Abstract
Spinal muscular atrophy (SMA) is a progressive neuromuscular disorder characterized by respiratory muscle weakness and impaired functional capacity. This study aimed to evaluate the effects of respiratory exercises, inspiratory muscle training (IMT), and trunk control training in children with SMA. Thirty-eight children (10.21 ± 4.56 years) with SMA were randomly assigned to two groups. Pulmonary rehabilitation group (PRG, n = 19), performed diaphragmatic, pursed-lip, and segmental breathing exercises plus IMT at 30% maximal inspiratory pressure, twice daily for 8 weeks. Trunk control group (TCG, n = 19) received the same respiratory program with additional trunk exercises, three times a week. Primary outcomes were respiratory muscle strength (MIP, MEP), pulmonary function (FVC, FEV1, PEF), and peak cough flow (PCF). Secondary outcomes were trunk control (TCMS), upper limb function (RULM), and motor performance (HFMSE). Both groups demonstrated significant within-group improvements in respiratory strength and motor outcomes (p < 0.05). MIP increased with large effect sizes (PRG: d = 1.039; TCG: d = 1.556). Notably, TCG showed additional significant gains in static sitting balance (p < 0.05), selective movement control (p < 0.001), and RULM (p < 0.05). Although between-group differences were not statistically significant, effect sizes indicated clinically relevant advantages favoring the combined intervention. Combining IMT with trunk control training appears safe, feasible, and clinically beneficial in children with SMA.
Similar content being viewed by others
Introduction
Spinal muscular atrophy (SMA) is a severe neuromuscular disorder caused by a deficiency of the survival motor neuron (SMN) protein due to a homozygous deletion or mutation in the SMN1 gene localized to 5q11.2-q13.31. The incidence of SMA is estimated to be approximately 1 in 6,000–12,000 live births2. The disease is characterized by progressive degeneration of α-motor neurons, leading to symmetrical and proximal muscle weakness, particularly in the axial and upper and lower limb muscles1. SMA subtypes are defined as type 1, type 2, type 3, and type 4 based on age of onset, clinical severity, and motor milestones achieved3. Beyond this clinical classification, the functional classification of SMA is crucial for understanding disease progression and treatment planning. Functionally, SMA patients are classified as non-sitters, sitters, and walkers, according to the individual’s ability to sit and walk independently4.
Respiratory complications, such as respiratory muscle weakness, hypoventilation, and impaired secretion clearance, are among the leading causes of morbidity and mortality in individuals with SMA5. As the disease progresses, vital capacity and cough strength decrease significantly, leading to early respiratory failure in SMA type 1 and the development of chronic pulmonary complications in type 2 and 3 patients. This deterioration is associated with ineffective airway clearance, recurrent pulmonary infections, restrictive lung function due to weak or inadequate chest wall and lung growth, and alveolar hypoventilation, potentially resulting in chronic respiratory failure and early mortality in the long term6,7. Although recent pharmacological advances, including gene replacement therapy and SMN2 splicing modulators, have improved motor outcomes and survival in SMA patients8,9. However, their effects on pulmonary function are limited, particularly in patients with type 2 and 3 SMA10,11.
Given these limitations, non-pharmacological strategies such as pulmonary rehabilitation and inspiratory muscle training (IMT) have gained increasing attention. However, there is currently insufficient research on the efficacy, safety, and optimal dosing of IMT in SMA populations. It is important that IMT protocols are carefully structured with adequate rest intervals due to the risk of fatigue12.
In addition to respiratory muscle weakness, postural instability and kyphoscoliosis are frequently observed in SMA patients. These thoracic deformities further increase the mechanical disadvantage of weakened respiratory muscles, which can cause an increase in respiratory load, reducing tidal volume and accelerate alveolar hypoventilation13. Moreover, SMA type 2 and 3 patients are known to perform trunk movements with limited and low muscle activity. Trunk joint torques in these patients are reduced by approximately 45% compared with those healthy peers, which may contribute to scoliosis progression and earlier deterioration of motor functions14. In seated activities, the trunk is indispensable as it interacts with upper limb movement as part of the kinematic chain and provides a stable base for these movements. Therefore, trunk control cannot be considered in isolation from upper limb function15,16. A study conducted in children with Duchenne muscular dystrophy (DMD) indicated that a trunk-focused exercise program may improve not only trunk control but also upper limb and respiratory functions17.
Respiratory and trunk muscles work synergistically to maintain both ventilation and postural stability. The diaphragm plays a dual role, contributing to breathing while stabilizing the spine and pelvis through coordinated activation with abdominal and paraspinal muscles. This coordinated recruitment regulates intra-abdominal pressure and facilitates efficient load transfer between the thorax and pelvis18. Conversely, trunk instability and axial weakness can impair respiratory mechanics by altering thoracic alignment and limiting diaphragmatic excursion. Evidence from neurological populations further supports this bidirectional relationship: trunk control is positively correlated with respiratory parameters such as FVC, FEV₁, MIP, and MEP, and respiratory training significantly enhances trunk stability19. Based on the synergistic relationship between respiratory and trunk muscles, we hypothesized that integrating trunk control training with IMT would yield greater improvements in respiratory strength, pulmonary function, and motor performance than IMT alone.
Recent studies on exercise in SMA often focus on the effects of isolated training modalities. For example, studies have demonstrated the benefits of aerobic exercise on functional capacity and fatigue in patients with SMA type 320, while others have shown that IMT alone can improve respiratory muscle strength and cough efficiency in children with various NMDs21. While these studies confirm the value of unimodal interventions, they do not address the potential synergistic benefits of a combined approach. This focus on isolated modalities represents a significant knowledge gap given the fundamental biomechanical link between respiratory mechanics and trunk stability. A recent study on physiotherapy practice has also highlighted the urgent need for updated, evidence-based guidelines that meet the evolving and complex needs of patients with SMA22. Therefore, investigating the effectiveness of a combined intervention is a critical next step in optimizing rehabilitation for this population.
