Abstract
Dengue virus (DENV) is a major health concern throughout the world infecting up to 390 million people globally each year. Infection with any one of the four DENV serotypes produces a spectrum of clinical illness ranging from a mild undifferentiated febrile disease through to severe dengue involving fever and haemorrhage. There is currently no antiviral treatment for dengue and only one licensed vaccine with limited distribution. This study characterises the kinetics of the serological dengue biomarker, NS1, and the appearance of anti-NS1 IgG, anti-E IgM and anti-E IgG responses in patients with primary and secondary infections. Blood samples were collected daily from a cohort of 52 Vietnamese patients during the acute phase of disease. NS1 was detected in 85% of patient samples from disease onset with detection decreasing throughout the acute phase of disease. Anti-NS1 IgG detected from the fourth day of illness and anti-E IgM and IgG from the third day of illness, were all observed to increase throughout the course of the disease. During secondary infection, NS1 levels rapidly decrease with the increasing levels of anti-NS1 IgG, suggesting the possibility of NS1 immune complex formation and a potential role in the pathogenesis of the severe forms of disease associated with secondary infections.
Similar content being viewed by others
Introduction
Dengue virus (DENV) is a major public health concern throughout the tropical regions of the world, infecting up to 390 million people each year1. DENV is a single stranded RNA virus belonging to the genus Flavivirus of the family Flaviviridae and is spread primarily by mosquitoes to humans by a bite of the domestic mosquito A. aegypti. Infection with any of the four DENV serotypes (DENV1–4) can produce a spectrum of clinical disease, ranging from a mild undifferentiated febrile illness through to the life-threatening forms of disease associated with secondary infection, characterized by increased vascular permeability, thrombocytopenia and hemorrhage2,3.
There is currently no antiviral therapy for dengue infection. Treatment is purely supportive, such as fluid replacement for patients. During the natural course of infection, an immune response is generated against viral antigens such as the DENV non-structural protein, NS1 which is often observed to be at elevated levels in patient plasma during the early stages of disease, particularly in secondary infection4. As a result of these observations, NS1 has been proposed as a surrogate marker for viremia and a predictor of progression to severe disease in secondary infected patients5. Furthermore, studies have identified NS1 as a viral toxin that contributes to an overactive innate immune response and endothelial cell dysfunction early in infection. The presence of NS1 in the serum of infected patients has been exploited as a useful early diagnostic biomarker6,7,8.
Primary dengue infection is characterized by rising levels of IgM appearing at day 3–5 and persisting for up to a period of months. Patients undergoing a secondary infection typically experience an anamnestic IgG antibody response with peak detection 6–9 days after the onset of symptoms. The DENV NS1 protein is secreted from infected cells during the natural course of infection with elevated levels of circulating secreted NS1 in patient plasma often observed during the early stages of disease9,10.
The kinetics of these dengue biomarkers have been intensively researched, and many studies have investigated various combinations of biomarkers in either primary, secondary infections or both. Several key studies by Papa et al. and Koraka et al. characterized NS1 and anti-NS111,12, while the study by Hu et al. determined the kinetics of NS1, anti-dengue E IgM and IgG13. While these studies provide solid evidence for the kinetics of the appearance of these biomarkers, the combined story has to be inferred by outbreaks from different geographic locations, outbreaks, dengue serotypes and times using various methods. However, no single prospective study has been performed to characterize the kinetics of NS1, anti-NS1, anti-E IgM, and anti-E IgG in a single patient cohort. In the study presented in this manuscript, we investigated the kinetics of these biomarkers in a single prospective study of primary and secondary infected patients from a single dengue outbreak occurring in Vietnam where dengue virus types 1 and 2 were circulating. Here we characterise the kinetics of serological dengue biomarkers during the acute phase of disease. Levels of free NS1 circulating during the course of infection in patient sera were determined by quantitative capture ELISA. Titres were determined for anti-NS1 antibodies circulating in sera by indirect ELISA. Anti-E IgM and IgG levels were assessed by capture IgM and IgG ELISA, respectively9.
