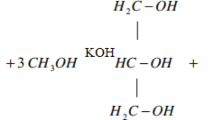Abstract
This study investigates the utilization of biodiesel derived from castor seeds and enhanced with 2% of diethyl ether and CeO2 NPs to improve engine performance and reduce emissions. Various fuel blends, including biodiesel-diesel mixtures with and without nanoparticles, were tested under fixed load conditions. Results indicate that adding CeO2 nanoparticles improved combustion and reduced emissions, particularly CO and NOx. Furthermore, biodiesel blends showed in increasing thermal efficiency and reduced BSFC. From the blends B10 showed the highest increase in thermal efficiency. Results indicate that adding CeO2 NPs improved combustion efficiency, leading to a 22.2% increase in thermal efficiency for the B10 blend. Furthermore, brake power increased by 18% for B10 and 19% for B15 when CeO2 was added, while brake-specific fuel consumption decreased by up to 26.02% for B25. Emissions were significantly reduced: CO emissions dropped by 12% for B5, and HC emissions decreased by 37.7% for B5 with CeO2 addition. However, NOx emissions increased by 4.3% for B10 due to improved combustion. Overall, this research suggests that sustainable biodiesel-diesel blends with CeO2 nanoparticles can effectively enhance engine performance while mitigating emissions, offering a promising solution for cleaner diesel engine operation.Please make justify the abstract to make smart lookup
Similar content being viewed by others
Introduction
Growing concerns over fossil fuel depletion, escalating oil prices, and environmental degradation have spurred interest in alternative fuels. Diesel engines, favoured for their efficiency and low emissions, are extensively used across industries1,2. The automotive industry is experiencing significant growth to meet the demands of both urban and rural areas, becoming an integral part of daily life3. However, this growth has led to increased reliance on petroleum diesel fuels, impacting both economic development and environmental sustainability4,5. Transport vehicles, particularly in countries like Ethiopia, heavily depend on petroleum diesel, necessitating a shift towards cleaner and more sustainable energy sources6.
Reducing diesel imports is crucial for conserving foreign exchange, but it is equally important to identify viable alternatives. However, the finite nature of fossil fuels and their environmental impact necessitate exploring sustainable alternatives7. Biodiesel emerges as a significant alternative energy source, offering a renewable option to replace non-renewable fuels and contribute to environmental conservation8,9. In this context, exploring and implementing alternate energy sources are imperative steps towards achieving sustainable development goals10.
Biodiesel, derived from various feedstocks including non-edible oils like castor oil, emerges as a promising substitute4,5. Castor seeds, known for their high oil yield and resilience to harsh climates for biodiesel production11,12. Although raw castor oil’s viscosity and water content render it unsuitable for direct engine use, conversion to biodiesel addresses this issue. Additionally, alternative fuels like diethyl ether (DEE) show potential for reducing emissions and enhancing engine performance.
Despite limited research on castor oil biodiesel, its potential warrants further investigation, especially regarding its performance when blended with conventional diesel with DEE and CeO2. Biodiesel derived from oilseed crops such as sunflower, canola, and soybean offers significant benefits as a renewable fuel source13. However, the combustion of non-renewable petroleum products remains a major contributor to environmental pollution and global warming14,15. While diesel engines emit lower levels of CO and HC compared to spark ignition engines, they produce substantial amounts of fine particulate matter, composed mainly of carbonaceous material, soluble organic fractions, sulfates, and trace metals16.
To address these environmental concerns and the growing demand for cleaner energy, alternative fuels like biodiesel are being increasingly explored. Castor oil, known for its excellent solubility in methanol, presents a hopeful option for biodiesel making due to its potential for cost-effective transesterification17. With the reduction of petrol reserves and rising oil costs, the quest for alternative fuels has become imperative for both environmental and economic reasons. Additives such as Al2O3, CeO2, and copper oxide, when combined with biodiesel, enhance combustion by increasing power and torque production, attributed to their oxygenated nature. Cerium oxide, in particular, exhibits remarkable redox properties, facilitating complete fuel combustion by generating oxygen that reacts with carbon atoms, hydrocarbon molecules, and carbon monoxide in soot18. When mixed with nanoparticles, vegetable oils demonstrate improved combustion efficiency, reduced emissions, and other favourable properties, highlighting their potential as clean fuel alternatives19,20.
In recent years, extensive research has been conducted to enhance diesel engine performance and reduce emissions through the use of biodiesel blends and nanoparticle additives. Cerium oxide (CeO2) nanoparticles, in particular, have garnered attention for their catalytic properties that promote complete combustion and lower emissions. A study by21,22 investigated the impact of CeO2 nanoparticle size on the performance and emission characteristics of diesel engines fuelled with biodiesel blends. The researchers found that smaller CeO2 nanoparticles significantly improved combustion efficiency, leading to reduced carbon monoxide (CO) and hydrocarbon (HC) emissions. However, an increase in nitrogen oxides (NOx) emissions was observed, attributed to higher combustion temperatures resulting from enhanced oxidation processes.
Similarly,23 explored the effects of adding CeO2 nanoparticles to waste cooking oil biodiesel on diesel engine performance. Their findings indicated that a 50 ppm concentration of CeO2 in B20 blends (20% biodiesel, 80% diesel) enhanced brake thermal efficiency and reduced CO and HC emissions. Nonetheless, a rise in NOx emissions was reported, consistent with the increased in-cylinder temperatures associated with the catalytic activity of CeO2.
In contrast,24 conducted a comparative study on the effects of CeO2 and aluminum oxide (Al2O3) nanoparticles in castor biodiesel blends. The study revealed that while both nanoparticles improved engine performance and reduced CO and HC emissions, CeO2 exhibited a more pronounced effect. However, the increase in NOx emissions was more significant with CeO2 compared to Al2O3, suggesting that the choice of nanoparticle influences the extent of NOx formation.