Despite this, to our knowledge, no randomized controlled trials have evaluated the combined use of IMT, respiratory exercises, and structured trunk control training in individuals with SMA. This study aimed to evaluate the effects of an eight-week intervention program combining respiratory exercises, IMT, and trunk control exercises on respiratory muscle strength, pulmonary function, cough efficiency, trunk control, upper limb function, and motor ability in children with SMA. We hypothesize that a multicomponent rehabilitation program including IMT, respiratory exercises, and trunk control training will improve the overall respiratory and motor function more than IMT and respiratory exercises alone.
Methods
Study design
This randomized controlled clinical trial included 40 children with SMA. Participants were recruited between February 2023 and December 2024 at the Pediatric Pulmonology Clinic of İstanbul Bağcılar Medipol Mega University Hospital (Istanbul, Turkey).
After eligibility screening, participants who met inclusion criteria were randomly allocated (1:1) to the Pulmonary Rehabilitation Group (PRG) or the Trunk Control Group (TCG), using a stratified randomization method based on SMA type (types 1, 2, and 3) and age group (5–7, 8–12, and 13–18 years). These stratification criteria were chosen for their clinical relevance, as both age and disease severity are known to influence respiratory and motor function outcomes in SMA. The predefined age ranges reflect functionally distinct developmental stages commonly used in pediatric rehabilitation research.
The randomization sequence was generated by an independent researcher who was not involved in participant recruitment, intervention delivery, or data collection. Random numbers were first generated using an Excel-based random number generator (Microsoft Corporation; RAND function), followed by allocation within each stratum using the Research Randomizer application (https://www.randomizer.org/). Randomization was completed prior to baseline assessments and the start of the intervention to prevent potential bias in group allocation.
Due to the nature of the intervention, blinding of the physiotherapist and participants was not feasible. However, allocation concealment was maintained, as randomization was performed by an independent researcher using a secure, pre-generated sequence, and group assignments were revealed only after participant enrollment. No deviations from the registered protocol, prespecified outcomes, or statistical analysis plan were made after trial commencement.
The study was prospectively registered at ClinicalTrials.gov (NCT06178653; https://clinicaltrials.gov/study/NCT06178653) on 21/02/2024, and approved by the Istanbul Medipol University Non-Interventional Research Ethics Committee (E-10840098-772.02.02.02–198). All methods were performed in accordance with the relevant guidelines and regulations. All methods were carried out in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants and their legal guardians.
Participants
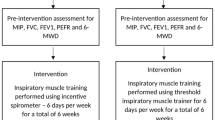
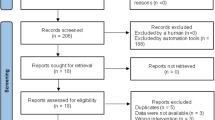
The inclusion criteria were: genetically confirmed 5q SMA (homozygous gene deletion or compound heterozygous mutation), age 5–18 years, maximum inspiratory pressure (MIP) less than 60 cmH2O, ability to sit unsupported for at least five seconds, and no recent upper respiratory infection within the past 3 weeks. The exclusion criteria included a history of upper limb or spine surgery, co-existing orthopedic or neurological conditions, cognitive, visual, or auditory impairments that could interfere with assessment, exercise discontinuation, and a history of premature birth. Details of recruitment and randomization are presented in the study flow diagram (see Fig. 1).
Outcome measures
Demographic and clinical characteristic data were recorded at baseline.
The primary outcomes included respiratory muscle strength, pulmonary function, and cough efficiency. MIP and maximal expiratory pressure (MEP) were measured using a portable digital respiratory pressure monitor (RP Check; MD Diagnostics Ltd, Chatham, UK) following American Thoracic Society (ATS)/European Respiratory Society (ERS) guidelines. Measurements were taken while the participants were seated with a nose clip and a sealed mouthpiece; the highest value from three trials with < 5% variability was recorded23. Pulmonary function was measured via spirometry (COSMED Pony FX®, COSMED; Italy), including forced vital capacity (FVC), forced expiratory volume in one second (FEV₁), FEV₁/FVC ratio, and peak expiratory flow (PEF), according to ATS/ERS standards24. Cough effectiveness was evaluated via peak cough flow (PCF) with a handheld flow meter (Expirite® DL-F03). The highest value from the three efforts was recorded25. Oxygen saturation (SpO2) and heart rate were monitored during assessments via a pulse oximeter (Freely SHO-3002). Patients were allowed to rest for one minute between attempts, and verbal encouragement was provided during the test.
The secondary outcomes included trunk control, upper limb function, and motor performance. Trunk control was assessed via the Trunk Control Measurement Scale (TCMS), a validated and reliable tool for children with various neuromotor disorders, which is scored from 0 to 58 (higher = better control) and is based on static and dynamic sitting balance26. Upper limb function was evaluated via the Revised Upper Limb Module (RULM), which is designed for SMA. It consists of 19 items scored 0–2, with a maximum total score of 37 points. Higher scores reflect greater functional ability27. The Hammersmith Functional Motor Scale-Expanded (HFMSE) evaluates changes in the motor functions of children with SMA who can sit and walk, as well as the motor milestones of these children. It consists of 33 items, each scored on a scale of 0 to 2, with a maximum total score of 66 points. Higher scores indicate better motor function4.
Interventions
All participants continued to receive their pharmacological treatments and routine standard care, including multidisciplinary follow-up, throughout the study. No changes were made to their ongoing treatment plans.
All interventions were delivered by a licensed physiotherapist with postgraduate training. Supervised physiotherapy sessions were conducted weekly. During these sessions, parents were provided with detailed information about the home-based program.
