Abstract
Purpose. Increased CD73 expression has been associated with progression in various cancer types. Results of the AZURE and other trials suggest that, in postmenopausal breast cancer patients, adjuvant bisphosphonates inhibit bone relapses and prolong overall survival. Based on these findings, adjuvant bisphosphonates (typically zoledronic acid) are standard-of-care in postmenopausal patients with high-risk early breast cancer. However, biomarkers are needed for improved patient selection. The aim of this study was to investigate the association of primary tumor CD73 expression with later development of bone metastases. Methods. To determine whether CD73 levels correlated with tumor parameters (hormone receptor status, tumor stage and grade), patient outcomes (bone metastases and survival) or other patient characteristics (menopausal status, chemotherapy or statin use), we analyzed primary breast tumor CD73 expression immunohistochemically in tumor microarray samples from the AZURE (BIG01/04) trial. Results. In the AZURE control arm, high CD73 score are significantly prognostic for overall survival (p-value = 0.03, HR = 1.87, 95% CI = 1.06–3.29), disease-free survival (p-value = 0.06, HR = 1.66, 95% CI = 0.982–2.8) and time to first metastasis to bone (p-value = 0.04, HR = 2.23, 95% CI = 1.04–4.81), as compared with low CD73 scores. However, high CD73 score did not display an association with time to non-bone metastasis or first recurrence to a non-skeletal site. In the zoledronate arm, high CD73 score did not have association with patient outcomes, first metastasis to bone, nor with bone recurrence at any time (distant recurrence, including skeletal) or first non-skeletal recurrence. In multivariate testing, CD73 had no significant association with age, ER status, tumor stage, histological grade, menopausal status, chemotherapy or statin use in either arm. Conclusions. High CD73 expression is associated with development of bone metastases. Zoledronate counteracts this effect. These results suggest that CD73 expression might serve as a biomarker for adjuvant zoledronic acid use.
Similar content being viewed by others
Introduction
Disease recurrence remains a significant problem among patients with early breast cancer that have been treated with curative intent. Approximately 70–80% of patients who do relapse, develop bone metastases1. Bone metastases may linger in a senescent stage for years, possibly decades, before they grow into symptomatic macroscopic metastases but, once manifest, they may cause pain, pathological fractures, hypercalcemia and spinal cord compression. However, time to relapse depends greatly on breast cancer subtype. Patients with triple-negative breast cancer (TNBC) typically relapse within the first five years after treatment, whereas those with hormone receptor-positive subtype may relapse many years later2,3.
Dormant cells play a critical role in the recurrence of cancer, particularly in breast cancer. These cells, known as disseminated tumor cells, can remain in a dormant state within the bone marrow for years, after the primary tumor has been surgically removed. Dormant disseminated tumor cells in the bones depend on pro-survival signals from the microenvironment and develop complex immune evasion mechanisms, leading to disease recurrence4. In ER-positive breast cancer patients and those with larger tumor sizes, pre-operative dormant tumor cells were a significant predictor of late systemic recurrences4. For cancers that commonly spread to bones, such as breast cancer, treatment plans could be based on prognostic markers to improve long-term outcomes and prevent late recurrences. Currently, bisphosphonates are the only standard treatment that may prevent the formation of bone metastases. When given in the adjuvant setting to post-menopausal patients with early breast cancer with high risk of relapse, they may prevent bone metastases, but this benefit does not extend to pre-menopausal patients. The reason for this is currently unknown, but it may be immunologically regulated5. However, although a number of potential biomarkers to assess the risk of recurrence and predict response to bisphosphonates have been identified6,7,8,9, none are currently in routine clinical use for identification of risk and response, and the number needed to be treated remains substantially higher than the numbers who will benefit. Although bisphosphonate drugs are relatively safe and well understood, it would be beneficial to have biomarkers to guide their clinical use.
