Abstract
The impact of hypoxic repetitive sprint training on the overall performance of team sports remains controversial due to the specific nature of the exercise capacity required for team sports. While taurine and caffeine are widely utilized as supplements for repetitive sprint exercise in normoxic environments, their efficacy in hypoxic environments remains to be fully understood. Therefore, additional research is needed to explore the role of supplementation in hypoxic conditions. This study was to investigate the effects of caffeine (C), taurine (T), caffeine, and taurine co-ingestion (TC) or placebo (P) on repetitive sprint exercise performance and related physiological responses after exhaustion exercise in team athletes under simulated hypoxic conditions. A double-blind crossover randomized controlled experimental design was employed. 16 male participants (Age:23.69 ± 2.15 years, Body mass: 75.04 ± 7.79 kg, Height:1.78 ± 0.06 m) volunteered to receive four different supplement ingestions to complete the exercise tests: (1) placebo (5 mg/kg maltodextrin), (2) taurine (50 mg/kg), (3) caffeine (5 mg/kg), (4) taurine + caffeine (50 mg/kg + 5 mg/kg). All selected participants were university football players who had undergone rigorous training regimens (85–95% of maximum heart rate, duration of 60 min, with more than five training sessions per week). All participants completed an exhaustion test and subsequent repetitive sprint exercise in a simulated hypoxic environment (A simulation of a soccer game in sports mode). Time to exhaustion (TTE), peak power (PP), and mean power (MP) were recorded at the end of the exhaustion test and during the repetitive sprint exercise, respectively. This study designed an exercise protocol for repetitive sprinting after exhaustion exercise based on the pattern of play in football. The following variables were monitored throughout the experiments: heart rate (HR), blood lactate (B[La]), arterial oxygen saturation (SpO2), dyspnea, and rating of perceived exhaustion (RPE). The Stroop Test was administered at three separate time points: pre-test, mid-test, and post-test, throughout the exercise trial. The countermovement jump test (CMJ) was carried out at three specific time points: before the test, 3 min after the test, and 6 min after the test. The caffeine (C:618.56 + 42.50 s, p = 0.027, d = 0.996) and taurine + caffeine (TC: 613.69 + 37.74 s, p = 0.041, d = 0.902) groups significantly improved time to exhaustion compared to the placebo group. Blood lactate was significantly higher in the taurine + caffeine group than in the placebo group after repetitive sprint exercise (P: 9.87 ± 1.97, TC: 12.31 ± 2.54, p = 0.016). The caffeine group significantly reduced dyspnea, and rating of perceived exhaustion after repetitive sprint exercise (p < 0.05). The taurine (T: 43.42 ± 3.46, p = 0.005), caffeine (C: 44.11 ± 4.72, p < 0.001), and taurine + caffeine (TC: 43.04 ± 3.30, p = 0.011) groups all showed an increase in pre-exercise countermovement jump height. The caffeine group significantly reduced the consistent response time (p = 0.023) and inconsistent response time (p < 0.001) in the Stroop Test compared to the placebo group. Caffeine, along with combined taurine, significantly prolonged the duration of exhaustion exercise in a hypoxic environment; however, it did not affect subsequent repetitive sprint performance. Additionally, caffeine supplementation had a positive impact on cognitive performance during hypoxic training.
Similar content being viewed by others
Introduction
Repeated sprint training in hypoxia (RSH), defined as the repetition of several short (≤ 30 s) ‘all-out’ exercise bouts in hypoxia interspersed with incomplete recoveries (exercise-to-rest ratio < 1:4)1. RSH was developed as a solution to the limited hypoxic stimulation provided by traditional hypoxic training methods2,3, and it is commonly employed in team sports4. Hypoxic training is a form of training that improves the body’s capacity for adaptation by restricting the supply of oxygen. Curiously, the impact of RSH on enhancing team athlete-specific performance improvement, as depicted in the literature, remains somewhat inconclusive3,5. The hypoxic environment contributes to the inability of the athlete to maintain the appropriate intensity, which results in insufficient stimulation of the organism by hypoxic training. Combined with the characteristics of the exercise program, the inconsistent results are mainly due to the multifaceted nature of team sports, which require a mixture of aerobic and anaerobic activities, and the considerable differences between different training protocols6,7,8. Hence, extensive research is still needed to further advance the effects of RSH on team sports.
In addition, current international competitions and high-altitude training present greater demands on athletes regarding hypoxic training (eg: FIFA World Cup qualifiers in La Paz, High-Altitude Training Camps). Hypoxic environments will inevitably affect the performance of team athletes in training or competition9,10. While taurine and caffeine are frequently utilized as ergogenic aids (EA) to enhance team sport performance in normoxic environments11,12,13,14, their effectiveness in hypoxic conditions has received comparatively less attention in research. The ability to perform repeated sprints plays a crucial role in determining the competition level of athletes in team sports such as football and rugby15. However, as exercise fatigue develops during a match, especially in the second half, repeated sprints after fatigue become more important16. Therefore, this study designed an exercise protocol for repetitive sprinting after exhaustion exercise based on the pattern of play in football.
Caffeine (CAF) is a xanthine alkaloid compound and the main ingredient in many sports drinks on the market17. The beneficial impact of CAF on aerobic endurance and sprinting in normoxic conditions has been substantiated through numerous studies18,19. Yet, the effects of CAF in hypoxic conditions have received less attention. Fulco et al. demonstrated that 4 mg kg− 1 CAF significantly improved time to exhaustion during cycling at 80% of maximal oxygen uptake under acute hypoxic (4300 m above sea level) conditions20. However, Stadheim et al. demonstrated that CAF (4.5 mg kg− 1) was not found to improve overall performance of cross-country skiers in an 8-km cross-country double pole time trial at a simulated altitude of 2000 m above sea level21. In addition, it has been shown that CAF can partially counteract these negative effects of neuromuscular fatigue by decreasing activity in the premotor and motor areas of the cortex required for exercise22. However, whether the hypoxic environment, as an additional stimulus, affects the effects of CAF still needs to be further investigated. Enhancing athletic performance involves not only advancements in physical abilities, but also improvements in mental and executive functioning during exercise23. CAF is a central nervous system stimulant, which enhances executive function by augmenting attention and inhibitory transitions24. This plays an important role in the overall performance of team sports25. Therefore, a better understanding of the physiological, perceptual, and performance effects of CAF during team sports in hypoxic conditions is necessary.