MIP values were measured during the weekly supervised sessions, and training load was adjusted weekly to 30% of the current MIP according to clinical evidence and manufacturer recommendations28. The pulmonary rehabilitation was prescribed for twice daily, five days per week, and the trunk control exercises were scheduled three days per week. On supervised days, exercises were performed under direct observation of the physiotherapist; on unsupervised days, caregivers administered the home-based protocol. Adherence and session timing were recorded in an exercise diary provided to each family.
Pulmonary rehabilitation group
The PRG included diaphragmatic breathing exercises to increase ventilation and SpO2, and reduce breathing frequency29, pursed-lip breathing exercises to increase alveolar ventilation30, and segmental breathing exercises31. Exercises were performed in three sets of 10 repetitions with rest intervals of 5–6 tidal volumes to prevent respiratory muscle fatigue and hyperventilation.
Subsequently, the IMT protocol was administered using a handheld threshold-loading IMT device (Orygen® Inspiratory Valve Muscle Trainer), which provides a constant inspiratory resistance independent of airflow. Before each supervised session, the device was calibrated to zero resistance in accordance with manufacturer guidelines, and valve stability was verified.
Each session consisted of three sets of 10 breaths against an external load at an intensity of 30% of the MIP value32, totaling 30 breaths. Rest intervals of 30–40 s were provided. Participants were instructed to perform slow, controlled inspirations and expirations while maintaining an upright seated posture. The target breathing rate was approximately 15–20 breaths per minute. The training intensity was dynamically adjusted each week to 30% of the newly measured MIP value, allowing for a natural progression of inspiratory load as respiratory muscle strength improved. Parents were instructed to observe their children’s home sessions and contacted by the research team at least twice weekly to ensure compliance. Additionally, the participants were asked to record video footage of their sessions to confirm that the exercises were performed correctly.
Trunk control group
In addition to the PRG protocol, exercises were performed to enhance trunk muscle activation, pelvic control, proximal stabilization, and trunk and gluteal muscle strengthening. The intensity of the exercises gradually increased. The exercises progressed from firm to soft to dynamic surfaces to stimulate proprioceptive and vestibular systems. Performing exercises on a dynamic surface was found to be more effective, as it triggers the response of the muscles to maintain the desired posture against possible postural deterioration33.
The training content was designed to promote neutral spine alignment, improve trunk extensor strength, and enhance core stabilization. Functional trunk movements were incorporated, including reaching, trunk rotation, and weight-shifting activities over the pelvis, as well as sitting balance and postural control exercises to support dynamic stability (see Supplementary File 1).
Exercise intensity began at a light level (Borg RPE 2–4), with two sets of 10 repetitions per session. To prevent fatigue, 20-second rests were provided between sets and 1-minute breaks between exercises. The physiotherapist offered verbal and tactile cues as needed to ensure correct execution and posture.
Safety considerations
No adverse events or side effects were reported by participants or their caregivers throughout the intervention period. To mitigate potential fatigue and ensure safety, structured rest periods were incorporated into all sessions, and initial intensities were set conservatively.
Safety monitoring included continuous observation of oxygen saturation (SpO₂) and clinical symptoms during supervised sessions. Prior to each session, the IMT device was checked for proper valve function and resistance stability to prevent excessive inspiratory loading. A predefined stopping criterion was applied if SpO₂ dropped below 92%, or if fatigue, dyspnea, dizziness, or discomfort occurred; the session was paused until physiological parameters normalized.
Participant safety and comfort were continuously monitored throughout the intervention via direct clinical observation by a licensed physiotherapist, with caregiver supervision during home-based sessions to promptly identify any signs of distress or overexertion.
Sample size considerations
The sample size was calculated using G*Power 3.1, based on the study conducted by Koessler et al.34. Accordingly, a sample of 36 participants was required to achieve a statistical power of 80.6% at a 5% significance level, based on an effect size of f = 0.874. To account for a potential 10% dropout rate due to factors such as medical complications or study withdrawal, a final sample size of 40 participants was targeted and successfully enrolled.
Adherence and participation
Participation and adherence to the 8-week intervention were closely monitored through weekly follow-ups, exercise diaries, and caregiver reports. Although participants who missed ≥ 3–4 consecutive weeks were to be excluded according to the study plan, all children and families attended the sessions consistently and completed the full program. This high adherence reflected both the feasibility and perceived benefit of the intervention, and therefore no participants were excluded due to noncompliance.
Statistical analysis
Statistical analyses were conducted using the IBM SPSS Statistics v22 (IBM Corp., Armonk, NY, USA). Descriptive statistics for quantitative variables were reported as mean ± standard deviation (SD), and qualitative variables were expressed as the number of participants (n) and frequency (%). The normality of the distribution was assessed by the Shapiro–Wilk test. Depending on data distribution, between-group comparisons were performed via the independent samples t-tests or the Mann–Whitney U test. Within-group differences were analysed using the Paired samples t-test or the Wilcoxon signed-rank test. Categorical variables were compared via the chi-square or Fisher’s exact test. Within-group effect sizes were calculated using Cohen’s d (small: d = 0.2; medium: d = 0.5; large: d = 0.8)35. Where applicable, 95% confidence intervals (CI) were reported alongside effect sizes A significance level of α = 0.05 was used for all two-tailed hypothesis tests, and p-values < 0.05 were considered statistically significant.
Results
Demographic and clinical characteristics of the participants
A total of 77 children were assessed for eligibility, of whom 40 were randomized (1:1) to PRG (n = 20) or TCG (n = 20). Two participants were lost to follow-up (one per group), resulting in 38 completers included in the primary per-protocol analyses. Reasons for exclusion and loss to follow-up are detailed in Fig. 1. No post-randomization protocol deviations occurred.