The present study distinguishes itself by focusing on the use of CeO2 nanoparticles specifically in castor oil biodiesel blends, an area less explored in existing literature. Key research gaps include optimizing CeO2 concentration, understanding CeO2-DEE interactions, and conducting durability studies. Future research should focus on optimizing nano-additive formulations, developing hybrid fuel strategies, using computational models for combustion analysis, and assessing long-term engine impacts. While previous research has predominantly examined waste cooking oil or other biodiesel sources, this study aims to evaluate the performance and emission characteristics of castor biodiesel with CeO2 additives. This research work assessed performance, emissions and combustion behaviour of diesel engines using diesel, castor biodiesel, and 2% DEE with and without 75 ppm cerium oxide, in particular, with the goal of reducing pollutants and optimizing engine operation.
Novelty of the current study are; first to study CeO2 nanoparticle effects on castor biodiesel blends (B0-B25) in a CI engine. Secondly combines CeO2 with diethyl ether (DEE) to further enhance combustion and efficiency. Thirdly, provides a comprehensive performance analysis, including brake power, torque, BSFC, and emissions, across multiple blends and quantifies a 22.2% increase in thermal efficiency with CeO2, which is higher than previously reported improvements with other nanoparticles.
Material and methods
The material required from production to the experimental analysis are shown in Table 1 below.
Experimental procedure
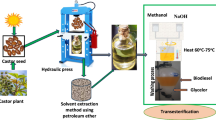
The experiment utilized a Gunt Model CT110 engine test stand equipped with a data logger, exhaust gas analyser, pressure gauge, and other instruments to assess engine performance and emissions. Castor biodiesel was produced by extracting oil from castor seeds using a hydraulic press, followed by transesterification with methanol and NaOH as a catalyst, and purification through washing and solvent removal. Fuel blends were prepared by mixing diesel, biodiesel, 2% of diethyl ether (DEE), and 75 ppm CeO2 nanoparticles to enhance combustion and reduce emissions. The engine was tested for combustion characteristics at 1800, 2400, and 3000 rpm, while performance and emission parameters were analysed over a speed range of 1700–3000 rpm under fixed load conditions, measuring cylinder pressure, heat release rate, brake power, thermal efficiency, and emissions such as CO, NOx, and HC. The overall experimental process flow starting from collecting castor seed to conducting experiment to the CT110 engine test stand is indicated in Fig. 1.
Extraction and determination of oil content
For bulk oil production, a hydraulic press was utilized. From 34 kg of castor seeds, 12.5 L of oil were obtained, yielding 367.6 ml/kg. The extraction efficiency was 0.28 v/w for the solvent method and 0.51v/w for the hydraulic press method. The oil yield was expressed as a percentage of the sample’s dry mass weight25.
Filtration and transesterification process
The castor oil extraction was carried out in both solvent and hydraulic press method. Then the crude oil was saturated for 48 h. after enthused regularly and wait up to settle. Cotton rag was used for filtering. Filtration and drying process is indicated in the process flow diagram in Fig. 2.
During transesterification process, 25 g of NaOH as a base catalyst per a litter of methanol that dissolved to generate metha-oxilate. To eliminate the soap, the methyl ester underwent three rounds of washing. By heating the biodiesel to 1300 °C, to remove the moisture content. The process flow diagrams in Fig. 3 illustrate the overall transesterification process.
Experimental setup
Experimental investigation of the research was done based on the proposed experimental on the CT110 engine test stand under different parameters and conditions with the fully established engine dynamometers and engine consisting of pressure gage, data loggers, charge amplifiers, data acquisition system, air pressure, and temperature gauge, fuel tank for both diesels, Petro blends with DEE and CeO2 NPs, injectors for both direct injection and exhaust gas analyser. The selection of 75 ppm CeO2 nanoparticles in this study was based on their catalytic role in enhancing combustion efficiency while minimizing emissions. CeO2 acts as an oxygen donor, promoting complete combustion and reducing unburned hydrocarbons (HC) and CO. Higher concentrations (> 100 ppm) can cause agglomeration, reducing fuel atomization and efficiency, while lower doses (25–50 ppm) may not sufficiently enhance oxidation. Additionally, excessive oxygen availability at higher doses increases NOx emissions. Prior studies, such as those by26,27, support 75 ppm as the optimal balance, improving brake thermal efficiency without excessive NOx formation. The expermental program for the study is summerized on Table 2.
Result and discussion
Physio-chemical properties of diesel and biodiesel blends
The properties of baseline diesel and biodiesel-diesel blends were measured and analyzed, revealing that all castor oil biodiesel-diesel blends met the ASTM D6751 specifications.
As summarized in Table 3, the physico-chemical properties of the blends were within the acceptable limits for diesel engine fuel. The flash points of B25, B15, B10, and B5 were 79.1 °C, 76.7 °C, 75.5 °C, and 73.4 °C, respectively, all higher than that of pure diesel (72.3 °C), making biodiesel safer for storage and handling. However, the cloud point of biodiesel was above 0 °C, higher than that of pure diesel, indicating the need for additives to lower its freezing point for use in colder climates. As the proportion of castor biodiesel increases in the blend, the cetane number generally decreases exept for B10 and the calorific value, demonstrating a decreasing trend with higher biodiesel content28,29. These trends are attributed to the higher oxygen content and unique fatty acid composition of castor biodiesel, which influence combustion characteristics and energy content. Due to its higher density, biodiesel is safer for storage and handling than petro-diesel, as shown in Table 3. Additionally, its higher initial boiling point can affect engine startup at lower temperatures, leading to increased fuel consumption and unburned hydrocarbon emissions at low engine speeds.
