Abstract
This study investigates malaria vector dynamics in Lare and Asendabo, southwestern Ethiopia, areas with limited entomological information. From July to December 2021, mosquitoes were collected from 56 randomly selected houses using Pyrethrum Spray Catches, CDC light traps, and Human Landing Catches. Blood meal sources (human, bovine, goat) and circumsporozoite proteins (CSP) were detected via Enzyme-linked immunosorbent assays (ELISA), and mosquitoes were analyzed for physiological state, and Plasmodium infection. The entomological inoculation rates (EIR) for Anopheles gambiae s.l. and An. funestus were calculated, and species identification was performed by PCR on CSP-positive and randomly selected samples. A total of 2,565 mosquitoes from four Anopheles species were collected, with An. gambiae s.l. dominating. Indoor collected anophelines (53%) surpassed outdoor (47%). PCR confirmed An. gambiae s.l. as An. arabiensis and An. funestus as An. funestus and An. leesoni. Twenty two An. funestus tested positive for Plasmodium while Plasmodium-positive pools also included, An. gambiae s.l. (15), An. coustani (2), and An. pharoensis (1). The P. falciparum EIR for An. gambiae s.l. in Asendabo was 1.39 bites/person/month, while An. funestus had the highest bite rate (10.52 bites/person/month). Mixed blood meals and zoophilic behaviour were common, underscoring the need for species-specific, spatio-temporal vector control strategies.
Similar content being viewed by others
Introduction
Globally, nearly half of the world population lives in areas at risk of malaria transmission whose health and work capacity could be significantly affected by the disease due to its vast geographic distribution, morbidity, mortality and socioeconomic impact1. Despite the significant declines in morbidity and mortality due to malaria in the last two decades, the disease remains the major public health and socioeconomic problem in Ethiopia. Approximately 75% of the total area in Ethiopia is malarious2,3,4, and more than 60% of the country’s population is estimated to be at risk of the disease2,5,6.
Malaria transmission in Ethiopia is affected by altitude and rainfall, with lag times varying from a few weeks before the beginning of the rainy season to more than a month after the end of the rainy season8,9. Transmission occurs mainly at altitudes less than 2000 m, although it has been reported at altitudes above 2000 m in the fringe highlands of the country2,10,11. In most parts of the country, malaria transmission is highly seasonal and peaks biannually, the major transmission occurring from September to December, following the main rainy season of June to September5. The short transmission season is from April to May following the short rains of March–April in central and most eastern parts of the country12. As the transmission seasons coincide with the main harvesting seasons in most parts of the country, the disease has serious consequences, particularly to Ethiopia’s agriculture-driven economy. The diverse ecology of the country also supports a wide range of transmission intensities ranging from low-seasonal to high-perennial transmission13. Accordingly, there is marked seasonal interannual and spatial variability in risk and transmission intensity in most regions of the country14,15,16. Because of the unstable nature of malaria infection in Ethiopia, both adults and children are at risk of malaria infection and disease.
The coexistence of P. falciparum and P. vivax as major parasite species in Ethiopia also makes malaria control more complicated than in most African countries, where P. vivax has low or no endemicity7,13. Such co-infections with multiple Plasmodium species, and their particular dynamics impacts persistence of the infections and the success of the treatment due to differences in biology and genetics of the parasites17, necessitating different diagnosis, and treatment approaches to target such complex parasite dynamics. Plasmodium falciparum accounts for 60–70% of malaria cases in Ethiopia while the rest malaria cases are caused by P. vivax18,19,20.
Members of the Anopheles gambiae complex are important malaria vectors in sub-Saharan Africa21. Anopheles arabiensis occupies more than 70% of sub-Saharan Africa, dominating both arid and highland areas22,23, and it is the main malaria vector in Ethiopia, occupying a wide geographical distribution2,14,24,25,26. The other malaria vectors in the country that play secondary roles are An. funestus, An. pharoensis and An. nili2,25,26. Although An. coustani has a role in malaria transmission as a secondary vector in some sub-Saharan African countries27, its importance in Ethiopia is mostly unclear2. However, a recent membrane feeding assay28 revealed that the Ethiopian populations of An. coustani are susceptible to both P. falciparum and P. vivax. An exotic invasive malaria vector, An. stephensi has also been detected in several parts of Ethiopia, where its geographical distribution has expanded rapidly29,30.
In Ethiopia, insecticide-treated bed nets (ITNs) and indoor residual spraying (IRS) are the two main malaria intervention tools13. However, the effectiveness of these interventions varies due to the vector species and their behavioural responses to the intervention tools31. Therefore, malaria control interventions can be more effective by understanding the spatiotemporal transmission dynamics of vectors and their feeding preferences32 coupled with accurate identification of anopheline species33. The incidence of malaria can also be estimated from several factors, including entomological34, parsitological35, clinical and immunological parameters36,37.
The entomological inoculation rate (EIR) is one of the most common parameters used to measure the intensity of malaria transmission in a particular setting34,38. The EIR is the product of the human biting rate (the number of bites per person per day by vector mosquitoes), and the sporozoite rate, the fraction of vector mosquitoes that are infectious39. The Human Biting Rate (HBR) is a key epidemiological indicator used to measure the frequency with which mosquitoes bite human hosts over a defined period, and can be estimated combined with the sporozoite rate (SR) with information on the prevalence of Plasmodium infection in the salivary glands of mosquitoes40. The HBR provides critical information about the intensity of human-mosquito contact, which directly influences the risk of vector-borne diseases, especially malaria. The EIR is influenced by HBR and its estimation is also important for quantifying the potential level of human exposure to infected mosquitoes and for assessing the impact of interventions on malaria transmission in a given area34,41 .
The blood feeding and host-seeking behaviour of a mosquito play pivotal role in determining its pathogen transmission, called vectorial capacity42. Specifically, the feeding preference of anopheline mosquitoes is a key determinant in the transmission of malaria parasites43. Accurately identifying the preferred hosts of malaria vectors is essential, as it determines the frequency of human-vector interactions, a measure that reflects both human exposure risk44 and vectorial capacity. Female Anopheles mosquitoes require blood meals for the production and development of their eggs45. Furthermore, the prevalence of malaria is strongly influenced by mosquito host choice behaviours, which can be studied through blood meal analysis46. Although mosquitoes may have an intrinsic preference for particular host species due to genetic factors irrespective of the availability of hosts47, environmental conditions and the availability of hosts may lead to adaptation to other host species48. In malaria-endemic regions, understanding the blood meal sources of Anopheles mosquitoes is crucial for identifying malaria vectors49. This knowledge is vital, as it provides insights into the exposure of human host to malaria and other vector-borne diseases. Evidence indicates that malaria transmission increases when mosquitoes preferentially feed on humans rather than other non-human hosts50. Therefore, understanding the spatio-temporal dynamics of vector populations and their feeding preferences is essential for vector control decision-making.
Local transmission of malaria can be sustained by mosquitoes within endemic areas, creating hotspots that facilitate widespread transmission of the disease51. These hotspots often persist even during dry seasons due to spatiotemporal heterogeneity, where mosquito breeding and transmission continue in restricted areas52. Such areas can act as sources from which malaria transmission may spread to broader regions when conditions are conducive53. Effective malaria control is more likely when interventions are tailored to specific geographic dynamics in transmission, which is influenced by mosquito species, eco-epidemiological factors, and blood meal sources. In unstable transmission areas, the distribution of malaria is characterized by this spatiotemporal variability54, necessitating the need for monitoring transmission dynamics in specific localities for assessing the effectiveness of malaria control strategies.
A retrospective study conducted from clinical data between 2011 and 2021 across nine health facilities in Lare district highlighted a high malaria burden, with fluctuating patterns over the years55. As a result, Gambella region in general and Lare in particular were identified as regions with the highest malaria prevalence in the country. Additional studies on mosquito species composition and insecticide susceptibility in Lare56 and the neighbouring Itang district57 have confirmed occurrence of both primary and secondary malaria vectors of Ethiopia in the region, some populations of these vectors developed resistance to some classes of insecticides. While a decline in malaria cases was observed in Asendabo town over a decade (2007–2017) based on malaria trend analysis58, recent reports particularly post-COVID-19 indicated an increase in the number of malaria cases among patients visiting the Asendabo Health Center, which serves both the town and catchment areas . However, there is insufficient or paucity of updated data, especially regarding key entomological indices such as Entomological Inoculation Rate (EIR), Human Biting Rate (HBR), and blood meal sources of malaria vectors in both study sites. In malaria-endemic regions, including the current study sites, microepidemiologic variations can constantly sustain malaria incidence and transmission. Therefore, investigating spatiotemporal transmission along with host choice and feeding behaviour of vectors is crucial for properly checking and implementing effective vector control interventions at the locality level at the right time.