The baseline demographic characteristics of participants who completed the study are presented in Table 1, and SMA-related clinical data are summarized in Table 2. No significant between-group differences were observed at baseline in terms of age, SMA type, respiratory function, or functional performance measures (p > 0.05).
Within-group changes in respiratory outcomes
The post-intervention findings regarding respiratory muscle strength, pulmonary function, and cough capacity are shown in Table 3. Significant improvements in all respiratory parameters were observed within both groups (p < 0.05). Large within-group effect sizes were noted, particularly in the PRG for MIP (d = 1.039), FEV₁ (d = 1.175), and PEF (d = 2.300); and in the TCG, large effect sizes were also revealed for MIP (d = 1.556), FEV1 (d = 1.057), and PEF (d = 1.960).
Mean MIP increased by + 18.15 ± 9.67 cmH₂O in the PRG and + 24.21 ± 14.42 cmH₂O in the TCG, corresponding to approximately 35–40% gains from baseline. In practical terms, these improvements likely reflect enhanced inspiratory reserve and cough efficiency, consistent with the concurrent increases observed in PEF and PCF, which are key determinants of ventilation stability and airway clearance in children with SMA.
Although minimal clinically important differences (MCIDs) for respiratory strength parameters have not yet been established in pediatric SMA, within-group changes were interpreted using a distribution-based framework (Cohen’s d ≥ 0.8 indicating a large effect) and the relative percentage improvement from baseline values, both of which suggest clinically meaningful gains.
Between-group comparisons in respiratory outcomes
Although both groups demonstrated significant within-group improvements in respiratory outcomes, no statistically significant differences were found in between-group comparisons (p > 0.05). The 95% confidence intervals for between-group comparisons support these findings. Specifically, the 95% CIs for the mean differences were: MIP (–14.13 to 2.03 cmH₂O), MEP (–8.52 to 3.99 cmH₂O), FVC (0.79 to 0.81 L), FEV₁ (–0.31 to 0.52 L), FEV₁/FVC (–14.66 to 14.90%), PEF (–1.37 to 1.17 L/s), and PCF (0.89 to 0.90 L/min). These findings suggest that both interventions yielded similar effects on respiratory function.
Within-group changes in functional outcomes
The pre- and post-intervention results regarding trunk control, upper limb function, and motor performance are summarized in Table 4. In the PRG, significant post-intervention improvements were observed in TCMS total scores (d = 0.111; p = 0.009) and HFMSE scores (d = 0.050; p = 0.014), whereas RULM scores did not change significantly (d = 0.020; p = 0.331). On the other hand, the TCG showed significant improvements across the TCMS subdomains (static sitting balance: d = 0.287; p = 0.004; selective movement control: d = 0.372; p < 0.001), total TCMS (d = 0.317; p < 0.001), HFMSE (d = 0.102; p = 0.020), and RULM (d = 0.103; p = 0.011).
Between-group comparisons in respiratory outcomes
No statistically significant differences were observed between PRG and TCG in post-interventional scores on the TCMS, RULM, or HFMSE (p > 0.05). The 95% confidence intervals for the mean differences support these findings: TCMS total score (–3.86 to 0.18), static sitting balance (–1.65 to 0.59), selective movement control (–2.85 to 0.22), dynamic reach (–0.53 to 0.53), RULM (–1.63 to 0.37), and HFMSE (–1.59 to 0.96). Even though these intervals hold the value zero, or no statistical significance, the direction and magnitude of change observed on the TCG indicate a probable additive advantage of including trunk control exercises in conjunction with respiratory training.
Discussion
To our knowledge, this is the first randomized controlled trial evaluating the combined effects of breathing exercises, IMT, and trunk control exercises on respiratory and motor functions in sitter and/or ambulatory children with SMA. Our findings suggest that IMT increases respiratory muscle strength and that combining trunk control exercises with IMT improves respiratory and motor outcomes in children with SMA.
Respiratory muscle weakness leads to reduced vital capacity due to impaired inspiration and expiration, along with decreased lung compliance23. Respiratory muscles share common structural, physiological, and biochemical features of skeletal muscles, enabling hypertrophy under load36. Therefore, strengthening these muscles is expected to improve pulmonary function. Most studies on respiratory muscle training have focused on chronic pulmonary diseases such as chronic obstructive pulmonary disease, asthma, and cystic fibrosis37. However, evidence on the efficacy of IMT in patients with NMD, particularly SMA, remains scarce, as most studies do not report SMA-specific outcomes. In our protocol, IMT led to significant improvements in MIP and MEP in both groups. The increase in MEP, was possibly due to enhanced airway diameter, reduced airflow limitation, and decreased dynamic hyperinflation38. Additionally, the threshold loading intensity of IMT may contribute to its effectiveness. Unlike breathing exercises, IMT provides direct resistance loading, inducing structural adaptations such as hypertrophy of the diaphragm and accessory muscles, and increasing the proportion of type I fibres and the cross-sectional area of type II fibers39,40. These findings are consistent with previous reports from other NMDs, supporting the feasibility and efficacy of IMT in SMA21,34. From a neurophysiological perspective, repetitive inspiratory loading may also enhance motor unit recruitment efficiency and improve the central coordination of respiratory and postural muscle activation patterns, contributing to more synchronized breathing and trunk stability. Although SMA primarily involves lower motor neuron degeneration, such adaptive reorganization within the remaining neural pathways may still facilitate more efficient respiratory control and postural synergy. Although no statistically significant difference was found in the between-group comparison, effect size analyses revealed greater improvements in the TCG. The progressive and surface-varied nature of trunk control exercises may enhance respiratory outcomes by improving postural stability and neuromuscular coordination.