Fourier transform infrared test (FTIR)
From 34 kg of castor seeds, 12.5 L of oil were obtained, yielding 367.6 ml/kg. Finally, a Infrared Fourier Transform test (FTIR) was conducted to confirm whether the oil after transesterification methyl ester as shown in Fig. 4. The FTIR test showed that the conversion of the parent oil to the methyl ester after transesterification process was monitored by carbon -oxygen double bond with ester stretches at wave number of (1740.38 cm−1) that showed a strong ester group and the existence of biodiesel. This result was consistence with the finding of other research work30.
Combustion analysis
In the study of combustion heat release rate and pressure rise rate are the two critical parameters31.
The combustion pressure (CP) and heat release rate (HRR) were evaluated at engine speeds of 1800, 2400, and 3000 rpm. At 80% load and 1800 rpm, the peak CP and HRR for B0, B5, B10, B15, and B25 were recorded as 65.51 bar @377.3 CA, 67.9 bar @375.4 CA, 70 bar @373.9 CA, 67.5 bar @375.7 CA, and 67.4 bar @370.4 CA, and 17.1 J/CA @369.3 CA, 17.24 J/CA @372 CA, 17.82 J/CA @369.8 CA, 18.1 J/CA @369 CA, and 17.7 J/CA @372 CA, respectively, as shown in Fig. 5a.
These results indicate that higher biodiesel blends exhibit slightly higher CP and HRR due to their higher oxygen content, which enhances combustion efficiency. However, when 2% diethyl ether and 75 ppm CeO2 nanoparticles were added, the maximum CP and HRR decreased to 57.12 bar @372.4 CA, 58.3 bar @370.6 CA, and 47.1 bar @361.6 CA, and 17.6 J/CA @373.4 CA, 21.7 J/CA @365.2 CA, and 13.5 J/CA @349.8 CA for B0, B5, and B10, respectively, as shown in Fig. 5b. This reduction is due to the improved combustion characteristics and faster energy release facilitated by diethyl ether’s high cetane number and the catalytic effect of CeO2 nanoparticles, which promote more complete combustion and optimize heat release phasing.
At an engine speed of 2400 rpm, the peak combustion pressure (CP) and heat release rate (HRR) were observed for B0, B5, B10, B15, and B25 as 60.1 bar @371.8 CA, 61.4 bar @372.1 CA, 61.76 bar @373.8 CA, 65.6 bar @370.8 CA, and 64.5 bar @368 CA, and 17.1 J/CA @362 CA, 17.14 J/CA @371 CA, 17.8 J/CA @369.8 CA, 17.3 J/CA @366.8 CA, and 17.5 J/CA @365.7 CA, respectively, as shown in Fig. 6a. These results indicate that higher biodiesel blends generally produce slightly higher CP and HRR due to their increased oxygen content, which enhances combustion efficiency. However, when CeO2 nanoparticles were added, the peak CP and HRR changed to 60.1 bar @378 CA, 58.5 bar @373.8 CA, and 53.6 bar @376 CA, and 21.6 J/CA @370.8 CA, 17.3 J/CA @373.4 CA, and 15.1 J/CA @353.7 CA for B0, B5, and B10, respectively, as shown in Fig. 6b. The incorporation of CeO2 nanoparticles led to a reduction in CP and HRR due to their catalytic properties, which promote more complete combustion and better oxidation reactions, thereby optimizing combustion phasing and heat release.
At 3000 rpm, the maximum cylinder pressure (CP) and heat release rate (HRR) for B0, B5, B10, B15, and B25 were recorded as 61.5 bar at 367.1°CA, 63.4 bar at 369.3°CA, 60.9 bar at 376.3°CA, 69.8 bar at 377.2°CA, and 64.4 bar at 376.3°CA, with corresponding HRR values of 17 J/°CA at 367.1°CA, 20.9 J/°CA at 365.5°CA, 19.2 J/°CA at 368.2°CA, 22.2 J/°CA at 374.4°CA, and 20.9 J/°CA at 364.6°CA as indicated on (Fig. 7a). When CeO2 nanoparticles were introduced, the maximum CP and HRR for B0, B5, and B10 were 47.9 bar at 350.5°CA, 62 bar at 373.2°CA, and 49.7 bar at 374°CA, with corresponding HRR values of 17 J/°CA at 343.8°CA, 24 J/°CA at 372°CA, and 19.1 J/°CA at 359.7°CA, respectively as illustrated on (Fig. 7b). CeO2 nanoparticles enhance combustion by reducing HRR and CP due to their high cetane number. Additionally, the increased oxygen content in biodiesel leads to higher CP and HRR as the blend ratio increases. This observation aligns with the findings32.
The Fig. 8 illustrates the ignition delay (ID) in crank angle degrees (°CA) for various biodiesel blends (B0, B5, B10, B15, and B25) with and without CeO2 nanoparticles, revealing key trends. Ignition delay increases with higher biodiesel blends, with B0 showing the shortest ID at 8°CA and B25 the longest at 12°CA. This is due to the higher viscosity and lower volatility of biodiesel, which hinder atomization and evaporation, delaying ignition. The higher oxygen content and lower calorific value of biodiesel also contribute to slower ignition characteristics. Conversely, the addition of CeO2 nanoparticles consistently reduces the ignition delay across all blends; for example, B0 with CeO2 drops from 8 to 7°CA, and B25 with CeO2 decreases from 12 to 9°CA. This reduction is due to the catalytic activity of CeO2, which enhances combustion by promoting better fuel–air mixing and accelerating oxidation reactions. CeO2 nanoparticles also improve pre-combustion reactions by facilitating faster energy release and increasing combustion temperature.
