Abstract
The World Health Organization has confirmed that asbestos fibres are carcinogenic, claiming that asbestos-related diseases should be eradicated worldwide. Actinolite, amosite, anthophyllite, chrysotile, crocidolite, and tremolite are regulated asbestiform mineral phases. However, in nature, asbestos minerals occur either in a fibrous and asbestiform (original morphology characterized by high length-to-width ratio and provided of high tensile strength and flexibility) or fibrous but not asbestiform appearance. This study used human epithelial cancer cells (A549) and a mouse fibroblast cell line (Balb/c 3T3) to compare the genotoxic and carcinogenic effects of a sample of amphibole asbestos with samples of fibrous not asbestiform named cleavage fragments (CV) obtained by grinding non-asbestiform amphiboles. The results showed that exposure of alveolar lung cells to asbestos and elongated mineral particles, in the habit of cleavage fragments (CF) derived from the grinding of non-asbestiform amphiboles and serpentines, causes cytotoxic effects, oxidative stress and genotoxic damage. Moreover, CF obtained from an actinolite schist induces a transformation effect in the Balb/c 3T3 model. Together, these findings highlight the importance of considering CF as a potential threat to human health since it can cause genotoxic damage by triggering cellular transformation processes that overlap with the mechanisms involved in the carcinogenesis processes of asbestos.
Similar content being viewed by others
Introduction
In 2012, the International Agency for Research on Cancer (IARC) confirmed that chrysotile, crocidolite, amosite, tremolite, actinolite, and anthophyllite are cancerogenic to humans (Group 1)1. All forms of asbestos may be causing extrapleural and pleural mesothelioma and cancer in the lung, larynx, ovary, and other sites, and WHO declares that exposure-related diseases should be eradicated2. Epidemiology data announced by Globocan reported that world mesothelioma mortality in 2020 equals 26,278 cases, 18,621 in men and 7,597 in women. In addition, it is currently estimated that changes in deaths from 2020 to 2040 will increase by 77.9% globally, 38.5% in the EU, and 36.1% in Italy3.
A recent appraisal discriminating fibrous mineral phases minerals has been provided by Goodman4. The elongated mineral particles include both asbestiform and non-asbestiform minerals. The asbestiform habit is connate and gives the fiber bundles flexibility, lengthwise split, and mechanical properties; the non-asbestiform minerals are elongated particles that lack the mechanical properties of the asbestiform analogue. They originate with massive, prismatic, needle-like habitus and might produce cleavage fragments by grinding5.
The regulated definition of asbestos fibre excludes several other fibrous minerals that may possess an asbestiform habitus but do not exhibit all of the properties of asbestos, such as sepiolite, erionite and nemolite6. Currently, only six natural minerals (actinolite, amosite, anthophyllite, chrysotile, crocidolite, and tremolite) are regulated as asbestiform7, which happened because they were used commercially during the establishment period of the regulations. In particular, amosite and crocidolite are commercial names for the mineral species grunerite and riebeckite, respectively.
Asbestiform minerals are fibrous silicates that can separate into thin, flexible, tensile, heat-resistant, and chemically inert fibres. Several mineral phases as the serpentine polymorph antigorite and lizardite, the fibrous zeolite erionite, or the phyllosilicate sepiolite-palygorskite, as well as the non-asbestiform amphiboles, are not regulated as asbestos minerals despite a high length: width ratio corresponding to the definition of Elongated Mineral Particle (EMP) habitus5.
The toxicity of asbestos fibres is mainly due to dimension, shape, and bio-persistence8. In this regard, it is worth remembering that airborne particles are breathable if they fall within the definition of fibre. According to WHO, “breathable fibre” particles showed the following parameters: length > 5 μm, width < 3 μm or have an Aspect Ratio (length of the particles divided by the width, A/R) of at least 3:19.
The National Research Council (1984) reported that the crystal structure defines mineral asbestiform and non-asbestiform habitus10. The asbestiform habitus refers to a crystallization habit of a mineral when the crystals are thin, hairlike fibres. The fibres are thin, long, and like needle-shaped elements with a unidirectional growth. On the other hand, in the non-asbestiform habitus, the structure is characterized by elongated prisms with a lozenge-shaped cross-section. The crystalline growth is not unidirectional.
In nature, minerals such as serpentinite and amphibole can have an asbestos or non-asbestos appearance, depending on their crystalline structure. These differences lead to significant variations in physical and toxicological properties11. In this regard, as reported in different studies12,13, morphological analysis of particles is of fundamental importance since mechanisms associated with toxicity are related to the width, length, and aspect ratio, as well as the surface chemical composition and reactivity of the individual fiber/ELM8.
