Abstract
Aerosol deposition in the human respiratory tract significantly impacts drug delivery, pollutant exposure, and radiological protection. While existing models, such as the Multiple-Path Particle Dosimetry (MPPD) and the Human Respiratory Tract Model (HRTM) from International Commission on Radiological Protection (ICRP) provide valuable insights, their reliance on simplified geometries and flow dynamics, limits their ability to accurately predict particle deposition within realistic anatomies. This study integrates Mesh-type Reference Computational Phantoms (MRCPs) with computational fluid-particle dynamics (CFPD) to address these limitations. Our simulations reveal the influence of complex anatomical features, including nasal cavity, trachea, and bronchial regions, on aerosol deposition patterns. For ambient aerosol particles in the diffusion-dominated regime (< 0.5 μm), CFPD results reveal enhanced nasal deposition fractions than ICRP predictions, while, above this size, the ICRP semi-empirical model shows overestimations. In the extrathoracic (ET) airways, deposition distribution varied significantly between ET1 and ET2, with ET2 receiving 65–75% of deposits (near the junction of ET1 and ET2) under certain flow conditions. In the bronchial bifurcation (BB1), deposition efficiency varies with Stokes number and Reynolds number, revealing localized preferential deposition. These findings enhance our understanding of aerosol behaviour and paves the way for more accurate therapeutic and safety models in radiological protection.
Similar content being viewed by others
Introduction
Aerosol deposition in the human respiratory tract is fundamental to several critical areas, including the development of aerosol drug therapies, evaluation of health risks from inhaled contaminants, and the assessment of radiological protection strategies. The biological effects of aerosols depend largely on their deposition location within the respiratory system and their inherent properties, such as radioactivity, chemical toxicity, or pathogenicity. The location and amount of aerosol deposition influence both therapeutic efficacy and potential health risks. For radioactive aerosols, risks extend beyond localized effects due to radiation transport within tissues, potentially leading to increased doses in surrounding tissues over time. Understanding the precise regional distribution of these particles is thus critical for evaluating health risks and optimizing interventions such as aerosol-based therapies.
Experimental and theoretical research has enhanced the understanding of particle deposition across different regions of the respiratory airways. Studies have utilized in vivo, in vitro, and in silico approaches to describe airflow and aerosol deposition within the human respiratory tract. In vivo studies, including radiolabelled imaging and plasma concentration measurements, are often constrained by specific flow conditions, particle sizes, and the need to ensure safety in controlled environments1,2,3,4. In vitro studies, involving experimental visualization using realistic nasal airway geometries, have significantly advanced knowledge of factors influencing drug delivery to the nasal passages5,6,7,8, as well as particle deposition in the upper respiratory and tracheobronchial regions9,10,11,12,13. In silico studies employ computational fluid dynamics (CFD) models, combined with Lagrangian and Eulerian aerosol transport models—collectively termed computational fluid-particle dynamics (CFPD)—to investigate deposition in the upper airways, bronchial, and pulmonary regions14,15,16,17,18,19,20,21,22,23. The effect of turbulence and mesh size on the deposition profile, specifically in the upper airways, was also analysed through in silico studies24,25,26.
Despite advancements, there is limited research on coupling respiratory deposition profiles with radiation dosimetry. Talaat et al.27 simulated aerosol deposition under mouth-breathing conditions and observed significant differences between regional (head and tracheobronchial) and total deposition when compared to the MPPD model. These differences were more pronounced under conditions of low physical activity. The Multiple Path Particle Dosimetry (MPPD) model28,29 and the ICRP Human Respiratory Tract Model (HRTM)30,31 are widely used to predict aerosol deposition and clearance in the respiratory tract. These models divide the respiratory tract into compartments based on radiation sensitivity, assuming uniform cylindrical geometries for each compartment. Deposition is calculated using semi-empirical equations derived from experimental data, factoring in flow rates and particle size. However, these models often fail to account for non-uniform aerosol distribution along the respiratory tract, potentially leading to higher localized radiation doses. Additionally, these models employ simplified, uniform geometries that may not fully capture the complex, non-uniform aerosol distributions within the respiratory system. Such limitations highlight the need for more advanced models that incorporate realistic anatomical details.
Recent advances in computational modeling, particularly through the use of Mesh-type Reference Computational Phantoms (MRCPs) introduced in ICRP Publication 14532, offer more anatomically accurate representations of human organs. These phantoms, derived from high-resolution CT imaging data, are constructed using either polygon mesh (PM) or tetrahedral mesh (TM) formats, providing a superior depiction of complex internal structures, including the respiratory tract. Currently, MRCPs are primarily used for radiation dose estimation in various exposure scenarios via Monte Carlo (MC) simulations.
This study utilizes anatomically detailed MRCP-based geometries for CFPD simulations to capture non-uniform aerosol distribution patterns more accurately, thereby enhancing the precision of radiation dose predictions and therapeutic aerosol delivery efficiency. The objectives are to investigate regional and local deposition patterns of aerosols, particularly in nasal, tracheal, and first bronchial bifurcation regions, and quantify how particle size, airflow conditions, and anatomical features such as the nasal septum affect deposition efficiency. Also, integrating MRCPs into CFPD simulations allows for coupling the resulting deposition profiles with MC simulations to estimate radiation doses from deposited particles. This approach moves beyond the assumption of uniform deposition across regions, significantly improving the accuracy of dose estimates and optimizing aerosol-based therapies.
Methods
Governing equations for continuous and dispersed phases
The fluid flow within the respiratory system is governed by the Navier-Stokes equations, which describe key parameters such as velocity, pressure, density, and viscosity of air as it navigates the complex geometry of the nasal passages, trachea, and bronchi. Aerosol particles, comprising the dispersed phase, are modelled using separate equations that capture forces including drag, gravity, and electrostatic interactions, although the latter were excluded in this study. To investigate local aerosol deposition patterns, we utilized the coupled fluid-particle transport code Aerosolved (v2.0)33,34,35,36, integrated into the OpenFOAM (v1906) platform. Aerosolved operates within an Eulerian-Eulerian framework as a sectional, internally mixed, multi-species aerosol model. The model discretizes particle size distribution (PSD) into sections and solves the transport equation for particle number density, incorporating aerosol evolution source terms. The time-dependent equations are solved using the compressible PISO (Pressure-Implicit with Splitting of Operators) algorithm. Simulations were performed on a parallel computing system comprising 320 nodes, requiring 120 h to simulate 3 s of physical time, at which point convergence was attained.
The model considers an aerosol mixture within a volume V, consisting of two phases: the continuous phase (gas or fluid) and the dispersed phase (droplets or particles) with the latter treated as internally mixed spherical particles. Mass conservation for this mixture follows the continuity equation:
where \(\:\rho\:\) is the mixture density and \(\:\varvec{u}\) the mixture velocity.