Despite the fact that three-fourths of the country is malarious, there are few studies on the EIR59,60,61,62, sporozoite rate/SR61,63,64 and blood meal origin61,65 in different parts of the country. Most of these studies focused on An. arabiensis, the principal malaria vector in the country. However, the disease burden is still high in the country and there is a need for updated information on species composition, infection rate, and blood meal origin of both the principal and secondary malaria vectors due to climate change, land use and cover change, environmental modification, changes in feeding and resting preferences, density, vector species shifts and disease transmission dynamics. Therefore, this study was to investigate species composition, abdominal status, circumsporozoite rate (CSP) and patterns of blood-feeding behaviour of Anopheles mosquitoes from a moderate malaria transmission Asendabo study site of Omonada district in the Oromia Regional State and Lare, a malaria hotspot district of the Gambella Regional State, Ethiopia.
Methods
Study sites
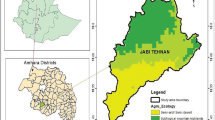
The study was conducted in Asendabo town, Omo Nada district of Jimma Zone (Oromia Regional State), and Lare town, Lare district of Nuer Zone (Gambella Region State) in southwestern Ethiopia (Fig. 1). Both study sites are known to be malaria endemic, with Lare experiencing particularly high malaria transmission rates55. The incidence per 1,000 individuals in Lare has varied over the years, ranging from 18.0 per 1,000 individuals in 2011 to 61.12 per 1,000 in 2021 with notable fluctuations and there was peak incidence in 2016, reaching 151.6 cases per 1,000 individuals55. Lare is located at the southwestern tip of Ethiopia approximately 780 km from the capital, Addis Ababa, while Asendabo is located 299 km from the capital towards southwestern Ethiopia and 55 km from Jimma town on the way to Addis Ababa.
Jimma Zone is located between 7◦15′ to 8◦45′ N latitude and 35◦30′ to 37◦30′E longitudes. In Ethiopia, traditional approach is one of the systems of classifying climatic condition which mainly relies on altitude and temperature variations66,67. Accordingly, a highland refers to areas with altitudes approximately between 2300–3200 m above sea level (a.s.l.) while midland refers to a range of altitudes from 1500–2300 m a.s.l, and lowland lies between 500–1500 m a.s.l. Jimma Zone is mainly divided into three agro-ecological zones: highland (15%), midland (67%), and lowland (18%). The Jimma zone is one of the major coffee growing areas in the Oromia region and is well endowed with natural resources that significantly contribute to the national economy. The Zone also receives good rains, ranging from 1,200–2,800 mm per annum. In most cases, the rainy season extends from February to October. The mean annual temperature ranges between approximately 14 °C and 24 °C. Jimma town, the capital of the zone, is located 354 km southwest of Addis Ababa, Oromia regional state. Jimma zone lies west of the Great Rift Valley generally approximately at altitudes between 1000 m a.s.l and 3300 m a.s.l.
Gambella is one of the twelve (12) regional states of Ethiopia. The region is situated in the Baro and Akobo River basin. Lare is one of districts in the Nuer Zone of Gambella Regional State. Detail description of Lare is stated elsewhere33.
Ethical approval
Scientific and ethical clearance was granted by Institutional Review Board (IRB) of the Institute of Health, Jimma University (Reference number: IHRPGD/787/2019). Written informed consent was obtained from all individuals who participated in mosquito collections. Families were requested through individual discussions and group meetings prior to the selection of their house for mosquito collection in the study. Permission was requested and obtained from inhabitants to conduct mosquito collections both indoors and outdoors, following standard protocols. All methods were performed in accordance with the relevant guidelines and regulations.
Mosquito collection
Adult female Anopheles mosquitoes were collected monthly from Lare and Asendabo study sites between July 2021 and December 2021 using pyrethrum spray catches (PSCs), CDC light traps (BioQuip Model 2836BQ, USA), and human landing catches (HLC). The mosquitoes were sorted by genus and sex, morphologically identified using an identification key68 and individually stored in 1.5 µl Eppendorf tubes. The samples were subsequently transported to Jimma University Tropical and Infectious Diseases Research Center (TIDRC) laboratory and preserved over silica gel, and then kept at 4 °C until further processing.
CDC Light trap collection
Mosquito samples were collected monthly from eight randomly selected houses (four from each of Lare and Asendabo) at the study sites. Each of the selected houses was visited for two consecutive nights every month, making 4 trap-nights per house/month (16 trap-nights per month at each study site). The CDC light traps (CDC LT) were suspended both indoors and outdoors approximately 1.5 m above the floor. No bait was used with the CDC LTs. The traps were switched on at 18:00 PM and switched off at 6:00 AM. Traps were collected from each house every morning at 6:00 AM, and mosquitoes were killed using cotton wool treated with chloroform.
Pyrethrum spray catches (PSCs)
Indoor resting mosquitoes were collected by PSC once a month from 6:00 am to 7:00 am from a total of 40 randomly selected houses (20 houses from each of the study sites) during the study period. Informed consent was obtained from the heads of the houses. People in the selected houses were kindly informed to stay outside, and food items were removed from each house; windows were closed before the operation. Then, the floor and furniture were covered with a white sheet, and Aerosol insecticide (Baygon, SC Johnson & Son, Inc., Racine, Wisconsin, USA) was sprayed in the room, after which the house was closed for 10 min. The knocked-down mosquitoes were retrieved from the sheets, morphologically identified, transferred to 1.5 µl labeled Eppendorf tubes and preserved over silica gel for further analysis.
Human landing catches (HLC)
Mosquito collection was also conducted using human landing catches (HLC) from eight other randomly selected houses (four houses from each of Lare and Asendabo) during the study period. HLC was performed both indoors and outdoors for two consecutive nights per house (16 person-nights per month at each study site) during each surveyed month. Two teams of four trained collectors consented to the HLC operation. Two collectors collected the samples indoors, and two were outdoors. HLC was performed between 6:00 PM and 6:00 AM. Details of the HLC method are described elsewhere33. The collectors put on long-sleeved shirts during the collections to prevent mosquitoes from landing and biting on their arms69. The collectors were seated on stools with their legs exposed from foot to knee to capture mosquitoes using a flashlight and mouth aspirator as soon as they landed on the exposed legs before they commenced blood feeding70. The collectors were given chemoprophylaxis (mefloquine) as per the Ethiopian National Malaria Diagnosis and Treatment Guidelines71.
The gonotrophic status of each female Anopheles mosquito was scored as unfed, freshly fed, half gravid or gravid. The anopheline mosquitoes were then morphologically identified68, individually kept in 1.5 µl labeled Eppendorf tubes and preserved over silica gel in a fridge at 4 °C for further analysis in the Tropical and Infectious Diseases (TIDRC) laboratory at Sekoru Research Center, Jimma University.
Mosquito DNA extraction
Chelex suspension method of DNA extraction from the legs and wings of individual CSP-positive An. funestus and An. gambiae s.l. samples was used as described by Wooden et al.72. Moreover, 27 additional subsamples of An. funestus from Lare study site and 100 An. gambiae s.l. (50 subsamples from each study site) were randomly selected for identification of mosquito species using species-specific PCR. The wings and legs were crushed using sterile conical pestles and incubated overnight at 4 °C after mixing well with 900 µl of 1 × PBS and 50 µl of 10% saponin. The mixture was centrifuged at 12,000 rpm for 12 min at room temperature and then washed with 100 µl of 1 × PBS after discarding the supernatant, followed by removal of any remnants by adding 1 ml of PBS and spinning at 12,000 rpm for 6 min. This was followed by discarding the PBS and drying the DNA for 15 min at room temperature. Then, 150 µl of the 20% Chelex suspension and 100 µl of deionized distilled water were added to extract the DNA by incubating at 95 °C for 10 min in a water bath while vortexing every two minutes during the process of incubation and extraction. Finally, the mixture was centrifuged at 12,000 rpm for 1 min, after which the DNA was transferred to sterile 1.5 ml prelabeled tubes and stored at -20 °C until processing.
Molecular identification of Anopheles species
Molecular identification of female An. gambiae s.l. and An. funestus positive for the CSP-ELISA test was conducted using species-specific PCR at the Molecular Biology Laboratory, Tropical and Infectious Diseases Research Centre (TIDRC), Sekoru, Jimma University. Moreover, 50 additional subsamples of An. gambiae s.l. (morphological) from each of the two study sites and 27 An. funestus subsamples collected during different months of the study period were randomly selected based on availability and molecularly identified via PCR.