The large (N = 3300), academic, multi-center, randomized phase III AZURE (BIG01/04) study evaluated the addition of the bisphosphonate zoledronic acid (4 mg) to standard therapy (neo/adjuvant chemotherapy and/or endocrine therapy) for 5 years10,11. In this and other adjuvant bisphosphonate studies, analyzed in a large (N = 18 700) meta-analysis, the benefits of treatment were limited to post-menopausal women and the benefit remained rather low at 3.3% for breast cancer mortality and 2.2% for recurrence at 10 years (EBCTCG). Currently, the most promising predictive biomarker for adjuvant bisphosphonate use is the MAF (mesenchymal aponeurotic fibrosarcoma) gene. Specifically, patients with MAF-negative breast cancers appear to benefit from adjuvant bisphosphonates. The predictive ability of c-MAF amplification to determine the outcome of treatment with bisphosphonates (either zoledronic acid or clodronate) was recently outlined in two key studies9,12. Despite these promising studies, further validation of potential biomarkers prognostic for breast cancer spread to bone and metastasis treatment are required before these markers are considered for routine clinical use.
CD73 is a membrane-bound receptor, the main function of which is to hydrolyze ribonucleoside 5′-monophosphates (AMP, uridine monophosphate, inosine monophosphate and cytidine monophosphate) to extracellular adenosine and inorganic phosphate13. Enzymatic activity of CD73, due to adenosine production, is associated with immunosuppressive events in cells14. As a regulator of tumor immunity, CD73 is also a novel target for immunotherapies. There are several ongoing phase I (NCT03875573, NCT03616886, NCT05431270) or phase II15 trials to investigate the efficacy of CD73 inhibition in breast cancer, as well as in other types of cancer16. We and others have demonstrated previously that CD73 expression is associated with metastases17,18, especially among TNBC19. The aim of this study was to investigate whether primary tumor CD73 expression is prognostic specifically for bone metastases, using tissue samples and the associated clinical database from the AZURE trial.
Materials and methods
Sample information
AZURE study
AZURE (BIG01/04) was an academic, prospective, randomized controlled phase III, open-label multi-national and multi-center trial (ISRCTN79831382). Patients with histologically confirmed invasive breast cancer of any biological subtype, with either pathologically confirmed axillary lymph node metastases or a T3/T4 primary tumor were enrolled. Patients were not eligible for this study if there was clinical or imaging evidence of distant metastases prior to study entry or current or recent (previous year) use of bisphosphonates or pre-existing bone disease likely to require bone-targeted treatment11,20. All patients gave written informed consent for this study. Between September 2003 and February 2006, 3360 patients from 174 centers were randomized on a 1:1 basis, using a central automated 24-hour computer-generated telephone randomization system to receive (neo)adjuvant systemic therapy with zoledronic acid or without (Control) zoledronic acid. A minimization method ensured balance in key prognostic and treatment variables across the two groups11. AZURE patients were reviewed annually and followed for 10 years. All analyses on patient samples were ethically approved (Ethics reference number: 55/03/182). Analyses for correlation of CD73 levels with risk of bone metastasis were performed on tissue microarrays (TMAs) constructed from primary tumors from a subset of patients within the AZURE trial (n = 689). Due to the relatively high prevalence of bone metastatic outcome within this group, and the long follow-up (median 117 months [interquartile range 70.4–120.4]), these TMAs provide an excellent resource for validation of protein biomarkers emerging from our histological analysis.
Immunohistochemistry (IHC)
Protein expression was assessed on TMAs using immunohistochemistry. Briefly, 5 μm serial tissue sections were dewaxed in xylene and rehydrated through graded alcohols. Endogenous peroxidases were blocked (3% H2O2 for 10 min), and antigens retrieved by microwaving the slides in sodium citrate buffer, pH 6 (800 W for 7 min). After cooling and washing, slides were blocked with normal goat serum for 1 h at room temperature, after which primary antibodies against CD73 (CST, 13160, 1:1000) were applied (overnight, 4 °C). The slides were incubated with biotinylated secondary rabbit antibodies (BA-2000 Vector, UK) at room temperature for 1 h, followed by incubation with avidin/biotinylated enzyme complex (ABC) for 1 h, 3,3′-diaminobenzidine (DAB) for 1 min and counterstaining with hematoxylin for 10 s (both Vector laboratories, UK). Slides were scanned using Pannoramic 250 slide scanner (3DHISTECH Ltd, Hungary).
