualified laboratory diagnosticians at the Epidemiological and Clinical Research Laboratory of the Voivodeship Sanitary and Epidemiological Station in Olsztyn.
Ethics approval and consent to participate
The study protocol followed the ethical guidelines of the 2013 Declaration of Helsinki. Each person, both from the HR and LR groups, voluntarily expressed their willingness to participate and consented to the use of biological material in this study. The study was approved by the Bioethics Committee of the University of Warmia and Mazury in Olsztyn (No. 35/2024).
Statistical analysis
A chi-square (chi2, χ²) test (with post-hoc Bonferroni test) and 95% confidence intervals (95% CI) were used to compare Rickettsia infection rates according to the developmental stage of ticks, subregions, habitats, and study years. Rickettsia spp. prevalence in nymphs (tested in pools) was presented as the Minimum Infection Rate (MIR). MIR was calculated as the percentage obtained by dividing the number of Rickettsia-positive pools by the total number of nymphs tested, assuming the presence of only one infected nymph per positive pool. The seroprevalence of Rickettsia and Borrelia was statistically compared within the human study group (HR vs. LR groups, women vs. men, and the Jedwabno vs. Olsztynek forest inspectorates) based on the chi-square (chi2, χ²) test and 95% confidence intervals (95% CI). In the analysis, equivocal serological results for the tested pathogens were classified as negative. Statistical analyses were performed using SPSS software, v. 27.0 for Windows (SPSS Inc., Chicago, IL, USA). A p-value of < 0.05 was considered statistically significant in all analyses.
Results
Prevalence of Rickettsia spp. In ticks
The presence of Rickettsia DNA was confirmed in 7.5% (308/4090) of the tested isolates. Rickettsia spp. was detected in at least 6.5% (MIR, minimum infection rate) of I. ricinus nymphs, 11.3% (43/381) of females and 11.8% (53/450) of males (χ²=24.296, p < 0.001) (Table 2). The proportion of Rickettsia-positive ticks was significantly higher in 2016 (8.4%, 183/2188) than in 2017 (6.6%, 125/1902) (χ²=4.269, p = 0.03). The highest infection rate (9.5%, 129/1351) was recorded in the central subregion of Warmia and Mazury. The type of habitat did not affect the Rickettsia spp. prevalence, which was 7.3% (127/1750) in forest areas and 7.7% (181/2340) in ecotones.
Rickettsia species diversity in ticks
To confirm Rickettsia species, 46 (15%) randomly selected PCR amplicons of the fragment of gltA gene (~ 730 bp) were sequenced and compared with GenBank accessions. Sequence analysis showed the presence of R. helvetica and R. monacensis (our GenBank submission: PQ119728 and PQ119729, respectively). Both Rickettsia species are known as human pathogens. The phylogenetic relationships between the Rickettsia species identified in this study and reference accessions from GenBank are presented in the Fig. 2. All obtained nucleotide sequences of R. helvetica (n = 45) were similar and showed 100% identity to the isolate C9P9 of R. helvetica (GenBank: U59723). The same nucleotide sequence was also detected from I. ricinus in north-eastern (GenBank: OM927745) and central (GenBank: MH018978) Poland as well as from Serbia (GenBank: MH618386) and Italy (GenBank: MN226407) (Fig. 2). One obtained sequence identified as R. monacensis showed 100% identity with gltA sequences of the R. monacensis strain IrR/Munich from Germany (GenBank: LN794217) and from questing I. ricinus ticks from north-eastern (GenBank: MW595238) and central Poland (GenBank: MH018982) (Fig. 2).
Length analysis of the rrl-rrf ITS PCR products showed the presence of amplificons of ~ 530 bp in 259 Rickettsia-positive DNA samples and ~ 350 bp in three samples. Such lengths of ITS fragments are characteristic for R. helvetica and R. monacensis , respectively. In order to confirm the specificity of PCR-screening, a randomly selected PCR product corresponding to R. helvetica and all sequences corresponding to R. monacensis were sequenced and deposited in GenBank under the accession numbers PV528813 and PV528814, respectively. The 505 bp sequence (our submission: PV528813) was 100% identical to the sequence of isolate BO144 of R. helvetica (GenBank: OZ018776) and R. helvetica previously identified in I. ricinus ticks in Poland (GenBank: JQ796866) and Austria (GenBank: EU057990). The nucleotide sequences of PCR products initially identified as R. monacensis were monomorphic (our submission: PV528814) and showed 100% identity to the IrR/Munich strain of R. monacensis (GenBank: LN794217) and sequences isolated from I. ricinus in Poland (GenBank: JQ796867), Bosnia and Herzegovina (GenBank: PV231331) and Switzerland (GenBank: JQ670878).
