Abstract
Residents of long-term care facilities (LTCFs) are at a high risk of severe COVID-19. Our study analyzed the COVID-19 outbreaks, associated cases and case fatality ratio (CFR) in LTCFs from May-2022 to September-2024 in Québec, Canada. An ecological analysis was conducted including all active LTCFs during the study period (n = 471). Regression analyses using generalized estimating equations were used to model odds of an active COVID-19 outbreak, confirmed COVID-19 cases incidence rate ratio (IRR) and ratio of CFR over different periods (wave 7, 2022-23 season, 2023-24 season), regions and LTCF bed capacity. A total of 2,501 outbreaks were recorded, corresponding to 39,089 COVID-19 cases (among residents and health care workers). The odds of an active outbreak, IRR of COVID-19 cases, and ratio of CFR declined significantly during the 2023-24 season compared to wave 7 (-35%, -39% and -24%, respectively; p-values < 0.0001). While larger LTCFs (≥ 40 beds) were up to 7 times more likely to have an active outbreak vs.10–39 beds, COVID-19 IRR and ratio of CFR were comparable across LTCFs’ bed-capacity. In summary, the burden of COVID-19 on LTCF residents has declined from May 2022 to September 2024. Bed capacity seems to predict outbreak occurrence but not virus transmission within LTCFs.
Similar content being viewed by others
Introduction
As of November 24th, 2024, the World Health Organization has estimated that approximately 777 million cases and 7 million deaths to COVID-19 occurred globally since the beginning of the pandemic1. It is well established that older adults are particularly vulnerable to severe COVID-19 illness and death2,3,4. Residents of long-term care facilities (LTCFs) are at even higher risk because of their frailty and the presence of multiple chronic health conditions5. Close living quarters and communal environments inherent to LTCFs facilitate the rapid spread of infectious diseases, making LTCFs hotspots for respiratory infection outbreaks3. A meta-analysis conducted before the widespread use of COVID-19 vaccines showed that the hospitalization and case fatality proportions for SARS-CoV-2 laboratory-confirmed infection among aged care facilities were 37% and 23%, respectively6. An international study (across 22 high-income countries) estimated that the number of care home resident COVID-19 deaths represented 41% of all COVID-19 deaths in 2020–20217. In Canada, from the beginning of the pandemic until July 24th, 2021, the proportion of LTCF residents who died from COVID-19 was 1,640 per 100,000 (number of LTCF beds were used as proxy for number of residents), corresponding to a COVID-19 mortality rate of about 1,100 per 100,000 person-years and to 73.3% of all deaths among elderly (≥ 65 years)8. In the province of Québec, an early report showed that 64% of total COVID-19 deaths occurred in LCTF residents by late November 20209.
Several measures have been implemented in Québec to mitigate SARS-CoV-2 spread in LTCFs, including visitation restrictions, reduced crowding, social distancing, caregiver training, and vaccination10,11,12. Vaccination has drastically reduced the burden of COVID-19 disease among both residents and healthcare workers (HCWs)13,14. For instance, in Québec, the incidence rate of COVID-19 cases and deaths in LTCFs decreased by 92% and 95%, respectively, between February and April 2021, following the administration of the first vaccine dose15.
However, despite high vaccination coverages and a lower virulence of the virus, highly transmissible Omicron variants (such as BA.1 and BA.5)16,17 continued to cause outbreak-associated morbidity and mortality in LTCFs in different countries16,18. According to the European Centre for Disease Prevention and Control, based on data from seven countries, the average case-fatality ratios (CFR) declined from 21.3% during the fourth quarter of 2020 to 3–4% throughout 2022 and until the first quarter of 202316. This is consistent with the lower risk of severe COVID-19 illness due to Omicron variants compared to pre-Omicron variants in the general population19. For example, a large Canadian retrospective study among patients presenting for bloodwork showed a decrease of CFR from mid-July to December 2022 among those aged 70–79 years (0.11%) and ≥ 80 years (1.27%), compared to the period marked by the co-circulation of the Delta and Omicron BA.1 variants (0.97% for 70–79 years and 3.32% for ≥ 80 years)20.
The present study analyzed the outbreaks in LTCFs from May 2022 (epiweek 22) to September 2024 (epiweek 38) in Québec (Canada). The main goal of this ecological study was to estimate the odds of COVID-19 outbreak on any given week, the incidence rate of COVID-19 and COVID-19 CFR in LTCFs in Québec, Canada over different periods, regions and LTCF bed capacity. This study is informative regarding the fact that the last assessment was conducted in Québec before the Omicron surge. Furthermore, to our knowledge, no studies have been conducted during the same surveillance period, or overlapping with it, that have examined the association between LTCF characteristics, and the incidence of COVID-19 outbreak associated morbidity.
Methods
Study design and population
About 9.3% of the Québec population is aged 75 and over21. In 2021, in Québec, there were 54 LTCF beds per 1000 persons aged 75 years or more22. The average LTCF length of stay has been estimated at 823 days (2.3 years) in 2020-2122. Our study includes all LTCFs in Québec active (providing health and personal care services) at any time between May 2022 and September 2024, totaling 471 LTCFs (93.2% were active during the entire study period) and 45,653 beds. Provincial Nosocomial Infection Surveillance Information System (Système d’information sur la surveillance provinciale des infections nosocomiales or SI-SPIN)23 data on COVID-19 active outbreaks were used, aggregated by LTCF and surveillance week (designated as epiweek).