Taurine (TAU) is a sulphur-containing amino acid that plays a beneficial role in various exercise metabolic and physiological processes12. Such as glucose26 and lipid regulation27, energy metabolism, anti-inflammatory regulation and antioxidant effects28. TAU has been extensively studied in aerobic endurance and sprint exercise under normoxic conditions12,29,30,31. A meta-analysis reported that oral TAU (1–6 g) significantly improved endurance sport performance26. Yusuf Buzdağlı et al. showed that acute TAU supplementation enhanced 30-s WanT in speed skaters without altering neuromuscular recovery29. Since the exact mechanism by which TAU affects exercise performance is not clear, the effects of different exercise intensities and doses on exercise performance are still controversial12. In addition, some animal studies have shown that TAU can help to maintain the stability of cell membranes under acute hypoxic conditions, and TAU has antioxidant effects, which can neutralise free radicals, reduce oxidative damage and protect cells from oxidative stress32. However, there is a lack of research on human experiments in hypoxic environments.
TAU and CAF as one of the main ingredients are usually found together in common sports drinks on the market33. Extensive research has demonstrated that combining these two ingredients under normoxic conditions improves endurance34, repetitive sprinting35, and cognitive performance13, which plays an important role in team sports performance. However, the components except TAU and CAF were not fully isolated in some of the studies, so the effect of the combination of the two still needs to be explored in a large number of studies36,37,38. Also, the combination of the two in hypoxic environments has not been studied. In summary, we have a bold hypothesis that TAU combined with CAF in a hypoxic environment can be effective in improving repetitive sprint exercise performance and cognitive after exhaustion exercise. Therefore, the present experiment was conducted to investigate the effects of TAU combined with CAF on team sports performance in a hypoxic environment through physiological, perceptual and performance parameters.
Methods
Study design
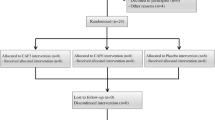
The study adopted a double-blind randomized crossover design. The participants reported to the laboratory on five occasions at the same time of day. The initial visit was for familiarization with the experimental equipment and procedures, while the last four visits consisted of actual experimental trials (Fig. 1). Each visit was separated by no more than 3 days for each participant, which was deemed sufficient to wash out taurine and caffeine29,35. All participants gave written informed consent. Ethical approval was provided by the institutional ethics committee, which was conducted in accordance with the 1964 Helsinki Declaration.
Familiarization and standardizations
On the first visit, participants completed the Physical Activity Readiness Questionnaire (PAR-Q) and informed consent. The researchers maintained the laboratory temperature at 20–22 °C, activated the hypoxia generator (HYP123 Altitude Generator, Hypoxico, American) to simulate a hypoxic environment equivalent to 2500 m above sea level, and monitored the 15.3% oxygen concentration in the hypoxic room. Participants were fitted to an electronically braked cycle ergometer, where saddle and handlebar position was recorded for all subsequent trials. The test protocol was explained to the participants in detail, and was demonstrated by one of the research team. The participants were also shown the RPE and dyspnea scale, and provided with instructions of how to interpret their score. Participants then completed an exhaustion test and a 6 × 10-s full-speed sprint exercise on a bicycle. Standardized encouragement and feedback were given to the participants in all trials by the same researcher. Participants were asked to avoid strenuous exercise for 48 h prior to testing. In addition, the food consumed by each participant during 24 h before the familiarizing experiment was recorded. Participants were asked to replicate this diet prior to subsequent trials. To ensure optimal conditions for experimental testing, participants should not eat for two hours prior to testing. The participants were also provided with an extensive list of dietary sources containing caffeine and taurine, which they were instructed to avoid throughout the test period.
Procedure
This study designed an exercise protocol for repetitive sprinting after exhaustion exercise based on the pattern of play in football31,35,39,40. On the experimental trial days, resting heart rate, blood lactate, RPE, and dyspnoea were measured 60 min after taking the supplement; then, they started the prescribed warm-up exercise, consisting of a 5-min period of submaximal cycling (70 rpm, 60 W) followed by three 3-s maximal sprints. At the end of the warm-up, participants performed the countermovement jump (CMJ) test and the Stroop test. Immediately after a wash-in period of 5-min breathing hypoxic air, an incremental exercise test (Starting at 50W and increasing by 30W per minute) was performed in hypoxia until exhaustion on a cycle ergometer. A rest period of 15 min and a Stroop test in between, followed by six 10-s full sprints with 50 s between each31. The CMJ and the Stroop test were repeated at the corresponding time points after the sprint exercise (As shown in Fig. 2).
Participants
Given the typical effect sizes (Cohen’s d = 0.25) reported with caffeine35 and taurine31 across the various dependent variables in this study, we used G ∗ Power41 (version 3.1.9.7) to determine an a priori sample size of 11. This was considered adequate to detect differences between groups with a power of 0.85. The possibility of type I and II errors was set at 5%. Effect size = 0.25, statistical test = repeated measures, with-between interaction, number of groups = 4. Nonetheless, we selected 16 participants for this study to avoid insufficient data due to sample attrition. Considering the potential for shedding or injury, we recruited 16 male university football players. In this study, the basic information of the participants was excluded by means of a questionnaire. The inclusion criteria were as follows: 2) participants were healthy with no diagnoses of cardiovascular, respiratory, or metabolic diseases that may impair muscle biology; 2) participants had not been in a hypoxic simulated environment or on a plateau for the 6 months preceding the experiment; 3) No chronic CAF use habits; 4) they were right-handed, and they could perform high-intensity exercise and had normal executive functions. Participants were indeed provided with compensation and incentives. These measures not only helped improve participant compliance but also ensured a higher quality of research design. The details are shown in Table 1.