Given the capacity of the code to model multispecies in both phases, this equation is expanded for the jth species in the continuous and dispersed phases:
where, \(\:{Y}_{j}\) and \(\:{Z}_{j}\) represent the continuous mass fraction and the dispersed mass fraction subject to the constraint
The momentum conservation equation for the mixture is governed by the Navier-Stokes equation:
where the mixture viscous stress tensor \(({\varvec{\tau\:}}_{\varvec{m}})\) is given by
Here, \(\:\mu\:\) is the mixture viscosity, \(\:p\) is the mixture pressure, \(\:\varvec{I}\) is the identity tensor, and \(\:{\varvec{\tau\:}}_{\varvec{m}}^{\varvec{t}\varvec{u}\varvec{r}\varvec{b}}\) and \(\:{\varvec{\tau\:}}_{\varvec{m}}^{\varvec{d}\varvec{r}\varvec{i}\varvec{f}\varvec{t}}\:\)account for the mixture’s turbulent and drift-induced stresses, respectively.
The mixture energy transport equation formulated in terms of the temperature T is
with \(\:{c}_{v}\) representing heat capacity at constant volume, \(\:{\kappa\:}_{m}\) is the mixture thermal conductivity, \(\:{\kappa\:}_{m}^{turb}\) is the mixture turbulent thermal conductivity, kinetic energy \(\:K=\frac{1}{2}{\left|\varvec{u}\right|}^{2}\), \(\:\dot{Q}\) is the heat source. In the present case, no temperature difference between the phases was assumed, focusing solely on the mixture temperature (298.15 K).
Particle size plays a crucial role in modelling aerosol evolution mechanisms, such as inertial drift, Brownian motion, and interfacial mass and energy transfer. Particle size distributions (PSDs) are often poly-disperse due to the processes that form the particles. In the Eulerian formulation, the PSD, \(\:n(s,\varvec{x},t\)), provides information about the number of particles with mass\(\:\:s\) at position \(\:x\) and time \(\:t\). The product n(s) ds represent the total number of particles per unit volume within the mass range \(\:\left(s,\:s+ds\right)\). This distribution adheres to the population balance equation (PBE) given by37:
The particle size distribution \(\:n\left(s,\varvec{x},t\right)\) is also related to \(\:Z\equiv\:\sum\:_{j}{Z}_{j}\) as follows:
In the sectional formulation, the PSD \(\:n(s,\varvec{x},t\)) is discretized into \(\:\wp\:\) sections allowing the continuous distribution to be approximated as:
where \(\:{N}_{i}\left(\varvec{x},t\right)\) represents the total number of particles per unit volume in the ith section, and \(\:\delta\:\) is the Dirac delta function. In this format, the PBE given in Eq. (8) can be written as:
The source term \(\:J(s,\varvec{x},t)\) or \(\:{\mathcal{J}}_{{M}_{i}}\:\)represents mechanisms such as growth by condensation and coagulation, breakup and nucleation, as well as, contributions to particle transport due to diffusion and drift.
In this study, two primary particle transport mechanisms were considered: diffusion (Brownian motion) and drift (particle inertia):
where, \(\:\mathbb{D}\left(s\right)\) is the size dependent diffusion coefficient and is computed from Stokes-Einstein equation, considering the Brownian diffusivity of a spherical particle as
with \(\:{k}_{B}\) being the Boltzmann constant, \(\:{C}_{c}\) is Cunningham correction factor, \(\:\mu\:\) is mixture viscosity, \(\:d\) being the diameter of particle corresponding to mass \(\:s\).
The relative velocity or drift velocity of the dispersed phase of a mass \(\:s\) with respect to the motion of continuous phase is represented as \(\:\varvec{V}\:\left(s\right)\) such that the particle velocity \(\:\varvec{v}\left(s\right)\:\)for each mass s can be written in the form
Drift velocity model for the particle
The partial differential equation (PDE) describing the motion of the dispersed phase (particle) is given by
The gravity force acting on the particle is given by
with particle volume \(\:{V}_{p}\) and gravitational acceleration \(\:\varvec{g}\).
The drag force \(\:{\varvec{F}}_{D}\) is represented by
with \(\:{A}_{d}\:\)being the cross-section of particle and \(\:{C}_{D}\) the drag coefficient, \(\:{\rho\:}_{g}\) the density of the continuous phase (air in this case).
For sufficiently low particle Reynolds number, i.e., \(\:{Re}_{d}<1,\:{C}_{D}\) takes the form of Stokes drag as,
where
\(\:{\rho\:}_{g}\) and \(\:{\mu\:}_{g}\:\)being the density and viscosity of continuous phase (1.1837 kg m-3 and 1.837E-05 kg m-1 s-1 respectively).
Modifications in the high particle Reynolds number regime
Another module was introduced in the code with the name ‘snDrift’, to incorporate the drag force for high particle Reynolds number following the empirical ‘Schiller-Naumann’ drag coefficient which is valid for \(\:{Re}_{d}<800\:\)38,
Additionally, the drag force is adjusted by introducing the dynamic shape factor (\(\:\chi\:\)) accounting for the effect of particle shape. The dynamic shape factor is defined as the ratio of the actual resistance force on a non-spherical particle to the resistance force on a spherical particle of the same volume and velocity39.
Thus, Eq. (15) reduces to
\(\:\gamma\:\) being the ratio of the continuum density and dispersed phase (aerosol particle) density and \(\:\tau\:\) is the particle relaxation time
In the present simulation, we considered \(\:\chi\:=1\), thus considering a spherical particle.
Integrating Eq. (12) over a size – section (i) and using the expression of \(\:{M}_{i},\) the total sectional number flux (m-2 s-1) at a wall can be written as
This equation is linear with respect to \(\:{M}_{i}\:(=\frac{{N}_{i}}{\rho\:})\). Multiplying this by mass at each section \(({s}_{i}),\) gives the total mass of aerosol particle in section i passing per unit area per unit time, i.e.,
The value of \(\:{\text{F}\text{l}\text{u}\text{x}}_{i}\) can be computed at the walls, inlet, outlet and other regions, to analyse deposition in different respiratory regions.
The above set of equations along with the new modules were solved in the modified Aerosolved code.
Deposition fraction and efficiency calculations
The regional deposition of aerosols was quantified in terms of the deposition fraction (DF) and deposition efficiency (DE) within specific regions of the respiratory tract. The DF was calculated as the ratio of the number of particles depositing on the walls of a region to the number entering the nostril inlet, while the DE was determined by analyzing the fraction of particles that deposited within each region relative to the total number of particles entering that region. These metrics provide a comprehensive understanding of how particles are distributed throughout the respiratory tract, allowing for the identification of regions where deposition is most likely to occur.
In BB1 region, the deposition is mainly governed by inertial impaction and gravitational sedimentation and the combined deposition efficiency (DEi+s) can be expressed in terms of linear or non-linear term of the individual deposition efficiencies (DEi, DEs) with the following form40:
where DE is the impaction and/or sedimentation induced deposition efficiency for a given section and p = 1–3. Yeh and Schum41 and ICRP 6630 proposed another form of
Furthermore, DEi and DEs can be expressed as a function of Stokes number (Stk) and Stk/Re2, where Re is fluid Reynolds number as given by10,42,
where \(\:{a}_{1},\) \(\:{a}_{2},\) \(\:{a}_{3},\) \(\:{a}_{4}\) are fitting parameters and \(\:Stk=\frac{{\rho\:}_{p}{d}^{2}U{C}_{c}\:}{18{\mu\:}_{g}D}\).