The consistent reduction in ignition delay for blends with CeO2 demonstrates its effectiveness as an additive for improving combustion performance and efficiency in diesel engines. These findings align with the results reported by33,34,35,36, confirming the enhanced combustion characteristics and reduced ignition delay achieved with CeO2 nanoparticles.
The Fig. 9 shows the combustion duration (CD) in crank angle degrees (°CA) for different biodiesel blends (B0, B5, B10, B15, and B25) with and without CeO2 nanoparticles. The results indicate that combustion duration increases with higher biodiesel blends, with B0 showing the shortest CD at 75°CA and B25 the longest at 86°CA. This is mainly due to the slower burning characteristics of biodiesel, which arise from its higher oxygen content and lower volatility, leading to prolonged combustion. Conversely, the addition of CeO2 nanoparticles reduces the combustion duration across all blends. For example, B0 with CeO2 drops from 75 to 72°CA, and B10 with CeO2 decreases from 81 to 68°CA. This reduction is attributed to the catalytic properties of CeO2, which enhance combustion efficiency by improving fuel–air mixing and accelerating oxidation reactions.
The nanoparticles facilitate better combustion by increasing the rate of oxidation and promoting more complete combustion, resulting in shorter combustion durations. The consistent reduction in combustion duration with CeO2 addition demonstrates its effectiveness as a combustion enhancer. These findings align with the results reported by37,38,39, confirming that CeO2 nanoparticles improve combustion efficiency and reduce combustion duration in biodiesel blends.
The Fig. 10 shows the CA50 (the crank angle at which 50% of the fuel mass is burned) for different biodiesel blends (B0, B5, B10, B15, and B25) with and without CeO2 nanoparticles. The results indicate that CA50 is delayed with higher biodiesel blends, with B0 at 370°CA and B25 at 382°CA. This delay is due to the slower burning rate and longer ignition delay of biodiesel, which are linked to its higher viscosity and lower volatility, resulting in slower combustion and increased mixing time. In contrast, the addition of CeO2 nanoparticles shifts the CA50 slightly earlier for all blends. For example, B0 with CeO2 moves from 370 to 368°CA, and B10 with CeO2 shifts from 376 to 374°CA. This advancement is due to the catalytic action of CeO2, which accelerates the oxidation process and improves combustion phasing by enhancing fuel–air mixing and promoting faster heat release. The nanoparticles also contribute to better combustion efficiency by optimizing the timing of energy release, leading to improved thermal efficiency and more favourable combustion phasing. The consistent shift to earlier CA50 with CeO2 addition demonstrates its effectiveness in enhancing combustion timing. These findings are consistent with the results reported by40,41, confirming that CeO2 nanoparticles improve combustion phasing and thermal efficiency in biodiesel blends.
Engine performance
Engine performance was performed for all blended fuels with and without a 75 ppm CeO2 NPs for comparison at 80% load, from 1700 to 3000 rpm.
Brake power
As the proportion of biodiesel increase, the brake power also increased linearly up to 2700 rpm and start decreasing after 2700 rpm for both cases. From the Fig. 8a and b with the exception of B25, at 270 rpm the break power decreased marginally compared to diesel fuel performance due to the biodiesel blends’ increased viscosity and lower calorific value. The similar result was also demonstrated by other researchers42,43,44. The brake power showed an average increment of 15%, 12%, 18%, 19%, and 9% for B0, B5, B10, B15, and B25, respectively, when CeO2 was added. Furthermore, in both scenarios, the maximum power was at speed of 2700 rpm. The plot in Fig. 11a and b illustrates how the addition of CeO2 NPs resulted in a steady power output that dropped at maximum speed throughout a wide speed operating range.
Brake torque
Addition of CeO2 to the biodiesel blends suggested a considerable increase in engine torque for all blends.
During the addition of CeO2, the braking torque increased on average by 1 Nm, 0.6 Nm, 2.4 Nm, 2.5 Nm, and 1.3 Nm for B0, B5, B10, B15, and B25, in that order. Furthermore, at lower engine speeds, the maximum torque output was measured. The addition of CeO2 NPs resulted in a smooth torque output in a wide speed reduction as engine speed rose, as seen in Fig. 12a and b. According to recent investigations23,45,46,47,48, the outcomes were likewise comparable.
Brake-specific fuel consumption
The two cases of BSFC for the biodiesel blends are displayed in Fig. 13a and b. The study’s findings demonstrated that, for all blends with or without CeO2 the BSFC was lowest at the test engine’s economical speed range, or intermediate engine speed. Nonetheless, including CeO2 has decreased in this speed range by 16.71%, 16.32%, 12.01%, 23.14%, and 26.02% for B0, B5, B10, B15, and B25, in that order.
According to the results, B25 has the highest BSFC reduction rate and B10 has the lowest. Prior research has demonstrated that when CeO2 NPs are combined with biodiesel, the percentage of biodiesel has a greater reduction rate of BSFC49,50. Furthermore, when the blends’ biodiesel ratio rose, as seen in Fig. 13a, the fuel consumption decreased. This is consistent with a prior study by51,52.
Brake thermal efficiency
B15 and B25 have greater engine thermal efficiency over the whole engine operating speed range, as demonstrated in Fig. 14a except engine running at 2700 rpm. A previous study51 found that utilising castor biodiesel increases brake thermal efficiency compared to diesel fuel. Previous studies have demonstrated that when the percentage of biodiesel in the blends increased, the thermal efficiency also increased. Also, resent research findings52,53,54,55 showed the blend ratio and thermal efficiency have linear relation. As illustrated in Fig. 14a and b, the brake thermal efficiency with the addition of CeO2 NPs demonstrated the largest increment, particularly at low speed. For B10 with CeO2 addition, the average maximum gain in thermal efficiency was 22.21%.