The study period was divided into three distinct surveillance periods defined as follows: May 29th, 2022 - September 3rd, 2022 (wave 7), September 4th, 2022 - August 26th, 2023 (2022-23 season), and August 27th, 2023 - September 21st, 2024 (2023-24 season). Although SARS-CoV-2 seasonality is not established, we followed respiratory viruses’ seasons starting from the 2022-23 season. Data extraction was performed on September 23rd, 2024. Outbreaks that were still active on that date were censored. Data entry in this surveillance system is very rapid (within a week). For COVID-19 weekly cases in the general population (all age groups), we used data from Québec’s COVID-19 surveillance system24.
Omicron BA.4, BA.5* and BQ.1 were the predominant variants during wave 7, while the other two periods had different co-circulating variants (BQ.1*, XBB.1.5*, XBB.1.9* during the 2022-23 season; JN.1*, KP.2*, KP.3* during the 2023-24 season; see Fig. S1).
Variables
Definition of COVID-19 case in a LTCF
A COVID-19 case (including residents and HCWs) was identified through either laboratory testing (polymerase chain reaction or PCR), rapid antigen detection test (RAT), or when an epidemiological link was established with another lab-confirmed or probable (RAT or epidemiological link) case25,26. An epidemiological link is established when a criterion of time, place or person is compatible with transmission from a confirmed case to a susceptible individual17. Specifically, an epidemiological link is established if users (residents or HCW) stayed in the same environment (e.g., room) less than two meters away and without barrier measures in place at any time during the contagious period of the COVID-19 case for 10 min (the criteria of 10 min does not apply between residents). The definition of an LTCF-acquired SARS-CoV-2 infection was modified during the study period. Before July 19th, 2023, a SARS-CoV-2 infection was considered acquired in the LTCF when it occurred 7 days or more after admission. After that date, the delay was shortened to 72 h to account for the shortened incubation period26.
Starting on July 19th, 2023, SARS-CoV-2 screening tests were no longer available for asymptomatic LTCF residents at admission. Asymptomatic LTCF residents were tested if they shared a room with a COVID-19 patient27. Asymptomatic HCWs were tested if they had a close contact with a confirmed case during an outbreak. Definitions for COVID-19-like symptoms are provided in Table S128. A timeline of SARS-CoV-2 testing indications in LTCFs is provided in Table S229,30,31,32,33,34,35.
SARS-CoV-2 outbreak definition
An outbreak was defined as two cases of COVID-19 acquired in an LTCF unit (including both residents and HCWs) occurring within a 10-day period (14 days prior to July 19th, 2023) and linked epidemiologically25. An outbreak was considered over if no new cases linked to the outbreak had been reported in the last 10 days. If two or more outbreaks in different units overlapped in time within the same LTCF, they were treated as a single outbreak for this study, since bed-days were available by LTCF rather than by care unit.
Definition of COVID-19 case in the general population
All general population cases were laboratory-confirmed by PCR. The population eligible for SARS-CoV-2 detection tests was inconsistent over the study period, targeting both symptomatic and asymptomatic individuals (unknown proportions). Of note, reported cases among the general population comprise SARS-CoV-2 outbreak cases, including LTCFs, hospitals and private seniors’ residences36.
Definition of COVID-19 death cases
Any death among LCTF COVID-19 cases that occurred during an outbreak was considered as a COVID-19 death.
LTCF characteristics
Québec health regions were grouped as follows: Montréal, the Greater Montréal Area (Montérégie, Lanaudière, Laurentides, Laval), other urban regions (Capitale-Nationale, Mauricie-Centre-du-Québec, Estrie, Outaouais, Chaudières-Appalaches, Saguenay-Lac-St-Jean) and less populated regions (Bas-St-Laurent, Abitibi-Témiscamingue, Côte-Nord, Nord-du-Québec, Gaspésie-Îles-de-la-Madeleine, Terres-Cries-de-la-Baie-James and Nunavik). This grouping is based on population density, proximity to Montréal and level of urbanicity. LTCF bed capacity was categorized by number of beds as follows: 10–39; 40–64; 65–99; 100–149; ≥1509.
Statistical analyses
For descriptive analysis, we reported the proportion of LTCF with at least one outbreak, the median outbreak duration, the median outbreak size and the median weekly associated cases in all LTCFs stratified by surveillance period, regions and LTCF bed-capacity. The cross-sectional association between the number and size (including both residents and HCWs cases) of outbreaks with LTCF characteristics was assessed using the Wilcoxon rank sum test.
The optimal lag between the weekly number and size of outbreaks in LTCFs (outcomes) and weekly COVID-19 cases in the general population (exposure variable) was identified through cross-correlation analysis. Linear regression models were then used to estimate association between the outcomes and the exposure variable for the three periods (wave 7, 2022-23 season and 2023-24 season)37. To compare the level of association between COVID-19 outbreaks and associated cases with COVID-19 cases in the general population across the three periods, models with an interaction term between total number of COVID-19 in Québec and periods were used.