The Occupational Safety and Health Administration (OSHA) in 1994 indicated that fibres with not-asbestiform aspects are “cleavage fragments (CF)”, referring to amphibole or serpentine that split into fragments with dimensions similar to asbestos fibers14. Although CFs are not asbestiform, they have the same composition as asbestos minerals and aspect ratios falling in the definition of fibers. The chemical-physical similarities between asbestiform and non-asbestiform fibres leave gaps in understanding their role in lung carcinogenesis. In this regard, the indications are unclear, and there is no exhaustive definition of fibres15. However, increasing evidence for a comprehensive approach to risk factors models points to a direct correlation between asbestiform amphibole and variance in mesothelioma potency16, considering the impact of cleavage fragments is not significant.
In this regard, the mechanical action exerted on non-asbestos-like amphibole rocks could generate, through crushing, CFs with aspect ratios of asbestos and, therefore, capable of triggering a carcinogenic process.
In humans, the cancerogenic multiple stages process inducted by asbestos fiber is induced by different mechanisms, including oxidative stress, cell proliferation, aneuploidy, and other chromosomal changes. Non-asbestos silicates during cleavage processes could determine the formation of EMPs, which, if inhaled, could trigger multistep processes similar to asbestos.
In the present study, we investigated amphibole genotoxic and transformation properties by comparing the biological effects of a sample recognized as asbestos, according to current legislation (F3), with not asbestiform fibrous amphiboles of the tremolite-actinolite series (A5, A1, A4) and a sample of fibrous lizardite (A3). CF used in this research were obtained by grinding different non-asbestiform amphiboles derived from different rocks. Resulting samples were tested at varying dosages and exposure times in A549 human epithelial cancer cell line for cytotoxicity, redox status, and DNA damage and in the Balb/c 3T3 cells for transformation assay.
The results comparing the effects of CF exposure to amphibole asbestos revealed CF’s ability to induce genotoxic damage and transformation effects, similar to the carcinogenesis mechanisms of asbestos.
Results
Fibres morphology
The microtextural analysis of the samples provides evidence that the grinding of massive prismatic or elongated amphiboles yielded cleavage fragments without originating asbestiform phases. The subsequent grain size characterization of mineral powders used in the experiments consists of measuring the length (L) and diameter (D) of 100 elongated mineral particles in each sample under Scanning Electron Microscope at 5000x. The L: D was reported in Table 1 and correlated with seven frequency classes. Samples A1, A3, A4, A5 contain cleavage fragments habit with average L: W between 9 and 108. Sample F3 is an asbestiform amphibole, with L: D between 6 and 139. Only two Elongated Mineral Particles on 500 measured have L: D ratio < 3 (from samples A4 and A5). The samples A1, A3, A4, A5 have variable amounts of fibers shorter than 5 μm: 28, 45 and 14% respectively. All EPM in sample F3 are fibers.
The prevailing size class frequency for the asbestiform amphibole is between 10 and 50 μm. Cleavage fragment amphibole samples (A1, A4, A5) show prevailing frequence between 10 and 50 μm.
The % frequence for all samples is consistent with mechanical properties and preferred partings for the corresponding selected natural occurrence.
The supplementary materials implement the geometric characterization of Table 1 by describing the textural setting of addressed samples, supported by the SEM images and EDX analyses of materials source for CFs.
Mineral chemistry
The chemical compositions of the asbestiform and non-asbestiform fragments of amphiboles are reported in Table 2. This analysis confirms the belonging of the amphibole class to the subgroup of calcium amphiboles, as reported by the IMA classification17. Furthermore, according to the classification reported by Leake et al.18, the calcium amphiboles themselves of the analyzed samples belong to the tremolite-actinolite series.
ζ-potential
The ζ-potentials of asbestiform and non-asbestiform amphiboles showed negative values when analyzed in deionized water at 37 °C. The most negative measure is reached by sample F3, which shows − 30.25 ± 0.99 mV at 37 °C. Sample A4, on the other hand, has the most negligible negative value of −20.37 ± 0.43 mV. Noteworthy are the potential values of samples A3 (−25.76 ± 0.37 mV) and A5 (−23.58 ± 1.36 mV), which are lower than F3 but higher than samples A1 (-21.63 ± 0.27 mV) and A4.
Clonogenic assay
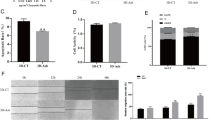
The results of 48 h exposure of Balb/c 3T3 at different concentrations of CF tested are reported in Fig. 1.
The linear model describes the clonogenicity trend for each sample tested. Briefly, it is possible to appreciate a clonogenic capacity decrease per all samples tested, consequently correlating to the concentration of CF to which the cell cultures are exposed. The regression coefficients were highly significant for samples A1, A3 and A5 (P < 0.001), with R2 values of 0.982, 0.959, and 0.905, respectively. Otherwise, samples A4 and F3 reported a P-value < 0.01 with R2 values of 0.829 and 0.832.
The regression line of each sample was used to calculate the no-effects dose and IC50 concentrations. The results obtained are shown in Table 3. Noteworthy, the IC50 in the tested samples is achieved at different CF concentrations except the F3 and A3 samples, where no-effects dose and IC50 values overlap.
