Abstract
Online hemodiafiltration (Ol-HDF) and expanded hemodialysis (HDx; using medium cut-off dialyzers) have shown superior removal of diverse uremic toxins, particularly middle molecules and inflammatory cytokines, compared to conventional hemodialysis (HD). However, the relative efficacy of toxin removal between HDx and Ol-HDF remains unclear. This study aimed to compare these two techniques. A randomized controlled trial was conducted among thrice-weekly hemodialysis patients. Participants were randomized into either a post-dilution Ol-HDF group or an HDx group using two types of medium cut-off dialyzers (Theranova 500® and Elisio HX21®) over 8 weeks. Efficacy was assessed by reduction ratios (RR) and pre-dialysis toxin levels. Statistical analysis used T-tests and generalized estimating equations. A total of 40 patients were enrolled (mean age 64.6 ± 12.0 years; 82.5% male; dialysis vintage 53.4 ± 38.0 months). The Ol-HDF group, with a mean convection volume of 24.7 L, had significantly higher RR for beta-2 microglobulin (82.8% vs. 74.6%; p < 0.001), parathyroid hormone (80.4% vs. 60.6%; p = 0.007), homocysteine (58.0% vs. 50.0%; p = 0.009), and kappa- and lambda-free light chains (73.3% vs. 64.6%; p = 0.0001 and 62.5% vs. 52.0%; p = 0.0026, respectively). Pre-dialysis toxin levels at the end of the study were similar between groups. These findings highlight that Ol-HDF demonstrated superior removal of uremic toxins, while HDx was comparable to Ol-HDF in maintaining pre-dialysis levels of middle molecules and inflammatory cytokines.
Trial registration This study was registered in the Thai Clinical Trials Registry (TCTR) on the first posted date 10/02/2023 with the registration number TCTR20230210004.
Similar content being viewed by others
Introduction
Patients with end-stage kidney disease (ESKD) undergoing hemodialysis (HD) face a high risk of cardiovascular morbidity and mortality. These cardiovascular risks stem from both non-dialysis related factors (e.g., diabetes, hypertension, dyslipidemia) and dialysis related factors (e.g., uremic toxins, chronic inflammation, and mineral and bone disorder)1. Uremic toxins are classified into small molecules (< 500 Da), middle molecules (> 500 Da) categorized into small- (500–15,000 Da), medium- (15,000–25,000 Da), and large-middle molecules (25,000–58,000 Da), as well as protein-bound uremic toxins (PBUT)2. Although conventional HD effectively removes small-molecule uremic toxins through diffusion, its capability to clear middle-molecule uremic toxins and PBUT is limited thereby impacting cardiovascular and all-cause mortalities3. Online hemodiafiltration (Ol-HDF) integrates diffusion and convection mechanisms, enhancing clearance of middle molecules uremic toxins, leading to reduced all-cause and cardiovascular mortalities4,5. However, implementing Ol-HDF in general HD units is challenging due to the need for specialized equipment and trained staff.
A medium cut-off (MCO) membrane dialyzer with larger pore sizes has been developed to improve the removal of medium- and large-middle molecule uremic toxins6. Expanded HD (HDx) using MCO dialyzers with conventional HD machines significantly improves middle-molecule uremic toxin clearance compared to conventional HD7. However, studies comparing the efficacy of uremic toxins and inflammatory cytokine clearances between HDx and Ol-HDF remain inconclusive8,9,10. Therefore, the primary objective of this study was to assess the efficacy of HDx in uremic toxin removal compared with post-dilutional Ol-HDF, assessed by pre- and post-dialysis reduction ratio (RR) for various-sized uremic toxins and inflammatory cytokines and changes in pre-dialysis uremic toxin levels from baseline to 4 and 8 weeks of treatment. Additionally, we aim to compare the efficacy between two types of MCO dialyzers (Theranova 500® and Elisio HX21®).