Logistic regression models were applied to assess the odds ratio of an active outbreak in LTCFs at any given week over periods (reference: wave 7), region (reference: Montréal) and LTCF bed capacity (reference: 10–39 beds); generalized estimating equations (GEE) were used to account for repeated observations per LTCF. GEE Poisson regression models were applied to measure the association between COVID-19 case incidence rates in LTCFs with aforementioned characteristics, using the log of the number of bed-days as the offset. A sensitivity analysis included only cases among LTCF residents, i.e. excluding cases among HCWs. Both logistic and Poisson regression were carried out using both censored and uncensored outbreaks.
GEE logit-binomial regression models were used to measure the association between CFR in LTCFs (only for uncensored outbreaks and including only residents) and the aforementioned characteristics.
Multivariate analyses always included all adjustment variables.
Multiple covariance matrix structures (exchangeable, independent and autoregressive) were tested for the GEE models. Their performance was assessed using the Quasi-likelihood under the Independence model Criterion (QIC), with the lowest QIC value indicating the optimal correlation structure38. The independent structure was deemed optimal and applied to all regression models.
All statistical analyses were carried out using R Studio software (2024.12.0 Build 467). Type I error risk was 0.05. Results were presented as point estimates with 0.95 confidence intervals.
Results
A total of 2,501 outbreaks were recorded, corresponding to 39,089 COVID-19 associated outbreak cases between May 29, 2022 (epiweek 22) and September 21st, 2024 (epiweek 38). Of these, 83.3% (n = 32,581) were outbreaks-associated cases among residents and 16.7% (n = 6,508) among HCWs. About 94.6% of outbreaks-associated cases (n = 36,997) were confirmed by PCR, 4.5% (n = 1,754) by rapid antigenic test and 0.9% (n = 338) by epidemiological link.
As of September 21st, 2024, 95 active outbreaks-associated with 1063 COVID-19 cases were identified and censored. Median number of cases among censored outbreaks was 8 [interquartile range: 5–14].
Figure 1 shows the trend of weekly active outbreaks along with COVID-19 cases in the general population.
Weekly number of COVID-19 active outbreaks in long-term care facilities and total weekly number of outbreaks associated COVID-19 cases and Québec, Canada (epiweek 22-2022 to 38-2024). Total number of COVID-19 cases among the general population in Québec includes COVID-19 outbreak cases in LTCFs, hospitals, private seniors’ residence, and other settings. The number of COVID-19 cases in LTCFs was rescaled (multiplied by 10) to match the same range of values as the total number of COVID-19 cases in the province, allowing them to be displayed on the same y-axis.
COVID-19 outbreak characteristics
Only 54% of LTCFs had at least one outbreak during wave 7, which was expected as this surveillance period was shorter than the other two periods (Table 1). The proportion of LTCFs with at least one outbreak was around 90% for the other two surveillance periods.
Median outbreak duration was 20 days over the entire study period. It increased during the 2022-23 season (24 days vs. 22 days during wave 7, p = 0.0033) but then dropped to its lowest level during the 2023-24 season (17 days, p < 0.0001) (Table 1 and Fig. S2). The outbreak duration increased with bed capacity, with 21 days for those with ≥ 150 beds compared to 19 days for those with 10–49 beds (p < 0.0001).
The median number of reported COVID-19 cases (residents and HCWs) per outbreak was 10 [IQR: 5–21] over the entire study period. Compared to wave 7, outbreak size did not change in the 2022-23 season (p = 0.11) but and, again, declined in the 2023-24 season (p < 0.0001). Outbreak size increased with LTCF bed capacity (8 cases during wave 7 vs. 11 cases during 2023-24 season, p = 0.0002).
The median number of weekly new outbreaks associated COVID-19 cases in all LTCFs over the entire study period was 298 [IQR: 121–418]. Despite a downward trend in the median number of weekly outbreaks-associated cases, there was no significant difference between wave 7 and the 2023-24 season (p = 0.14). Median weekly outbreak-associated cases were significantly lower in the 2023-24 season than in the 2022-23 season (p = 0.047), while significant differences were found between regions (Fig. S2).
Correlation between COVID-19 outbreaks in LTCFs and associated cases and COVID-19 cases in the general population
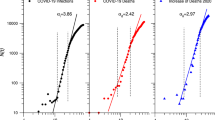
There was a significant correlation (R ≥ 0.89; p-values < 0.0001) between the weekly number of COVID-19 active outbreaks in LTCFs and COVID-19 cases in the general population. A significant correlation (R ≥ 0.85; p-values < 0.0001) was also observed between the number of outbreaks-associated COVID-19 cases in LTCFs and COVID-19 cases in the general population. For the weekly number of outbreaks in LTCFs, the slopes observed during wave 7 and 2022-23 season were comparable (p = 0.69); however, the slope of wave 7 was lower than that of 2023-24 season (p = 0.023). For weekly outbreaks-associated COVID-19 cases in LTCFs, all slopes were comparable (p = 0.42 for wave 7 vs. 2022-23 season; p = 0.34 for wave 7 vs. 2023-24 season). An increase of 5,000 weekly cases in the general population was associated with 53, 119, and 140 additional outbreaks (Fig. S3), and 168, 532, and 508 additional cases in LTCFs (Fig. S4) during wave 7, the 2022-23 season and the 2023-24 season, respectively.
Association between LTCF characteristics, period of study and active COVID-19 outbreak status
Odds of an active outbreak during the 2022-23 season were comparable to those in wave 7 (adjusted OR = 0.91 [95% CI: 0.81–1.03]) but were smaller during the 2023-24 season compared to wave 7 (adjusted OR = 0.65 [95% CI: 0.58–0.73]) (Table 2).