Redox status
A time course of DCF shows the magnitude of the redox status, reported as DCF fluorescence intensity, in A549 cell lines, either untreated (Ctrl), exposed for the entire duration of the treatment to varying concentrations of CFs (A1, A3, A4, and A5), or asbestos (F3). In general, A549 cells exposed to sample A5 were more susceptible to redox alterations than other samples. Noteworthy, the results obtained and represented in Fig. 2 describe a similar trend for all samples tested.
Fluorescence units referred to DCF emitted intensity in A549 cells. The redox status was evaluated either untreated (Ctrl) or exposed for 24 h to IC50, of corresponding concentrations of varying CFs (A1, A3, A4, and A5) or asbestos (F3). The results are expressed as the average increase of fluorescence (FU) compared to Ctrl, and a t-test was conducted to compare differences between different groups. P values < 0.05 were considered statistically significant.
In particular, the curves show a first phase corresponding to the third hour, in which there is a slight decrease in ROS. From the third to the 6th hour, an increase in reactive oxygen species (ROS) is possible, a time beyond which a plateau is observed. The significance values reached at the 6 h show a P < 0.01 for all the tested samples. After 18 h, the ROS levels maintained almost equal values for all the samples except F3, where a positive trend was observed (P < 0.001). The results obtained at 24 h indicate a slight negative decline for samples A3 (P < 0.05) and A1 (P < 0.01), maintenance of the observed ROS values for samples A4 (P < 0.05) and A5 (P < 0.001) and a further positive trend for F3 (P < 0.001).
Genotoxic damage
Figure 3 reports the percentage of DNA in the tail (TDNA %) as an indicator of DNA damage in A549 cells when tested at no-effects dose and IC50.
The results show a significant increase in DNA damage only in the cells treated with A3, A5, and F3 samples. Sample A3 induced DNA damage only at the highest concentration corresponding to IC50 and signed as + (P < 0.001). At the same time, sample A5 induced DNA damage both at the lowest tested concentrations, corresponding to no-effects dose and signed as – (P < 0.01), and at the highest concentrations corresponding to IC50 (P < 0.001). Noteworthy, the genotoxic damage induced by F3 sample, corresponding to amphibole, followed the same trend as the A5 sample, albeit with higher damage levels (F3 vs. A5 P < 0.01).
% Tail DNA, as evaluated by the Comet test, in A549 cells after exposure for 12 h to the no-effects dose indicated as - and IC50 concentrations indicated as + (µg/cm2) of CFs or asbestos (F3). The columns report the means + SD of triplicate analyses. Statistical analysis: *P < 0.05, **P < 0.01 and ***P < 0.001 vs. controls.
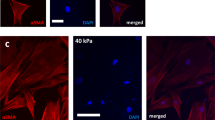
Moreover, fiber morphology (A1, A3, A4, A5, and F3) in the context of the Balb/c 3T3 cells interaction was obtained by SEM analysis and reported in Fig. 4. This microphotography highlights the morphology change of treated cells compared to untreated cells (Ctrl).
Figure 4- F3, representing the in vitro exposure to asbestos, shows a dense network of needle-like fibres incorporated within the entire cellular structure. The images of samples A4 and A5, while showing the CFs inside the cell body, show a class of morphologically heterogeneous particles in which, however, an EMP with a fibrous appearance could justify a lower reproductive viability. Photomicrographs A1 and A3 show a scarcely fibrous appearance that seems to adhere to the surface of the cell membrane, englobing its entire structure.
Transforming effect of CFs
The possible transforming activity of CFs was investigated using the in vitro transformation model of Balb/c 3T3 cells. The range of concentrations used in the transformation tests was chosen based on the results obtained from the cytotoxicity and genotoxicity tests. Specifically, transformation tests were performed only on samples F3, A3 and A5. This choice arises from the awareness that the carcinogenesis process requires mutational events that ROS and genotoxic damage could trigger. The results expressed as the number of foci transformed were related to the number of foci obtained for the negative control sample (Ctrl-), untreated cells, and those present in the cells in contact with the F3 sample (mineral Ctrl+). The latter can be considered a positive control given the recognized carcinogenic activity of asbestos. In addition, a positive control (3-methylchloranthene) was added to the test system to demonstrate the test sensitivity and verify the transforming action.
The results (Fig. 5) show a highly significant effect induced by 3-methylchloranthene (P < 0.001) when compared with the control sample (Ctrl-) as a sign of the high sensitivity of the system.
Furthermore, cell contact with sample F3 at doses between 0.4 and 15.2 µg/cm2 shows a highly transforming effect at doses of 1.9 µg/cm2 (P < 0.001) corresponding to the IC50 and 3.8 µg/cm2, although the latter less significantly (P < 0.01).