Phylogenetic relationships between Rickettsia species identified in the study and accessions from GenBank, based on the sequences of the gltA gene. The phylogram was constructed with MEGA 11 software (https://www.megasoftware.net) using the Maximum Likelihood method and the Kimura 2-parameter method as a distance method. The percentage of replicate trees in which the associated taxa are clustered together in the bootstrap test (1,000 replicates) is shown next to the branches. The tree is drawn to scale, with branch lengths measured in the number of base substitutions per site. The sequences obtained in this study are labelled with color symbols.
Rickettsia spp. and Borrelia spirochaete co-infections in adult ticks
In a previous study27, an infection rate of Borrelia spirochaetes in adult I. ricinus ticks was 18.2% (151/831) and was higher compared to the level of Rickettsia spp. infection 11.6% (96/831) (Table 2). A total of 27.1% (225/831) of adult I. ricinus ticks tested positive for Rickettsia spp. and/or B. burgdorferi s.l. Co-infections with both bacteria were detected in 2.6% (22/831) of the examined ticks, which was significantly less frequent compared to non-infections and mono-infections (χ²=645.28, p < 0.001) (Table 3). The co-infection rate in females was 2.6% (10/381) and did not differ significantly from that in males, which was 2.7% (12/450). In the I. ricinus population from the central part of the Warmia-Mazury province, the level of co-infection was significantly higher compared to the tick population from the western part, with rates of 4.7% (13/279) and 0.5% (1/208), respectively (Table 3). The co-infection rate of Rickettsia spp. and Borrelia spirochaetes in adult I. ricinus ticks was not significantly influenced by either the year of the study or the habitat (Table 3).
Rickettsia spotted-fever group seroprevalence in humans
Of the 155 serum samples tested with ELISA, positive results of IgG antibodies against Rickettsia spp. were found in 38.7% (60/155) of the total human study group (Table 4). The overall seroprevalence was significantly different between men 56.4% (53/94) and women 11.5% (7/61) (χ2 = 31.445, p < 0.001). The IgG seroprevalence for Rickettsia SFG was significantly higher in the HR group (forestry workers) than the LR group (city residence), 52.9% (54/102) and 11.3% (6/53) (χ2 = 25.465, p < 0.001), respectively (Table 4). In both examined groups the positive results were significantly higher in men (61.3% - HR and 28.6% - LR) than women (22.7% - HR and 5.1% - LR) (Table 4). Among forestry workers (HR), no significant differences in Rickettsia-positive results between Jedwabno (55.7%) and Olsztynek (48.8%) (χ2 = 0.476, p = 0.490) forest inspectorates were noted.
Borrelia spirochaetes seroprevalence in humans
The overall Borrelia IgG seroprevalence was 21.3% (33/155) which was nearly two times lower than the Rickettsia seroprevalence (Table 4). Significantly more positive results for Borrelia-IgG were found in males (33.0%, 31/94) than females (3.3%, 2/61) (χ2 = 19.473, p < 0.001). The IgG anti-Borrelia rate was significantly higher in the HR group (30.4%, 31/102) compared to the LR group (3.8%, 2/53) (Table 4). In both the HR and LR groups, Borrelia-positive results were significantly more frequent in men than in women, 36.3% and 14.3% vs. 9.1 and 0.0%, respectively (Table 4). 29.5% (18/61) of workers from the Jedwabno forest inspectorate and 31.7% (13/41) from the Olsztynek forest inspectorate were IgG Borrelia-positive (χ2 = 0.056, p < 0.813).
Co-exposure in humans
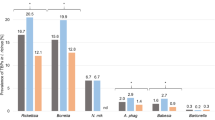
IgG antibodies against SFG Rickettsia and/or B. burgdorferi s.l were detected in 43.9% (68/155) of the total study group. Co-exposure to both bacteria was detected in 16.1% (25/155) of the participants. The IgG seroprevalence rate for co-infection with both pathogens was significantly higher among forest workers (HR group) than among residents of Olsztyn city (LR group) (23.5% [24/102] vs. 1.9% [1/53]) (Fig. 3).
