Abstract
Cricothyroidotomy is a key technique for securing airways when tracheal intubation and oxygenation are difficult to achieve. In recent years, it has become clear that ultrasonography is more accurate than palpation for identifying the cricothyroid membrane. However, it is unclear how ultrasound should be administered. We investigated whether real-time or static ultrasound techniques were more useful for needle cricothyrotomy in patients with simulated anatomical abnormalities of the neck. Three cervical simulators with anatomical abnormalities were created. After education and hands-on training in needle cricothyroidotomy, 48 participants (27 junior residents, 12 anesthesia residents, and nine anesthesiologists) performed needle cricothyroidotomy using either a real-time or static ultrasound technique (crossover study). Fisher’s exact test was used to assess the success rate and accuracy of the puncture (p < 0.05 was considered statistically significant). The success rate of cricothyrotomy puncture was significantly higher with the real-time ultrasound technique than with the static ultrasound technique. The real-time ultrasound technique led to significantly higher puncture accuracy and shorter procedure time than the static ultrasound technique. Needle cricothyroidotomy using real-time ultrasonography may be more useful than that using static ultrasonography.
Similar content being viewed by others
Introduction
Cricothyroidotomy is the last resort in a situation of “cannot intubate, cannot oxygenate (CICO).” However, this procedure is not easy for anesthesiologists. A survey conducted in the United Kingdom (The Fourth National Airway Project: NAP4) reported that the success rate of emergent needle cricothyroidotomy performed by anesthesiologists was less than 50%, whereas surgical cricothyroidotomy performed by surgeons was almost universally successful1. Therefore, surgical cricothyroidotomy is recommended over needle cricothyroidotomy by many guidelines in CICO situations2,3,4,5. However, surgical cricothyroidotomy may be associated with a risk of bleeding from the arteries (superior thyroid or cricothyroid arteries) around the cricothyroid membrane (CTM). Percutaneous needle cricothyroidotomy may be beneficial in preventing complications associated with heavy bleeding if the CTM can be accurately punctured.
Accurate identification of the CTM and adequate skills for needle handling are essential for successful percutaneous needle cricothyroidotomy. Many studies have shown that ultrasound examination is more accurate than the digital palpation technique to identify CTM6,7. However, it remains unclear whether accurate identification of the CTM leads to successful needle puncture. In particular, patients with CICO often have obesity or concomitant anatomical abnormalities of the neck (malposition, deviation, and rotation of the trachea), and it is doubtful whether accurate CTM identification will lead to accurate CTM puncture in such patients.
Ultrasound-guided needle puncture can be classified into two types: static and real-time (or dynamic) ultrasound techniques. In the static ultrasound technique, ultrasound is used to identify the target location, and then a needle puncture is performed using this information. By contrast, all needle puncture procedures are performed under ultrasound observation in the real-time ultrasound technique. In this simulation study, we examined whether static or real-time ultrasound techniques were more useful for needle cricothyroidotomy in patients with cervical anatomical abnormalities.
Materials and methods
This study was conducted in accordance with the ethical principles that have their origins in the Declaration of Helsinki and its subsequent amendments, and was approved by the local ethics committee on (Faculty of Medicine Research Ethics Committee; approval number, 1044). This study was registered in the University Hospital Medical Information Network Center Clinical Registration System, identifcation number UMIN000032106, first trial registration on 10/04/2018.
This was a randomized, prospective, crossover simulation study. Participants were recruited from among junior residents, anesthesia residents, and anesthesiologists. Exclusion criteria were past experiments of cricothyroidotomy and refusal to participate (Fig. 1). Written informed consent was obtained from all the participants. Participants were recruited and data were collected between July 2020 and December 2022 .
Anatomically abnormal neck simulator for CTM puncture
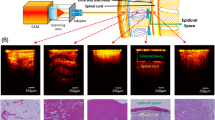
Three types of anatomically abnormal neck simulators were created for this study (Alfa Bio Co., Gumma, Japan) (Fig. 2). These simulators reproduced the anatomically abnormal necks of three clinically encountered patients in which the tracheas deviated and rotated. The simulators included the thyroid, cricoid, and tracheal cartilages, which were then filled with a thick ultrasound-transparent gel. Therefore, it is difficult to palpate the CTM from the surface but possible to observe the cartilage and trachea by ultrasound (Fig. 3). Three simulators were used to ensure that information about laryngeal deviation was not communicated to other participants (see Supplemenary Fig. S1 online).