No transforming effect is observable at doses below 1.9 µg/cm2, while at concentrations above 3.8 µg/cm2, a toxic action is observed that also inhibits the formation of a homogeneous cell mat in the plates in which the cells were cultured.
Although less significant, a transforming action was observed for sample A5 at higher concentrations than sample A3. For sample A5, the transforming effect is seen at concentrations of 3.8 µg/cm2 (P < 0.05), reaching the peak transforming effect at concentrations of 7.6 µg/cm2 (P < 0.01).
Higher concentrations prevented the formation of a complete and uniform cell monolayer and, thus, transformation foci for A5. On the other hand, no transforming effect was observed for sample A3 at the different doses used.
Discussion and conclusions
Our in vitro study evaluated the effects of CFs as EMPs, overlapping them with the effects of exposure to asbestiform fibres. In particular, the present results assessed some EMPs’ ability to induce cytotoxic and genotoxic effects, redox disequilibrium in lung epithelial cells, and a transforming effect on Balb/c 3T3 cells.
Carcinogenesis is a long and complex multi-step process requiring different DNA mutations, transforming a healthy cell into a cancer cell. If, on one side, DNA mutations could disable the repair system, on the other hand, they may induce cell death. The mutated cells, evading the apoptotic process, start an accelerated mitotic process that can give rise to a tumour mass. Several factors are involved in the multiphasic carcinogenesis process, including redox imbalance with the production of Reactive Oxygen-Nitrogen Species (RNOs), inflammatory reaction with release of proinflammatory mediators such as IL6, TNF-α, TGF-β, the growth factor VEGF promoters angiogenesis, to name a few19.
The results showed that in vitro exposure of alveolar cells to asbestos, besides causing cytotoxic effects, oxidative stress, and genotoxic damage, induced a transforming effect in the Balb/c 3T3 model.
Asbestos carcinogenesis depends on numerous factors, including inhaled dose, dimension, physical durability, surface features, crystallinity, and chemical composition. All these factors have been linked to the formation of RNOs, the release of cytokines and growth factors promoting chronic inflammation, leukocyte invasion, and DNA damage20,21,22. Moreover, zeta potential data show that EMPs exhibit negative potential. The surfaces of negatively charged CFs promote HO° radical formation through reaction with peroxide, Ca2+ sequestration that impairs the mitochondrial apoptotic pathway, and activation of agglomeration processes responsible for chronic tumourigenic inflammation23.
Clonogenic assay performed in cell exposure to asbestiform amphibole (F3), or CFs confirms that fibres can generate widespread injury in the cellular system. However, it is essential to highlight that IC50, after CFs exposure, has always been reached at higher concentrations than the asbestiform amphibole sample except for the CFs obtained by grinding the actinolite schist A3, which showed a response overlapping with asbestiform amphibole, probably from mechanistic effects different from those attributable to the action of asbestos.
The ROS production in cultured cells treated with asbestos or CFs highlights the capacity to trigger oxidative stress by contact with the cell epithelium. Albeit a redox imbalance was observed in all the cells treated with the different CF samples, the A5 sample triggered a remarkable increase in ROS. It is worth noting that the redox imbalance determined by the treatment of cells with sample A5 cannot be related to the dimensions of the fibres themselves or their ratio, which does not differ from the other samples tested except sample F3. Most likely, the surface properties of A5 characterising actinolite from the toxicological point of view have been proven to be very effective in the generation of reactive oxygen species (ROS) and the activation of biological reactivity by an iron-catalysed Haber-Weiss cycle24. However, the mineral chemistry of amphibole in A5 is balanced by assessing Fe2+ to C and B sites. Additionally, the differences in reactive oxygen species (ROS) and other toxic effects observed in samples A1 and A5 could be attributed to the size of the fibers or their ratio, which was more significant for sample A5. Noteworthy, the highest levels of ROS highlighted for sample A5 could be referred to as the almost total absence of fibers with a length less than 5 μm (short fibers), which, unlike the other samples, are at values close to zero as for sample F3. It is well known that the length of the fibers significantly impacts the genesis of stressogenic processes in the cellular comparts due mainly to frustrated phagocytosis25.
Several studies have demonstrated the prominent role of ROS as a second messenger in asbestos toxicity mediation26, mainly in the pulmonary mesothelium27. Moreover, oxidative stress can lead to chronic inflammation, triggering chronic diseases like cancer. Oxidative stress is most likely a prominent player in the induction of genotoxic damage. Different authors reported that asbestos induces DNA damage by directly or indirectly breaking it by triggering ROS production28,29,30. The Comet test highlighted DNA damage in the cells exposed to asbestiform amphibole (F3) and in those exposed to CFs obtained by grinding samples of actinolite schist (A5) and serpentinised peridotite with lizardite vein (A3). In this regard, several observations are noteworthy since the damage is present in the cells exposed to A3 samples at the highest concentration, corresponding to IC50. In contrast, sample A5 caused high levels of damage at the dose corresponding to the no-effects dose. In this regard, the genotoxic damage after A5 exposure reaches values like those reported for cells exposed to asbestiform amphiboles.