IHC scoring
A standard IHC approach of semi-quantitative scoring based on operator assessment of weak to intense staining was performed using a simple scoring system of 0 (no expression), 1 (weak expression), 2 (intermediate expression) and 3 (strong expression). For antibody optimization, IHC staining was performed using a range of primary antibody dilutions. The dilution that gave the widest range of cytoplasmic staining intensities across different TMA samples was chosen.
This optimized approach was then applied to TMAs from the AZURE study. IHC staining was scored as either level 0, 1, 2 or 3 as described above. Scoring was performed blindly by two trained operators. Scoring was based on staining intensity only, and not on percentage of positive cells. This was appropriate for CD73, since when staining was present, it was of a similar intensity throughout the tumor cells, rather than being patchy or focal in nature, and therefore the number of stained cells was not an issue. Kappa coefficients were calculated for the two independent scores demonstrating very significant agreement (Kappa coefficient > 0.85). Discrepant scores were assessed by an independent adjudicator and values assigned. CD73 IHC scores were submitted via patient identification numbers to the clinical database of the AZURE study for alignment to the AZURE clinical data and statistical analyses (Walter Gregory).
Statistical analysis
CD73 scoring data was analyzed within Excel to determine the frequency of CD73 scores within defined patient subgroups (e.g. tumor stage) for each arm of the trial. The distribution of CD73 scores between different subgroups of each such variable was then tested for association by use of a heterogeneity Chi-squared test, with degrees of freedom equal to the number of categories for that variable minus 1. Immunohistochemical analyses followed REMARK guidelines21. Statistical analyses evaluated the associations between CD73 protein expression and relevant clinical and pathological variables (e.g. ER/PR/HER2 status) using Fisher’s exact test (categorical variables) and the Kruskal–Wallis test (continuous variables), before assessing prognostic and predictive associations with time-to-event data (time to first distant recurrence, time to first skeletal recurrence, time to first non-skeletal recurrence) using Cox proportional hazards regression, the Kaplan–Meier estimate of the survival function and the log-rank test. Time to first distant recurrence was defined as the time from the date of randomization to the date of the distant recurrence. In analyses, other types of events were censored. For example, if a local recurrence occurred prior to any distant recurrence, the patient would be censored at the date of the local recurrence. Time to first skeletal recurrence and first non-skeletal recurrence were defined similarly. Time to first skeletal recurrence irrespective of all other previous recurrences was also investigated. Time to event analysis was first performed within treatment arms to identify prognostic associations with CD73.
Multivariate analysis looking for potential correlation of CD73 levels with patient variables (including menopausal status, ER, HER2 and PR status) were performed using Mann-Whitney U-test. The predictive heterogeneity of effect between treatment arms for time to distant events was then assessed in multivariable analysis by including an interaction term in the Cox proportional hazard regressions for treatment arm and biomarker (while adjusting for systemic therapy plan, ER status and lymph node involvement). All significance tests were two-sided and were designated significant at the 5% level.
Ethics
The study was performed in accordance with ethical principles originating in the Declaration of Helsinki. This research study at the University of Turku was approved by the Finnish National Board on Research Integrity TENK (ISBN 978-952-5995-88-6). Data were anonymized for statistical analyses and handled in a manner that meets the EU General Data Protection Regulation 2016/679 (GDPR) on data protection.
Results
Clinical association of CD73 expression with patient characteristics in breast cancer patients in the AZURE trial
In the study, CD73 staining intensities were analyzed from a TMA cohort from the AZURE (BIG/01/04) trial. All analyses comparing CD73 scores with patient variables, or with time-to-event data, employed data from IHC staining of tumor cores, where CD73 staining intensity was scored as either 0, 1, 2 or 3 (Fig. 1). Dichotomized staining scores were used to compare high CD73 expression (Score = 3) with low CD73 expression (score = 1 or 2). We observed that most cores exhibited low CD73 expression, with negligible instances of high expression. Cores which were not of sufficient quality to score accurately were assigned as non-scorable (NS). The possible association of CD73 with outcome was tested for 422 patients (204 in the Control Arm and 218 in the Zoledronate Arm). The demographic data for the patients with assessable cores and the overall AZURE population were similar (Table 1).