Pulmonary function decline in SMA closely parallels progressive muscle weakness and is considered a clinical key marker, particularly in early adulthood41. Spirometry is a reliable tool for assessing pulmonary function in pediatric and young adult SMA patients41,42. Reduced FVC and FEV1 values observed in our sample were consistent with previous studies and may reflect underlying respiratory muscle weakness, limited thoracic expansion, and scoliosis progression41. In our study, both groups exhibited statistically significant improvements in PFT parameters after eight weeks. Breathing exercises and IMT increase the expansion capacity of the lungs by strengthening the diaphragm and intercostal muscles in individuals with SMA, thus contributing to improvements in the FVC and FEV1 values. IMT may reduce the effects of restrictive breathing patterns by facilitating greater inspiratory volume. Additionally, progressive trunk exercises may have amplified these effects by enhancing postural muscle strength and supporting expiratory and accessory inspiratory function. The observed increase in the FEV1/FVC ratio supports the positive effect of pulmonary rehabilitation on restrictive lung dysfunction. Although the between-group differences were not statistically significant, the effect size analyses yielded clinically important and relevant insights. TCG had greater clinical effects on the FVC and FEV1/FVC ratio, whereas PRG exhibited higher clinical effects on the FEV1 and PEF values. These values may reflect the complex nature of the combined intervention, requiring a higher level of physical and cognitive coordination, and this may cause additional challenges for younger type 1 and 2 participants. Moreover, the primary purpose of trunk control training is to support general respiratory mechanics by creating a stable base that will support the activation of respiratory muscles. However, this may not have directly led to improved parameters reflecting acute airflow, such as FEV₁ and PEF. Nonetheless, both interventions showed high efficacy, and the combined protocol had greater effects on certain respiratory parameters, reinforcing its potential value in clinical settings.
Cough efficacy is an essential component of airway clearance and requires the coordinated activity of both inspiratory and expiratory muscles. A PCF of 160–200 L/min is considered the threshold for mucus expectoration, and 250–270 L/min is typically needed to prevent pneumonia in patients with NMD43. Prior studies have reported that children with SMA usually have PCF values below 270 L/min44. Our findings confirm this, supporting the notion that airway clearance capacity is significantly limited in SMA and highlighting their vulnerability to respiratory infections. Resistance-based IMT may have increased lung expansion capacity, enabling higher expiratory flow rates and thereby contributing to improvements in PEF and PCF values45. These enhancements facilitate more effective coughing, improve airway clearance, and potentially lower infection risk. Although differences between groups were not statistically significant, effect size analyses suggest that the combined intervention resulted in greater improvement in PCF.
A strong relationship exists between respiratory muscle activation and the timing of trunk muscle contraction during limb movements. The diaphragm not only supports breathing but also contributes to trunk-limb synergy46,47. While the literature on trunk and neck muscle strength in SMA remains limited, previous studies have reported reduced trunk muscle activity in SMA types 2 and 348. After eight weeks, the total TCMS score increased significantly in the PRG; however, no statistically significant difference was observed in the subscales. In contrast, the TCG showed significant improvements in the total score, the static sitting balance, and total selective movement control subscales. No notable changes were observed in dynamic reaching, possibly due to ceiling effects (mean: 8.10/10) or difficulties experienced by some participants in performing tasks on unstable surfaces. The observed improvements in selective movement control are likely attributable to the targeted effects of trunk-strengthening exercises. Considering the contribution of the diaphragm in trunk stability, pulmonary rehabilitation may indirectly support; however, the lack of significant change in the TCMS subscales suggests that trunk stability should be supported with targeted, specific exercises.
Children with SMA types 2 and 3 typically exhibit progressive upper limb muscle weakness, limiting independence in terms of quality of life and social participation49. Studies conducted on DMD patients found a linear association between trunk control and upper limb functions50, and trunk training was shown to increase upper limb function17. In our study, RULM scores improved significantly only in the TCG, suggesting that trunk stabilization enhances upper limb functionality. The lack of significant changes in the PRG can be explained by the lack of direct targeting of upper limb musculature. Improved trunk stability may have facilitated more effective upper limb functions, suggesting that comprehensive rehabilitation strategies in SMA should incorporate trunk stabilization to support upper limb functionality. Greater improvements may be attainable with longer interventions or higher training intensity.
Motor function outcomes following modern treatment approaches constitute an important dimension of SMA rehabilitation research51,52,53. In our study, both groups showed significant improvements in HFMSE scores following the intervention, indicating gains in overall motor performance. However, no statistically significant difference emerged between groups. Effect size analyses revealed that the greater improvement in the TCG was more pronounced, supporting a potential adjunctive role for trunk training in motor development. The lack of statistical significance between groups may be related to the relatively short duration of the intervention. Moreover, the decreased sensitivity of HFMSE in weaker subtypes, as induced by floor effects and high-motor-demand tasks (e.g., stair climbing), may have masked further improvements54. These findings highlight the importance of selecting outcome measures that align with patients’ functional levels to capture actual progress.
The differing response patterns between the two intervention groups can be attributed to the complementary but distinct physiological mechanisms targeted by each intervention. While IMT primarily improves respiratory pump strength through increased muscle force-generating capacity, the addition of trunk control training enhances neuromuscular coordination, postural stabilization, and load transfer efficiency across the thoraco-abdominal region. Improved trunk stiffness and anticipatory postural adjustments reduce mechanical load on the diaphragm, allowing respiratory muscles to operate more efficiently. This synergistic interaction likely explains the greater improvements observed in MIP, selective trunk control, and upper limb function within the trunk control group. From a clinical standpoint, these findings suggest that an optimal rehabilitation protocol for SMA should integrate respiratory muscle training with targeted trunk stabilization exercises. Such a multi-component approach not only enhances respiratory efficiency but also supports motor performance and functional independence, aligning with the holistic demands of SMA management.