Results
Baseline characteristics and dialysis treatment of the study population
A total 40 patients were enrolled with a mean age of 64.6 ± 12.0 years, 82.5% were male, and the main etiology of ESKD was diabetic nephropathy (57.5%) with a mean dialysis vintage of 53.4 ± 38.0 months. The type of vascular access was arteriovenous fistula in all participants. After the 2-week washout period, baseline characteristics and pre-dialysis laboratory data at the first session of the intervention showed no significant differences between groups (Table 1). The baseline equilibrated Kt/V and nPCR were determined at the last session of washout period which revealed similar levels (equilibrated Kt/V; 1.6 ± 0.2 vs.1.5 ± 0.2 and nPCR; 1.1 ± 0.3 vs. 1.1 ± 0.2 g/kg/day, all p-values > 0.05 in the Ol-HDF and HDx groups, respectively).
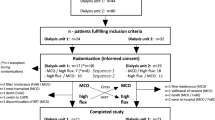
All participants received dialysis intervention for 8 weeks of study period (Fig. 1). Both Ol-HDF and HDx groups received comparable prescriptions for dialysis settings, including similar 240 min of dialysis session time, ultrafiltration volume (2.4 ± 0.9 vs. 2.4 ± 1.2 L/session), blood flow rates (322.5 ± 34.3 vs. 316 ± 32.7 mL/min), and dialysate flow rates (590 ± 141.0 vs. 620 ± 150.8 mL/min) as demonstrated in Table S1. In the Ol-HDF group, the mean convective volume was 24.7 ± 2.2 L/session and substitution volume was 22.2 ± 2.6 L/session.
Efficacy on uremic toxin and inflammatory cytokine clearance
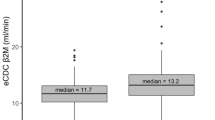
In terms of uremic toxins removal assessed at the first dialysis session, the Ol-HDF group exhibited significantly higher equilibrated urea Kt/V values compared to the HDx group (1.8 ± 0.3 vs. 1.5 ± 0.2; p = 0.0002). Furthermore, the RRs for various uremic toxins were significantly higher in the Ol-HDF group compared to the HDx group, including parathyroid hormone (PTH; 80.4 ± 12.3% vs. 60.6 ± 28.1%; p = 0.007), beta-2 microglobulin (β2-MG; 82.8 ± 3.5% vs. 74.6 ± 5.0%; p < 0.0001), kappa-free light chain (κ-FLC; 73.3 ± 6.0% vs. 64.6 ± 6.3%; p = 0.0001), lambda-free light chain (λ-FLC; 62.5 ± 8.3% vs. 52.0 ± 12.0%; p = 0.003), and homocysteine (58.0 ± 5.9% vs. 50.0 ± 11.6%; p = 0.009) as shown in Fig. 2A.
Pre- to post-dialysis reduction ratios of small-molecule, middle-molecule, and protein-bound uremic toxins, (A) compared between online-HDF and HDx with medium cut-off dialyzers, (B) compared between two types of MCO dialyzer (Theranova 500® and Elisio HX21®). PTH parathyroid hormone, B2-M beta-2 microglobulin, KFLC kappa free light chain, λFLC lambda free light chain, IL-6 interleukin-6, CRP C-reactive protein.
Analyzing the HDx group, there were no statistically significant differences observed in the RRs of all uremic toxins between the two types of MCO dialyzers (Fig. 2B). When comparing the Ol-HDF group with subgroups of HDx utilizing two types of MCO dialyzers, however, significantly greater uremic toxin removal was observed in the Ol-HDF group than both types of MCO dialyzer. i.e.,Theranova 500® and Elisio HX21®, including β2-MG, κ-FLC, λ-FLC and homocysteine (Fig. S1 and Table S2).
