Abstract
Body temperature (BT) monitoring is critical for the management of critically ill patients, and numerous studies have demonstrated that abnormal BT in ICU patients is linked to adverse outcomes. However, evidence regarding the association between admission BT and 28-day mortality in pediatric intensive care unit (PICU) patients is limited. This study aims to clarify the association between admission BT and 28-day mortality in critically ill pediatric patients. This retrospective analysis utilized the pediatric intensive care (PIC) database, comprising 7,350 patients. The primary outcome was 28-day mortality, while 90-day mortality and in-hospital mortality were assessed as secondary outcomes. Multivariate Cox regression analysis and smooth curve fitting were used to evaluate the relationship between BT and mortality. Ultimately, the 28-day mortality rate in the PICU was 3.5%. Severe hypothermia (HR 1.89, 95% CI 1.35–2.63) and severe hyperthermia (HR 1.97, 95% CI 1.28–3.05) were identified as independent risk factors for 28-day mortality. Curve fitting analysis indicated a U-shaped correlation between BT and 28-day mortality (inflection point = 37.2 °C). Therefore, we conclude that the severity of temperature abnormalities at admission, whether hypothermia or hyperthermia, is directly associated with an increased risk of mortality in pediatric patients.
Similar content being viewed by others
Introduction
Body temperature (BT) is a critical physiological parameter reflecting the health status of critically ill patients and serving as a biomarker for infection detection1. Temperature abnormalities, including hypothermia and hyperthermia, represent distinct adaptive mechanisms influenced by physiological adaptability, nutritional status, ambient temperature, and immunological challenges, and are strongly associated with adverse clinical outcomes2,3,4,5,6.
Extensive studies have shown that temperature abnormalities, including hypothermia and hyperthermia, are strongly associated with adverse outcomes in ICU patients7,8,9. Both conditions increase mortality risk, with death rates rising significantly for each 1 °C deviation from normal BT10,11. However, findings remain inconsistent, as some studies report protective effects of elevated BT in infections12, while others find hypothermia consistently linked to higher mortality in sepsis patients13,14. Despite these variations, most evidence supports the association of both hypothermia and hyperthermia with increased mortality rates15,16.
The impact of temperature variations on disease prognosis remains controversial, especially in pediatric populations, where existing studies are scarce and largely focused on adults, highlighting the need to evaluate its predictive value in critically ill children. Therefore, this study aimed to investigate the relationship between admission BT and 28-day mortality in pediatric intensive care unit (PICU) patients.
Methods
Database
For this study, we utilized the Pediatric Intensive Care (PIC) database, a publicly accessible resource specializing in pediatric ICU admissions (http://pic.nbscn.org/)17. The database, spanning 2010 to 2018, contains data from 12,881 pediatric patients admitted to various intensive care units at the Children’s Hospital of Zhejiang University School of Medicine, China, resulting in a total of 13,941 hospitalizations. We selected patient demographics, diagnostic findings, initial laboratory test results, and patient outcomes for analysis, focusing on variables relevant to the study objective. Ethical approval was granted by the Institutional Review Board of the Children’s Hospital, Zhejiang University School of Medicine, Hangzhou, China. The requirement for explicit patient consent was waived due to the study’s non-interventional and retrospective nature and the use of anonymized data. This waiver complies with the Declaration of Helsinki and national guidelines for secondary research using anonymized health data, ensuring the protection of patient rights and data confidentiality.
Study population
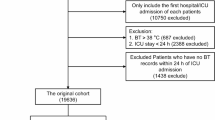
This study employed a retrospective cohort design and included all patients under 18 years of age. For patients with multiple hospital admissions, only data from their first hospitalization were included in the analysis. The body temperature assessed was the first temperature measurement taken within the first 24 h after PICU admission. Patients were excluded if their temperature data were missing or if their readings fell outside the predefined acceptable range for valid measurements. As a result, the final dataset comprised 7,350 patients. Based on their temperature profiles and supported by clinical evidence, patients were divided into three groups12,18,19,20,21: hypothermia (BT < 36.5 °C), normothermia (36.5 °C ≤ BT < 37.5 °C), and hyperthermia (BT ≥ 37.5 °C). No restrictions were imposed regarding the timing of in-hospital mortality. The patient selection process is illustrated in Fig. 1. To further investigate the impact of atypical body temperatures on 28-day mortality, we subdivided the hypothermia group into two subcategories: mild hypothermia (36.0 °C ≤ BT < 36.5 °C) and severe hypothermia (BT < 36.0 °C). Similarly, the hyperthermia group was divided into mild hyperthermia (37.5 °C < BT ≤ 38.0 °C) and severe hyperthermia (BT > 38.0 °C).
