Abstract
Agatston score, the degree of lumen narrowing categorized by CAD-RADS, high-risk atherosclerotic plaque features and pericoronary adipose tissue attenuation (PCAT) are parameters, which can be assessed non-invasively by coronary computed tomography angiography (CCTA) and aid risk stratification in patients with chronic coronary syndromes (CCS). However, few studies have so far compared the prognostic value of all those parameters together. To develop and test the prognostic value of a composite CCTA score, derived from Agatston score, CAD-RADS, high-risk plaques and PCAT in patients undergoing CCTA due to CCS. Consecutive patients with clinical indication for CCTA and available clinical follow-up of ≥ 6 months after the CCTA examination were included. (i) Agatston score, (ii) CAD-RADS, (iii) the number of plaques with at least one high-risk feature and (iv) PCAT in the proximal 4 cm of the right coronary artery (RCA) were measured, and a composite CCTA score was generated considering all four parameters. The primary endpoint encompassed all-cause mortality, myocardial infarction, and coronary revascularization (> 60 days after the CCTA scan) during follow-up. In total, 759 patients (median age 68.0 (IQR 59.0–76.0) years, 352 (46.4%) female) were included. During a median follow-up of 591.5 (IQR 505.5-686.8) days, 39 (5.1%) patients reached the primary endpoint. Cox-proportional regression demonstrated that the Agatston score, the number of high-risk plaques and CAD-RADS predicted the primary endpoint, independent of age and conventional cardiovascular risk factors. The number of high-risk plaques per patient provided the most robust prediction of the primary endpoint (HR = 2.74, 95%CI = 1.56–4.80, p < 0.001), whereas the composite CCTA score outperformed all other parameters (HR = 1.54, 95%CI = 1.19–1.98, p < 0.001). The Agatston score, CAD-RADS and high-risk plaque features may provide complementary prognostic information in patients with CCS. A composite CCTA score, derived by these imaging markers may identify high-risk individuals, who may benefit from more intensified treatment and clinical follow-up in future studies.
Similar content being viewed by others
Introduction
Coronary computed tomography angiography (CCTA) has become a well-established non-invasive imaging modality for the diagnostic work-up of patients with chronic coronary syndrome (CCS)1. In this regard, recent landmark studies have demonstrated its ability to reduce unnecessary invasive procedures and thus periprocedural complications, and to improve patient outcomes2,3. Apart from the presence, extent and severity of coronary calcification and lumen narrowing, as assessed by the Agatston score and the Coronary Artery Disease Reporting And Data System (CAD RADS), CCTA enables detection of low-attenuation non-calcified plaques and of plaques with other high-risk features, potential predecessors of plaque rupture and future acute coronary syndromes (ACS)4,5,6.
Furthermore, recent studies suggested that the pericoronary adipose tissue attenuation index (PCAT) is linked to atherosclerosis progression, as a marker of coronary inflammation7, and is a prognostic marker for cardiovascular (CV) events8. An association between PCAT, plaque burden, and high-risk plaque features has been reported9. In addition, low-attenuation plaque burden and PCAT have demonstrated complementary value for the risk-stratification of patients with CCS10.
Few studies, however, combined information derived from (i) Agatston score, (ii) high-risk plaque features, (iii) CAD-RADS and (iv) PCAT for the risk stratification of patients with CCS. In addition, the prognostic value of PCAT was recently questioned when compared to other CCTA-derived markers, such as the Agatston score, plaque volumes, high-risk plaque features, and the presence of obstructive CCS11,12. We therefore sought to develop a composite CCTA score, derived from all the above mentioned parameters (i)-(iv) for the risk stratification of patients with CCS.