Comparison of pre-dialysis levels during the study period
Pre-dialysis levels of various-sized uremic toxins at week 4 and week 8 of the study exhibited comparability between the Ol-HDF and HDx groups. Additionally, pre-dialysis levels of uremic toxins at intervention initial (week 0) and changes over the 8-week study period appeared similar between both study arms (Fig. 3 and Table S3). Furthermore, the results of the generalized estimating equations (GEE) analysis conducted over the study duration also indicated no significant differences in overall pre-dialysis uremic toxin levels between these two groups.
Safety of the study interventions
In terms of safety outcomes, the HDx group had a significantly higher incidence of intradialytic hypotension compared to the Ol-HDF group, but other intradialytic adverse events were similar between groups. The albumin reduction ratio was higher in the Ol-HDF group compared to the MCO group, although this difference was not statistical significance (9.73 ± 7.70% vs. 5.15 ± 6.67%, p = 0.051). However, the pre-dialysis albumin levels at the end of study were similar between 2 groups (4.02 ± 0.24 vs. 4.00 ± 0.23 g/dL, p = 0.74) (Table S4).
Discussion
HDx represents an innovative approach to HD with potential broad applicability in general HD units equipped with ultrapure-quality dialysis water. Prior studies established HDx as a method that significantly surpasses high-flux HD in terms of effectively clearing middle-molecule uremic toxins, positioning it as a highly comparable alternative to Ol-HDF11,12,13,14. The MCO dialyzer’s design enhances permeability and convective clearance of middle-molecule uremic toxins through a modified sieving profile. The high-retention onset of the MCO dialyzer improves the clearance of medium to large uremic toxins. Additionally, membrane modifications—such as lengthening the fibers in the Elisio Hx dialyzer to increase the end-to-end pressure drop or reducing their diameters in the Theranova dialyzer to enhance the wall shear rate—contribute to elevate the retrofiltration and, consequently, improve the clearance of middle-molecule uremic toxins through convection. The combination of hydraulic permeability and the geometric structure of the fibers further enhances the processes of internal filtration and back-filtration providing remarkable convective clearance of. However, the effectiveness of Ol-HDF depends on variables such as blood flow rate and convective volume, which can enhance removal efficiency more than HDx15.
This study revealed that post-dilution Ol-HDF had significantly higher RR of several uremic toxins compared to HDx excepted phosphate and IL-6. The efficacy of Ol-HDF depends on the convective volume which is influenced by vascular access flow, filtration fraction, and the ultrafiltration coefficient of the dialyzer. A previous study suggested that the minimum convective volume in HDF required to surpass HDx efficacy was 17.6 L/session at a blood flow rate of 400 mL/min and 19.2 L/session at 350 mL/min16. Another study demonstrated that Ol-HDF with a high convective volume (mean 28 ± 8 L/session) had higher RR for middle-molecule uremic toxins than MCO group11. However, a previous study of Hadad-Arrascue et al. found similar RR of middle molecules uremic toxins [β2-MG, fibroblast growth factor-23 (FGF-23), chitinase-3-like protein 1 (YKL-40), κ-FLC, and λ-FLC] and inflammatory biomarkers [C-reactive protein (CRP), interleukin-6 (IL-6), pentraxin-3] between HDx and HDF treatment, though HDx had a larger dialyzer surface area and lower dialysate flow rate17. While Kirsch et al. reported that HDx using Theranova 400® had a more superior RR of YKL-40, α1-microglobulin, complement factor D, myoglobin and λ-FLC than HDF, although their study had higher blood flow rate (400 mL/min) and longer dialysis session time (4.4 ± 0.24 h) compared with ours18.
Recently, a meta-analysis of 18 trials reported that HDx is less effective for removing β2-MG than HDF but better for larger molecular toxins like κ-FLC and λ-FLC. However, there were different dialyzers for HDx or HDF, also with different dialysis prescriptions, which may have affected the results19. Thus, the effectiveness of removing middle-molecule uremic toxins and inflammatory cytokines depends not only on the dialyzer and technique but also on factors like dialysis treatment time and volume of dialysate.