Primary and secondary outcomes
To refine and validate the data, unique identifying codes for subjects, admissions, and ICU stays were used to remove duplicates and resolve any ambiguities. Data collected included demographic information, vital signs, ICU admission type, ICD-10 diagnostic codes, in-hospital mortality, and results of initial laboratory assessments following ICU admission (Table 1). Age was calculated as the difference between the date of birth and the PICU admission date, while the length of PICU stay was defined as the interval between PICU admission and discharge. The primary outcome assessed was 28-day mortality, with secondary outcomes including 90-day mortality and in-hospital mortality. The 28-day mortality rate was defined as any death occurring within 28 days of PICU admission, including deaths after discharge from the PICU. Mortality within 90 days of PICU admission was classified as 90-day mortality. Deaths occurring during hospitalization were classified as in-hospital mortality.
Statistical analysis
Categorical variables were summarized as frequencies and percentages. For continuous variables, normally distributed data were expressed as the mean with standard deviation (SD), while skewed data were reported as the median and interquartile range (IQR). Statistical methods applied included the chi-square test for categorical variables, one-way ANOVA for continuous variables with a normal distribution, and the Kruskal–Wallis test for non-normally distributed data. Survival probabilities were compared using Kaplan–Meier curves to examine the effect of BT variations on patient outcomes.
Univariate and multivariate Cox regression models were used to assess the associations between BT and 28-day and 90-day mortalities. Logistic regression was used to evaluate the association between BT and in-hospital mortality. Variables were selected for inclusion based on statistical significance (P < 0.05) from univariate analysis, clinical relevance, or their potential to change the effect estimate by more than 10%. The multivariate models were structured as follows: Model I was unadjusted; Model II adjusted for age and gender; Model III further adjusted for conditions such as pre-admission cardiopulmonary resuscitation (CPR), pneumonia, sepsis, congenital heart disease (CHD), neurological disorders, digestive system disorders, and cardiovascular diseases. Model IV included additional adjustments for laboratory parameters, including white blood cell count (WBC), neutrophil count (NEUT), hemoglobin (HGB), platelet count (PLT), red cell distribution width (RDW), C-reactive protein (CRP), total protein (TP), total cholesterol (TC), lactate dehydrogenase (LDH), lactate (Lac), pH, partial pressure of carbon dioxide (PCO2), potassium (K), and calcium levels (Ca).
To elucidate the correlation between BT and the risk of 28-day mortality, we employed a smoothing technique for curve fitting that incorporated potential confounding factors as outlined in the Cox regression analysis. A piecewise Cox regression model with smoothing was also used to evaluate the threshold effect of BT on 28-day mortality, incorporating the covariates from Model IV. Furthermore, a segmented Cox regression model with smoothing techniques was used to examine the effect of BT on 28-day mortality, incorporating the covariates specified in Model IV.
Moreover, potential variations in the association between BT and 28-day mortality were examined, taking into account factors such as sex, age, CPR, pneumonia, sepsis, CHD, neurological disorders, gastrointestinal diseases, and cardiovascular conditions. Subgroup analyses were specifically conducted based on sepsis status (sepsis vs. non-sepsis groups) and the presence of congenital heart disease (CHD vs. non-CHD groups) to evaluate whether these factors influenced the relationship between BT and 28-day mortality. Multivariate Cox regression was used to analyze heterogeneity across subgroups, and the significance of interactions between these subgroups and BT was assessed using likelihood ratio tests.
In this study, missing values for the extracted variables were less than 15% of the total data. Mean imputation was used for normally distributed variables, whereas median imputation was applied to variables exhibiting skewed distributions. For variables with more than 15% missing values, a complete case analysis was performed by excluding these data.
The analysis was performed using R version 4.2.1 (http://www.Rproject.org; The R Foundation, Vienna, Austria) and Free Statistics software version 1.9.2 (Beijing FreeClinical Medical Technology Co. Ltd., Beijing, China). Statistical significance was set at a p-value of less than 0.05 (two-sided).