Results
Analysis of NS1 protein and anti-NS1 IgG kinetics in infected patients
In primary patients, NS1 could be detected in blood from the onset of illness through to 9 days post infection, with peak NS1 levels occurring between day 1–4. In secondary patients, NS1 could be detected from day 1 to 8 post onset of illness. However, lower NS1 levels were observed later in infection when compared to primary patients with NS1 peaking on days 3 and 4. NS1 levels ranged from undetectable to 7.8 µg/ml. Patients with DENV-1 had higher levels of NS1 during primary infection, than those patients with primary DENV-2 infection whose NS1 levels were consistently low (Fig. 1a).
Daily patient samples tested for NS1 and anti-NS1 levels. (a) Primary patient NS1 levels and (b) Secondary patient NS1 levels during the acute phase of illness. (c) Primary patient anti-NS1 IgG responses. (d) Secondary patient anti-NS1tested by indirect ELISA and plotted as 50% titres. Anti-NS1 IgG titre plotted against NS1 levels in (e) primary and (f) secondary patients. Blue circles: DENV-1 DF (Dengue Fever) patient, Red circles: DENV-2 DF patient, Blue squares: DENV-1 DHF (Dengue Haemorrhagic Fever) patient, Red squares: DENV-2 DHF patient, LoD limit of detection.
NS1 was successfully detected throughout the acute phase of the disease with detection above 80% for the first four days of illness in primary patients. However, detection was lower in secondary infections, ranging from 50 to 63% during the first 4 days of infection. NS1 was detected in 100% of primary DENV-1 patients during the first 3 days of illness and above 80% through the rest of the acute period. However, the results for primary DENV-2 patients revealed lower detection rates ranging from 50 to 67% during the first 4 days of illness. During DENV-1 secondary infection, NS1 was detected from day 1 for only one patient, with detection decreasing through to day 8 where no NS1 was detected. DENV-2 secondary infection patients had no detectable NS1 on day 1 (n = 1), with detection rising to 83% on day 2 and peaking at 85% detection on day 3 before steadily decreasing throughout infection until NS1 was undetectable by day 8 (Fig. 1b).
Primary DENV patients had poor early anti-NS1 IgG responses, as expected for an initial antigen exposure with NS1 specific IgG only detected in one patient on day 4 during acute illness and at 8% detection in the follow-up samples (Fig. 1c). In contrast, and as expected of an anamnestic response, secondary patients showed a rapid anti-NS1 IgG response. Anti-NS1 IgG was detected as early as 4 days post onset of illness (Fig. 1d). Anti-NS1 IgG titers and detection increased during infection, with 88% of secondary patients having detectable anti-NS1 IgG levels in the follow-up samples. Primary patients had high levels of free circulating NS1 while having undetectable anti-NS1 IgG titers, whereas secondary infected patients had decreasing levels of NS1 at the same time that anti-NS1 IgG levels were rising (Fig. 1e). The appearance of anti-NS1 IgG was inversely correlated (Pearsons two tailed t test, 95% CI) with NS1 levels in secondary patients, suggesting the formation of NS1/IgG immune complexes or IgG-mediated NS1 clearance from circulation (Fig. 1f).
Appearance of anti-E IgM and IgG in patient serum samples
The virion surface E protein is a major target of an infected patient’s immune response. The presence of anti-E IgM and IgG was determined using IgM and IgG specific capture ELISAs (Fig. 2). The ELISAs were performed as per the manufacturer’s instructions. None of the samples from the 3 negative patients tested positive for anti-E IgM or IgG antibodies. IgM was detected from the onset of symptoms with detection levels increasing throughout infection, reaching 100% of patients by 7 days post onset of illness (Fig. 2a). In secondary infected patients, IgM was detected from the third day post onset of symptoms (26.67%) with detection rapidly rising on day 4 (78.95%). Detection of IgM in secondary patients was maintained at ≥ 80% for the remainder of the study period. The anti-E IgM response in DENV-2 patients was delayed by 2 days compared to DENV-1 primary infected patients, however in secondary infection the anti-E IgM kinetics were similar for both DENV-1 and 2 infected patients (Fig. 2b).