LTCFs located in ‘Other urban and less populated regions’ were more likely to report an outbreak during the study period compared to those in the Montréal region (adjusted OR = 1.21 [95% CI: 1.07–1.38]). Large (≥ 150 beds) LTCFs were 7.23 times (95% CI) more likely to report an active outbreak compared to smaller (10–39 beds) LTCFs.
Association between outbreak-associated COVID-19 cases incidence rate ratio in LTCFs and characteristics
During the 2023-24 season, outbreak-associated COVID-19 cases adjusted incidence rate ratio (IRR) decreased by 39% [95% CI: 31–46] compared to wave 7, while the 2022-23 season showed a comparable incidence rate (adjusted IRR = 0.96 [95% CI: 0.84–1.09]) (Table 3). Adjusted IRR were significantly higher in all regions (from + 21% to + 39%; p values < 0.0001) compared to Montréal. There was no association between COVID-19 adjusted IRR and LTCF bed capacity (multivariate risk reduction= -9% to + 5%; p values > 0.05). A sensitivity analysis including only outbreak-associated cases in LCTF residents and excluding cases in HCWs yielded similar results (Table S3).
Association between case fatality ratio in LTCFs and characteristics
Compared to wave 7, the CFR was 18% [95% CI: 1–33] and 24% [95% CI: 8–38] lower during the 2022-23 and 2023-24 seasons, respectively (Table 4). Higher CFR were found in all regions (adjusted ratio of CFRs = 1.88 to 2.08; p values < 0.0001) compared to Montréal, but there was no statistically significant difference in CFR observed across LTCF bed capacity categories.
Discussion
This is a comprehensive study of all COVID-19 outbreaks, outbreak-associated COVID-19 cases, and COVID-19 deaths that occurred in LTCFs from May 2022 to September 2024 in Québec, Canada. Over those two years, 2,501 outbreaks and 39,089 COVID-19 cases were recorded, representing 9.2% of total cases (n = 423,899) reported in Québec among the general population for the same period. Nearly all LTCFs faced at least one outbreak during the study period. We found that the odds of experiencing an active COVID-19 outbreak on any given week, associated COVID-19 cases incidence and CFRs (COVID-19 deaths) all declined over the study period.
The odds of outbreaks and incidence rate of outbreaks-associated COVID-19 cases declined significantly during the 2023-24 season compared to wave 7. Several factors could have contributed to the phenomenon, such as virological properties of the circulating Omicron variants (e.g. S protein mutations at the N-terminal domain (NTD) and receptor-binding domain)39,40, updated vaccination41, high vaccine coverage against SARS-CoV-242, and the development of population-level immunity from previous infections43. Despite increased evasion of immune response and higher transmission fitness of successive Omicron variants44, transmission might have been hampered by a relative decrease of viral shedding period, which was observed for BA.1 and BA.2 compared to Delta45. However, the decline of viral shedding period may have been inconsistent during the study period46.
Contextual factors might have also contributed to the reported declining trends of COVID-19 in LTCFs. For example, COVID-19 screening tests were unavailable for asymptomatic LTCF residents starting from July 2023, except for those considered close contacts during an outbreak (more details are available in Table S2). This change might have resulted in an under-reporting of COVID-19 outbreaks and associated cases in LTCFs because a significant proportion of elderly individuals could have acquired an asymptomatic or paucisymptomatic SARS-CoV-2 infection despite an ageing immune system and/or the presence of chronic health conditions; this still reflects an improvement of the situation47. A study conducted in Belgium during the beginning of the pandemic (April 8th to May 18th, 2020) showed that 75% of LTCF residents with a positive PCR test were asymptomatic47. Although the cross-sectional design of the survey may have led to an underestimation of the prevalence of residents who may develop symptoms later, a significant proportion of asymptomatic individuals may still be present. Considering that the Omicron variants cause less severe disease than pre-Omicron variants, it is expected that the number of asymptomatic cases would have risen since the Omicron predominance period, which would increase the underreporting48. Finally, it is unlikely that the declining trend in outbreaks-associated COVID-19 cases can be attributed to the exclusion of most asymptomatic HCWs at the mid-study period. A sensitivity analysis showed that the results stand when including only LTCF residents and excluding HCWs (Table S3).
We reported a decline in CFR during the study period. This finding is consistent with population-level surveillance reports in Québec49, other Canadian provinces50, and other countries51,52. For instance, in Canada and the United States, deaths where COVID-19 is listed as an underlying or contributing cause declined from 2022 to 2023 among those aged 65 years and older by nearly 58%53 and 65%54, respectively. Globally, according to the World Health Organization, deaths per 1000 hospitalizations declined from 253 in June 2021 to 41 by November 202451. Studies carried out since the emergence of the BA.5 variant and onward have shown a relatively stable symptoms presentation and comparable or lower severity of illness over time55,56,57,58. However, it is worth mentioning that COVID-19 incidence rate is most likely subject to surveillance bias59. This bias could be observed because users were no longer tested unless they were a close contact of a COVID-19 case starting from the mid-study period. Then, the surveillance bias might have increased the underreporting of cases, decreasing the incidence of COVID-19 outbreak and associated cases; however, our case definition of symptomatic patient is broad (Table S1), allowing the detection of paucisymptomatic residents. For example, a resident with nasal congestion and headache could be systematically being tested for SARS-CoV-2 infection. Also, our case definition includes atypical symptoms such as nausea, vomiting, and abdominal pain which are frequent (29%) among older adults60. This would also apply to COVID-19 deaths, but to a lesser extent and despite this, a decreasing CFR was observed59.