Representative images of immunostaining for CD73 in TMA cores from patients in the AZURE study. (A) Images of protein expression obtained using the anti-CD73 antibody and visualized at a magnification of 20x. Score 0–3 categories were determined by the intensity of staining in the cytoplasmic compartment of tumor cells only. Scale bar = 200 μm. (B) 40x magnification view of from the boxed regions depicted in panel A. Scale bar of core view = 100 μm. Scale bar of 40x = 50 μm.
In multivariate testing, CD73 had no significant associations with age, ER status, tumor stage, histological grade, menopausal status, chemotherapy or statin use (Table 2). The measurement of HER2 status was not mandated within the AZURE trial and HER2 measurements are available for 307 participants in total, of which 168 of these patients had assessable cores for CD73 staining. Within this analysis, CD73 did not associate significantly with HER2 status. In addition to these analyses within the control arm of AZURE, no significant associations of CD73 level with clinical variables were observed within the zoledronic acid arm (Table 2).
Association of CD73 expression with patient outcome
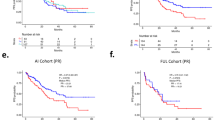
Kaplan-Meier analysis of overall survival showed significant association of high CD73 expression with worse prognosis compared with those for low CD73 expression (p-value = 0.03, HR = 1.87, 95% CI = 1.06–3.29) in control arm but not in the zoledronic acid arm (Fig. 2A, B; Table 3). Association between high CD73 expression and worse disease-free survival (DFS) almost reached significance compared with those with low CD73 expression (p-value = 0.06, HR = 1.66, 95% CI = 0.982–2.8) in the control arm but not in the zoledronic acid arm (Fig. 2C, D; Table 3).
Prognostic value of CD73 levels in clinical events. Kaplan-Meier association of CD73 expression level in Overall Survival (OS) in (A) control and (B) zoledronic acid arms. Relationship between level of expression of CD73 in Disease-Free-Survival (DFS) in (C) control and (D) zoledronic acid arms. P-value is obtained from the log-rank test for testing quality of survival Q15 functions.
CD73 expression is associated with distant recurrence
Kaplan-Meier estimates for the survival function for the time to distant recurrence confirmed that high CD73 expression is a significantly prognostic factor for time to first metastasis to bone (p-value = 0.04, HR = 2.23, 95% CI = 1.04–4.81) within the control arm (Fig. 3A; Table 3). Expression of CD73 did not correlate with any first recurrence (including skeletal) (p < 0.06, HR = 1.77, 95% CI = 0.97–3.21) or to a non-skeletal site (p < 0.17, HR = 1.74, 95% CI = 0.791–3.82) within control arm (Fig. 3C, E; Table 3). We did not observe an association between CD73 level and first skeletal event whether other distant events had occurred first (p < 0.06, HR = 1.86, 95% CI 0.97-3.55) within control arm (Table 3). The inclusion of metastasis to another site thus would appear to reduce the association seen during the bone only analysis.
Kaplan-Meier association of CD73 levels with clinical metastatic events. Relationship between CD73 expression to time to first bone recurrence, – where cancer spread to bone is the first recorded event in (A) control and (B) zoledronic acid arm. Relationship between level of CD73 expression to spread to bone at any time (for which patients may have had spread to non-bone sites first) in (C) control and (D) zoledronic acid arm. Relationship between CD73 expression to time to spread to non-bone sites in (E) control and (F) zoledronic acid arm. P-value is obtained from the log-rank test for testing quality of survival Q15 function.
In the zoledronic acid arm, we did not observe association between high CD73 levels and time to first metastasis to bone (p-value = 0.92, HR = 0.945, 95% CI 0.316–2.83), bone recurrence at any time (distant recurrence including skeletal) (p-value = 0.50, HR = 0.717, 95% CI 0.275–1.87) or first non-skeletal recurrence (p-value = 0.70, HR = 0.85, 95% CI 0.374–1.93) (Fig. 3B, D, F; Table 3). These results suggest that the increased risk for first metastasis to bone patients with high tumor CD73 expression is counteracted by treatment with zoledronate.
Discussion
Breast cancer metastasis to bone is associated with increased patient morbidity with development of bone-specific effects, including bone fractures, spinal cord compression and hypercalcemia. In the current study, we have demonstrated that high levels of CD73 expression within primary breast tumors are associated with a greater risk of cancer spread first to bone.