PCR amplification was conducted according to the methods of Scott et al.73 for An. gambiae s.l. although this method has a limitation because of its inability to distinguish between An. coluzzii and An. gambiae. Briefly, the extracted DNA product was amplified via PCR using a universal primer (UN: 5’-GTGTGCCCCTTCCTCGATGT-3’) and species-specific primers for An. gambiae s.s. (GA: 5’-CTGGTTTGGTCGGCACGTTT-3’), An. arabiensis (AR: 5’- AAGTGTCCTTCTCCATCCTA-3’) and An. amharicus/formerly An. quadriannulatus B (QD: 5’-CAGACCAAGATG GTTAGTAT-3’), 7.5 µL of nuclease-free water and 1 µL of template DNA. PCR was started with initial 95 °C denaturation for five minutes, followed by 30 cycles of 94 °C for 30 s, 50 °C for 30 s, and 72 °C for 30 s and a final extension of 10 min at 72 °C for An. gambiae complex. To confirm successful amplification, 8 µL of the amplicon was electrophoresed on a 2% agarose gel stained with 1.5 µL ethidium bromide. Previously confirmed An. arabiensis from Sekoru Insectary colony was used as a positive control, and nuclease-free water was used as a negative control.
Preprepared GoTaq® Green Master Mix was used for An. funestus group according to the supplier’s instructions, and 22 µL of an aliquot of the Master Mix solution was used per reaction. Species-specific sequences of primers previously described by Koekemoer and others74 was used for An. funestus group as indicated in Table 1, and the amplification procedure began with an initial hot start at 94 °C for 5 min followed by; 40 cycles of denaturation at 94 °C for 30 s, annealing at 50 °C for 30 s and extension at 72 °C for 40 s; and a final extension at 72 °C for 10 minutes74. Nuclease-free PCR water was used as a negative control, while a 100 bp ladder was used as a marker.
Circumsporozoite enzyme-linked immunosorbent assay (CSP-ELISA)
Dried heads and thoraxes of female Anopheles mosquitoes from preserved samples were analyzed for CSPs of P. falciparum and the two variants of P. vivax (Pv-210 and Pv-247) using a sandwich enzyme-linked immunosorbent assay (ELISA)75,. The CSPs of An. gambiae s.l. and An. funestus were performed on randomly selected samples collected between July 2021 and December 2021. For the CSP analysis of An. pharoensis and An. coustani, both pooled samples collected during same period (July 2021-December 2021) and additional previous collections (2018–2020) from the same study sites were randomly selected and analyzed. Individual heads and thoraces of An. funestus, pools of five heads and thoraces of An gambiae s.l. and pools of ten heads and thoraces for both An. pharoensis and An. coustani were homogenized in a labeled 1.5 ml centrifuge grinding tube using 50 μl of grinding buffer (PBS, pH 7.4 containing IGEPAL CA-603 (Sigma Aldrich) and 0.5% casein, 0.02% phenol red) and a digital pestle. Then, 100 μL of grinding buffer was added twice to bring the final volume to 250 μL per grinding tube, and the homogenate was stored overnight at 4 °C. Each well of a flexible polyvinyl chloride (PVC) microtitration plate was coated with 50 μL of a phosphate-buffered saline (PBS) solution containing capture antibody and incubated for 30 min (Pf) or 60 min (Pv-210 & Pv-247) at room temperature. A circum-sprozoite protein sandwich ELISA using 50 μL/well of the homogenate was performed in 96-well microtiter plates coated with monoclonal antibodies against P. falciparum and P. vivax 210 (Pv-210) and P. vivax 247 (Pv-247) at room temperature for 2 h. Four wells (A12-D12) were used for each of the positive controls (Pf, Pv-210 and Pv-247), and the other four wells (E12-H12) were used for the heads and thoraces of male Anopheles mosquitoes as negative controls.
After 2 h of incubation, the plates were washed twice with 200 μl PBS-Tween 20. Then, mouse peroxidase (MAb)-conjugated monoclonal antibody was added to each plate, and after 1 h of incubation, the plates were washed 3 times with 200 µl PBS-Tween 20. Finally, 100 μL of peroxidase substrate ABTS [2,2’-azino-di-(3-ethylbenzthiazoline-6-sulfonate)] was added to the wells, and the mixture was incubated for 30 min (Pf) or 60 min (Pv). The plates were observed visually for green fluorescence, and the optical density was also determined at 405 nm in a microplate ELISA reader. Absorbance values two times the mean of negative controls were considered the cutoff for sample positivity 75.
Blood meal origin assay
The blood meal sources of samples dried and preserved freshly fed Anopheles mosquitoes collected by CDC light traps (CDC LT), PSCs and HLC were assayed by direct ELISA using human, bovine and goat antibodies75 in different microtiter well-plates. Briefly, the abdomens of Anopheles mosquitoes were cut transversely between the thorax and the abdomen. The posterior part containing the blood meal was individually placed and ground in labeled Eppendorf tubes, homogenized in 50 μL of phosphate-buffered saline (PBS) solution (pH 7.4) and diluted to a volume of 200 μL with PBS. ELISA plates were coated with the homogenate (100 μL/well) or with the positive or negative controls and incubated for 2 h at room temperature in the dark. PBS containing a Tween-20 solution was used to wash each well twice. Then, 50 μL of anti-host (IgG) conjugate against human, bovine or goat proteins was added to each well of separate 96-microtiter plates and incubated for 1 h. Human blood-fed mosquitoes (An gambiae s.l.) and bovine/goat blood were used as positive controls, while PBS was used as a negative control. After the contents were discarded and the wells were washed, 100 µL of ABTS substrate was added, and the mixture was incubated for 30 min. The intensity of the color produced was measured for human, bovine and goat blood meal using an ELISA reader at 405 nm. To record the results as positive, two times the absorbance value of the mean of four negative controls was considered the cutoff value75.
Data analysis
The following entomological parameters were used: a) human biting rate (HBR), calculated as the average number of bites received per person per night of collection; b) sporozoite infection rate, measured as the proportion of mosquitoes positive for a specific species of Plasmodium (P. falciparum, P. vivax) parasite antigen by ELISA; and c) the entomological inoculation rate (EIR), calculated as the product of the mean monthly human biting rate and the sporozoite rate75,76. The human blood index (HBI) was calculated from the proportion of mosquitoes feeding on humans out of all identified blood meals for each species by the following formula77:
The bovine blood index (BBI) and ovine blood index (OBI) were also calculated as the proportions of Anopheles samples with bovine blood and ovine (goat) blood, respectively (with possibly other species sources), out of the total number of Anopheles that fully blood-fed and samples whose blood meals have been identified77. Blood-meal origin data were checked for normality and log-transformed using log (x + 1). A chi-square test was used to compare the mean density of indoor and outdoor mosquito catches. The human blood index (HBI), bovine blood index (BBI) and/or ovine blood index (OBI) of the Anopheles mosquitoes were also compared using the chi-square test. Mean monthly density differences among mosquito species were evaluated by analysis of variance (ANOVA) and when significant variation was detected, Tukey’s-test (HSD) was used to compare the differences between means, adjusted with the Bonferroni correction for multiple comparisons. The data were analyzed using IBM SPSS statistics for Windows v.20.0 (IBM Corp, Armonk, NY, USA). In all tests, values were considered to be significant at p < 0.05.
Results
Anopheles mosquito fauna and dynamics
A total of 2,565 Anopheles mosquitoes were collected in pools using CDC LT (47%), HLC (39%) and PSC (14%) collections from the two study sites from July to December 2021. Four species of Anopheles mosquitoes: An. funestus, An. gambiae s.l., An. pharoensis and An. coustani were collected from Lare study site, while An. funestus was not recorded from Asendabo study site. Among these, An. gambiae s.l. and An. funestus s.l. are of particular medical importance due to their role as malaria vectors. However, these species complexes consist of several morphologically indistinguishable members, making accurate identification essential for understanding their biology, ecology, behaviour, and vector competence33 for designing targeted and effective vector control strategies. Of all, 60% (n = 1,531) of the mosquitoes were collected from Lare study site, while the remaining 40% (n = 1,034) were from Asendabo study site. Among the mosquitoes from the two study sites, 49% (n = 1,244) were An. gambiae s.l. A large proportion of the An. gambiae s.l. samples were collected from both study sites using CDC light traps. Of the 2,565 Anopheles collected during the study period, 53% (n = 1,354) were collected indoors, and 47% (n = 1,211) were collected outdoors (Table 2). Among the 1,531 anophelines sampled from Lare, 51% (n = 779) were collected outdoors, while 49% (n = 752) were collected indoors. In Asendabo, of the 1,034 anophelines sampled, 58% (n = 602) were collected indoors, while 42% (n = 432) were collected outdoors. However, there was no significant difference in the overall mean density of Anopheles mosquito species between those collected outdoors and those collected indoors (x2 = 0.007, df = 3, p = 0.994).