Seroprevalence of IgG antibodies in single infection and coinfection with Rickettsia spp. and B. burgdorferi s.l. in adult residents of north-eastern Poland. chi2 test, p < 0.001, a vs. b - statistically significant difference between groups; a vs. a or b vs. b - no statistically significant (post-hoc Bonferroni test); HR - the high-risk group (forest workers); LR - the low-risk group - residences of Olsztyn city.
Discussion
Bacteria of the genus Rickettsia are the second most frequently detected microorganisms in questing ticks and those feeding on hosts in urban and natural areas of north-eastern Poland, following Borrelia spirochaetes28,29. The overall prevalence of SFG Rickettsia, determined at 7.5% (including MIR in nymphs), falls within the lower values of the reported prevalence range (4.4–42.3%) observed in other regions of Poland30,31,32,33,34. In the studied I. ricinus tick population, we observed significant variations in SFG Rickettsia prevalence between the study years and among the subregions of the Warmia-Mazury province. A highly variable prevalence of Rickettsia spp. in I. ricinus ticks, ranging from 0.5% to 66%, has also been reported in other European countries35,36,37. Differences in the prevalence of Rickettsia spp. in I. ricinus ticks are influenced by a number of factors, such as geographic region, habitat type, and changes in environmental conditions throughout the year30,32,34,38. However, microclimatic factors37,39,40 and the availability of suitable hosts that maintain and spread Rickettsia spp. in ecosystems41,42 are also important.
In the studied I. ricinus tick population, the SFG Rickettsia infection rate in adults was almost twice as high as in nymphs (11.6% vs. 6.5%). Higher infection rates in females and males may result from more frequent contact with infected hosts during a greater number of blood meals compared to nymphs32,40. All developmental stages of I. ricinus can harbor SFG Rickettsia, including the most frequently identified species in Europe such as R. helvetica and R. monacensis35,41. Transstadial transmission of these species should be taken into account, as well as the transovarial transmission of R. helvetica observed under field conditions43. It is therefore possible that the vector I. ricinus, like the vertebrate hosts, serves as a reservoir for these two Rickettsia species18,35,44.
The influence of geographical, ecological, and environmental factors on the variability in Rickettsia spp. prevalence in I. ricinus populations highlights the need for systematic monitoring of pathogens (or potentially pathogenic species) transmitted by ticks, other than Borrelia spirochetes and the tick-borne encephalitis virus. From a medical point of view, data on the diversity of microorganism species and their prevalence in ticks as their vectors are of great importance. Molecular analyses revealed the presence of only two species, R. helvetica and R. monacensis, in the I. ricinus tick population in northeastern Poland. These two species seem to be typical for the I. ricinus microbiota in Poland30,31,34,45. Only a few scientific reports have indicated the occurrence of another species Candidatus R. mendelii30,45 and R. slovaca46 in Polish I. ricinus ticks. R. helvetica and R. monacensis are also most frequently identified in other I. ricinus populations from Europe35. Rickettsia helvetica found in I. ricinus ticks occurs from 0.5% (Islands in the Baltic Sea) to a maximum as high as 66% in the Netherlands13. Similarly wide fluctuations in prevalence have been observed for R. monacensis, varying from 1% in Germany to 57% in Italy10. Rickettsia helvetica is considered the predominant species in I. ricinus in central and northern Europe, whereas R. monacensis is more frequently detected in southern Europe13,37. Notably, both species have been recognized as human pathogens6,47,48,49. In patients suspected of infection with these species, a relatively mild clinical course has typically been observed, including symptoms such as headache, rash, and occasionally an inoculation eschar3. In some cases, neurological complications such as meningitis and meningoencephalitis have been diagnosed50,51. Although both R. helvetica and R. monacensis have been isolated from symptomatic patients, no experimental studies have yet been conducted to unequivocally confirm their transmission through the tick-to-host route52.
In Poland, spotted fever and other rickettsioses are subject to national epidemiological surveillance. However, the number of reported cases does not exceed 10 per year53 (accessed April 30, 2025). The data do not specify whether the infections were caused by R. helvetica or by other Rickettsia species.