In our study, no significant difference was found in pre-dialysis levels of middle molecules and inflammatory cytokines between HDx and Ol-HDF groups at week 4 and week 8, nor change from baseline to week 8 in either group. This aligns with a previous study that reported similar pre-dialysis levels of middle molecules uremic toxins (YKL-40, FLCs, κ-FLC, and λ-FLC) and inflammatory markers (CRP, IL-6, and pentraxin-3) at baseline, and their changes over the 24-week were not different between the HDx and Ol-HDF groups, except for β2-MG and FGF-23 which significantly decreased in the HDx group17. Furthermore, a long-term study in 57 HD patients with a 12-month study period found that HDx with the MCO dialyzer (Theranova 400®) provided excellent RR of β2-MG, κ-FLC and λ-FLC but there was no significant change in pre-dialysis serum levels of these uremic toxins after 1-year treatment (β2-MG from 25.6 ± 9.6 to 28.4 ± 4.8 mg/L; κ-FLC from 168.2 ± 58.5 to 167.7 ± 65.8 mg/L and λ-FLC from 135.7 ± 39.9 to 132.0 ± 39.1 mg/L)20. Previous findings reported by Ward et al. indicated that despite the high clearance rate observed in HDF, there was insufficient long-term reduction of β2-MG due to its rebound after the HD session. They suggested that alternative strategies, such as increasing treatment times or frequency, are necessary to effectively reduce plasma β2-MG concentrations21. In the present study, pre-dialysis PTH levels were comparable between the two groups. However, PTH is not considered a reliable marker for dialysis clearance, as its secretion is influenced by fluctuations in calcium and phosphorus levels. Previous studies have demonstrated that PTH levels fluctuate during hemodialysis due to changes in ionized calcium from dialysate and an increase in pH during procedure22. In our study, the majority of participants (80% in the Ol-HDF group and 70% in the MCO group) used dialysate with a calcium concentration of 3.0 mEq/L. Pre-dialysis serum calcium levels were comparable between both groups at intervention initiation (8.6 ± 0.6 vs. 8.9 ± 0.9 mg/dL, p = 0.19), at 4th week (8.8 ± 0.6 vs. 8.9 ± 0.8 mg/dL, p = 0.59) and at 8th week (8.7 ± 0.6 vs. 8.9 ± 0.9 mg/dL, p = 0.27). Additionally, the percentage changes in pre-dialysis calcium levels from initial to week 8 were similar between the 2 groups (1.2 ± 6.7 vs. 0.9 ± 8.8%, p = 0.88), as shown in Table S5. Given the inconclusive evidence regarding the efficacy of MCO dialyzers in reducing absolute exposure to uremic toxins and its potential impact on clinical outcomes, further research is warranted.
In this study, the two types of MCO dialyzer showed similar efficacy in the clearances of inflammatory cytokines and middle-molecule uremic toxins, both inferior to Ol-HDF treatment. Similar results were reported in a study by Maduell F et al., which examined the efficacy of uremic toxin removal compared between HDx with four types of MCO dialyzer (Phylther 17-SD®, Vie-18X®, Elisio HX19®, and Theranova 400®), Ol-HDF, and conventional HD with high-flux dialyzer (FX80 Cordiax®). The results revealed similar efficacy of clearance of middle molecules uremic toxins (myoglobin, prolactin, α1-microglobulin, κ-FLC, and λ-FLC) among four types of MCO dialyzer, all superior to conventional HD but inferior to Ol-HDF treatment23. In a randomized controlled trial comparing two MCO dialyzers shown that Elisio HX17® had significantly lower RR for β2-MG, myoglobin and κ-FLC than Theranova 400®, however, the small sample size and laboratory assay discrepancies were limitations of this study24.