To explore the deposition patterns within the bronchial bifurcations, the DE in the BB1 region was analyzed as a function of the Stokes number (Stk), a dimensionless parameter that quantifies the relative importance of particle inertia to viscous forces. The Stokes number was numerically calculated using the average velocity (U) and diameter (D) of the BB1 region, providing a measure of the tendency of particles to deviate from the airflow due to their inertia. This analysis was critical for understanding the role of particle inertia in determining deposition patterns within the bifurcating airways, where the flow is highly complex and the potential for particle deposition is influenced by the interactions between the flow and the airway walls.
Regression analysis for optimization of parameters
To establish a relationship between DE and Stk for the first bifurcation region, it was needed to determine the values of the fitting parameters (\(\:{a}_{1},\) \(\:{a}_{2},\) \(\:{a}_{3},\) \(\:{a}_{4}\) and p as given in Eqs. (25, 27 and 28). Thus, we performed parameter optimization using a robust statistical approach to enhance accuracy. A Python-based routine was developed, and the process involved three steps:
-
(1)
Defining a robust loss function used in regression problems, which was accomplished by employing the Huber loss function,
-
(2)
Defining an objective function to minimize the Huber loss between observed and predicted values while incorporating a regularization term to mitigate overfitting, and finally,
-
(3)
Employing an optimization algorithm tailored for large-scale optimization problems. The L-BFGS-B method was utilized, which is particularly advantageous when dealing with high-dimensional parameter spaces, as it retains a limited amount of historical information about past iterations to build a gradient approximation.
The analysis involved two primary datasets: (a) Dataset 1: Flow rate at nostril: 15 L/min, average Reynolds number (Re) at the outlet of trachea = 903 (b) Dataset 2: Flow rate at nostril: 7 L/min, average Reynolds number (Re) at the outlet of trachea = 412. Both datasets consisted of simulated values for DE and corresponding average Stk and Re values. An objective function was defined to minimize the Huber loss between observed and predicted values while incorporating a regularization term to mitigate overfitting.
The Huber loss43 is defined as follows:
where, y is the true value, f(x) is the predicted value, δ is a threshold parameter that determines the sensitivity of the loss function to outliers. For values of the prediction close to the true value (within δ), the loss is quadratic, while for larger residuals, it becomes linear, thus mitigating the influence of outliers.
In our study, the objective function was crafted to evaluate the discrepancies between the observed deposition efficiency values and those predicted by our model. The objective function was expressed as follows
Additionally, a regularization term was incorporated to penalize large values of the parameters, thus preventing overfitting. This term is typically structured as \(\:\lambda\:\sum\:_{i}{r}_{i}^{2},\) where \(\:{r}_{i}\) represents the model parameters, and λ is a regularization coefficient. The combination of the Huber loss and regularization in the objective function enabled a balance between fitting the data well and maintaining model simplicity.
Input parameters and initial conditions
The volume of air inhaled or exhaled per minute, known as minute ventilation (\(\:\dot{{V}_{n}}\)), is determined by the respiratory rate (\(\:{f}_{R}\)) in breaths per minute and the tidal volume (\(\:{V}_{T}\)), which is the amount of air moved in or out of the lungs with each breath. The relationship is expressed as \(\:\dot{{V}_{n}}={f}_{R}{V}_{T}\). For an average adult at rest, the typical respiratory parameters are a respiratory rate of 12 breaths per minute and a tidal volume of 500 ml per breath, resulting in a minute ventilation of 6 L per minute. As the level of physical activity increases, minute ventilation also rises. In this study, we examined two different minute ventilations that represented low to moderate activity levels, specifically \(\:\dot{{V}_{n}}\) values of 7 L/min and 15 L/min. At the inlet, we considered a polydisperse aerosol (water droplet) size distribution characterized by a log-normal distribution with a count median diameter (CMD) of 1 μm and a geometric standard deviation \(\:\left({\sigma\:}_{g}\right)\) of 2.5, with a total particle concentration of 1010 particles/m3 representing typical ambient aerosol number concentrations44,45,46. The density of these aerosols was assumed to be 1000 kg/m3, equivalent to that of water droplets. The sectional model was employed to resolve the particle size distribution, using 15 sections representing particle diameters ranging from approximately 35.5 nm to 12.67 μm.
Boundary conditions and numerical schemes for airflow and aerosol deposition simulation
To capture the effects of turbulent flow, the Reynolds-averaged Navier-Stokes (RANS) shear stress transport (SST) k-ω turbulence model was applied, where k is the turbulent kinetic energy and ω is the specific turbulent dissipation rate. The use of the SST k-ω model was particularly important for accurately resolving the boundary layer flow near the walls of the respiratory tract, where the velocity gradients are highest. A zero-gradient boundary condition implied no flux and, consequently, no absorption of the gas phase through the walls. The no-slip condition indicated that the flow velocity was zero at the wall. For particle deposition, the zero-gradient extrapolation model (ZGM) boundary condition was implemented to determine the inertial particle velocity at the wall, assuming that the particle velocity at the wall equalled that at the computational cell centre near the wall, provided the velocity was directed towards the wall. Deposition due to diffusion was modelled under the assumption of a perfectly absorbing boundary, meaning that when a particle contacts the wall, it would be immediately deposited. These two conditions are collectively referred to as the deposition boundary condition (Deposition BC) in the simulation.
Given the low dispersed mass fraction, there was only one-way coupling between the continuous and dispersed phases since the particle concentration has a negligible effect on fluid flow or deposition patterns. Each CFPD simulation began at t = 0 and continued until t = 3 s, when the system reached a steady state condition. Particles were injected at t = 0 as well and continued throughout the simulation time. All gradient and Laplacian terms were discretized using a second-order central difference scheme. It should be noted that during unsteady simulations, oscillations were observed in the solution (as indicated by the residuals); therefore, a first-order upwind scheme was initially implemented. Once stability in the solution was achieved, higher-order numerical schemes were applied for subsequent time steps.
Construction of 3D respiratory geometry using MRCP
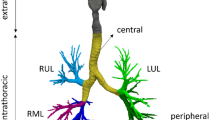
The MRCP-based respiratory geometry utilized in this study was derived from the adult male MRCP dataset, as provided by the International Commission on Radiological Protection32. MRCPs offer a substantial improvement over traditional voxel-based phantoms by providing a more detailed and anatomically accurate representation of human anatomy. These phantoms are constructed by converting voxel-type ICRP reference models into a high-resolution polygon mesh (PM) and tetrahedral mesh (TM) format, facilitating the precise depiction of complex structures, such as the respiratory tract. Respiratory regions within the MRCPs are defined according to the morphometric classification, based on radiation sensitivity (Fig. 1a), as described by the Human Respiratory Tract Model (HRTM) in ICRP Publication 6630. Figure 1c shows the organs in the adult male MRCP, which has a height of 176 cm and a weight of 73 kg, consistent with reference values. The male phantom consists of 2.5 million triangular facets in PM format and 8.2 million tetrahedrons in the TM format. Notably, the respiratory geometry in the MRCP is provided up to the first-generation bronchi (BB1).
In this study, we focused on the respiratory regions extending from the nasal passages (ET1 and ET2) through the trachea (Generation 0, G0), to the first-generation bronchi (BB1, Generation 1, G1). Respiratory geometry files (Fig. 1b) in .obj format (from the PM data) were imported into FreeCAD (v 1.0.0) software for initial processing, including filtering and scaling. The nasal septum, absent in the default MRCP geometry, was explicitly added during this stage to capture its impact on airflow and aerosol deposition.