One approach to estimating the burden and risk of tick-borne diseases (TBDs), as well as the circulation of tick-borne pathogens in the environment, involves serological testing of the general population or healthy groups, such as blood donors, to detect specific antibodies in blood serum produced as a result of contact with antigens of microorganisms transmitted by ticks20. However, the distribution of TBDs is clearly linked to the occurrence of ticks in specific biotopes. The results of serological tests in the general population may be unreliable and very expensive because most people are not commonly exposed to tick bites. Therefore, groups at increased risk of contact with ticks, such as forest workers, farmers or patients already diagnosed with TBD as well as people working, living or engaging in recreational activities in habitats favourable to tick occurrence20, are indicated as populations that can act as “sentinels”. Among them, the emergence of new or rare tick-borne pathogens will be identified before they spread in the general population. This strategy can also reveal the co-occurrence of two or more TBDs in the same patient20,52.
Although cases of SFG rickettsiae infections in humans are not commonly reported in the temperate region of Europe19, studies on the presence of anti-spotted fever group rickettsiae antibodies indicate frequent contact with bacteria of the genus Rickettsia, especially in groups occupationally exposed to tick bite54,55. In Poland, previous serological surveys among such high-risk groups have shown antibody prevalence rates ranging from 14.7% to 39% in forestry and agricultural workers, depending on the region33,56,57,58. Our serological study also confirmed the reaction to Rickettsia antigen in residents of north-eastern Poland, both the high- and low-risk groups of exposure to tick bites. The IgG seroprevalence for SFG Rickettsia in forestry workers was almost five times higher than in the city residence group (52.9% vs. 11.3%). A very high percentage (~ 50%) of positive serological results confirming contact with Rickettsia bacteria was noted in the group of forestry workers performing outdoor work33,57. In other European countries, the seroprevalence rates for SFG Rickettsia have been reported to be 3.9–27% for similar occupational groups48,54,55. Although the overall level of infection with Borrelia spirochetes and SFG Rickettsia in the same population of I. ricinus ticks studied by us was comparable (8.1%23 vs. 7.5%), the overall seroprevalence of SFG Rickettsia in the group of residents of north-eastern Poland was nearly twice as high as that of Borrelia (38.7% vs. 21.3%). We also demonstrated similar large differences when analysing separately subgroups of residents with high and low risk of exposure to ticks. Such large differences in the frequency of seropositive reactions to SFG rickettsiae and Borrelia spirochetes undoubtedly result from the fact that I. ricinus is not the only vector of rickettsiae. Our multi-year research has demonstrated the presence of D. reticulatus in northeastern Poland, which is a vector of the human pathogenic R. raoultii59,60, responsible for DEBONEL/TIBOL (tick-borne lymphadenopathy/erythematous necrosis-lymphadenopathy) cases in Europe, including Poland61,62,63. It is noteworthy that in both high- and low-risk groups of residents from northeastern Poland, men showed significantly higher rates of Rickettsia-positive and Borrelia-positive results compared with women. These observations suggest that men may experience more frequent exposure to tick bites, most likely due to occupational activities or outdoor recreation.
However, it should be emphasized that SFG rickettsial infection is not straightforward and novel serological and molecular approaches are still in development for diagnostic applications64,65. At present, the detection of antibodies against rickettsiae in the serum by indirect immunofluorescence assay (IFA) is the gold standard for diagnosis of tick-borne rickettsioses11,66 but ELISA tests based on the purified whole bacteria or antigens, such as lipopolysaccharides from bacterial cultures (used in this study) have also been applied56,67,68. Depending on the purpose of the study, the use of ELISA is advantageous over IFA as a large number of samples can be screened in a single run64. However, a significant limitation of both methods is the inability to identify the SFG rickettsia species as a potential factor causing seroconversion and/or clinical symptoms66.
In the context of research on ticks and tick-borne diseases, increasing attention is being given to the possibility of co-infection with genetically distinct pathogens or variants within the same species69. In our study, the co-infection rate of SFG Rickettsia with Borrelia spirochaetes in questing adult I. ricinus ticks was low (2.6%) compared to a significantly higher rate (16.1%) observed based on the presence of immunological responses in the study group of residents from north-eastern Poland. The high proportion of individuals with simultaneous IgG antibodies against antigens of both tick-borne microorganisms may result not only from the bite of a single tick infected with multiple pathogens, but also from bites by more ticks either simultaneously or following sequential transmission events. This scenario is supported by our serological results, which indicate that co-infection with SFG Rickettsia and Borrelia spirochaetes among forest workers (23.5%) occupationally exposed to multiple tick bites was significantly higher than in the group of urban residents (1.9% - low-risk group). Despite the limitations of serological testing in diagnosing co-infections with multiple tick-borne pathogens in humans, numerous studies do not exclude their occurrence in symptomatic patients following tick bites. Moreover, simultaneous infection with multiple tick-borne agents can be associated with greater clinical severity than would be expected from each infection occurring independently70,71. Co-infections in ticks and their impact on the pathogenesis of tick-borne diseases are intricate and not yet fully understood. Nevertheless, co-infections should be taken into account in clinical practice, particularly in patients with immunodeficiencies70,71,72.