Results
The selection of participants
The initial PIC database contained 13,941 records. To ensure each patient was represented only once, 1,060 duplicate cases were removed. From the remaining 12,881 hospitalizations, 5,531 cases were excluded due to missing or invalid body temperature (BT) data. Consequently, the final analysis included 7,350 pediatric cases, as shown in Fig. 1.
Baseline characteristics
Among the patients, 2,654 (36.1%) were classified as hypothermia, 799 (10.9%) with hyperthermia, and 3,897 (53.0%) with normal body temperature. The mean age of the patients was 9.4 months, with 4,089 (55.6%) being male. The mean body temperature was 36.6 ± 0.9 °C. Patients in the hyperthermia group were older and had higher systolic and diastolic blood pressures, along with elevated levels of white blood cells, total protein, serum albumin, neutrophils, C-reactive protein, alanine aminotransferase, total cholesterol, and blood glucose. Conversely, the hypothermia group was younger and exhibited significantly higher levels of hemoglobin, hematocrit, red cell distribution width, total bilirubin, direct bilirubin, aspartate aminotransferase, gamma-glutamyl transferase, alkaline phosphatase, serum creatinine, lactate dehydrogenase, and partial pressure of carbon dioxide. The PICU had a higher proportion of patients with hyperthermia, whereas the NICU and CICU had a greater number of patients with hypothermia. Additionally, the hyperthermia group had a higher prevalence of pneumonia and neurological disorders, while congenital heart disease was more common in the hypothermia group. Table 1 provides a summary of these baseline characteristics.
By the end of the study, the mortality rates were 3.5% at 28 days, 3.9% at 90 days, and 4.0% during the in-hospital period. The average hospitalization lasted 13.0 days, with an ICU stay of 2.0 days on average. Patients with either hyperthermia or hypothermia had significantly higher mortality rates at both 28 and 90 days, along with longer ICU stays, compared to those with normal BT.
Association between BT and clinical outcomes
The univariate and multivariate Cox regression analyses, as shown in Supplementary Table S1 and Table 2, identified significant associations. When using the normal temperature group as a reference, both hypothermia and hyperthermia were significantly linked to increased 28-day mortality across all models. Specifically, hypothermia was consistently associated with elevated mortality rates (Model I: HR 1.59, 95% CI 1.21–2.09; Model II: HR 1.58, 95% CI 1.20–2.09; Model IV: HR 1.43, 95% CI 1.08–1.90). Similarly, hyperthermia was associated with increased mortality risks (Model I: HR 2.32, 95% CI 1.64–3.46; Model III: HR 2.28, 95% CI 1.58–3.28). These associations were also observed for 90-day mortality and in-hospital mortality (Supplementary Table S2).
In various analytical frameworks, severe hypothermia and severe hyperthermia consistently emerged as major predictors of 28-day mortality, a pattern that was also evident for 90-day mortality and in-hospital mortality (Table 3). To further explore the impact of BT on patient outcomes, we analyzed the correlation between the average BT recorded during the first 24 h of PICU admission and both 28-day and 90-day mortality, as well as overall in-hospital mortality. The results of this analysis confirmed a significant association, underscoring BT’s role as an independent risk factor for mortality (Supplementary Table S3).
Results of non-linearity of BT and 28-day mortality
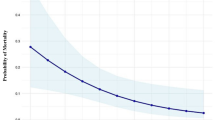
The association between BT and 28-day mortality exhibited a U-shaped pattern, as indicated by multivariate Cox regression and curve fitting analyses, adjusted using Model IV (Fig. 2). Using a segmented Cox regression model with two distinct slopes, we were able to more precisely characterize this relationship. The likelihood ratio test confirmed the statistical significance of this model with a P-value well below 0.001 (Table 4). The analysis revealed a critical temperature threshold of 37.2 °C, beyond which the hazard ratio for mortality increased markedly. Specifically, below this threshold, the hazard ratio was 0.794 (95% CI 0.693–0.909, P < 0.001), whereas above 37.2 °C, it rose to 2.953 (95% CI 1.937–4.501, P < 0.001). These results were corroborated by the significant correlation between the initial 24-h average BT following ICU admission and 28-day mortality.