In parallel, when samples capable of triggering genotoxic damage were tested by transformation assay performed in Balb/c 3T3 cells, it was possible to observe a carcinogenic potential in both asbestiform and non-asbestiform amphibole. The assay is based on changing the phenotypic features of cells undergoing the first conversion steps from normal cells to neoplastic-like cell foci with oncogenic properties.
In vitro Balb/c 3T3 CTA, highlights a change in the phenotypic features of cells undergoing the first conversion steps from normal cells to neoplastic-like cell foci after amphibole CFs exposure. Different studies have shown that the process follows a multistage process closer to the model stages of in vivo carcinogenesis.
In conclusion, the results of this in vitro study demonstrate that EMPs like asbestos can trigger adverse effects such as cytotoxicity and redox imbalances. However, EMPs obtained by grinding prismatic amphiboles significantly increase genotoxic damage and the transformation potentiality, highlighting typical asbestos mechanisms of the carcinogenesis process.
Considering these results, further in vivo studies are needed to understand EMPs’ actions better. In addition to clarifying the etiopathological mechanisms induced by their inhalation, a reassessment of current legislation would eventually be necessary to accurately identify the danger and risk due to exposure to these particles produced in a workplace.
Materials and methods
Minerals samples
The tested minerals (Table 4) represent non-asbestiform fibrous amphiboles from different host rocks, showing heterogeneous mineralogical and petrographic characteristics.
Sample F3 identifies an asbestiform amphibole, while samples A1, A3, A4, and A5 contain non-asbestiform amphiboles.
Mineralogic and petrographic characterization
Mineral and petrographic characterization was based on reflected and transmitted optical microscopy (OM), SEM/EDS, and µ-Raman spectroscopy to identify the fibrous and EPM minerals and distinguish the phase morphology, composition, and microtextures.
Scanning electron microscopy and energy dispersive spectroscopy
The amphibole mineralogical classification was issued from the quantitative in situ analyses on polished and graphite-coated thin-sections by SEM-EDS methodology using a Vega 3 XML TESCAN (Brno, Czech Republic) equipped with an energy-dispersive X-ray spectroscopy AZtec 2.4 by Oxford Instruments, installed at the Department of Earth, Environment and Life Sciences, University of Genoa. The elemental analysis of minerals was acquired with a counting time of 30 s.
The morphometric qualitative analysis on the ground, gold-coated dust dispersed on membranes was conducted at 2500× magnification, 20 kV of acceleration voltage.
Micro-Raman
Due to the variegated microtextures of sample A3 µ-Raman spectroscopy were performed. Micro-Raman scattering measurements were performed by a single monochromator Horiba Jobin-Yvon Explora_Plus spectrometer (HORIBA, Longjumeau, France) (with a grating of 2400 grooves/mm) equipped with an Olympus BX41 microscope (HORIBA, Longjumeau, France). Raman spectra were excited from the 532 nm line, and the silicon peak was calibrated at 520.5 cm−1, spectral resolution was ~2 cm−1, and instrumental accuracy was ~0.56 cm−1. Raman spectra were collected at 100–1100 cm−1 and 3000–3800 cm−1 for 15 s, with an average of 10 accumulations. In addition, two analysis points were performed on the vein/rock interface and vein.
ζ-potential by dynamic light scattering
The ζ-potential of mineral fibres was investigated to enlighten the possible relationship between surficial charge and fibre activity (Pollastri et al., 2014). Analyses were conducted using a Zetasizer Nano ZS instrument (Malvern Panalytical, Malvern, UK). Each sample was dispersed in deionized water at a concentration of 0.1% (w/w) and analyzed at 37 °C. The results were expressed as an average Z-potential value of six replicates, each consisting of 20 runs per replica.
Preparation of CF
The CF and asbestos samples addressed to in vitro tests were obtained following the protocol below. The amphibole and the lizardite veins were separated from the host rock, described in supplementary materials, with a scalpel and ground by hand in an agate mortar. The CFs were sieved through a 63 μm mesh, then ground by a McChrone micronizer for 20 min at 4 RPM speed, and finally resuspended in 5 ml of distilled water. Subsequently, the suspension was dehydrated by freeze-drying, weighed, resuspended in deionized water at a known concentration, sterilized in an autoclave at 121 °C for 15 min, and sonicated for 5 min before being inoculated into cells.
Cell cultures and treatment conditions
All tests were performed in A549 and Balb/c 3T3 cells obtained from the IRCCS Hospital Policlinico San Martino Cell Bank (Genoa, Italy). Cells lines were grown as monolayer and maintained in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% (v/v) fetal bovine serum (FBS), 100 IU/mL penicillin, and 100 µg/mL streptomycin in a humidified atmosphere containing 5% CO2 at 37 °C. The cells were periodically subcultured or used for the experiments until 80–90% confluence was reached. All tests were conducted by exposing cells to CF obtained after grinding non-asbestiform amphiboles (samples A1-A5) and using a positive control (F3). Untreated cells were considered as a negative control.