Determination of abdominal status in Anopheles mosquitoes
The abdominal status of Anopheles mosquitoes from different collection sites are shown in Table 2. Overall, freshly fed Anopheles mosquitoes were relatively dominant (1,104/2, 565, 43%), followed by gravid (618/2,565, 24%) and unfed (616/2,565, 24%), and the percentage of half-gravid mosquitoes was only 8.85% (227/2,565). Of the 1,104 freshly fed Anopheles mosquitoes, 55.5% (n = 613) were from Lare site, and the remaining 44.5% (n = 491) were from Asendabo site. In terms of species composition, 48.2% (n = 532) of the freshly fed Anopheles mosquitoes were An. gambiae s.l., most of which (396/532 = 74.44%) were from Asendabo site. The other freshly fed anophelines included An. pharoensis (n = 275), followed by An. funestus (n = 180) from the Lare site and An. coustani (n = 117). The ratio of freshly fed An. gambiae s.l. was greater in PSCs (57/66, 86.36%) from Asendabo than in those recorded from Lare (43/127, 33.86%). Similarly, for CDC light traps, a greater (80.07%, 237/296) proportion was from Asendabo than from Lare (57/267, 21.35%). With respect to HLCs, the ratio of freshly fed An. gambiae s.l. was smaller in Lare (36/184, 19.60%) than in Asendabo (102/129, 79%). A relatively larger proportion of the freshly fed An. pharoensis (200/275, 72.73%) and An. coustani (97/117, 83%) were from the Lare site. Of all An. funestus mosquitoes sampled in this study, 55% (180/326) was freshly fed.
Among the 1,104 freshly fed Anopheles mosquitoes collected from the two study sites, the majority, 54.2% (n = 598) were collected using CDC light traps, while 28.35% (n = 313) were sampled using HLC, and the remaining 17.48% (n = 193) were collected using PSC. Of the 598 CDC LT-collected anophelines, 50.5% (n = 302) were from Lare site, while 49.5% (n = 296) were from Asendabo site. The dominant (636/1,104, 57.61%) freshly fed anophelines were collected indoors, and the majority (332/636, 52.20%) of them were from the Asendabo study site.
Monthly dynamics of Anopheles mosquitoes
The mosquito species and abundances that dominate Lare and Asendabo study sites were different, as confirmed by the monthly sampling between July 2021 and December 2021 (Fig. 2). Although there was variation in abundance, all four species of Anopheles mosquito species: An. gambiae s.l., An. pharoensis, An. coustani and An. funestus were recorded every month between July 2021 and November 2021 at Lare site (Fig. 2A). Anopheles pharoensis and An. gambiae s.l. were dominant from July 2021 to October 2021, and An. funestus and An. coustani dominated in November 2021. Only An. funestus and An. coustani recorded in December 2021, with the highest number of An. funestus being collected during the month. At the Asendabo study site, An. gambiae s.l. was dominant throughout the entire six months, and An. pharoensis was also recorded in all the sampling months with variable numbers (Fig. 2B). The presence of An. coustani in Asendabo was relatively low (n = 26), all recorded exclusively during July–August 2021.
Molecular identification of An. gambiae complex and An. funestus group
Of the 113 (62 from Asendabo and 51 from Lare) samples of An. gambiae complex that were tested for species identification by PCR, 95% (n = 108) were successfully amplified, and all of them were found to be An. arabiensis, and 4.42% (n = 5) could not be amplified, hence, their identity was not determined. Cropped part of the gel electrophoresis result is presented in Fig. 3 while a full-length gel result is included in supplementary material 1. All (n = 13) pools of P. falciparum-positive An. gambiae s.l. samples were also An. arabiensis, the main malaria vector in Ethiopia.
Out of the total 49 An. funestus group subsamples tested for species identification by PCR, 37 (75.51%) were amplified, whereas 12 could not be amplified (Fig. 4). Of the 37 successfully amplified An. funestus group, 94.59%) (n = 35), were An. funestus funestus, and the remaining 5.41% (n = 2) were An. leesoni. Of the 22 An. funestus subsamples that were positive for Plasmodium parasites (six P. falciparum-positive and sixteen P. vivax 247-positive); 20 were successfully amplified, and 95% (19%) of these were An. funestus funustus, and one (n = 1) was An. leesoni.
Anopheles mosquito sporozoite rates
A total of 4,936 female Anopheles mosquitoes, including An. funestus (n = 305), approximately 142 pools of An. gambiae s.l. (n = 709), 248 pools of An. pharoensis (n = 2,478), and 144 pools of An. coustani (n = 1,444), were tested for Plasmodium CSP (Table 3). Among these, 22 individual An. funestus mosquitoes tested positive for CSP, with 16 positive for Plasmodium vivax-247 and 6 for Plasmodium falciparum. Additionally, 15 pools of An. gambiae s.l., 2 pools of An. coustani, and 1 pool of An. pharoensis were also positive for CSP. Remarkably, 14 of the 15 positive pools (93.3%) of An. gambiae s.l. were collected from Asendabo, while only one positive pool came from Lare. Among the 15 positive pools of An. gambiae s.l., 13 (86.7%) were positive for P. falciparum, and 2 pools (13.3%) were positive for P. vivax-247. Of the 13 P. falciparum-positive pools, 12 (92%) were from Asendabo, with only one pool from Lare. Furthermore, two pools of An. coustani (one from each site, Lare and Asendabo) were positive for P. vivax-247, and a pool of An. pharoensis from Asendabo was positive for both P. vivax-247 and P. vivax-210. None of the An. coustani or An. pharoensis pools tested positive for P. falciparum from either site.
In terms of sampling methods, of the 4,936 Anopheles mosquitoes tested for CSP by ELISA, 72% (n = 3,551) were collected using CDC light traps, 22% (n = 1,080) by human landing catch (HLC), and the remaining 6% (n = 305) were sampled using the pyrethrum spray catch (PSC) method.
Entomological inoculation rates
The P. falciparum EIR of An. gambiae s.l. was 1.39 infection bites/person-per month (ib/p/m) at the Asendabo study site. The P. falciparum EIR of An. gambiae s.l. and An. funestus in Lare was equal (0.92 ib/p/m) (Table 4). Anopheles funestus was the most aggressive species, with an overall monthly biting rate of 10.52 bites/person-month (ib/p/m) for P. vivax-247 at the Lare site (Table 5). Pf EIR by An. gambiae s.l. from PSCs was greater (1.19 ib/p/m) at Asendabo than that from An. gambiae at Lare sampled with CDC LT (0.92 ib/p/m). The Pf EIR from HLC-sampled An. gambiae s.l. from Asendabo site was 0.03 ib/p/m. An. funestus from Lare site had a Pf EIR of 0.85 ib/p/m from the PSC and 0.03 and 0.04 ib/p/m from the HLC and CDC LT, respectively. Although CSP-positive An. coustani and An. pharoensis were recorded, their EIRs were not calculated because the subsamples were pooled from both those collected during July 2021-December 2021 and in 2018–2020 and preserved samples from the same study sites.
The P. vivax-247 entomological inoculation rate (Pv-247 EIR) for An. funestus sampled using CDC was the highest (7.34 ib/p/m), while it was 3.18 ib/p/m the HLC-sampled An. funestus (Table 5). The Pv-247 EIR for An. gambiae s.l. was 0.90 ib/p/m (Lare site, CDC), and there was no EIR from An. gambiae s.l. subsamples at Asendabo.
Blood meal sources
From a total of 1,104 engorged Anopheles mosquitoes collected between July and December 2021, randomly selected samples of 528 fully bloodfed female mosquitoes, representing four species: An. gambiae s.l. (n = 176), An. funestus (n = 176), An. pharoensis (n = 88), and An. coustani (n = 88) were used for blood meal analysis. Of the 88 An. coustani samples used for blood meal analysis, 35 sub-samples were previous (2018–2020) collections from Asendabo. The blood meal identification was performed using ELISA, with 344 mosquitoes collected from Lare and 184 from Asendabo. Of the blood meals, 64% (n = 337) were successfully identified, while 36% (n = 191) did not react to any of the three antibodies tested (Fig. 5). The proportion of unidentified blood meals varied between locations, with 28% (n = 146) in Lare and 8.5% (n = 45) in Asendabo. Mixed blood meals were common at both study sites, with approximately 19% (n = 101) of blood meals taken from two or three of the vertebrate hosts. More than 35% (n = 62) of the blood meals from An. gambiae s.l. collected outdoors using CDC light traps were not positive for any of the three antibodies tested. Among mosquitoes with identified blood meals, an overall human blood index (HBI) of 6% (n = 7) was observed for An. gambiae s.l. (An. arabiensis). The majority of An. gambiae s.l. mosquitoes exhibited a strong preference for animal hosts, with an overall ovine blood index (OBI) of 79% (n = 90) and a bovine blood index (BBI) of 15% (n = 17). An. funestus, which was detected only from Lare and its blood meal indices were determined out of mosquito populations collected from Lare site with identified blood meals, exhibited a HBI of 8.33%, with a BBI of 10.71%, and an OBI of 80.95%. On the other hand, a significant portion of the An. funestus (n = 92/176, 52.27%) showed no reactivity to the three antibodies tested while 43.75% (77/176) were found to be zoophilic, feeding primarily on bovine or goat blood and only 3.9% (7/176) were anthropophilic (Fig. 5). An. pharoensis and An. coustani also showed a marked preference for bovine and ovine blood, indicating a predominantly zoophilic feeding behaviour. Overall, the human blood indices (HBI) for mosquitoes from Lare and Asendabo were 15% (30/198) and 40% (55/139), respectively. Out of 176 An. gambiae s.l., 60.79% (n = 107) were zoophilic, feeding on bovine or ovine. Of these, 38.63% (n = 68) was from Asendabo and 22% (n = 39) from Lare. No anthropophilic An. pharoensis was observed in Asendabo, while 30% (20/67) of the An. pharoensis from Lare were anthropophilic.