Mechanical efficiency
The mechanical efficiency of an internal combustion engine is significantly influenced by fuel composition, engine speed, and the presence of cerium oxide nanoparticles (CeO2 NP). As shown in Fig. 15a and b, mechanical efficiency is high (85–95%) at lower to mid-range speeds (1700–2700 rpm) but decreases at higher speeds due to increased frictional and thermal losses.
Biodiesel blends show slightly lower efficiency than pure diesel (B0) due to higher viscosity and lower calorific value, which increase frictional resistance. However, the addition of CeO2 NP enhances efficiency across all blends by promoting more complete combustion, reducing energy losses, and minimizing frictional wear. Notably, higher blends (B15, B25) see the most improvement, with efficiency gains of 3.2% and 3.6%, respectively, due to the catalytic and lubricating effects of CeO2. These findings align with previous studies showing that CeO2 NP reduces frictional losses by up to 15%, improves combustion efficiency by 5–10%, and stabilizes mechanical efficiency at higher speeds56,57. Additionally, mechanical efficiency generally decreases with increasing engine speed, as reported by47,52,58,59.
Emission characteristics
Carbon monoxide emission
The carbon monoxide (CO) emission lower than that of diesel fuel, from idle speed to the maximum speed as illustrated in Fig. 16a. As engine speed increases, when the CeO2 NP added to biodiesel causes an increase in CO emissions Fig. 16b. The largest average percentage reduction of CO emission for B5 and B10, respectively, was 12% and 10.2% when CeO2 NPs were added. In contrast, CO emissions increased by 1.5%, 30%, and 27% for the blends of B0, B15, and B25, respectively. According to previous studies52,60,61,62, the increase in CO emissions with engine speed was consistent.
Hydrocarbon emission
Figure 17a and b illustrate the correlation between engine speed and hydrocarbon (HC) emissions for blends containing and excluding CeO2 nano-additives. As the percentage of biodiesel grew, as indicated by Fig. 17a, the emission of HC decreased, except for B10 at the maximum engine speed. Furthermore, as speed rose, HC emission decreased for all blends, both with and without CeO2 nano additions. Hydrocarbon (HC) emissions in the engine cylinder are reduced due to the higher cetane number, the enhanced oxygen content of biodiesel, and the inclusion of CeO2 in the biodiesel-diesel blends63,64. For B0, B5, B10, B15, and B25, the addition of CeO2 nanoparticles resulted in an average HC emission reduction of 32.3%, 37.7%, 32.6%, 33%, and 32.8%, respectively. When NPs were employed, the percentage of HC emission decrease was likewise the same as in previous studies51,52,65.
Nitrogen oxide emission
Nitrogen oxides (NOx) in diesel engines primarily form due to high combustion temperatures and excess oxygen, with thermal NOx (Zeldovich mechanism) being the dominant contributor. In this study, NOx emissions increased for B10, B15, and B25 blends with CeO2 due to higher combustion temperatures, shorter ignition delay, increased cylinder pressure, and excess oxygen availability from biodiesel and CeO2. However, a slight NOx reduction was observed in B0 and B5 blends, attributed to more complete combustion, better fuel atomization, and improved air–fuel mixing, which helped reduce localized high-temperature zones.
Figure 18a and b illustrate the variation of NOx emissions as a function of engine speed for fuel blends without and with CeO2 nanoparticles, respectively. The data indicate that the maximum power and torque are achieved at an engine speed of 2900 rpm when CeO2 nanoparticles are introduced. In contrast, the highest NOx emissions were recorded at 2700 rpm in the absence of CeO2. A general increasing trend in NOx emissions was observed with rising engine speed. The incorporation of CeO2 nanoparticles led to a reduction in NOx emissions for B0 and B5 blends, with decreases of 5.5% and 2.4%, respectively. However, a contrary effect was observed for higher biodiesel blends, where NOx emissions increased by 4.3%, 3.3%, and 8.5% for B10, B15, and B25, respectively. These findings are consistent with prior studies, which have also reported a reduction in NOx emissions upon the addition of nano-additives47,65,66. This behaviour is likely attributed to the catalytic influence of CeO2, which enhances combustion efficiency and modifies the thermal characteristics of the flame, thereby affecting NOx formation mechanisms.
Data availability
Data supporting the conclusions are available upon reasonable request from the corresponding author.
Change history
10 September 2025
The original online version of this Article was revised: The original version of this Article contained errors in References 3 and 52. The correct References now accompany the original Article.
Abbreviations
- B0, B5, B10, B15, B25:
-
Diesel-biodiesel blends (0%, 5%, 10%, 15%, 25% biodiesel)
- BSFC:
-
Brake-Specific Fuel Consumption (kg/kWh)
- BTE:
-
Brake Thermal Efficiency (%)
- CA:
-
Crank Angle (°)
- CA50:
-
Crank Angle at 50% Heat Release (°)
- CD:
-
Combustion Duration (°CA)
- CeO2 :
-
Cerium Oxide Nanoparticles
- CI Engine:
-
Compression Ignition Engine
- CO:
-
Carbon Monoxide (%)
- CP:
-
Cylinder Pressure (bar)
- DEE:
-
Diethyl Ether
- HC:
-
Hydrocarbons (ppm)
- HRR:
-
Heat Release Rate (J/°CA)
- ID:
-
Ignition Delay (°CA)
- NOx:
-
Nitrogen Oxides (ppm)
- NPs:
-
Nanoparticles
- η m :
-
Mechanical Efficiency (%)
- η th :
-
Thermal Efficiency (%)
- ρ :
-
Density (kg/m3)
- µ :
-
Dynamic Viscosity (mm2/S)
- LHV:
-
Lower Heating Value (MJ/kg)
- P :
-
Cylinder Pressure (bar)
- V :
-
Cylinder Volume (m3)
- T :
-
Temperature (K)
- γ :
-
Ratio of Specific Heats (Cp/Cv)
- \(\dot{m}\) :
-
Mass Flow Rate (kg/s)
- \(\dot{Q}\) :
-
Heat Transfer Rate (kW)
References
Aydin, H. & Ilkılıc, C. J. A. T. E. Effect of ethanol blending with biodiesel on engine performance and exhaust emissions in a CI engine. Appl. Therm. Eng. 30(10), 1199–1204 (2010).