Supplementation interventions
Participants were measured to calculate taurine and caffeine doses before the start of each experiment. The dosages of caffeine17 (ALLMAX, 200 mg, Purity:100%) and taurine31 (YONGAN, 100 g, Purity: ≥ 98.5% ) followed the recommendations of recent studies. All of the supplements were formulated as powders, quantified with an analytical balance (JI MING, China), and taken in gelatine capsules. Participants were randomly assigned to one of the four groups (T, C, TC, or P) using a computer-generated randomization list. This minimized selection bias. The administration of capsules was conducted by personnel not involved in the data analysis. They provided the capsules to participants without disclosing the contents.The capsules contained one of the following: taurine (T) (50 mg/kg BM), caffeine (C) (5 mg/kg BM), caffeine + taurine (TC) (5 mg/kg BM + 50 mg/kg BM) or a placebo (P) (maltodextrin) (5 mg/kg BM). Participants were supplemented 1 h before the experiment, based on the time of peak plasma utilization of both taurine12 and caffeine42 after oral administration. Throughout the experiment, we conducted regular checks to ensure that the blinding process was maintained, with all personnel reminded of the importance of not disclosing group information.
Measures and data collection
Primary outcome
Exhaustion test. The participants underwent an incremental cycling test using a ramp protocol on a cycle ergometer (LODE 906900, Netherlands). The test began at a workload of 60 W and increased by 30 W every minute until volitional exhaustion, with a pedaling cadence of 70 rpm. All participants must satisfy at least one of the following two exhaustion criteria: a) the participant has reached or exceeded his maximum heart rate; b) The test was terminated when the pedaling cadence dropped by more than 5 rpm below 70 rpm for more than 10 s, despite strong verbal encouragement. Time to exhaustion (TTE) is recorded immediately at the end of the exhaustion test.
Repeat sprint ability test (RSA). The repetitive sprint protocol consisted of a series of six 10-s sprints at 0.075 × body weight resistance, with each sprint interspersed with 50 s of recovery. Participants were asked to keep their hips on the seat during the riding, and the experimenter continuously gave verbal encouragement and time cues to the participants. The performance variables measured on the ergometer were: peak power (maximum power output reached during each 10-s full-speed sprint, PP); mean power (average power output during each 10-s full-speed sprint, MP). The peak and mean power outcomes were calculated via Monark Anaerobic Testing software (Ergomedic 894E, Sweden) after every test.
Countermovement jump (CMJ). The countermovement jump (CMJ) is the vertical jump test that is more frequently used to assess the jumping performance and the neuromuscular status43. The vertical jump height was obtained by applying vertical jump pad (Omegewave, Finland). Participants start in an upright static position with their arms crossed. The participants have to be instructed to squat by bending the knees at approximately 90◦ angles as quickly as possible. Then, they should jump as high as possible, keeping the legs straight, and landing with both feet together. Repeat the jump three times, taking the average height44.
Stroop test. The Stroop task is widely used to evaluate selective attention, cognitive flexibility, and processing speed45. The task was programmed and performed using E-prime 1.0 software (Psychology Software Tools, Pittsburgh, PA, USA). Every trial was shown with a fixed cross in the middle of the screen for 500 ms, and then a stimulus for another 500 ms. Congruent and incongruent stimuli were present dis equal measure. Three colour words—green, blue, and red—with font colours that matched each word’s colour were displayed in the congruent condition. The same three words were displayed in the incongruent condition, but the font colour did not correspond to the colour the word suggested (for example, the word “green” was displayed in a red or blue font). On a numerical keypad, the participants had to select the presenting colour of each word; the numbers 1, 2, and 3 stood for blue, green, and red, respectively. The participants used their index, middle, and ring fingers of their right hand to press the keys, which were situated in the left-to-right order of 1, 2, and 3. The RT and accuracy rate (ACC) were measured.
Physiological and perceptual measures
Participants underwent baseline index (BL) collection 60 min after taking the supplement. [blood lactate (B[La]), heart rate (HR), rating of perceived exertion (RPE), and Dyspnea]. Using the Polar RS 800 CX telemetric heart rate monitor (Polar H10, Finland), HR was measured continuously for 5 min, with the final 1 min reported. Capillary blood samples were taken from the participant’s earlobes using a blood collection needle and B[La] was measured using a Sirius R lactate tester (SensLab GmbH, Germany). For all B[La] measurements, two samples were taken and the mean calculated. Using finger-clip oximetry (Haier 82A1, China), we monitored the arterial oxygen saturation (SpO2) of the subjects in real time to assess acute altitude sickness and facilitate the timely implementation of appropriate protective measures. The second, third, and fourth measurements of HR, B[La], RPE (Borg 6–20), and Dyspnea (Borg 0–10) were taken respectively at the end of the exhaustion test (END1), at the middle of the interval (MID), and at the end of the repeated sprint exercise (END2).
Statistics and analysis
Data are presented as the mean ± standard deviation (SD). The Shapiro–Wilk test was used to determine whether all the variables obtained showed a normal distribution. Sphericity was tested using Mauchly’s test, and in cases of violation, the Greenhouse–Geisser correction was applied. TTE were analyzed using one-way ANOVA with Bonferroni correction following confirmation of normality via the Shapiro–Wilk test. CMJ, Stroop test, PP, MP HR, B[La], RPE, and Dyspnea were subjected to two-way repeated measures analysis of variance (RM-ANOVA) with condition (P, T, C, TC) and time as independent variables. Bonferroni correction was used for performing post-hoc analyses where appropriate. All data were analyzed using SPSS software (IBM SPSS statistics 27, New York, USA). The statistical significance level was set at P ≤ 0.05.
Results
Primary outcome
Time to exhaustion (TTE)
TTE at the point of exhaustion was recorded for P (M = 566.94, SD = 54.35), T (M = 584.19, SD = 67.59), C (M = 618.56, SD = 42.50), and TC (M = 613.69, SD = 37.74) groups respectively. Data show significant differences in TTE between interventions, F (3,33.71) = 3.86, MSE = 2688.28, p = 0.02, r = 0.37. Follow-up multiple comparisons showed that the TTE of group P was significantly lower than that of groups C [t (60) = 0.154, p = 0.027, d = 0.996] and TC [t (60) = 0.139, p = 0.041, d = 0.902] (As shown in Fig. 3).