There were significant differences between the HBI and BBI at both the Lare (x2 = 20.000, df = 6, p = 0.003) and Asendabo (x2 = 16.000, df = 4, p = 0.003) study sites. Similarly, there were significant differences in HBI and OBI (x2 = 16.000, df = 4, p = 0.003) at the Asendabo and Lare sites (x2 = 30.000, df = 9, p < 0.0001). From all the 528 Anopheles mosquitoes tested for blood meal analysis, 55.49% (n = 293) fed on goats followed by bovine (16.47%) and humans. From the mosquitoes collected via HLC, only 6.63% (35/528) were engorged and suitable for blood meal analysis.
Discussions
This study investigated species composition, circumsporozoite rate, entomological inoculation rate, physiological status, and blood meal sources of Anopheles mosquitoes at two study sites in southwestern Ethiopia. Four distinct Anopheles species were identified, with An. gambiae s.l. (confirmed as An. arabiensis) dominating the population. An. funestus was absent in Asendabo while two member species of An. funestus group: An. funestus s.s. and An. leesoni were detected in Lare. Of these, An. funestus s.s. was the predominant sibling species, accounting over 94% of the group. These findings are consistent with prior research in Lare, which documented similar Anopheles species, including An. gambiae s.l., An. funestus, An. pharoensis, and An. coustani56.
As revealed from the present study, the estimated EIRs depended on the sampling methodology used (human landing catches, CDC LT or pyrethrium spray catches), species composition and study sites. For instance, despite providing good estimates of the human biting rate, low sporozoite infection rates were recorded from HLC at the Asendabo study site, and no Pv-247 sporozoite infection was detected from samples collected from either of the study sites using PSC, suggesting the possibility of various factors to be responsible for heterogeneous transmission intensity of malaria in space and time.
The findings from the present study of monthly mosquito dynamics demonstrate temporal variation in the relative abundance and species composition of Anopheles mosquitoes across Lare and Asendabo. In Lare, there was a marked shift in the dominance of Anopheles species over time. From July to October, An. gambiae s.l. (An. arabiensis) and An. pharoensis were the predominant species, suggesting that these vectors could play a major role in malaria transmission during these months. This was followed by a shift in dominance from November to December, with An. funestus and An. coustani becoming more abundant, particularly in December when An. funestus appeared as the most dominant species in Lare. Such temporal variation aligns with previous report by Woyessa and Yewhalaw56, who documented an increase in density of An. funestus populations in Lare from November to January, a period that likely contributes to residual malaria transmission in the area. The observed shifts in species dominance in Lare suggest the need for malaria control interventions to be dynamically adapted to seasonal changes in vector composition and behaviour. From July to October, when Anopheles gambiae s.l. (presumably An. arabiensis) and An. pharoensis are predominant, interventions such as long-lasting insecticidal nets (LLINs) and indoor residual spraying (IRS) are likely to be most effective. However, from November onwards, a shift in vector dominance to An. funestus and An. coustani necessitates a recalibration of control strategies to address these species. The persistence of An. funestus and An. coustani beyond the peak malaria transmission season, after the primary malaria vectors have gone or significantly declined may pose a challenge to malaria control efforts. This is particularly concerning given that some samples these species have tested positive for CSP, indicating their role in residual malaria transmission. To address these challenges, a multi-pronged approaches such as larval source management (LSM) to target aquatic stages78, outdoor residual spraying (ORS) to control outdoor-resting populations of An. funestus and An. coustani79, and community-based environmental management to reduce breeding habitats80 are essential. Complementary strategies including, repelling or killing mosquitoes outdoors, targeting livestock or sugar-feeding adults should also be integrated into control programs as reviewed by Killeen81.
In contrast, the species composition at Asendabo remained relatively stable over the six-month period. An. gambiae s.l. and An. pharoensis were consistently recorded from July to December, with An. arabiensis emerging the predominant vector throughout this duration. Meanwhile, An coustani was recorded only during July and August. Overall, the variations in Anopheles species composition and abundance observed from the two study sites highlight the need for local specific vector control strategies.
The overall proportion of fed Anopheles mosquitoes was approximately 1.68 times greater than that of unfed Anopheles mosquitoes. Although the proportion of blood-fed Anopheles mosquitoes collected indoors was higher compared to unfed mosquitoes, the fact that majority exhibited of zoophagic behaviour suggests that they likely fed on animals outdoors before resting indoors. The zoophilic behaviour of Anopheles mosquitoes observed in this study is consistent with other findings from Ethiopia59 that revealed zoophilic behaviour of An. arabiensis59. Moreover, An. arabiensis with high bovine blood index and CSP positive has been reported from central Ethiopia65, indicating its role in malaria transmission.
Our detail analysis of blood meal sources revealed that mixed blood meals from two or three vertebrate hosts were more common than single blood meals from one host. This trend is consistent with the results of Adugna et al.82, who also reported a similar predominance of mixed blood meals in northwestern Ethiopia. In contrast, a study by Gueye et al.50. in Senegal revealed that single blood meals from either humans or animals were more common than mixed blood meals. Among the identified blood meals in the present study, the majority were of animal origin, with only 25% (85/337) of identified meals, or 16% (85/528) of the total tested meals, originating from humans. Further analysis of the identified potential animal hosts revealed that ovine (goat) was the most frequently bitten host (with 178 in Lare and 115 in Asendabo), followed by bovine (with 13 in Lare and 74 in Asendabo). Our findings are consistent with previous study by Massebo et al.58 in Chano village of southwestern Ethiopia, which documented a zoophagic feeding behaviour in Anopheles mosquitoes though they used only human and bovine antibodies for blood meal identification, unlike the current study, which additionally incorporated ovine antibodies. The strong zoophilic preference by majority of the Anopheles mosquitoes in both study sites in the present study could be due to the impacts of long-lasting insecticidal nets (LLINs) and indoor residual spraying (IRS), the major vector control interventions that have been used in malaria-endemic parts of the country since decades.
Anopheles gambiae s.l., identified as An. arabiensis via PCR analysis using samples from both study sites, exhibited a high infection rate for Plasmodium falciparum and was also infected with P. vivax-247. However, the human blood index (HBI) for An. arabiensis was relatively low, with values of 7.14% in Lare and 5.55% in Asendabo. In contrast, the ovine blood index for An. arabiensis was significantly higher, 92.86% in Lare and 70.83% in Asendabo, confirming a strong tendency of zoophilic behaviour. This finding aligns with previous studies in Ethiopia and elsewhere22,59, which also reported low HBI and a high degree of zoophily in An. arabiensis, across various environmental settings. Moreover, approximately 36% of all Anopheles mosquitoes, and more than 35% of An. gambiae s.l. samples in this study, showed no reaction to the three antibodies tested, suggesting that those which were non-reactive to human, bovine, and goat antibodies would possibly have other host choices such as sheep and pig21. However, it is worth to note that although zoophilic behaviour dominates, the fact that the An. arabiensis was tested positive for CSP from both study sites could enable the vector capable of sustaining endemic malaria transmission whenever feed on humans as reported in other study83. Evidences from other studies also show that Anopheles gambiae s.l., An. coluzzii, and An. funestus shifting from indoor, human-preferring behaviours to outdoor feeding and non-human hosts as a result of vector control measures84,85.
In the present study, molecular identification of An. funestus group from the Lare study site showed that the predominant (94.87%) sibling species was An. funestus, supporting the finding of previous report from the same study site33, where An. funestus s.s. was the dominant sibling species circulating in Lare. However, occurrence of additional sibling species, An. leesoni (n = 2) was also recorded in the present study from subsamples of An. funestus group collected from Lare site and analyzed via PCR in contrast to the previous finding. These differences could be due to the larger sample size (n = 49) used in the present study compared to the previous study (n = 20). In another previous study from North Wollo district of the northern part of Ethiopia, An. leesoni was reported to be the dominant (93.2%) sibling species of An. funestus group84. The predominance of An. funestus s.s. in Lare district, compared to An. leesoni in northern part of Ethiopia may be due to the influence of local ecological or environmental factors on species distribution. This variation may lead to differences in malaria transmission dynamics, necessitating the need for local-specific control strategies. To better understand and predict vector distribution, further studies should focus on longitudinal surveillance to monitor species composition and abundance over time, as well as vector bionomics across broader geographical areas so as to refine malaria control measures tailored to specific vector species in each region.