The processed geometry was subsequently imported into ANSYS Workbench (version 2023 R2) software for further refinement, including mesh generation and the creation of a continuous fluid domain necessary for simulation. The irregular shapes of the respiratory regions, as captured by the MRCP, were preserved, providing a more realistic representation of the respiratory tract than the uniform cylindrical geometries used in traditional models. Since MRCP is constructed from realistic CT voxels, the geometry is inherently irregular. For example, in the HRTM, the trachea is modelled as a uniform cylinder with a length of 0.09 m, an inner diameter of 0.017 m, and a total volume of 2E-5 m3. In contrast, the MRCP trachea is not uniform; it has a maximum height of 0.092 m with a diameter varying approximately between 0.015 m and 0.024 m, and a total volume of 2.52E-5 m3. An overview of these regions is provided in Fig. 2 (a-d).
(a) Schematic Diagram of HRTM classification, (b) Present PM geometry of respiratory regions in adult male MRCP [visualized in FreeCAD (v 1.0.0), www.freecad.org] (c) An overall view of adult male MRCP [visualized in FreeCAD (v 1.0.0), www.freecad.org]
Different respiratory region in adult male MRCP (PM format) [visualized in ANSYS Workbench (version 2023 R2), www.ansys.com]
Modifications in nasal airway geometry
Inspired air entered through the anterior nares into the nasal vestibule and passes into the nasal cavity, which was divided by the nasal septum. The nasal septum, composed of bone and cartilage, separated the cavity into two nostrils and extends from the external nose to the pharynx. This structure increased airflow resistance, slowing inhaled air and promoting the deposition of larger aerosol particles in the upper nasal cavity, thereby reducing their penetration into deeper regions.
In the MRCP, the default nasal airway geometry (ET1 and ET2) does not include the nasal septum. For this study, we modified the geometry to introduce the nasal septum, by cutting anterior and posterior sections with planes, and adjusting the mesh accordingly. The septum was designated as a “wall” boundary, while the two nostrils were defined as the “inlet”. Although the modified septum is not an exact anatomical representation, its inclusion illustrated the effect of increased surface area and altered airflow patterns on particle deposition and fluid dynamics.
Mesh generation and refinement
To capture the complex flow dynamics within the respiratory geometry, unstructured tetrahedral meshes were generated using the ANSYS Workbench Meshing tool. Special attention was given to the near-wall regions, where boundary layers were implemented to accurately capture the flow dynamics close to the walls of the respiratory tract. Eight near-wall prism layer mesh elements were employed, with the thickness of these boundary layers growing progressively towards the bulk region, ensuring that the local velocity profiles were accurately represented.
A grid independence study was conducted by generating five different mesh densities, ranging from course to highly refined, and evaluating their impact on key flow variables. For this, incompressible steady-state simulations were carried out using the SIMPLE algorithm (Semi-Implicit Method for Pressure Linked Equations), which solves the Navier-Stokes equations for fluid flow. Once we achieved an optimal mesh for the present geometry, aerosol transport studies were carried out with the optimal mesh by implementing the transient PISO algorithm.
Results and discussion
Mesh independence
To ensure the reliability and robustness of our simulations, we conducted an extensive mesh independence study, a critical step to verify that the results are not influenced by the discretization of the computational domain. Five different mesh densities were tested across key regions of the respiratory tract, including the nasal passages, trachea, and first-generation bronchi (BB1). These densities ranged from coarse (0.65E6 and 0.71E6 cells) to highly refined (1.26E6, 2.43E6, and 8.86E6 cells), balancing computational efficiency with the ability to capture detailed flow dynamics.
Velocity profiles at different locations along various axes were compared to ensure mesh independence. One such example along the x-axis is illustrated in Fig. 3. The plot compared the velocity magnitude along different line profiles on various cross-sectional planes of the respiratory geometry at time t = 3s, when steady state had already been achieved. The plot demonstrated that the coarser meshes (Mesh 1 and Mesh 2) were inadequate for accurately capturing the complex flow patterns within the respiratory tract, particularly in regions with irregular geometry, such as around the nasal septum and bronchial bifurcations. These meshes exhibited significant discrepancies in velocity profiles. In contrast, the finer meshes (Mesh 3, Mesh 4, and Mesh 5) provided stable and consistent results, with deviations of less than 5% in key flow parameters across critical planes. Mesh 4, comprising approximately 2.43 million cells, was selected for all subsequent simulations, offering an optimal balance between accuracy and computational efficiency. This mesh effectively captured essential flow features, such as the formation of secondary vortices in the bronchial bifurcations and complex flow patterns within the nasal passages.
Computed velocity magnitude at t = 3s for five different unstructured tetrahedral meshes along different line profiles (x direction shown as red horizontal line) on various cross-sectional planes [plotted in python3 code, www.python.org].
Airflow dynamics across respiratory regions
The airflow within the respiratory tract exhibited distinct patterns that varied significantly across different regions, influenced by the complex anatomy of the MRCP-based geometry. In the nasal cavity, airflow entered from the bottom and moved upward, guided by the nasal septum as shown in Fig. 4. The direction of gravity is also indicated in the figure which is the downward when the person is in an upright position. The velocity profiles in the nasal passages revealed the presence of two separate nasal chambers (Fig. 4a), with airflow velocities that varied depending on the proximity to the septum. The non-identical spaces within each nasal cavity, resulting from the use of straight planes to separate the septum from the upper airways, led to slightly higher velocities in one nasal cavity compared to the other. However, this effect diminishes upon reaching the trachea. At the end of the posterior nasal passage, the airway bends towards the pharynx (Fig. 4b). The downward airflow direction in this area is influenced by the anatomical transition from the nasal cavity to the throat i.e. the naso-pharynx region. In this bent region, recirculation zones were observed, influencing the local particle deposition.
In the trachea and first-generation bronchi (BB1), the flow transitioned to a more complex pattern, with the formation of secondary vortices and regions of recirculation (Fig. 4c and d). The tracheal flow was characterized by a zone of high velocity, with lower velocities near the walls. The main feature of this tracheal flow was the formation of non-identical boundary layers (Fig. 5a). The peak velocity was seen to be shifted to one side, resulting in a thicker boundary layer at the other wall. This local velocity pattern differs if a straight tracheal geometry with a 90-degree bend from the oral geometry is assumed. Figure 6a and b illustrated the velocity profiles along the central sagittal plane for inhalation flow rates of 7 L/min and 15 L/min. Two distinct acceleration regions were identified: one prominently located in the in the posterior nasal part, especially in the pharynx and larynx region of ET2 and another at the entrance of trachea. Similarly, the peak turbulent kinetic energy (kmax) was observed at the end of the nasal septum and, more prominently, at the entrance of trachea. This peak was higher for more intense physical activity levels, corresponding to higher flow rates (Fig. 6c and d). These findings are consistent with the numerical simulations conducted on oral model by Wedel et al.47.
At the junction of the trachea and the bronchi, the flow became more complex, with the formation of secondary vortices that influenced the local deposition of aerosols (Fig. 4d). The presence of these vortices is particularly important in determining the deposition patterns of larger particles, which are more susceptible to inertial impaction. Figure 5b revealed the velocity profile at BB1, just before the bifurcation begins. The flow showed a central peak velocity with the flow gradually splitting towards the two bifurcating branches. Figure 5c revealed that the velocity distribution was not identical in the two branches of BB1 due to their non-identical alignment with respect to gravity and varied geometry. As the air stream splits at the junction of the trachea and BB1, new boundary layers were formed near the inner walls of the daughter branches. Counter-rotating vortices were observed at the right bronchi planes compared to the left bronchi due to their difference in alignment with respect to gravity.