CD73 is a cell surface protein found on normal epithelial and stromal cells22,23,24,25,26 and tumor cells27,28,29,30,31,32. CD73 has prognostic value in TNBC33. Specifically, low tumor cell expression of CD73 is associated with prolonged disease-free survival in TNBC patients. Furthermore, high CD73 expression on epithelial cells was negatively associated with tumor infiltrating lymphocytes, suggesting an immunoregulatory mechanism behind the survival difference33. In the present study, we found a correlation between high CD73 expression and OS or DFS in breast cancer patients from control arm of the AZURE trial. However, we did not observe such correlation in patients treated with adjuvant zoledronate.
In normal tissues, CD73-derived adenosine exerts tissue protective functions, but in cancer, it may help tumors escape immune surveillance5. Because of this aspect in tumor immunology, CD73 has emerged as a possible target for cancer therapy. High tumor CD73 expression has also been associated with poor response to anthracyclines34 and NACT27. The poor response may be also associated with tumor immunity, as various chemotherapeutic agents were shown to induce upregulation of PD-L1 and other checkpoint inhibitors35. In our recent study, silencing of CD73 expression (using shRNA) within breast cancer cells was demonstrated to increase the sensitivity of these cells to the growth inhibitory actions of nitrogen-containing bisphosphonates in vivo5.
CD73 has been so have several roles within the bone microenvironment. High CD73 expression level was demonstrated to promote osteoblastic differentiation in mice36. Osteoblasts play a key role in the maintenance of breast cancer cell dormancy within the bone marrow, enabling cancer cells to evade cytotoxic drugs that target rapidly proliferation cells. Later, these dormant cells may emerge as macro-metastases37. The importance of CD73 in breast cancer spread to bone was also demonstrated in a study targeting both CD73 and transforming-growth-factor-β (TGF-β) in mouse models of TNBC38. Xing and coworkers demonstrated that high expression of CD73 was associated with epithelial-to-mesenchymal transition, as well as the development of a fibrotic stroma and an immune-tolerant tumor micro-environment. Dual blockade of CD73 and TGFβ promoted a multifaceted inflammatory tumor microenvironment with diminished levels of myeloid-derived suppressor cells (MDSCs) and M2-macrophages, as well as increased levels of activated dendritic cells, cytotoxic T cells, and B cells. Their study suggested that dual targeting of CD73 and TGF-β may serve as a potential therapeutic option within TNBC38.
Our study has several limitations. The low frequency of high CD73 expression in the cohort may limit the generalizability of our findings. Additionally, the incomplete data on HER2 status, due to its non-mandatory measurement in the AZURE trial, is another limitation that may impact the comprehensiveness of our analysis. We demonstrated that high CD73 expression did not show a significant association with patient outcomes in the zoledronic acid arm. However, we did not investigate whether CD73 expression is affected by zoledronate treatment in patient samples but it was not regulated in mouse TNBC cells upon zoledronate in vitro, according to our previous study5. In tumor microenvironment, CD73 expression is regulated by various factors, such as hypoxia19, signaling pathways PI3K, MAPK39, transcription factors40 or miRNAs41. However, studies, whether CD73 protein expression is affected by anti-cancer treatment when it is not specific CD73 inhibitor42,43 are limited.
CD73 showed association with metastases in different cancers. High tumor CD73 expression significantly correlated with worse outcome of patients with colorectal cancer liver metastases18 or metastatic melanoma44. A comprehensive meta-analysis of 2951 cancer cases revealed that high CD73 expression is significantly associated with shorter overall survival in various cancers, including breast cancer and is strongly associated with lymph node metastases45. We show here that high CD73 expression in the primary tumors is significantly associated with the development of bone metastases in the control arm of the study. Furthermore, Kaplan-Meier analysis revealed a significant prognostic value of high CD73 expression for time to first metastasis to bone in the control arm, which was not evident in the zoledronic acid arm. These findings suggest a potential role for CD73 as a prognostic marker on bone metastasis. However, additional investigation is necessary to uncover the underlying mechanisms and assess the clinical implications of targeting CD73 in breast cancer.