Future studies should explore variations in IMT intensity, including progressive loading protocols or higher thresholds (e.g., 40–50% of MIP), to determine the optimal balance between efficacy and fatigue prevention in SMA. The combined application of IMT with expiratory muscle training may further enhance cough efficiency and airway clearance by strengthening both inspiratory and expiratory pumps. Additionally, integrating physiological assessment tools such as diaphragm ultrasound (for muscle thickness and thickening ratio) and surface electromyography could provide valuable insights into respiratory muscle activation patterns and adaptation mechanisms during training. These methodological refinements would contribute to developing more individualized, evidence-based rehabilitation protocols for SMA populations.
From a clinical perspective, the present findings provide practical guidance for the implementation of these interventions in routine SMA management. Inspiratory muscle training at 30% of MIP, performed twice daily for eight weeks, was shown to be feasible and well-tolerated, supporting its safe integration into home-based or supervised programs. Weekly reassessment of MIP ensured individualized load adjustment to maintain optimal intensity. Incorporating targeted trunk control exercises that emphasize neutral alignment, postural stabilization, and dynamic reaching further enhances respiratory and postural synergy and promotes functional independence. For clinical translation, clinicians may consider combining both components within a structured, multidisciplinary framework, with ongoing caregiver education, adherence monitoring, and safety precautions (SpO₂ <92% as a stopping criterion). This protocol can be feasibly integrated into pediatric rehabilitation programs for SMA to improve both respiratory and functional outcomes.
A major strength of this study is that it represents the first randomized controlled trial to evaluate the combined effects of IMT, respiratory rehabilitation, and structured trunk control exercises in children with SMA. The intervention results were analysed not only for statistical significance but also for clinical effect sizes, thereby assessing the real-world applicability of the findings. Methodological rigor was supported by a randomized design and the use of multidimensional, validated outcome measures. Additionally, by employing a stratified randomization strategy to balance SMA subtypes (types 1, 2, and 3), the trial addressed sample heterogeneity, a natural challenge in rare disease research, and enhanced between-group comparability.
Nonetheless, this study has several limitations. Owing to the nature of the intervention and the characteristics of the participants, double-blinding was not feasible for interventionists or outcome assessors. Although this introduces a potential for observer or performance bias, standardized procedures and objective measurement tools were used to minimize its impact. The relatively short duration of the eight-week program may limit the observation of long-term respiratory and functional adaptations. Moreover, although the sample size met the a priori power calculation, the rarity and heterogeneity of SMA restrict the generalizability of the findings, particularly to non-sitter or type 1 populations. Future studies with longer follow-up periods and larger, subtype-specific samples are warranted to confirm the durability and broader applicability of these results.
Conclusions
This randomized controlled study demonstrated that integrating the IMT into pulmonary rehabilitation is a safe, feasible, and effective approach for children with SMA who can sit independently. Over the eight-week multidimensional intervention program, MIP increased by approximately 35–40% from baseline, accompanied by parallel gains in expiratory performance and pulmonary function indices. Combining IMT with trunk control exercises yielded synergistic improvements in respiratory strength, pulmonary capacity, trunk stability, upper limb function, and overall motor performance. These findings support comprehensive rehabilitation protocols tailored to individual needs and highlight the potential for incorporating such strategies into routine clinical practice for ambulatory or sitter-type SMA populations.
Data availability
The datasets used and analysed during the current research are available from the corresponding author (G.A.) upon reasonable request.
Abbreviations
- DMD:
-
Duchenne Muscular Distrophy
- FEV1 :
-
Forced Expiratory Volume in 1 s
- FVC:
-
Forced Vital Capacity
- HFMSE:
-
Hammersmith Functional Motor Scale Expanded
- IMT:
-
Inspiratory Muscle Training
- MIP:
-
Maximal Inspiratory Pressure
- MEP:
-
Maximal Expiratory Pressure
- PCF:
-
Peak Cough Flow
- PEF:
-
Peak Expiratory Flow
- PRG:
-
Pulmonary Rehabilitation Group
- RULM:
-
Revised Upper Limb Module for SMA
- SMA:
-
Spinal Muscular Atrophy
- SMN:
-
Survival Motor Neuron
- SpO2 :
-
Oxygen Saturation
- TCG:
-
Trunk Control Group
- TCMS:
-
Trunk Control Measurement Scale
References
Lefebvre, S. et al. Identification and characterization of a spinal muscular atrophy-determining gene. Cell 80, 155–165 (1995).
Verhaart, I. E. C. et al. Prevalence, incidence and carrier frequency of 5q-linked spinal muscular atrophy - A literature review. Orphanet J. Rare Dis. 12, 1–15 (2017).
Mercuri, E. et al. Diagnosis and management of spinal muscular atrophy: part 1: recommendations for diagnosis, rehabilitation, orthopedic and nutritional care. Neuromuscul. Disord. 28, 103–115 (2018).
Coratti, G. et al. Determining minimal clinically important differences in the Hammersmith functional motor scale expanded for untreated spinal muscular atrophy patients: an international study. Eur. J. Neurol. 31, 1–10 (2024).
Finkel, R. S. et al. Diagnosis and management of spinal muscular atrophy: part 2: pulmonary and acute care; medications, supplements and immunizations; other organ systems; and ethics. Neuromuscul. Disord. 28, 197–207 (2018).
Gozal, D. & Thiriet, P. Respiratory muscle training in neuromuscular disease: long-term effects on strength and load perception. Med. Sci. Sports Exerc. 31, 1522–1527 (1999).