High EIRs were recorded for P. falciparum and P. vivax-247 from An. funestus in the Lare district challenges the previous thought of considering An. funestus as a secondary vector in Ethiopia86, as it may be an efficient vector for malaria transmission at least at the locality of the current study site. Of the 22 An. funestus group, from which CSPs were detected in this study; 21 (95.54%) were from the An. funestus sibling member species, while the remaining (n = 1, 4.46%) was from An. leesoni, indicating that at least two sibling species of An. funestus group are occurring as possibly important malaria vectors in the Lare district of Ethiopia. An. funestus was reported to be a highly efficient vector of malaria in another study87. As confirmed by direct ELISA, 43.75% of the An. funestus were zoophilic (bovine or goat), while only 5.6% were anthropophilic. In contrast to the current study, it was previously reported in another study that An. funestus is the most anthropophilic and endophilic member of the group87.
The high CSP and EIR recorded from An. funestus may indicate that this vector could be important in disease transmission in such malaria hotspot districts such as Lare in Ethiopia. This can avert the traditional consideration of the vector having only a secondary role in malaria transmission in the country. Although some false-positive circumsporozoite ELISA could not be uncommon as we did not confirm with PCR, information on CSP and the EIR from the current study calls for targeting malaria control efforts to hotspots to efficiently reduce malaria transmission in a wider area and eliminate transmission in a relatively small geographical area83,.
The other anopheline mosquitoes observed in both Lare and Asendabo study sites were An. pharoensis and An. coustani. An. pharoensis has previously been identified as a secondary malaria vector in Ethiopia, as to that of An. funestus, whereas the role of An. coustani in malaria transmission has remained unclear2. Although the circumsporozoite protein (CSP) detection rates in An. pharoensis and An. coustani were lower than those observed in An. arabiensis and An. funestus, their presence in the current study suggests they may still contribute to malaria transmission. It is important to note that even low levels of CSP detection should not exclude the possibility of transmission, as malaria can still be efficiently transmitted at low entomological inoculation rates41. Therefore, integrating these Anopheles species into Ethiopia’s national malaria vector surveillance and control efforts is essential, particularly in regions and seasons where they are prevalent. Furthermore, the National Malaria Control Programs (NMCP) should continuously monitor EIRs from both primary and secondary vectors, with a focus on transmission hotspots to achieve malaria pre-elimination stage and eventual elimination.
While An. coustani was highly anthropo-zoophilic at the Asendabo site, there was no human blood recorded from An. coustani subsamples analyzed from the Lare site. On the other hand, samples of An. pharoensis did not feed on human blood at the Asendabo site, while anthropo-zoophilic behaviour was recorded for the same species at the Lare study site, indicating the existence of variation among the same Anopheles species in terms of host preference according to locality. Moreover, there is variation in terms of host preference among the same mosquito species as for An. coustani and An. pharoensis from the two study sites in the present study which could be due to behavioural changes as a result of special host distributions, such as human/animal ratios42,.
Of remarkable results from our study is that An. coustani exhibited a high degree of anthropo-zoophilic behaviour from samples in the Asendabo site, with blood meal analysis revealing that the majority of its blood meals were from humans (92.73%) and livestock (bovines/goats) (98.2%). Although the sample size for An. coustani blood meal analysis from Asendabo was relatively small (n = 55), the strong preference for human blood observed in this study may be an important factor contributing to its efficiency in parasite transmission, despite the low (< 1%) CSP detection rate. Further studies with larger sample sizes from broader endemic sites are needed to confirm these findings and better understand the role of An. coustani in malaria epidemiology.
There is also a sizable proportion (29%) of An. pharoensis that fed on human in the Lare site, as confirmed by blood meal analysis. However, no human blood was detected in An. coustani samples from Lare as well as in An. pharoensis samples from Asendabo. Such variability in blood-feeding patterns among different anopheline species, such as An. funestus and An. gambiae s.l., has also been reported in Senegal, where differences in blood meal preferences were found even between houses within the same village88.
Conclusions
Molecular analysis confirmed that all An. gambiae s.l. samples from both Lare and Asendabo were identified as An. arabiensis, while two sibling species: An. funestus funestus and An. leesoni, were detected in Lare within the An. funestus group. The high sporozoite and entomological inoculation rates observed in An. funestus from Lare underscore its role in malaria transmission in this area, challenging the species' previous classification as a secondary vector in Ethiopia. The presence of An. funestus in substantial numbers, particularly toward the end of the peak malaria season, combined with its zoophilic feeding behaviour and detection of both Plasmodium falciparum and Plasmodium vivax 247, highlights the potential of this vector for sustaining malaria transmission. These findings suggest that An. funestus may be responsible for residual malaria transmission and warrant its inclusion in national vector surveillance and control programs. The detection of CSP in two other species, An. pharoensis and An. coustani, which were previously considered secondary and suspected vectors, respectively, indicates that these species may also have a role in malaria transmission. This also necessitates the need for comprehensive and ongoing surveillance across endemic areas of the country to better understand their contribution to disease transmission and design targeted vector control strategies. At both the Asendabo and Lare sites, An. arabiensis tested positive for P. falciparum, while P. vivax 247 was detected in this species only at the Lare site. Blood meal analysis revealed a generally zoophilic tendency among mosquitoes at both study sites, with varying degrees of host preference, indicating significant local variation in host attraction. Spatial variation in host preferences was also observed between and within species of An. arabiensis s.l., An. pharoensis, and An. coustani. Finally, there was also spatial variation in sporozoite rates both between and within different Anopheles species, further emphasizing the complexity of malaria transmission dynamics across different geographic areas Supplementary information.
Data availability
The data used are included in the manuscript and/or available from the corresponding author upon request.
Change history
28 April 2025
A Correction to this paper has been published: https://doi.org/10.1038/s41598-025-98342-z
References
WHO. World malaria report 2023. Geneva. World Health Organization (2023).
FMOH. National Malaria Gudelines, Federal Democratic Republic of Ethiopia, 4th ed, Addis Ababa, Ethiopia (2018).
Girum, T., Shumbej, T. & Shewangizaw, M. Burden of malaria in Ethiopia, 2000–2016: Findings from the Global Health Estimates 2016. Trop. Dis. Travel Med. Vaccines. 5, 11. https://doi.org/10.1186/s40794-019-0090-z (2019).
Adugna, T., Getu, E. & Yewhalaw, D. Species diversity and distribution of Anopheles mosquitoes in Bure district. Northwestern Ethiopia. Heliyon. 6, e05063 (2020).
FMoH. Federal Ministry of Health. National strategic plan for malaria prevention, control and elimination in Ethiopia, 2011–2015. Addis Ababa: Ministry of Health of Ethiopia. (2010).
Ayele, D. G., Zewotir, T. T. & Mwambi, H. G. Prevalence and risk factors of malaria in Ethiopia. Malar J. 11, 195 (2012).
WHO. World malaria report 2017. Geneva: World Health Organization (2017).
Tulu, N. A. Malaria. The Ecology of Health and Disease in Ethiopia. Edited by: Kloos, H. Zein, A. Z. Boulder, USA: Westview Press Inc, 341–352. 2 (1993).
Deressa, W., Ali, A. & Enqusellassie, F. Self-treatment of malaria in rural communities, Butajira, southern Ethiopia. Bull. World Health Organization. 81, 261–268 (2003).
Delil, R. Magnitude of malaria and factors among febrile cases in low transmission areas of Hadiya zone, Ethiopia: a facility based cross sectional study. PLoS One. 11, e0154277 (2016).
Yalew, W. G. et al. Current and cumulative malaria infections in a setting embarking on elimination: Amhara. Ethiopia. Malar J. 16, 242 (2017).
Berhanu, B., Seleshi, Y. & Melesse, A. M. Surface water and groundwater resources of ethiopia: potentials and challenges of water resources development. Nile River Basin: Ecohydrol. Challenges, Climate Change Hydropolitics https://doi.org/10.1007/978-3-319-02720-3_6 (2014).
Taffese, H. S. et al. Malaria epidemiology and interventions in Ethiopia from 2001 to 2016. Infect Dis. Poverty. 7, 103. https://doi.org/10.1186/s40249-018-0487-3 (2018).
Gebre, B. & Negash, Y. Severe malaria among children in Gambella, western Ethiopia. Ethiop. J. Health Dev. 16, 61–70. https://doi.org/10.4314/EJHD.V16I1.9827 (2002).
Aregawi, M. et al. Time series analysis of trends in malaria cases and deaths at hospitals and the effect of antimalarial interventions, 2001–2011. Ethiopia. PLoS One. 9, e106359. https://doi.org/10.1371/journal.pone.0106359 (2014).