Conclusions
In summary, our findings provide insight into the prevalence of SFG Rickettsia in I. ricinus ticks and their seroprevalence among residents of north-eastern Poland. The prevalence in ticks, comparable to that of Borrelia spirochetes, together with high levels of antibodies in humans, confirms the circulation of these pathogens in the environment and human exposure to infection. Although limited by the assessment of minimum prevalence in nymphs, the developmental stage of I. ricinus most frequently biting humans, and by the serological assay, which did not allow precise identification of the specific SFG Rickettsia species responsible for seroconversion, our results emphasize the need for surveillance of tick-borne infections, other than Lyme borreliosis and TBE.
Data availability
The datasets used and analyzed during this study are available from the corresponding author (K.K.) upon reasonable request. Sequence data that support the findings of this study have been deposited in the GenBank database under the accession numbers PQ119728-729 and PV528813-814.
References
Stewart, A. G. & Stewart, A. G. A. An update on the laboratory diagnosis of Rickettsia spp. Infection. Pathogens 10, 1319 (2021).
Diop, A., El Karkouri, K., Raoult, D. & Fournier, P. E. Genome sequence-based criteria for demarcation and definition of species in the genus Rickettsia. Int. J. Syst. Evol. Microbiol. 70, 1738–1750 (2020).
Piotrowski, M. & Rymaszewska, A. Expansion of tick-borne rickettsioses in the world. Microorganisms 8, 1906 (2020).
Zhang, Y. Y. et al. Mapping the global distribution of spotted fever group rickettsiae: a systematic review with modelling analysis. Lancet Digit. Heal. 5, e5–e15 (2023).
Parola, P., Paddock, C. D. & Raoult, D. Tick-borne rickettsioses around the world: emerging diseases challenging old concepts. Clin. Microbiol. Rev. 18, 719–756 (2005).
Jado, I. et al. Rickettsia monacensis and human disease, Spain. Emerg. Infect. Dis. 13, 1405–1407 (2007).
Raoult, D., Berbis, P., Roux, V. & Xu, W. Maurin, M. A new tick-transmitted disease due to Rickettsia Slovaca. Lancet 350, 112–113 (1997).
Hajduskova, E. et al. 'Candidatus Rickettsia mendelii’, a novel basal group rickettsia detected in Ixodes ricinus ticks in the Czech Republic. Ticks Tick. Borne Dis. 7, 482–486 (2016).
Schötta, A. M. et al. Identification and characterization of ‘Candidatus rickettsia thierseensis’, a novel spotted fever group Rickettsia species detected in Austria. Microorganisms 8, 1670 (2020).
Parola, P. et al. Update on tick-borne rickettsioses around the world: geographic approach. Clin. Microbiol. Rev. 26, 657–702 (2013).
Fang, R., Blanton, L. S. & Walker, D. H. Rickettsiae as emerging infectious agents. Clin. Lab. Med. 37, 383–400 (2017).
Tomassone, L., Portillo, A., Nováková, M., De Sousa, R. & Oteo, J. A. Neglected aspects of tick-borne rickettsioses. Parasites Vectors. 2018 111 (11), 263 (2018).
Oteo, J. A. & Portillo, A. Tick-borne rickettsioses in Europe. Ticks Tick. Borne Dis. 3, 271–278 (2012).
Springer, A. et al. Tick hazard in a central European country: mapping europe’s principal tick-borne disease vector across Germany. Ticks Tick. Borne Dis. 16, 102485 (2025).
Zortman, I. et al. A social-ecological systems approach to tick bite and tick-borne disease risk management: exploring collective action in the occitanie region in Southern France. One Heal. 17, 100630 (2023).
Banović, P. et al. Emerging tick-borne spotted fever group rickettsioses in the Balkans. Infect. Genet. Evol. 107, 105400 (2023).
Brouqui, P. et al. Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin. Microbiol. Infect. 10, 1108–1132 (2004).
Gray, J., Kahl, O. & Zintl, A. Pathogens transmitted by Ixodes ricinus. Ticks Tick. Borne Dis. 15, 102402 (2024).