Non-linear relationship between BT and 28-day PICU mortality. Hazard ratios (HRs) were adjusted for gender, age, CPR, Pneumonia, sepsis, CHD, and Neurologic disease, Digestive disease, Cardiovascular-disease, white blood cell count, neutrophil count, hemoglobin, platelet count, red blood cell distribution width, C-reactive protein, total protein, total cholesterol, lactate dehydrogenase, glucose, lactate, PH, partial pressure of carbon dioxide, potassium, calcium.
Hazard ratios (HRs) were adjusted for gender, age, CPR, Pneumonia, sepsis, CHD, and Neurologic disease, Digestive disease, Cardiovascular-disease, white blood cell count, neutrophil count, hemoglobin, platelet count, red blood cell distribution width, C-reactive protein, total protein, total cholesterol, lactate dehydrogenase, glucose, lactate, PH, partial pressure of carbon dioxide, potassium, calcium.
Kaplan–Meier curve and sensitivity analysis
To analyze survival outcomes across varying body temperatures, we categorized patients into tertiles based on their BT and constructed 28-day survival curves (Fig. 3). Kaplan–Meier survival analysis indicated a significant decrease in 28-day survival rates for the hyperthermia group compared to the normothermia group (P < 0.001). This trend was also observed in the 90-day survival curves shown in Supplementary Fig. S2.
Additionally, stratified analyses were conducted to examine the association between BT and 28-day mortality. No significant interactions were observed in subgroups other than the pneumonia subgroup. The results are shown in Supplementary Fig. S3. To better evaluate the influence of confounding factors, subgroup analyses were conducted based on sepsis status (sepsis vs. non-sepsis groups) and the presence of congenital heart disease (CHD vs. non-CHD groups). In the sepsis group, hyperthermia was significantly associated with an increased risk of mortality (HR 5.22, 95% CI 2.21–12.35, P < 0.001). In the non-CHD group, both hypothermia and hyperthermia were significantly associated with an increased risk of mortality (HR 1.49, 95% CI 1.07–2.08, P = 0.019; HR 1.6, 95% CI 1.02–2.5, P = 0.039). Detailed results are provided in Supplementary Tables S6 and S7.
To assess the robustness of our findings, we conducted a sensitivity analysis after excluding participants with missing data (Supplementary Table 5). The results showed that after excluding missing data from the entire cohort (n = 7,350), the remaining 5892 patients still demonstrated a robust relationship between BT and 28-day mortality. In the sensitivity analysis of this study, the use of vasoactive drugs was included as a covariate to assess its impact on mortality. The results remained stable after adjusting for vasoactive drug use, with detailed data provided in Supplementary Table 10.
Discussion
This retrospective cohort study, conducted in PICU patients, investigated the association between admission BT at admission and 28-day mortality. A U-shaped relationship was observed, where each 1 °C decrease in BT below 37.2 °C was associated with a 20% reduction in 28-day mortality risk. Conversely, each 1 °C increase above 37.2 °C significantly elevated mortality risk (HR 2.95, 95% CI 1.93–4.50). Additionally, our study confirmed that severe hypothermia (< 36.0 °C) and severe hyperthermia (> 38.0 °C) were independent risk factors for both 28-day and 90-day mortality, highlighting the need for early temperature monitoring in critically ill pediatric patients.
Our results align with multiple prior studies that highlight the impact of temperature abnormalities on ICU mortality. Ralf Erkens et al. demonstrated that hypothermia increased ICU mortality fourfold (24%, OR 4.05, 95% CI 3.38–4.85), while hyperthermia doubled the risk (12%, OR 1.80, 95% CI 1.43–2.26)19. Similarly, Wang et al. found that in pediatric sepsis patients, severe temperature deviations significantly increased in-hospital mortality (OR for severe hypothermia: 14.72; OR for severe hyperthermia: 5.15)18. Additionally, Xu et al. reported that mean BT during the first 24 h of ICU admission was significantly associated with 28-day and in-hospital mortality in patients with diastolic heart failure22. Furthermore, neonatal mortality studies have confirmed a nonlinear relationship between BT and survival, identifying 36.5 °C to 37.5 °C as the optimal BT range associated with the lowest risk23,24,25. Consistent with these findings, our study identified 37.2 °C as the temperature threshold with the lowest mortality rate, reinforcing the importance of early BT assessment as an independent prognostic factor in critically ill pediatric patients.