Clonogenic assay
Balb/c 3T3 transformation assay (CTA) was conducted to evaluate simultaneously both the cytotoxicity and provide insights into the carcinogenic potential of the tested substances based on their ability to induce morphological changes and affect clonogenicity in vitro. The preliminary inhibition of colony formation (CFE) was performed to evaluate the cytotoxicity by counting the number of colonies formed after CF exposure compared with non-treated cells. Overall, the CFE assay was performed by seeding 250 cells in plates of 60 mm (five replicas by treatment) and maintained in standard growth conditions for 48 h (Minimum Essential Medium (MEM), 10% FBS, 100 IU/mL penicillin, and 100 µg/mL streptomycin). Subsequently, the cells were treated under different concentrations of CF for 48 h, washed three times with PBS and maintained in culture for 10–12 days, changing the medium every 3–4 days. At the end of the incubation period, the cells were washed with PBS, fixed with methanol, and stained with Giemsa solution. Clusters of cells were indicated as colonies if visible macroscopically and containing more than 50 cells. The concentration, which reduces clonal efficiency by 50% (IC50), was calculated by linear regression from the interpolation line equation.
Cells transformation assay
Cells transformation assay (CTA) was conducted to provide insight into the carcinogenic potential of the tested substances based on their ability to induce a morphological change of Balb/c 3T3 in-vitro. The CTA highlights the change in phenotypic characteristics of cells undergoing early conversion steps from normal cells to neoplastic-like cell foci with oncogenic properties. CTA was conducted according to published guidelines (IARC/NCI Working Group, 1985) as amended by Hayashi31.
Overall, Balb/c 3T3 cells in the exponential growth phase were seeded at a density of 2 × 104 cells in 10-mm diameter plates, 4 replicates for each concentration tested, and incubated in MEM containing 10% FBS at 37 °C and 5% CO2. Twenty-four hours after seeding, the cells were exposed to different concentrations of CFs (A3, A5) and fibrous amphibole samples (F3), as reported in Fig. 5. Untreated cells were considered negative controls, while cells treated with 3-methylchloranthene (4µg/ml) were positive controls. After 48 h of incubation, the plates were washed with PBS three times and kept in a fresh culture medium (MEM + 10% FBS) for five days to identify the initiation of the carcinogenesis process. From day 6 to day 17, the cell cultures were maintained in DMEM/F12 with 2% FBS and enriched with insulin (5 µg/ml) to initiate the promotion process. From day 18 to the end of treatment (day 31), the cells were maintained in a DMEM/F12 with 2% FBS. Throughout the test, the medium was changed biweekly. On day 31, the cells were fixed with methanol and stained with Giemsa for examination to assess the formation of transformation foci. Only unequivocally positive foci, greater than 1 mm in diameter and intensely basophilic, were counted, consisting of randomly arranged, multilayered cells with an infiltrating phenotype toward the surrounding monolayer exhibiting contact inhibition. The transforming activity of CF was expressed as the mean number of foci per plate ± SD.
Prooxidant effect of CF
The prooxidant effects were evaluated using 2′,7′-dichlorofluoresceindiacetate (DCF-DA) in A549 cells. DCF-DA is a highly liposoluble probe that easily crosses cell membranes, hydrolyses by cell esterases, and responds quickly to ROS. A549 cells (4 × 103 per well) were seeded in 96-well plates. After 24 h, the cells were loaded with 1 µM of DCF-DA in DMEM (2% FCS), incubated at 37 °C for 30 min, washed twice and treated for each EMPs tested with IC50 concentration and using 8 replicates. Fluorescence analysis was performed at time 0 and again following the time course protocol after incubation at 37 °C using a Perkin-Elmer LS3B spectrophotometer. The fluorescence in each well was recorded at 495 nm Ex and 530 nm Em, the change in redox status was expressed as a unit of fluorescences (FU), and results were reported as mean + SD of the different replications.
Alkaline comet assay
According to previous studies, DNA damage was evaluated by single-cell gel electrophoresis20. Overall, A549 cells were seeded in 6-well plates and treated in duplicate (12 h) with the respective IC50 and NOEL concentrations of different CFs.
After incubation, the cells were removed (0.25% trypsin and 1 mM EDTA), and the viability was assessed by trypan blue exclusion. A viability of at least 80% was a prerequisite to proceed. Subsequently, an aliquot of 10,000 cells was assayed in duplicate, and electrophoresis was performed for 30 min at 300 mA and 25 V (0.86 V cm−1). Dyeing was done using ethidium bromide (2 µg mL−1), and acquisitions by fluorescence microscope equipped with a digital camera at 200× magnification. One hundred randomly selected cell nuclei were taken per sample. Analysis was performed and submitted to CASP (Comet assay software project, http://www.casp.sourceforge.net). The results were expressed as TDNA % reporting DNA percentage in the comet tail.