Larger LTCF bed capacity was associated with increased odds of an active outbreak. LTCFs with a bed capacity of 150 or more were about seven times more likely to report an active outbreak at a given time than those with 10–39 beds over all study periods. One of the hypotheses would be that larger LTCFs are more prone to imported COVID-19 cases from the general population than smaller ones, as they tend to have more visitors (restrictions had been progressively relaxed over time between 2022 and 2024) and caregivers61,62. For example, a pre-Omicron study by Orlando et al. reported that LTCFs with ≥ 15 beds had 5.3 times [95% CI: 1.58–22.8] higher odds of reporting an outbreak compared to those with less than 15 over the study period61. Similarly, Lambardo et al. found that LTCFs with > 60 beds were 1.57 times (95% CI: 1.17–2.09) more at risk of experiencing an outbreak compared to those with ≤ 60 beds between March and April 202062. However, even though larger LTCFs were more likely to report an outbreak during the study period, COVID-19 outbreak cases incidence were comparable. Even if larger LTCFs have probably more imported infections (visitors and HCW), SARS-CoV-2 transmission within facilities remained comparable across LTCF bed capacity. The stringent application of the preventive and control measures in the different LTCFs might have contributed to limiting the spread of the virus once it reached a facility.
Interestingly, outbreaks-associated COVID-19 cases incidence rate and CFR were not associated with LTCF bed capacity, while during the first pandemic wave (February 23rd to July 11th, 2020), LCTF bed capacity was strongly correlated with COVID-19 cases incidence rate9. During the first two months of the second wave (August 23rd to November 21st, 2020), incidence rates were relatively comparable across LTCF bed capacity9. Infection prevention and control measures (hand washing, use of personal protective equipment, environment disinfection, proving single isolation room, if possible, with all amenities, and standardised procedure for the management of outbreaks) probably mitigated the association between LTCF bed capacity and virus transmission; in other word irrespective of the LTCF bed capacity, the transmission of the virus was similarly controlled.
Strengths
Our study includes all LTCFs located in Québec, thereby maximizing representativeness. Most outbreaks-associated COVID-19 cases (95%) were identified using PCR tests rather than rapid antigen tests, ensuring high validity. While the ecological study design remains limited for causal inference at the individual level, it remains appropriate to follow trends over time.
Limitations
First, the eligibility for SARS-CoV-2 screening was inconsistent during the study period. Indeed, during the mid-period of the study, asymptomatic residents and HCW (with few exceptions as mentioned previously) (Table S2) were no longer tested. Second, only COVID-19 cases identified during an outbreak were reported here, thus sporadic cases were not considered. This would underestimate the total incidence of COVID-19 cases in LTCFs, but this bias is presumed to be consistent throughout the study period. Third, censored outbreaks were included while analysing the association between outbreak-associated cases incidence and LTCF characteristics. The analysis was rerun in February 2025 with complete data (detailed data not shown), and associations remained consistent. Therefore, the missed cases did not bias our analysis. Forth, starting from July 2023, definitions of an LTCF-acquired SARS-CoV-2 infection and an outbreak were adapted: (i) an infection was considered as LTCF-acquired if the resident tested positive ≥ 3 days after admission (instead of ≥ 7 days prior to July 2023). New admissions in LTCFs are less frequent than in acute care settings, so imported cases from such events are expected to be rare. Consequently, it is unlikely that changes in the LTCF-acquired infection definition for new arrivals would have a significant impact on reported trends; (ii) The time interval without any new case required to close an outbreak was shortened from 14 to 10 days. It is unlikely that this shorter time interval (10 days) will lead to misleadingly declaring an outbreak over, as the viable virus shedding time (5 days from the symptoms onset45 remains far shorter. However, probably this adjustment of the time interval to 10 days could underestimate the CFR, which is defined based on the LTCF COVID-19 deaths that occurred while the outbreak is still active.
Information on the crowding index was not available. However, the occupancy proportion of LTCFs in Québec (either public or private) is, on average, very high (97%)22, suggesting low crowding variability across LCTFs. Also, other predictors of COVID-19 cases were not available, e.g. the number of HCW per resident62 and other LTCF characteristics (e.g. design and use of multi-occupant rooms)63. Finally, also because the currently admitted residents are older, frail adults having lower functional levels. Then, even if information on the health status of admitted residents in LTCFs is limited, we are not expecting a significant variability across different settings.
Conclusion
In summary, 95% of the LTCFs have experienced at least one outbreak during the study period. We reported a progressive decline in active outbreak odds, associated COVID-19 cases and deaths in LTCFs from May 2022 to September 2024. The death burden in LCTFs declined significantly compared to the first year of the pandemic, before vaccines were first administered in this population. As expected, larger LTCFs were more likely to have an active outbreak at any given time during the study period, while incidence rate of outbreaks-associated COVID-19 cases within facilities were comparable, irrespective of LTCFs’ bed capacity. This suggests that probably the preventive and control measures were implemented in a comparable manner, which reduced variability of SARS-CoV-2 transmission within LTCFs irrespective of their bed capacity.