Moerbeck, L., Domingos, A. & Antunes, S. Tick-borne rickettsioses in the Iberian Peninsula. Pathogens 11, 1377 (2022).
Vu Hai, V. et al. Monitoring human tick-borne disease risk and tick bite exposure in Europe: Available tools and promising future methods. Ticks and Tick-borne Diseases. 5, 607–619 at (2014). https://doi.org/10.1016/j.ttbdis.2014.07.022
Estrada-Peña, A., Mihalca, A. D. & Petney, T. N. Ticks of Europe and North Africa. A Guide To Specimen Identification (Springer, 2018). https://doi.org/10.1007/978-3-319-63760-0
Nowak-Chmura, M. Fauna Kleszczy (Ixodida) Europy Środkowej (Wydawnictwo Naukowe Uniwersytetu Pedagogicznego, 2013).
Roux, V., Rydkina, E., Eremeeva, M. & Raoult, D. Citrate synthase gene comparison, a new tool for phylogenetic analysis, and its application for the rickettsiae. Int. J. Syst. Bacteriol. 47, 252–261 (1997).
Vitorino, L., Zé-Zé, L., Sousa, A., Bacellar, F. & Tenreiro, R. rRNA intergenic spacer regions for phylogenetic analysis of rickettsia species. Ann. N Y Acad. Sci. 990, 726–733 (2003).
Hall, T. A. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 95–98 (1999).
Tamura, K., Stecher, G. & Kumar, S. MEGA11: molecular evolutionary genetics analysis version 11. Mol. Biol. Evol. 38, 3022–3027 (2021).
Kubiak, K., Szymańska, H., Dmitryjuk, M. & Dzika, E. Abundance of Ixodes ricinus ticks (Acari: Ixodidae) and the diversity of Borrelia species in northeastern Poland. Int. J. Environ. Res. Public. Health. 19, 7378 (2022).
Kubiak, K., Dmitryjuk, M., Dziekońska-Rynko, J., Siejwa, P. & Dzika, E. The risk of exposure to ticks and tick-borne pathogens in a spa town in northern Poland. Pathogens 11, 542 (2022).
Michalski, M. M., Kubiak, K., Szczotko, M., Chajęcka, M. & Dmitryjuk, M. Molecular detection of Borrelia burgdorferi sensu lato and Anaplasma phagocytophilum in ticks collected from dogs in urban areas of north-eastern Poland. Pathogens 9, 1–11 (2020).
Kowalec, M. et al. Rickettsiales occurrence and co-occurrence in Ixodes ricinus ticks in natural and urban areas. Microb. Ecol. 77, 890–904 (2019).
Kirczuk, L., Piotrowski, M. & Rymaszewska, A. Detection of tick-borne pathogens of the genera Rickettsia, Anaplasma and Francisella in Ixodes ricinus ticks in Pomerania (Poland). Pathogens 10, (2021).
Dyczko, D., Błażej, P. & Kiewra, D. The influence of forest habitat type on Ixodes ricinus infections with Rickettsia spp. in south-western Poland. Curr. Res. Parasitol. Vector-Borne Dis. 6, 100200 (2024).
Stańczak, J. et al. Kampinos National Park: a risk area for spotted fever group rickettsioses, central Poland? Exp. Appl. Acarol. 70, 395–410 (2016).
Zając, Z. et al. Disparate dynamics of pathogen prevalence in Ixodes ricinus and Dermacentor reticulatus ticks occurring sympatrically in diverse habitats. Sci. Rep. 13, 10645 (2023).
Rizzoli, A. et al. Ixodes ricinus and its transmitted pathogens in urban and peri-urban areas in europe: new hazards and relevance for public health. Front. Public. Heal. 2, 251 (2014).
Oechslin, C. P. et al. Prevalence of tick-borne pathogens in questing Ixodes ricinus ticks in urban and suburban areas of Switzerland. Parasites Vectors. 10, 558 (2017).
Knoll, S. et al. Regional, seasonal, biennial and landscape-associated distribution of Anaplasma phagocytophilum and Rickettsia spp. infections in Ixodes ticks in northern Germany and implications for risk assessment at larger Spatial scales. Ticks Tick. Borne Dis. 12, 101657 (2021).
Welc-Falȩciak, R. et al. Rickettsiaceae and Anaplasmataceae infections in Ixodes ricinus ticks from urban and natural forested areas of Poland. Parasites Vectors 7, 121 (2014).