The safety profile of MCO dialyzers is maintained through the regulation of pore sizes, thereby restricting albumin loss to below 5 g per session based on the hypothesis for hepatic synthesis compensation25. Although MCO membranes result in higher albumin loss than conventional HD, findings are inconsistent compared to HDF12,26,27. A recent systematic review found no significant difference in albumin loss between HDx and HDF19. In our study, minimal albumin loss was observed with MCO dialyzers, and serum albumin levels at the end of study did not differ between Ol-HDF and HDx groups. This implies that HDx with MCO dialyzers may initially induce a small, albeit non-significant, decrease in serum albumin levels. A higher incidence of intradialytic hypotension was noted in the HDx group, suggesting that Ol-HDF may mitigate dialysis-related hypotension, though the precise mechanism remains unclear5,28. Proposed mechanisms include reduced Na removal, the effect of low-temperature replacement fluid and reduced small-molecule solute removal during HDF29.
This study has some limitations. Firstly, the RR were only collected at the initial intervention session, limiting our ability to assess changes over time. Additionally, some uremic toxins were not measured due to laboratory constraints, but blood samples were collected for future investigation. Further long-term studies are warranted to assess the sustained effects of both Ol-HDF and HDx on uremic toxin clearance and their broader impact on patient outcomes, including survival rates, cardiovascular events, and quality of life. Additionally, future research should consider expanding the range of analyzed toxins, particularly those with higher molecular weights, including protein-bound uremic toxins. This approach may provide a more comprehensive understanding of the capabilities and limitations of both HDx and Ol-HDF in toxin removal.
Conclusion
High convection volume post-dilutional Ol-HDF demonstrated superior removal of various uremic toxins compared to HDx with MCO dialyzers. However, HDx maintained pre-dialysis levels of middle molecules and inflammatory cytokines similarly to Ol-HDF throughout the 8-week study period. Both MCO dialyzers showed similar effectiveness in removing uremic toxins, making HDx a viable alternative for patients unable to undergo high convective volume Ol-HDF.
Materials and methods
Study design and population
The single-center open-label randomized controlled trial was conducted among thrice-weekly HD patients at Srinagarind Hospital, Khon Kaen University, from December 2022 to February 2023. The inclusion criteria comprised participants aged between 18 and 80 years who had undergone adequate HD (spKt/V > 1.2 per session) for more than 3 months utilizing arteriovenous fistula or graft (AVF or AVG) for vascular access. Exclusion criteria encompassed pregnancy, contraindications for online HDF, advanced-stage cancer or cardiovascular diseases, severe comorbidities with a life expectancy of less than 1 year, prior severe infections or hospitalization within 3 months, receipt of immunosuppressive drugs, and a history of severe dialyzer allergy.
Sample size calculation
The sample size was calculated based on the RR of β2-MG clearances, observed to be 74.7% with HDx utilizing the MCO membrane and 81.2% with Ol-HDF11. The study required 16 participants per group for a power of 80% to detect a difference between groups with 95% confidence. Accounting for a 10% dropout rate, 18 participants per group were needed, totaling 36 participants.
Study intervention
All 40 participants underwent the 2-week washout period with treatments of high-flux conventional HD using Elisio 210 H® dialyzer, 3 sessions/week for balance the two groups before randomization 1:1 ratio into the post-dilution Ol-HDF group or the HDx group by block of 4 randomization. After the washout period, the patients of Ol-HDF group were dialyzed using Elisio 210 H® dialyzer with Fresenius 5008 H® machine with post-dilution automatic infusion flow. The HDx group used either Theranova 500® or Elisio HX21® dialyzers with Fresenius 4008 H® machine (Table S6). Each patient received thrice-weekly 4-hour dialysis sessions adhering to their regular prescriptions. Intradialytic monitoring and management were overseen by trained dialysis nurses, and intradialytic medications were reviewed and adjusted by nephrologists according to hospital protocol. Both groups utilized ultrapure-quality dialysis water.