Data availability
These datasets generated and/or analysed during the current study are not publicly available because they are a property of the Ministère de la Santé et des Services sociaux du Québec (MSSS); but are available from the MSSS upon reasonable request and with permission. The access can be requested from the Direction Générale de la gouvernance et des affaires institutionnelles through the following email address [responsable.acces@msss.gouv.qc.ca]. More details are available through the following link: https://www.quebec.ca/en/gouvernement/ministeres-organismes/sante-services-sociaux/cadre-legal-transparence/access-information-protection-personal-information.
References
World Health Organization. COVID-19 cases - WHO COVID-19 dashboard. (2024). https://data.who.int/dashboards/covid19/cases
Taylor, C. A. COVID-19–Associated hospitalizations among U.S. Adults Aged ≥ 18 Years — COVID-NET, 12 States, October 2023–April 2024. MMWR Morb Mortal. Wkly. Rep. 73, 869–875 (2024).
COVID-19 Forecasting Team. Variation in the COVID-19 infection-fatality ratio by age, time, and geography during the pre-vaccine era: a systematic analysis. Lancet. 399, 1469–1488 (2022).
Bartholomew, S. et al. Aging and chronic diseases: a profile of Canadian seniors. https://www.canada.ca/en/public-health/services/publications/diseases-conditions/aging-chronic-diseases-profile-canadian-seniors-report.html (2020).
Honinx, E. et al. Dying in long-term care facilities in europe: the PACE epidemiological study of deceased residents in six countries. BMC Public. Health. 19, 1199. https://doi.org/10.1186/s12889-019-7532-4 (2019).
Hashan, M. R. et al. Epidemiology and clinical features of COVID-19 outbreaks in aged care facilities: A systematic review and meta-analysis. EClinicalMedicine 33, 100771. https://doi.org/10.1016/j.eclinm.2021.100771 (2021).
Comas-Herrera, A. et al. Mortality associated with COVID-19 in care homes: international evidence. (2021). https://ltccovid.org/wp-content/uploads/2021/02/LTC_COVID_19_international_report_January-1-February-1-2.pdf.
Sepulveda, E. R., Stall, N. M. & Sinha, S. K. A comparison of COVID-19 mortality rates among long-term care residents in 12 OECD countries. J. Am. Med. Dir. Assoc. 21, 1572–1574e3 (2020).
Fortin, É., De Serres, G., Gilca, R., Deceunick, G. & Brousseau, N. Portrait de la maladie à coronavirus 2019 (COVID-19) chez les résidents des centres d’hébergement et de soins de longue durée (CHSLD) du Québec. (2020). https://www.inspq.qc.ca/publications/3088-portrait-chsld-covid19.
Ministère de la Santé et des Services sociaux. Directive concernant la gestion des cas et des contacts dans les milieux de vie, réadaptation et d’hébergement. https://publications.msss.gouv.qc.ca/msss/document-003369/?&txt=chsld&msss_valpub&date=DESC (2022).
Ministère de la Santé et des Services sociaux du Québec. Directives CHSLD (DGAPA-007.REV5). https://publications.msss.gouv.qc.ca/msss/document-003206/ (2021).
Ministère de la Santé et des Services sociaux du Québec. Directives applicables à partir du 31 Janvier 2022 Dans les CHSLD, RPA et RI SAPA concernant notamment les personnes proches Aidantes et les visiteurs (DGAPA-021.REV4). https://publications.msss.gouv.qc.ca/msss/document-003308/ (2022).
Canadian Institute for Health Information. COVID-19’s impact on long-term care. https://www.cihi.ca/en/covid-19-resources/impact-of-covid-19-on-canadas-health-care-systems/long-term-care.
Salcher-Konrad, M., Smith, S. & Comas-Herrera, A. Emerging evidence on effectiveness of COVID-19 vaccines among residents of long-term care facilities. J. Am. Med. Dir. Assoc. 22, 1602–1603 (2021).
Fortin, É. et al. Impact of the first vaccine dose on COVID-19 and its complications in long-term care facilities and private residences for seniors in Québec, Canada. Can. Commun. Dis. Rep. Releve Mal Transm Au Can. 48, 164–169 (2022).
European Centre for Disease Prevention and Control. Surveillance of COVID-19 in Long-Term Care Facilities in the EU/EEA 2020–2023. https://www.ecdc.europa.eu/en/publications-data/surveillance-covid-19-long-term-care-facilities-eueea-2021-2023 (2024).
Institut national de santé publique du Québec. Données sur les éclosions de COVID-19 au Québec. https://www.inspq.qc.ca/covid-19/donnees/eclosions (2024).
Breznik, J. A. et al. Early Omicron infection is associated with increased reinfection risk in older adults in long-term care and retirement facilities. EClinicalMedicine 63, 102148 (2023).
Relan, P. et al. Severity and outcomes of Omicron variant of SARS-CoV-2 compared to delta variant and severity of Omicron sublineages: a systematic review and metanalysis. BMJ Glob Health. 8, e012328 (2023).
Skowronski, D. M. et al. Risk of hospital admission and death from first-ever SARS-CoV-2 infection by age group during the delta and Omicron periods in British Columbia, Canada. CMAJ. 195, E1427–E1439 (2023).