Velocity streamlines at different regions for an inhalation flow rate of 7 L/min. [visualized in ParaView, version 5.12.1. https://www.paraview.org]
Air flow direction at selected planes in trachea-bronchial region for steady inhalation flow rate of 7 L/min and 15 L/min [visualized in ParaView, version 5.12.1. https://www.paraview.org].
Impact of nasal septum in the regional deposition
Initially, two sets of simulations were conducted: one with the nasal septum included and the other without it, both for a flow rate of 7 L/min. The inclusion of the nasal septum in the MRCP geometry resulted in a notable shift in the particle deposition patterns, particularly in the ET1 and ET2 regions. The septum altered airflow dynamics, increased the pressure drop across the nasal passage, and enhanced particle deposition, especially for larger particles (> 0.5 μm in diameter) in the anterior and posterior regions of the nasal cavity. CFPD simulations incorporating the septum showed approximately three times more nasal deposition than those without it. Additionally, the septum increased the surface area available for deposition, particularly in regions with heightened airflow velocities. However, deposition in the trachea and BB1 remained largely unaffected, indicating that the septum’s influence is confined to the nasal passages. This highlights the critical role of anatomical features influencing aerosol behaviour. Hence, further simulations and subsequent discussions were carried out for MRCP geometry with the inclusion of the nasal septum region.
Regional aerosol deposition
A detailed analysis of aerosol deposition patterns was performed, focusing on the impact of particle size and inhalation flow rates. The deposition fraction (DF) of aerosol particles was analyzed across various regions of the respiratory tract, including the nasal passages (ET1 and ET2), trachea, and first-generation bronchi (BB1). Figure 7a revealed a characteristic “V” shape in the deposition fraction curves when plotted on a log-log scale, which is consistent with the well-known behaviour of aerosol deposition in the respiratory tract. For smaller particles (with diameters less than 0.5 μm), diffusion was the dominant deposition mechanism, leading to higher deposition fractions in the upper airways. Larger particles (with diameters greater than 0.5 μm) were more influenced by inertial impaction and gravitational settling. A closer view of the deposition pattern at trachea is shown in Fig. 7b, where the deposition for particles with a size (\(\:d\)) of 0.19 μm was almost uniform (minimal depositions are observed at regions where flow is minimal), whereas particles having a size of 3.6 μm were dragged to one side of the trachea which is consistent with the local flow profile as shown earlier in Figs. 5a and 6a. The deposition profile on BB1 for \(\:d=\) 0.19 μm was shown in Fig. 7c which revealed a non-identical deposition pattern at the front and back sides of this region. The deposition profile for \(\:d=\) 12.67 μm in ET1 and ET2 was shown in Fig. 7d, which revealed highly nonuniform deposition in this region.
Nasal deposition due to impaction
For aerosols with particle diameters above 0.2 μm, various authors have reported experimental studies on nasal deposition; however, due to differences in experimental methodologies or evaluation procedures, not all data sets are directly comparable. For example, Lippmann48 determined nasal deposition by external detection of deposited radioactivity after inhalation of radio-labelled particles. In contrast, other experiments measured the change in particle concentration as aerosols were inhaled through the nose and exhaled through the mouth during breath-holding conditions49,50. Rudolf51 measured the total respiratory tract deposition during different breathing situations, i.e., pure nose breathing, pure mouth breathing, nose-in mouth-out and mouth-in nose-out conditions. Since nasal deposition of particles within this aerodynamic size range is due to impaction, the mean aerodynamic deposition efficiency of the nose on inhalation is often plotted as a function of the impaction parameter, denoted in terms of \(\:{d}_{ae}^{2}\dot{{V}_{n}}\), where \(\:{d}_{ae}\) is the aerodynamic diameter of the inhaled particle and \(\:\dot{{V}_{n}}\) is the volumetric flow rate through the nose. This variation revealed a slow increase in deposition efficiency for low values of \(\:{d}_{ae}^{2}\dot{{V}_{n}}\), and it was assumed that the value asymptotically approaches unity at high impaction parameter values. Based on this behaviour, Pattle52 provided an expression for deposition efficiency with the following general hyperbolic form which was later adopted by ICRP Task Group on Lung Dynamics
where \(\:R={d}_{ae}^{2}\dot{{V}_{n}}\), \(\:a\) and \(\:p\) are dimensionless constants, the values of which are given in ICRP 66, 1994a30 for depositions in ET1 and ET2 regions. This expression serves as an average of the experimental data scattered across subjects.
The variability in experimental data stems from differences between subjects, including both inter-subject and intra-subject variability; Rudolf51 provided a more detailed analysis of these effects. Hounam et al.50 conducted experiments with several subjects at flow rates of 5, 10, 20, 30, and 37 L/min using particles of four diameters ranging from 1.6 to 7.3 μm. They established a relationship between deposition efficiency and pressure differences across the nose and mouth, debating whether pressure difference or flow rate should be the primary factor in describing deposition efficiency. Stahlhofen et al.53 also concluded that inter-subject variability and flow-rate-induced intra-subject variability could not be eliminated if deposition efficiency was expressed solely in terms of \(\:{d}_{ae}^{2}\dot{{V}_{n}}\). Instead, they recommended that the pressure drop \(\:{\Delta\:}p\), across the nasal passage be considered a more appropriate parameter governing particle deposition. Consequently, a revised form of aerodynamic inspiratory nasal efficiency was proposed with the form53
where, \(\:N={d}_{ae}^{2}{{\Delta\:}p}^{2/3}\), \(\:b\) and \(\:m\) are dimensionless parameters with values 3.5E-3 and 1 respectively, when \(\:{\Delta\:}p\) is expressed in units of mm of water gauge and \(\:{d}_{ae}\) in µm.
Unfortunately, the nasal pressure drop for subjects involved in other experimental studies is often not reported, and its estimation in real-life scenarios can be challenging. However, to assess the reliability of Eq. (31) and Eq. (32), a similar investigation was conducted in the present study using CFPD simulations based on the MRCP geometry. First, the nasal deposition efficiency (combining contributions from ET1 and ET2) was plotted as a function of impaction parameter \(\:{d}_{ae}^{2}\dot{{V}_{n}}\). Figure 8 revealed that the simulated deposition efficiency was less as compared to experimental values by Lippmann48 and Rudolf51, likely influenced by subject variability. To account for this, the simulated nasal deposition efficiency was compared with the expressions proposed by Stahlhofen et al.53 which incorporated the pressure drop between the inlet nostrils and the ET2 region (Fig. 9a and b for an inhalation flow rate of 7 L/min and 15 L/min respectively). Additionally, the deposition efficiency based on ICRP 6630 assumptions was plotted, where the numerical expression involved only the flow variable (\(\:\dot{{V}_{n}}\)). These figures revealed a significant difference between CFPD results and ICRP HRTM predictions, with the discrepancy being more pronounced at lower levels of physical activity (i.e., lower inhalation flow rates). For instance, at an inhalation flow rate of 7 L/min and aerosol sizes of 1, 2.36, 5.47 and 12.67 μm, the nasal deposition efficiencies (combining ET1 and ET2) obtained from CFPD simulations were 0.22%, 1.14%, 5.92% and 26.05% respectively, while for 15 L/min, these values were 0.44%, 2.37%, 11.88% and 41.32% respectively. In contrast, the corresponding ICRP HRTM predictions using Eq. (31) for an inhalation flow rate of 7 L/min were 2.49%, 12.98%, 47.17%, and 84.66% of inhaled aerosols, while for 15 L/min, the values were 5.46%, 25.4%, 67.5%, and 92.8%, respectively. This observation corroborates recent studies comparing CFPD results with multiple-path particle dosimetry (MPPD) predictions27,54.