Data availability
Detailed data analysis and statistical analysis are provided within the manuscript. The raw data includes sensitive patient information that cannot be fully anonymized, and we are bound by confidentiality agreements and institutional regulations that restrict data sharing.
References
Cummings, M. C. et al. Metastatic progression of breast cancer: insights from 50 years of autopsies. J. Pathol. 232, 23–31. https://doi.org/10.1002/path.4288 (2014).
Sopik, V., Sun, P. & Narod, S. A. Predictors of time to death after distant recurrence in breast cancer patients. Breast Cancer Res. Tr. 173, 465–474. https://doi.org/10.1007/s10549-018-5002-9 (2019).
Dent, R. et al. Pattern of metastatic spread in triple-negative breast cancer. Breast Cancer Res. Tr. 115, 423–428. https://doi.org/10.1007/s10549-008-0086-2 (2009).
Tjensvoll, K. et al. Detection of disseminated tumor cells in bone marrow predict late recurrences in operable breast cancer patients. BMC Cancer. 19 https://doi.org/10.1186/s12885-019-6268-y (2019).
Petruk, N. et al. CD73 regulates zoledronate-induced lymphocyte infiltration in triple-negative breast cancer tumors and lung metastases. Front. Immunol. 14 https://doi.org/10.3389/fimmu.2023.1179022 (2023).
Westbrook, J. A. et al. CAPG and GIPC1: breast Cancer biomarkers for bone metastasis development and treatment. JNCI-J Natl. Cancer I. 108 https://doi.org/10.1093/jnci/djv360 (2016).
Westbrook, J. A. et al. Identification and validation of DOCK4 as a potential biomarker for risk of bone metastasis development in patients with early breast cancer. J. Pathol. 247, 381–391. https://doi.org/10.1002/path.5197 (2019).
Pavlovic, M. et al. Enhanced MAF oncogene expression and breast Cancer bone metastasis. JNCI-J Natl. Cancer I. 107 https://doi.org/10.1093/jnci/djv256 (2015).
Coleman, R. et al. Effect of amplification on treatment outcomes with adjuvant Zoledronic acid in early breast cancer: a secondary analysis of the international, open-label, randomised, controlled, phase 3 AZURE (BIG 01/04) trial. Lancet Oncol. 18, 1543–1552. https://doi.org/10.1016/S1470-2045(17)30603-4 (2017).
Wilson, C. et al. Adjuvant Zoledronic acid reduces fractures in breast cancer patients; an AZURE (BIG 01/04) study. Eur. J. Cancer. 94, 70–78. https://doi.org/10.1016/j.ejca.2018.02.004 (2018).
Coleman, R. E. et al. Benefits and risks of adjuvant treatment with Zoledronic acid in stage II/III breast cancer. 10 years follow-up of the AZURE randomized clinical trial (BIG 01/04). J. Bone Oncol. 13, 123–135. https://doi.org/10.1016/j.jbo.2018.09.008 (2018).
Paterson, A. H. G. et al. MAF amplification and adjuvant clodronate outcomes in early-stage breast cancer in NSABP B-34 and potential impact on clinical practice. JNCI Cancer Spectr. 5 https://doi.org/10.1093/jncics/pkab054 (2021).
Silva-Vilches, C., Ring, S. & Mahnke, K. ATP and its metabolite adenosine as regulators of dendritic cell activity. Front. Immunol. 9 https://doi.org/10.3389/fimmu.2018.02581 (2018).
Schneider, E. et al. Generation and function of non-cell-bound CD73 in inflammation. Front. Immunol. 10 https://doi.org/10.3389/fimmu.2019.01729 (2019).
De Caluwé, A. et al. Neo-CheckRay: Radiation therapy and adenosine pathway Blockade to increase benefit of immuno-chemotherapy in early stage luminal B breast cancer, a randomized phase II trial. BMC Cancer. 21 https://doi.org/10.1186/s12885-021-08601-1 (2021).
Xia, C. L., Yin, S. H., To, K. K. W. & Fu, L. W. CD39/CD73/A2AR pathway and cancer immunotherapy. Mol. Cancer. 22 https://doi.org/10.1186/s12943-023-01733-x (2023).