Anti-dengue IgM and IgG responses. (a) Daily primary patient anti-E IgM responses. (b) Daily secondary patient anti-E IgM responses. (c) Daily primary patient anti-E IgG responses. (d) Daily secondary patient anti-E IgG responses. Blue circles: DENV-1 DF patient, Red circles: DENV-2 DF patient, Blue squares: DENV-1 DHF patient, Red squares: DENV-2 DHF patient. Limit of detection (LoD) set at 12 and 23.4 Panbio units for IgM and IgG respectively.
As expected, anti-E IgG detection in primary patients was low in comparison with IgM with a positive response detected in only 3 patients during the acute disease phase. Anti-E IgG was detected on days 6 and 7 in 19% and 17% of patients, respectively. Higher rates of detection were observed in the follow-up samples with IgG detection in 44% of patients (Fig. 2c). In secondary infected patients, anti-E IgG was detected from day 3 in 6.67% of patients, with detection rapidly rising to ≥ 87% from day 5–10 (Fig. 2d). Follow-up samples (day 16–45) showed that all patients mounted a detectable anti-E IgG response. In summary, primary patients infected with either DENV-1 or DENV-2 had similarly low levels of anti-E IgG responses during the acute phase of disease. In contrast, secondary responses to each DENV serotype had higher detection rates with increasing detection from day 4, reflecting a strong anamnestic antibody response to cross-reactive epitopes.
Discussion
In this prospective study, we examined the kinetics of appearance of NS1, anti-NS1 IgG, anti-E IgM and IgG in the serum of 51 patients infected with dengue during epidemics in Vietnam from 2005 to 2007. Patients enrolled in this study either experienced DENV-1 or DENV-2 infections (both primary and secondary), with levels of disease outcome varying from mild infection to severe disease with or without vascular leak. Samples were collected daily during the acute disease period then at convalescence.
NS1 was detected in both primary and secondary DENV patients during the acute phase of disease from the first day of illness. In primary infections detection was retained through to at least 9 days post illness, while detection in secondary cases was found to decline by days 6–7. Peak detection for both primary and secondary patients was on days 3–414,15,16. NS1 was detected in ≥ 72% of samples during the first 4 days of illness ranging from undetectable to 7.8 µg/ml. By day 5, detection dropped to 50%, then decreased throughout the remainder of the disease period. These results are consistent with the findings of others who have used in-house and commercial NS1 capture ELISAs to detect NS117,18,19,20,21,22,23,24,25,26,27,28. As with the current analysis, these studies found that NS1 capture ELISAs had high sensitivity and specificity early during the acute period with detection decreasing during infection.
Primary DENV-2 patients had lower NS1 levels overall with 133 ng/ml the highest NS1 level recorded, as opposed to DENV-1 primary patients which peaked at 7.8 µg/ml. The lower levels of NS1 associated with DENV-2 infection has also been observed in other studies in Vietnam29. While the use of dengue 2 virus NS1 as the sole standard for NS1 quantification for all samples likely impacts absolute values, the trends in relative levels of NS1 within individual patients, the main focus of this study, will not be impacted. Interestingly, secondary DENV-2 patients showed comparable levels of NS1 to secondary DENV-1 patients, with peak levels detected at 5.5 µg/ml. The lower levels of NS1 in primary DENV-2 patients may simply reflect an overall lower viral load14. The higher relative levels of NS1 in secondary DENV-2 infected patients may be a consequence of antibody-dependent enhanced infection, with these higher levels shown to be a predictive marker of progression to more severe disease4.