Repeat sprint ability test (RSA)
For PP, there was a Supplementation by Time interaction [F(5,58) = 2.679; p = 0.03; η2partial = 0.188] as well as a Time effect [F(5,56) = 121.415; p < 0.001; η2partial = 0.916] but no Supplementation effect [F(3,60) = 0.31; p = 0.818; η2partial = 0.015]. Post-hoc analysis revealed that PP declined as the number of sprints increased in the four groups. And there was no significant difference between the four supplements (As shown in Fig. 4).
For MP, there was a Supplementation by Time interaction [F(5,58) = 9.165; p < 0.001; η2partial = 0.441] as well as a Time effect [F(5,56) = 147.469; p < 0.001; η2partial = 0.929] but no Supplementation effect [F(3,60) = 1.673; p = 0.182; η2partial = 0.077]. Post-hoc analysis revealed that MP declined as the number of sprints increased in the four groups. And there was no significant difference between the four supplements (As shown in Fig. 4).
Countermovement jump (CMJ)
There was a Supplementation by Time interaction [F(3,60) = 12.214; p < 0.001; η2partial = 0.379] as well as a Time effect [F(3,59) = 175.185; p < 0.001; η2partial = 0.856] but no Supplementation effect [F(3,60) = 0.684; p = 0.565; η2partial = 0.033]. Post hoc multiple comparisons revealed significant reductions in 3MIN and 6MIN after the exercise test for all four groups. Additionally, the T (T: 43.42 ± 3.46, p = 0.005), C (C: 44.11 ± 4.72, p < 0.001), and TC (TC: 43.04 ± 3.30, p = 0.011) groups exhibited significantly higher values than the P (P:38.58 ± 3.86) group at BL (As shown in Fig. 5).
Stroop test
Consistent conditions
For RT, this study analysis unveiled an effect of time [F(2,59) = 156.75; p < 0.001; η2partial = 0.723], supplementation[F(3,60) = 2.53; p = 0.066; η2partial = 0.112], and a supplementation × time interaction [F(3,60) = 27.545; p < 0.001; η2partial = 0.579]. Post hoc multiple comparisons revealed a gradual decrease in RT as the number of tests increased. Also, RT was significantly lower in group C compared to groups P(p = 0.025) and T(p = 0.023) at MID. RT was significantly lower in group C compared to groups P(p < 0.001) and T(p < 0.001) at END. For ACC, there was no significant supplementation, time, and supplementation × time interaction effect ( as shown in Tables 2 and 3).
Inconsistency conditions
For RT, this study analysis unveiled an effect of time [F(2,59) = 31.07; p < 0.001; η2partial = 0.513], supplementation [F(3,60) = 8.64; p < 0.001; η2partial = 0.302], and a supplementation × time interaction [F(3,60) = 11.35; p < 0.001; η2partial = 0.362]. Post hoc multiple comparisons revealed a gradual decrease in RT as the number of tests increased. Also, RT was significantly lower in group C compared to groups P (p < 0.001), T (p < 0.001) and T (p < 0.001) at MID. RT was significantly lower in group C compared to groups P(p = 0.001), T (p < 0.001) and TC (p = 0.008) at END. For ACC, there was no significant supplementation, time, and supplementation × time interaction effect ( as shown in Tables 2 and 3).
Physiological and perceptual measures
Heart rate (HR)
There was a time main effect of HR change in the four groups during the exercise [F(3,58) = 2280.048; p < 0.001; η2partial = 0.992]; however, there were no main effects for supplementation[F(3,60) = 1.37; p = 0.261; η2partial = 0.064], nor was there an interactions[F(3,60) = 2.872; p = 0.044; η2partial = 0.126]. HR at the end of the two exercises was significantly higher than BL and MID (p < 0.05) (As shown in Fig. 6).
Blood lactate (B[La])
This study analysis unveiled an effect of time [F(3,58) = 505.276; p < 0.001; η2partial = 0.963], supplementation [F(3,60) = 4.507; p = 0.006; η2partial = 0.184], and a supplementation × time interaction [F(3,60) = 4.094; p = 0.01; η2partial = 0.170]. Post hoc multiple comparisons revealed significantly higher B[La] at END1(p < 0.05) and END2(p < 0.05) than at MID. Also, the TC group had higher B[La] compared to the P group at END2(P: 9.87 ± 1.97, TC: 12.31 ± 2.54, p = 0.016) (As shown in Fig. 7).
Arterial oxygen saturation (SpO2)
This study analysis unveiled an effect of time [F(3,58) = 505.276; p < 0.001; η2partial = 0.979], supplementation [F(3,60) = 2.782; p = 0.049; η2partial = 0.122], and a supplementation × time interaction [F(3,60) = 2.176; p = 0.05; η2partial = 0.120]. Post hoc multiple comparisons revealed significantly higher SpO2 at END1(p < 0.05) and END2(p < 0.05) than at MID. Also, the C group had higher SpO2 compared to the P group at END1(P: 83.94 ± 2.11, C: 86.5 ± 2.16, p = 0.01) (As shown in Fig. 8).
Rating of perceived exertion (RPE)
This study analysis unveiled an effect of time [F(3,58) = 1091.923; p < 0.001; η2partial = 0.983], supplementation [F(3,60) = 2.662; p = 0.05; η2partial = 0.117], and a supplementation × time interaction [F(3,60) = 8.463; p < 0.001; η2partial = 0.297]. Post hoc multiple comparisons revealed significantly higher RPE at END1(p < 0.05) and END2(p < 0.05) than at MID. Also, the C (C: 16.88 ± 1.15) group had lower B[La] compared to the P (P: 18.44 ± 1.15, p = 0.001), T (T: 18.56 ± 1.03, p < 0.001), and TC (TC: 18.5 ± 1.10, p < 0.001) groups at END2 (As shown in Fig. 9).