Furthermore, it was observed that, the deposition efficiency predicted by Eq. (32) closely matched the CFPD simulations, with only slight deviations due to difference in the geometry under consideration. These findings confirm that the parameter \(\:N={d}_{ae}^{2}{{\Delta\:}p}^{2/3}\) provides a more reliable estimation of nasal deposition than the parameter \(\:R={d}_{ae}^{2}\dot{{V}_{n}}\), which does not account for the effect of the nasal passage’s complex structure, where pressure drop is the primary driver of deposition. Although the ICRP formula may give a conservative estimate of nasal deposition, resulting in higher deposition values in this region, it can lead to significant underestimation of particle deposition in subsequent airway branches, especially during lower physical activity. In this context, deposition efficiency expressed as Eq. (32) is a more reliable method for estimating nasal deposition. In the adult male MRCP geometry, with the present septum configuration, the value of parameter b in Eq. (32) was found to range from 4.94E-3 to 4.21E-3 for inhalation flow rates of 7 L/min and 15 L/min. More accurate parametric optimization can be done with precise nasal septum geometry, which determines the pressure drop in this region. This observation thus highlights the necessity to include a realistic nasal septum in the MRCP model for more accurate predictions of nasal deposition.
Nasal deposition due to diffusion
For particle sizes below 0.5 μm, nasal deposition is primarily governed by Brownian diffusion. However, in this sub-micron size domain, nasal deposition has not been extensively studied in human subjects. Therefore, nasal deposition studies were conducted using surrogate measurements in hollow casts of the nasal passages, pharynx, and larynx regions. Based on various experiments55, the experimental data were plotted as a function of the parameter \(\:D{\left(\dot{{V}_{n}}\right)}^{-1/4}\), where \(\:D\) is the diffusion coefficient associated with the particle size. The thermodynamic deposition in this domain was expressed with a general form of
where \(\:R=D{\left(\dot{{V}_{n}}\right)}^{-1/4}\), \(\:a\) and \(\:p\) are dimensionless constants, the values of which are provided in ICRP 66, 1994a30 for depositions in the ET1 and ET2 regions.
When comparing the present CFPD simulations with the ICRP predictions, it was observed that the thermodynamic deposition in the nasal region predicted by the ICRP HRTM using Eq. (33) was lower than the results obtained from CFPD simulations. For instance, at an inhalation flow rate of 7 L/min, the deposition fractions in the upper airways for aerosol sizes of 36, 54, 82, 125, and 190 nm yielded values of 1.46%, 0.76%, 0.38%, 0.20%, and 0.11% from CFPD simulations, compared to HRTM predictions of 0.05%, 0.03%, 0.03%, 0.04%, and 0.08%, respectively. The observed differences may be attributed to the influence of pressure differences in this region and the effects of turbulence in the flow profile.
Distribution of deposits in the ET airways between regions ET1 and ET2
Another significant deviation from the ICRP HRTM was observed in the distribution of deposits in the ET airways, specifically between regions ET1 and ET2. ICRP Publication 13031 recommends a redistribution of depositions in the nasal region, allocating 65% to ET1 and 35% to ET2 from the total depositions in the ET region. In contrast, our CFPD simulations revealed a reverse trend, with 25–35% of depositions occurring in ET1 and 65–75% in ET2. The ICRP proposed values were determined from in-vivo experiments56 that measured the clearance rate of inhaled particle through four clearance routes (a) clearance by nose blowing, (b) clearance to the GI tract via (i) prompt clearance by swallowing, (ii) rapid mucociliary clearance and (iii) slow mucociliary clearance. Subjects performed nose blows within the first 24 to 48 h, representing a clearance of 50% and 90% of the initial extrathoracic deposits, respectively. For mucociliary clearance, a time period ranging from 1 to 13 h was monitored. These clearance rates were used to define particle retention in the nasal airways, thereby providing an estimate of deposition fractions in ET1 and ET2. However, this experimental scenario differs from the present simulation, where the deposition profile was obtained at a time when both the flow parameters and deposition had reached a steady-state distribution—achieved within 3 s of inhalation. Several factors may contribute to this discrepancy:
-
1.
Natural biological variability, both intra-subject and inter-subject, could explain part of the discrepancy. For monitoring periods ranging from 1 h to several hours, this variability may significantly impact deposition patterns.
Our simulations showed that the majority of deposition occurs at the junction of ET1 and ET2, with a shift towards the ET2 (see Fig. 7d). This location, often known as turbinate zone, is very crucial in determining the local deposition in nasal region. In nose-blowing experimental scenarios, the forceful exhalation may dislodge particles deposited in ET2, especially near the junction. Given that deposition primarily occurs at this junction, it is important to carefully define the boundaries between ET1 and ET2 to ensure accurate assignment of particle distribution.
-
2.
Another significant source of discrepancy could arise from variations in breathing patterns, both between different subjects and between consecutive breaths by the same subject.
-
3.
The head’s orientation during inhalation may also affect deposition fractions in ET1 and ET2. Recent studies of nasal deposition from nasal sprays revealed that deposition patterns in ET1 and ET2 are influenced by the inhalation plume angle. For instance, Foo et al.5 conducted experiments with three different spray devices on a human nasal replica, reconstructed from an MRI of a healthy, non-smoking 53-year-old male. They varied the angle between the nasal cavity base and the spray device tip. Their findings showed that deposition in the anterior region increased with a larger inhaled plume angle, while deposition in the turbinate region increased with a smaller plume angle. In our study, the direction of airflow into the nose was perpendicular to the nostril plane, representing a smaller plume angle, consistent with these experimental observations.
In light of these considerations, while no single factor can fully account for the observed discrepancy, the present study underscores the importance of accurately segmenting the anatomy in this region and highlights key parameters that influence deposition patterns.
Deposition patterns in BB1 region and stokes number analysis
To further understand the deposition behaviour within the bronchial bifurcations, we analyzed deposition efficiencies in the BB1 region as a function of the Stokes number (Stk), a dimensionless parameter that quantifies the relative importance of particle inertia to viscous forces and flow Reynolds number (Re). Figure 10 revealed that the deposition efficiency increased with Stokes number and also was a function of Re (dotted points on the plot), following a non-linear trend that was well-described by a logistic function (marked with smooth line) with parameters \(\:\:{a}_{1}=0.23\), \(\:{a}_{2}=0.64\), \(\:{a}_{3}=1.52\), \(\:{a}_{4}=0.26\) and p = 3 (Eqs. 25, 27 and 28), considering average values of other variables at the entrance of BB1 region. It is to be noted that although the overall trend can be fitted with a logistic function, the local deposition profiles can only be revealed through CFPD simulations.