Anatomically abnormal neck simulator. The simulator is based on computed tomography data from a patient with a thyroid tumor. The larynx is deviated 20 mm from the midline and rotated clockwise at 4°, and the skin-cricothyroid membrane distance is 25 mm. (a) Appearance of the simulator; (b) Position of the larynx and trachea inside the simulator. (c) Computed tomography image of the patient with the thyroid tumor. We created two additional simulators based on computed tomography images of the larynges from two different patients. One additional simulator was created based on the computed tomography of a patient with a neck abscess in which the larynx deviated to the left side 20 mm from the midline, rotated clockwise at 7°, and the skin-cricothyroid membrane distance was 25 mm. The other additional simulator was made based on a patient with a tumor in which the larynx deviated to the right by 25 mm, rotated clockwise at 5°, and the skin-cricothyroid membrane distance was 25 mm.
Ultrasound views of the cricothyroid membrane in the simulator. The ultrasound images of the cricothyroid membrane, thyroid cartilage, and cricoid cartilage in an abnormal neck simulator are shown in Fig. 1.
Education for needle cricothyroidotomy
The participants received a lecture to review the relevant anatomy and details of the needle cricothyroidotomy procedure based on an educational video (Cricothyroidotomy Technique, Medscape)8.
Hands-on training for ultrasound-guided CTM identification
An ultrasound machine used in this study was SonoSite Edge II with ultrasound probe HFL38xi (13-6 MHz, SonoSite, Inc., Bothell, WA, USA). For ultrasound-guided CTM identification, we used a transverse ultrasound view, called the TACA method9, in which the neck is scanned from the cephalad to the caudal and then back to the cephalad to identify the thyroid cartilage (T), airline line (A), cricoid cartilage (C), and airline. The airline indicates the CTM (Fig. 4). After the lecture on CTM identification, an ultrasound expert (HN) demonstrated the CTM using the TACA method. The participants were trained in ultrasound-guided CTM identification for 30 min. The instructor (HN) confirmed that all participants could identify the CTM using ultrasound guidance.
Ultrasound-guided identification of cricothyroid membrane. To identify the cricothyroid membrane, we used a transverse technique called TACA, named after the way the technique is performed: T and C represent the initials of the thyroid and cricoid cartilages, respectively, and A represents the air border. The thyroid cartilage was identified on the basis of its triangular roof-like appearance on ultrasonography. When the probe was moved caudally and the thyroid cartilage disappeared, a bright white line appeared at the midline. This is the cricothyroid cartilage membrane, and the white line is due to the reflection of ultrasound waves from air on the inner surface of the trachea. Similarly, the cricothyroid cartilage was recognizable and resembled a horseshoe shape. In the TACA technique, the cricothyroid cartilage-air border-cricothyroid cartilage is scanned from the cephalad side to the caudal side, and the air border (cricothyroid membrane) is scanned again to the head side.
Ultrasound-guided CTM puncture
This study compared two types of ultrasound-guided CTM puncture: static and real-time (dynamic) ultrasound techniques. Which technique was performed first was determined randomly using a sealed envelope system. The order of the CTM puncture was randomized in the three simulators using a computer-generated random number table.
In the static ultrasound technique (Fig. 5a), the participants attempted to identify the CTM using the TACA method and marked the location of the CTM on the simulator’s neck skin. After marking the CTM, it was punctured with a 23G needle (cathelin needle, length 70 mm) using a 2.5 ml syringe. In the real-time ultrasound technique (Fig. 5b), the participants identified the CTM using the TACA method and then commenced the CTM puncture using an out-of-plane approach under ultrasound guidance. The participants were able to confirm whether the CTM puncture was successful by pulling the plunger of the syringe to draw air. The participants attempted CTM puncture until they achieved a successful puncture within 180 s. The success or failure of the CTM puncture was assessed using a fiberscope set in the trachea. Success was defined as the placement of the needle tip in the trachea within 180 s after starting the CTM identification and till finishing puncture. Failure was defined as the needle tip not being placed in the trachea within the time limit of 180 s. The punctures that were deemed successful were divided into two categories: “high accuracy,” defined as a cricothyrotomy expected to be performed without complications, and “low accuracy,” defined as the needle tip placed in the trachea but the catheter is not easily inserted. The accuracy of the puncture was evaluated on a 2-point scale according to the puncture position (Fig. 6). The success rate, including puncture accuracy and procedure time, was recorded. Procedure time was defined as the time from the start of handling the ultrasound probe to the end of the CTM puncture. The examinations were set up and handled by an examiner (HW), and all data were collected and evaluated by the instructor (HN).