Ma, X. L. et al. CD73 promotes hepatocellular carcinoma progression and metastasis via activating PI3K/AKT signaling by inducing Rap1-mediated membrane localization of P110 and predicts poor prognosis. J. Hematol. Oncol. 12 https://doi.org/10.1186/s13045-019-0724-7 (2019).
Messaoudi, N. et al. Prognostic value of CD73 expression in resected colorectal cancer liver metastasis. Oncoimmunology 9 https://doi.org/10.1080/2162402x.2020.1746138 (2020).
Petruk, N. et al. CD73 facilitates EMT progression and promotes lung metastases in triple-negative breast cancer. Sci. Rep-Uk. 11 https://doi.org/10.1038/s41598-021-85379-z (2021).
D’Oronzo, S. et al. Natural history of stage II/III breast cancer, bone metastasis and the impact of adjuvant zoledronate on distribution of recurrences. J. Bone Oncol. 28 https://doi.org/10.1016/j.jbo.2021.100367 (2021).
Sauerbrei, W., Taube, S. E., McShane, L. M., Cavenagh, M. M. & Altman, D. G. Reporting recommendations for tumor marker prognostic studies (REMARK): an abridged explanation and elaboration. Jnci-J Natl. Cancer I. 110, 803–811. https://doi.org/10.1093/jnci/djy088 (2018).
Mediero, A., Wilder, T., Shah, L. & Cronstein, B. N. Adenosine A receptor (A2AR) stimulation modulates expression of semaphorins 4D and 3A, regulators of bone homeostasis. Faseb J. 32, 3487–3501. https://doi.org/10.1096/fj.201700217R (2018).
Shih, Y. R. V. et al. Dysregulation of ectonucleotidase-mediated extracellular adenosine during postmenopausal bone loss. Sci. Adv. 5 https://doi.org/10.1126/sciadv.aax1387 (2019).
Sutton, N. R. et al. CD73 promotes Age-Dependent accretion of atherosclerosis. Arterioscl Throm Vas. 40, 61–71. https://doi.org/10.1161/Atvbaha.119.313002 (2020).
Eichin, D. et al. CD73 contributes to anti-inflammatory properties of afferent lymphatic endothelial cells in humans and mice. Eur. J. Immunol. 51, 231–246. https://doi.org/10.1002/eji.201948432 (2021).
Hara, T. et al. CD73(+) epithelial progenitor cells that contribute to homeostasis and renewal are depleted in eosinophilic esophagitis. Cell. Mol. Gastroenterol. Hepatol. 13, 1449–1467. https://doi.org/10.1016/j.jcmgh.2022.01.018 (2022).
Cerbelli, B. et al. CD73 expression and pathologic response to neoadjuvant chemotherapy in triple negative breast cancer. Virchows Arch. 476, 569–576. https://doi.org/10.1007/s00428-019-02722-6 (2020).
Tahkola, K. et al. Prognostic impact of CD73 expression and its relationship to PD-L1 in patients with radically treated pancreatic cancer. Virchows Arch. 478, 209–217. https://doi.org/10.1007/s00428-020-02888-4 (2021).
He, X. D. et al. Impact of intratumoural CD73 expression on prognosis and therapeutic response in patients with gastric cancer. Eur. J. Cancer. 157, 114–123. https://doi.org/10.1016/j.ejca.2021.08.006 (2021).
Tripathi, A. et al. Prognostic significance and immune correlates of CD73 expression in renal cell carcinoma. J. Immunother. Cancer. 8 https://doi.org/10.1136/jitc-2020-001467 (2020).
Chen, Y. H., Lu, H. I., Lo, C. M. & Li, S. H. CD73 promotes tumor progression in patients with esophageal squamous cell carcinoma. Cancers 13 https://doi.org/10.3390/cancers13163982 (2021).
Rocha, P. et al. CD73 expression defines immune, molecular, and clinicopathological subgroups of lung adenocarcinoma. Cancer Immunol. Immun. 70, 1965–1976. https://doi.org/10.1007/s00262-020-02820-4 (2021).
Buisseret, L. et al. Clinical significance of CD73 in triple-negative breast cancer: multiplex analysis of a phase III clinical trial. Ann. Oncol. 29, 1056–1062. https://doi.org/10.1093/annonc/mdx730 (2018).