Conclusion
This study developed a respiratory deposition model that integrates the adult male Mesh-type Reference Computational Phantom (MRCP) with computational fluid-particle dynamics (CFPD), providing a detailed assessment of aerosol deposition in the upper respiratory tract. Quantitative analysis revealed notable deviations in deposition patterns compared to the ICRP Human Respiratory Tract Model (HRTM). Specifically, nasal deposition efficiencies obtained from CFPD simulations for aerosol sizes of 1, 2.36, 5.47, and 12.67 μm at an inhalation flow rate of 7 L/min were 0.22%, 1.14%, 5.92%, and 26.05%, respectively, compared to HRTM predictions of 2.49%, 12.98%, 47.17%, and 84.66%. These differences, also observed at 15 L/min, underscore the role of pressure drop over inhalation flow rate in determining nasal deposition efficiency. A revised parametric value ranging from 4.94E-3 to 4.21E-3 for nasal deposition efficiency was proposed for incorporation into the formula with pressure drop as a variable, as opposed to the flow rate variable used in the ICRP semi-empirical formula.
CFPD simulations revealed significant regional variations in deposition within the extrathoracic (ET) airways, with 25–35% of deposits occurring in ET1 and 65–75% in ET2, contrary to the ICRP-recommended allocation of 65% to ET1 and 35% to ET2. Given that deposition primarily occurs at the junction (often referred as turbinate zone), it is important to carefully define the boundaries between ET1 and ET2 to ensure accurate assignment of particle distribution. These findings were attributed to the influence of nasal septum geometry, which induces flow separation, boundary layer development, and turbulence, all of which significantly affect deposition patterns.
At the first bronchial bifurcation (BB1), the deposition efficiency was found to be dependent on Stokes number and Reynolds number, following a non-linear trend well-characterized by a logistic function. This demonstrates the necessity of CFPD-based simulations to capture localized deposition profiles, particularly in anatomically complex regions.
Despite the fidelity of the MRCP-based model, the ICRP MRCP polygon mesh data is limited to BB1. Extending this geometry to distal airways in future research will enable a more comprehensive understanding of aerosol behaviour across the entire respiratory tract. Integrating these deposition profiles into Monte Carlo radiation transport simulations will enhance dose estimation accuracy for inhaled radioactive aerosols, addressing the limitations of uniform deposition assumptions.
Future work should simulate dynamic respiratory conditions, such as varying tidal volumes and breathing rates, and incorporate particle shape factors linked to fractal dimensions to better predict the deposition of non-spherical aerosols. These refinements will extend the model’s applicability for accurate dose estimates and therapeutic strategies.
Data availability
The data generated in this study, along with new modules introduced in the Aerosolved (v 2.0) code can be available from the corresponding author on reasonable request.
References
Cheng, K. H. et al. In vivo measurements of nasal airway dimensions and ultrafine aerosol deposition in the human nasal and oral airways. J. Aerosol Sci. 27, 785–801 (1996).
Newman, S. P., Pitcairn, G. R., Hirst, P. H. & Rankin, L. Radionuclide imaging technologies and their use in evaluating asthma drug deposition in the lungs. Adv. Drug Deliv. Rev. 55, 851–867 (2003).
Newman, S. P., Pitcairn, G. R. & Dalby, R. N. Drug delivery to the nasal cavity: In vitro and in vivo assessment. Crit. Rev. Ther. Drug Carrier Syst. 21 (1), 21–66 (2004).
Cryan, S. A., Sivadas, N. & Garcia-Contreras, L. In vivo animal models for drug delivery across the lung mucosal barrier. Adv. Drug Deliv. Rev. 59, 1133–1151 (2007).
Foo, M. Y., Cheng, Y. S., Su, W. C. & Donovan, M. D. The influence of spray properties on intranasal deposition. J. Aerosol Med. 20 (4), 495–508 (2007).
Xi, J., Yuan, J. E., Zhang, Y., Nevorski, D. & Wang, Z. Z. Visualization and quantification of nasal and olfactory deposition in a sectional adult nasal airway cast. Pharm. Res. 33 (6), 1527–1541 (2016).
Warnken, Z. N. et al. Personalized medicine in nasal delivery: The use of patient-specific administration parameters to improve nasal drug targeting using 3D-printed nasal replica casts. Mol. Pharm. 15, 1392–1402 (2018).
Chen, J. Z., Kiaee, M. & Martin, A. F. In vitro assessment of an idealized nose for nasal spray testing: Comparison with regional deposition in realistic nasal replicas. Int. J. Pharm. 582, 119341 (2020).
Grgic, B., Finlay, W., Burnell, P. & Heenan, A. In vitro intersubject and intrasubject deposition measurements in realistic mouth–throat geometries. J. Aerosol Sci. 35 (8), 1025–1040 (2004).
Chong, S., Kim & Donald, M. F. Deposition Characteristics of Aerosol Particles in sequentially bifurcating Airway models. Aerosol Sci. Technol. 31 (2–3), 198–220 (2010).
Asgari, M. et al. Development of a realistic human respiratory tract cast representing physiological thermal conditions. Aerosol Sci. Technol. 10, 1–11 (2019).
Warren, H., Finlay, Dino, J., Farina, S., Tavernini & Andrew, R. M. In Vitro Estimation of Tracheobronchial and alveolar doses using filters. Front. Drug Deliv.. 2, 901289 (2022).
Jedelsky, J., Lizal, F. & Jicha, M. Characteristics of turbulent particle transport in human airways under steady and cyclic flows. Int. J. Heat. Fluid Flow. 35, 84–92 (2012).
Banko, A. J., Coletti, F., Elkins, C. J. & Eaton, J. K. Oscillatory flow in the human airways from the mouth through several bronchial generations. Int. J. Heat. Fluid Flow 61, 45–57 (2016).
Darquenne, C. A realistic two-dimensional model of aerosol transport and deposition in the alveolar zone of the human lung. J. Aerosol Sci. 32, 1161–1174 (2001).
Foo, M. Y., Sawant, N., Overholtzer, E. & Donovan, M. D. A simplified geometric model to predict nasal spray deposition in children and adults. AAPS Pharm. SciTech. 19 (7), 2767–2777 (2018).
Kim, J., Xi, J., Si, X., Berlinski, A. & Su, W. C. Hood nebulization: Effects of head direction and breathing mode on particle inhalability and deposition in a 7-month-old infant model. J. Aerosol Med. Pulm D. 27, 209–218 (2014).
Frank, D. O., Kimbell, J. S., Pawar, S. & Rhee, J. S. Effects of anatomy and particle size on nasal sprays and nebulizers. Otolaryngol. Head Neck Surg. 146 (2), 313–319 (2012).
Frederix, E. et al. Simulation of size-dependent aerosol deposition in a realistic model of the upper human airways. J. Aerosol Sci. 115, 29–45 (2018).
Kolanjiyil, A. V. & Kleinstreuer, C. Computationally efficient analysis of particle transport and deposition in a human whole-lung-airway model. Part I: Theory and model validation. Comput. Biol. Med. 79, 193–204 (2016).
Kolanjiyil, A. V. & Kleinstreuer, C. Computational analysis of aerosol-dynamics in a human whole-lung airway model. J. Aerosol Sci. 114, 301–316 (2017).