BT abnormalities at admission may reflect immune dysfunction, metabolic stress, or systemic inflammation, all of which are key contributors to patient outcomes4,5,26. Fever, as part of the acute-phase response, is often triggered by infectious and non-infectious inflammatory stimuli27,28. While moderate fever can enhance immune function by inhibiting bacterial growth and promoting antibody and cytokine production29,30,31, excessive hyperthermia increases metabolic demands and oxygen consumption, potentially worsening neurological outcomes32,33,34. In a study of 10,503 hospitalized COVID-19 patients, a strong positive correlation was found between peak BT and mortality, particularly when BT exceeded 38 °C11. Similarly, the subgroup analysis in our study revealed that hyperthermia in the sepsis group was significantly associated with an increased risk of early mortality. In children with pneumonia, the risk of early mortality significantly increased with rising body temperature (HR 4.24, 95% CI 2.59–6.94). The lack of a significant association between mild temperature abnormalities and mortality in our study may be attributed to the limited physiological impact of mild deviations and the greater physiological resilience of pediatric patients compared to adults. Furthermore, confounding factors, such as infection severity and organ dysfunction, may have mitigated the effects of mild hypothermia and hyperthermia on mortality4,12,19.
Beyond 28-day mortality, both 90-day mortality and in-hospital mortality followed a similar pattern, further emphasizing the long-term prognostic value of admission BT. Several studies have demonstrated that fever may have differential effects depending on infection status. Young et al. found that fever was associated with reduced in-hospital mortality among infected ICU patients, but excessively high fever (> 39 °C) increased mortality risk in non-infected patients12. Other studies have reinforced that temperature management strategies should be tailored based on individual patient conditions13,35. The consistency of our findings across multiple mortality endpoints suggests that monitoring and controlling extreme temperature deviations could help optimize pediatric ICU care.
Hypothermia is often linked to sepsis, systemic inflammation, metabolic failure, or thermoregulatory dysfunction, indicating severe disease progression26,36,37,38. Prior research has suggested that inflammatory responses leading to hypothermia may be indicative of poor prognosis 7,12,18,19,39. Our study highlights the importance of early recognition of hypothermia and hyperthermia at ICU admission, supporting temperature regulation as a crucial aspect of pediatric critical care.
While our study provides evidence for the relationship between admission BT and early mortality in pediatric ICU patients, caution must be exercised when generalizing these findings to adult ICU patients. The physiological characteristics of children differ significantly from those of adults, which may influence the thresholds and clinical impacts of temperature abnormalities. For instance, children have a higher metabolic rate, and their immune systems are not fully developed, which may lead to different responses to infections and temperature abnormalities compared to adults. These differences suggest that the thresholds and clinical outcomes associated with temperature abnormalities in pediatric populations may not be directly applicable to adult ICU settings. Future research should focus on investigating these differences in temperature response between pediatric and adult ICU patients, and on developing tailored management strategies for each age group. However, our findings have significant clinical implications for real-time interventions and management strategies in the PICU. Early identification and management of temperature abnormalities, especially cases of severe deviation from normal body temperature, can reduce mortality risk and improve patient outcomes. Regular and continuous temperature monitoring should be a key component of ICU care for critically ill pediatric patients. Early interventions targeting hyperthermia and hypothermia should be prioritized based on the specific clinical condition of the patient to optimize survival rates and minimize complications.
This study is limited by its retrospective design, which may introduce selection and confounding biases. While we made adjustments for potential confounders and conducted subgroup analyses, the inherent limitations of retrospective data must still be acknowledged. Additionally, although our study included a diverse pediatric population with various diseases, which enhances generalizability, differences in disease severity and underlying conditions may still introduce certain limitations. Furthermore, the single-center nature of the study may introduce additional selection bias, highlighting the need for multicenter studies to validate our results. The use of the PIC database, which had missing clinical variables such as weight and oxygen saturation (SpO₂) for some patients, necessitated the exclusion of these variables from the regression models to minimize the impact of missing data. Notably, this exclusion did not affect the stability of the models, as demonstrated in Supplementary Table 8. Additionally, the lack of data on the use of mechanical ventilation may limit the comprehensiveness of our analysis. Lastly, while temperature fluctuations in ICU patients can arise from various causes, our study did not distinguish between these etiologies beyond those identified. Future studies could benefit from incorporating more detailed etiological data to further refine our understanding of temperature abnormalities in ICU patients. Despite the inherent limitations, our study underscores the critical role of admission BT as a predictor of overall mortality, reaffirming its significance as an independent prognostic factor in ICU patients. A comparative summary of our findings and prior research is presented in Supplementary Table S9. This table outlines the key similarities, differences, and the unique contributions of our study to the existing body of knowledge. Notably, this study provides unique value by being the first to explore the relationship between admission BT and 28-day mortality in critically ill pediatric patients.