In a clinical setting, timing and choice of what diagnostic test to employ is critical, as each method described in this study has an ideal window in which it is at its most effective. The NS1 capture ELISA is at its most effective early in infection, i.e. from the onset of symptoms, whereas IgM and IgG ELISAs rely on the patients induced immune response, which in most cases appears from day 3 or 430,31. By the time IgM ELISAs are performing at a high diagnostic rate, NS1 detection may be rapidly declining, particularly in those patients experiencing a secondary infection. This highlights the need for complementary testing of both NS1 and antibody levels, as this testing regime was found to cover the disease period with greater than 80% successful diagnosis (in this study day 2 was an exception with 73% detection)32,33. Performing concurrent tests on patient sera for NS1 capture as well as anti-E IgM and IgG capture would allow additional determination of primary versus secondary infection30.
Anti-NS1 IgG 50% titers were determined by indirect ELISA, with both detection and titers varying in correlation with the infection status of the patient. Previous studies have suggested that NS1 specific antibodies may play a role in the development of severe disease and vascular leak, as anti-NS1 antibodies are seldom seen in primary infection but detected in most secondary infected patients32,34,35,36,37. All primary infected patient titers were below the negative sample cut-off values, with the exception of a small number of patients (n = 3). In secondary infected patients, specific anti-NS1 IgG was detected from day 4, with titers and sensitivity of detection rising throughout the course of the infection to ≥ 81% detection in convalescent sera. All secondary infected patients with positive anti-NS1 IgG had no or rapidly decreasing NS1 levels. It was observed in most patients that decreasing levels of NS1 correlated with rising levels of anti-NS1 IgG. In other patients with no NS1 detected, high levels of anti-NS1 IgG were present. The logical interpretation of these observations is the formation of immune complexes, which render the bound NS1 unable to be detected in the capture ELISA.
One limitation of the NS1 capture ELISA employed in this study is that detection is limited to free-circulating NS1. In this study and others, it has been noted that the ability to detect NS1 diminishes from day 4–5. It has been hypothesized that this loss of NS1 detection in secondary infection is due to the presence of NS1 specific IgG removing NS1 from circulation and leading to the formation of immune complexes. These immune complexes are then presumed to be cleared from the circulatory system or deposited onto the surface of endothelial cells possibly contributing to vascular leak. In this study we have confirmed an inverse correlation between decreasing NS1 levels and increasing anti-NS1 IgG over the course of infection within individual patients. Note added in proof: while this manuscript was under review, our findings were confirmed by similar observations reported by Papa et al.12.
Speculation on the role of antibodies directed against NS1 has, at times provided a somewhat confusing and contradictory narrative. Historically, NS1 has been viewed as a potential vaccine candidate that would give rise to a protective immune response that does not have the same intrinsic ADE risks associated with E-based vaccines10. Our group identified a role for an anti-dengue NS1 monoclonal antibody in protection of mice from dengue virus or Zika virus challenge38 and there is a wealth of literature demonstrating that NS1 vaccination elicits protective antibodies by targeting dengue-infected cells for destruction via complement-mediated lysis and other mechanisms39,40,41,42,43,44,45. However, NS1 vaccination has also been shown to produce inappropriate immune responses with antibodies that are host reactive via self-mimicry apparently enhancing disease46,47,48,49,50. Furthermore, the protective efficacy of the anti-NS1 response needs to be balanced against its role in pathogenesis. As demonstrated in our manuscript and others, NS1 is rapidly cleared from circulation during a secondary infection by anti-NS1 IgG. The loss of free circulating NS1 through the formation of immune complexes aligns with the onset of severe dengue disease. We speculate that the formation of immune complexes and deposition onto endothelial cell walls rather than clearance, likely plays a role in the vascular leak characteristic of severe dengue. Based on our observations, this hypothesis is limited by the relatively small number of patients experiencing secondary dengue and severe disease. Therefore, the sample size needs to be expanded in future studies to conclusively determine the role that immune complexes may be playing in severe disease.