Institut de la statistique du Québec. Population du Québec selon l’âge et le genre. https://statistique.quebec.ca/vitrine/egalite/dimensions-egalite/demographie/population-quebec-selon-age-et-sexe (2025).
Dubé-Linteau, A. & Brouard, M. È. Rapport d’appréciation - La performance du système de soins et services aux ainés en CHSLD (Québec, 2022).
Boulais, A. & Desjardins, F. Vigie du SRAS-Cov-2 et des autres virus respiratoires dans les milieux de soins au Québec: Volet éclosions. (2025).
Institut national de santé publique du Québec. Données COVID-19 au Québec. https://www.inspq.qc.ca/covid-19/donnees (2025).
Ministère de la Santé et des Services Sociaux du Québec. Définition de cas de COVID-19-Québec. https://www.msss.gouv.qc.ca/professionnels/documents/coronavirus-2019-ncov/definition-nosologique-COVID-19_2021-07-05.pdf (2021).
Richard, C. SRAS-CoV-2: Définitions des termes en prévention et contrôle des infections dans les milieux de soins. https://www.inspq.qc.ca/publications/3212-definitions-pci-milieux-soins (2023).
Richard, C., Anctil, G., Massicotte, J., Villeneuve, J. & SRAS-CoV-2 Gestion des travailleurs de la santé en milieux de soins. https://www.inspq.qc.ca/sites/default/files/publications/3141-gestion-travailleurs-sante-milieux-soins-v6.pdf (2023).
Ministère de la Santé et des Services sociaux du Québec. Directive concernant la gestion des cas de COVID-19 et des contacts des cas de COVID-19 dans les milieux de vie, réadaptation et d’hébergement - (DGCRMAI-004 REV.5). https://publications.msss.gouv.qc.ca/msss/document-003516/ (2022).
Ministère de la Santé et des Services sociaux du Québec. Tableau des indications d’accès aux TAAN (tests PCR). https://www.cisss-lanaudiere.gouv.qc.ca/fileadmin/internet/cisss_lanaudiere/Professionnels/Biologie_medicale__laboratoires_/Biologie_medicale_Sud/Serologie_et_virologie/Tableau_des_indications_d_acces_aux_TAAN__tests_PCR__2023-07-19.pdf (2023).
Ministère de la Santé et des Services sociaux du Québec. Directive sur la levée de l’isolement des travailleurs de la santé et des services sociaux (DGSP-018.REV11). https://publications.msss.gouv.qc.ca/msss/document-003446/?&txt=DGSP-018&msss_valpub&index=directives-covid-19&date=DESC (2022).
Ministère de la Santé et des Services sociaux du Québec. Directive sur la levée de l’isolement des travailleurs de la santé des établissements du réseau de la santé et des services sociaux (DGSP-018.REV10). https://publications.msss.gouv.qc.ca/msss/document-003396/?&txt=DGSP-018&msss_valpub&index=directives-covid-19&date=DESC (2022).
Ministère de la Santé et des Services sociaux du Québec. Directive sur l’utilisation des tests de dépistage TAAN de la COVID-19 (DGSP-001.REV9). https://publications.msss.gouv.qc.ca/msss/document-003517/ (2022).
Ministère de la Santé et des Services sociaux du Québec. Directive concernant La gestion des Cas et des contacts Dans les milieux de vie, réadaptation et d’hébergement (DGCRMAI-004.REV3). https://publications.msss.gouv.qc.ca/msss/document-003369/ (2022).
Ministère de la Santé et des Services sociaux du Québec. Directive concernant la gestion des cas et des contacts dans les milieux de vie, réadaptation et d’hébergement (GCRMAI-004 REV.4) ERRATUM. https://publications.msss.gouv.qc.ca/msss/fichiers/directives-covid/archives/dgcrmai-004-rev4.pdf (2022).
Ministère de la Santé et des Services sociaux du Québec. Directive concernant la gestion des cas de COVID19 et des contacts des cas de COVID-19 dans les milieux de soins de longue durée et de soins aigus (DGCRMAI-004. REV6). https://publications.msss.gouv.qc.ca/msss/fichiers/directives-covid/archives/dgcrmai-004-rev6.pdf (2023).
Ministère de la Santé et des Services Sociaux du Québec. Directive sur l’utilisation des tests selon le palier d’alerte. https://publications.msss.gouv.qc.ca/msss/document-002853/ (2020).
Zeileis, A. Econometric computing with HC and HAC covariance matrix estimators. J. Stat. Softw. 11, 1–17 (2004).
Pan, W. Akaike’s information criterion in generalized estimating equations. Biometrics 57, 120–125 (2001).
Xia, S., Wang, L., Zhu, Y., Lu, L. & Jiang, S. Origin, virological features, immune evasion and intervention of SARS-CoV-2 Omicron sublineages. Signal. Transduct. Target. Ther. 7, 241 (2022).
Parsons, R. J. & Acharya, P. Evolution of the SARS-CoV-2 Omicron Spike. Cell. Rep. 42, 113444 (2023).
Institut national de santé publique du Québec. Données de vaccination contre la COVID-19 au Québec. https://www.inspq.qc.ca/covid-19/donnees/vaccination (2025).
Brousseau, N., Kiely, M. & Carazo Perez, S. Vaccination contre la COVID-19: Recommandations pour l’automne 2024. 1–15 (2024). https://www.inspq.qc.ca/publications/3516.