Conclusion
In summary, this retrospective cohort study identifies a U-shaped relationship between admission BT and 28-day mortality in PICU patients. Severe hypothermia (< 36.0 °C) and severe hyperthermia (> 38.0 °C) were associated with the highest risk of mortality. These findings highlight the prognostic significance of admission BT, emphasizing the need for early monitoring and appropriate intervention to improve outcomes in critically ill children.
Data availability
Data are available upon reasonable request. The full data set used in this study is available from the first author at changjie@stu.hebmu.edu.cn. However, reanalysis of the full data for other use requires approval by the PIC Institute.
References
O’Grady, N. P. et al. Society of critical care medicine and the infectious diseases society of America guidelines for evaluating new fever in adult patients in the ICU. Crit. Care Med. 51, 1570 (2023).
Dallimore, J. et al. Effect of active temperature management on mortality in intensive care unit patients. Crit. Care Resusc. J. Australas. Acad. Crit. Care Med. 20, 150–163 (2018).
Drewry, A. & Mohr, N. M. Concise definitive review: Temperature management in the ICU. Crit. Care Med. 50, 1138–1147 (2022).
Ganeshan, K. et al. Energetic trade-offs and hypometabolic states promote disease tolerance. Cell 177, 399-413.e12 (2019).
Steiner, A. A. & Romanovsky, A. A. Energy trade-offs in host defense: Immunology meets physiology. Trends Endocrinol. Metab. TEM 30, 875–878 (2019).
Wang, A. & Medzhitov, R. Counting calories: The cost of inflammation. Cell 177, 223–224 (2019).
Xu, F., Zhang, C., Liu, C., Bi, S. & Gu, J. Relationship between first 24-h mean body temperature and clinical outcomes of post-cardiac surgery patients. Front. Cardiovasc. Med. 8, 746228 (2021).
Niven, D. J. & Laupland, K. B. Pyrexia: Aetiology in the ICU. Crit. Care Lond. Engl. 20, 247 (2016).
Laupland, K. B. et al. Occurrence and outcome of fever in critically ill adults. Crit. Care Med. 36, 1531–1535 (2008).
Kiekkas, P. et al. Peak body temperature predicts mortality in critically ill patients without cerebral damage. Heart Lung J. Crit. Care 39, 208–216 (2010).
Uchiyama, S., Sakata, T., Tharakan, S. & Ishikawa, K. Body temperature as a predictor of mortality in COVID-19. Sci. Rep. 13, 13354 (2023).
Young, P. J. et al. Early peak temperature and mortality in critically ill patients with or without infection. Intensive Care Med. 38, 437–444 (2012).
Fever and Antipyretic in Critically ill patients Evaluation (FACE) Study Group et al. Association of body temperature and antipyretic treatments with mortality of critically ill patients with and without sepsis: multi-centered prospective observational study. Crit. Care 16, R33 (2012).
Megged, O., Yinnon, A. M., Raveh, D., Rudensky, B. & Schlesinger, Y. Group a streptococcus bacteraemia: comparison of adults and children in a single medical centre. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 12, 156–162 (2006).
Greer, D. M., Funk, S. E., Reaven, N. L., Ouzounelli, M. & Uman, G. C. Impact of fever on outcome in patients with stroke and neurologic injury: a comprehensive meta-analysis. Stroke 39, 3029–3035 (2008).
Saxena, M. et al. Early temperature and mortality in critically ill patients with acute neurological diseases: trauma and stroke differ from infection. Intensive Care Med. 41, 823–832 (2015).
Zeng, X. et al. PIC, a paediatric-specific intensive care database. Sci. Data 7, 14 (2020).
Wang, H. et al. Hypothermia or hyperthermia, which is associated with patient outcomes in critically ill children with sepsis? —A retrospective study. BMJ Open 13, e067716 (2023).
Erkens, R. et al. Admission body temperature in critically Ill patients as an independent risk predictor for overall outcome. Med. Princ. Pract. 29, 389–395 (2020).
Rincon, F. et al. Significance of admission temperature and impact on mortality in critically ill neurological patients. Crit. Care 15, 1–190 (2011).