Methods
Study design and patient population
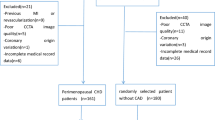
Between September 2020 and August 2022, consecutive patients who underwent CCTA for known or suspected CCS and stable chest pain and with clinical follow-up information available for ≥ 6 months, were enrolled in a single-center retrospective study. Patients with ACS and patients undergoing CCTA for other indications such as planning structural heart procedures were excluded from analysis. Since PCAT may be influenced by the presence of systemic inflammatory conditions, such as for example psoriasis or acute and chronic infections, Creactive protein (CRP) was assessed in cases of clinical suspicion of infection or other inflammatory conditions at the time of baseline CCTA and patients with known inflammatory diseases or increased CRP were excluded from the analysis. Clinical and CCTA data from the patients included in this study were previously reported by Giesen et al.9 who described an association between plaque composition and PCAT at a time point when clinical follow-up data were not available.
Ethical approval of the protocol was obtained from the ethic committee of the Medical Faculty of the University of Heidelberg (S-526/2016) for inclusion of the patients to the study and collection of outcome data. All patients gave written informed consent. In addition, all methods were performed in accordance with the relevant guidelines and institutional regulations.
Conventional CV risk factors (CVRF), such as arterial hypertension, hyperlipidemia, current or prior smoking, diabetes mellitus, family history of CAD as well as history of CAD, myocardial infarction, or percutaneous coronary intervention (PCI) were recorded. Laboratory markers, including hemoglobin, serum creatinine and the estimated glomerular filtration rate (eGFR), were analyzed.
CCTA protocol
CCTA was performed using a third-generation dual-source CT (SOMATOM Force, Siemens Healthineers, Forchheim, Germany). This 192-slice dual-source CT scanner offers a high-pitch spiral acquisition mode (table feed 737 mm/s; pitch 3.2), a minimal gantry rotation time of 250 ms resulting in 66 ms temporal resolution and a refined detector system allowing 0.24 mm spatial resolution.
Coronary calcium scans were performed before CCTA and without administering contrast agent. The scan range extended from the proximal ascending aorta to the caudal aspect of the heart. The parameters for native calcium scoring were as follows: Tube voltage of 120 kV, 80 QrefmAs, reconstruction matrix = 512 × 512, reconstructed slice thickness = 3 mm, reconstructed slice interval = 1.5 mm, reconstruction kernel = Qr36.
Patients were given metoprolol intravenously in individually adjusted doses to attain a heart rate of < 65 bpm. The acquisition protocol was selected based on patient specific parameters, including heart rate and rhythm. Most patients underwent ECG-triggered prospective axial acquisition. Patients with heart rates > 80 bpm despite beta-blocker administration or with frequent ectopic beats, were scanned using an ECG-triggered prospective arrhythmia protocol with axial acquisition or spiral acquisitions with retrospectively ECG-gated image reconstruction. Patients with heart rates < 65 bpm and body-mass-index (BMI) < 28 kg/m2 were examined using prospectively ECG-triggered high-pitch spiral acquisition.
The parameters of the ECG-triggered sequential CCTA scan protocol were as follows: Tube voltage of 80–120 kV depending on patients’ body mass index (BMI), tube current of 290–560 mA, reconstruction matrix of 512 × 512, reconstructed slice thickness of 0.8 mm, reconstructed slice interval of 0.4 mm. A bolus of iodine contrast media lopromide (Ultravist®, Bayer Healthcare, Berlin, Germany) of 50–60 ml was injected into the antecubital vein at a rate of 5 ml/s followed by a 50 ml saline bolus chaser at the same flow rate to provide contrast enhancement. The parameters of ECG-triggered sequential CCTA scan protocol are as follows: Tube voltage of 80–120 kVp depending on BMI, tube current of 290–560 mA, reconstruction matrix = 512 × 512, reconstructed slice thickness = 0.8 mm, reconstructed slice interval = 0.4 mm.
CCTA image analysis
Coronary CT angiograms were analyzed independently by experienced readers with each > 5 years of experience in CCTA, level 3 certified by the German Society of Cardiology, which is equivalent to the clinical competence training level 3 of the Society of Cardiovascular Computed Tomography13.