Park, S. H., Cha, J. & Lee, C. S. J. A. E. Impact of biodiesel in bioethanol blended diesel on the engine performance and emissions characteristics in compression ignition engine. Appl. Energy 99, 334–343 (2012).
Tamrat, S., Ramayya, V., Nallamothu, R. B., & Seifu, Y. CFD Analysis of Sector Combustion Simulation in Four Stroke Direct Injection Diesel Engine. Engineering And Technology Journal, 8(3), 2062–2068. https://doi.org/10.47191/etj/v8i3.10 (2023).
Yadav, A. S. et al. Potential utilization of turpentine oil as an alternative fuel. Mater. Today: Proc. 63, A1–A8 (2022).
Mori, R. Replacing all petroleum-based chemical products with natural biomass-based chemical products: A tutorial review. RSC Sustain. 1(2), 179–212 (2023).
Shrirame, H. Y., Panwar, N. L. & Bamniya, B. R. Biodiesel from castor oil aGreen energy option. Low Carbon Econ. 2(1), 1–6 (2011).
Shahriar, S. & Erkan, T. When will fossil fuel reserves be diminished?. Energy Policy 37(1), 181–189 (2009).
Khan, D. & Goga, G. A review on performance and emission characteristics of a diesel engine fueled with gaseous & liquid fuels. Mater. Today: Proc. 80, A14–A22 (2023).
Ali, S. S. et al. Recent advances in the life cycle assessment of biodiesel production linked to azo dye degradation using yeast symbionts of termite guts: A critical review. Energy Rep. 8, 7557–7581 (2022).
Gebru, H. T. & Gupta, N. Investigation of the performance and emission character biodiesel-diesel blends in direct injection diesel engines. Ethiop. J. Sci. Sustain. Dev. 10(2), 60–73 (2023).
Sana, K., Muhammad, H. & Mustafa, A. Mussel shell based CaO nano-catalyst doped with praseodymium to enhance biodiesel production from castor oil. Fuel 330, 125480 (2022).
Tibesigwa, T., Iezzi, B., Lim, T. H., Kirabira, J. B. & Olupot, P. W. Life cycle assessment of biodiesel production from selected second-generation feedstocks. Clean. Eng. Technol. 13, 100614 (2023).
Ramezani, K., Rowshanzamir, S. & Eikani, M. H. Castor oil transesterification reaction: A kinetic study and optimization of parameters. Energy 35(10), 4142–4148 (2010).
Mahmud, S. et al. Bioethanol and biodiesel blended fuels—feasibility analysis of biofuel feedstocks in Bangladesh. Energy Rep. 8, 1741–1756 (2022).
Nayab, R. et al. "Sustainable biodiesel production via catalytic and non-catalytic transesterification of feedstock materials–A review. Fuel 328, 125254 (2022).
Park, B. et al. Hazard and risk assessment of a nanoparticulate cerium oxide-based diesel fuel additive—a case study. Inhal. Toxicol. 20(6), 547–566 (2008).
Rajagopal, D. Rethinking current strategies for biofuel production in India, Energy and Resources Group, University of California, Berkeley, 2011–2012.
Bailey, B., Eberhardt, J., Goguen, S., Erwin, J. J. S. T. Diethyl ether (DEE) as a renewable diesel fuel, 1578–1584, 1997
Tamrat, S., Ancha, V. R., Gopal, R. & Nallamothu, R. B. J. F. C. Study on the effect of dimethyl ether and diesel-castor biodiesel blends on emission and combustion characteristics. Fuel Commun. 17, 100098 (2023).
Tamrat, S., Ancha, V. R., Gopal, R., Nallamothu, R. B. & Seifu, Y. J. S. R. Emission and performance analysis of diesel engine running with CeO2 nanoparticle additive blended into castor oil biodiesel as a substitute fuel. Sci. Rep. 14(1), 7634 (2024).
Surendhiran, S., Balu, K. S., Karthik, A. & Rajendran, V. Biogenic synthesis of CeO2 nanoparticles via moringa oleifera seed extract: Photocatalytic and biological activity for textile dye degradation. J. Indian Chem. Soc. 101(10), 101302 (2024).
Surendhiran, S., Balu, K. S., Karthik, A. & Rajendran, V. "Biogenic synthesis of CeO2 nanoparticles via moringa oleifera seed extract: Photocatalytic and biological activity for textile dye degradation. J. Indian Chem. Soc. 101(10), 101302 (2024).
El-Seesy, A. I., Waly, M. S., El-Batsh, H. M. & El-Zoheiry, R. M. Enhancement of the diesel fuel characteristics by using nitrogen-doped multi-walled carbon nanotube additives. Process Saf. Environ. Prot. 171, 561–577 (2023).
Kassa, G. M. et al. "Prevalence, trends, and distribution of hepatitis C virus among the general population in sub-Saharan Africa: A systematic review and meta-analysis. Liver Int. 44(12), 3238–3249 (2024).
Fredrick, F. Y., Abdullahi, A. M., Ibrahim, M. N., Chiroma, M. B. & Girbo, A. A. Extraction, characterization and epoxidation of castor seed oil. Fudma J. Sci. 7(2), 284–288 (2023).