Loi, S. et al. CD73 promotes anthracycline resistance and poor prognosis in triple negative breast cancer. Proc. Natl. Acad. Sci. USA. 110, 11091–11096. https://doi.org/10.1073/pnas.1222251110 (2013).
Samanta, D. et al. Chemotherapy induces enrichment of CD47/CD73/PDL1 immune evasive triple-negative breast cancer cells. P Natl. Acad. Sci. USA. 115, E1239–E1248. https://doi.org/10.1073/pnas.1718197115 (2018).
Takedachi, M. et al. CD73-generated adenosine promotes osteoblast differentiation. J. Cell. Physiol. 227, 2622–2631. https://doi.org/10.1002/jcp.23001 (2012).
Shupp, A. B., Kolb, A. D., Mukhopadhyay, D. & Bussard, K. M. Cancer metastases to bone: Concepts, mechanisms, and interactions with bone osteoblasts. Cancers 10 https://doi.org/10.3390/cancers10060182 (2018).
Xing, Y. et al. Therapeutic efficacy and mechanism of CD73-TGFβ dual-blockade in a mouse model of triple-negative breast cancer. Acta Pharmacol. Sin. 43, 2410–2418. https://doi.org/10.1038/s41401-021-00840-z (2022).
Zhan, J. H. et al. Regulation of CD73 on NAD metabolism: Unravelling the interplay between tumour immunity and tumour metabolism. Cell. Commun. Signal. 22 https://doi.org/10.1186/s12964-024-01755-y (2024).
Hasmim, M. et al. SNAI1-dependent upregulation of CD73 increases extracellular adenosine release to mediate immune suppression in TNBC. Front. Immunol. 13 https://doi.org/10.3389/fimmu.2022.982821 (2022).
Kordass, T., Osen, W. & Eichmüller, S. B. Controlling the immune suppressor: transcription factors and MicroRnAs regulating CD73/NT5E. Front. Immunol. 9 https://doi.org/10.3389/fimmu.2018.00813 (2018).
Wurm, M. et al. A novel antagonistic CD73 antibody for Inhibition of the immunosuppressive adenosine pathway. Mol. Cancer Ther. 20, 2250–2261. https://doi.org/10.1158/1535-7163.Mct-21-0107 (2021).
Kurago, Z. et al. Inhibitors of the CD73-adenosinergic checkpoint as promising combinatory agents for conventional and advanced cancer immunotherapy. Front. Immunol. 14 https://doi.org/10.3389/fimmu.2023.1212209 (2023).
Turiello, R. et al. Serum CD73 is a prognostic factor in patients with metastatic melanoma and is associated with response to anti-PD-1 therapy. J. Immunother. Cancer 8 https://doi.org/10.1136/jitc-2020-001689 (2020).
Jiang, T. et al. Comprehensive evaluation of NT5E/CD73 expression and its prognostic significance in distinct types of cancers. BMC Cancer. 18 https://doi.org/10.1186/s12885-018-4073-7 (2018).
Acknowledgements
NP was funded by Turku Doctoral Programme of Molecular Medicine and Emil Aaltonen Foundation, Finland. SW, MO and ALG were funded by Weston Park Hospital Cancer Charity (WPCC), Breast Cancer Now (BCN). Biocenter Finland and Institute of Biomedicine Imaging Center are acknowledged for imaging instrumentation. In memory of Dr. Johanna Tuomela, PhD, who dedicated her research investigating breast cancer and died of stage IV breast cancer while working on these studies.
Author information
Authors and Affiliations
Contributions
Design – K.S. Materials – J.B. Analysis – N.P, A.L. and M.O. Statistical analysis – W.G, S.W. Data curation – S.W, J.B. Methodology – J.S and A.J. Writing the original draft – N.P, S.W, J.B and K.S. Writing and editing the manuscript – N.P, S.W, J.S, A.J, J.B and K.S. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Petruk, N., Wood, S.L., Gregory, W. et al. Increased primary breast tumor expression of CD73 is associated with development of bone metastases and is a potential biomarker for adjuvant bisphosphonate use. Sci Rep 15, 9449 (2025). https://doi.org/10.1038/s41598-025-92841-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-92841-9