Zhou, G. et al. Analysis of asymptomatic and clinical malaria in urban and suburban settings of southwestern Ethiopia in the context of sustaining malaria control and approaching elimination. Malar J. 15, 250 (2016).
Souleiman, Y., Ismail, L. & Eftimie, R. Modeling and investigating malaria P. Falciparum and P. Vivax infections: application to Djibouti data. Infect. Dis. Model. 9, 1095–1116 (2024).
Assefa, A. The third Ethiopian Malaria Indicator Survey 2015 (EMIS-2015). 28th Annual conference (2016).
FMoH. Federal Ministry of Health> National Malaria Elimination Roadmap. Addis Ababa, Ethiopia (2017).
Daba, C. et al. A retrospective study on the burden of malaria in northeastern ethiopia from 2015 to 2020: implications for pandemic preparedness. Infect. Drug Resist. 16, 821–828 (2023).
Mahande, A., Mosha, F., Mahande, J. & Kweka, E. Feeding and resting behaviour of malaria vector, Anopheles arabiensis with reference to zooprophylaxis. Malar J. 6, 100. https://doi.org/10.1186/1475-2875-6-100 (2007).
Onyabe, D. Y. & Conn, J. E. Population genetic structure of the malaria mosquito Anopheles arabiensis across Nigeria suggests range expansion. Mol. Ecol. 10, 2577–2591 (2001).
Mutero, C. M. et al. A transdisciplinary perspective on the links between malaria and agroecosystems in Kenya. Acta Trop. 89, 171–186 (2004).
Abose, T. et al. Re‑Orientation and definition of the role of malaria vector control in Ethio‑ pia; the epidemiology and control of malaria with special emphasis to the distribution, behaviour and susceptibility to insecticides of anopheline vectors and chloroquine resistance in Ziway, Central Ethiopia and other areas. Addis Ababa (1998).
FMoH. Entomological profile of malaria in Ethiopia. Addis Ababa: Federal Ministry of Health (2007).
PMI. Africa IRS (AIRS) Project Indoor Residual Spraying (IRS 2) Task Order Six. Ethiopia 2016 End of spray report. Bethesda, MD: Abt Associates Inc (2016).
Afrane, Y. A., Bonizzoni, M. & Yan, G. Secondary Malaria Vectors of Sub-Saharan Africa: Threat to Malaria Elimination on the Continent?. Current Topics in Malaria. InTech; 2016. Available from: https://doi.org/10.5772/65359.
Tsegaye, A. et al. Susceptibility of primary, secondary and suspected vectors to Plasmodium vivax and Plasmodium falciparum infection in Ethiopia. Parasit Vectors. 15, 384. https://doi.org/10.1186/s13071-022-05467-5 (2022).
Carter, T. E. et al. First detection of Anopheles stephensi Liston, 1901 (Diptera: Culicidae) in Ethiopia using molecular and morphological approaches. Acta Trop. 188, 180–186 (2018).
Balkew, M. et al. Geographical distribution of Anopheles stephensi in eastern Ethiopia. Parasit Vectors. 13, 35. https://doi.org/10.1186/s13071-020-3904-y (2020).
Assa, A., Eligo, N. & Massebo, F. Anopheles mosquito diversity, entomological indicators of malaria transmission and challenges of morphological identification in southwestern Ethiopia. Trop. Med. Health. 51, 38. https://doi.org/10.1186/s41182-023-00529-5 (2023).
Ogola, E. et al. Composition of Anopheles mosquitoes, their blood-meal hosts, and Plasmodium falciparum infection rates in three islands with disparate bed net coverage in Lake Victoria. Kenya. Malar J. 16, 360. https://doi.org/10.1186/s12936-017-2015-5 (2017).
Woyessa, D. et al. Species composition, infection rate and detection of resistant alleles in Anopheles funestus (Diptera: Culicidae) from Lare, a malaria hotspot district of Ethiopia. Malar J. 22, 233. https://doi.org/10.1186/s12936-023-04667-3 (2023).
Shaukat, A., Breman, J. G. & McKenzie, F. E. Using entomological inoculation rate to assess the impact of vector control on malaria parasite transmission and elimination. Malar J. 9, 122 (2010).
Vitor-Silva, S. et al. Declining malaria transmission in rural Amazon: changing epidemiology and challenges to achieve elimination. Malar J. 15, 266 (2016).
Bretscher, M. T. et al. Measurement of Plasmodium falciparum transmission intensity using serological cohort data from Indonesian schoolchildren. Malar J. 12, 21 (2013).
Kerkhof, K. et al. Implementation and application of a multiplex assay to detect malaria specific antibodies: a promising tool for assessing malaria transmission in Southeast Asian preelimination areas. Malar J. 14, 338 (2015).
Kelly-Hope, L. A. & McKenzie, F. E. The multiplicity of malaria transmission: a review of entomological inoculation rate measurements and methods across sub-Saharan Africa. Malar. J. 8, 19. https://doi.org/10.1186/1475-2875-8-19.PMID:19166589;PMCID:PMC2656515 (2009).
Hay, S. I., David, J. R., Jonathan, F. T. & Robert, W. Annual Plasmodium falciparum entomological inoculation rates (EIR) across Africa: literature survey, internet access and review. T. Roy Soc. Trop. Med. H. 94, 113–127 (2000).
Smith, D. L. & Ellis McKenzie, F. Statics and dynamics of malaria infection in Anopheles mosquitoes. Malar. J. 3, 13. https://doi.org/10.1186/1475-2875-3-13 (2004).
Ulrich, J., Naranjo, D. P. & Alimi, T. O. How much vector control is needed to achieve malaria elimination?. Trends Parasitol. 29, 104–109 (2013).
Jeyaprakasam, N. K. et al. Blood meal analysis of Anopheles vectors of simian malaria based on laboratory and field studies. Sci. Rep. 12, 354. https://doi.org/10.1038/s41598-021-04106-w (2022).
Mwangangi, J. M. et al. Blood-meal analysis for anopheline mosquitoes sampled along the Kenyan coast. J. Am. Mosq. Control Assoc. 19, 371–375 (2003).
Garrett-Jones, C., Boreham, P. F. L. & Pant, C. P. Feeding habits of anophelines (Diptera: Culicidae) in 1971–78, with reference to the human blood index: a review. Bull. Entom. Res. 70, 165–185. https://doi.org/10.1017/S0007485300007422 (1980).
Hansen, I. A., Attardo, G. M., Rodriguez, S. D. & Drake, L. L. Four-way regulation of mosquito yolk protein precursor genes by juvenile hormone-, ecdysone-, nutrient-, and insulin-like peptide signaling pathways. Front. Physiol. https://doi.org/10.3389/fphys.2014.00103 (2014).
Bashar, K., Tuno, N., Ahmed, T. U. & Howlader, A. J. Blood-feeding patterns of Anopheles mosquitoes in a malaria-endemic area of Bangladesh. Parasit Vectors. 5, 39. https://doi.org/10.1186/1756-3305-5-39 (2012).
Takken, W. & Verhulst, N. O. Host preferences of blood-feeding mosquitoes. Ann. Rev. Entomol. https://doi.org/10.1146/annurev-ento-120811-153618 (2013).
Stone, C. & Gross, K. Evolution of host preference in anthropophilic mosquitoes. Malar J. https://doi.org/10.1186/s12936-018-2407-1 (2018).
Escobar, D. et al. Blood meal sources of anopheles spp in malaria endemic areas of honduras. Insects. 16(11), 450. https://doi.org/10.3390/insects11070450 (2020).
Gueye, A. et al. Host feeding preferences of malaria vectors in an area of low malaria transmission. Sci. Rep. 13, 16410. https://doi.org/10.1038/s41598-023-43761-z (2023).
Bousema, T. et al. The impact of hotspot-targeted interventions on malaria transmission in rachuonyo south district in the western kenyan highlands: a cluster-randomized controlled trial. PLoS Med. 13, e1001993. https://doi.org/10.1371/journal.pmed.1001993 (2016).
Bousema, T. et al. The impact of hotspot-targeted interventions on malaria transmission: study protocol for a cluster-randomized controlled trial. Trials. 14, 36. https://doi.org/10.1186/1745-6215-14-36 (2013) (PMID:23374910).
Oesterholt, M. J. et al. Spatial and temporal variation in malaria transmission in a low endemicity area in northernTanzania. Malar. J. 5, 98 (2006) (PMID: 17081311).
Knudson, A. et al. Spatio-temporal dynamics of Plasmodium falciparum transmission within a spatial unit on the Colombian Pacific Coast. Sci. Rep. 10, 3756. https://doi.org/10.1038/s41598-020-60676-1 (2020).
Haileselassie, W. et al. International border malaria transmission in the Ethiopian district of Lare, Gambella region: implications for malaria spread into South Sudan. Malar J. 22, 64. https://doi.org/10.1186/s12936-023-04479-5 (2023).