CAD-RADS 2.0 assessment and plaque burden quantification
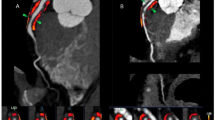
Coronary calcium scans were used for the quantification of the Agatston score (total calcium score). The CAD-RADS 2.0 was used to categorize the degree of lumen narrowing as follows: (0) none (0%) or (1) minimal (1–24%), (2) mild (25–49% stenosis), (3) moderate (50–69% stenosis), (4) severe (70–99% stenosis), (5) chronic total occlusions (100%).14 Coronary plaques was defined as structures located within or adjacent to the coronary artery lumen with or without causing coronary obstruction. The presence of high-risk plaque features i.e., (i) low-attenuation plaque, defined as a focal central area of plaque with an attenuation density of < 30 Hounsfield Units15, (ii) positive remodeling, (iii) spotty calcification, or (iv) the napkin ring sign were evaluated in each patient6. Patients were separated in those exhibiting none, 1–4 and ≥ 5 plaques with high-risk characteristics, as reported previously9.
PCAT quantification
PCAT was measured using the syngo.via (Frontier CT Coronary Plaque Analysis, version 5.0.2, Siemens Healthineers, Forchheim Germany research software), and was reported in HU as described previously8. For measurement of PCATRCA, the proximal 4 cm of the right coronary artery (RCA) were traced and perivascular fat was defined as the adipose tissue within a radial distance from the outer vessel wall being equal to the diameter of the vessel8. To adjust for differences in attenuation between scans performed at different tube voltages, the PCAT attenuation for scans performed with less than 120kVp was divided by conversion factors to be comparable to scans performed at 120 kVp i.e., conversion factor of 1.11485 for scans performed at 100 kVp and of 1.267 for scans performed at 80 kVp, as previously reported9.
Composite CCTA score
A composite score was calculated by adding the following:
-
(i)
Agatston score by quartiles (0–3 points),
-
(ii)
The CAD-RADS score 2.0 by groups (0–1: Group A (1 point); 2–3: Group B (2 points) and 4–5: Group C (3 points)),
-
(iii)
The number of plaques with high-risk features by groups (A-C), (group A( 1 point) = no high-risk plaques; group B (2 points): 1–4 high-risk plaques and group C (3 points): >5 high-risk plaques) and
-
(iv)
PCATRCA by quartiles (0–3 points).
The composite score (1–12 points) was compared to all other semi-quantitative and quantitative parameters for the prediction of the clinical endpoint.
Endpoints
Personnel blinded to the CCTA results contacted each subject or an immediate family member. The date of this contact was used to calculate the duration of follow-up. A standardized questionnaire was used to collect outcome data obtained from patient interviews in the outpatient clinic or by telephone. Reported clinical events were confirmed by reviewing the corresponding medical records in our electronic hospital information system and by contacting the general practitioner, the referring cardiologist, or the treating hospital if the patients were not treated at our institution.
The primary clinical endpoint included (i) all-cause mortality, (ii) myocardial infarction, and (iii) coronary revascularization by PCI or CABG, performed either urgently due to unstable angina or elective due to coronary artery stenosis ≥ 90% or a fractional flow reserve ≤ 0.8016, which was measured in case of stenosis between 40% and 90%.
Since CCTA may have triggered coronary revascularization procedures, we excluded procedures, occurring within 60 days after the CCTA examination.
Statistical analysis
Analysis was performed using the MedCalc 20.009 software (MedCalc software, Mariakerke, Belgium). Continuous normally distributed variables were expressed as mean ± standard deviation, whereas continuous non-normally distributed variables were reported as medians with interquartile range (IQR). Normal distribution was assessed using the Shapiro-Wilk test. Categorical variables were reported as numbers and proportions and were compared using χ2 tests. Quartiles were calculated for PCATRCA and the Agatston score. Receiver Operating Characteristics (ROC) analysis was performed to assess the ability of CCTA parameters to predict the primary clinical endpoint. In addition, Cox proportional hazard regression was used to compare the prognostic value of CCTA parameters for the prediction of the primary endpoint after adjustment for age, gender and other CVRF. The ANOVA test was used for comparing three or more normally distributed groups with the Scheffé test for post-hoc analysis16, whereas for not normally distributed continuous variables, non-parametric tests (Mann-Whitney U-tests or Kruskal-Wallis tests) were performed. Intraclass correlation coefficients were used to analyze the agreement between observers for CAD-RADS and the presence of plaques with high-risk features. Differences were considered statistically significant at p < 0.05.