While the kinetics of dengue biomarker appearance has been intensively investigated, to our knowledge, this is the first prospective study to examine the kinetics of NS1, anti-NS1, anti-E IgM and IgG in a single dengue patient cohort. Overall, our findings confirm what has otherwise been inferred by numerous previous studies reporting the kinetics of dengue biomarkers in various patient cohorts. The identification of a viral toxin activity for NS1 that impacts vascular integrity of the endothelial barrier 3 days post intravenous injection in mouse studies further highlights the importance of early NS1 detection6,7. Taken together, these results support the suggestion that both soluble NS1 and its antibody bound immune complex may be contributing factors in the pathogenesis of severe disease.
Materials and methods
Clinical samples and study objectives
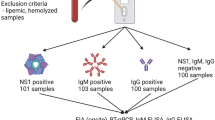
We selected 303 serum samples from 52 dengue patients (confirmed positive by qPCR) enrolled in research studies at the Hospital for Tropical Diseases in Ho Chi Minh City, Vietnam (reviewed by the ethics committee and approved by the internal review board of the hospital), during outbreaks that occurred between 2005 and 2007. Daily serum samples were taken from patients on presentation to healthcare facilities during the course of disease development (1–9 days) and convalescent serum samples were taken several weeks after illness resolution. The patient cohort contained primary and secondary patients experiencing DENV-1 and 2 infections, with symptoms ranging from mild dengue fever through to severe dengue involving haemorrhage and shock (Table 1). Patients were classified as having a primary vs. secondary infection based on RT-qPCR results coupled with IgM and IgG analysis. A negative IgG ELISA result in the first 72 h with no increase in dengue-reactive IgG by day 7 defined a primary infection, and a positive indirect ELISA result within 72 h and/or an equivocal response, with a clear increase in dengue-reactive IgG greater than the IgM response by day 7, defined a secondary infection. This study examines the kinetics of NS1 production, as well as the appearance of anti-NS1 IgG, anti-dengue IgM and anti-dengue IgG in patients with primary and secondary infections.
Study design
Diagnostic ELISAs: NS1, anti E IgM and anti-E IgG
Evaluation of NS1, anti-dengue IgM and anti-dengue IgG levels were assessed using the Panbio Early dengue ELISA, Panbio Dengue IgM capture ELISA and Panbio IgG capture ELISA IgG kits as per manufacturer’s instructions (Abbott). Quantitation of NS1 in patient samples was made possible by the addition of a baculovirus expressed recombinant dengue virus 2 NS1 protein standard curve from which quantitative values were determined using GraphPad Prism. The authors note that the use of a single serotype NS1 will likely impact absolute protein values attributed to other serotypes. However the findings of this study relate to the relative rise and fall in levels of circulating NS1 within patients.
Indirect anti-NS1 IgG
Nunc maxisorp plates were coated with 50 µl of 100 ng/ml of NS1 in carbonate buffer pH 9.6 and incubated at 4 °C overnight. After coating, the plates were blocked with 150 µl of a solution of 1% milk powder, 1% sucrose, 0.3% Tween 20 in PBS at room temperature for 2 h. Following blocking, plates were washed and 50 µl of 1 in 10 dilutions of patient plasma samples were serially diluted in a 1:10 series in blocking solution and incubated for 1 h at 37 °C. After antibody binding, the plates were washed 3 times and anti-human IgG HRP at 1:100 in 0.3% Tween 20 in PBS (PBST-20) was added to the plate for 1 h at 37 °C, then washed a further 6 times. Fifty µl of K-Blue TMB substrate (ELISA systems) was added, and the colour reaction was developed for 5 min in the dark. The reaction was stopped by the addition of 25 µl of 2 M H2SO4, with plates read spectrophotometrically at 450 nm.
Data availability
The authors declare that all data supporting the findings of this study are available within the paper.
References
Bhatt, S. et al. The global distribution and burden of dengue. Nature 496 (7446), 504–507 (2013).
Halstead, S. B. Pathogenesis of dengue: challenges to molecular biology. Science 239 (4839), 476–481 (1988).
Guzmán, M. G. & Kourí, G. Dengue: an update. Lancet Infect. Dis. 2 (1), 33–42 (2002).