Statistical analyses
The analyses were performed by JMP software (version 17. SAS Institute Inc., Cary, NC, 1989–2023). The results regarding multiple individual experiments were expressed as means ± SD, and data were analysed by one-way analysis of variance (ANOVA) with post hoc testing using the Bonferroni test. A P value of < 0.05 was considered statistically significant.
Data availability
Data is provided within the manuscript.
References
A review of human carcinogens. Part C: Arsenic, metals, dusts, and fibres. Lyons, International Agency for Research on Cancer, 2009 (IARC monographs on the evaluation of carcinogenic risks to humans, Vol. 100 C). http://monographs.iarc.fr/ENG/Monographs/vol100C/mono100C.pdf (Accessed 13 April 2013) (2009).
WHO World Health Organization, Geneva. (2006).
THE GLOBAL CANCER OBSERVATORY. (https://gco.iarc.fr/tomorrow/en).
Goodman, J. E. et al. Non-asbestiform elongate mineral particles and mesothelioma risk: Human and experimental evidence. Environ. Res. 1 (230) 114578.https://doi.org/10.1016/j.envres.2022.114578 (2023).
Militello, G. M., Gaggero, L. & La Maestra, S. Asbestiform amphiboles and cleavage fragments analogues: overview of critical dimensions, aspect ratios. Exposure Health Eff. Min. 11, 525. https://doi.org/10.3390/min11050525 (2021).
Ross, M., Kuntze, R. A. & Clifton, R. A. in B. Levadie, ed., Definition for Asbestos and Other Health Related Silicates, ASTMSTP 834, American Society for Testing and Materials, Philadelphia, Pa.139–147 (1984).
IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Lyon (FR): International Agency for Research on Cancer (2012).
Aust, A., Cook, P. & Dodson, R. Morphological and chemical mechanisms of elongated mineral particle toxicities. J. Toxicol. Environ. Health B Crit. Rev. 14, 40–75. https://doi.org/10.1080/10937404.2011.556046 (2011).
WHO. Determination of airborne fibre number concentrations. A recommended method, by phase contrast optical microscopy (Membrane Filter Method), World Health Organization, Geneva. (1996).
National Research Council (US) Committee on Nonoccupational Health Risks of Asbestiform Fibers. Asbestiform Fibers: Nonoccupational Health Risks (National Academies Press (US), 1984).
Langer, A. M., Nolan, R. P. & Addison, J. Distinguishing between amphibole asbestos fibers and elongate cleavage fragment of their non-asbestos analogues. In: Mechanism in Fibre Carcinogenesis. 253–267. (New York: Plenum, 1991).
Meeker, G. et al. The composition and morphology of amphiboles from the rainy creek complex near Libby. Mont. Am. Mineral. 88, 1955–1969. https://doi.org/10.2138/am-2003-11-1239 (2003).
Buck, B. J., Goossens, D., Metcalf, R. V., McLaurin, B. & Ren, M. F. Naturally occurring asbestos: potential for human exposure, Southern Nevada. U. S. A. Soil. Sci. Soc. Am. J. 77, 2192–2204. https://doi.org/10.2136/sssaj2013.05.0183 (2013).
Occupational Safety and Health Administration. Rules and regulations, Department of Labor: 29 CFR Parts 1910, 1915 and 1926, 59 FR 40964, RIN: 1218-AB25; occupational exposure to asbestos; August -final rule; appendix B of 1910.1001 (1), (1994).
Keane, M. J., Stephens, J. W., Zhong, B. Z., Miller, W. E. & Wallace, W. E. A study of the effect of chrysotile fiber surface composition on genotoxicity in vitro. J. Toxicol. Environ. Health Part. A 57, 529–541. https://doi.org/10.1080/009841099157494 (1999).
Wylie, A. G., Korchevskiy, A., Segrave, A. M. & Duane, A. Modeling mesothelioma risk factors from amphibole fiber dimensionality: mineralogical and epidemiological perspective. J. Appl. Toxicol. 40, 515–524. https://doi.org/10.1002/jat.3923 (2020).
Hawthorne, F. C. et al. Welch M.D. nomenclature of the amphibole supergroup. Am. Mineral. 97, 2031–2048 (2012).
Leake, B. E. et al. Nomenclature of amphiboles; Report of the subcommittee on amphiboles of the international mineralogical association commission on new minerals and mineral names. Mineral. Mag. 61, 295–310. https://doi.org/10.1180/minmag.1997.061.405.13 (1997).
Aguilar-Cazares, D. et al. Contribution of angiogenesis to inflammation and cancer. Front. Oncol. 12, 1399. https://doi.org/10.3389/fonc.2019.01399 (2019).