Results
Demographic data and baseline characteristics
Between September 2020 and August 2022, 759 patients, who underwent clinically indicated CCTA for CCS and had a complete follow-up, were analyzed.
Median age was 68.0 (IQR 59.0–76.0) years. Of 759 patients, 352 (46.4%) patients were female. 91 (12.0%) patients had type 2 diabetes mellitus. Patients had a median of 2.0 (IQR 1.0–2.0) CVRF (Table 1). Data on radiation exposure of the patients during CCTA are shown in the suppl. Table 1. CAD-RADS of 0 was observed in 36 (4.7%) patients, of 1 in 121 (15.9%), of 2 in 270 (35.6%), of 3 in 169 (22.3%), of 4 in 129 (17.0%), and of 5 in 34 (4.5%) patients.
During a median follow-up of 591.5 (IQR 505.5-686.8, range 285.0-1188.0) days, six (0.8%) patients died, seven (0.9%) experienced non-fatal myocardial infarction and 66 (8.7%) underwent coronary revascularization either by PCI (n = 61) or by CABG (n = 5). Forty revascularization procedures occurred within the first 60 days after the CCTA examination and were therefore excluded from further analysis.
Association of PCAT, high-risk plaques and Agatston score with outcomes
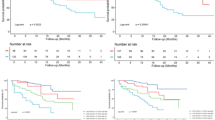
Agatston score quartiles and the total number of high-risk coronary plaques per patient was associated with the primary clinical endpoint (Fig. 1A,B). However, PCATRCA by quartiles (Fig. 1C) and by selecting a cut-off of -70.1 HU for the PCATRCA, as provided by the CRISP CT study8, was not predictive of the primary endpoint (Fig. 1D).
Cox-proportional hazard regression models
Cox proportional regression model showed that PCATRCA, Agatston score, the number of high-risk coronary plaques, and the CAD-RADS derived score predicted the primary endpoint, independent of age and CVRF (Table 2). PCATRCA was not predictive, whereas the number of high-risk plaques provided the most robust individual prediction of the primary endpoint (HR = 2.74, 95%CI = 1.56–4.80, p < 0.001).
Multiparametric assessment of the primary endpoint by the composite score
ROC analysis showed that the composite score, combining Agatston score, CAD-RADS, high-risk plaque features and PCATRCA, provided higher accuracy for the prediction of the pre-defined primary clinical endpoint, compared to all other parameters separately (AUC = 0.82, 95%CI = 0.80–0.85, p < 0.05 versus all other parameters, Fig. 2).
The corresponding sensitivities and specificities are provided in Table 3.
The composite score outperformed clinical parameters i.e., age and the total number of CVRF, as well as the Agatston score, CAD-RADS, high-risk plaque features and PCATRCA for the prediction of the combined clinical endpoint (HR = 1.54, 95%CI = 1.19–1.98, p < 0.001), (Table 4).
Reproducibility analysis for plaque burden
High intra-class coefficients of 0.85 and 0.89 were found for CAD-RADS and the presence or absence of high-risk atherosclerotic plaque features.
Discussion
The present study shows that a composite CCTA score derived from the Agatston score, CAD-RADS, the presence of plaques with high-risk morphology, and PCATRCA provides robust prediction of adverse events, including death, myocardial infarction, and coronary revascularization in patients with CCS. PCATRCA both by quartiles and by the cut-off values provided by the CRISP CT study8, however, did not predict the primary clinical endpoint. On the other hand, the number of plaques with high-risk features provided the most robust prediction of the predefined clinical endpoint. In addition, a composite score, exceeded the predictive value of conventional risk factors as well as the single CTTA-derived features, thus paving the way for multiparametric prediction of clinical endpoints by CCTA in future multi-center studies.