Libraty, D. H. et al. High Circulating levels of the dengue virus nonstructural protein NS1 early in dengue illness correlate with the development of dengue hemorrhagic fever. J. Infect. Dis. 186 (8), 1165–1168 (2002).
Alcon, S. et al. Enzyme-linked immunosorbent assay specific to dengue virus type 1 nonstructural protein NS1 reveals circulation of the antigen in the blood during the acute phase of disease in patients experiencing primary or secondary infections. J. Clin. Microbiol. 40 (2), 376–381 (2002).
Beatty, P. R. et al. Dengue virus NS1 triggers endothelial permeability and vascular leak that is prevented by NS1 vaccination. Sci. Transl Med. 7 (304), 304ra141 (2015).
Modhiran, N. et al. Dengue virus NS1 protein activates cells via Toll-like receptor 4 and disrupts endothelial cell monolayer integrity. Sci. Transl Med. 7 (304), 304ra142–304ra142 (2015).
Watterson, D., Modhiran, N. & Young, P. R. The many faces of the flavivirus NS1 protein offer a multitude of options for inhibitor design. Antiviral Res. 130, 7–18 (2016).
Young, P. R. et al. An antigen capture enzyme-linked immunosorbent assay reveals high levels of the dengue virus protein NS1 in the Sera of infected patients. J. Clin. Microbiol. 38 (3), 1053–1057 (2000).
Muller, D. A. & Young, P. R. The flavivirus NS1 protein: molecular and structural biology, immunology, role in pathogenesis and application as a diagnostic biomarker. Antiviral Res. 98 (2), 192–208 (2013).
Koraka, P. et al. Detection of immune-complex-dissociated nonstructural-1 antigen in patients with acute dengue virus infections. J. Clin. Microbiol. 41 (9), 4154–4159 (2003).
Premazzi Papa, M. et al. Dengue NS1 antibodies are associated with clearance of viral nonstructural protein-1. J. Infect. Dis. 230 (6), e1226–e1234 (2024).
Hu, D. et al. Kinetics of non-structural protein 1, IgM and IgG antibodies in dengue type 1 primary infection. Virol. J. 8, 47 (2011).
Duyen, H. T. L. et al. Kinetics of plasma viremia and soluble nonstructural protein 1 concentrations in dengue: differential effects according to serotype and immune status. J. Infect. Dis. 203 (9), 1292–1300 (2011).
Nainggolan, L. et al. Association of viral kinetics, infection history, NS1 protein with plasma leakage among Indonesian dengue infected patients. PLoS One 18 (5), e0285087 (2023).
Tricou, V. et al. Kinetics of viremia and NS1 antigenemia are shaped by immune status and virus serotype in adults with dengue. PLoS Negl. Trop. Dis. 5 (9), e1309–e1309 (2011).
Blacksell, S. D. et al. Evaluation of the Panbio dengue virus nonstructural 1 antigen detection and Immunoglobulin M antibody enzyme-linked immunosorbent assays for the diagnosis of acute dengue infections in Laos. Diagn. Microbiol. Infect. Dis. 60 (1), 43–49 (2008).
Bessoff, K. et al. Comparison of two commercially available dengue virus (DENV) NS1 capture enzyme-linked immunosorbent assays using a single clinical sample for diagnosis of acute DENV infection. Clin. Vaccine Immunol. 15 (10), 1513–1518 (2008).
Chaiyaratana, W. et al. Evaluation of dengue nonstructural protein 1 antigen strip for the rapid diagnosis of patients with dengue infection. Diagn. Microbiol. Infect. Dis. 64 (1), 83–84 (2009).
Kumarasamy, V. et al. Evaluation of a commercial dengue NS1 antigen-capture ELISA for laboratory diagnosis of acute dengue virus infection. J. Virol. Methods. 140 (1–2), 75–79 (2007).
Kumarasamy, V. et al. Evaluating the sensitivity of a commercial dengue NS1 antigen-capture ELISA for early diagnosis of acute dengue virus infection. Singap. Med. J. 48 (7), 669–673 (2007).