Špitalská, E., Boldiš, V., Derdáková, M. & Selyemová, D. Rusňáková Tarageľová, V. Rickettsial infection in Ixodes ricinus ticks in urban and natural habitats of Slovakia. Ticks Tick. Borne Dis. 5, 161–165 (2014).
Arz, C., et al. Spotted fever group rickettsiae in ticks and small ammmals from grassland and forest habitats in central Germany. Pathogens 12, 933 (2023).
Heglasová, I., Víchová, B. & Stanko, M. Diversity and prevalence of spotted-fever group rickettsiae in Ixodid ticks across Slovakia, central Europe. Ticks Tick. Borne Dis. 16, 102490 (2025).
Glass, A., Springer, A. & Strube, C. A 15-year monitoring of rickettsiales (Anaplasma phagocytophilum and Rickettsia spp.) in questing ticks in the city of Hanover, Germany. Ticks Tick. Borne Dis. 13, 101975 (2022).
Hauck, D. et al. Transovarial transmission of Borrelia spp., Rickettsia spp. and Anaplasma phagocytophilum in Ixodes ricinus under field conditions extrapolated from DNA detection in questing larvae. Parasites and Vectors 13, 176 (2020).
Sprong, H. et al. Ixodes ricinus ticks are reservoir hosts for Rickettsia helvetica and potentially carry flea-borne Rickettsia species. Parasit. Vectors 2, 41 (2009).
Stańczak, J., Biernat, B., Racewicz, M., Zalewska, M. & Matyjasek, A. Prevalence of different rickettsia spp. In Ixodes ricinus and dermacentor reticulatus ticks (Acari: Ixodidae) In north-eastern Poland. Ticks Tick. Borne Dis. 9, 427–434 (2018).
Chmielewski, T., Podsiadly, E., Karbowiak, G. & Tylewska-Wierzbanowska, S. Rickettsia spp. in ticks, Poland. Emerg. Infect. Dis. 15, 486–488 (2009).
Nilsson, K., Lindquist, O. & Påhlson, C. Association of Rickettsia helvetica with chronic perimyocarditis in sudden cardiac death. Lancet 354, 1169–1173 (1999).
Fournier, P. E., Grunnenberger, F., Jaulhac, B., Gastinger, G. & Raoult, D. Evidence of Rickettsia helvetica infection in humans, eastern France. Emerg. Infect. Dis. 6, 389–392 (2000).
Kim, Y. S. et al. First isolation of Rickettsia monacensis from a patient in South Korea. Microbiol. Immunol. 61, 258–263 (2017).
Nilsson, K., Elfving, K. & Påhlson, C. Rickettsia helvetica in patient with meningitis, Sweden, 2006. Emerg. Infect. Dis. 16, 490 (2010).
Nilsson, K., Wallménius, K. & Påhlson, C. Coinfection with Rickettsia helvetica and Herpes simplex virus 2 in a young woman with meningoencephalitis. Case Rep. Infect. Dis. 2011, 469194 (2011).
Azagi, T., Hoornstra, D., Kremer, K., Hovius, J. W. R. & Sprong, H. Evaluation of disease causality of rare Ixodes ricinus-borne infections in Europe. Pathogens 9, 1–22 (2020).
National Institute of Public Health – National Institute of Hygiene – Department of Epidemiology Infectious diseases and poisonings in Poland. National Inst. Public. Health – Natl. Inst. Hygiene – Department Epidemiology (accessed April 30, 2025) http://www.pzh.gov.pl/oldpage/epimeld/index_p
Wölfel, S. et al. High Seroprevalence for Indigenous spotted fever group rickettsiae in forestry workers from the federal state of Brandenburg, Eastern Germany. Ticks Tick. Borne Dis. 8, 132–138 (2017).
Cinco, M., Luzzati, R., Mascioli, M., Floris, R. & Brouqui, P. Serological evidence of Rickettsia infections in forestry rangers in north-eastern Italy. Clin. Microbiol. Infect. 12, 493–495 (2006).
Borawski, K. et al. Prevalence of spotted fever group Rickettsia in north-eastern Poland. Infect. Dis. (Auckl). 51, 810–814 (2019).
Zajac, V. et al. Study on tick-borne rickettsiae in Eastern Poland. II. Serological response of occupationally exposed populations. Ann. Agric. Environ. Med. 20, 280–282 (2013).
Podsiadły, E., Chmielewski, T., Karbowiak, G., Kȩdra, E. & Tylewska-Wierzbanowska, S. The occurrence of spotted fever rickettsioses and other tick-borne infections in forest workers in Poland. Vector-Borne Zoonotic Dis. 11, 985–989 (2011).