Previous studies
(i) Calcium scoring: The Agatston score can be assessed at low cost (typically less than 100 €), low radiation of less than one mSv and without contrast injections17,18. It provides incremental risk stratification over conventional CVRF in intermediate asymptomatic patients, and can guide lipid-lowering therapies based on current recommendations19. In addition, recent studies demonstrated the prognostic value of an Agatston score of zero, in that it was associated with a low rate of future adverse events in symptomatic patients with CCS20. However, its ability was lower than that provided by CCTA. Furthermore, Agatston score did not provide incremental information over myocardial perfusion abnormalities in symptomatic patients with suspected CAD21.
(ii) CAD-RADS: CAD-RADS classifies patients based on the highest grade of coronary artery stenosis, ranging from zero for normal coronary arteries up to 5 for patients with at least one occlusion22. CAD-RADS was introduced to standardize the communication of CCTA findings and to harmonize the decision-making process for patient management. Recently, patients with higher CAD-RADS were reported to have an increased risk of myocardial infarction, suggesting prognostic information provided by this classification system5. However, the value of CAD-RADS may be limited in patients with known CAD23, whereas data on its incremental value beyond that provided by PCATRCA or high-risk plaque features are still lacking.
(iii) High-risk plaque features: Several studies have already demonstrated the role of high-risk plaque features as potential precursors of plaque rupture causing cardiac events24,25,26. In low-risk patients with stable chest pain and non-obstructive CAD, such plaque features were shown to provide superior prognostic value compared to total plaque burden for the prediction of clinical events25. In addition, the presence of low-attenuation high-risk plaques outperformed the value provided by lumen narrowing for the prediction of cardiac events4,6. In the same direction, the risk for future ACS was substantially higher in plaques with high-risk features (OR = 12.1) in a meta-analysis of six studies, highlighting the value of this parameter for robust risk-stratification in patients with CCS27.
Epicardial adipose tissue is associated with coronary plaque parameters and cardiac outcomes28. Furthermore, PCAT, measured in perivascular adipose tissue, is linked to atherosclerosis progression, as a marker and regulator of coronary inflammation7, and also provides risk stratification of patients with CCS8,10,29. In addition, increased PCAT attenuation was identified as a predictor of non-calcified plaque progression in serial CCTA studies30, whereby PCAT was increased around culprit coronary plaques in patients with ACS, compared to non-culprit lesions and to lesions in patients with stable CAD. This has been attributed to a higher degree of inflammation around the vulnerable coronary lesions that are prone to rupture31,32. In addition, high-risk plaque characteristics and PCAT showed complementary value in predicting cardiac events in patients with CCS10,33. However, to our knowledge, the comparative prognostic value of PCAT and plaque burden to more basic CCTA parameters, such as calcium score and CAD-RADS has not been systematically investigated so far.
Current findings
In the present study, we used a multiparametric approach and systematically assessed the value of Agatston score, CAD-RADS, high-risk plaques, and PCATRCA for the prediction of all-cause mortality, myocardial infarction, and coronary revascularization. Most of the above parameters separately predicted the combined clinical endpoint and outperformed conventional parameters such as age and other CVRF. Interestingly, PCATRCA was not predictive, whereas the number of high-risk plaques per patient exhibited the most robust prediction of future cardiac events, compared to all other CCTA imaging parameters (Table 2). In addition, the composite score outperformed all four parameters separately for the prediction of the primary clinical endpoint. This could be demonstrated both by ROC analysis (Fig. 2) and by cox-proportional hazard regression models (Table 4). Notably, the median age of our patient cohort of 68 years was older than in previous CCTA studies2,3. This may be attributed to the liberal implementation of CCTA in our institution as the first-line diagnostic test for patients with suspected and known CAD, as recommended by current guidelines1, as well as to the diagnostic potential of the third-generation dual-source CT scanners, which allow precise diagnosis of CAD, even in patients with coronary calcification and after stent placement34. In addition, high inter-observer reproducibility was found in all CCTA-derived parameters, including high-risk plaque features and CAD-RADS. Finally, BMI was not associated with future cardiac events.