Data collection and outcomes measures
Baseline characteristics and demographic data were collected through interviews, physical examinations, and medical record reviews. Baseline laboratory data were collected at the end of a 2-week washout period with high-flux HD which equilibrated Kt/V and nPCR values were determined at the last session of washout period and the other baseline pre-dialysis laboratory data were obtained during the first session of intervention initiation (week 0). The efficacy of removing various-sized uremic toxins including homocysteine (135 Da), a protein-bound uremic toxin, phosphorus (30 Da), urea (60 Da), creatinine (113 Da), PTH (9,400 Da), β2-MG (11,800 Da), κ-FLC (22,500 Da), IL-6 (24,500 Da), λ-FLC (45,000 Da), and albumin (66000 Da) were assessed by RR at the first dialysis session intervention, pre-dialysis serum uremic toxin levels at the week 4 and week 8 and change from baseline to week 8 of the study period. All blood sample were collected at the first dialysis session of the week when after the longest inter-dialytic interval for maximizing assessments of uremic toxin RRs and capturing the highest pre-dialysis uremic toxin levels in patients.
The RR was calculated by the following equations according to Bergström and Wehle30:
where,
“Cpre” and “Cpost” denote the concentrations measured before and after dialysis sessions, respectively. “cCpost” represents the corrected post-dialysis concentration from ultrafiltration, while “∆BW” signifies the weight lost during the HD session and “BWpost” denotes the body weight at the end of the HD session.
Techniques for measuring serum solute levels included molecular absorption spectrometry (for urea and creatinine), immunoturbidimetric assay (for β2-MG and CRP), electrochemiluminescence immunoassay (for IL-6), enzymatic assay (for homocysteine), and latex particle enhanced immunoturbidimetric method (for κ-FLC and λ-FLC).
Statistical analysis
Continuous data were expressed as means ± standard deviation (SD) or median (interquartile range, IQR), and categorical data were expressed as percentages. The two groups were compared using Student’s t-test or Mann-Whitney U test for continuous variables and Chi-square test or Fisher’s exact test for categorical variables. One-way analysis of variance (ANOVA) and linear regression analyses were used for comparisons among more than two groups. GEE evaluated clinical parameters with repeated measurements. Statistical analyses were performed using STATA version 17.0, with a p-value < 0.05 considered statistically significant.
Data availability
The datasets generated and analyzed during the current study are not publicly available due to confidentiality agreements but are available from the corresponding author on reasonable request.
References
Cozzolino, M. et al. Cardiovascular disease in dialysis patients. Nephrol. Dial Transplant. 33 (Suppl 3), 28–34. https://doi.org/10.1093/ndt/gfy174 (2018).
Rosner, M. H. et al. Classification of uremic toxins and their role in kidney failure. Clin. J. Am. Soc. Nephrol. 16 (12), 1918–1928. https://doi.org/10.2215/cjn.02660221 (2021).
Vanholder, R. C., Eloot, S. & Glorieux, L. R. L. Future avenues to decrease uremic toxin concentration. Am. J. Kidney Dis. 67 (4), 664–676. https://doi.org/10.1053/j.ajkd.2015.08.029 (2016).
Blankestijn, P. J. et al. Effect of hemodiafiltration or hemodialysis on mortality in kidney failure. N. Engl. J. Med. 389 (8), 700–709. https://doi.org/10.1056/nejmoa2304820 (2023).
Peters, A. E. et al. Haemodiafiltration and mortality in end-stage kidney disease patients: a pooled individual participant data analysis from four randomized controlled trials. Nephrol. Dial Transplant. 31 (6), 978–984. https://doi.org/10.1093/ndt/gfv349 (2016).
Boschetti-de-Fierro, A., Voigt, M., Storr, M. & Krause, B. MCO membranes: enhanced selectivity in high-flux class. Sci. Rep. 5 (1), 18448. https://doi.org/10.1038/srep18448 (2015).
Fiorentino, M., La Fergola, F. & De Rosa, S. Medium cut-off dialyzer for middle molecular uremic toxins in AKI and chronic dialysis. J. Nephrol. 37 (1), 23–37. https://doi.org/10.1007/s40620-023-01771-0 (2024).