Longest, P. W. & Xi, J. Effectiveness of direct lagrangian tracking models for simulating nanoparticle deposition in the upper airways. Aerosol Sci. Technol. 41, 380–397 (2007).
Longest, P. W. & Xi, J. Computational investigation of particle inertia effects on submicron aerosol deposition in the respiratory tract. J. Aerosol Sci. 38 (1), 111–130 (2007).
Zhang, Z., Kleinstreuer, C. & Kim, C. Micro-particle transport and deposition in a human oral airway model. J. Aerosol Sci. 33, 1635–1652 (2002).
Longest, P. W. & Holbrook, L. T. In silico models of aerosol delivery to the respiratory tract—development and applications. Adv. Drug Deliv. Rev. 64 (4), 296–311 (2012).
Koullapis, P. G. et al. Regional aerosol deposition in the human airways: The SimInhale benchmark case and a critical assessment of in silico methods. Eur. J. Pharm. Sci. 113, 1–18 (2017).
Talaat, K. et al. Radiation Dosimetry of Inhaled Radioactive aerosols: CFPD and MCNP Transport Simulations of radionuclides in the lung. Sci. Rep. 9, 17450 (2019).
Anjilvel, S. & Asgharian, B. A multiple-path model of particle deposition in the rat lung. Fund Appl. Toxicol. 28, 41–50 (1995).
Asgharian, B., Hofmann, W. & Bergmann, R. Particle deposition in a multiple-path model of the human lung. Aerosol Sci. Technol. 34, 332–339 (2001).
ICRP. Human Respiratory Tract Model for Radiological Protection. vol. 24(1–3) (ICRP Publication 66. Ann. ICRP, 1994).
ICRP. Occupational Intakes of Radionuclides: Part 1. vol. 44(2) (ICRP Publication 130. Ann. ICRP, 2015).
ICRP. Adult Mesh-type Reference Computational Phantoms. vol. 49(3) (ICRP Publication 145. Ann. ICRP, 2020).
Frederix, E. M. A. et al. Extension of the compressible piso algorithm to single-species aerosol formation and transport. Int. J. Multiph. Flow. 74, 184–194 (2015).
Frederix, E. M. A., Stanic, M., Kuczaj, A. K., Nordlund, M. & Geurts, B. J. Characteristics-based sectional modeling of aerosol nucleation and condensation. J. Comput. Phys. 326, 499–515 (2016).
Frederix, E. et al. Application of the characteristics-based sectional method to spatially varying aerosol formation and transport. J. Aerosol Sci. 104, 123–140 (2017).
Francesco Lucci, E., Frederix, A. K. & Kuczaj. AeroSolved: Computational fluid dynamics modeling of multispecies aerosol flows with sectional and moment methods. J. Aerosol Sci. 159, 105854 (2022).
Friedlander, S. Smoke Dust and Haze: Fundamentals of Aerosol Dynamics (2nd ed.) (Oxford University Press, 2000).
Clift, R., Grace, J. R. & Weber, M. E. Bubbles Drops and Particles. (Academic press, 1978).
Hinds, W. C. Aerosol Technology. (2nd ed.) (John Wiley & Sons, Inc., 1999).
Clement, K., Zhang, Z., Chong, S. & Kim. Combined inertial and gravitational deposition of microparticles in small model airways of a human respiratory system. J. Aerosol Sci. 38 (10), 1047–1061 (2007).
Yeh, H. C. & Schum, G. M. Models of human lung airways and their application to inhaled particle deposition. Bull. Math. Biol. 42, 461–480 (1980).
Ou Cuiyun, H., Jian, D. & Qihong Particle deposition in human lung airways: Effects of Airflow, particle size, and mechanisms. Aerosol Air Qual. Res. 20, 2846–2858 (2020).
Huber Peter, J. Robust estimation of a location parameter. Ann. Stat. 53 (1), 73–101 (1964).
Diapouli, E., Chaloulakou, A. & Koutrakis, P. Estimating the concentration of indoor particles of outdoor origin: A review. J. Air Waste Manag. Assoc. 63 (10), 1113–1129 (2013).
Hussein et al. Indoor particle concentrations, size distributions, and exposures in Middle Eastern Microenvironments. Atmosphere 11 (1), 41 (2020).
Ridolfo, S., Amato, F. & Querol, X. Particle number size distributions and concentrations in transportation environments: A review. Environ. Int. 187, 108696 (2024).
Jana Wedel, P. et al. Anatomy matters: The role of the subject-specific respiratory tract on aerosol deposition — A CFD study. Comput. Methods Appl. Mech. Eng. 401 (Part A), 115372 (2022).
Lippmann, M. Deposition and clearance of inhaled particles in the human nose. Ann. Otol. Rhinol. Laryngol.. 79 (3), 519–528 (1970).
Landahl, H. D. & Tracewell, T. Penetration of air-borne particulates through the human nose. J. Ind. Hyg. Toxicol. 31 (1), 55–59 (1949).
Hounam, R. F., Black, A. & Walsh, M. The deposition of aerosol particles in the nasopharyngeal region of the human respiratory tract. J. Aerosol Sci. 2 (1), 47–61 (1971).
Rudolf, G. Deposition von Aerosolteilchen in der Nase. Diploma thesis. (University Frankfurt, 1975).
Pattle, R. E. The retention of gases and particles in the human nose. Inhaled Particles and Vapours: Proceedings of an International Symposium 302–311 (Pergamon Press, 1961).
Stahlhofen, W., Rudolf, G. & James, A. C. Inter comparison of experimental regional aerosol deposition data. J. Aerosol Med. 2 (3), 285–308 (1989).
Richard, A. et al. New Approach Methodology for Assessing Inhalation Risks of a Contact Respiratory Cytotoxicant: Computational Fluid Dynamics-Based Aerosol Dosimetry Modeling for Cross-Species and In Vitro Comparisons. T182oxicol.Sci.182 (2), 243–259 (2021).
Swift, D. L. et al. Inspiratory deposition of ultrafine particles in human nasal replicate cast. J. Aerosol Sci. 23, 65–72 (1992).
Smith, J. R. H. et al. An experimental study of clearance of inhaled particles from the human nose. Exp. Lung Res. 37, 109–129 (2011).
Acknowledgements
The authors gratefully acknowledge the supercomputer facilities of the Variable Energy Cyclotron Centre (VECC) and Bhabha Atomic Research Centre (BARC). They also extend their gratitude to VECC for providing the ANSYS Workbench (version 2023 R2) platform, which was used to carry out the mesh preparation for the geometry. The authors also appreciate the fruitful discussions with Dr Francesco Lucci regarding the present version of the AeroSolved code.
Author information
Authors and Affiliations
Contributions
S Anand and H K Patni planned and supervised the project. Riya Dey carried out the simulations, analyzed the data, and prepared the manuscript. All authors discussed the results and contributed to the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dey, R., Patni, H.K. & Anand, S. Improved aerosol deposition predictions in human upper respiratory tract using coupled mesh phantom-based computational model. Sci Rep 15, 14260 (2025). https://doi.org/10.1038/s41598-025-86458-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86458-1
This article is cited by
-
Inhaled biologics for respiratory diseases: clinical potential and emerging technologies
Drug Delivery and Translational Research (2025)