Limitations
Our study has some limitations. First, the study was conducted in a single center. A sensitivity analysis was not performed, whereas diagnostic accuracies were calculated within the same patient cohort, possibly overestimating the value of the parameters tested. In addition, a primary analysis of hard cardiac endpoints like death and myocardial infarction was not performed due to the limited number of hard endpoints, which would not have allowed for meaningful statistical analysis. Multicenter studies are now warranted in this context, including a larger number of patients with longer follow-up duration for the assessment of the prognostic value of different CCTA parameters. Importantly, most patients who underwent CCTA were at low risk for obstructive CAD, as recommended by current guidelines35. This, however, resulted in a low number of hard cardiac events during follow-up, whereas the primary endpoint was substantially triggered by coronary revascularization procedures (n = 66, 83.5%), which is a limitation. Importantly, all events after CCTA were considered, so that some coronary revascularization procedures may have been triggered by the CCTA results. However, events occurring > 60 days after the CCTA were excluded from analysis. In addition, although previous studies attempted subsection analysis between fibrous and fibro-fatty components within non-calcified plaques36, this was not assessed in the present study due to overlaps in HU cut-off values. Furthermore, some of the investigated CCTA parameters like plaque features and the Agatston score may have been more decisive for the predictive value of the composite CCTA score than others, like for example PCAT, which was not predictive for the primary endpoint. In the same direction, our study was not powered to assess the prognostic value of each CCTA variable separately for the prediction of cardiac events. In addition, CCTA variables may act differently and be modifiable to different extents during treatment with lipid-lowering or anti-inflammatory agents. Thus, the current findings are rather hypothesis-generating for future trials and may not allow for individual patient management based on the proposed CCTA score.
Conclusions
Our study highlights that a multiparametric strategy may provide complementary prognostic information in patients with CCS. Such a composite CCTA score, combining multiple imaging markers may identify high-risk individuals, who may benefit from more intensified treatment and more intensive clinical follow-up. Multi-center studies are now warranted to specifically address the contribution of each parameter for all-cause mortality, infarction, and coronary revascularization separately and the role of pharmacologic treatment for the prevention of future CV events.
Data availability
Availability of data and material: The dataset used and/or analyzed is available from the corresponding author upon reasonable request.
References
Knuuti, J. et al. 2019 ESC guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 41 (3), 407–477 (2020).
Investigators, S-H. et al. Coronary CT angiography and 5-year risk of myocardial infarction. N. Engl. J. Med. 379 (10), 924–933 (2018).
Group, D. T. et al. CT or invasive coronary angiography in stable chest Pain. N. Engl. J. Med. 386 (17), 1591–1602 (2022).
Williams, M. C. et al. Low-attenuation noncalcified plaque on coronary computed tomography angiography predicts myocardial infarction: Results from the multicenter SCOT-HEART trial (Scottish Computed Tomography of the HEART). Circulation 141 (18), 1452–1462 (2020).
Williams, M. C. et al. Standardized reporting systems for computed tomography coronary angiography and calcium scoring: A real-world validation of CAD-RADS and CAC-DRS in patients with stable chest pain. J. Cardiovasc. Comput. Tomogr. 14 (1), 3–11 (2020).
Gitsioudis, G. et al. Combined assessment of high-sensitivity troponin T and noninvasive coronary plaque composition for the prediction of Cardiac outcomes. Radiology 276 (1), 73–81 (2015).
Oikonomou, E. K., West, H. W. & Antoniades, C. Cardiac computed tomography: Assessment of coronary inflammation and other plaque features. Arterioscler. Thromb. Vasc Biol. 39 (11), 2207–2219 (2019).
Oikonomou, E. K. et al. Non-invasive detection of coronary inflammation using computed tomography and prediction of residual cardiovascular risk (the CRISP CT study): A post-hoc analysis of prospective outcome data. Lancet 392 (10151), 929–939 (2018).
Giesen, A. et al. Pericoronary adipose tissue attenuation is associated with non-calcified plaque burden in patients with chronic coronary syndromes. J. Cardiovasc. Comput. Tomogr. 17 (6), 384–392 (2023).