Khirani, S. et al. Longitudinal course of lung function and respiratory muscle strength in spinal muscular atrophy type 2 and 3. Eur. J. Paediatr. Neurol. 17, 552–560 (2013).
Finkel, R. S. et al. Nusinersen versus Sham control in Infantile-Onset spinal muscular atrophy. N Engl. J. Med. 377, 1723–1732 (2017).
Audic, F. et al. Effects of Nusinersen after one year of treatment in 123 children with SMA type 1 or 2: A French real-life observational study. Orphanet J. Rare Dis. 15, 1–10 (2020).
Paul, G. R., Gushue, C., Kotha, K. & Shell, R. The respiratory impact of novel therapies for spinal muscular atrophy. Pediatr. Pulmonol. 56, 721–728 (2021).
Heitschmidt, L. et al. Nusinersen does not improve lung function in a cohort of children with spinal muscular atrophy – A single-center retrospective study. Eur. J. Paediatr. Neurol. 31, 88–91 (2021).
Human, A. & Morrow, B. M. Inspiratory muscle training in children and adolescents living with neuromuscular diseases:a pre-experimental study. South. Afr. J. Physiother. 77, a1577 (2021).
Fauroux, B. et al. Respiratory management of children with spinal muscular atrophy (SMA). Arch. Pédiatrie. 27, 7S29–7S34 (2020).
Peeters, L., Janssen, M., Kingma, I., van Dieen, J. & de Groot, I. Patients with spinal muscular atrophy use high percentages of trunk muscle capacity to perform seated tasks. Am. J. Phys. Med. 98, 1110–1117 (2019).
Flatters, I. et al. The relationship between a child’s postural stability and manual dexterity. Exp. Brain Res. 232, 2907–2917 (2014).
Peeters, L., de Groot, I. & Geurts, A. Trunk involvement in performing upper extremity activities while seated in neurological patients with a flaccid trunk - A review. Gait Posture. 62, 46–55 (2018).
Güneş Gencer, G. & Yılmaz, Ö. The effect of trunk training on trunk control, upper extremity, and pulmonary function in children with Duchenne muscular dystrophy: A randomized clinical trial. Clin. Rehabil. 36, 369–378 (2022).
Amiri, B. & Zemková, E. Trunk stability and breathing exercises superior to foam rolling for restoring postural stability after core muscle fatigue in sedentary employees. Sci. Rep. 15, 1–15 (2025).
Pai, H. C. & Li, C. C. Relationship between trunk control ability and respiratory function in stroke patients: A scoping review and Meta-Analysis. Asian Nurs. Res. (Korean Soc. Nurs. Sci). 17, 61–69 (2023).
Mergen Kilic, S. et al. The effect of aerobic exercise training on patients with type III spinal muscular atrophy. J. Clin. Med. 14, 1–15 (2025).
Human, A., Corten, L., Lozano-Ray, E. & Morrow, B. M. Inspiratory muscle training in children with neuromuscular disorders. South. Afr. J. Physiother. 80, 1–11 (2024).
Vogt, S. et al. Physiotherapeutic management of patients with SMA: A questionnaire-based online survey among physiotherapists within the smartcare network. J. Neuromuscul. Dis. 1, 1–9 (2025).
Society, A. T. S. R. ATS/ERS statement on respiratory muscle testing. Am. J. Respir Crit. Care Med. 166, 518–624 (2002).
Miller, M. R. et al. Standardisation of spirometry. Eur. Respir J. 26, 319–338 (2005).
Dohna-Schwake, C., Ragette, R., Teschler, H., Voit, T. & Mellies, U. Predictors of severe chest infections in pediatric neuromuscular disorders. Neuromuscul. Disord. 16, 325–328 (2006).
Ozal, C., Ari, G. & Gunel, M. K. Inter–intra observer reliability and validity of the Turkish version of trunk control measurement scale in children with cerebral palsy. Acta Orthop. Traumatol. Turc. 53, 381–384 (2019).
Maggi, L. et al. Nusinersen safety and effects on motor function in adult spinal muscular atrophy type 2 and 3. J. Neurol. Neurosurg. Psychiatry. 91, 1166–1174 (2020).
Hill, K., Cecins, N. M., Eastwood, P. R. & Jenkins, S. C. Inspiratory muscle training for patients with chronic obstructive pulmonary disease: A practical guide for clinicians. Arch. Phys. Med. Rehabil. 91, 1466–1470 (2010).
Fernandes, M., Cukier, A. & Feltrim, M. I. Z. Efficacy of diaphragmatic breathing in patients with chronic obstructive pulmonary disease. Chron. Respir Dis. 8, 237–244 (2011).
Ugalde, V. et al. Pursed lips breathing improves ventilation in myotonic muscular dystrophy. Arch. Phys. Med. Rehabil. 81, 472–478 (2000).
Macêdo, T. M., Freitas, D. A., Chaves, G. S., Holloway, E. A. & Mendonça, K. M. Breathing exercises for children with asthma. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD011017.pub2 (2016).
Human, A., Honey, E. & Morrow, B. Inspiratory muscle training in severe spinal muscular atrophy: A case report. Int. J. Ther. Rehabil. 26, 1–19 (2019).
Reddy, S. & Balaji, G. K. Dynamic surface exercise training in improving trunk control and gross motor functions among children with quadriplegic cerebral palsy: A single Center, randomized controlled trial. J. Pediatr. Neurosci. 15, 214–219 (2020).
Koessler, W. et al. 2 years’ experience with inspiratory muscle training in patients with neuromuscular disorders. Chest 120, 765–769 (2001).
Schober, P. & Schwarte, L. A. Correlation coefficients: appropriate use and interpretation. Anesth. Analg. 126, 1763–1768 (2018).