Static and real-time ultrasound techniques. (a) In the static ultrasound technique, the participants tried to identify the cricothyroid membrane using the TACA method and marked the location of the cricothyroid membrane on the simulator’s neck skin. The cricothyroid membrane is marked and punctured. (b) In the real-time ultrasound technique, the participants identified the cricothyroid membrane using the TACA method and then started the cricothyroid membrane puncture using an out-of-plane approach under ultrasound guidance. Participants were able to confirm whether the cricothyroid membrane puncture was successful by pulling the syringe plunger to draw air.
The accuracy of the cricothyroid membrane puncture. The accuracy of the cricothyroid membrane puncture was rated on a 2-point scale (high or low accuracy) according to the distance from the center of the cricothyroid membrane where the needle penetrated it. The transverse diameter was divided into six segments, and the anterior wall corresponding to the central 4 segments was determined by considering the laryngeal rotation. Success with high accuracy was defined as needle penetration into the anterior wall within the four segments of the larynx. Low accuracy was defined as needle penetration from a site corresponding to one division at each end (without the four segments). The ultrasound expert instructor (HN), who did not observe which ultrasound technique was used, evaluated and judged whether the puncture needle entered the trachea (whether the puncture was successful) and where the puncture needle penetrated the cricothyroid membrane. The point at which the puncture needle enters the cricothyroid membrane is compared to the evaluation chart created before the examination (taking into account the rotation of the trachea in each simulator), and if there is any doubt as to whether the accuracy is high or low, the accuracy is judged to be low.
Participants performed the puncture using the assigned technique and then attempted using the other technique (a crossover study).
Statistical analysis
Numerical values were expressed as ratios (%) or as the mean ± standard deviation for normal distributions and as the median [interquartile range] for non-normal distributions. The success rate and accuracy of the puncture were evaluated using Fisher’s exact test. The presence or absence of carryover effects was evaluated using one-way ANOVA. Statistical significance was considered at p < 0.05. All data were analyzed using GraphPad Prism ver. 7.0 (GraphPad Software Inc. Boston, USA).
Power analysis
To date, no study has compared ultrasound-guided CTM puncture techniques. Therefore, assuming a clinically significant difference, we calculated ɑ = 0.05 and estimated the sample size needed to obtain 80% power to be 40 participants.
Results
Twenty-seven junior residents, twelve anesthesia residents, and nine anesthesiologists participated in the study (Fig. 1). The success rate of CTM puncture was significantly higher with the real-time ultrasound technique than with the static ultrasound technique (Table 1). Puncture accuracy, indicating a high accuracy rate, was significantly higher with the real-time ultrasound technique (Table 1). The procedure time was shorter for the real-time ultrasound technique than for the static ultrasound technique (Table 1).
The carry-over effect occurred when either the real-time or static ultrasound technique was performed first, was not observed (real-time ultrasound technique: first vs. second trial, p = 0.13). Static ultrasound technique: first versus second trial, p = 0.38).
Subgroup analysis
In junior residents, there was no significant difference in the success rate between the static and real-time techniques (Table 1). Among the anesthesia residents, there were no significant differences in the success rate, higher rate of accuracy, and procedure time. There was no significant difference in success rate among the anesthesiologists.
There were no significant differences between the three types of simulators in terms of success rate, high accuracy rate, or procedure time (see Supplementary Table S1 online). No carryover effects were observed that would have occurred if either the real-time or static ultrasound techniques had been performed first (Supplementary Table S2 online).