Lindgren, A., Fjellstedt, E. & Christensson, A. Comparison of hemodialysis using a medium cutoff dialyzer versus hemodiafiltration: a controlled cross-over study. Int. J. Nephrol. Renovasc. Dis. 13, 273–280. https://doi.org/10.2147/ijnrd.s263110 (2020).
Kim, T. H. et al. Removal of large middle molecules via haemodialysis with medium cut-off membranes at lower blood flow rates: an observational prospective study. BMC Nephrol. 21 (1), 2. https://doi.org/10.1186/s12882-019-1669-3 (2019).
Eiamcharoenying, J. et al. Comparative effectiveness between expanded hemodialysis (hemodialysis using a medium cut-off dialyzer) and mixed-dilution online hemodiafiltration using a high-flux dialyzer in removing middle-molecule uremic toxins. Blood Purif. 51 (11), 923–931. https://doi.org/10.1159/000521886 (2022).
García-Prieto, A. et al. Evaluation of the efficacy of a medium cut-off dialyser and comparison with other high-flux dialysers in conventional haemodialysis and online haemodiafiltration. Clin. Kidney J. 11 (5), 742–746. https://doi.org/10.1093/ckj/sfy004 (2018).
Belmouaz, M. et al. Comparison of the removal of uraemic toxins with medium cut-off and high-flux dialysers: a randomized clinical trial. Nephrol. Dial Transplant. 35 (2), 328–335. https://doi.org/10.1093/ndt/gfz189 (2020).
Maduell, F. et al. High-permeability alternatives to current dialyzers performing both high-flux hemodialysis and postdilution online hemodiafiltration. Artif. Organs. 43 (10), 1014–1021. https://doi.org/10.1111/aor.13480 (2019).
Rambabova, B. I. et al. Efficacy of medium cut-off dialyzer and comparison with standard high-flux hemodialysis. Blood Purif. 50 (4–5), 492–498. https://doi.org/10.1159/000511983 (2021).
Ronco, C., Marchionna, N., Brendolan, A., Neri, M. & Lorenzin, A. Martínez Rueda, A.J. expanded haemodialysis: from operational mechanism to clinical results. Nephrol. Dial Transplant. 33 (suppl_3), 41–47. https://doi.org/10.1093/ndt/gfy202 (2018).
Maduell, F. et al. Determining factors for hemodiafiltration to equal or exceed the performance of expanded hemodialysis. Artif. Organs. 44 (10), E448–E458. https://doi.org/10.1111/aor.13700 (2020).
Hadad-Arrascue, F., Nilsson, L. G., Rivera, A. S., Bernardo, A. A. & Cabezuelo Romero, J. B. Expanded hemodialysis as effective alternative to on-line hemodiafiltration: a randomized mid-term clinical trial. Ther. Apher Dial. 26 (1), 37–44. https://doi.org/10.1111/1744-9987.13700 (2022).
Kirsch, A. H. et al. Performance of hemodialysis with novel medium cut-off dialyzers. Nephrol. Dial Transplant. 32 (1), 165–172. https://doi.org/10.1093/ndt/gfw310 (2017).
Zhao, Y., Gan, L., Niu, Q., Ni, M. & Zuo, L. Efficacy and safety of expanded hemodialysis in hemodialysis patients: a meta-analysis and systematic review. Ren. Fail. 44 (1), 541–550. https://doi.org/10.1080/0886022x.2022.2048855 (2022).
Cho, N. J. et al. Long-term effect of medium cut-off dialyzer on middle uremic toxins and cell-free hemoglobin. PLoS ONE. 14 (7), e0220448. https://doi.org/10.1371/journal.pone.0220448 (2019).