Jolley, C. J., Moxham, J. R. & Muscles Chest Wall, Diaphragm, and other. Encycl Respir Med. 1–4, 632–643 (2006).
Gosselink, R. et al. Impact of inspiratory muscle training in patients with COPD: what is the evidence? Eur. Respir J. 37, 416–425 (2011).
Duruturk, N., Acar, M. & Doğrul, M. I. Effect of inspiratory muscle training in the management of patients with asthma: A RANDOMIZED CONTROLLED TRIAL. J. Cardiopulm. Rehabil Prev. 38, 198–203 (2018).
Beaumont, M. et al. Inspiratory muscle training during pulmonary rehabilitation in chronic obstructive pulmonary disease. Chron. Respir Dis. 12, 305–312 (2015).
Shei, R. J., Paris, H. L. R., Wilhite, D. P., Chapman, R. F. & Mickleborough, T. D. The role of inspiratory muscle training in the management of asthma and exercise-induced bronchoconstriction. Phys. Sportsmed. 44, 327–334 (2016).
Wijngaarde, C. A. et al. Natural history of lung function in spinal muscular atrophy. Orphanet J. Rare Dis. 15, 1–11 (2020).
Trucco, F. et al. Respiratory trajectories in type 2 and 3 spinal muscular atrophy in the iSMAC cohort study. Neurology 96, E587–E599 (2021).
Morrow, B. M. et al. The utility of using peak expiratory flow and forced vital capacity to predict poor expiratory cough flow in children with neuromuscular disorders. South. Afr. J. Physiother. 75, 1–8 (2019).
Veldhoen, E. S. et al. Natural history of respiratory muscle strength in spinal muscular atrophy: a prospective National cohort study. Orphanet J. Rare Dis. 17, 1–11 (2022).
Anan, B. & Karthikbabu, S. Effects of additional inspiratory muscle training on mobility capacity and respiratory strength for school-children and adolescents with cerebral palsy: a randomized controlled trial. Brazilian J. Phys. Ther. 25, 891–899 (2021).
Gandevia, S. C., Butler, J. E., Hodges, P. W. & Taylor, J. L. Balancing acts: respiratory sensations, motor control and human posture. Clin. Exp. Pharmacol. Physiol. 29, 118–121 (2002).
Saunders, S. W., Rath, D. & Hodges, P. W. Postural and respiratory activation of the trunk muscles changes with mode and speed of locomotion. Gait Posture. 20, 280–290 (2004).
Stępień, A., Osiak, T., Rekowski, W. & Wit, A. Neck and trunk muscle strength in children with spinal muscular atrophy is lower than in healthy controls and depends on disease type. Front. Neurol. 12, 1–15 (2021).
Janssen, M. M. H. P., Peeters, L. H. C. & De Groot, I. J. M. Quantitative description of upper extremity function and activity of people with spinal muscular atrophy. J. Neuroeng. Rehabil. 17, 1–13 (2020).
Bulut, N., Alemdaroğlu-Gürbüz, I., Topaloğlu, H., Yılmaz, Ö. & Karaduman, A. The association between trunk control and upper limb functions of children with Duchenne muscular dystrophy. Physiother Theory Pract. 38, 46–54 (2022).
Bieniaszewska, A., Sobieska, M., Steinborn, B. & Gajewska, E. Examination of Upper Limb Function and the Relationship with Gross Motor Functional and Structural Parameters in Patients with Spinal Muscular Atrophy. Biomedicines 11, 1005 (2023).
Cavaloiu, B., Simina, I. E., Vilciu, C., Trăilă, I. A. & Puiu, M. Nusinersen improves motor function in type 2 and 3 spinal muscular atrophy patients across time. Biomedicines 12, 1–16 (2024).
Bieniaszewska, A., Sobieska, M. & Gajewska, E. Functional and Structural Changes in Patients with Spinal Muscular Atrophy Treated in Poland during 12-Month Follow-Up: A Prospective Cohort Study. J Clin. Med 13, 4232 (2024).
O’Hagen, J. M. et al. An expanded version of the Hammersmith functional motor scale for SMA II and III patients. Neuromuscul. Disord. 17, 693–697 (2007).
Acknowledgements
We sincerely thank all the children with SMA and their families for their invaluable participation in this study. We also gratefully acknowledge the support of the Pediatric Pulmonology Clinic staff at Mega University Hospital for their collaboration and assistance.This research was supported by the Scientific and Technological Research Council of Turkey (TUBITAK) under the 1002–Short-Term Support Module (Project No: 223S072).
Funding
This research was supported by the Scientific and Technological Research Council of Turkey (TUBITAK) under the 1002- Short-Term Support Module (Project No: 223S072).
Author information
Authors and Affiliations
Contributions
G.A. conceptualized the study and wrote the main manuscript text, and prepared all main and supplementary file. N.T.v.D.B. advised the research. E.D. contributed to data interpretation. S.K.Y. and S.O. contributed to the acquisition of data. S.K.Y. also contributed to the writing project and revised the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declarations
The study was prospectively registered at ClinicalTrials.gov (NCT06178653; https://clinicaltrials.gov/study/NCT06178653) on 21/02/2024, and approved by the Istanbul Medipol University Non-Interventional Research Ethics Committee (E-10840098-772.02-198). All methods were performed in accordance with the relevant guidelines and regulations. All methods were carried out in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants and their legal guardians.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ata, G., Tunalı Van Den Berg, A.N., Dansuk, E. et al. Inspiratory muscle training and trunk control exercises on respiratory strength and motor function in spinal muscular atrophy: randomized controlled trial. Sci Rep 16, 2700 (2026). https://doi.org/10.1038/s41598-025-32566-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32566-x