Dyspnea (Borg 0–10)
There was a Supplementation by Time interaction [F(3,60) = 7.131; p < 0.001; η2partial = 0.263] as well as a Time effect [F(3,58) = 691.587; p < 0.001; η2partial = 0.973] but no Supplementation effect [F(3,60) = 1.845; p = 0.149; η2partial = 0.084]. Post hoc multiple comparisons revealed significantly higher Dyspnea at END1(p < 0.05) and END2(p < 0.05) than at MID. Also, the C group had lower Dyspnea compared to the TC group at END2(C: 6.13 ± 1.31, TC: 7.50 ± 1.27, p = 0.011) (As shown in Fig. 10).
Discussion
The efficacy of hypoxic repetitive sprint training for team sports remains a topic of debate due to the specificity of the specialized abilities required for team sports4. In team competitions, repetitive sprinting often occurs following fatigue or during a state of incomplete recovery46. Therefore, the present study was specifically designed to investigate the effects of repetitive sprinting exercise after exhaustion exercise. Meanwhile, in order to enhance exercise performance during hypoxic training or competition, we have introduced taurine and caffeine, which are commonly used and studied in normoxic environments, as ergogenic aids12,17,47, but the effects of these two supplements in hypoxic environments need to be further investigated.
In this study, we found that both the C group and TC group exhibited significantly prolonged fatigue performance under hypoxic conditions. However, there were no significant differences among the four groups in subsequent repeated sprint performance. Additionally, caffeine had effects on CMJ, Stroop task, RPE, and Dyspnea during exercise, which may contribute to enhancing overall team performance. However, the T group did not show a significant advantage in the exhaustion test and repeated sprint performance.
In partial support of our hypothesis, the present study demonstrated that TTE was significantly higher in the C and TC groups compared to the P group in the hypoxic environment. Consistent with previous studies, Yinhang Cao et al. showed that 4 mg/kg caffeine supplementation in a moderately hypoxic environment (2500 m above sea level) improved performance in the VO2peak by 80% and the Incremental cycling test22. This study concluded that moderate doses of caffeine enhance the ventilatory response to hypoxia, which may be the main reason for improving exercise performance in hypoxic environments. From the SpO2 at the end of the exhaustion test in our study, it was found that the group C was significantly higher than the group P. Therefore, the reason for this phenomenon in our study may be consistent with that study, due to caffeine supplementation increasing the ventilatory response thereby increasing SpO2.
Unlike previous studies conducted in normoxic environments, the current study revealed that taurine in hypoxic environments did not result in a significant improvement in TTE. It has been shown that taurine has antioxidant properties, promotes its stabilizing effect in the mitochondrial matrix and increases the efficiency of ATP turnover in myocytes, which in turn promotes endurance exercise performance30,48. However, exercise in a hypoxic environment inevitably disrupts this physiological homeostasis49. Research has indicated that long-term taurine supplementation may enhance the body’s ability to utilize oxygen in hypoxic environments. This adaptation could potentially provide performance benefits over time, suggesting that while acute taurine supplementation may not yield immediate performance enhancements.Also, the dosage of taurine utilised in our study is often used to improve exercise performance in normoxic conditions, However, further research is needed to select a dosage of taurine that better adapts to or counteracts the effects of hypoxic environments on exercise. Surprisingly, our research revealed that the TC group significantly increased TTE. Previous research has demonstrated a beneficial synergistic effect of combining taurine and caffeine in normoxic environments13,14,50. Additionally, animal studies have indicated that taurine can enhance calcium processing in muscle fibers when combined with physiological doses of caffeine51. However, the investigation of this combination in hypoxic environments has received comparatively less attention. In contrast to the effect of caffeine, the present study did not identify any significant influencing factors from various physiological indicators. This outcome could potentially be attributed to the predominant contribution of caffeine in the combined supplementation. Whether there is any accumulation or interaction in the hypoxic environment still needs further study.
Regrettably, despite previous studies showcasing the effects of taurine and caffeine supplementation on repetitive sprint performance in team sports under normoxic conditions, the present study did not observe significant differences in repetitive sprint performance among the four groups in the hypoxic environment following exhaustion exercise. These contrasting outcomes may be attributed to variations in exercise protocols, intensities, and dose–response mechanisms employed in different studies13,14,34,35,36. Consistent with previous research, Mark Waldron et al. demonstrated that 50 mg/kg taurine improves exhaustion exercise performance, but this effect may not be sustained over a longer period of exercise and may require longer recovery, resulting in a non-significant effect in subsequent repetitive sprint exercise31. Therefore, it is important to focus on the duration of the effect of ergogenic aids52, particularly for team sports that require maintaining repetitive sprint performance in a fatigued state. In addition, our study also found that B[La] was significantly higher in the C, TC group than in the P group at the end of the repetitive sprint exercise. Compared to other studies, the combined supplementation significantly improved exercise performance, resulting in higher B[La]34,35,38. However, this study failed to find a significant improvement in repetitive sprinting with combined supplementation. Due to the specificity of the exercise protocol in this study, fewer studies have focused on repeated sprint performance after fatigue, making it more difficult to interpret. The present study suggests that a possible explanation is that the C and TC groups experienced a greater degree of peripheral fatigue to sustain the subsequent sprint performance due to the significantly prolonged duration of exhaustion, resulting in an elevation of B[La].
The CMJ has been one of the most used tests for monitoring neuromuscular status in individual, and team sports, as well as the military44. This study found that before exhaustion exercise, the T, C, and TC group had a significant increase in CMJ, but there was no significant difference in CMJ after exercise compared to group P. Consistent with previous studies, Yusuf Buzdağlı et al. showed that acute taurine supplementation significantly increased CMJ before anaerobic exercise in speed skaters, but had no significant effect on CMJ after exercise29. Research has shown that TAU has a positive effect on intramuscular and intermuscular coordination, resulting in improved jumping performance53. However, how taurine affects muscle activity after fatigue is not clear still further research is needed. Furthermore, Jørgen Jensen et al. showed that caffeine increased CMJ at rest; however, the positive effects of caffeine were no longer significant after maximal effort intensity training54. And this study indicates that the consumption of caffeine can enhance exercise performance, resulting in heightened fatigue and increased physiological indications of stress, which in turn affect the effects of caffeine. Compared to caffeine and taurine supplementation, our study did not find additional effects of combined supplementation on neuromuscular status. Several studies have reported that both taurine and caffeine have the ability to modulate intracellular calcium ion handling and influence the sensitivity of myogenic fibers in skeletal muscle to calcium ions55,56,57. And, in vitro studies have shown that taurine’s effects on peak force (~ 29%) and rate of force development (~ 28%) is augmented in the presence of a physiological dose of caffeine58. However, the exact mechanism underlying these effects remains unclear, and further research is needed to gain a comprehensive understanding of potential cumulative or reciprocal outcomes following fatigue.