La Maestra, S., Micale, R. T., Ferretti, M., Izzotti, A. & Gaggero, L. Attenuation of oxidative stress and chromosomal aberrations in cultured macrophages and pulmonary cells following self-sustained high temperature synthesis of asbestos. Sci. Rep. 10, 8581. https://doi.org/10.1038/s41598-020-65620-x (2020).
Huang, S. X., Jaurand, M. C., Kamp, D. W., Whysner, J. & Hei, T. K. Role of mutagenicity in asbestos fiber-induced carcinogenicity and other diseases. J. Toxicol. Environ. Health B Crit. Rev. 14, 179–245. https://doi.org/10.1080/10937404.2011.556051 (2011).
Kim, H. N. et al. Changes in DNA 8-hydroxyguanine levels, 8-hydroxyguanine repair activity, and hOGG1 and hMTH1 mRNA expression in human lung alveolar epithelial cells induced by crocidolite asbestos. Carcinogenesis 22, 265–269. https://doi.org/10.1093/carcin/22.2.265 (2001).
Pollastri, S. et al. The zeta potential of mineral fibres. J. Hazard. Mater. 15 469–79. https://doi.org/10.1016/j.jhazmat.2014.05.060 (2014).
GualtieriAF, AndreozziGB, Tomatis, M. & Turci, F. Iron from a geochemical viewpoint. Understanding toxicity/pathogenicity mechanisms in iron-bearing minerals with a special attention to mineral fibers. Free Radic. Biol. Med. 133, 21–37. https://doi.org/10.1016/j.freeradbiomed (2018).
Kuroda, A. Recent progress and perspectives on the mechanisms underlying asbestos toxicity. Genes Environ. 12 (43), 46. https://doi.org/10.1186/s41021-021-00215-0 (2021).
Chew, S. H. & Toyokuni, S. Malignant mesothelioma as an oxidative stress-induced cancer: an update. Free Radic. Bio Med. 86, 166–178. https://doi.org/10.1016/j.freeradbiomed.2015.05.002 (2015).
Gào, X. & Schöttker, B. Reduction oxidation pathways involved in cancer development: a systematic review of literature reviews. Oncotarget 8, 51888–51906. https://doi.org/10.18632/oncotarget.17128 (2017).
Walker, C., Everitt, J. & Barrett, J. C. Possible cellular and molecular mechanisms for asbestos carcinogenicity. Am. J. Ind. Med. 21, 253–273. https://doi.org/10.1002/ajim.4700210214 (1992).
Kamp, D. W., Graceffa, P., Pryor, W. A. & Weitzman, S. A. The role of free radicals in asbestos-induced diseases. Free Radic Biol. Med. 12, 293–315. https://doi.org/10.1016/0891-5849(92)90117-y (1992).
Upadhyay, D. & Kamp, D. W. Asbestos-induced pulmonary toxicity: role of DNA damage and apoptosis. Exp. Biol. Med. 228, 650–659. https://doi.org/10.1177/153537020322800602 (2023).
Hayashi, K. et al. Technical modification of the Balb/c 3T3 cell transformation assay: the use of serum-reduced medium to optimize the practicability of the protocol. Altern. Lab. Anim. 36, 653–665. https://doi.org/10.1177/026119290803600609 (2008).
Acknowledgements
The authors thank Elisa Sanguineti, Adrian Yus Gonzalez, and Samanta Annis for the dimensional characterization of amphibole powders used in the experiments under SEM.
Funding
This research received no external funding and was carried out with the financial support of the Analisi delle proprietà microstrutturali, chimico-fisiche di materiali inorganici; determinazioni quantitative della composizione mineralogica di materiali naturali e delle proprietà tecniche dei materiali litici Laboratory funds, DISTAV, University of Genoa. The authors acknowledge the PRIN 2020 POEM (POligEnetic Mélanges: anatomy, significance and societal impacts - CUP 1 D33C22000350001).
Author information
Authors and Affiliations
Contributions
SLM: Conceptualization, writing – planning, methodology, formal analysis, and editing. GM: Conceptualisation, methodology, formal analysis, and writing. SA: methodology. MB: methodology. LG: Supervision, conceptualization, methodology, writing, editing, funding. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
La Maestra, S., Militello, G.M., Alberti, S. et al. Evaluation of the genotoxic and transformation potential induced by asbestos compared to cleavage fragments. Sci Rep 15, 3613 (2025). https://doi.org/10.1038/s41598-025-86325-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86325-z
Keywords
This article is cited by
-
Microbial-Mediated Synthesis of Silver Nanoparticles Using Tilapia Gut E. coli: A Sustainable Approach for Biomedical Applications
BioNanoScience (2026)
-
Cleavage fragments and asbestos fibers: better characterization of minerals and improved cell models are needed to elucidate toxicity differences
Archives of Toxicology (2025)