Ahmed, A. et al. Environment-friendly novel fuel additives: Investigation of the effects of graphite nanoparticles on performance and regulated gaseous emissions of CI engine. Energy Convers. Manag. 211, 112748 (2020).
Hoang, A. T. et al. A feasible and promising approach for diesel engine fuelled with a blend of biodiesel and low-viscosity Cinnamon oil: a comprehensive analysis of performance, combustion, and exergy. J. Clean. Prod. 401, 136682 (2023).
Keera, S. T. El Sabagh, S. M., Taman, A. R. Castor oil biodiesel production and optimization, Egyptian Journal of Petroleum, 27(4) 979–984 (2018).
Busari, R., Olaoye, J., Fadeyibi, A., Akpenpuun, T., Iyanda, M., Adebayo, S. Characterization of castor biodiesel blended with conventional diesel fuels, 25, 1–17 (2021).
Lamichhane, G., Khadka, S., Adhikari, S., Koirala, N. & Poudyal, D. P. Biofuel production from waste cooking oils and its physicochemical properties in comparison to petrodiesel. Nepal J. Biotechnol. 8(3), 87–94 (2020).
Elkelawy, M., AlmElDin Mohamad, H., Abo-Samra, S. & Abd-Elhay, E. I. Nanoparticles additives for diesel/biodiesel fuel blends as a performance and emissions enhancer in the applications of direct injection diesel engines: A comparative review. J. Eng. Res. 7(1), 112–121 (2023).
Zhu, L., Cheung, C., Zhang, W. & Huang, Z. J. F. Combustion, performance and emission characteristics of a DI diesel engine fueled with ethanol–biodiesel blends. Fuel 90(5), 1743–1750 (2011).
Prasad, R. D., Prasad, N. R., Prasad, N., Prasad, R. S., Prasad, S. R., Shrivastav, O. P., Prasad, R. B., Prasad, R. A., Prasad, R. G., Saxena, P. J. E. G. A Review on Aspects of Nanotechnology in Environmental Science and Engineering, (2025).
Shahir, V., Jawahar, C., Suresh, P. J. R. & Reviews, S. E. Comparative study of diesel and biodiesel on CI engine with emphasis to emissions—a review. Renew. Sustain. Energy Rev. 45, 686–697 (2015).
Selvaraj, S. et al. Cerium oxide nanoparticles and their polymeric composites: Advancements in biomedical applications. J. Inorg. Organometal. Polym. Mater. 34, 5691–5717 (2024).
Mohan, B., Yang, W., Yu, W., Tay, K. L. & Chou, S. K. J. A. E. Numerical investigation on the effects of injection rate shaping on combustion and emission characteristics of biodiesel fueled CI engine. Appl. Energy 160, 737–745 (2015).
Bittle, J., Knight, B., Jacobs, T. J. E. & Fuels,. Interesting behavior of biodiesel ignition delay and combustion duration. Energy Fuels 24(8), 4166–4177 (2010).
Shu, J., Fu, J., Liu, J., Zhang, L. & Zhao, Z. Experimental and computational study on the effects of injection timing on thermodynamics, combustion and emission characteristics of a natural gas (NG)-diesel dual fuel engine at low speed and low load. Energy Convers. Manag. 160, 426–438 (2018).
Zhang, W. et al. A review on performance, combustion and emission of diesel and alcohols in a dual fuel engine. J. Energy Inst. https://doi.org/10.1016/j.joei.2024.101760 (2024).
Zhang, L. et al. and Management, Effective transesterification of castor oil to biodiesel catalyzed by novel carbon-based calcium composite. Energy Convers. Manag. 307, 118368 (2024).
Youssef, A. & Ibrahim, A. J. C. E. A numerical investigation into the effect of altering compression ratio, injection timing, and injection duration on the performance of a diesel engine fuelled with diesel–biodiesel–butanol blend. Clean Energy 8(5), 73–96 (2024).
Carraretto, C., Macor, A., Mirandola, A., Stoppato, A. & Tonon, S. Biodiesel as alternative fuel: Experimental analysis and energetic evaluations. Energy 29(12–15), 2195–2211 (2004).
Akram, S. et al. Impact of cerium oxide and cerium composite oxide as nano additives on the gaseous exhaust emission profile of waste cooking oil based biodiesel at full engine load conditions. Renew Energy 143, 898–905 (2019).
Gajera, K. N., Rawal, R., I. I. I. Education, Effects of addition of various nanoparticles on performance and emission properties of compression ignition engine with diesel and biodiesel blends as a fuel–A review study, 4(1), 562–8, (2018).
Aalam, C. S., Saravanan, C. G. & Kannan, M. Experimental investigations on a CRDI system assisted diesel engine fuelled with aluminium oxide nanoparticles blended biodiesel. Alex. Eng. J. 54(3), 351–358 (2015).
Chinnasamy, C., Tamilselvam, P. & Ranjith, R. Influence of aluminum oxide nanoparticle with different particle sizes on the working attributes of diesel engine fueled with blends of diesel and waste plastic oil. Environ. Sci. Pollut. Res. 26(29), 29962–29977 (2019).
Hoang, A. T. Combustion behavior, performance and emission characteristics of diesel engine fuelled with biodiesel containing cerium oxide nanoparticles: A review. Fuel Process. Technol. 218, 106840 (2021).
Sam Sukumar, R., Muralidhara Rao. M., Gopala Krishna, A. Synthesis and characterization of Cerium Oxide nanoparticles and its applicability as fuel catalyst, Recent Advances in Material Sciences Select Proceedings of ICLIET 2018, 2019, 627–635: Springer.