In team sports, it is important to maintain optimal cognitive performance during training and competition. Our study found that Group C significantly improved cognitive performance during exercise (consistent and inconsistent RT). Consistent with previous research, Souissi et al. found that caffeine ingestion at 6 mg/kg significantly improved cognitive function related to aspects of attention during repetitive sprint exercise59. Furthermore, previous research has demonstrated that the performance in the Stroop task can be clarified by the fact that caffeine, acting as a stimulant for the central nervous system, intensifies neural activity and boosts local cerebral blood flow, stimulating the prefrontal cortex, which is linked to cognitive functioning60. The impact of caffeine on cognitive function in hypoxic environments has received relatively limited investigation. However, our study revealed that the effect of caffeine on cognitive function remained unaffected by hypoxic stimulation. This finding suggests a potential novel complementary measure for training and competition in hypoxic environments. In addition, caffeine has been extensively studied and found to reduce perceived fatigue and enhance endurance performance. In hypoxic conditions, this effect may be even more important, as limited oxygen supply can increase feelings of fatigue. Taurine, on the other hand, may alleviate fatigue by reducing lactic acid accumulation and enhancing antioxidant capacity. This could be the primary reason for the differing effects observed.
In this study, we found that heart rate was significantly higher in all 4 groups after exhaustion and repetitive sprint exercise in a hypoxic environment, but there was no significant difference between the 4 groups. Consistent with most studies in normoxic environments, both Ben Desbrow and Raci Karayigit et al. did not find an effect of taurine and caffeine on heart rate changes during exercise in their studies14,61. Under the normoxic environment, a study has shown that taurine can reduce the pre-exercise heart rate31, in addition, taurine combined with caffeine has a potential protective effect on the cardiovascular system62. However, the specific mechanism of action is still unclear, and the effects of taurine and caffeine on the cardiovascular system under different exercise intensities and exercise environments still need to be further studied.
The present study found that RPE and Dyspnea after repeated sprints were significantly lower in group C compared to group P in the hypoxic environment. This is similar to previous studies, where Yinhang Cao et al. found that 4 mg/kg caffeine intake significantly reduced RPE and Dyspnea during exercise20. And the study indicates that a decrease in subjective perception may serve as one of the primary mechanisms through which caffeine enhances exercise performance. Furthermore, Samuele M. Marcora et al. demonstrated that caffeine consumption improved the duration of high-intensity cycling under moderate hypoxic conditions by diminishing the perception of effort22. Although our study did not find improvements in repeated sprint performance in the C group, further research is needed to investigate whether it is due to caffeine reducing RPE and dyspnea, thereby maintaining repeated sprint performance after prolonged exhaustive exercise.
This study has several limitations that should be acknowledged. Firstly, the relatively small sample size of 16 participants may limit the statistical power of our findings. Secondly, the hypoxic conditions simulated at 2500 m may not accurately reflect the varying environmental factors encountered in real-world team sports, such as temperature, humidity, and altitude fluctuations. Thirdly, the reliance on self-reported dietary intake presents limitations due to potential inaccuracies in participants’ recall. To mitigate this, we provided participants with a dietary checklist; however, the inherent biases of self-reporting remain a concern. Finally, there is limited literature on taurine supplementation in hypoxic environments, which constrains our ability to discuss certain aspects in detail. Future research should explore the effects of caffeine and taurine supplementation across diverse demographic groups, including female athletes and non-athletes, to better understand population-specific responses. In addition, there is a need for long-term studies to assess the chronic effects of caffeine and taurine supplementation under hypoxic conditions, which could provide valuable insights into the sustainability of performance benefits over time.
In conclusion, although no significant improvement was found with taurine or caffeine on repetitive sprint performance after exercise in a hypoxic environment, the present study found that caffeine supplementation had an effect on TTE, CMJ, Stroop task, RPE and dyspnoea during exercise, which may be helpful in improving overall team sport performance. Additionally, for team sports, the improvement of exercise performance after fatigue was not addressed in this study, and much future research is needed to find optimal nutritional supplementation strategies.
Conclusion
In the hypoxic environment, both the C and TC groups exhibited a significant prolongation of exhaustion exercise performance. However, there were no significant differences among the four groups in the subsequent repetitive sprint performance. By all metrics, caffeine provided certain improvements in overall team sport performance but did not address the key issue, which is repeated sprint performance after fatigue. Continued exploration for optimal supplementation strategies is warranted in the future.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Ramos-Campo, D. J. et al. Effect of high-intensity resistance circuit-based training in hypoxia on aerobic performance and repeat sprint ability. Scand. Med. Sci. Sports 28, 2135–2143 (2018).
Millet, G., Girard, O., Beard, A. & Brocherie, F. Repeated sprint training in hypoxia – an innovative method. Dtsch. Z. Sportmed. 2019, 115–122 (2019).
Hamlin, M. J., Olsen, P. D., Marshall, H. C., Lizamore, C. A. & Elliot, C. A. Hypoxic repeat sprint training improves rugby player’s repeated sprint but not endurance performance. Front. Physiol. 8 (2017).
Faiss, R., Girard, O. & Millet, G. P. Advancing hypoxic training in team sports: from intermittent hypoxic training to repeated sprint training in hypoxia.
Inness, M. W. H., Billaut, F. & Aughey, R. J. Team-sport athletes’ improvement of performance on the Yo-Yo intermittent recovery test level 2, but not of time-trial performance, with intermittent hypoxic training. Int. J. Sports Physiol. Perform. 11, 15–21 (2016).