Tzolos, E. et al. Pericoronary adipose tissue attenuation, low-attenuation plaque burden, and 5-year risk of myocardial infarction. JACC Cardiovasc. Imaging. 15 (6), 1078–1088 (2022).
Bengs, S. et al. Quantification of perivascular inflammation does not provide incremental prognostic value over myocardial perfusion imaging and calcium scoring. Eur. J. Nucl. Med. Mol. Imaging. 48 (6), 1806–1812 (2021).
van Diemen, P. A. et al. Prognostic value of RCA pericoronary adipose tissue CT-attenuation beyond high-risk plaques, plaque volume, and ischemia. JACC Cardiovasc. Imaging. 14 (8), 1598–1610 (2021).
Choi, A. D. et al. 2020 SCCT Guideline for training cardiology and radiology trainees as independent practitioners (level II) and advanced practitioners (level III) in cardiovascular computed tomography: A statement from the Society of Cardiovascular Computed Tomography. JACC Cardiovasc. Imaging. 14 (1), 272–287 (2021).
Cury, R. C. et al. An expert consensus document of the Society of Cardiovascular Computed Tomography (SCCT), the American College of Cardiology (ACC), the American College of Radiology (ACR) and the North America Society of Cardiovascular Imaging (NASCI). J. Am. Coll. Radiol. 19 (11), 1185–1212 (2022). CAD-RADS 2.0–2022 Coronary Artery Disease - Reporting and Data System.
Williams, M. C. et al. Coronary artery plaque characteristics associated with adverse outcomes in the SCOT-HEART study. J. Am. Coll. Cardiol. 73 (3), 291–301 (2019).
Hofmann, N. P. et al. Quantitative myocardial blush grade reserve during pharmacologic hyperaemia: A way to perform a real wireless fractional flow reserve measurement in patients with coronary artery disease and intermediate coronary lesions. EuroIntervention 12 (18), e2219–e27 (2017).
Budoff, M. J. et al. Long-term prognosis associated with coronary calcification: Observations from a registry of 25,253 patients. J. Am. Coll. Cardiol. 49 (18), 1860–1870 (2007).
Andre, F. et al. Simultaneous assessment of heart and lungs with gated high-pitch ultra-low dose chest CT using artificial intelligence-based calcium scoring. Eur. J. Radiol. Open. 10, 100481 (2023).
Grundy, S. M., Stone, N. J., Bailey, A. L. & et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: A report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. Circulation 139 (25), e1082–e143 (2019).
Budoff, M. J. et al. Prognostic value of coronary artery calcium in the PROMISE study (prospective Multicenter Imaging Study for evaluation of chest pain). Circulation 136 (21), 1993–2005 (2017).
Patel, K. K. et al. Prognostic relationship between coronary artery calcium score, perfusion defects, and myocardial blood flow reserve in patients with suspected coronary artery disease. Circ. Cardiovasc. Imaging. 15 (4), e012599 (2022).
Cury, R. C. et al. Coronary artery Disease - Reporting and Data System (CAD-RADS): An expert consensus document of SCCT, ACR and NASCI: Endorsed by the ACC. JACC Cardiovasc. Imaging. 9 (9), 1099–1113 (2016).
Oeing, C. U. et al. Coronary artery disease grading by cardiac CT for predicting outcome in patients with stable angina. J. Cardiovasc. Comput. Tomogr. 17 (5), 310–317 (2023).
Motoyama, S. et al. Plaque characterization by coronary computed tomography angiography and the likelihood of acute coronary events in mid-term follow-up. J. Am. Coll. Cardiol. 66 (4), 337–346 (2015).
Taron, J. et al. Risk stratification with the use of coronary computed tomographic angiography in patients with nonobstructive coronary artery disease. JACC Cardiovasc. Imaging. 14 (11), 2186–2195 (2021).
Korosoglou, G., Chatzizisis, Y. S. & Raggi, P. Coronary computed tomography angiography in asymptomatic patients: Still a taboo or precision medicine? Atherosclerosis 317, 47–49 (2021).