Klaassen, F. et al. Changes in population immunity against infection and severe disease from severe acute respiratory syndrome coronavirus 2 Omicron variants in the united States between December 2021 and November 2022. Clin. Infect. Dis. 77, 355–361 (2023).
Guo, H. et al. SARS-CoV-2 omicron: viral evolution, immune evasion, and alternative durable therapeutic strategies. Viruses 16, 697 (2024).
Wu, Y. et al. Duration of viable virus shedding and polymerase chain reaction positivity of the SARS-CoV-2 Omicron variant in the upper respiratory tract: a systematic review and meta-analysis. Int. J. Infect. Dis. 129, 228–235 (2023).
Wannigama, D. L. et al. Increased faecal shedding in SARS-CoV-2 variants BA.2.86 and JN.1. Lancet Infect. Dis. 24, e348–e350 (2024).
Hoxha, A. et al. Asymptomatic SARS-CoV-2 infection in Belgian long-term care facilities. Lancet Infect. Dis. 21, e67 (2021).
Wu, Y. et al. Elder and booster vaccination associates with decreased risk of serious clinical outcomes in comparison of Omicron and delta variant: A meta-analysis of SARS-CoV-2 infection. Front. Microbiol. 14, 1051104 (2023).
Institut de la statistique du Québec. Causes de décès (liste détaillée) selon le sexe, Québec, 2000–2023. https://statistique.quebec.ca/fr/produit/tableau/causes-de-deces-liste-detaillee-selon-le-sexe-quebec (2025).
Government of Canada. COVID-19 daily epidemiology update: Current situation. (2020). https://health-infobase.canada.ca/covid-19/current-situation.html.
World Health Organization. COVID-19 Epidemiological Update. https://cdn.who.int/media/docs/default-source/documents/emergencies/20241224_covid-19_epi_update_special-edition.pdf?sfvrsn=b0a6ddaf_1&download=true (2024).
Konetzka, R. T., Grabowski, D. C. & Mor, V. Four years and more than 200,000 deaths later: lessons learned from the COVID-19 pandemic in US nursing homes. Health Aff. 43, 985–993 (2024).
Government of Canada. Leading causes of death, total population, by age group. https://www150.statcan.gc.ca/t1/tbl1/en/tv.action?pid=1310039401 (2021).
Ahmad, F. B., Cisewski, J. A. & Anderson, R. N. Mortality in the united States - Provisional Data, 2023. MMWR Morb Mortal. Wkly. Rep. 73, 677–681 (2024).
Kirsebom, F. C. M. et al. Vaccine effectiveness against hospitalisation estimated using a test-negative case-control study design, And comparative odds of hospital admission And severe outcomes with COVID-19 sub-lineages BQ.1, CH.1.1. And XBB.1.5 in England. Lancet Reg. Health Eur. 35, 100755 (2023).
Prost, N. et al. Clinical phenotypes and outcomes associated with SARS-CoV-2 Omicron variant JN.1 in critically ill COVID-19 patients: a prospective, multicenter cohort study. Ann. Intensive Care. 14, 101 (2024).
World Health Organization. Initial risk evaluation of JN.1. https://www.who.int/docs/default-source/coronaviruse/18122023_jn.1_ire_clean.pdf?sfvrsn=6103754a_3.
World Health Organization. EG.5 updated risk evaluation. (2023). https://www.who.int/docs/default-source/coronaviruse/eg5-risk-evaluation.pdf?sfvrsn=6e9690e0_4 (2023).
Tancredi, S., Cullati, S. & Chiolero, A. Surveillance bias in the assessment of the size of COVID-19 epidemic waves: a case study. Public. Health. 234, 98–104 (2024).
Tavares, J. et al. Atypical presentation of COVID-19 in older adults: A scoping review. Port J. Public. Health. 41, 198–217 (2023).
Orlando, S. et al. Characteristics of nursing homes and early preventive measures associated with risk of infection from COVID-19 in Lazio region, italy: a retrospective case-control study. BMJ Open. 12, e061784 (2022).
Lombardo, F. L. et al. Adverse events in Italian nursing homes during the COVID-19 epidemic: A National survey. Front Psychiatry 11, (2020).
Stall, N. M. et al. COVID-19 and ontario’s long‐term care homes. J. Elder. Policy. 1, 65–110 (2021).
Acknowledgements
The authors are thankful to Dr Jasmin Villeneuve, a medical advisor at the INSPQ, for checking all information reported in this paper regarding infection control and preventive measures related long-term care facilities. This work was supported by the Ministère de la Santé et des Services sociaux du Québec.
Author information
Authors and Affiliations
Contributions
RD, ER, EF: Writing original draft; All authors: Writing – reviewing & editing; RD, EF: Conceptualization; RD: Data curation; RD: Formal analysis; All authors: Methodology; EF: Supervision; RD: Software; All authors: Validation; All authors: Visualization.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics declarations
As we were operating under the surveillance mandate of the national director of public health, participant consent and approval were automatically waived by the national director. All methods were carried out in accordance with relevant guidelines and regulations.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Doggui, R., Ouakki, M., Boulais, A. et al. SARS-CoV-2 outbreaks in long-term care facilities during the Omicron era in Québec, Canada. Sci Rep 16, 3122 (2026). https://doi.org/10.1038/s41598-025-32967-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-32967-y