Goods, P. S. R., Dawson, B. T., Landers, G. J., Gore, C. J. & Peeling, P. Effect of different simulated altitudes on repeat-sprint performance in team-sport athletes. Int. J. Sports Physiol. Perform. 9, 857–862 (2014).
Girard, O., Brocherie, F. & Millet, G. P. Effects of altitude/hypoxia on single- and multiple-sprint performance: A comprehensive review. Sports Med. 47, 1931–1949 (2017).
Rojas-Valverde, D. et al. Exploring the impact of training methods on repeated sprints in hypoxia training effects. Oxygen 3, 366–373 (2023).
Böning, D. Altitude and hypoxia training—a short review. Int. J. Sports Med 18, 565–570 (1997).
Carr, A. J. et al. Training to compete at altitude: Natural altitude or simulated live high: Train low?. Int. J. Sports Physiol. Perform. 14, 509–517 (2019).
Lazić, A. et al. Acute effects of caffeine on overall performance in basketball players-a systematic review. Nutrients 14, 1930 (2022).
Kurtz, J. A., VanDusseldorp, T. A., Doyle, J. A. & Otis, J. S. Taurine in sports and exercise. J. Int. Soc. Sports Nutr. 18, 39 (2021).
Ozan, M. et al. Does single or combined caffeine and taurine supplementation improve athletic and cognitive performance without affecting fatigue level in elite boxers? A double-blind, placebo-controlled study. Nutrients 14, 4399 (2022).
Karayigit, R. et al. Combined but not isolated ingestion of caffeine and taurine improves wingate sprint performance in female team-sport athletes habituated to caffeine. Sports 9, 162 (2021).
Spencer, M., Bishop, D., Dawson, B. & Goodman, C. Physiological and metabolic responses of repeated-sprint activities: Specific to field-based team sports. Sports Med. 35, 1025–1044 (2005).
Noakes, T. D. Physiological models to understand exercise fatigue and the adaptations that predict or enhance athletic performance: Physiological models to study exercise. Scand. J. Med. Sci. Sports 10, 123–145 (2000).
Guest, N. S. et al. International society of sports nutrition position stand: caffeine and exercise performance. J. Int. Soc. Sports Nutr. 18, 1 (2021).
Wellington, B. M., Leveritt, M. D. & Kelly, V. G. The effect of caffeine on repeat-high-intensity-effort performance in rugby league players. Int. J. Sports Physiol. Perform. 12, 206–210 (2017).
Brown, S. J., Brown, J. & Foskett, A. The effects of caffeine on repeated sprint performance in team sport athletes—a meta-analysis. Sport Sci. Rev. 22, 25–32 (2013).
Lei, T.-H. Caffeine intake enhances peak oxygen uptake and performance during high-intensity cycling exercise in moderate hypoxia. Eur. J. Appl. Physiol.
Stadheim, H. K., Nossum, E. M., Olsen, R., Spencer, M. & Jensen, J. Caffeine improves performance in double poling during acute exposure to 2,000-m altitude. J. Appl. Physiol. 1985(119), 1501–1509 (2015).
Smirmaul, B. P. C., de Moraes, A. C., Angius, L. & Marcora, S. M. Effects of caffeine on neuromuscular fatigue and performance during high-intensity cycling exercise in moderate hypoxia. Eur. J. Appl. Physiol. 117, 27–38 (2017).
Bello, M. L., Walker, A. J., McFadden, B. A., Sanders, D. J. & Arent, S. M. The effects of TeaCrine® and caffeine on endurance and cognitive performance during a simulated match in high-level soccer players. J. Int. Soc. Sports Nutr. 16, 20 (2019).
De Almeida, R. F. et al. Effects of acute caffeine ingestion on cognitive performance before and after repeated small-sided games in professional soccer players: A placebo-controlled, randomized crossover trial. Nutrients 15, 3094 (2023).
Krenn, B., Finkenzeller, T., Würth, S. & Amesberger, G. Sport type determines differences in executive functions in elite athletes. Psychol. Sport Exerc. 38, 72–79 (2018).
Carneiro, E. M. et al. Taurine supplementation modulates glucose homeostasis and islet function. J. Nutr. Biochem. 20, 503–511 (2009).
Haidari, F., Asadi, M., Mohammadi-Asl, J. & Ahmadi-Angali, K. Evaluation of the effect of oral taurine supplementation on fasting levels of fibroblast growth factors, β-Klotho co-receptor, some biochemical indices and body composition in obese women on a weight-loss diet: A study protocol for a double-blind, randomized controlled trial. Trials 20, 315 (2019).
Yatabe, Y., Miyakawa, S., Ohmori, H. & Adachi, H. M. T. Effects of taurine administration on exercise. In Taurine 7 Vol. 643 (eds Azuma, J. et al.) 245–252 (Springer, 2009).
Buzdağlı, Y. et al. Taurine supplementation enhances anaerobic power in elite speed skaters: A double-blind, randomized, placebo-controlled, crossover study. Biol. Sport 40, 741–751 (2022).
Waldron, M., Patterson, S. D., Tallent, J. & Jeffries, O. The effects of an oral taurine dose and supplementation period on endurance exercise performance in humans: A meta-analysis. Sports Med. 48, 1247–1253 (2018).
Waldron, M., Knight, F., Tallent, J., Patterson, S. & Jeffries, O. The effects of taurine on repeat sprint cycling after low or high cadence exhaustive exercise in females. Amino Acids 50, 663–669 (2018).
Liu, Z. et al. Role of taurine supplementation to prevent exercise-induced oxidative stress in healthy young men. Amino Acids 26, 203–207 (2004).
Kerksick, C. M. et al. ISSN exercise & sports nutrition review update: Research & recommendations. J. Int. Soc. Sports Nutr. 15, 38 (2018).
Quinlivan, A. et al. The effects of red bull energy drink compared with caffeine on cycling time-trial performance. Int. J. Sports Physiol. Perform. 10, 897–901 (2015).