Kalaimurugan, K., Karthikeyan, S., Periyasamy, M. & Mahendran, G. Experimental investigations on the performance characteristics of CI engine fuelled with cerium oxide nanoparticle added biodiesel-diesel blends. Mater. Today: Proc. 33, 2882–2885 (2020).
Akhtar, M. T. et al. Sustainable production of biodiesel from novel non-edible oil seeds (Descurainia sophia L.) via green nano CeO2 catalyst. Energies 16(3), 1534 (2023).
Ganapathy, T., Gakkhar, R. P. & Murugesan, K. Influence of injection timing on performance, combustion and emission characteristics of Jatropha biodiesel engine. Appl. Energy 88(12), 4376–4386 (2011).
Tamrat, S., Ancha, V. R., Gopal, R. & Nallamothu, R. B. Study of emission characteristics of a diesel engine using cerium oxide nanoparticle blended pongamia methyl ester. Fuel Commun. 41(5), 524–527 https://doi.org/10.1016/j.jfueco.2023.100098 (2020).
Bibin, C., Seeni, K. P. & Devan, P. Performance, emission and combustion characteristics of a direct injection diesel engine using blends of punnai oil biodiesel and diesel as fuel. Therm. Sci. 24(1A), 13 (2020).
Bibin, C., Kannan, P. S., Devan, P. & Rajesh, R. Performance and emission characteristics of a DI diesel engine using diestrol blends and diesel as fuel. Int. J. Enterpr. Netw. Manag. 10(2), 91–108 (2019).
Attia, A. M. A., Kulchitskiy, A. R., Nour, M., El-Seesy, A. I. & Nada, S. A. The influence of castor biodiesel blending ratio on engine performance including the determined diesel particulate matters composition. Energy 239, 121951 (2022).
Karthikeyan, K., Nithya, A. & Jothivenkatachalam, K. Photocatalytic and antimicrobial activities of chitosan-TiO2 nanocomposite. Int. J. Biol. Macromol. 104, 1762–1773 (2017).
Qi, J.-J., Lin, J.-Q. & Fu, H.-Q. One-step production of biodiesel from waste cooking oil catalysed by SO 3 H-functionalized quaternary ammonium ionic liquid. Curr. Sci. 110, 2129–2134 (2016).
Dhanasekar, K., Sridaran, M., Arivanandhan, M. & Jayavel, R. A facile preparation, performance and emission analysis of pongamia oil based novel biodiesel in diesel engine with CeO2: Gd nanoparticles. Fuel 255, 115756 (2019).
Pullagura, G., Prasad Vanthala, V. S., Vadapalli, S., Bikkavolu, J. R. & Chebattina, K. R. R. The effect of thermal conductivity and stably dispersed graphene nanoplatelets on Sterculia foetida biodiesel–diesel blends for the investigation of performance, emissions, and combustion characteristics on VCR engine. Biofuels 15(4), 449–460 (2024).
Gad, M. et al. Performance and emissions characteristics of CI engine fueled with palm oil/palm oil methyl ester blended with diesel fuel Egypt. J. Pet. 27(2), 215–219 (2018).
Ibrahim, S., Abed, K. & Gad, M. Experimental investigation of diesel engine performance burning preheated jatropha oil. World Appl. Sci. J. 31(7), 1231–1236 (2014).
Praveena, V., Ashwath Narayanan, S., Hariskousik, G. & Balavinayagam, K. Effect of aluminium oxide hydroxide Nano fluid in castor oil biodiesel fuelled diesel engine. IOP Conf. Ser. Mater. Sci. Eng. 402(1), 012036 (2018).
Shukla, P. C., Gupta, T., Labhsetwar, N. K. & Agarwal, A. K. J. F. Trace metals and ions in particulates emitted by biodiesel fuelled engine. Fuel 188, 603–609 (2017).
Agarwal, A. K., Gupta, T., Shukla, P. C. & Dhar, A. J. E. C. Particulate emissions from biodiesel fuelled CI engines. Energy Convers. Manag. 94, 311–330 (2015).
Al-Dawody, M. F. & Edam, M. S. Experimental and numerical investigation of adding castor methyl ester and alumina nanoparticles on performance and emissions of a diesel engine. Fuel 307, 121784 (2022).
Fayad, M. A. et al. Reducing soot nanoparticles and NOX emissions in CRDI diesel engine by incorporating TiO2 nano-additives into biodiesel blends and using high rate of EGR. Energies 16(9), 3921 (2023).
Acknowledgements
The authors thank Adama Science and Technology University and Jimma University Institute of Technology for their support, and Balewgize Amare Zeru (Ass. Prof) for his invaluable assistance during the experiment.
Funding
This research received no specific funding from public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
S. T Performed experiments, analyzed data, and wrote the paper. V. R . A, R.G and R.B.N: Supervision, writing, and editing. Y.S. writing, and editing the paper. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tamrat, S., Ancha, V.R., Gopal, R. et al. Study on the effect of CeO2 NP on combustion, emission and performance in Gunt CT110 diesel engine running with castor biodiesel blend. Sci Rep 15, 11225 (2025). https://doi.org/10.1038/s41598-025-95384-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-95384-1
Keywords
This article is cited by
-
Validation of experimental and gradient boosting regressor model for predicting performance, combustion, emission, and biomedical implications of cerium oxide (CeO₂)-augmented B20 biodiesel blends derived from used temple oil
BMC Chemistry (2025)
-
Performance and emission interaction analysis of biodiesel–diesel blends with CeO₂ nanoparticles and diethyl ether in a compression ignition engine
Scientific Reports (2025)
-
Impact of biodiesel concentration and temperature on fuel properties: a comprehensive analysis of diesel and biodiesel mixtures
International Journal of Energy and Water Resources (2025)