Ward, R. A., Greene, T., Hartmann, B. & Samtleben, W. Resistance to intercompartmental mass transfer limits beta2-microglobulin removal by post-dilution hemodiafiltration. Kidney Int. 69 (8), 1431–1437. https://doi.org/10.1038/sj.ki.5000048 (2006).
Rudnicki, M., Frølich, A., Haaber, A., Tvedegaard, E. & Thode, J. Serum ionized calcium, parathyroid hormone and phosphate in uremic patients during and between hemodialysis. Clin. Nephrol. 40 (4), 225–229 (1993).
Maduell, F. et al. Comparison of four medium cut-off dialyzers. Clin. Kidney J. 15 (12), 2292–2299. https://doi.org/10.1093/ckj/sfac167 (2022).
Krieter, D. H., Rüth, M., Lemke, H. D. & Wanner, C. Clinical performance comparison of two medium cut-off dialyzers. Ther. Apher Dial. 27 (2), 284–292. https://doi.org/10.1111/1744-9987.13919 (2023).
Potier, J., Queffeulou, G. & Bouet, J. Are all dialyzers compatible with the convective volumes suggested for postdilution online hemodiafiltration? Int. J. Artif. Organs. 39 (9), 460–470. https://doi.org/10.5301/ijao.5000525 (2016).
Maduell, F. et al. Medium cut-off dialyzer versus eight hemodiafiltration dialyzers: comparison using a global removal score. Blood Purif. 48 (2), 167–174. https://doi.org/10.1159/000499759 (2019).
Boschetti-de-Fierro, A., Voigt, M., Storr, M. & Krause, B. Extended characterization of a new class of membranes for blood purification: the high cut-off membranes. Int. J. Artif. Organs. 36 (7), 455–463. https://doi.org/10.5301/ijao.5000220 (2013).
Locatelli, F. et al. Hemofiltration and hemodiafiltration reduce intradialytic hypotension in ESRD. J. Am. Soc. Nephrol. 21 (10), 1798–1807. https://doi.org/10.1681/asn.2010030280 (2010).
Kawanishi, H. Is there enough evidence to prove that hemodiafiltration is superior? Blood Purif. 46 (1), 3–6. https://doi.org/10.1159/000487917 (2018).
Bergström, J. & Wehle, B. No change in corrected beta 2-microglobulin concentration after cuprophane haemodialysis. Lancet 1 (8533), 628–629. https://doi.org/10.1016/s0140-6736(87)90266-2 (1987).
Acknowledgements
The authors would like to thank participants and all staff working at the hemodialysis unit of Srinagarind Hospital, Faculty of Medicine, Khon Kaen University, for their active participation in the project.
Funding
This study was supported by funding from the Kidney Foundation of Thailand (604/2565) and the Faculty of Medicine, Khon Kaen University (funding number IN66046).
Author information
Authors and Affiliations
Contributions
Conceptualization, Project administration: E.L., S.A.; Data curation: E.L., H.K., M.P., P.P.; Formal analysis: S.A.; Investigation: E.L., H.K., M.P., P.P.; Writing–original draft: E.L., H.K.; Supervision or mentorship: S.A. All the authors provided critical feedback, commented on the manuscript and agreed to the final version of the manuscript for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki that was approved by the Ethics Committee for Human Research, Faculty of Medicine, Khon Kaen University (KKU), Thailand (HE 651368). All the participants provided written informed consent before the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lukkanalikitkul, E., Kidkaem, H., Phonrat, M. et al. A randomized trial comparing medium cut-off membrane dialyzers with online hemodiafiltration for uremic toxins clearance in hemodialysis patients. Sci Rep 15, 5467 (2025). https://doi.org/10.1038/s41598-025-89197-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-89197-5
Keywords
This article is cited by
-
Selected tryptophan metabolites and inflammatory molecules in hemodialyzed patients
Pharmacological Reports (2026)
-
Evaluating mechanical and thermodynamic properties of asphalt mixtures containing anti freezing agents against low temperature cracking
Scientific Reports (2025)