Kubiak, K. et al. Dermacentor reticulatus ticks (Acari: Ixodidae) distribution in north-eastern Poland: an endemic area of tick-borne diseases. Exp. Appl. Acarol. 75, 289–298 (2018).
Kubiak, K. et al. Tick-borne pathogens in questing adults Dermacentor reticulatus from the Eastern European population (north-eastern Poland). Sci Rep 14, 698 (2024).
Chmielewski, T., Rudzka, D., Fiecek, B., Maczka, I. & Tylewska-Wierzbanowska, S. Case of TIBOLA/DEBONEL (tick-borne lymphadenopathy/Dermacentor spp.-borne necrosis-erythema-lymphadenopathy) in Poland. Przegl Epidemiol. 65, 583–586 (2011).
Koczwarska, J., Pawełczyk, A., Dunaj-Małyszko, J. & Polaczyk, J. Welc-Falęciak, R. Rickettsia species in Dermacentor reticulatus ticks feeding on human skin and clinical manifestations of tick-borne infections after tick bite. Sci. Rep. 13, 1–8 (2023).
Świtaj, K., Chmielewski, T., Borkowski, P., Tylewska-Wierzbanowska, S. & Olszynska-Krowicka, M. Spotted fever rickettsiosis caused by Rickettsia raoultii - case report. Przegl Epidemiol. 66, 37–39 (2012).
Husin, N. A., AbuBakar, S. & Khoo, J. J. Current tools for the diagnosis and detection of spotted fever group Rickettsia. Acta Trop. 218, 105887 (2021).
Willson, R. et al. Development of a rapid antigen-based lateral flow assay for tick-borne spotted fever rickettsioses. PLoS One. 20, e0312819 (2025).
Blanton, L. S. The rickettsioses: a practical update. Infect. Dis. Clin. North. Am. 33, 213–229 (2019).
Cheran, C. A. et al. Identifying new areas of endemicity and risk factors for Rickettsia conorii subsp. conorii infection: serosurvey in rural areas of Romania. Pathogens 13, 783 (2024).
Ganbold, D. et al. Seroprevalence and risk factors of Borrelia burgdorferi sensu Lato and Rickettsia species infection in humans in Mongolia, 2016–2020. PLoS One. 18, e0289274 (2023).
Cutler, S. J. et al. Tick-borne diseases and co-infection: current considerations. Ticks Tick. Borne Dis. 12, 101607 (2021).
Diuk-Wasser, M. A., Vannier, E. & Krause, P. J. Coinfection by Ixodes tick-borne pathogens: ecological, epidemiological, and clinical consequences. Trends Parasitol. 32, 30–42 (2016).
Swanson, S. J., Neitzel, D., Reed, K. D. & Belongia, E. A. Coinfections acquired from Ixodes ticks. Clin. Microbiol. Rev. 19, 708–727 (2006).
Pawełczyk, A. et al. Seroprevalence of six pathogens transmitted by the Ixodes ricinus ticks in asymptomatic individuals with HIV infection and in blood donors. Sci. Rep. 9, 1–10 (2019).
Acknowledgements
The study was approved by the Bioethics Committee of the University of Warmia and Mazury in Olsztyn (No. 35/2024). The authors would like to thank Tański A. (Department of Medical Biology, UWM in Olsztyn) for drawing the map (Fig. 1).
Funding
This work was supported by the statutory fund of the School of Public Health, Collegium Medicum, University of Warmia and Mazury in Olsztyn.
Author information
Authors and Affiliations
Contributions
Conceptualization: K.K., methodology: K.K.; formal analysis: K.K. and H.S., investigation: K.K., H.S., S.K. B.W-B., B.D., M.Ł., A.B. P.K.; resources: K.K., H.S., S.K., B.W-B., A.B.; writing—original draft preparation: K.K.; writing—review and editing: K.K., H.S., S.K. B.W-B.; visualization: K.K. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
The study was approved by the Bioethics Committee of the University of Warmia and Mazury in Olsztyn (No. 35/2024).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kubiak, K., Szymańska, H., Krzętowska, S. et al. Molecular and serological evidence of spotted fever group rickettsiae and Borrelia co-infection in Ixodes ricinus and humans in north-eastern Poland. Sci Rep 16, 1053 (2026). https://doi.org/10.1038/s41598-025-30618-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-30618-w