Thomsen, C. & Abdulla, J. Characteristics of high-risk coronary plaques identified by computed tomographic angiography and associated prognosis: A systematic review and meta-analysis. Eur. Heart J. Cardiovasc. Imaging. 17 (2), 120–129 (2016).
Tesche, C. et al. Association of epicardial adipose tissue with coronary CT angiography plaque parameters on cardiovascular outcome in patients with and without diabetes mellitus. Atherosclerosis 363, 78–84 (2022).
Sagris, M. et al. Pericoronary fat attenuation index-a new imaging biomarker and its diagnostic and prognostic utility: A systematic review and meta-analysis. Eur. Heart J. Cardiovasc. Imaging. 23 (12), e526–e36 (2022).
Goeller, M. et al. Relationship between changes in pericoronary adipose tissue attenuation and coronary plaque burden quantified from coronary computed tomography angiography. Eur. Heart J. Cardiovasc. Imaging. 20 (6), 636–643 (2019).
Goeller, M. et al. Pericoronary adipose tissue computed tomography attenuation and high-risk plaque characteristics in acute coronary syndrome compared with stable coronary artery disease. JAMA Cardiol. 3 (9), 858–863 (2018).
Kuneman, J. H. et al. Pericoronary adipose tissue attenuation in patients with acute coronary syndrome versus stable coronary artery disease. Circ. Cardiovasc. Imaging. 16 (2), e014672 (2023).
Oikonomou, E. K. et al. Perivascular fat attenuation index stratifies cardiac risk associated with high-risk plaques in the CRISP-CT study. J. Am. Coll. Cardiol. 76 (6), 755–757 (2020).
Ochs, M. M. et al. Limits of the possible: Diagnostic image quality in coronary angiography with third-generation dual-source CT. Clin. Res. Cardiol. 106 (7), 485–492 (2017).
Vrints, C. et al. 2024 ESC guidelines for the management of chronic coronary syndromes. Eur. Heart J. (2024).
van Rosendael, A. R. et al. Association of statin treatment with progression of coronary atherosclerotic plaque composition. JAMA Cardiol. 6 (11), 1257–1266 (2021).
Acknowledgements
Clinical and CCTA data of patients included in this study were reported previously by Giesen et al.9. The previous study described an association of plaque composition with PCAT, without presenting any follow-up data. In this study, we focused on the ability of CCTA parameters including PCAT and plaque composition for the risk stratification of patients with CCS.
Funding
Our work was supported by the German Foundation for Heart Research (Deutsche Stiftung für Herzforschung), Application Number: F/17/21. The project was performed in the Cardiac Imaging Center, Weinheim, which is supported by the Hector Foundation in Weinheim. AAG is supported by grants provided by Promedica Stiftung and Iten-Kohaut Foundation in collaboration with the USZ Foundation. FA received research support from Siemens Healthineers und Philips Healthcare.
Author information
Authors and Affiliations
Contributions
GK, AG, SL and DM designed the study, performed the analysis, wrote, and reviewed the manuscript, and provided important intellectual input, all others reviewed the manuscript and provided important intellectual input. All authors approved the final version of this paper.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Ethical approval of the protocol was obtained from the ethic committee of the Medical Faculty of the University of Heidelberg (S-526/2016). All patients gave written informed consent.
Competing interests
GK: Received modest honoraria from Philips Healthcare, Siemens Healthineers, Cordis, Boston scientific and BARD Peripheral Vascular Inc., and institutional grants from Philips Healthcare and Bard Peripheral Vascular Inc. NF: Speaker fees from Daiichi Sankyo, Novartis, and Pfizer. MR: received proctor fees from Boston Scientific. All other authors have no competing interests to declare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, S., Giesen, A., Mouselimis, D. et al. Composite cardiac computed tomography angiography score for improved risk assessment in chronic coronary syndromes. Sci Rep 15, 3089 (2025). https://doi.org/10.1038/s41598-025-87118-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-87118-0