Nakajima, Y. Controversies in the temperature management of critically ill patients. J. Anesth. 30, 873–883 (2016).
Tan, D. J. et al. Association of body temperature and mortality in critically ill patients: An observational study using two large databases. Eur. J. Med. Res. 29, 33 (2024).
Lyu, Y. et al. Association between admission temperature and mortality and major morbidity in preterm infants born at fewer than 33 weeks’ gestation. JAMA Pediatr. 169, e150277 (2015).
Cavallin, F. et al. Non-linear association between admission temperature and neonatal mortality in a low-resource setting. Sci. Rep. 10, 20800 (2020).
Guadagno, C. et al. Relationship between admission temperature and risk of cerebral palsy in infants admitted to special care unit in a low resource setting: a retrospective single-center study. Children 9, 352 (2022).
Garami, A., Steiner, A. A. & Romanovsky, A. A. Fever and hypothermia in systemic inflammation. Handb. Clin. Neurol. 157, 565–597 (2018).
Walter, E. J., Hanna-Jumma, S., Carraretto, M. & Forni, L. The pathophysiological basis and consequences of fever. Crit. Care 20, 200 (2016).
Herlihy, J. M. & D’Acremont, V. Hay Burgess, D. C. & Hamer, D. H. Diagnosis and treatment of the febrile child. In Reproductive, Maternal, Newborn, and Child Health: Disease Control Priorities, Third Edition (Volume 2) (eds Black, R. E. et al.) (The International Bank for Reconstruction and Development/The World Bank, 2016).
Villar, J. et al. Induction of the heat shock response reduces mortality rate and organ damage in a sepsis-induced acute lung injury model. Crit. Care Med. 22, 914–921 (1994).
Kluger, M. J., Kozak, W., Conn, C. A., Leon, L. R. & Soszynski, D. The adaptive value of fever. Infect. Dis. Clin. North Am. 10, 1–20 (1996).
Mackowiak, P. A. Fever: Blessing or curse? A unifying hypothesis. Ann. Intern. Med. 120, 1037–1040 (1994).
Manthous, C. A. et al. Effect of cooling on oxygen consumption in febrile critically ill patients. Am. J. Respir. Crit. Care Med. 151, 10–14 (1995).
Hypothermia after Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N. Engl. J. Med. 346, 549–556 (2002).
Bernard, S. A. et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N. Engl. J. Med. 346, 557–563 (2002).
Ahkee, S., Srinath, L. & Ramirez, J. Community-acquired pneumonia in the elderly: Association of mortality with lack of fever and leukocytosis. South. Med. J. 90, 296–298 (1997).
Mégarbane, B., Axler, O., Chary, I., Pompier, R. & Brivet, F. G. Hypothermia with indoor occurrence is associated with a worse outcome. Intensive Care Med. 26, 1843–1849 (2000).
Brivet, F. et al. Hypothermia, a pertinent clinical prognostic factor in severe systemic inflammatory response syndrome. Crit. Care Med. 22, 533 (1994).
Clemmer, T. P. et al. Hypothermia in the sepsis syndrome and clinical outcome. Crit. Care Med. 20, 1395 (1992).
Kushimoto, S. et al. Body temperature abnormalities in non-neurological critically ill patients: A review of the literature. J. Intensive Care 2, 14 (2014).
Acknowledgements
We thank Jie Liu of the Department of Vascular and Endovascular Surgery, Chinese PLA General Hospital, for his contribution to statistical support, study design consultations, and comments regarding the manuscript.
Funding
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
J.C. was responsible for conceiving and designing the study, extracting and analyzing the data, and drafting the manuscript. L.P.L. collected data. Z.Y.H. critically reviewed and revised the manuscript and, as the guarantor, is accountable for the overall content.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study involves human participants and this project has been approved by the Institutional Review Committee of the Children’s Hospital of Zhejiang University School of Medicine (ethics reference number: 2019-IRB-052). Because this study was a retrospective study and did not affect clinical decision-making, the need for informed consent from patients was waived. Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Consent for publication
All authors have reviewed and approved the manuscript for publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chang, J., Liu, L. & Han, Z. Association between hypothermia and hyperthermia and 28-day mortality in pediatric intensive care unit patients: a retrospective cohort study. Sci Rep 15, 9141 (2025). https://doi.org/10.1038/s41598-025-93862-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-93862-0