Warnock, R., Jeffries, O., Patterson, S. & Waldron, M. The effects of caffeine, taurine, or caffeine-taurine coingestion on repeat-sprint cycling performance and physiological responses. Int. J. Sports Physiol. Perform. 12, 1341–1347 (2017).
Eckerson, J. M. et al. Acute ingestion of sugar-free red bull energy drink has no effect on upper body strength and muscular endurance in resistance trained men. J. Strength Cond. Res. 1. https://doi.org/10.1519/JSC.0b013e31827e14f2 (2012).
Seidl, R., Peyrl, A., Nicham, R. & Hauser, E. A taurine and caffeine-containing drink stimulates cognitive performance and well-being. Amino Acids 19, 635–642 (2000).
Jeffries, O., Hill, J., Patterson, S. D. & Waldron, M. Energy drink doses of caffeine and taurine have a null or negative effect on sprint performance. J. Strength Cond. Res. 34, 3475–3481 (2020).
Fan, Y., Zhang, B., Wang, Y. & Wu, H. Different humidity environments do not affect the subsequent exercise ability of college football players after aerobic high-intensity interval training. Sci. Rep. 14, 16205 (2024).
Waldron, M., Patterson, S. D. & Jeffries, O. Oral taurine improves critical power and severe-intensity exercise tolerance. Amino Acids 51, 1433–1441 (2019).
Faul, F., Erdfelder, E., Buchner, A. & Lang, A.-G. Statistical power analyses using G*Power 3.1: Tests for correlation and regression analyses. Behav. Res. Methods 41, 1149–1160 (2009).
Sökmen, B. et al. Caffeine use in sports: Considerations for the athlete. J. Strength Cond. Res. 22, 978–986 (2008).
Alba-Jiménez, C., Moreno-Doutres, D. & Peña, J. Trends assessing neuromuscular fatigue in team sports: A narrative review. Sports 10, 33 (2022).
Claudino, J. G. et al. The countermovement jump to monitor neuromuscular status: A meta-analysis. J. Sci. Med. Sport 20, 397–402 (2017).
Scarpina, F. & Tagini, S. The stroop color and word test. Front. Psychol. 8 (2017).
Wang, C., Zhu, Y., Dong, C., Zhou, Z. & Zheng, X. Effects of various doses of caffeine ingestion on intermittent exercise performance and cognition. Brain Sci. 10, 595 (2020).
Baltazar-Martins, J. G., Brito De Souza, D., Aguilar, M., Grgic, J. & Del Coso, J. Infographic. The road to the ergogenic effect of caffeine on exercise performance. Br. J. Sports Med. 54, 618–619 (2020).
Ward, R., Bridge, C. A., McNaughton, L. R. & Sparks, S. A. The effect of acute taurine ingestion on 4-km time trial performance in trained cyclists. Amino Acids 48, 2581–2587 (2016).
Ballmann, C. et al. Exercise-induced oxidative stress and hypoxic exercise recovery. Eur. J. Appl. Physiol. 114, 725–733 (2014).
Jiménez, S. L., Díaz-Lara, J., Pareja-Galeano, H. & Del Coso, J. Caffeinated drinks and physical performance in sport: A systematic review. Nutrients 13, 2944 (2021).
Imagawa, T. F. et al. Caffeine and taurine enhance endurance performance. Int. J. Sports Med. 30, 485–488 (2009).
Takahashi, Y. & Hatta, H. Effects of taurine administration on exercise-induced fatigue and recovery. JPFSM 6, 33–39 (2017).
Dutka, T. L., Lamboley, C. R., Murphy, R. M. & Lamb, G. D. Acute effects of taurine on sarcoplasmic reticulum Ca2+ accumulation and contractility in human type I and type II skeletal muscle fibers. J. Appl. Physiol. 117, 797–805 (2014).
Tangen, D. S., Nielsen, S. R., Kolnes, K. J. & Jensen, J. Caffeine increases vertical jumping height in young trained males before but not after a maximal effort strength training session. J. Sci. Sport Exerc. 2, 145–153 (2020).
Domaszewski, P., Pakosz, P., Konieczny, M., Bączkowicz, D. & Sadowska-Krępa, E. Caffeine-induced effects on human skeletal muscle contraction time and maximal displacement measured by tensiomyography. Nutrients 13, 815 (2021).
Kowsari, E., Moosavi, Z. A., Rahimi, A., Faramarzi, M. & Haghighi, M. M. The effect of short-term taurine amino acid supplement on neuromuscular fatigue, serum lactate level and choice reaction time after maximal athletic performance. J. Res. Med. Dent. Sci. 6 (2018).
McLeay, Y., Stannard, S. & Barnes, M. The effect of taurine on the recovery from eccentric exercise-induced muscle damage in males. Antioxidants 6, 79 (2017).
Hamilton, E. J., Berg, H. M., Easton, C. J. & Bakker, A. J. The effect of taurine depletion on the contractile properties and fatigue in fast-twitch skeletal muscle of the mouse. Amino Acids 31, 273–278 (2006).
Souissi, Y., Souissi, M. & Chtourou, H. Effects of caffeine ingestion on the diurnal variation of cognitive and repeated high-intensity performances. Pharmacol. Biochem. Behav. 177, 69–74 (2019).
Dixit, A., Goyal, A., Thawani, R. & Vaney, N. Effect of caffeine on information processing: Evidence from stroop task. Indian J. Psychol. Med. 34, 218–222 (2012).
Desbrow, B. et al. The effects of different doses of caffeine on endurance cycling time trial performance. J. Sports Sci. 30, 115–120 (2012).
Schaffer, S. W. et al. Effect of taurine and potential interactions with caffeine on cardiovascular function. Amino Acids 46, 1147–1157 (2014).
Author information
Authors and Affiliations
Contributions
W.C.R. conceived and supervised the study. J.L. and W.C.R. carried out the experiments. J.L. and W.C.R. wrote and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, J., Rong, W. Effects of taurine combined with caffeine on repetitive sprint exercise performance and cognition in a hypoxic environment. Sci Rep 15, 5386 (2025). https://doi.org/10.1038/s41598-025-89680-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-89680-z