Discussion
It is debated whether percutaneous or surgical cricothyroidotomy is superior in emergency front-of-neck airways in CICO case4,9,10,11. The Difficult Airway Society of the United Kingdom recommends surgical cricothyroidotomy (scalpel bougie tube cricothyroidotomy)5. The American Society of Anesthesiologists does not recommend a specific approach3. The Japanese Society of Anesthesiologists and the Canadian Anesthesiologists Society support both needle and surgical cricothyroidotomy412. Risk of bleeding is a major concern during surgical cricothyroidotomy. A recent cadaveric report described some thyroid ima arteries with multiple branching patterns over the trachea, which may have a high risk for excessive bleeding during surgical cricothyroidotomy13. Surgical cricothyroidotomy has been reported to cause more bleeding than percutaneous cricothyroidotomy in animal studies using pigs14. In patients with tracheal deviation, misidentification of the CTM may cause unexpected hemorrhage. Therefore, the ultrasound identification of the CTM should be more useful than the digital palpation CTM identification technique for needle cricothyroidotomy in patients with anatomically abnormal necks.
Among the anesthesiologists, the success rate of CTM puncture did not differ between the static and real-time ultrasound techniques. The same trend was observed among the anesthesia residents. Although peripheral nerve blocks are usually performed under ultrasound guidance, epidural and spinal anesthesia are still usually performed using the anatomic landmark technique, either blindly or depending on spatial ability. Therefore, anesthesiologists not only routinely practice ultrasound guidance training but also use spatial grasping training. However, junior residents do not have this training environment and would greatly benefit from real-time ultrasound technology, which allows them to immediately understand ultrasound-guided needle projection.
We conclude that real-time ultrasound is more useful than static ultrasound for CTM punctures. However, the success rate of the real-time technique is only 60%. This result indicates that ultrasound-guided CTM puncture may not be suitable for emergency cricothyroidotomy. However, in the subgroup analysis, the anesthesiologist reported a success rate of 78% when using the real-time ultrasound technique. Considering that the simulator was designed for a difficult case of cricothyrotomy, ultrasound-guided needle cricothyroidotomy performed by an anesthesiologist may be clinically feasible.
After needle cricothyroidotomy, oxygen is typically supplied via low-pressure ventilation or jet ventilation15. Alternatively, a cannula can be inserted using the Seldinger technique. Puncture of the limbus of the CTM can result in difficult insertion of the cannula into the trachea and possible misplacement outside the trachea. In this study, the procedure in which a puncture needle was placed at the center of the simulated trachea was defined as highly accurate. The accuracy rate of the real-time ultrasound technique was significantly higher than that of the static ultrasound technique. In the subgroup analysis, both junior residents and anesthesiologists had a significantly higher rate of accuracy with the real-time ultrasound technique than with the static ultrasound technique. In this study, cannula insertion was not performed after puncture. Even if the puncture is performed correctly, ease of cannula insertion may depend on the quality of the puncture kit. Therefore, further studies are needed before the results of this study can be directly applied in clinical practice.
Some guidelines state that awake intubation should be selected in cases with a high risk of difficult mask ventilation and tracheal intubation. In patients with anticipated difficult airways, it is important to plan to secure the airway safely. Usually, awake intubation or surgical front-of-neck access is used for difficult airways. In particular, awake intubation will require a backup plan called “double setup airway intervention,” which involves preparing simultaneously for both awake intubation and surgical cricothyroidotomy4. If the results of this study were to be integrated into the current guidelines, ultrasound would be used for CTM identification before surgical cricothyroidotomy, and guidewire16 would be inserted through a thin puncture needle using ultrasound guidance. This guidewire will increase the success rate of cricothyroidotomies.
Study limitation
Three subsets (junior residents, anesthesiology residents, and anesthesiologists) were enrolled in this study. It is undeniable that this heterogeneity may have affected the results. In particular, the inexperience of the residents may have contributed to the lower overall success rate. However, cricothyroidotomy in patients with cervical abnormalities is a challenging procedure for anyone, not just junior residents. Therefore, it is essential to find a way to improve its safety using ultrasound guidance.
Another problem is that there is currently no commercially available cricothyroidotomy kit suitable for real-time ultrasound technique. In order to take advantage of the results of this study, a new cricothyroidotomy kit needs to be developed.
In conclusion, CTM puncture using the real-time ultrasound technique may be more useful than the static ultrasound technique, especially for naïve operators. However, further research is required to translate these results into clinical practice.
Data availability
The datasets generated in the current study are available from the corresponding author upon request.