Woyessa, D. & Yewhalaw, D. Seasonal dynamics, resting behaviour and insecticide susceptibility of Anopheles mosquitoes across two differentially malaria-endemic regions in Ethiopia. Curr. Res. Parasitol Vector-Borne Dis. https://doi.org/10.1016/j.crpvbd.2024.100224 (2024).
Chanyalew, et al. Composition of mosquito fauna and insecticide resistance status of Anopheles gambiae sensu lato in Itang special district, Gambella, Southwestern Ethiopia. Malar J https://doi.org/10.1186/s12936-022-04150-5 (2022).
Jemal, A. & Ketema, T. A declining pattern of malaria prevalence in Asendabo Health Center Jimma zone. Southwest Ethiopia. BMC Res Notes 12, 290. https://doi.org/10.1186/s13104-019-4329-6 (2019).
Massebo, F., Balkew, M., Gebre-Michael, T. & Lindtjørn, B. Blood meal origins and insecticide susceptibility of Anopheles arabiensis from Chano in south-west Ethiopia. Parasit Vectors. 6, 44 (2013).
Abraham, M., Massebo, F. & Lindtjørn, B. High entomological inoculation rate of malaria vectors in area of high coverage of interventions in southwest Ethiopia: Implication for residual malaria transmission. Parasite Epidemiol. Control. 2, 61–69. https://doi.org/10.1016/j.parepi.2017.04.003 (2017).
Getachew, D. et al. Species composition, blood meal hosts and Plasmodium infection rates of Anopheles mosquitoes in Ghibe River Basin, southwestern Ethiopia. Parasit Vectors. 12, 257. https://doi.org/10.1186/s13071-019-3499-3 (2019).
Animut, A., Balkew, M., Gebre-Michael, T. & Lindtjørn, B. Blood meal sources and entomological inoculation rates of anophelines along a highland altitudinal transect in south-central Ethiopia. Malar. J. 12, 76. https://doi.org/10.1186/1475-2875-12-76 (2013).
Kenea, O. et al. Human-biting activities of Anopheles species in south-central Ethiopia. Parasit Vectors https://doi.org/10.1186/s13071-016-1813-x (2016).
Degefa, T., Githekob, A. K., Leec, M., Yanc, G. & Yewhalawa, D. Patterns of human exposure to early evening and outdoor biting mosquitoes and residual malaria transmission in Ethiopia. Acta Trop. https://doi.org/10.1016/j.actatropica.2021.105837 (2021).
Eba, K. et al. Anopheles arabiensis hotspots along intermittent rivers drive malaria dynamics in semi-arid areas of Central Ethiopia. Malar J. 20, 154. https://doi.org/10.1186/s12936-021-03697-z (2021).
Hurni, H. Agroecological belts of Ethiopia: explanatory notes on three maps at a scale of 1: 1,000,000. Soil Conservation Research Program of Ethiopia. Addis Ababa, Ethiopia, 31 (1998).
Ayalew, M. The role of rainfall amount and distribution on agriculture systems and crop cropping systems of different agro-ecological regions of ethiopia: a review. Int. J. Agric. Res. 7, 26–40 (2020).
Gillies, M. T. & Coetzee, M. A. Supplement to the Anophelinae of Africa South of the Sahara (African region), Johannesburg: South Africa Institute for Medical Research (1987). file:///C:/Users/user/Downloads/190531%20 (7).pdf. Accessed 20 June, 2024.
Gimnig, J. E. et al. Incidence of malaria among mosquito collectors conducting human landing catches in Western Kenya. Am. J. Trop. Med. Hyg. 88, 301–308. https://doi.org/10.4269/ajtmh.2012.12-0209 (2013).
WHO. Malaria entomology and vector control. Geneva: World Health Organization (2013).
FMoH. National Malaria Gudelines, Federal Democratic Republic of Ethiopia, 3rd ed. Addis Ababa, Ethiopia (2012).
Wooden, J., Kyes, S. & Sibley, C. H. PCR and Strain Identification in Plasmodium falciparum. Parasitol Today. 9, 303–305 (1993).
Scott, J. A., Brogdon, W. G. & Collins, F. H. Identification of single specimens of the Anopheles gambiae complex by the polymerase chain reaction. Am. J. Trop. Med. Hyg. 49, 520–529. https://doi.org/10.4269/ajtmh.1993.49.520 (1993) (PMID: 8214283).
Koekemoer, L. L., Kamau, L., Hunt, R. H. & Coetzee, M. A cocktail polymerase chain reaction assay to identify members of the Anopheles funestus (Diptera: Culucidae) group. Am. J. Trop. Med. Hyg. 6, 804–811 (2002).
Beier, J. C. et al. Field evaluation of an enzyme-linked immunosorbent assay (ELISA) for Plasmodium falciparum sporozoite detection in anopheline mosquitoes from Kenya. Am. J. Trop. Med. Hyg. 36, 459–468. https://doi.org/10.4269/ajtmh.1987.36.459 (1987) (PMID: 3555134).
Bigoga, J. D. et al. Seasonal prevalence of malaria vectors and entomological inoculation rates in the rubber cultivated area of Niete. South Region Cameroon. Parasit Vectors. 5, 197. https://doi.org/10.1186/1756-3305-5-197 (2012).
Pappa, V., Reddy, M., Overgaard, H. J., Abaga, S. & Caccone, A. Estimation of the Human Blood Index in malaria mosquito vectors in Equatorial Guinea after indoor antivector interventions. Am. J. Trop. Med. Hyg. 84, 298–301 (2011).
World Health Organization (WHO). Larval Source Management: A Supplementary Measure for Malaria Vector Control. Geneva: World Health Organization (2013).
World Health Organization (WHO). Guidelines for Malaria Vector Control. Geneva: World Health Organization (2019).
Keating, J. et al. Community-based environmental management for malaria control in Africa. Malaria J. 2, 10 (2003).
Killeen, G. F. Characterizing, controlling and eliminating residual malaria transmission. Malar. J. 13, 330. https://doi.org/10.1186/1475-2875-13-330 (2014).
Adugna, T., Yewhelew, D. & Getu, E. Bloodmeal sources and feeding behaviour of anopheline mosquitoes in Bure district, northwestern Ethiopia. Parasites Vectors 14, 166. https://doi.org/10.1186/s13071-021-04669-7 (2021).
Eshetu, T., Eligo, N., Massebo, F. Cattle feeding tendency of Anopheles mosquitoes and their infection rates in Aradum village, North Wollo, Ethiopia: an implication for animal-based malaria control strategies. Malar J, 22, 81 https://doi.org/10.1186/s12936-023-04516-3 (2023).
Degefa, T. et al. Indoor and outdoor malaria vector surveillance in western Kenya: implications for better understanding of residual transmission. Malar J. 16, 443. https://doi.org/10.1186/s12936-017-2098-z (2017).
Ondeto, B. M. et al. Malaria vector bionomics and transmission in irrigated and non-irrigated sites in western Kenya. Parasitol Res. 121, 3529–3545. https://doi.org/10.1007/s00436-022-07678-2 (2022).
FMoH. Federal Minstry of Health. National Malaria Guidelines. Fourth Ed. Addis Ababa, Ethiopia. 1–108 (2017).
Dia, I., Guelbeogo, M. G. & Ayala, D. 2013. Advances and perspectives i the study of the malaria mosquito Anopheles funestus. In: Anopheles mosquitoes. (eds. Manguin S). (Springer, London)
Doucoure, S. et al. Anopheles arabiensis and Anopheles funestus biting patterns in Dielmo, an area of low level exposure to malaria vectors. Malar J. 19, 230. https://doi.org/10.1186/s12936-020-03302-9 (2020).
Funding
This work received financial support from the European Union’s Horizon 2020 Research and Innovation Programme under grant agreement No. 731060 (INFRAVEC2).
Author information
Authors and Affiliations
Contributions
DY and DW conceived and designed the study. DW performed the field and laboratory experiments, analyzed data and drafted the manuscript. DY critically reviewed the manuscript. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Scientific and ethical clearance was granted by the Institutional Review Board (IRB) of the Institute of Health, Jimma University (Reference number: IHRPGD/787/2019). Written informed consent was obtained from all individuals who participated in the mosquito collections. Heads of families were requested through individual discussions and group meetings prior to the selection of their house for mosquito collection in the study. Permission was requested and obtained from inhabitants to conduct mosquito collections both indoors and outdoors.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained several errors in the in-text citations to the reference list. Starting from reference 27, the in-text references were linked to the wrong references.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Woyessa, D., Yewhalaw, D. Anopheles mosquito fauna, blood meal sources and transmission intensity from high and moderate malaria endemic areas of Ethiopia. Sci Rep 15, 10636 (2025). https://doi.org/10.1038/s41598-025-94739-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-94739-y