Dussart, P. et al. Evaluation of two new commercial tests for the diagnosis of acute dengue virus infection using NS1 antigen detection in human serum. PLoS Negl. Trop. Dis. 2 (8), e280 (2008).
Dussart, P. et al. Evaluation of an enzyme immunoassay for detection of dengue virus NS1 antigen in human serum. Clin. Vaccine Immunol. 13 (11), 1185–1189 (2006).
Zainah, S. et al. Performance of a commercial rapid dengue NS1 antigen immunochromatography test with reference to dengue NS1 antigen-capture ELISA. J. Virol. Methods. 155 (2), 157–160 (2009).
Osorio, L. et al. Comparison of the diagnostic accuracy of commercial NS1-based diagnostic tests for early dengue infection. Virol. J. 7 (1), 361 (2010).
Chong, Z. L. et al. Diagnostic accuracy and utility of three dengue diagnostic tests for the diagnosis of acute dengue infection in Malaysia. BMC Infect. Dis. 20 (1), 210 (2020).
Kikuti, M. et al. Accuracy of the SD BIOLINE dengue duo for rapid point-of-care diagnosis of dengue. PLoS One 14 (3), e0213301 (2019).
Santoso, M. S. et al. Diagnostic accuracy of 5 different brands of dengue virus non-structural protein 1 (NS1) antigen rapid diagnostic tests (RDT) in Indonesia. Diagn. Microbiol. Infect. Dis. 98 (2), 115116 (2020).
Chau, T. N. et al. Clinical and virological features of dengue in Vietnamese infants. PLoS Negl. Trop. Dis. 4 (4), e657 (2010).
Muller, D. A., Depelsenaire, A. C. I. & Young, P. R. Clinical and laboratory diagnosis of dengue virus infection. J. Infect. Dis. 215 (suppl_2), S89–S95 (2017).
Pérez-Rodríguez, F. J. et al. Direct dengue virus genome sequencing from antigen NS1 rapid diagnostic tests: a proof-of-concept with the standard Q dengue duo assay. Viruses 15 (11) (2023).
Shu, P. Y. et al. Dengue virus serotyping based on envelope and membrane and nonstructural protein NS1 serotype-specific capture Immunoglobulin M enzyme-linked immunosorbent assays. J. Clin. Microbiol. 42 (6), 2489–2494 (2004).
Kit Lam, S. et al. Evaluation of a capture screening enzyme-linked immunosorbent assay for combined determination of Immunoglobulin M and G antibodies produced during dengue infection. Clin. Diagn. Lab. Immunol. 7 (5), 850–852 (2000).
Falkler, W. A. Jr., Diwan, A. R. & Halstead, S. B. Human antibody to dengue soluble complement-fixing (SCF) antigens. J. Immunol. 111 (6), 1804–1809 (1973).
Huang, J. L. et al. High-level expression of recombinant dengue viral NS-1 protein and its potential use as a diagnostic antigen. J. Med. Virol. 65 (3), 553–560 (2001).
Shu, P. Y. et al. Comparison of capture immunoglobulin M (IgM) and IgG enzyme-linked immunosorbent assay (ELISA) and nonstructural protein NS1 serotype-specific IgG ELISA for differentiation of primary and secondary dengue virus infections. Clin. Diagn. Lab. Immunol. 10 (4), 622–630 (2003).
Shu, P. Y. et al. Dengue NS1-specific antibody responses: isotype distribution and serotyping in patients with dengue fever and dengue hemorrhagic fever. J. Med. Virol. 62 (2), 224–232 (2000).
Modhiran, N. et al. A broadly protective antibody that targets the flavivirus NS1 protein. Science 371 (6525), 190–194 (2021).
Chung, K. M. et al. West nile virus nonstructural protein NS1 inhibits complement activation by binding the regulatory protein factor H. Proc. Natl. Acad. Sci. USA. 103 (50), 19111–19116 (2006).