References
Cook, T. M., Woodall, N., Harper, J., Benger, J. & Fourth National Audit Project. Major complications of airway management in the UK: Results of the Fourth National Audit Project of the Royal College of Anaesthetists and the Difficult Airway Society. Part 2: Intensive care and emergency departments. Br. J. Anaesth. 106(5), 632–642 (2011).
Rosenblatt, W. H. & Yanez, N. D. A decision tree approach to airway management pathways in the 2022 difficult airway algorithm of the American society of anesthesiologists. Anesth. Analg. 134(5), 910–915 (2022).
Apfelbaum, J. L. et al. 2022 American society of anesthesiologists practice guidelines for management of the difficult airway. Anesthesiology 136(1), 31–81 (2022).
Law, J. A. et al. Canadian airway focus group updated consensus-based recommendations for management of the difficult airway: Part 2. Planning and implementing safe management of the patient with an anticipated difficult airway. Can. J. Anaesth. 68(9), 1405–1436 (2021).
Frerk, C. et al. Difficult Airway Society 2015 guidelines for management of unanticipated difficult intubation in adults. J. Anaesth. 115(6), 827–848 (2015).
Siddiqui, N., Yu, E., Boulis, S. & You-Ten, K. E. Ultrasound is superior to palpation in identifying the cricothyroid membrane in subjects with poorly defined neck landmarks: A randomized clinical trial. Anesthesiology 129(6), 1132–1139 (2018).
You-Ten, K. E. et al. Practice of ultrasound-guided palpation of neck landmarks improves accuracy of external palpation of the cricothyroid membrane. Anesth. Analg. 127(6), 1377–1382 (2018).
Markowitz, J. E. Cricothyroidotomy (Cricothyrotomy) Technique (Clinical Procedures). Medscape https://emedicine.medscape.com/article/80241-technique?form=fpf. Accessed 14 Apr.
Kristensen, M. S., Teoh, W. H. & Rudolph, S. S. Ultrasonographic identification of the cricothyroid membrane: Best evidence, techniques, and clinical impact. Br. J. Anaesth. 117(Suppl_1), i39–i48 (2016).
Yeow, C., Greaney, L., Foy, C., King, W. & Patel, B. Evaluation of a novel cricothyroidotomy introducer in a simulated obese Porcine model: A randomised crossover comparison with scalpel cricothyroidotomy. Anaesthesia 73(10), 1235–1243 (2018).
Rees, K. A. et al. J. Time to oxygenation for cannula- and scalpel-based techniques for emergency front-of-neck access: a wet lab simulation using an ovine model. Anaesthesia 74(9), 1153–1157 .
Japanese Society of Anesthesiologists. JSA airway management guideline 2014: To improve the safety of induction of anesthesia. J. Anesth. 28(4), 482–493 (2014).
Totlis, T., Natsis, K., Achlatis, V., Pettas, T. & Piagkou, M. Thyroidea Ima artery multiple branching pattern over the trachea. Surg. Radiol. Anat. 45(7), 813–817 (2023).
Chen, Y. et al. Cricothyroid membrane puncture-guided tracheostomy: A new technique for emergency airway access. J. Oral Maxillofac. Surg. 76(6), 1248–1254 (2018).
Mariappa, V., Stachowski, E., Balik, M., Clark, P. & Nayyar, V. Cricothyroidotomy: Comparison of three different techniques on a Porcine airway. Anaesth. Intensive Care 37(6), 961–967 (2009).
Vora, J., Leslie, D. & Stacey, M. Awake tracheal intubation. BJA Educ. 22(8), 298–305 (2022).
Acknowledgements
The authors would like to thank all the participants of this study.
Funding
The University Department provided funding for this study.
Author information
Authors and Affiliations
Contributions
H.N substantially contributed to the study conceptualization. H.W. and H.N. significantly contributed to investigation, data curation and analysis. H.W. H.N. and J.T. substantially contributed to the manuscript drafting. J.T. and T.Y. supervised the conduct of this study. All authors reviewed the manuscript draft and revised it critically on intellectual content. All authors approved the final version of the manuscript to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This case report was written in accordance with institutional ethical guidelines.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Watanabe, H., Nakazawa, H., Tokumine, J. et al. Real-time vs. static ultrasound-guided needle cricothyroidotomy: a randomized crossover simulation trial. Sci Rep 15, 8112 (2025). https://doi.org/10.1038/s41598-025-92684-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-92684-4