Diamond, M. S., Pierson, T. C. & Fremont, D. H. The structural immunology of antibody protection against West nile virus. Immunol. Rev. 225, 212–225 (2008).
Schlesinger, J. J., Brandriss, M. W. & Walsh, E. E. Protection against 17D yellow fever encephalitis in mice by passive transfer of monoclonal antibodies to the nonstructural glycoprotein gp48 and by active immunization with gp48. J. Immunol. 135 (4), 2805–2809 (1985).
Schlesinger, J. J., Brandriss, M. W. & Walsh, E. E. Protection of mice against dengue 2 virus encephalitis by immunization with the dengue 2 virus non-structural glycoprotein NS1. J. Gen. Virol. 68 (Pt 3), 853–857 (1987).
Falgout, B. et al. Immunization of mice with Recombinant vaccinia virus expressing authentic dengue virus nonstructural protein NS1 protects against lethal dengue virus encephalitis. J. Virol. 64 (9), 4356–4363 (1990).
Avirutnan, P. et al. Antagonism of the complement component C4 by flavivirus nonstructural protein NS1. J. Exp. Med. 207 (4), 793–806 (2010).
Avirutnan, P. et al. Binding of flavivirus nonstructural protein NS1 to C4b binding protein modulates complement activation. J. Immunol. 187 (1), 424–433 (2011).
Falconar, A. K. Antibody responses are generated to immunodominant ELK/KLE-type motifs on the nonstructural-1 glycoprotein during live dengue virus infections in mice and humans: implications for diagnosis, pathogenesis, and vaccine design. Clin. Vaccine Immunol. 14 (5), 493–504 (2007).
Falconar, A. K. Monoclonal antibodies that bind to common epitopes on the dengue virus type 2 nonstructural-1 and envelope glycoproteins display weak neutralizing activity and differentiated responses to virulent strains: implications for pathogenesis and vaccines. Clin. Vaccine Immunol. 15 (3), 549–561 (2008).
Lin, C. F. et al. Expression of cytokine, chemokine, and adhesion molecules during endothelial cell activation induced by antibodies against dengue virus nonstructural protein 1. J. Immunol. 174 (1), 395–403 (2005).
Lin, C. F. et al. Endothelial cell apoptosis induced by antibodies against dengue virus nonstructural protein 1 via production of nitric oxide. J. Immunol. 169 (2), 657–664 (2002).
Lin, C. F. et al. Liver injury caused by antibodies against dengue virus nonstructural protein 1 in a murine model. Lab. Investig. 88 (10), 1079–1089 (2008).
Funding
This research was funded by Australian Research Council Linkage grant, (LP0668437).
Author information
Authors and Affiliations
Contributions
Conceptualization, D.A.M. and P.R.Y.; methodology, D.A.M. and P.R.Y.; investigation, C.M., H.T.L.D.; formal analysis, D.A.M.; writing—original draft preparation, D.A.M.; writing—review and editing, D.A.M., P.R.Y., C.M., J.J.Y.C., and B.W.; visualization, D.A.M.; funding acquisition, D.A.M. and P.R.Y. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Institutional review board statement
The study was conducted in accordance with the Declaration of Helsinki, and the use of serum samples routinely taken from patients as part of ongoing research projects was approved by the Ethics Committee of the Hospital for Tropical Diseases, Ho Chi Minh City, Vietnam.
Informed consent statement
Informed consent was obtained from all subjects involved in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Muller, D.A., Choo, J.J.Y., McElnea, C. et al. Kinetics of NS1 and anti-NS1 IgG following dengue infection reveals likely early formation of immune complexes in secondary infected patients. Sci Rep 15, 6684 (2025). https://doi.org/10.1038/s41598-025-91099-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-91099-5
This article is cited by
-
Dengue Fever Diagnosis: Performance Characteristics of Current Tests and Emerging Detection Technologies
Molecular Diagnosis & Therapy (2026)
-
Dengue virus infection and Guillain–Barré syndrome: a systematic review of clinical characteristics, outcomes, and predictors of severity
BMC Infectious Diseases (2025)